The Fine Characterization and Potential Photocatalytic Effect of Semiconducting Metal Minerals in Danxia Landforms
Abstract
1. Introduction
2. Materials and Methods
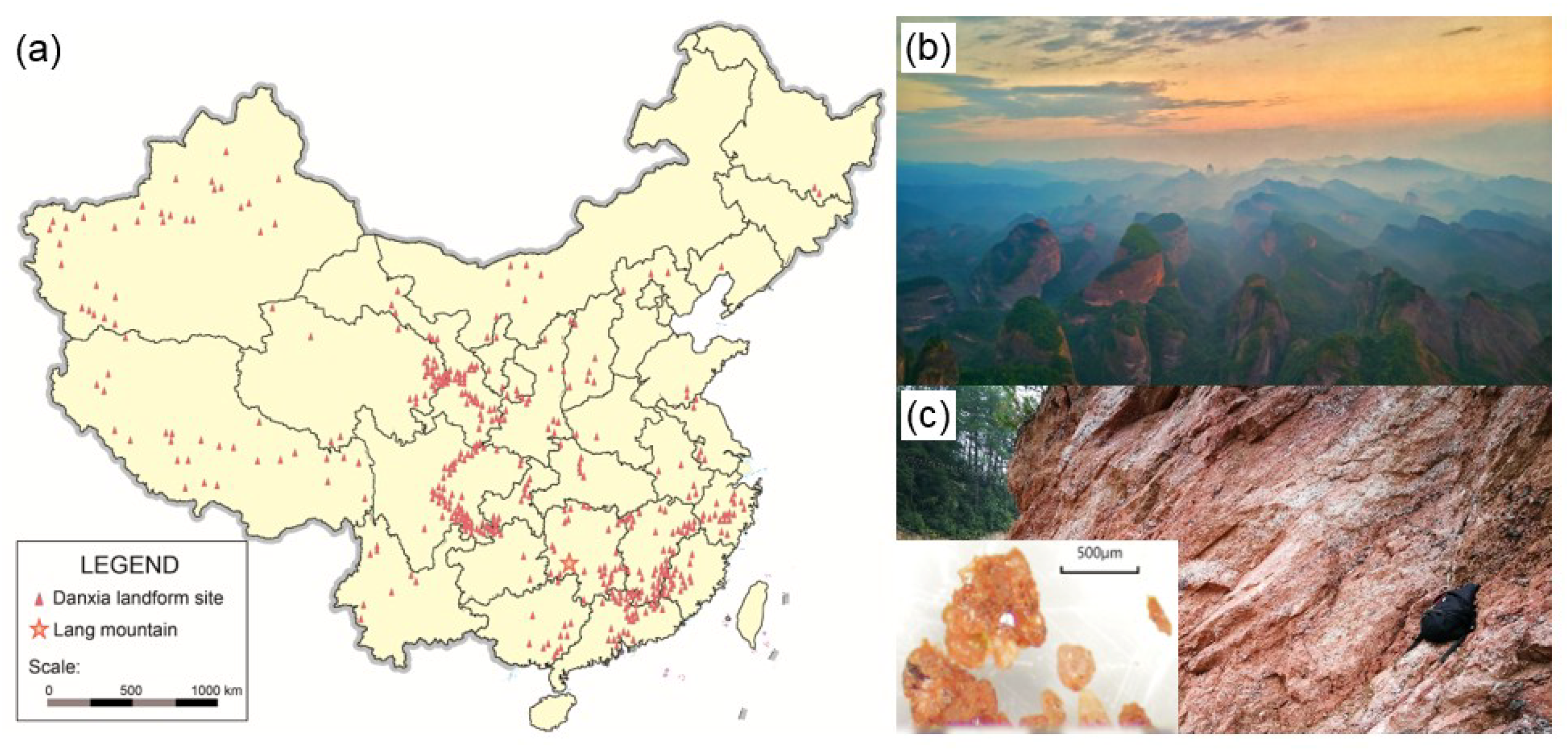
2.1. Sample Preparation
2.2. Characteristic Methods
