The Fate of Platinum-Group Minerals in the Exogenic Environment—From Sulfide Ores via Oxidized Ores into Placers: Case Studies Bushveld Complex, South Africa, and Great Dyke, Zimbabwe
Abstract
1. Introduction
2. Materials and Methods
3. Results
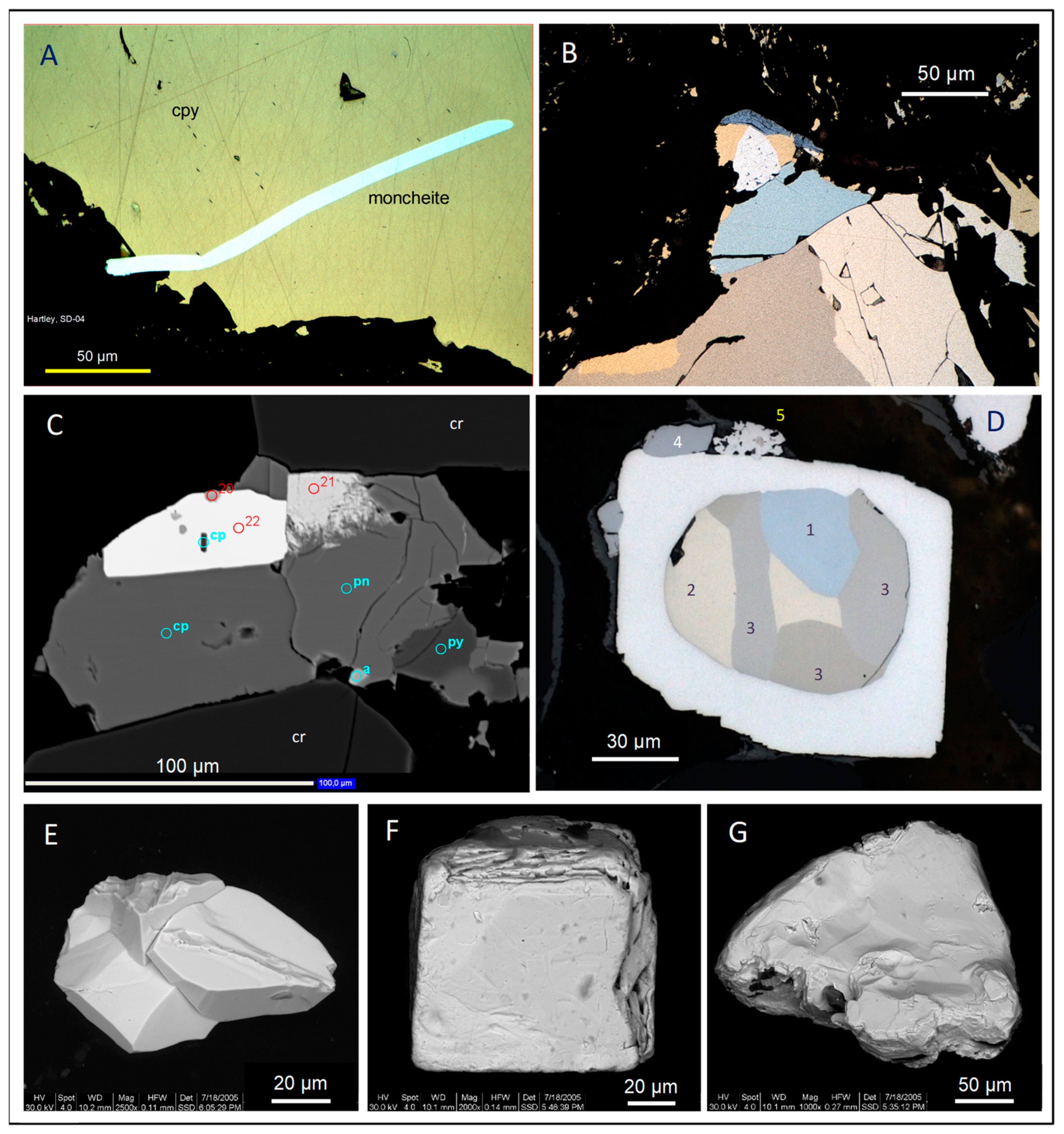
3.1. Pristine Sulfide Ores
- Bismuthides and tellurides mainly of Pt and Pd, called (Pt,Pd)-bismuthotellurides in the following, which show high degrees of Pt ↔ Pd and Te ↔ Bi substitution. A first group [MeX2] comprises moncheite [PtTe2], maslovite [PtBiTe], merenskyite [PdTe2], and michenerite [PdBiTe], and a second, rarer group [MeX] consists of kotulskite [PdTe]-sobolevskite [PdBi] (Figure 2A,B,G).
- Sulfides, encompassing the Pt–Pd monosulfides cooperate-braggite-vysotskite [PtS–(Pt,Pd,Ni)S–PdS] and called (Pt,Pd)-sulfides in the following (Figure 2B,C); malanite-cuprorhodsite solid solution [CuPt2S4–CuRh2S4] (Figure 2C); and laurite [RuS2], which often carries the bulk IPGE budget of the ores.
- Arsenides, the most ubiquitous representative being sperrylite [PtAs2] (Figure 2D,E).
- Sulfarsenides with limited substitution comprise the common Rh-mineral hollingworthite [RhAsS], rarer platarsite [PtAsS], irarsite [IrAsS], and ruarsite [RuAsS].
- Antimonides like stibiopalladinite [Pd5Sb2].
- Zvyagintsevite [Pd3Pb] and members of the solid solution rustenburgite-atokite [Pt3Sn–Pd3Sn] are locally abundant in the Bushveld ores.
- Alloys mainly of Pt with Fe, Cu, or Ni (Figure 2D,F), and inter-PGE alloys (e.g., Pt–Pd, Os–Ir–Ru).
