Abstract
The application of acidophilic sulfate-reducing bacteria (SRB) for the treatment of acidic mine water has been recently developed to integrate mine water remediation and selective biomineralization. The use of biogenic hydrogen sulfide (H2S) produced from the dissimilatory reduction of sulfate to fabricate valuable products such as metallic sulfide nanoparticles has potential applications in green chemistry. Here we report on the operation of a low-pH sulfidogenic bioreactor, inoculated with an anaerobic sediment obtained from an acid river in northern Chile, to recover copper via the production of copper sulfide nanoparticles using biogenic H2S. The laboratory-scale system was operated as a continuous flow mode for up to 100 days and the bioreactor pH was maintained by the automatic addition of a pH 2.2 influent liquor to compensate for protons consumed by biosulfidogenesis. The “clean” copper sulfide nanoparticles, produced in a two-step process using bacterially generated sulfide, were examined using transmission electron microscopy, dynamic light scattering, energy dispersive (X-ray) spectroscopy and UV-Vis spectroscopy. The results demonstrated a uniform nanoparticle size distribution with an average diameter of less than 50 nm. Overall, we demonstrated the production of biogenic H2S using a system designed for the treatment of acid mine water that holds potential for large-scale abiotic synthesis of copper sulfide nanoparticles.
1. Introduction
Dissimilatory sulfur-reducing bacteria (SRB) generate hydrogen sulfide (H2S) as result of a reductive metabolic process, using sulfate as a terminal electron acceptor. This metabolic activity can be used for the remediation of acid mine drainage (AMD), which is usually characterized as an acidic solution (pH < 4) containing elevated concentrations of dissolved metals, metalloids, and salts [1].
A main limitation to the use of sulfate-reducing bioreactors for the treatment of AMD is that most species are highly sensitive to acid. Therefore, it becomes necessary to avoid direct “contact” between acidic mine waters and the bacteria, which is normally achieved by using “off-line” systems. The Biosulfide and Thiopaq processes are successful industrial applications for the treatment of mine impacted water, and employ offline systems based on a second vessel where sulfide pumped from the bioreactors is in contact with an acidic metal solution [2]. In both systems, the oxidation of organic carbon (e.g., acetic acid) or hydrogen is coupled to the reduction of an oxidized source of sulfur, producing biogenic H2S that is passed to an anaerobic agitated contactor in which metals can be precipitated.
Biosulfidogenesis at low pH to remove sulfate in acidic liquors has the advantage that the final products are almost exclusively H2S and CO2. These compounds are readily transferred through waste gas streams from bioreactors to other reactor vessels for offline metal precipitation [3]. However, few successful applications using low-pH sulfidogenic reactors have been demonstrated for sulfate reduction in, and metal removal from, AMD [4,5,6]. The main reason for this challenge is that SRB preferentially grow between pH 6 and 8, even though sulfate reduction activity has been reported in low pH ecosystems, including acidic lakes, wetlands, and AMD [5]. Attempts to cultivate acidophilic or acid-tolerant SRB from environmental samples have been mostly unsuccessful; nevertheless, media formulations have proven successful when enriching mixed populations possessing acid-tolerant or acidophilic sulfate-reducing properties, e.g., a microbial mat collected from an abandoned copper mine in Spain [4]. This enrichment process has also been used to set up anaerobic bioreactors for the remediation of AMD and to selectively precipitate transition metals as sulfides [4,6,7].
Recently, H2S produced by a sulfidogenic metal remediation process was used to produce zinc sulfide quantum dots possessing optical properties analogous to those prepared commercially [3]. Biogenic production of metallic sulfide nanoparticles has been reported by using a range of different biological [8] and chemical approaches, including aqueous solutions [9], aqueous gels [10], and wet chemical routes using complexing agents [11]. The synthesis of copper sulfide nanoparticles has attracted increasing attention due to their important technological properties as advanced nanomaterials including novel applications in the biomedical area, e.g., as photothermal agents for cancer treatment [12]. Here we describe the operation of a low-pH, sulfate-reducing biofilm-bioreactor populated with novel acidophilic SRB designed to produce enough H2S to remediate a synthetic acidic mine water for the removal of copper and to synthesize copper sulfide nanoparticles.