2.3. Photoelectrochemical and Photocatalytic Experiments
3. Results and Discussion
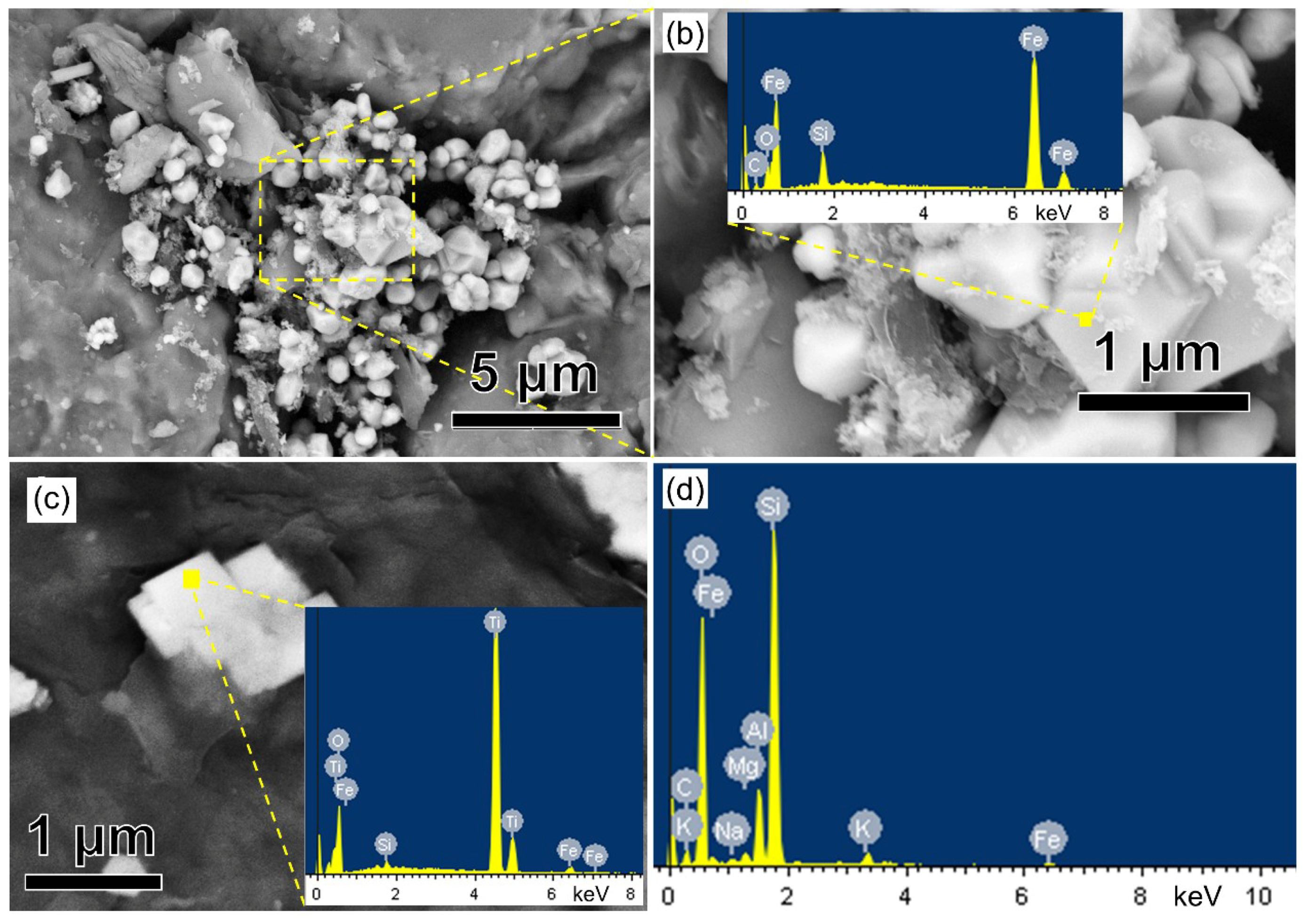
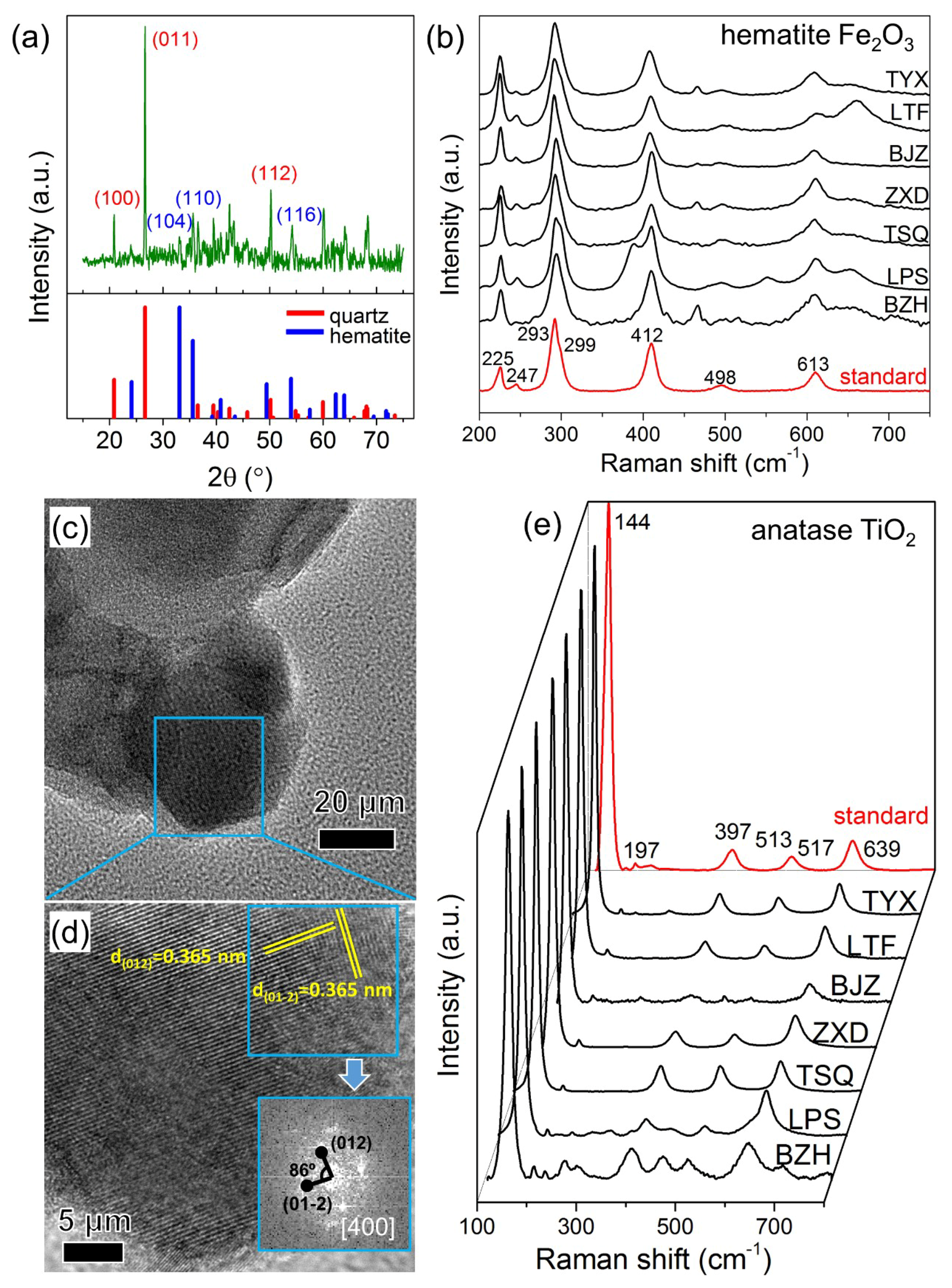
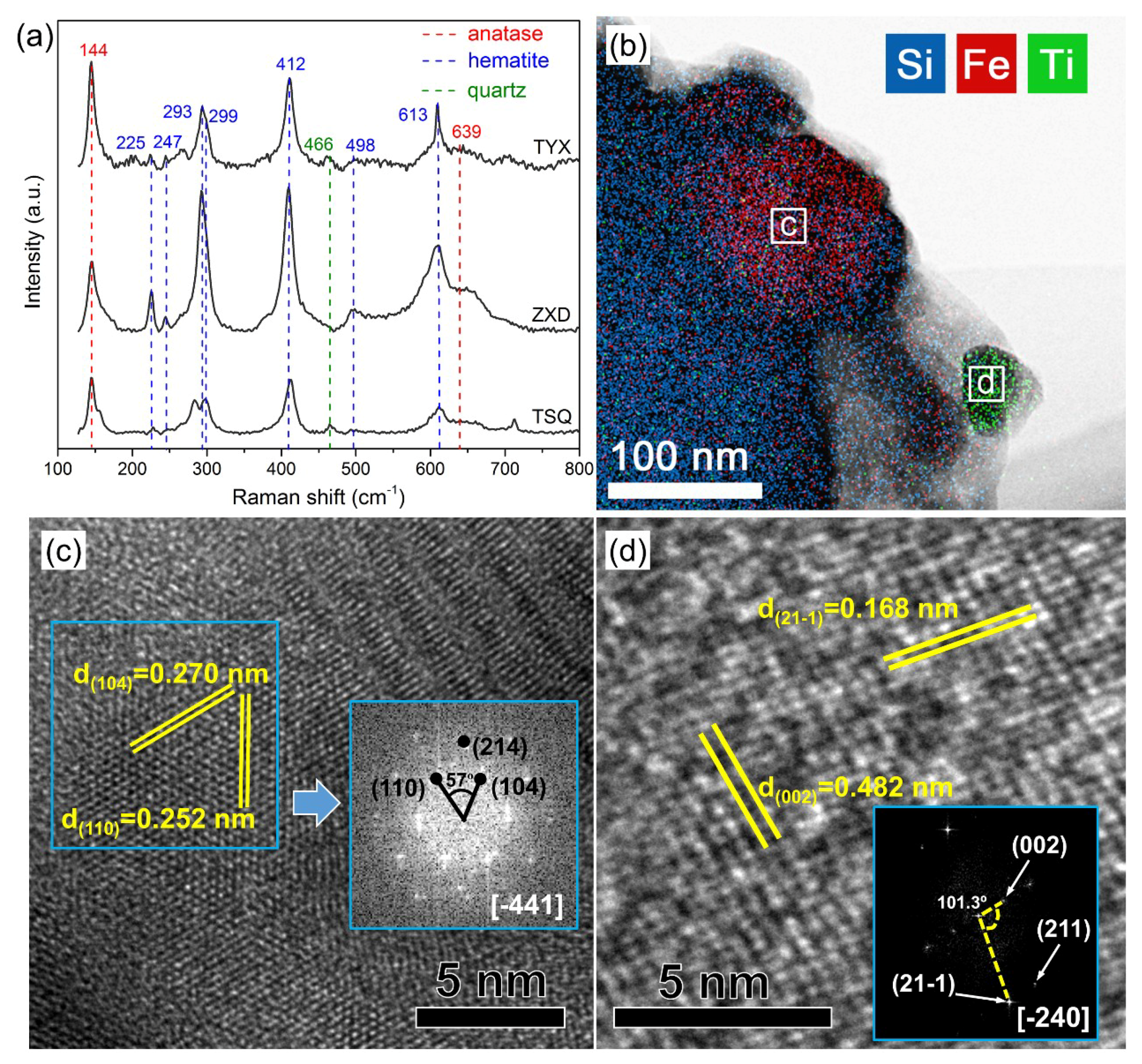
3.1. The Mineralogical Identification of Fe Oxides and Ti Oxides
