Mineralogical Characterization and Dissolution Experiments in Gamble’s Solution of Tremolitic Amphibole from Passo di Caldenno (Sondrio, Italy)
Abstract
1. Introduction
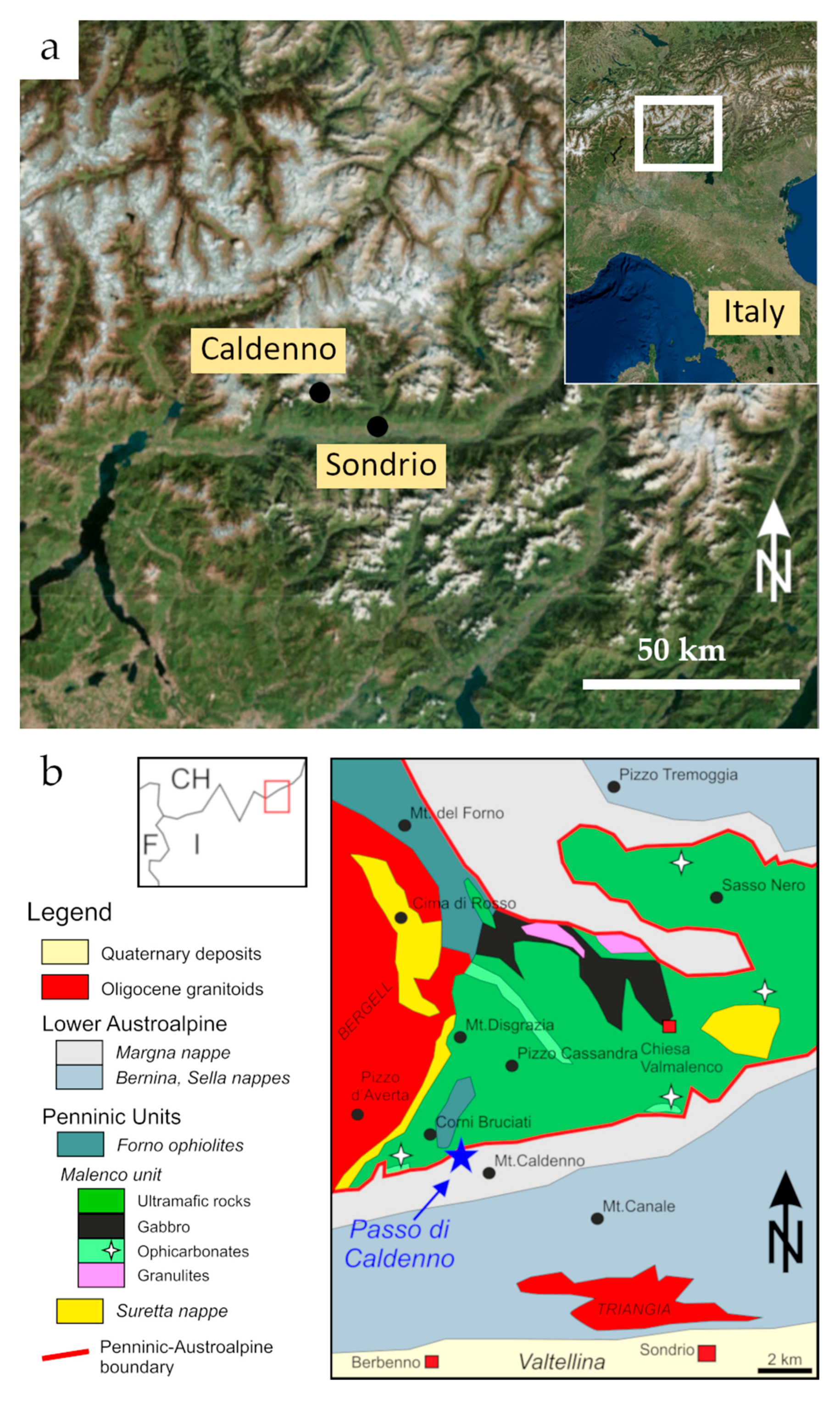
Geological Setting
2. Materials and Methods
2.1. VLM
2.2. XRPD
2.3. EPMA
2.4. µ-Raman and FTIR Spectroscopy
2.5. BET Surface Area Analysis
2.6. Laser Diffraction
2.7. FE-ESEM-EDXS
2.8. ICP-OES
3. Results
3.1. Naked-Eye and VLM Description of the Sample
3.2. XRPD
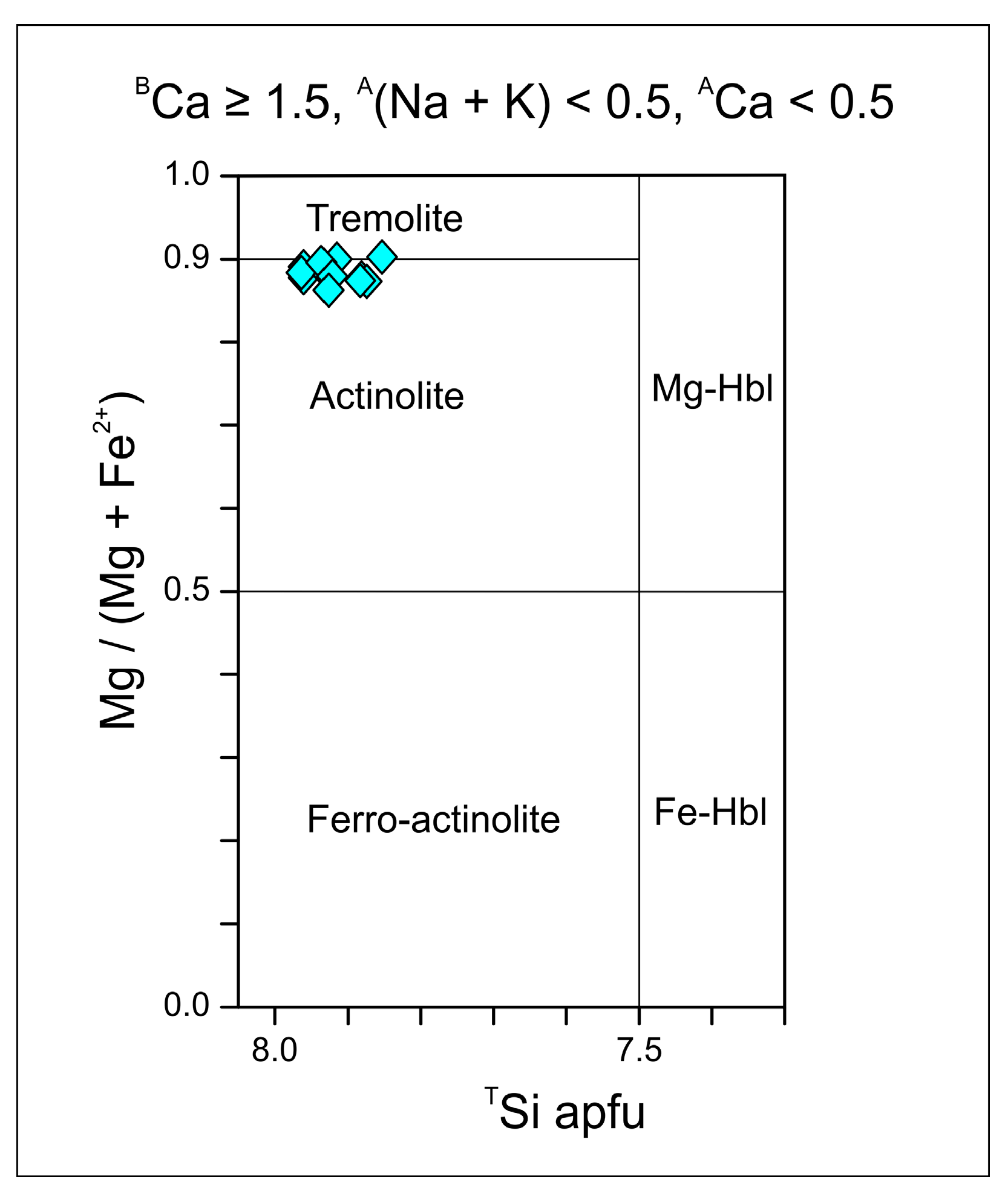
3.3. EPMA
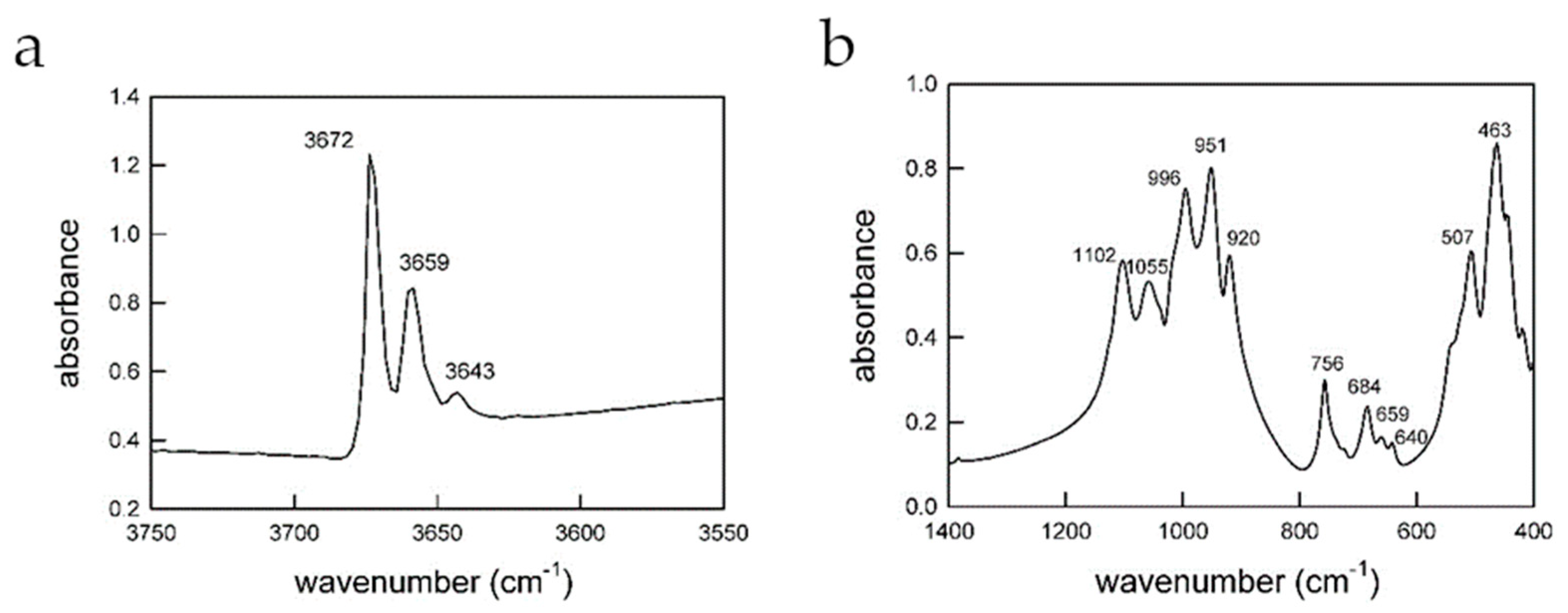
3.4. µ-Raman and FTIR Spectroscopic Observations
3.5. BET Surface Area Results
3.6. Particle-Size Distribution
3.7. Morphology as Observed by FE-ESEM for the Powdered Starting Material
3.8. Dimensional Results Obtained by FE-ESEM for the Powdered Starting Material
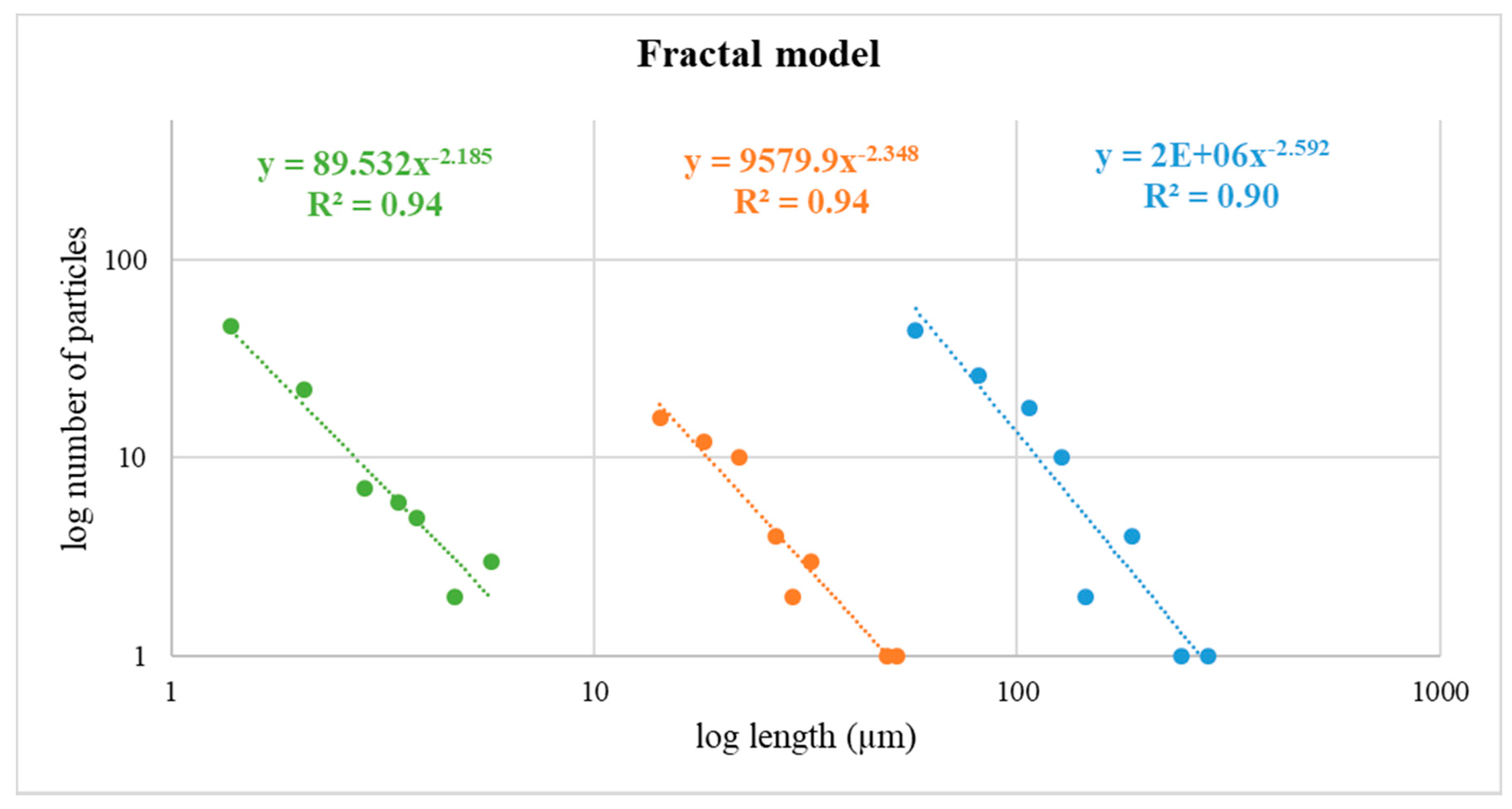
3.9. Fractal Model from FE-ESEM Data Obtained for the Powdered Starting Material
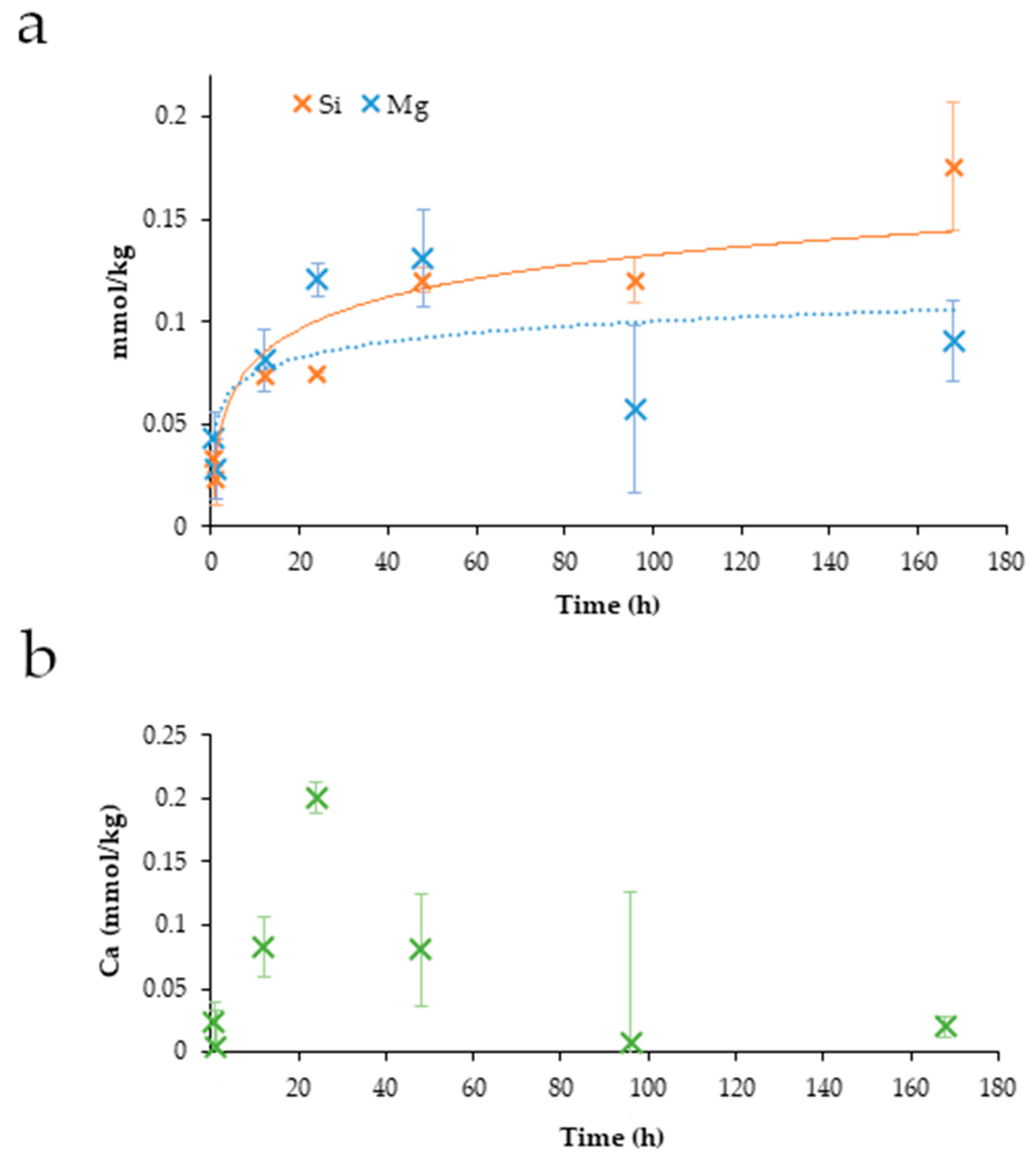
3.10. ICP-OES Data from Dissolution Experiment
3.11. Morphology as Observed by FE-ESEM for the Material After Dissolution Experiment
3.12. Dimensional Results Obtained by FE-ESEM for the Material after Dissolution Experiment
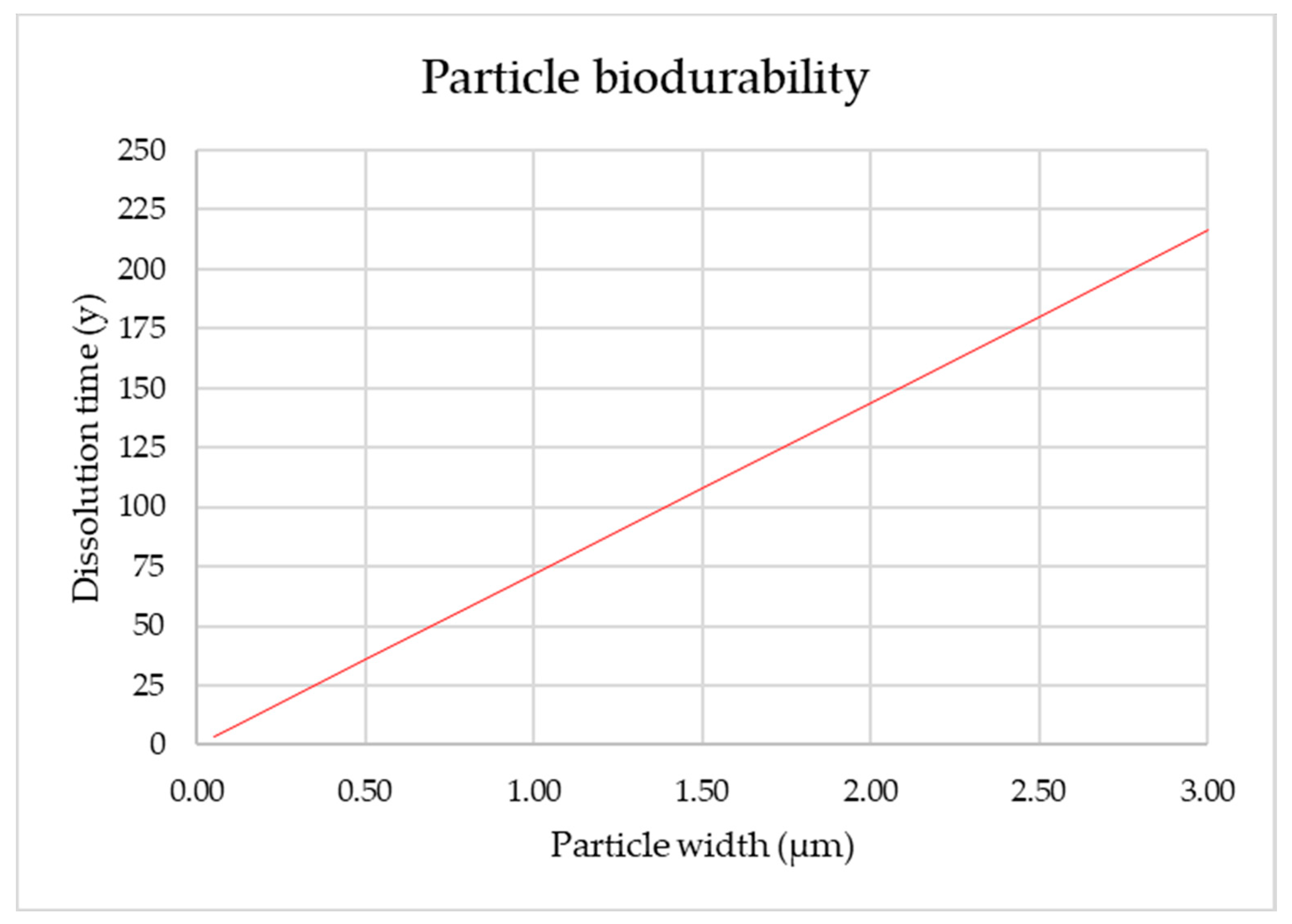
3.13. Estimation of Fiber Biodurability
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gamble, J.F.; Gibbs, G.W.; Nolan, R.P. An evaluation of the risk of lung cancer and mesothelioma from exposure to amphibole cleavage fragments. Regul. Toxicol. Pharmacol. 2008, 52, S154–S186. [Google Scholar] [CrossRef] [PubMed]
- Gualtieri, A.F. (Ed.) Introduction. In Mineral Fibres: Crystal Chemistry, Chemical-Physical Properties, Biological Interaction and Toxicity (EMU Notes in Mineralogy); European Mineralogical Union: London, UK, 2017; Volume 18, pp. 1–15. ISBN 9780903056656. [Google Scholar]
- Ferro, A.; Zebedeo, C.N.; Davis, C.; Ng, K.W.; Pfau, J.C. Amphibole, but not chrysotile, asbestos induces anti-nuclear autoantibodies and IL-17 in C57BL/6 mice. J. Immunotoxicol. 2014, 11, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Pfau, J.C.; Serve, K.M.; Noonan, C.W. Autoimmunity and asbestos exposure. Autoimmune Dis. 2014, 2014, 782045. [Google Scholar] [CrossRef] [PubMed]
- Meeker, G.P.; Bern, A.M.; Brownfield, I.K.; Lowers, H.A.; Sutley, S.J.; Hoefen, T.M.; Vance, J.S. The composition and morphology of amphiboles from the Rainy Creek complex, near Libby, Montana. Am. Mineral. 2003, 88, 1955–1969. [Google Scholar] [CrossRef]