2. Materials and Methods
2.1. Sulfidogenic Bioreactor
An upflow biofilm sulfidogenic bioreactor (Fermac 200; Electrolab Biotech, Tewkesbury, UK) with a working volume of 2.3 L, was set up and operated as a continuous flow biofilm reactor, based on the system described by Nancucheo and Johnson [4]. The bioreactor housed sulfidogenic bacteria obtained from an anaerobic sediment found in an acidic river located in northern Chile [13]. The mixed population was immobilized on 1–2 mm diameter porous beads made from recycled glass (Poraver Dennert GmbH, Schlüsselfeld, Germany), which occupied ~50% of the total volume of the bioreactor. The system was maintained at pH 4.5, 30 °C and operated at a 50 rpm stirring speed. The bioreactor was initially operated over a pre-trial period with SRB medium at pH 2.5 [7], containing 5 mM glycerol as electron donor and 0.01% (w/v) yeast extract. Automated flow into the sulfidogenic reactor was controlled by a pump linked to the pH unit of the control module. A continuous stream of oxygen-free nitrogen (OFN) was used to remove H2S produced by the bioreactor vessel which was delivered to an off-line vessel to mediate selective precipitation of copper from synthetic acidic mine water based on the chemical composition of a stream draining from a Chilean copper mine (Table 1). A second, more acidic (pH 2.2) form of mine water resulting from this partly-treated water was also supplemented with 5 mM glycerol and 0.01% yeast extract, and was also used as a feed liquor pumped into the base of the bioreactor. The processed liquor was removed at the same rate to maintain constant volumes in the reactor.

Table 1.
Composition of the synthetic mine water used in the present study.
2.2. Formation of Copper Sulfide Nanoparticles
The H2S produced by the low-pH sulfidogenic bioreactor was removed in a stream of OFN with a flow rate of 150 mL∙min−1 extracting 0.1% (v/v) H2S. The gas stream was fed into 100 mL serum bottles containing 100 mL of 2 mM CuSO4 in 50 mM of citrate buffer (pH 6.0), which were subsequently sealed, and head space air was displaced by passing nitrogen through the solution [3]. The copper solutions were sparged by this gas stream for 5, 15, 30, 45, and 60 min, with shaking between dosing, and then stored in the dark at 4 °C for characterization of the nanoparticles. Each experiment was carried out in duplicate.
2.3. Characterization of Nanoparticles
Particle sizes (hydrodynamic diameter) of putative copper sulfide nanoparticles collected from the suspended material were measured twice (before and after of vigorous shaking) by non-invasive dynamic light scattering (DLS) on a zeta-potentiometer (Nano-ZS90, Malvern Instruments, Westborough, MA, USA). The morphology and average size of the nanoparticles were determined using transmission electron microscopy (TEM; JEOL JEM 1200EX-II, Tokyo, Japan). A black precipitate was collected by centrifugation and washed with nanopure water (18.2 megohm-cm−1) to perform energy-dispersive X-ray analysis (EDS) using scanning electron microscopy (JEOL JSM-6380 LV). UV–visible spectroscopy was employed to characterize optical properties, using an EPOCH microplate reader (Bioteck Instrument, Winooski, VT, USA).
2.4. Physicochemical and Microbiological Analysis
Copper concentration was determined with a Perkin-Elmer model AAnalyst 200 atomic absorption spectrometer. Acetic acid was measured by HPLC coupled with a UV detector (Agilent Technologies, Santa Clara, CA, USA) [14], and glycerol was determined enzymatically using a commercial kit [15]. Bacteria were isolated from the bioreactor by streaking liquid samples onto overlay SRB medium to promote the growth of acidophilic SRB [7]. Phylogenetic analysis of the isolates was carried out using the method described by Rowe and colleagues [16].