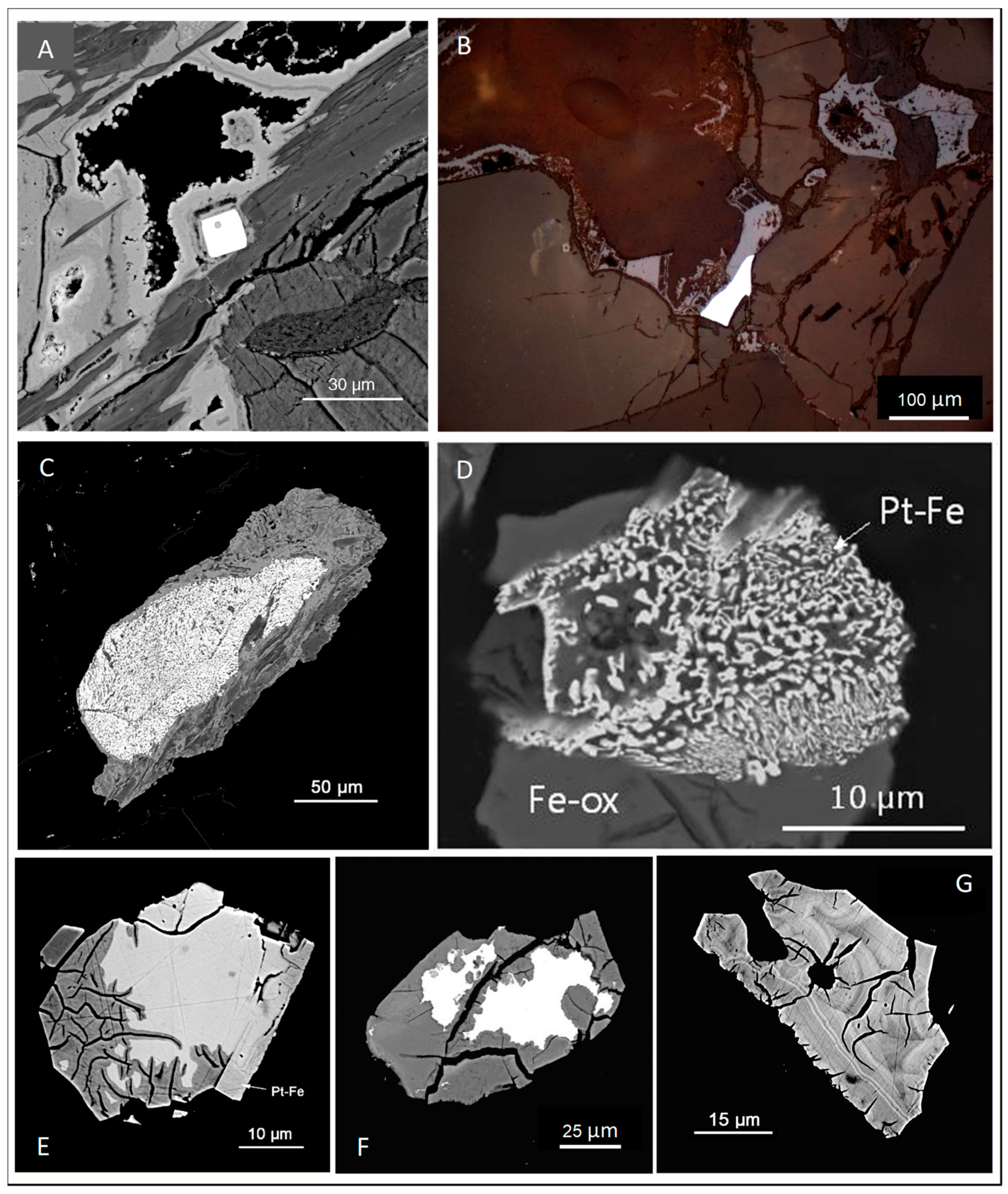
3.2. Oxidized Ores
Summary—PGM and PGE Carriers in Oxidized Ores
- (1)
- As relict primary PGM (mainly sperrylite, cooperite/braggite, and Pt–Fe alloys),
- (2)
- in solid solution in relict sulfides (dominantly Pd and Rh in pentlandite),
- (3)
- as secondary PGM neoformations (rare, mainly small grains, e.g., native Pt),
- (4)
- as PGE oxides/hydroxides that either replace primary PGM or represent neoformations,
- (5)
- in iron oxides/hydroxides (up to some thousand ppm Pt and Pd),
- (6)
- in iron/manganese oxides/hydroxides (up to 1.6% Pt and 1150 ppm Pd) [57], and
- (7)
- in secondary phyllosilicates (up to a few hundred ppm Pt and Pd).
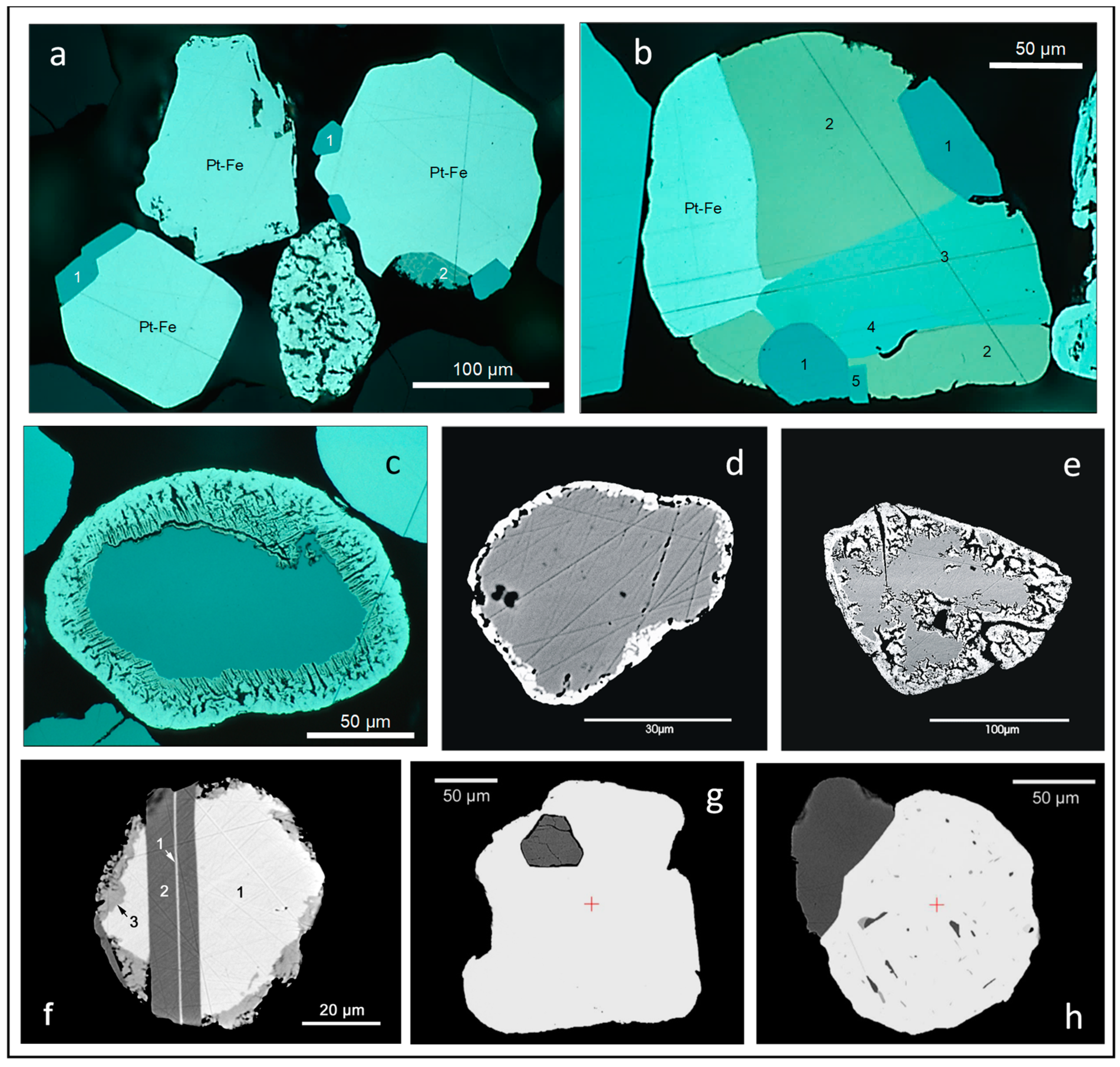
3.3. PGM Grains in Alluvial Sediments of Rivers Draining the Bushveld Complex and the Great Dyke
3.3.1. SEM Observations of Single Grains—Morphology, Intergrowths, Alteration, Modification
3.3.2. Detrital PGM: Intergrowths, Inclusions, Alteration, Modification
4. Discussion
- (1)
- the primary, pristine ores,
- (2)
- the near-surface oxidized/weathered ores, and,
- (3)
- associated placers.