- Boulanger, M.; Morlais, F.; Bouvier, V.; Galateau-Salle, F.; Guittet, L.; Maquignon, M.; Paris, C.; Raffaelli, C.; Launoy, G.; Clin, B. O32-1 Digestive cancers and occupational asbestos exposure: Significant associations in a French cohort of asbestos plant workers. Occup. Environ. Med. 2016, 73, A58. [Google Scholar] [CrossRef]
- Clin, B.; Thaon, I.; Boulanger, M.; Brochard, P.; Chamming’s, S.; Gislard, A.; Lacourt, A.; Luc, A.; Ogier, G.; Paris, C.; Pairon, J. Cancer of the esophagus and asbestos exposure. Am. J. Ind. Med. 2017, 60, 968–975. [Google Scholar] [CrossRef] [PubMed]
- Di Ciaula, A. Asbestos ingestion and gastrointestinal cancer: A possible underestimated hazard. Expert Rev. Gastroenterol. Hepatol. 2017, 11, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Paris, C.; Thaon, I.; Hérin, F.; Clin, B.; Lacourt, A.; Luc, A.; Coureau, G.; Brochard, P.; Chamming’s, S.; Gislard, A.; et al. Occupational asbestos exposure and incidence of colon and rectal cancers in French men: The asbestos-related diseases cohort (ARDCo-Nut). Environ. Health Perspect. 2017, 3, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Gunter, M.E.; Belluso, E.; Mottana, A. Amphiboles: Environmental and health concerns. Rev. Mineral. Geochem. 2007, 67, 453–516. [Google Scholar] [CrossRef]
- Martin, R.F. Amphiboles in the igneous environment. Rev. Mineral. Geochem. 2007, 67, 323–358. [Google Scholar] [CrossRef]
- Schumacher, J.C. Metamorphic amphiboles: Composition and coexistence. Rev. Mineral. Geochem. 2007, 67, 359–416. [Google Scholar] [CrossRef]
- Vignaroli, G.; Rossetti, F.; Belardi, G.; Billi, A. Linking rock fabric to fibrous mineralisation: A basic tool for the asbestos hazard. Nat. Hazard Earth Syst. 2011, 11, 1267–1280. [Google Scholar] [CrossRef]
- Vignaroli, G.; Ballirano, P.; Belardi, G.; Rossetti, F. Asbestos fibre identification vs. evaluation of asbestos hazard in ophiolitic rock mélanges; a case study from the Ligurian Alps (Italy). Environ. Earth Sci. 2014, 72, 3679–3698. [Google Scholar] [CrossRef]
- Trommsdorff, V.; Montrasio, A.; Hermann, J.; Müntener, O.; Spillman, P.; Gieré, R. The geological map of Valmalenco. Schweiz. Mineral. Petrogr. Mitt. 2005, 85, 1–13. [Google Scholar]
- Trommsdorff, V.; Evans, B.W. Antigorite-ophicarbonates: Contact metamorphism in Valmalenco, Italy. Contrib. Mineral. Petrol. 1977, 62, 301–312. [Google Scholar] [CrossRef]