3. Results
3.1. Operation of a Low Sulfidogenic Bioreactor
An off-line “acidophilic” sulfidogenic reactor was used successfully to over-produce H2S for the recovery of copper from low pH synthetic mine water. No pH adjustment was required to precipitate copper as a sulfide phase. H2S generated by a low-pH sulfidogenic bioreactor during the pre-trail period was delivered in a continuous OFN stream to a precipitation vessel containing synthetic mine water. Over 99% of the copper present in the mine water (200 mL) was removed from solution in 10 h. The copper-depleted mine water was sufficiently acidic (pH 2.2) to be used as feed for the bioreactor supplemented with glycerol and yeast extract. Over the first 15 days of bioreactor operation in continuous flow mode, the hydraulic retention times (HRTs) progressively decreased, though this period (from 285 to 71 h; Figure 1a) was determined to be a transient feature related to the increasing amounts of bacterial numbers in the upper phase in the bioreactor (Figure 1b). Between days 15 and 51, the HRTs decreased steadily, reaching an average HRT of ~63 h with typical rates of sulfate reduction of 0.6 g sulfate reduced L−1∙day−1. However, from day 56 to the end of the experiment (day 99), the HRTs increased dramatically, which was closely coupled to inhibition caused by increasing concentrations of acetic acid. The level of sulfate reduction was directly proportional to the bacterial numbers.
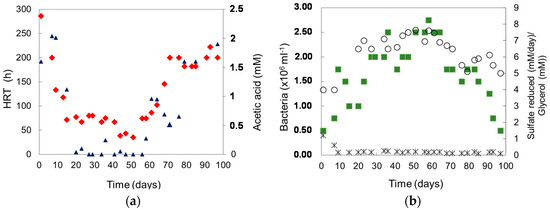
Figure 1.
(a) Changes in hydraulic retention times (HRTs) (♦) and acetic acid (▲) in the reactor during the operational phase; (b) Numbers of planktonic-phase bacteria (■), glycerol in the effluent (✱) and rate of sulfate reduction (⚪).
Liquid samples from the bioreactor at days 40 and 80 were spread onto overlay acidophilic SRB medium. Three isolates were obtained at day 40; one of these (isolate USS-CCA2) grew in colonies encrusted with ZnS, while the other two isolates (USS-CCA3 and USS-CCA4) grew as distinct colonies. At day 80, isolate USS-CCA2 was the only organism recovered. Using 16S rRNA analysis, isolate USS-CCA2 shared 99% similarity with the known sulfidogen Desulfosporosinus acididurans (Strain E, the closest related strain in GenBank), which was isolated from an enrichment containing acidic sediments from the Tinto River [17]. Isolate USS-CCA3 shared 97% gene similarity with an Actinobacteria (IR1, [4]), which was previously isolated from a sulfidogenic bioreactor. The third isolate (USS-CCA4) had a 16S rRNA gene sequence identical to Pandoraea thiooxydans strain ATSB16, originally isolated from a rhizosphere soil [18]. The 16S rRNA gene sequences of the current isolates have been deposited in GenBank, with the accession numbers MF595567.1 (the Desulfosporosinus isolate), MF629800.1 (the Actinobacterium isolate), and MG564634.1 (the Pandoraea isolate).
3.2. H2S Production and Formation of Copper Sulfide Nanoparticles
Hydrogen sulfide produced by the sulfidogenic bioreactor (Figure 2a) between days 20 and 42 was used in the synthesis of metallic copper sulfide nanoparticle experiment. A preliminary test, performed by adding biogenic H2S to the synthetic mine water, predominantly formed micrometer-scale copper sulfides. This size distribution was probably associated with the presence of high concentration of copper in the mine water, allowing for continued growth of initial mineral nuclei, precluding the formation of copper sulfides at the nanoscale. Subsequent efforts were focused in the synthesis of nanoparticles using waste H2S off-gas from the continuous sulfidogenic bioreactor using copper/citrate solutions. Testing was performed upon initial contact between the H2S and the copper solution in the serum bottles, which initially occurred as a brown, turbid suspension, turning black within an hour and settling after several days as a black precipitate (Figure 2b,c). Using UV-Vis spectroscopy, Figure 2d highlights the optical behavior of the suspensions at 15, 30, and 45 min of contact time between H2S with the copper solution. The absorption spectra of the initially formed material were generally similar, which increased from 300 nm to 400 nm and reached a maximum at about ~345 nm. A similar behavior was reported recently for Schaffie and Hosseini [8]. A maximum peak of absorption was obtained for the experiment carried out at 15 min, while no absorption spectra were detected at 5 and 60 min.