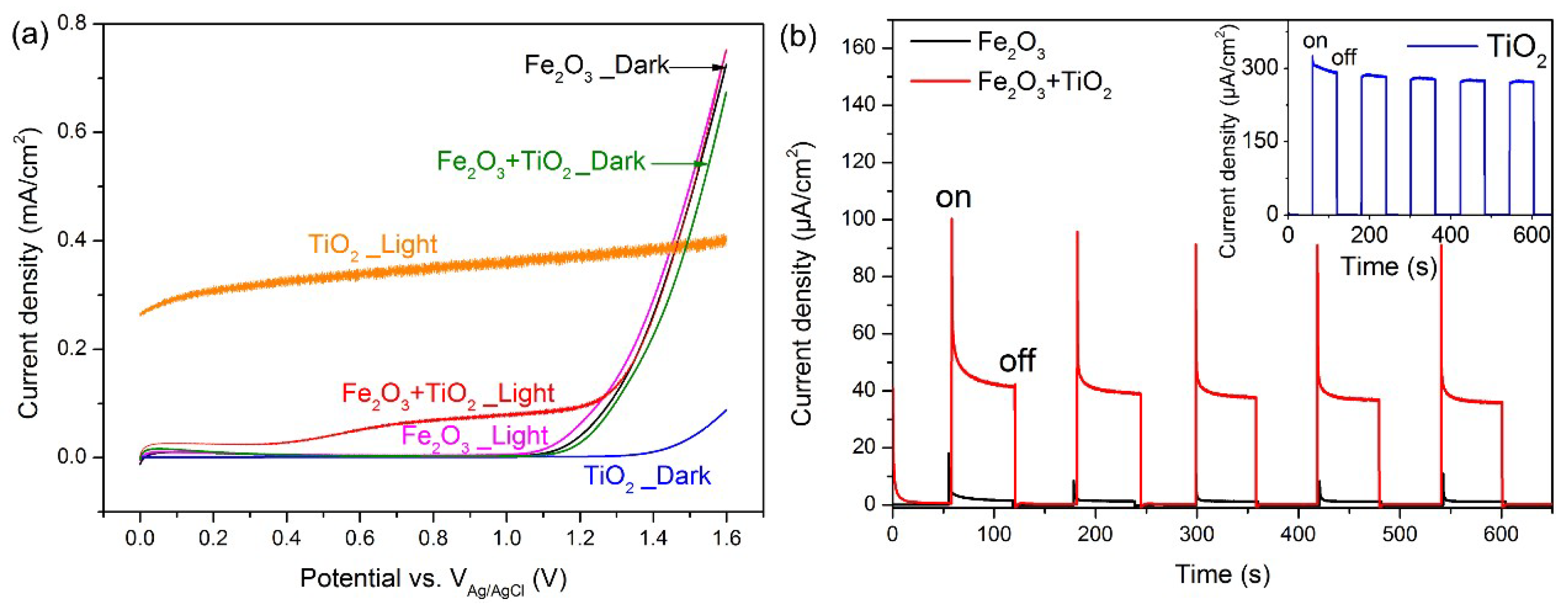
3.2. Simulated Photoelectrochemical Experiments of Fe Oxides and Ti Oxides
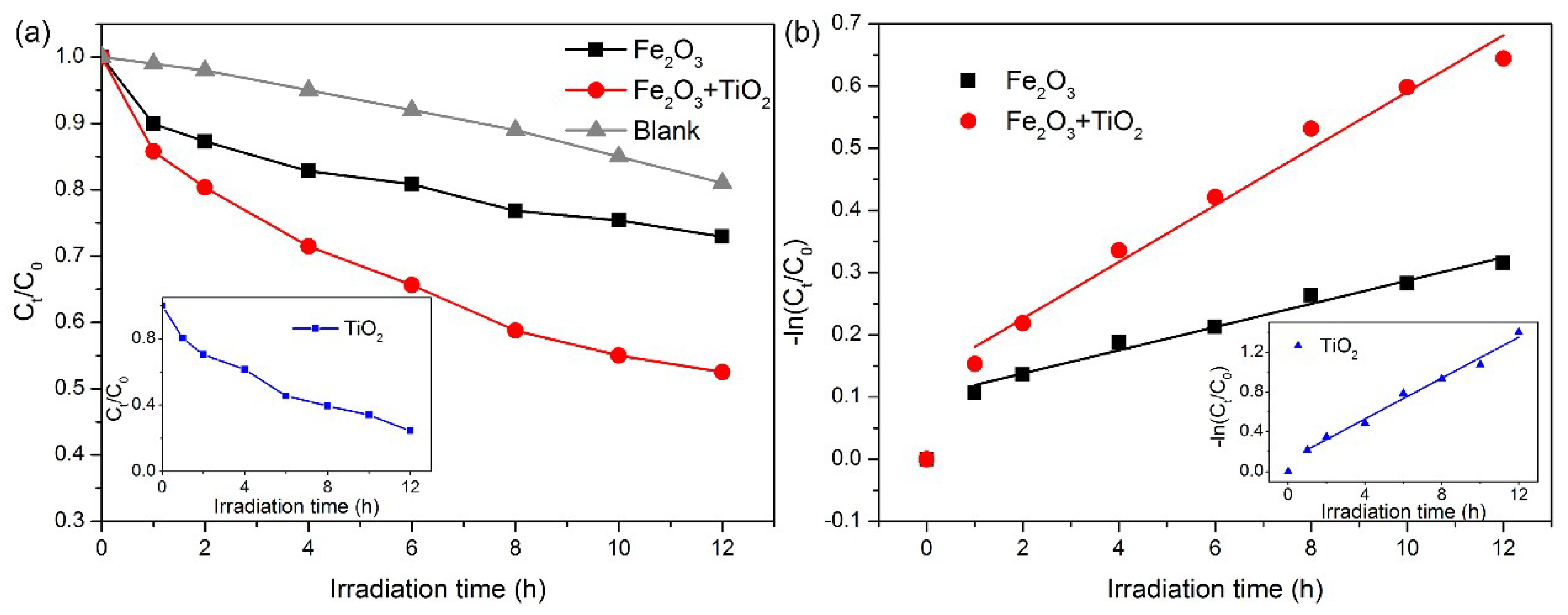
3.3. The Potential Environmental Effect of Fe Oxide and Ti Oxide Minerals on the Danxia Landform
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Peng, H. Danxia geomorphology of China: A review. Chin. Sci. Bull. 2001, 46, 38–44. [Google Scholar] [CrossRef]
- Hou, Y.; Peng, S.; Li, F.; Lin, Z.; Chen, B.; Peng, H. Characteristics and scientific values of ecological succession in Danxia landform of China. Acta Ecol. Sin. 2008, 28, 3384–3389. [Google Scholar]
- Zhang, W.; Hayakawa, Y.S.; Oguchi, T. DEM and GIS based morphometric and topographic-profile analyses of Danxia landforms. Geomorphometry Organ. 2001, 121–124. [Google Scholar]
- Peng, H.; Ren, F.; Pan, Z. A review of Danxia landforms in China. Zeitschrift für Geomorphologie. Suppl. Issues 2015, 59, 19–33. [Google Scholar]
- United Nations Educational, Scientific and Cultural Organization. Available online: http://whc.unesco.org/en/list/1335 (accessed on 29 November 2018).