- Hermann, J.; Müntener, O.; Trommsdorff, V.; Hansmann, W.; Piccardo, G.B. Fossil crust-to-mantle transition, Val Malenco (Italian Alps). J. Geophys. Res-Solid Earth 1997, 102, 20123–20132. [Google Scholar] [CrossRef]
- Bocchio, R.; Diella, V.; Adamo, I.; Marinoni, N. Mineralogical characterization of the gem-variety pink clinozoisite from Val Malenco, Central Alps, Italy. Rend. Fis. Acc. Lincei 2017, 28, 549–557. [Google Scholar] [CrossRef]
- Trommsdorff, V.; Evans, B.W. Titanian hydroxyl-clinohumite: Formation and breakdown in antigorite rocks (Malenco, Italy). Contrib. Mineral. Petrol. 1980, 72, 229–242. [Google Scholar] [CrossRef]
- Wojydr, M. Fityk: A general purpose peak fitting program. J. Appl. Crystallogr. 2010, 42, 1126–1128. [Google Scholar] [CrossRef]
- Marques, M.R.C.; Loebenberg, R.; Almukainzi, M. Simulated biological fluids with possible application in dissolution testing. Dissolut. Technol. 2011, 18, 15–28. [Google Scholar] [CrossRef]
- Pacella, A.; Fantauzzi, M.; Turci, F.; Cremisini, C.; Montereali, M.R.; Nardi, E.; Atzei, D.; Rossi, A.; Andreozzi, G.B. Dissolution reaction and surface iron speciation of UICC crocidolite in buffered solution at pH 7.4: A combined ICP-OES, XPS and TEM investigation. Geochim. Cosmochim. Acta 2014, 127, 221–232. [Google Scholar] [CrossRef]
- Della Ventura, G.; Vigliaturo, R.; Gieré, R.; Pollastri, S.; Gualtieri, A.F.; Iezzi, G. Infrared spectroscopy of the regulated asbestos amphiboles. Minerals 2018, 8, 413. [Google Scholar] [CrossRef]
- Oberti, R.; Cannillo, E.; Toscani, G. How to name amphiboles after the IMA 2012 report: Rules of thumb and a new PC program for monoclinic amphiboles. Periodico Mineral. 2012, 81, 257–267. [Google Scholar] [CrossRef]
- Locock, A.J. An Excel spreadsheet to classify chemical analyses of amphiboles following the IMA 2012 recommendations. Comput. Geosci. 2014, 62, 1–11. [Google Scholar] [CrossRef]
- Hawthorne, F.C.; Oberti, R.; Harlow, G.E.; Maresch, W.V.; Martin, R.F.; Schumacher, J.C.; Welch, M. IMA Report: Nomenclature of the amphibole supergroup. Am. Mineral. 2012, 9, 2031–2048. [Google Scholar] [CrossRef]
- Leake, B.E.; Woolley, A.R.; Arps, C.E.; Birch, W.D.; Gilbert, M.C.; Grice, J.D.; Hawthorne, F.C.; Kisch, H.J.; Krivovichev, V.G. Nomenclature of amphiboles: Report of the subcommittee on amphiboles of the International Mineralogical Association commission on new minerals and mineral names. Mineral. Mag. 1997, 61, 295–321. [Google Scholar] [CrossRef]