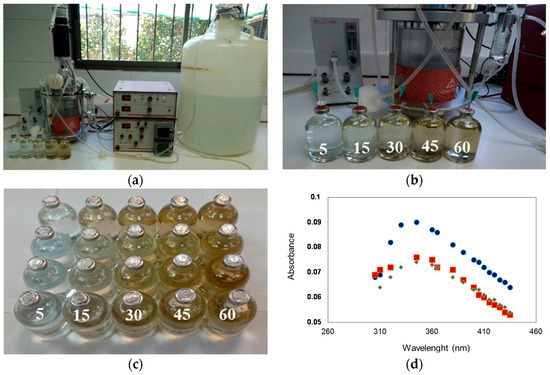
Figure 2.
(a) Bioreactor operating continuously for the synthesis of copper sulfide nanoparticles and (b,c) progression of the synthesis as a function of exposure time (up to 60 min); (d) Absorption spectra recorded of material formed at 15 (●), 30 (■) and 45 (♦) min.
Examination of the copper sulfide nanoparticles using DLS revealed unusual particle behavior. DLS values obtained after vigorous shaking showed the presence of large agglomerates with mean hydrodynamic diameters of 192, 596, and 667 nm at 15, 30, and 45 min exposure times, respectively (Figure 3a). In addition, suspended material obtained at 60 min without shaking showed a mean diameter of 28 nm; however, when the same sample was subjected to shaking, the mean diameter was 684 nm. No nanoparticles were “observed” using DLS for the samples obtained at 5, 15, 30, and 45 min without shaking.
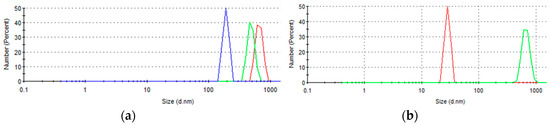
Figure 3.
Size distribution of copper sulfide nanoparticles formed at different contact times with biogenic H2S: 15 min (blue), 30 min (green), and 45 min (red), for samples obtained from the solution (a) upon vigorous shaking and (b) with and without shaking of material formed at 1 h of exposure time (black precipitate).
Figure 4 presents an image obtained using TEM of the shaken, synthesized copper sulfide nanoparticles at a contact time of 1 h with H2S, which reveals the presence of individual, euhedral nanosized particles occurring as an aggregated material. A nearly uniform nanoparticle size can be appreciated, with a mean size ca. 20 × 30 nm.

Figure 4.
Whole mount TEM micrograph of copper sulfides nanoparticles, showing particles with a size diameter less than 50 nm. Note: the aggregation of particles and the angular nature of the microcrystalline nanoparticles formed in this reaction system.
Using SEM (Figure 5a), the angular nature of the copper sulfides was evident. A concurrent EDS analysis (Figure 5b) showed patterns of copper and sulfur, confirming the formation of copper sulfide nanoparticles.
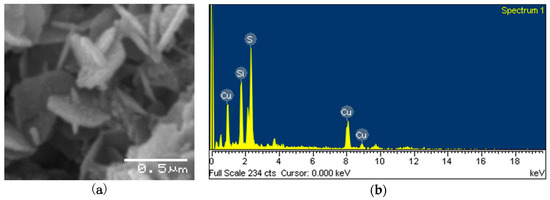
Figure 5.
(a) SEM image and (b) energy dispersive X-ray analysis (EDS) analysis of the nanoparticles. The Si peak was generated from the sample support matrix.
4. Discussion
Most of the approaches currently employed to remediate acidic mine waters produce metal-rich wastes that have to be removed, transported, and stored in specially designated landfills, because the potentially valuable metals are not removed in a form that facilitates their recovery and recycling. In contrast, as no pH adjustment is required to precipitate copper as a sulfide phase, off-line low pH sulfidogenic bioreactors developed to over-produce H2S hold potential as a technology to recover copper from AMD and mine process waters [2]. By implementing this strategy, copper contained in a Chilean synthetic mine water was readily precipitated upon contact with biogenic H2S producing copper sulfide using a single bioreactor.