- Peng, S.; Li, F.; Zhou, T.; Chen, L.; Chen, B.; Hua, P. Special ecological effects of ravines in danxia landform, china. Acta Ecol. Sin. 2008, 28, 2947–2953. [Google Scholar]
- Wu, J.; Peng, S.; Lin, Z.; Chen, L.; Chen, B.; Peng, H.; Hou, R. Ecological effect on the hilltop of danxia landform. Acta Ecol. Sin. 2008, 28, 3390–3400. [Google Scholar]
- Opdyke, N.D. The paleomagnetism of the Permian red beds of southwest Tanganyika. J. Geophys. Res. 1964, 69, 2477–2487. [Google Scholar] [CrossRef]
- Eriksson, P.G.; Cheney, E.S. Evidence for the transition to an oxygen-rich atmosphere during the evolution of red beds in the Lower Proterozoic sequences of southern Africa. Precambrian Res. 1992, 54, 257–269. [Google Scholar] [CrossRef]
- Hu, X.; Jansa, L.; Sarti, M. Mid-Cretaceous oceanic red beds in the Umbria–Marche Basin, central Italy: Constraints on paleoceanography and paleoclimate. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2006, 233, 163–186. [Google Scholar] [CrossRef]
- Turner, P.; Archer, R. The role of biotite in the diagenesis of red beds from the Devonian of northern Scotland. Sediment. Geol. 1977, 19, 241–251. [Google Scholar] [CrossRef]
- Cai, Y.; Hu, X.; Li, X.; Pan, Y. Origin of the red colour in a red limestone from the Vispi Quarry section (central Italy): A high-resolution transmission electron microscopy analysis. Cretac. Res. 2012, 38, 97–102. [Google Scholar] [CrossRef]
- Rose, A.W.; Bianchi-Mosquera, G.C. Adsorption of Cu, Pb, Zn, Co, Ni, and Ag on goethite and hematite; a control on metal mobilization from red beds into stratiform copper deposits. Econ. Geol. 1993, 88, 1226–1236. [Google Scholar] [CrossRef]
- Berner, R.A. Goethite stability and the origin of red beds. Geochim. Cosmochim. Acta 1969, 33, 267–273. [Google Scholar] [CrossRef]
- Kiipli, E.; Kallaste, T.; Kiipli, T. Hematite and goethite in Telychian marine red beds of the East Baltic. GFF 2000, 122, 281–286. [Google Scholar] [CrossRef]
- Schwertmann, U.; Taylor, R.M. Iron oxides. In Minerals in Soil Environments, 2nd ed.; Soil Science Society of America: Madison, WI, USA, 1989; pp. 380–395. [Google Scholar]
- Steila, D.; Pond, T.E. The Geography of Soils: Formation, Distribution, and Management, 2nd ed.; Rowman & Littlefield Publishers: Washington, DC, USA, 1989; pp. 13–47. [Google Scholar]
- Schoonen, M.A.; Xu, Y.; Strongin, D.R. An introduction to geocatalysis. J. Geochem. Explor. 1998, 62, 201–215. [Google Scholar] [CrossRef]
- Lu, A.; Wang, X.; Li, Y.; Ding, H.; Wang, C.; Zeng, C.; Hao, R.; Yang, X. Mineral photoelectrons and their implications for the origin and early evolution of life on Earth. Sci. China Earth Sci. 2014, 57, 897–902. [Google Scholar] [CrossRef]
- Xu, Y.; Schoonen, M.A. The absolute energy positions of conduction and valence bands of selected semiconducting minerals. Am. Mineral. 2000, 85, 543–556. [Google Scholar] [CrossRef]
- Chen, X.; Shen, S.; Guo, L.; Mao, S.S. Semiconductor-based photocatalytic hydrogen generation. Chem. Rev. 2010, 110, 6503–6570. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Lu, A. Mineral–microbe interactions and implications for remediation. Elements 2012, 8, 95–100. [Google Scholar] [CrossRef]
- Lu, A.; Li, Y.; Jin, S. Interactions between semiconducting minerals and bacteria under light. Elements 2012, 8, 125–130. [Google Scholar] [CrossRef]
- Bersani, D.; Lottici, P.P.; Montenero, A. Micro-Raman investigation of iron oxide films and powders produced by sol–gel syntheses. J. Raman Spectrosc. 1999, 30, 355–360. [Google Scholar] [CrossRef]