- Leake, B.E.; Woolley, A.R.; Birch, W.D.; Burke, E.A.; Ferraris, G.; Grice, J.D.; Hawthorne, F.C.; Kisch, H.J.; Krivovichev, V.G.; Schumacher, J.C.; et al. Nomenclature of amphiboles: Additions and revisions to the International Mineralogical Associations amphibole nomenclature. Mineral. Mag. 2004, 68, 209–215. [Google Scholar] [CrossRef]
- Lucci, F.; Della Ventura, G.; Conte, A.; Nazzari, M.; Scarlato, P. Naturally Occurring Asbestos (NOA) in Granitoid Rocks, A Case Study from Sardinia (Italy). Minerals 2018, 8, 442. [Google Scholar] [CrossRef]
- Rinaudo, C.; Belluso, E.; Gastaldi, D. Assessment of the use of Raman spectroscopy for the determination of amphibole asbestos. Mineral. Mag. 2004, 68, 455–465. [Google Scholar] [CrossRef]
- Apopei, A.I.; Buzgar, N.; Buzatu, A. Raman and infrared spectroscopy of kaersutite and certain common amphiboles. Analele Stiintifice ale Universitatii “Al. I. Cuza” din Iasi. Seria Geologie 2011, 57, 35–58. [Google Scholar]
- Della Ventura, G. The analysis of asbestos minerals using vibrational spectroscopies (FTIR, Raman): Crystal Chemistry, identification and environmental applications. In Mineral Fibres: Crystal Chemistry, Chemical-Physical Properties, Biological Interaction and Toxicity (EMU Notes in Mineralogy); Gualtieri, A.F., Ed.; European Mineralogical Union: London, UK, 2017; Volume 18, pp. 135–169. ISBN 9780903056656. [Google Scholar]
- Ishida, K.; Jenkins, D.M.; Hawthorne, F.C. Mid-IR bands of synthetic calcic amphiboles of tremolite-pargasite series and of natural calcic amphiboles. Am. Mineral. 2008, 93, 1112–1118. [Google Scholar] [CrossRef]
- Kouropis-Agalou, K.; Liscio, A.; Treossi, E.; Ortolani, L.; Pugno, N.M.; Palermo, V. Fragmentation and exfoliation of 2-dimensional materials: A statistical approach. Nanoscale 2014, 6, 5926–5933. [Google Scholar] [CrossRef] [PubMed]
- Gonda, I.; Abd El Khalik, A.F. On the calculation of aerodynamic diameters of fibers. Aerosol Sci. Technol. 1985, 4, 233–238. [Google Scholar] [CrossRef]
- Veblen, D.R.; Wylie, A.G. Mineralogy of amphiboles and 1:1 layer silicates. In Reviews in Mineralogy and Geochemistry; Guthrie, G.D., Jr., Mossman, B.T., Eds.; Mineralogical Society of America: Chantilly, VA, USA, 1993; Volume 28, pp. 61–137. ISSN 0275-0279. [Google Scholar]