The key biologic component of the system was an acidophilic anaerobic indigenous consortium obtained from acid drainage, collected from an abandoned sulfur mine in northern Chile [13]. This approach has been recently reported to facilitate the recovery and recycling of copper present in pregnant leach solution (PLS) from mine tailings, allowing for the recovery of a “clean” mineral product [19,20]. Hydrogen sulfide, generated through sulfate reduction by acidophilic SRB, has the advantage of being rapidly transferred from the acid liquor (bioreactor) toward a second vessel in order to promote off-line metal precipitation. However, most of the previously described SRB grow optimally at pH values between 6 and 8 [5]. Acidophilic SRB have been difficult to isolate and cultivate in vitro due to the challenges associated with enrichment and cultivation [7]. Two Desulfosporosinus spp. have been identified and described as acidophilic microorganisms: (1) D. acidophilus, which grows at pH range from pH 3.6–5.5, was isolated from a sediment sample collected in a decantation pond receiving AMD with pH around 3.0 [21]; and (2) D. acididurans, with a pH range for growth between 3.8 and 7.0 was isolated from acidic sediment [14]. In our work, the sequence analysis of one isolate obtained from the bioreactor revealed the presence of the recently described acidophilic bacterium D. acididurans. The roles and significance of isolates CCA3 and CCA4 in the sulfidogenic bioreactor cannot be ascertained at this stage, although they may be involved in syntrophic interactions with this SRB, given they tend to grow successfully with a “partner” acidophile [22]. D. acididurans has been described as an incomplete oxidizer of glycerol that generates acetic acid as a waste product, which can be highly toxic even at μM concentrations. To address this problem, the acidophilic heterotroph Acidocella aromatica was introduced to a sulfidogenic low pH bioremediation process, successfully decreasing the concentrations of acetic acid during operation of the bioreactor [6]. The generation of acetic acid is not desirable in a bioremediation context, since fewer electrons become available for sulfate reduction and the acetic acid generated increases the chemical oxygen demand of water discharged from the process [23]. This energetic issue was observed in the bioreactor during the last period of the experiment, detecting up to 2 mM of acetic acid when only Desulfosporosinus acididurans was found. Between 20 and 58 days, acetic acid was not detected, suggesting that CCA2 or CCA3, or both, had the ability to consume acetic acid.
Another approach to the commercial use of the H2S produced from full-scale bioremediation operations has been developed in the SulfaTeqTM process operated by Paques, in the Netherlands. In this system, the sulfide generated can be partially re-oxidized to elemental sulfur by using sulfide-oxidizing bacteria, while the solid product is removed and applied as fertilizer [24]. However, relatively little is known about the synthesis of metallic nanoparticles using biogenic H2S. The synthesis of nanoparticles of ZnS from a sulfate solution was recently reported by feeding biogenic H2S from the off-gas generated from a sulfidogenic metal remediation process [3]. The present study examined whether the H2S produced from this low-pH sulfidogenic reactor, set up for the removal of copper and sulfate from an acidic mine water, can be used to fabricate CuS nanoparticles. The rationale of using an off-line system is that H2S delivered toward a mine water-containing vessel allows a more controlled separation of metals via pH control. For example, at the Talvivaara mine in Finland, transition metals (e.g., copper and zinc) are selectively precipitated from their PLS using chemically generated H2S using a pH adjustment, enabling selective metal recovery [25]. In this study, the preliminary test using synthetic mine water demonstrated that the formation of copper sulfide particles initially occurred in the bulk solution, after which they tended to form aggregates that gradually settled due to gravity [26]. In order to avoid this aggregation process and to stabilize the nanoparticles, the copper solution was prepared with citrate buffer to suppress the formation of bulk copper sulfide. The results confirmed the presence of copper sulfide nanoparticles suspended with an approximate monomer size of less than 50 nm, though nanoaggregate formation occurred over several days, reaching a functional size of ~700 nm. The nanoparticles produced in this study were remarkably crystalline and homogenous compared with those of other synthetic, wet chemical routes [9,10,11].