- (1)
- originate from the primary ores (residual/allogenic/detrital grains), or,
- (2)
- developed during weathering/oxidation of the primary ores (authigenic grains), or,
- (3)
- were newly formed (authigenic grains/“neoformation”) in the alluvial placer environment.
4.1. Development of the Bushveld/Great Dyke PGM Assemblages in the Course of Weathering
4.2. Placer PGM Worldwide—An Overview
4.3. Placer PGM—Residual Grains (Allogenic/Detrital) or Authigenic (“Neoformation”)?
4.3.1. PGM in Laterites
4.3.2. PGM in Placers
- (i)
- Primary PGM formation within mafic/ultramafic intrusions, followed by weathering with no or only minor further alteration prior to alluvial concentration.
- (ii)
- Solution of the PGE during weathering of the source rocks, probably aided by organic reactions, followed by the supergene growth of the macroscopic grains within the weathering zone and final alluvial concentration.
4.3.3. PGE Mobility and PGM Neoformation—Concluding Remarks
5. Conclusions
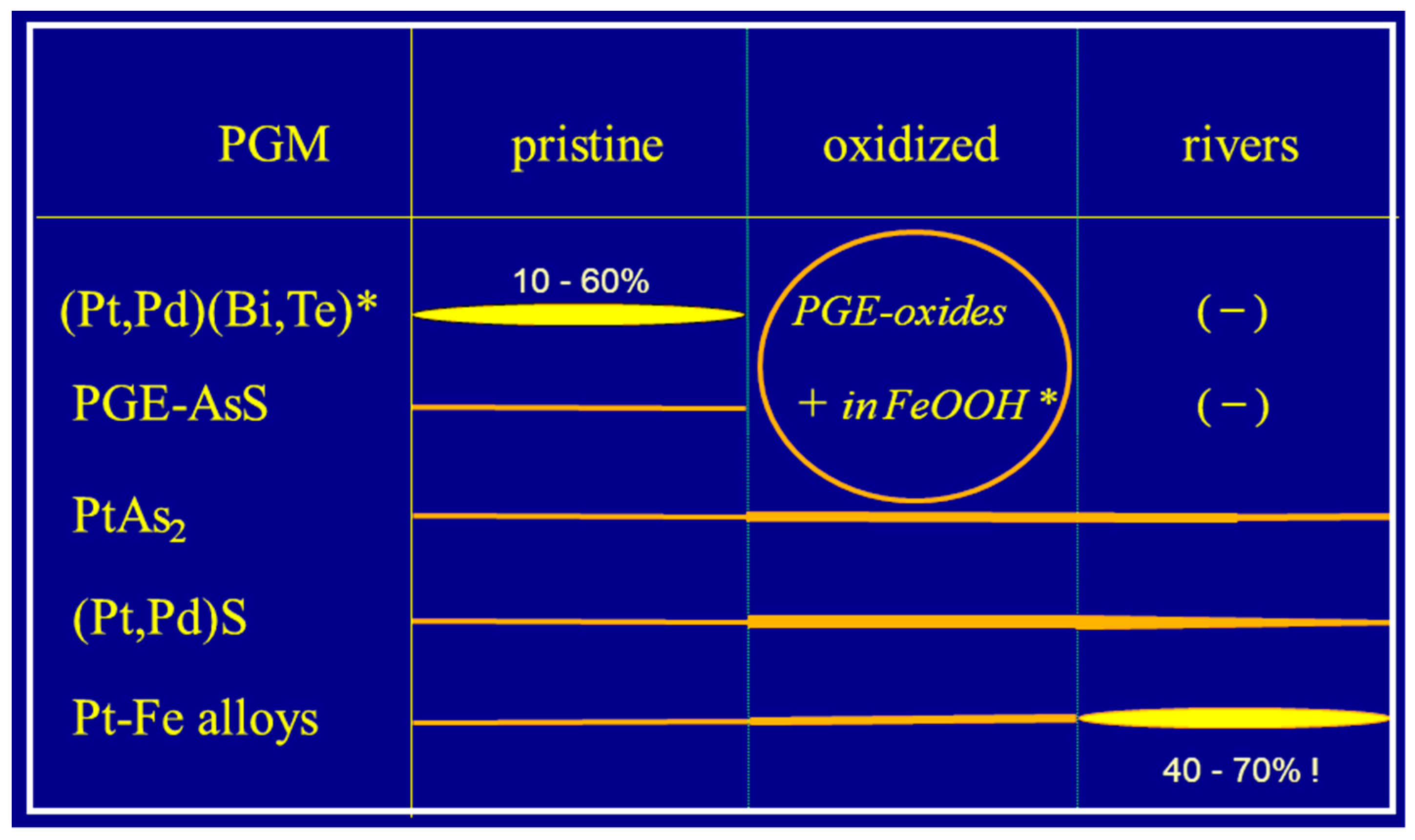
- In the pristine sulfide ores of both the Bushveld and the Great Dyke, the PGE are bimodally distributed: Pt and variable proportions of Pd and Rh are present in the form of discrete PGM, dominantly PGE-bismuthotellurides, -sulfides, -arsenides, -sulfarsenides, and -alloys, and substantial though variable proportions of Pd and Rh are hosted in pentlandite.
- Near surface, in the oxidized ores, the PGE become polymodally distributed: In the course of weathering of the ores, PGE-sulfides, -arsenides and Pt–Fe alloys remain the only relics of the pristine PGM assemblage. The base metal sulfides and the (Pt,Pd)-bismuthotellurides are destroyed, and ill-defined (Pt,Pd)-oxides or -hydroxides develop. Further, elevated contents of Pt and Pd are found in Fe/Mn/Co-oxides/hydroxides and smectites.
- In the alluvial sediments, the PGE are unimodally contained in discrete PGM. The assemblages of detrital PGM are characterized by partial alteration or destruction of most remaining PGE-sulfide grains, whereas sperrylite largely survives as a stable phase, and Pt–Fe alloy grains predominate.
- Accordingly, the order of decreasing stability in the supergene environment is as follows: (1) Pt–Fe and Os–Ir–Ru alloys (very stable) → (2) sperrylite (stable) → (3) cooperite/braggite (variably stable/”meta-stable”) → (4) PGE-bismuthotellurides, PGE-sulfarsenides, and PGE-oxides (unstable).