- National Institute for Occupational Safety and Health (NIOSH). Asbestos Fibers and Other Elongated Mineral Particles: State of the Science and Roadmap for Research; DHHS Publication No.2011-159; 2011. Available online: https://www.cdc.gov/niosh/docs/2011-159/pdfs/2011-159.pdf (accessed on 28 November 2018).
- Wylie, A.G. Modeling asbestos population: A fractal approach. Canad. Mineral. 1993, 30, 437–446. [Google Scholar]
- Rozalen, M.; Ramos, M.E.; Gervilla, F.; Kerestedjian, T.; Fiore, S.; Huertas, F.J. Dissolution study of tremolite and anthophyllite: pH effect on the reaction kinetics. Appl. Geochem. 2014, 49, 46–56. [Google Scholar] [CrossRef]
- Bernstein, D.M.; Castranova, V.; Donaldson, K.; Fubini, B.; Hadley, J.; Hesterberg, T.; Kaneg, A.; Laih, D.; McConnell, E.E.; Muhle, H.; et al. Testing of fibrous particles: Short-term assays and strategies. Inhal. Toxicol. 2005, 17, 497–537. [Google Scholar] [CrossRef] [PubMed]
- Hume, L.A.; Rimstidt, J.D. The biodurability of chrysotile asbestos. Am. Mineral. 1992, 77, 1125–1128. [Google Scholar]
- Rozalen, M.; Ramos, M.E.; Huertas, F.J.; Fiore, S.; Gervilla, F. Dissolution kinetics and biodurability of tremolite particles in mimicked lung fluids: Effect of citrate and oxalate. J. Asian Earth Sci. 2013, 77, 318–326. [Google Scholar] [CrossRef]
- Critelli, T.; Marini, L.; Schott, J.; Mavromatis, V.; Apollaro, C.; Rinder, T.; De Rosa, R.; Oelkers, E.H. Dissolution rates of actinolite and chlorite from a whole-rock experimental study of metabasalt dissolution from 2 ≤ pH ≤ 12 at 25 °C. Chem. Geol. 2014, 390, 100–108. [Google Scholar] [CrossRef]
- Pollastri, S.; Gualtieri, A.F.; Lasinantti Gualtieri, M.; Hanuskova, M.; Cavallo, A.; Gaudino, G. The zeta potential of mineral fibres. J. Hazard. Mater. 2014, 276, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Pacella, A.; Fantauzzi, M.; Turci, F.; Cremisini, C.; Montereali, M.R.; Nardi, E.; Atzei, D.; Rossi, A.; Andreozzi, G.B. Surface alteration mechanism and topochemistry of iron in tremolite asbestos: A step toward understanding the potential hazard of amphibole asbestos. Chem. Geol. 2015, 405, 28–38. [Google Scholar] [CrossRef]




| Oxides | Average (n = 12 with σn−1 in Parentheses) | Range |
|---|---|---|
| SiO2 (wt. %) | 56.68 (0.67) | 55.65–57.72 |
| TiO2 | <0.01 | - |
| Al2O3 | 0.46 (0.07) | 0.29–0.54 |
| FeOt | 5.37 (0.75) | 4.44–6.59 |
| MnO | 0.19 (0.04) | 0.13–0.26 |
| MgO | 21.16 (0.35) | 20.58–21.62 |
| CaO | 13.12 (0.29) | 12.58–13.74 |
| Na2O | 0.25 (0.08) | 0.03–0.36 |
| K2O | 0.03 (0.02) | 0.00–0.06 |
| F | 0.06 (0.01) | 0.00–0.22 |