5. Conclusions
The synthesis of copper sulfide nanoparticles through a simple one-step method based on biogenic H2S generated from a low-pH sulfidogenic bioreactor with minimal engineering complexity is possible. The modular system inoculated with an indigenous consortium can be configured to optimize bioremediation in order to remove sulfate and metals, while the excess waste-H2S generated as the by product is employed to produce high-value nanomaterials. Importantly, the sulfidogenic reactor can be easily scalable to produce “clean” nanoparticles free from bacterial cells.
Acknowledgments
This research was supported by Fondecyt, Chile (No.11150170). We thank the two anonymous reviewers for contributing to the presentation of this work.
Author Contributions
I.N. conceived the presented idea. I.N., G.S., P.O. and V.D. planned the experiments. C.C., D.G., V.D. and B.C. carried out the experiments. I.N. wrote the paper with support from G.S., P.O. and V.D.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Johnson, D.B.; Hallberg, K.B. The microbiology of acidic mine waters. Res. Microbiol. 2003, 154, 466–473. [Google Scholar] [CrossRef]
- Nancucheo, I.; Bitencourt, J.A.P.; Sahoo, P.K.; Alves, J.O.; Siqueira, J.O.; Oliveira, G. Recent developments for remediating acidic mine waters using sulfidogenic bacteria. Biomed. Res. Int. 2017. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.J.; Roussel, J.; Rolley, J.; Woodhall, F.; Mikheenko, I.P.; Johnson, D.B.; Gomez-Bolivar, J.; Merroun, M.L.; Macaskie, L.E. Biosynthesis of zinc sulfide quantum dots using waste off-gas from a metal bioremediation process. RSC Adv. 2017, 7, 21484–21491. [Google Scholar] [CrossRef]
- Nancucheo, I.; Johnson, D.B. Selective removal of transition metals from acidic mine waters by novel consortia of acidophilic sulfidogenic bacteria. Microb. Biotechnol. 2012, 5, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Widdel, F. Microbiology and ecology of sulfate-and sulfur-reducing bacteria. In Biology of Anaerobic Microorganisms; Zehnder, A.J.B., Ed.; John Wiley and Sons, Inc.: New York, NY, USA, 1988; pp. 469–585. ISBN 0471882267. [Google Scholar]
- Hedrich, S.; Johnson, D.B. Remediation and selective recovery of metals from acidic mine waters using novel modular bioreactors. Environ. Sci. Technol. 2014, 48, 12206–12212. [Google Scholar] [CrossRef] [PubMed]
- Nancucheo, I.; Rowe, O.F.; Hedrich, S.; Johnson, D.B. Solid and liquid media for isolating and cultivating acidophilic and acid-tolerant sulfate-reducing bacteria. FEMS Microbiol. Lett. 2016, 363, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Schaffie, M.; Hosseini, M.R. Biological process for synthesis of semiconductor copper sulfide nanoparticle from mine wastewaters. J. Environ. Chem. Eng. 2014, 2, 386–391. [Google Scholar] [CrossRef]
- Ajibade, P.A.; Botha, N.L. Synthesis and structural studies of copper sulfide nanocrystals. Results Phys. 2016, 6, 581–589. [Google Scholar] [CrossRef]
- Riyaz, S.; Parveen, A.; Azam, A. Microstructural and optical properties of CuS nanoparticles prepared by sol—Gel route. Perspect. Sci. 2016, 8, 632–635. [Google Scholar] [CrossRef]
- Pal, M.; Mathews, N.R.; Sanchez-Mora, E.; Pal, U.; Paraguay-Delgado, F.; Mathew, X. Synthesis of CuS nanoparticles by a wet chemical route and their photocatalytic activity. J. Nanopart. Res. 2015, 17, 301. [Google Scholar] [CrossRef]