- In the Bushveld/Great Dyke case, and in all probability also worldwide, “neoformation”, i.e., authigenic growth, of discrete, larger PGM in both oxidized ores and placers plays no substantial role. Dissolution and redistribution of PGE is taking place, however, the newly-formed products are nano-sized particles, small crystallites, or rarely µm-sized grains primarily sited on substrates of precursor detrital/allogenic PGM grains, and they are of subordinate significance. Any growth to larger crystals or nuggets (mm size) was not observed, and in the Bushveld/Great Dyke case, any proof of PGM “neoformation” in a grand style is missing.
- The final PGM suite which survived the weathering process on route from sulfide ores via oxidized ores into placers results from the continuous elimination of unstable PGM and the dispersion of soluble PGE. Therefore, the alluvial PGM assemblage represents a PGM rest spectrum of residual, detrital grains.
Funding
Acknowledgments
Conflicts of Interest
References
- Mertie, J.B. Economic Geology of the Platinum Metals. US Geol. Surv. Prof. Pap. 1969, 63, 120. [Google Scholar]
- Cabri, L.J.; Harris, D.C.; Weiser, T.W. The mineralogy and distribution of Platinum Group Mineral (PGM) placer deposits of the world. Explor. Min. Geol. 1996, 5, 73–167. [Google Scholar]
- Weiser, T.W. Platinum-group minerals (PGM) in placer deposits. In The Geology, Geochemistry, Mineralogy and Mineral Beneficiation of Platinum-Group Elements; Cabri, L.J., Ed.; Canadian Institute of Mining, Metallurgy and Petroleum (CIM): Westmount, QC, Canada, 2002; CIM Special Volume 54, pp. 721–756. [Google Scholar]
- Bundesanstalt für Geowissenschaften und Rohstoffe (BGR), Hannover, Germany. BGR Data Bank, PGE Production 2017. Available online: www.bgr.bund.de (accessed on 12 August 2018).
- Merensky, H. The Various Platinum Occurrences on the Farm Maandagshoek No. 148; Unpublished Memorandum to Lydenburg Platinum Syndicate; Archives of the Merensky Trust: Duivelskloof, South Africa, 1924. [Google Scholar]
- Merensky, H. Die neuentdeckten Platinfelder im mittleren Transvaal und ihre wirtschaftliche Bedeutung. Zeitschrift der Deutschen Geologischen Gesellschaft 1926, 78, 296–314. [Google Scholar]
- Wagner, P.A. The Platinum Deposits and Mines of South Africa; Oliver & Boyd: London, UK, 1929; 326p. [Google Scholar]
- Cawthorn, R.G. The discovery of the platiniferous Merensky Reef in 1924. S. Afr. J. Geol. 1999, 102, 178–183. [Google Scholar]
- Scoates, J.S.; Friedman, R.M. Precise Age of the platiniferous Merensky Reef, Bushveld Complex. South Africa, by the U-Pb zircon chemical abrasion ID-TIMS technique. Econ. Geol. 2008, 103, 465–471. [Google Scholar] [CrossRef]
- Viljoen, M. The Bushveld Complex—Host to the world’s largest platinum, chromium and vanadium resources. Episodes 2016, 39, 239–268. [Google Scholar] [CrossRef]
- Naldrett, T.; Kinnaird, J.; Wilson, A.; Chunnett, G. Concentration of PGE in the Earth’s Crust with Special Reference to the Bushveld Complex. Earth Sci. Front. 2008, 15, 264–297. [Google Scholar] [CrossRef]
- Oberthür, T.; Junge, M.; Rudashevsky, N.; de Meyer, E.; Gutter, P. Platinum-group minerals in the LG and MG chromitites of the Bushveld Complex, South Africa. Miner. Depos. 2016, 51, 71–87. [Google Scholar] [CrossRef]
- Oberthür, T.; Davis, D.W.; Blenkinsop, T.G.; Höhndorf, A. Precise U-Pb mineral ages, Rb-Sr and Sm-Nd systematics for the Great Dyke, Zimbabwe—Constraints on late Archean events in the Zimbabwe Craton and Limpopo Belt. Precambrian Res. 2002, 113, 293–305. [Google Scholar] [CrossRef]
- Worst, B.G. The Great Dyke of Southern Rhodesia; Bulletin 47; Southern Rhodesian Geological Survey: Salisbury, Zimbabwe, 1960. [Google Scholar]
- Prendergast, M.D.; Wilson, A.H. The Great Dyke of Zimbabwe—II: Mineralisation and mineral deposits. In Magmatic Sulphides—The Zimbabwe Volume; Prendergast, M.D., Jones, M.J., Eds.; Institution Mining and Metallurgy: London, UK, 1989; pp. 21–42. [Google Scholar]
- Oberthür, T. Platinum-Group Element Mineralization of the Main Sulfide Zone, Great Dyke, Zimbabwe. Rev. Econ. Geol. 2011, 17, 329–349. [Google Scholar]
- Oberthür, T.; Weiser, T.W.; Gast, L.; Kojonen, K. Geochemistry and mineralogy of the platinum-group elements at Hartley Platinum Mine, Zimbabwe. Part 1: Primary distribution patterns in pristine ores of the Main Sulfide Zone of the Great Dyke. Miner. Depos. 2003, 38, 327–343. [Google Scholar] [CrossRef]
- Naldrett, A. Magmatic Sulphide Deposits—Geology, Geochemistry and Exploration; Springer: Berlin, Germany, 2004; p. 728. [Google Scholar]
- Godel, B.; Barnes, S.-J.; Maier, W.D. Platinum-Group Elements in Sulphide Minerals, Platinum-Group Minerals, and Whole-Rocks of the Merensky Reef (Bushveld Complex, South Africa): Implications for the Formation of the Reef. J. Petrol. 2007, 48, 1569–1604. [Google Scholar] [CrossRef]
- Osbahr, I.; Klemd, R.; Oberthür, T.; Brätz, H.; Schouwstra, R. Platinum-group element distribution in base-metal sulfides of the Merensky Reef from the eastern and western Bushveld Complex, South Africa. Miner. Depos. 2013, 48, 211–232. [Google Scholar] [CrossRef]
- Osbahr, I.; Oberthür, T.; Klemd, R.; Josties, A. Platinum-group element distribution in base-metal sulfides of the UG2, Bushveld Complex, South Africa—A reconnaissance study. Miner. Depos. 2014, 49, 655–665. [Google Scholar] [CrossRef]
- Junge, M.; Wirth, R.; Oberthür, T.; Melcher, F.; Schreiber, A. Mineralogical siting of platinum-group elements in pentlandite from the Bushveld Complex, South Africa. Miner. Depos. 2015, 50, 41–54. [Google Scholar] [CrossRef]
- Weiser, T.; Oberthür, T.; Kojonen, K.; Johanson, B. Distribution of trace PGE in pentlandite and of PGM in the Main Sulfide Zone (MSZ) at Mimosa Mine, Great Dyke, Zimbabwe. In 8th Internat Platinum Symposium; South African Institute Mining Metallurgy; Symposium Series S 18; Johannesburg, South Africa, 1998; pp. 443–445. [Google Scholar]