| Cl | <0.01 | - |
| Total | 97.32 | 96.47–98.16 |
| Normalization Scheme: 13-CNK, all ferrous iron | ||
| Si, apfu | 7.92 | 7.85–7.96 |
| Al | 0.07 | 0.04–0.09 |
| ΣT | 7.99 | 7.94–8.00 |
| Al | 0.01 | 0.00–0.04 |
| Mn2+ | 0.01 | 0.00–0.02 |
| Fe2+ | 0.58 | 0.49–0.69 |
| Mg | 4.41 | 4.31–4.51 |
| ΣC | 5.00 | 5.00 |
| Mn2+ | 0.02 | 0.00–0.03 |
| Fe2+ | 0.05 | 0.00–0.12 |
| Ca | 1.96 | 1.89–1.99 |
| ΣB | 2.03 | 2.00–2.13 |
| Na | 0.07 | 0.01–0.09 |
| K | 0.01 | 0.00–0.01 |
| ΣA | 0.08 | 0.01–0.10 |
| OH | 1.98 | 1.90–2.00 |
| F | 0.02 | 0.00–0.10 |
| ΣW | 2.00 | 2.00 |
| Mg/(Mg+Fe2+) | 0.88 | 0.86–0.90 |
| Laser Diffraction Dimensional Distribution | Geometric | Logarithmic |
| µm | ɸ | |
| Mode 1: | 127.79 | 2.97 |
| Mode 2: | 73.02 | 3.78 |
| Mode 3: | - | - |
| D10: | 4.53 | 2.57 |
| Median or D50: | 49.78 | 4.33 |
| D90: | 168.75 | 7.79 |
| (D90/D10): | 37.25 | 3.03 |
| (D90-D10): | 164.22 | 5.22 |
| (D75/D25): | 6.45 | 1.83 |
| (D75-D25): | 90.44 | 2.69 |
| Folk & Ward Method | ||
| Geometric | Logarithmic | |
| µm | ɸ | |
| Mean | 39.21 | 4.67 |
| Sorting (σ) | 4.13 | 2.05 |
| Skewness (Sk) | −0.34 | 0.34 |
| Kurtosis (K) | 1.05 | 1.05 |
| Magnification 102× (n = 150) | ||||||||
| ESEM Dimensional Study | L | w | L/w | Areal | Acalc | Areal/Acalc | s (Areal) | Dae |
| Mean | 75.03 | 26.00 | 4.53 | 2141.08 | 2396.60 | 0.88 | 38.69 | 54.68 |
| Median | 61.56 | 17.75 | 3.18 | 1061.42 | 1181.89 | 0.91 | 32.58 | 44.09 |
| Mode | #N/D | 43.50 | #N/D | #N/D | #N/D | #N/D | #N/D | #N/D |
| Max | 336.27 | 162.91 | 33.37 | 31031.81 | 31038.24 | 1.00 | 176.16 | 256.19 |
| Min | 16.00 | 1.18 | 0.73 | 21.19 | 25.48 | 0.58 | 4.60 | 3.93 |
| Std. Dev. | 47.60 | 24.98 | 4.26 | 3660.95 | 3976.48 | 0.10 | 25.47 | 39.57 |
| Std. Error | 3.89 | 2.04 | 0.35 | 298.92 | 324.68 | 0.01 | 2.08 | 3.23 |
| Magnification 103× (n = 150) | ||||||||
| L | w | L/w | Areal | Acalc | Areal/Acalc | s (Areal) | Dae | |
| Mean | 11.80 | 3.92 | 5.24 | 57.15 | 66.36 | 0.86 | 5.86 | 8.34 |
| Median | 8.52 | 2.80 | 3.47 | 18.50 | 22.35 | 0.90 | 4.30 | 6.13 |
| Mode | 8.18 | 0.75 | #N/D | #N/D | #N/D | #N/D | #N/D | #N/D |
| Max | 54.63 | 23.67 | 33.75 | 1046.30 | 1082.56 | 1.00 | 32.35 | 46.28 |
| Min | 2.00 | 0.20 | 0.33 | 0.58 | 0.63 | 0.11 | 0.76 | 0.71 |
| Std. Dev. | 9.13 | 3.99 | 5.43 | 114.36 | 125.81 | 0.14 | 4.79 | 7.21 |
| Std. Error | 0.75 | 0.33 | 0.44 | 9.34 | 10.27 | 0.01 | 0.39 | 0.59 |
| Magnification 104× (n = 150) | ||||||||
