Case Studies for Clean Technology Development in the Chemical Industry Using Zeolite Based Catalysts
Abstract
1. Introduction
1.1. Nitrogen Oxides Emission
- Combustion of fuels: At high temperatures, the oxygen and nitrogen content of the air forms nitrogen oxides. Typical flue gas contains about 100–1500 ppm of nitrogen oxides.
- Nitric acid production in fertilizer manufacturing technology: Stack gases that cannot be absorbed usually contain 2–3% nitrogen oxides relative to the acid produced.
- Metal finishing operations: Several metal surface treatment procedures use nitrates, nitrites or nitric acid, which generate nitrogen oxides.
- Chemical processes: Many chemical technologies (explosives, plastics, nitrated materials, etc.) that use nitric acid, nitrates or nitrites result in the formation of nitrogen oxides. Cement kilns, glass manufacturers and power generating plants (where high processing temperatures are used) also produce NOx.
- Agriculture: Biochemical processes result in the generation of nitrous oxide in soils during nitrification, denitrification and decomposition of nitrogen compounds in livestock manure [23].
1.2. Biodiesel
2. Materials and Methods
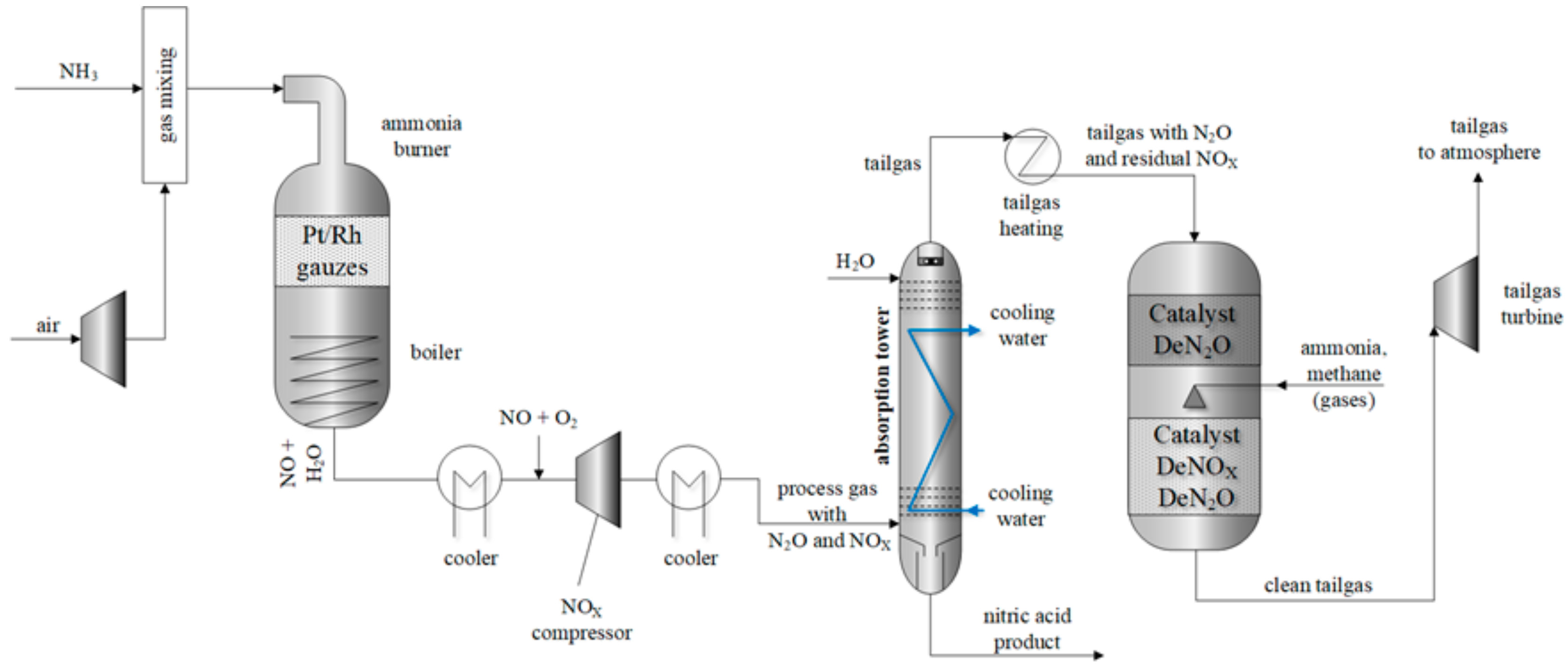
2.1. Selective Catalytic Reduction of Nitrogen Oxides with Ammonia
2.2. Biodiesel Production
3. Results
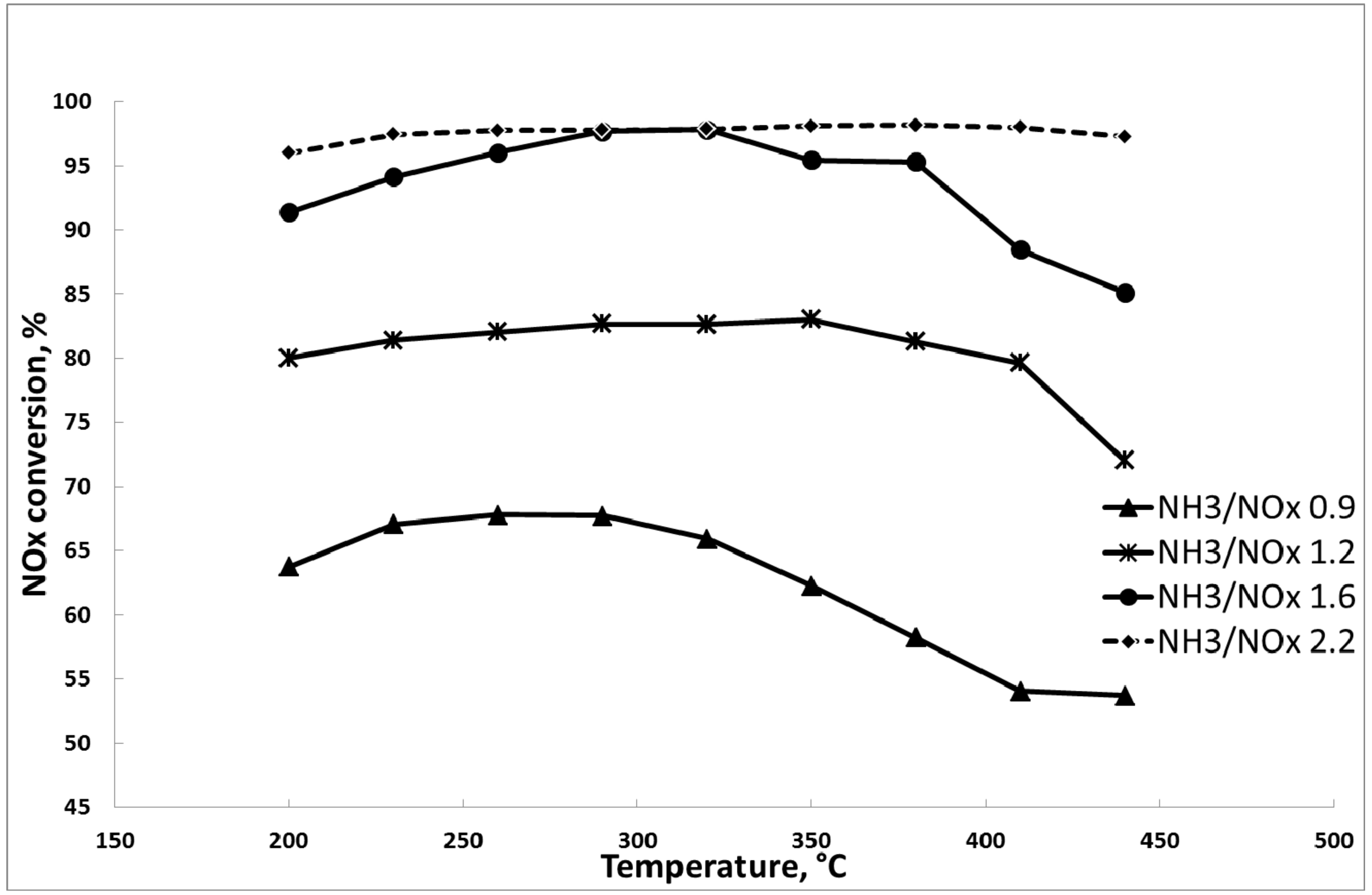
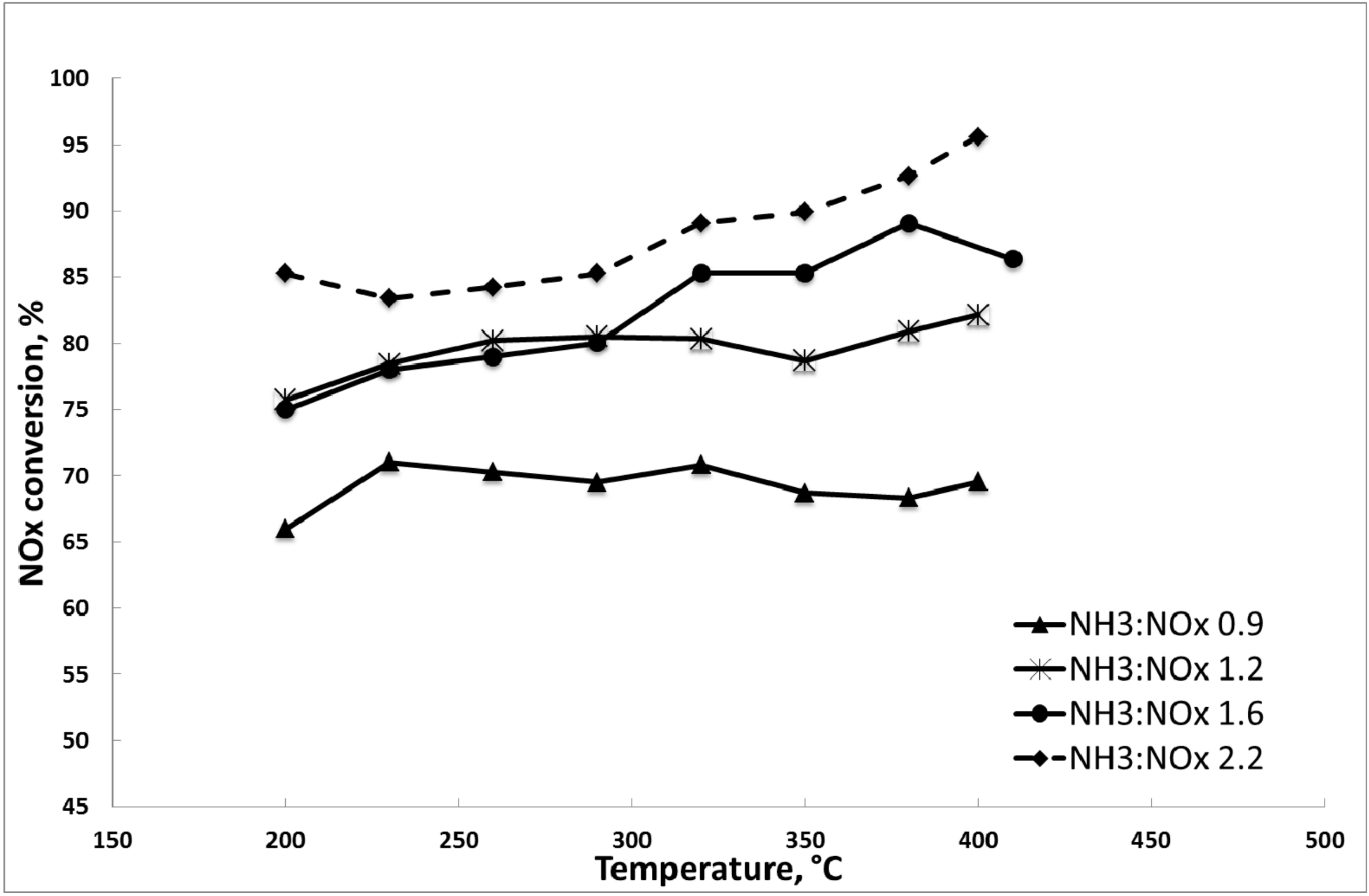
3.1. Results of the DeNOx Tests
3.1.1. Structural Properties of the Catalysts
3.1.2. Catalytic Tests
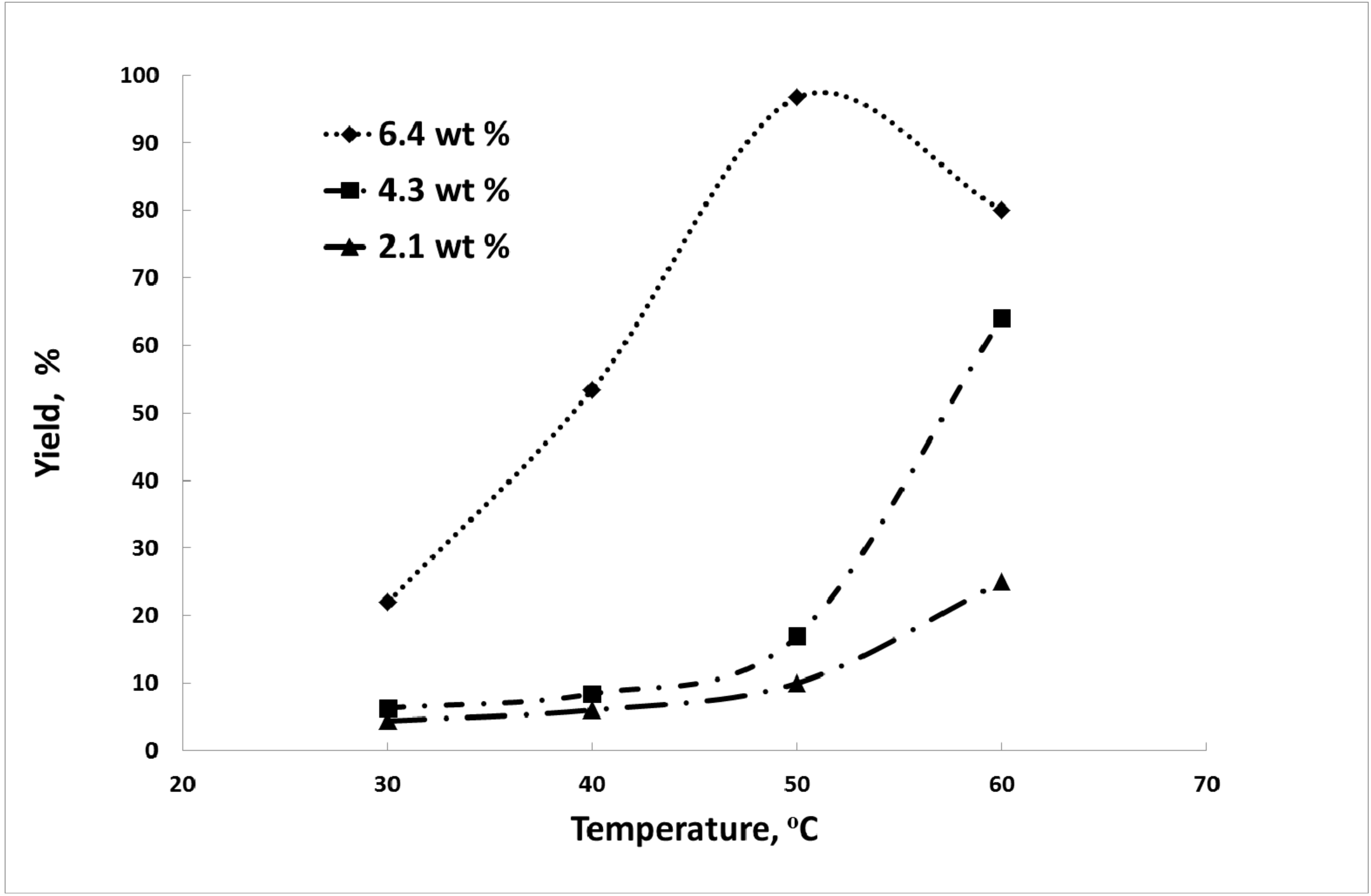
3.2. Biodiesel Production from Waste Vegetable Oil over Jordanian Zeolite as the Catalyst
3.2.1. Properties of the Zeolitic Tuff
3.2.2. Effect of Reaction Temperature
4. Discussion on Sustainability Issues
5. Conclusions
- The first case study aimed at decreasing the detrimental emissions of nitric acid production.
- The second case study dealt with the use of waste vegetable oil as a secondary raw material to produce biodiesel, making possible an important economy for diesel production.