- Oberthür, T.; Melcher, F.; Buchholz, P.; Locmelis, M. The oxidized ores of the Main Sulfide Zone, Great Dyke, Zimbabwe: Turning resources into minable reserves—Mineralogy is the key. J. South. Afr. Inst. Min. Metall. 2013, 133, 191–201. [Google Scholar]
- Junge, M. The Fate of Platinum-Group Elements during Weathering Processes—With Special Focus on Pristine and Weathered Platreef Ores at the Mogalakwena Mine in the Bushveld Complex. Ph.D. Thesis, Leibniz-Universität Hannover, Hannover, Germany, 2017. 265p. [Google Scholar]
- Korges, M. Supergene Mobilization and Redistribution of Platinum-Group Elements in the Merensky Reef, Eastern Bushveld Complex, South Africa. Master’s Thesis, Martin-Luther-Universität Halle-Wittenberg, Halle, Germany, 2015. 116p. [Google Scholar]
- Oberthür, T.; Weiser, T.W.; Gast, L. Geochemistry and mineralogy of the platinum-group elements at Hartley Platinum Mine, Zimbabwe. Part 2: Supergene redistribution in the oxidized Main Sulfide Zone of the Great Dyke, and alluvial platinum-group minerals. Miner. Depos. 2003, 38, 344–355. [Google Scholar] [CrossRef]
- Oberthür, T.; Melcher, F.; Gast, L.; Wöhrl, C.; Lodziak, J. Detrital platinum-group minerals in rivers draining the eastern Bushveld Complex, South Africa. Can. Mineral. 2004, 42, 563–582. [Google Scholar] [CrossRef]
- Oberthür, T.; Weiser, T.W.; Melcher, F.; Gast, L.; Wöhrl, C. Detrital platinum-group minerals in rivers draining the Great Dyke, Zimbabwe. Can. Mineral. 2013, 51, 197–222. [Google Scholar] [CrossRef]
- Oberthür, T.; Weiser, T.W.; Melcher, F. Alluvial and Eluvial Platinum-Group Minerals (PGM) from the Bushveld Complex, South Africa. S. Afr. J. Geol. 2014, 117, 255–274. [Google Scholar] [CrossRef]
- Naldrett, A.J.; Kinnaird, J.; Wilson, A.; Yudovskaya, M.; McQuade, S.; Chunnett, G.; Stanley, C. Chromite composition and PGE content of Bushveld chromitites: Part 1—the Lower and Middle Groups. Appl. Earth Sci. (Trans. Inst. Min. Metall. B) 2009, 118, 131–161. [Google Scholar] [CrossRef]
- Hazen, R.M.; Grew, E.S.; Downs, R.T.; Golden, J.; Hystad, G. Mineral ecology: Chance and necessity in the mineral diversity of terrestrial planets. Can. Mineral. 2015, 53, 295–324. [Google Scholar] [CrossRef]
- Schouwstra, R.; Kinloch, E.D.; Lee, C. A short geological review of the Bushveld Complex. Platin. Met. Rev. 2000, 44, 33–39. [Google Scholar]
- Kinloch, E.D. Regional Trends in the Platinum-Group Mineralogy of the Critical Zone of the Bushveld Complex, South Africa. Econ. Geol. 1982, 77, 1328–1347. [Google Scholar] [CrossRef]
- Kinloch, E.D.; Peyerl, W. Platinum-group minerals in various rocks of the Merensky Reef: Genetic implications. Econ. Geol. 1990, 85, 537–555. [Google Scholar] [CrossRef]
- Rose, D.; Viljoen, F.; Knoper, M.; Rajesh, H. Detailed assessment of platinum-group minerals associated with chromitite stringers in the Merensky Reef of the eastern Bushveld Complex, South Africa. Can. Miner. 2011, 49, 1385–1396. [Google Scholar] [CrossRef]
- Voordouw, R.J.; Gutzmer, J.; Beukes, N.J. Zoning of platinum group mineral assemblages in the UG2 chromitite determined through in situ SEM-EDS-based image analysis. Miner. Depos. 2010, 45, 47–159. [Google Scholar] [CrossRef]
- Junge, M.; Oberthür, T.; Melcher, F. Cryptic variation of chromite chemistry, platinum-group-element and -mineral distribution in the UG-2 chromitite—An example from the Karee Mine, western Bushveld Complex, South Africa. Econ. Geol. 2014, 109, 795–810. [Google Scholar] [CrossRef]
- Kottke-Levin, J.; Tredoux, M.; Gräbe, P.-J. An investigation of the geochemistry of the Middle Group of the eastern Bushveld Complex, South Africa Part 1—The chromitite layers. Appl. Earth Sci. Trans. Inst. Min. Metall. B 2009, 118, 111–130. [Google Scholar] [CrossRef]
- Junge, M.; Oberthür, T.; Osbahr, I.; Gutter, P. Platinum-group elements and minerals in the Lower and Middle Group chromitites of the western Bushveld Complex, South Africa. Miner. Depos. 2016, 51, 841–852. [Google Scholar] [CrossRef]
- McDonald, I.; Holwell, D.A.; Armitage, P.E.B. Geochemistry and mineralogy of the Platreef and ‘Critical Zone’ of the northern lobe of the Bushveld Complex, South Africa: Implications for Bushveld stratigraphy and the development of PGE mineralisation. Miner. Depos. 2005, 40, 526–549. [Google Scholar] [CrossRef]
- Kinnaird, J.; Hutchinson, D.; Schurmann, L.; Nex, P.A.M.; de Lange, R. Petrology and mineralization of the southern Platreef, northern limb of the Bushveld Complex, South Africa. Miner. Depos. 2005, 40, 576–597. [Google Scholar] [CrossRef]
- Holwell, D.A.; McDonald, I.; Armitage, P.E.B. Platinum-group mineral assemblages in the Platreef at the South-Central Pit, Sandsloot Mine, northern Bushveld Complex, South Africa. Mineral. Mag. 2006, 70, 83–101. [Google Scholar] [CrossRef]
- Manyeruke, T. Compositional and Lithological Variation of the Platreef on the Farm Nonnenwerth, Northern Lobe of the Bushveld Complex: Implications for the Origin of the Platinum-Group Element Mineralization. Ph.D. Thesis, University of Pretoria, Pretoria, South Africa, 2007. 248p. [Google Scholar]
- van der Merwe, F.; Viljoen, F.; Knoper, M. The mineralogy and mineral associations of platinum group elements and gold in the Platreef at Zwartfontein, Akanani Project, Northern Bushveld Complex, South Africa. Mineral. Petrol. 2012, 106, 25–38. [Google Scholar] [CrossRef]
- Klemd, R.; Junge, M.; Oberthür, T.; Herderich, T.; Schouwstra, R.; Roberts, J. Platinum-group element concentrations in base-metal sulphides from the Platreef, Mogalakwena Platinum Mine, Bushveld Complex, South Africa. S. Afr. J. Geol. 2016, 119, 623–638. [Google Scholar] [CrossRef]
- Tarkian, M.; Stumpfl, E. Platinum mineralogy of the Driekop mine, South Africa. Miner. Depos. 1975, 10, 71–85. [Google Scholar] [CrossRef]
- Stumpfl, E.; Rucklidge, J.C. The platiniferous dunite pipes of the eastern Bushveld. Econ. Geol. 1982, 77, 1419–1431. [Google Scholar]