| L | w | L/w | Areal | Acalc | Areal/Acalc | s (Areal) | Dae | |
| Mean | 1.87 | 0.59 | 4.03 | 1.07 | 1.40 | 0.87 | 0.87 | 1.26 |
| Median | 1.29 | 0.47 | 2.52 | 0.53 | 0.60 | 0.89 | 0.72 | 1.03 |
| Mode | 0.92 | 0.41 | #N/D | 0.20 | #N/D | #N/D | 0.45 | #N/D |
| Max | 41.56 | 2.35 | 59.73 | 10.38 | 28.92 | 1.00 | 3.22 | 5.18 |
| Min | 0.15 | 0.05 | 0.85 | 0.01 | 0.01 | 0.09 | 0.11 | 0.17 |
| Std. Dev. | 3.44 | 0.44 | 5.50 | 1.62 | 2.93 | 0.12 | 0.56 | 0.89 |
| Std. Error | 0.28 | 0.04 | 0.45 | 0.13 | 0.24 | 0.01 | 0.05 | 0.07 |
| Magnification | Acicular % | Bladed % | Columnar % | Equant % | Fiber % | Massive % | Platy % | Prismatic % |
|---|---|---|---|---|---|---|---|---|
| 102× | 7.33 | 16.00 | 12.67 | 10.67 | 21.33 | 6.67 | 4.00 | 21.33 |
| 103× | 14.00 | 20.67 | 6.67 | 10.00 | 15.33 | 6.00 | 12.00 | 15.33 |
| 104× | 11.33 | 29.33 | 13.33 | 9.33 | 6.67 | 9.33 | 4.00 | 16.67 |
| Atomic % Ratios | Powdered Starting Material (Bulk EPMA) | Powdered Starting Material (Bulk EDXS) | Powdered Sample after 168-h Exposure (Bulk EDXS) | Gamble’s Solution after 168-h Experiment (ICP-OES) |
|---|---|---|---|---|
| 0.56 (0.01) | 0.63 (0.1) | 0.64 (0.1) | 0.51 (0.04) | |
| 0.25 (0.01) | 0.29 (0.07) | 0.41 (0.2) | 0.11 (0.03) | |
| 0.08 (0.01) | 0.12 (0.09) | 0.22 (0.2) | 0.030 (0.01) |
| Magnification 102× (n = 150) | ||||||||
| ESEM Dimensional Study | L | w | L/w | Areal | Acalc | Areal/Acalc | s (Areal) | Dae |
| Mean | 115.18 | 31.09 | 5.34 | 3652.20 | 4201.40 | 0.85 | 52.48 | 72.56 |
| Median | 107.94 | 28.31 | 4.02 | 2762.68 | 3081.09 | 0.90 | 52.56 | 68.32 |
| Mode | #N/D | 6.25 | #N/D | 93.99 | #N/D | #N/D | 9.69 | #N/D |
| Max | 462.15 | 94.95 | 40.56 | 22437.60 | 30946.22 | 1.00 | 149.79 | 198.56 |
| Min | 13.52 | 1.40 | 1.06 | 15.63 | 19.87 | 0.12 | 3.95 | 4.28 |
| Std. Dev. | 71.68 | 21.77 | 5.15 | 3776.07 | 4436.00 | 0.16 | 30.07 | 43.61 |
| Std. Error | 5.85 | 1.78 | 0.42 | 308.31 | 362.20 | 0.01 | 2.46 | 3.56 |
| Magnification 103× (n = 150) | ||||||||
| L | w | L/w | Areal | Acalc | Areal/Acalc | s (Areal) | Dae | |
| Mean | 17.70 | 6.12 | 3.72 | 144.19 | 171.05 | 0.85 | 8.95 | 13.22 |
| Median | 10.66 | 4.37 | 2.63 | 33.27 | 41.13 | 0.88 | 5.77 | 8.90 |
| Mode | 4.44 | 1.90 | #N/D | 71.60 | #N/D | #N/D | 8.46 | #N/D |
| Max | 154.49 | 41.94 | 34.43 | 2188.56 | 2369.54 | 1.00 | 46.78 | 69.59 |
| Min | 1.57 | 0.19 | 1.04 | 0.58 | 0.60 | 0.08 | 0.76 | 0.62 |
| Std. Dev. | 19.39 | 6.11 | 3.76 | 298.25 | 343.70 | 0.13 | 8.03 | 11.98 |