- Zhou, N.Q.; Tian, L.J.; Wang, Y.C.; Li, D.B.; Li, P.P.; Zhang, X.; Yu, H.Q. Extracellular biosynthesis of copper sulfide nanoparticles by Shewanella oneidensis MR-1 as a photothermal agent. Enzyme Microb. Technol. 2016, 95, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Leiva, E.D.; Rámila, C.D.P.; Vargas, I.T.; Escauriaza, C.R.; Bonilla, C.A.; Pizarro, G.E.; Regan, J.M.; Pasten, P.A. Natural attenuation process via microbial oxidation of arsenic in a high Andean watershed. Sci. Total Environ. 2014, 466–467, 490–502. [Google Scholar] [CrossRef] [PubMed]
- Zamanova, M.K.; Glotova, V.N.; Izhenbina, T.N.; Krutas, D.S.; Novikov, V.T. Simultaneous HPLC-UV determination of lactic acid, glycolic acid, glycolide, lactide and ethyl acetate in monomers for producing biodegradable polymers. Procedia Chem. 2014, 10, 244–251. [Google Scholar] [CrossRef]
- Oliveira, B.M.; Barrio, E.; Querol, A.; Pérez-Torrado, R. Enhanced enzymatic activity of glycerol-3-phosphate dehydrogenase from the cryophilic Saccharomyces kudriavzevii. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Rowe, O.F.; Sánchez-España, J.; Hallberg, K.B.; Johnson, D.B. Microbial communities and geochemical dynamics in an extremely acidic, metal-rich stream at an abandoned sulfide mine (Huelva, Spain) underpinned by two functional primary production systems. Environ. Microbiol. 2007, 9, 1761–1771. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Andrea, I.; Stams, A.J.M.; Hedrich, S.; Nancucheo, I.; Johnson, D.B. Desulfosporosinus acididurans sp. nov.: An acidophilic sulfate-reducing bacterium isolated from acidic sediments. Extremophiles 2015, 19, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Anandham, R.; Indiragandhi, P.; Kwon, S.W.; Sa, T.M.; Jeon, C.O.; Kim, Y.K.; Jee, H.J. Pandoraea thiooxydans sp. nov., a facultatively chemolithotrophic, thiosulfate-oxidizing bacterium isolated from rhizosphere soils of sesame (Sesamum indicum L.). Int. J. Syst. Evol. Microbiol. 2010, 60, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Falagán, C.; Grail, B.M.; Johnson, D.B. New approaches for extracting and recovering metals from mine tailings. Miner. Eng. 2017, 106, 71–78. [Google Scholar] [CrossRef]
- Zepeda, V.J.; Nancucheo, I.; Guillen, M.; Becerra, E.; Escuti, C.; Cautivo, D.; González, D.; Colipai, C.; Demergasso, C.; Galleguillos, P.A. Biological production of copper sulfide concentrate from flotation tailings and low Grade ore. Solid State Phenom. 2017, 262, 202–206. [Google Scholar] [CrossRef]
- Alazard, D.; Joseph, M.; Battaglia-Brunet, F.; Cayol, J.L.; Ollivier, B. Desulfosporosinus acidiphilus sp. nov.: A moderately acidophilic sulfate-reducing bacterium isolated from acid mining drainage sediments. Extremophiles 2010, 14, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Kimura, S.; Hallberg, K.B.; Johnson, D.B. Sulfidogenesis in low pH (3.8–4.2) media by a mixed population of acidophilic bacteria. Biodegradation 2006, 17, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Nancucheo, I.; Johnson, D.B. Removal of sulfate from extremely acidic mine waters using low pH sulfidogenic bioreactors. Hydrometallurgy 2014, 150, 222–226. [Google Scholar] [CrossRef]
- Boonstra, J.; van Lier, R.; Janssen, G.; Dijkman, H.; Buisman, C.J.N. Biological treatment of acid mine drainage. In Biohydrometallurgy and the Environment Toward the Mining of the 21st Century (Process Metallurgy 9B); Amils, R., Ballester, A., Eds.; Elsevier: Elsevier, Amsterdam, The Nederland, 1999; pp. 559–567. ISBN 978-0-444-50193-6. [Google Scholar]
- Riekkola-Vanhanen, M. Talvivaara mining company—From a project to a mine. Miner. Eng. 2013, 48, 2–9. [Google Scholar] [CrossRef]
- Liyanage, D.D.; Thamali, R.J.K.A.; Kumbalatara, A.A.K.; Weliwita, J.A.; Witharana, S. An analysis of nanoparticle settling times in liquids. J. Nanomater. 2016, 2016. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).