- Melcher, F.; Lodziak, J. Platinum-group minerals of concentrates from the Driekop platinum pipe, Eastern Bushveld Complex—Tribute to Eugen F. Stumpfl. Neues Jahrbuch für Mineralogie Abhandlungen 2007, 183, 173–195. [Google Scholar] [CrossRef]
- Oberthür, T.; Melcher, F.; Sitnikova, M.; Rudashevsky, N.S.; Rudashevsky, V.N.; Cabri, L.J.; Lodziak, J.; Klosa, D.; Gast, L. Combination of Novel Mineralogical Methods in the Study of Noble Metal Ores—Focus on Pristine (Bushveld, Great Dyke) and Placer Platinum Mineralisation. In Proceedings of the Ninth International Congress for Applied Mineralogy, Brisbane, QLD, Australia, 8–10 September 2008; pp. 187–193. [Google Scholar]
- Coghill, B.M.; Wilson, A.H. Platinum-group minerals in the Selukwe subchamber, Great Dyke, Zimbabwe: Implications for PGE collection mechanisms and post-formational redistribution. Mineral. Mag. 1993, 57, 613–633. [Google Scholar] [CrossRef]
- Oberthür, T.; Cabri, L.J.; Weiser, T.; McMahon, G.; Müller, P. Pt, Pd and other trace elements in sulfides of the Main Sulfide Zone, Great Dyke, Zimbabwe—A reconnaissance study. Can. Mineral. 1997, 35, 597–609. [Google Scholar]
- Kuhlmann, G.; Oberthür, T.; Melcher, F.; Lodziak, J. Bushveld Komplex, Südafrika: UG2-Chromitit-Horizont, Mineralogisch—Geochemische Feinstratigraphie—Schwerpunkt Platinmetall-Verteilung; Unpublished Internal Report Tgb.-Nr. 11327/06; Bundesanstalt für Geowissenschaften und Rohstoffe (BGR): Hannover, Germany, 2006; 166p. [Google Scholar]
- Holwell, D.A.; McDonald, I. Distribution of platinum-group elements in the Platreef at Overysel, northern Bushveld complex: A combined PGM and LA-ICP-MS study. Contrib. Mineral. Petrol. 2007, 154, 171–190. [Google Scholar] [CrossRef]
- Fuchs, W.A.; Rose, A.W. The geochemical behaviour of platinum and palladium in the weathering cycle in the Stillwater Complex, Montana. Econ. Geol. 1974, 69, 332–346. [Google Scholar] [CrossRef]
- Hey, P. The effects of weathering on the UG-2 chromitite reef with special reference to the platinum-group minerals. S. Afr. J. Geol. 1999, 102, 251–260. [Google Scholar]
- Locmelis, M.; Melcher, F.; Oberthür, T. Platinum-Group Element Distribution in the Oxidized Main Sulfide Zone, Great Dyke, Zimbabwe. Miner. Depos. 2010, 45, 93–109. [Google Scholar] [CrossRef]
- Cousins, C.A.; Kinloch, E.D. Some observations on textures and inclusions in alluvial platinoids. Econ. Geol. 1976, 71, 1377–1398. [Google Scholar] [CrossRef]
- Schneiderhöhn, H.; Moritz, H. Die Oxydationszone im platinführenden Sulfidpyroxenit (Merensky-Reef) des Bushvelds in Transvaal. Zentralblatt Mineralogie Geologie Paláontologie 1939, Abteilung A, 1–12. [Google Scholar]
- Evans, D.M.; Buchanan, D.L.; Hall, G.E.M. Dispersion of platinum, palladium and gold from the Main Sulphide Zone, Great Dyke, Zimbabwe. Trans. Inst. Min. Metall. 1994, 103, B57–B67. [Google Scholar]
- Oberthür, T.; Melcher, F. PGE and PGM in the supergene environment: The Great Dyke, Zimbabwe. In Exploration for Platinum-Group Element Deposits; Mungall, J.E., Ed.; Short Course Series; Mineral Association of Canada: Quebec City, QC, Canada, 2005; Volume 35, pp. 97–112. [Google Scholar]
- Weiser, T. The quantitative proof of the existence of PGE-oxides. In Proceedings of the 6th Internat Platinum Symposium, Perth, Australia, 8–11 July1991; Abstract Volume 52. [Google Scholar]
- Jedwab, J. Oxygenated platinum-group element and transition metal (Ti, Cr, Mn, Co, Ni) compounds in the supergene domain. Chronique de la Recherche Minière 1995, 520, 47–53. [Google Scholar]
- Augé, T.; Legendre, O. Platinum-group element oxides from the Pirogues ophiolitic mineralization, New Caledonia: Origin and significance. Econ. Geol. 1994, 89, 1454–1468. [Google Scholar] [CrossRef]
- Augé, T.; Maurizot, P.; Breton, J.; Eberlé, J.-M.; Gilles, C.; Jézéquel, P.; Mézière, J.; Robert, M. Magmatic and supergene platinum-group minerals in the New Caledonia ophiolite. Chronique de la Recherche Miniere 1995, 520, 3–26. [Google Scholar]
- Evans, D.M.; Spratt, J. Platinum and palladium oxides/hydroxides from the Great Dyke, Zimbabwe, and thoughts on their stability and possible extraction. In Applied Mineralogy; AA Balkema: Rotterdam, The Netherlands, 2000; pp. 289–292. [Google Scholar]
- McDonald, I.; Ohnenstetter, D.; Ohnenstetter, M.; Vaughan, D.J. Palladium Oxides in Ultramafic Complexes Near Lavatrafo, Western Andriamena, Madagascar. Mineral. Mag. 1999, 63, 345–352. [Google Scholar] [CrossRef]
- Cabral, A.R.; Lehmann, B.; Kwitko, R.; Jones, R.; Pires, F.; Rocha Filho, O.; Innocentini, M. Palladium-oxygenated compounds of the Gongo Soco mine, Quadrilátero Ferrífero, central Minas Gerais, Brazil. Mineral. Mag. 2001, 65, 169–179. [Google Scholar] [CrossRef]
- Jedwab, J.; Criddle, A.J.; du Ry, P.; Piret, P.; Stanley, C.J. Rediscovery of palladinite (PdO tetrag.) from Itabira (Minas Gerais, Brazil) and from Ruwe (Shaba, Zaire). In Proceedings of the IAGOD Meeting, Orleans, France, 6 September 1994. [Google Scholar]
- Gregor, S. Evaluation of PGE Mineralization in the Oxidized Main Sulfide Zone at Mimosa Mine, Great Dyke, Zimbabwe; Unpublished Internal File 10748/04; Bundesanstalt für Geowissenschaften und Rohstoffe (BGR): Hannover, Germany, 2004; 55p. [Google Scholar]
- Melcher, F.; Oberthür, T.; Lodziak, J. Modification and alteration of detrital platinum-group minerals from the Eastern Bushveld Complex, South Africa. Can. Mineral. 2005, 43, 1711–1734. [Google Scholar] [CrossRef]
- Möller, P.; Kersten, G. Electrochemical accumulation of visible gold on pyrite and arsenopyrite surfaces. Miner. Depos. 1994, 29, 404–413. [Google Scholar] [CrossRef]
- Nixon, G.T.; Cabri, L.J.; Laflamme, J.H.G. Platinum-group element mineralization in lode and placer deposits associated with the Tulameen Alaskan-type complex, British Columbia. Can. Mineral. 1990, 28, 503–535. [Google Scholar]
- Malitch, K.N.; (Institute of Geology and Geochemistry, UB RAS, Ekaterinburg, Russia). Personal communication, 2016.