| Std. Error | 1.58 | 0.50 | 0.31 | 24.35 | 28.06 | 0.01 | 0.66 | 0.98 |
| Magnification 104× (n = 150) | ||||||||
| L | w | L/w | Areal | Acalc | Areal/Acalc | s (Areal) | Dae | |
| Mean | 3.64 | 1.38 | 3.12 | 6.19 | 7.66 | 0.83 | 1.95 | 3.00 |
| Median | 2.73 | 1.15 | 2.27 | 2.71 | 3.17 | 0.86 | 1.65 | 2.45 |
| Mode | 2.30 | 0.15 | #N/D | #N/D | #N/D | #N/D | #N/D | #N/D |
| Max | 19.99 | 6.85 | 15.25 | 74.16 | 102.53 | 1.00 | 8.61 | 14.88 |
| Min | 0.14 | 0.02 | 1.11 | 0.00 | 0.01 | 0.11 | 0.03 | 0.06 |
| Std. Dev. | 3.34 | 1.14 | 2.39 | 10.55 | 13.38 | 0.15 | 1.55 | 2.41 |
| Std. Error | 0.27 | 0.09 | 0.20 | 0.86 | 1.09 | 0.01 | 0.13 | 0.20 |
| Magnification | Acicular % | Bladed % | Columnar % | Equant % | Fiber % | Massive % | Platy % | Prismatic % |
|---|---|---|---|---|---|---|---|---|
| 102× | 10.67 | 19.33 | 13.33 | 9.33 | 22.67 | 8.67 | 0.67 | 15.33 |
| 103× | 10.67 | 24.67 | 10.67 | 12.00 | 14.67 | 8.00 | 4.00 | 15.33 |
| 104× | 10.67 | 26.00 | 19.33 | 11.33 | 6.00 | 10.67 | 0.67 | 15.33 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vigliaturo, R.; Della Ventura, G.; Choi, J.K.; Marengo, A.; Lucci, F.; O'Shea, M.J.; Pérez-Rodríguez, I.; Gieré, R. Mineralogical Characterization and Dissolution Experiments in Gamble’s Solution of Tremolitic Amphibole from Passo di Caldenno (Sondrio, Italy). Minerals 2018, 8, 557. https://doi.org/10.3390/min8120557
Vigliaturo R, Della Ventura G, Choi JK, Marengo A, Lucci F, O'Shea MJ, Pérez-Rodríguez I, Gieré R. Mineralogical Characterization and Dissolution Experiments in Gamble’s Solution of Tremolitic Amphibole from Passo di Caldenno (Sondrio, Italy). Minerals. 2018; 8(12):557. https://doi.org/10.3390/min8120557
Chicago/Turabian StyleVigliaturo, Ruggero, Giancarlo Della Ventura, Jessica K. Choi, Alessandra Marengo, Federico Lucci, Michael J. O'Shea, Ileana Pérez-Rodríguez, and Reto Gieré. 2018. "Mineralogical Characterization and Dissolution Experiments in Gamble’s Solution of Tremolitic Amphibole from Passo di Caldenno (Sondrio, Italy)" Minerals 8, no. 12: 557. https://doi.org/10.3390/min8120557
APA StyleVigliaturo, R., Della Ventura, G., Choi, J. K., Marengo, A., Lucci, F., O'Shea, M. J., Pérez-Rodríguez, I., & Gieré, R. (2018). Mineralogical Characterization and Dissolution Experiments in Gamble’s Solution of Tremolitic Amphibole from Passo di Caldenno (Sondrio, Italy). Minerals, 8(12), 557. https://doi.org/10.3390/min8120557