- Ohsaka, T.; Izumi, F.; Fujiki, Y. Raman spectrum of anatase, TiO2. J. Raman Spectrosc. 1978, 7, 321–324. [Google Scholar] [CrossRef]
- Tauc, J.; Grigorovici, R.; Vancu, A. Optical properties and electronic structure of amorphous germanium. Phys. Status Solidi B 1966, 15, 627–637. [Google Scholar] [CrossRef]
- Chen, X.; Li, C.; Grätzel, M.; Kostecki, R.; Mao, S.S. Nanomaterials for renewable energy production and storage. Chem. Soc. Rev. 2012, 41, 7909–7937. [Google Scholar] [CrossRef] [PubMed]
- Schoonen, M.; Smirnov, A.; Cohn, C. A perspective on the role of minerals in prebiotic synthesis. AMBIO J. Hum. Environ. 2004, 33, 539–551. [Google Scholar] [CrossRef]
- Lu, A.; Li, Y.; Jin, S.; Wang, X.; Wu, X.L.; Zeng, C.; Li, Y.; Ding, H.; Hao, R.; Lv, M.; et al. Growth of non-phototrophic microorganisms using solar energy through mineral photocatalysis. Nat. Commun. 2012, 3, 768. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lu, A.; Jin, S.; Wang, C. Photo-reductive decolorization of an azo dye by natural sphalerite: Case study of a new type of visible light-sensitized photocatalyst. J. Hazard. Mater. 2009, 170, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lu, A.; Li, Y.; Zhang, L.; Yip, H.Y.; Zhao, H.; An, T.; Wong, P.K. Naturally occurring sphalerite as a novel cost-effective photocatalyst for bacterial disinfection under visible light. Environ. Sci. Technol. 2011, 45, 5689–5695. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z. Fragile ecological zones and land desertification in China. J. Desert Res. 1991, 11, 11–22. (In Chinese) [Google Scholar]
- Peng, H.; Yan, L.; Chen, Z.; Simonson, S.; Luo, G. A preliminary study of desertification in red beds in the humid region of southern china. Acta Geogr. Sin. 2015, 70, 1699–1707. (In Chinese) [Google Scholar]
- Zent, A.P.; Ichimura, A.S.; Quinn, R.C.; Harding, H.K. The formation and stability of the superoxide radical (O2−) on rock-forming minerals: Band gaps, hydroxylation state, and implications for Mars oxidant chemistry. J. Geophys. Res. Planets 2008, 113. [Google Scholar] [CrossRef]
- Georgiou, C.D.; Sun, H.J.; McKay, C.P.; Grintzalis, K.; Papapostolou, I.; Zisimopoulos, D.; Panagiotidis, K.; Zhang, G.; Koutsopoulou, E.; Christidis, G.E.; et al. Evidence for photochemical production of reactive oxygen species in desert soils. Nat. Commun. 2015, 6, 7100. [Google Scholar] [CrossRef] [PubMed]
- Ren, G.; Ding, H.; Li, Y.; Lu, A. Natural Hematite as a Low-Cost and Earth-Abundant Cathode Material for Performance Improvement of Microbial Fuel Cells. Catalysts 2016, 6, 157. [Google Scholar] [CrossRef]




© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiao, Y.; Li, Y.; Ding, H.; Li, Y.; Lu, A. The Fine Characterization and Potential Photocatalytic Effect of Semiconducting Metal Minerals in Danxia Landforms. Minerals 2018, 8, 554. https://doi.org/10.3390/min8120554
Xiao Y, Li Y, Ding H, Li Y, Lu A. The Fine Characterization and Potential Photocatalytic Effect of Semiconducting Metal Minerals in Danxia Landforms. Minerals. 2018; 8(12):554. https://doi.org/10.3390/min8120554
Chicago/Turabian StyleXiao, Yuxiong, Yanzhang Li, Hongrui Ding, Yan Li, and Anhuai Lu. 2018. "The Fine Characterization and Potential Photocatalytic Effect of Semiconducting Metal Minerals in Danxia Landforms" Minerals 8, no. 12: 554. https://doi.org/10.3390/min8120554
APA StyleXiao, Y., Li, Y., Ding, H., Li, Y., & Lu, A. (2018). The Fine Characterization and Potential Photocatalytic Effect of Semiconducting Metal Minerals in Danxia Landforms. Minerals, 8(12), 554. https://doi.org/10.3390/min8120554