- Hagen, D.; Weiser, T.; Than, H. Platinum-group minerals in Quaternary gold placers in the upper Chindwin area of northern Burma. Mineral. Petrol. 1990, 42, 265–286. [Google Scholar] [CrossRef]
- Weiser, T.W.; Bachmann, H.-G. Platinum-group minerals from the Aikora River area, Papua New Guinea. Can. Mineral. 1999, 37, 1131–1145. [Google Scholar]
- Oberthür, T.; Melcher, F.; Weiser, T. Detrital platinum-group minerals and gold in placers of south-eastern Samar Island, Philippines. Can. Mineral. 2017, 54, 45–62. [Google Scholar] [CrossRef]
- Oberthür, T.; Melcher, F.; Goldmann, S.; Wotruba, H.; Dijkstra, A.; Gerdes, A.; Dale, C. Mineralogy and mineral chemistry of detrital heavy minerals from the Rhine River in Germany as evidence of their provenance, sedimentary and depositional history: Focus on platinum-group minerals and remarks on cassiterite, columbite-group minerals, and uraninite. Int. J. Earth Sci. 2016, 105, 637–657. [Google Scholar]
- Feather, C.E. Mineralogy of Platinum-Group Minerals in the Witwatersrand, South Africa. Econ. Geol. 1976, 71, 1399–1428. [Google Scholar] [CrossRef]
- O’Driscoll, B.; González-Jiménez, J.M. Petrogenesis of the Platinum-Group Minerals. Rev. Mineral. Geochem. 2016, 81, 489–578. [Google Scholar] [CrossRef]
- Salpeteur, I.; Martel-Jantin, B.; Rakotomanana, D. Pt and Pd mobility in ferralitic soils of the West Andriamina Area (Madagascar). Evidence of a supergene origin of some Pt and Pd Minerals. Chronique de la Recherches Minière 1995, 520, 27–45. [Google Scholar]
- Aiglsperger, T.; Proenza, J.A.; Zaccarini, F.; Lewis, J.F.; Garuti, G.; Labrador, M.; Longo, F. Platinum group minerals (PGM) in the Falcondo Ni laterite deposit, Loma Caribe peridotite (Dominican Republic). Miner. Depos. 2015, 50, 105–123. [Google Scholar] [CrossRef]
- Aiglsperger, T.; Proenza, J.A.; Font-Bardia, M.; Baurier-Aymat, S.; Galí, S.; Lewis, J.F.; Longo, F. Supergene neoformation of Pt-Ir-Fe-Ni alloys: Multistage grains explain nugget formation in Ni-laterites. Miner. Depos. 2017, 52, 1069–1083. [Google Scholar] [CrossRef]
- Ramdohr, P. A widespread mineral association, connected with serpentinization. Neues Jahrbuch Mineralogie Abhandlungen 1967, 107, 241–265. [Google Scholar]
- Eckstrand, O.R. The Dumont serpentinite: A model for control of nickeliferous opaque mineral assemblages by alteration reactions in ultramafic rocks. Econ. Geol. 1975, 70, 183–201. [Google Scholar] [CrossRef]
- Ahmed, Z.; Bevan, J.C. Awaruite, iridian awaruite and a new Ru-Os-Ir-Ni-Fe alloy from the Sakhakot-Qila complex, Malakand agency, Pakistan. Mineral. Mag. 1981, 44, 225–230. [Google Scholar] [CrossRef]
- Weiser, T.W. Platinum-Group Minerals (PGM) from placer deposits in the mineral collection of the Museum of Natural History, Vienna, Austria. Annalen Naturhist. Museum Wien 2004, 105A, 1–28. [Google Scholar]
- Malitch, K.N.; Thalhammer, O.A.R. Pt–Fe nuggets from clinopyroxenite–dunite massifs, Russia: A structural, compositional and osmium-isotope study. Can. Mineral. 2002, 40, 395–417. [Google Scholar] [CrossRef]
- Daouda Traoré, D.; Beauvais, A.; Augé, T.; Parisot, J.C.; Colin, F.; Cathelineau, M. Chemical and physical transfers in an ultramafic rock weathering profile: Part 2. Dissolution vs. accumulation of platinum group minerals. Am. Mineral. 2008, 93, 31–38. [Google Scholar] [CrossRef]
- Okrugin, A.V. Origin of platinum-group minerals in mafic and ultramafic rocks: From dispersed elements to nuggets. Can. Mineral. 2011, 49, 1397–1412. [Google Scholar] [CrossRef]
- Augusthitis, S.S. Mineralogical and geochemical studies of the platiniferous dunite-birbirite-pyroxenite complex of Yubdo, Birbir, W. Ethiopia. Chemie der Erde 1965, 24, 159–165. [Google Scholar]
- Ottemann, J.; Augusthitis, S.S. Geochemistry and origin of “platinum-nuggets” in laterite covers from ultrabasic rocks and birbirites of W. Ethiopia. Miner. Depos. 1967, 1, 269–277. [Google Scholar] [CrossRef]
- Stumpfl, E.F. The genesis of platinum deposits: Further thoughts. Miner. Sci. Eng. 1974, 6, 120–141. [Google Scholar]
- Bowles, J.F.W. The development of platinum-group minerals in laterites. Econ. Geol. 1986, 81, 1278–1285. [Google Scholar] [CrossRef]
- Bowles, J.F.W. Platinum–iron alloys, their structural and magnetic characteristics in relation to hydrothermal and low-temperature genesis. Mineral. Petrol. 1990, 43, 37–47. [Google Scholar] [CrossRef]
- Bowles, J.F.W. The development of platinum-group minerals (PGM) in laterites: Mineral morphology. Chronique de la Recherche Minière 1995, 520, 55–63. [Google Scholar]
- Bowles, J.F.W.; Suárez, S.; Prichard, H.M.; Fisher, P.C. The mineralogy, geochemistry and genesis of the alluvial platinum-group minerals of the Freetown Layered Complex, Sierra Leone. Mineral. Mag. 2018, 82, 223–246. [Google Scholar] [CrossRef]
- Bowles, J.F.W.; Suárez, S.; Prichard, H.M.; Fisher, P.C. Inclusions in an isoferroplatinum nugget from the Freetown Layered Complex, Sierra Leone. Mineral. Mag. 2018, 82, 577–592. [Google Scholar] [CrossRef]
- Mardock, C.L.; Barker, J.C. Theories on the transport and deposition of gold and PGM minerals in offshore placers near Goodnews Bay, Alaska. Ore Geol. Rev. 1991, 6, 211–227. [Google Scholar] [CrossRef]
- McClenaghan, M.B.; Cabri, L.J. Review of gold and platinum group element (PGE) indicator minerals methods for surficial sediment sampling. Geochem. Explor. Environ. Anal. 2011, 11, 251–263. [Google Scholar] [CrossRef]
- Zhmodik, S.M.; Nesterenko, G.V.; Airiyants, E.V.; Belyanin, D.K.; Kolpakov, V.V.; Podlipsky, M.Y.; Karmanov, N.S. Alluvial platinum-group minerals as indicators of primary PGE mineralization (placers of southern Siberia). Russ. Geol. Geophys. 2016, 57, 1437–1464. [Google Scholar] [CrossRef]
- Nesterenko, G.V.; Zhmodik, S.M.; Airiyants, E.V.; Belyanin, D.K.; Kolpakov, V.V.; Bogush, A.A. Colloform high-purity platinum from the placer deposit of Koura River (Gornaya Shoriya, Russia). Ore Geol. Rev. 2017, 91, 236–245. [Google Scholar] [CrossRef]
- Reith, F.; Zammit, C.-M.; Shar, S.S.; Etschmann, B.; Bottrill, R.; Southam, G.; Ta, C.; Kilburn, M.; Oberthür, T.; Ball, A.S.; et al. Biological role in the transformation of platinum-group-mineral grains. Nat. Geosci. 2016, 9, 294–298. [Google Scholar] [CrossRef]
- Yudovskaya, M.A.; Kinnaird, J.A.; Grobler, D.F.; Costin, G.; Abramova, V.D.; Dunnett, T.; Barnes, S.-J. Zonation of Merensky-Style Platinum-Group Element Mineralization in Turfspruit Thick Reef Facies (Northern Limb of the Bushveld Complex). Econ. Geol. 2017, 112, 1333–1365. [Google Scholar] [CrossRef]
- McCreesh, M.J.G.; Yudovskaya, M.A.; Kinnaird, J.A.; Reinke, C. Platinum-group minerals of the F and T zones, Waterberg Project, Far Northern Bushveld Complex: Implication for the formation of the PGE mineralisation. Mineral. Mag. 2018, 82, 539–575. [Google Scholar] [CrossRef]
- Tistl, M. Geochemistry of platinum-group elements of the zoned ultramafic Alto Condoto Complex, Northwest Colombia. Econ. Geol. 1994, 89, 158–167. [Google Scholar] [CrossRef]
- Burgath, K.P.; Salinas, R. The Condoto Complex in Chocó, Colombia: A Pt-bearing Alaskan-type intrusion. Zeitschrift für Angewandte Geologie 2000, Sonderheft SH 1, 163–170. [Google Scholar]
- Cabri, L.J. The platinum-group minerals. In Canadian Institute of Mining, Metallurgy and Petroleum, CIM Special Volume; Cabri, L.J., Ed.; CIM: Westmount, QC, Canada, 2002; Volume 54, pp. 13–129. [Google Scholar]
- Hattori, K.; Cabri, L.J.; Hart, S.R. Osmium isotope ratios of PGM grains associated with the Freetown Layered Complex, Sierra Leone, and their origin. Contrib. Mineral. Petrol. 1991, 109, 10–18. [Google Scholar] [CrossRef]
- Cabral, A.R.; Beaudoin, G.; Choquette, M.; Lehmann, B.; Polônia, J.C. Supergene leaching and formation of platinum in alluvium: Evidence from Serro, Minas Gerais, Brazil. Mineral. Petrol. 2007, 90, 141–150. [Google Scholar] [CrossRef]
- Cabral, A.R.; Lehmann, B.; Tupinambá, M.; Schlosser, S.; Kwitko-Ribeiro, R.; De Abreu, F.R. The platiniferous Au-Pd belt of Minas Gerais, Brazil, and genesis of its botryoidal Pt-Pd aggregates. Econ. Geol. 2009, 104, 1265–1276. [Google Scholar] [CrossRef]
- Fleet, M.E.; De Almeida, C.M.; Angeli, N. Botryoidal platinum, palladium and potarite from the Bom Sucesso stream, Minas Gerais, Brazil: Compositional zoning and origin. Can. Mineral. 2002, 40, 341–355. [Google Scholar] [CrossRef]
- Cabral, A.R.; Radtke, M.; Munnik, F.; Lehmann, B.; Reinholz, U.; Riesemeier, H.; Tupinambá, M.; Kwitko-Ribeiro, R. Iodine in alluvial Pt-Pd nuggets: Evidence for biogenic precious-metal fixation. Chem. Geol. 2011, 281, 125–132. [Google Scholar] [CrossRef]



| PGM Locality | Ru–Os–Ir Alloys | Pt–Fe Alloys | PtAs2 | RuS2–OsS2 | PGE Sulfides | PGE–AsS | Others | Ref. | |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Great Dyke Rivers | 42 | 46 | x | 7 | 5 | [29] | ||
| 2 | Bushveld Rivers | 54 | 33 | 11 | 2 | [30] | |||
| 3 | Tulameen, Canada | 2 | 98 | [73] | |||||
| 4 | Choco, Columbia | 1 | 97 | 1 | 1 | x | x | [2] | |
| 5 | Urals, Russia | 2 | 97 | 1 | [74] | ||||
| 6 | Chindwin, Burma | 96 | x | x | x | x | 4 | [75] | |
| 7 | Aikora River, PNG | 88 | 12 | [76] | |||||
| 8 | Samar, Philippines | 41 | 40 | 17 | 2 | [77] | |||
| 9 | Rhine River | 70 | 15 | 10 | x | 5 | [78] | ||
| 10 | Witwatersrand | 80 | 10 | 10 | x | x | [79] |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oberthür, T. The Fate of Platinum-Group Minerals in the Exogenic Environment—From Sulfide Ores via Oxidized Ores into Placers: Case Studies Bushveld Complex, South Africa, and Great Dyke, Zimbabwe. Minerals 2018, 8, 581. https://doi.org/10.3390/min8120581
Oberthür T. The Fate of Platinum-Group Minerals in the Exogenic Environment—From Sulfide Ores via Oxidized Ores into Placers: Case Studies Bushveld Complex, South Africa, and Great Dyke, Zimbabwe. Minerals. 2018; 8(12):581. https://doi.org/10.3390/min8120581
Chicago/Turabian StyleOberthür, Thomas. 2018. "The Fate of Platinum-Group Minerals in the Exogenic Environment—From Sulfide Ores via Oxidized Ores into Placers: Case Studies Bushveld Complex, South Africa, and Great Dyke, Zimbabwe" Minerals 8, no. 12: 581. https://doi.org/10.3390/min8120581
APA StyleOberthür, T. (2018). The Fate of Platinum-Group Minerals in the Exogenic Environment—From Sulfide Ores via Oxidized Ores into Placers: Case Studies Bushveld Complex, South Africa, and Great Dyke, Zimbabwe. Minerals, 8(12), 581. https://doi.org/10.3390/min8120581