Author Contributions
Funding
Conflicts of Interest
References
- Gavrilescu, M. Cleaner production as a tool for sustainable development. Environ. Eng. Manag. J. 2004, 3, 45–70. [Google Scholar] [CrossRef]
- Harja, M.; Ciocinta, R.C.; Cretescu, I.; Apostolescu, M.; Barbuta, M. Crystal growth of calcium carbonate with various morphologies from residual calcium chloride solution. Rev. Chim. 2014, 60, 1025–1031. [Google Scholar]
- Nowotny, J.; Dodson, J.; Fiechter, S.; Gür, T.M.; Kennedye, B.; Macyk, W.; Bak, T.; Sigmund, W.; Yamawaki, M.; RahmanA, K.A. Towards global sustainability: Education on environmentally clean energy technologies. Renew. Sustain. Energy Rev. 2018, 81, 2541–2551. [Google Scholar] [CrossRef]
- Zhai, Q.; Cao, H.; Zhao, X.; Yuan, C. Cost benefit analysis of using clean energy supplies to reduce greenhouse gas emissions of global automotive manufacturing. Energies 2011, 4, 1478–1494. [Google Scholar] [CrossRef]
- Fisher, B.S.; Nakicenovic, N.; Alfsen, K.; Corfee Morlot, J.; de la Chesnaye, F.; Hourcade, J.-C.; Jiang, K.; Kainuma, M.; La Rovere, E.; Matysek, A.; et al. Issues related to mitigation in the long term context. In Climate Change 2007: Mitigation; Metz, B., Davidson, O.R., Bosch, P.R., Dave, R., Meyer, L.A., Eds.; Contribution of Working Group III to 4th Assessment Report of the Inter-governmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2007. [Google Scholar]
- Verbruggen, A. Annex I: Glossary. In Climate Change 2007: Mitigation; Metz, B., Davidson, O.R., Bosch, P.R., Dave, R., Meyer, L.A., Eds.; Contribution of Working Group III to 4th Assessment Report of the Inter-governmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2007. [Google Scholar]
- Fu, Y.; Kok, R.A.W.; Dankbaar, B.; Ligthart, P.E.M.; van Riel, A.C.R. Factors affecting sustainable process technology adoption: A systematic literature review. J. Clean. Prod. 2018, 205, 226–251. [Google Scholar] [CrossRef]
- Development and International Co-operation: Environment. Report of the World Commission on Environment and Development: Our Common Future; Note by the Secretary-General, A/42/427; United Nation: Geneva, Switzerland, 1987. [Google Scholar]
- Gbededo, M.A.; Liyanage, K. Identification and alignment of the social aspects of sustainable manufacturing with the theory of motivation. Sustainability 2018, 10, 852. [Google Scholar] [CrossRef]
- Paulik, K. We have to be sincere to sustainable development. Innotéka 2016, 29, 31–36. (In Hungarian) [Google Scholar]
- Vaccaro, L. Green chemistry. Beilstein J. Org. Chem. 2016, 12, 2763–2765. [Google Scholar] [CrossRef] [PubMed]
- McKinney, M.L.; Schoch, R.M.; Yonavjak, L.; Mincy, G.A. Environmental Science. Systems and Solutions, 6th ed.; Jones and Bartlett Leaning: Burlington, MA, USA, 2018; ISBN 13-978-1284091700. [Google Scholar]
- Van Bueren, E. Environmental Policy. Encyclopædia Britannica, Inc. Available online: https://www.britannica.com/topic/environmental-policy (accessed on 22 April 2018).
- 2008 Annual Report; Blaszek I., Ed.; Nitrogénművek Zrt., Hungary, 2008. Available online: http://www.nitrogen.hu/ (accessed on 1 October 2018).
- Sustainability, Air Quality, Shell Global. Available online: https://www.shell.com/sustainability/environment/air-quality.html (accessed on 1 October 2018).
- Li, Y.; Li, L.; Yu, J. Application of zeolites in sustainable chemistry. Review. Chem. 2017, 3, 928–949. [Google Scholar] [CrossRef]
- Robu, B.M.; Caliman, F.A.; Betianu, C.; Gavrilescu, M. Methods and procedures for environmental risk assessment. Environ. Eng. Manag. J. 2007, 6, 573–592. [Google Scholar] [CrossRef]
- Cojocaru, C.; Cocârtă, D.M.; Istrate, I.A.; Cretescu, I. Graphical methodology of global pollution index for the environmental impact assessment using two environmental components. Sustainability 2017, 9, 593. [Google Scholar] [CrossRef]
- Skalska, K.; Miller, J.S.; Ledakowicz, S. Trends in NOx abatement. Sci. Total Environ. 2010, 408, 3976–3989. [Google Scholar] [CrossRef] [PubMed]
- Air Pollution Fact Sheet 2014: Hungary. European Environment Agency, EEA, Copenhagen K, Denmark. 2014. Available online: https://www.eea.europa.eu/themes/air/air-pollution-country-fact-sheets-2014/hungary-air-pollutant-emissions-country-factsheet (accessed on 9 September 2018).
- Ministerial Decree 4/2011 (I. 14.) VM on Air Pollution Thresholds and Emissions Ceilings for Located Air Pollutant Point, Hungary. Available online: https://net.jogtar.hu/jogszabaly?docid=a1100004.vm (accessed on 19 April 2018). (In Hungarian).
- Nitrogen Oxides (NOx) Abatement with Hydrogen Peroxide, USP Technologies, Solutions for a Clean Environment, Canada. Available online: http://www.h2o2.com/industrial/applications.aspx?pid=101 (accessed on 2 June 2018).
- Ussiri, D.; Lal, R. Soil Emission of Nitrous Oxide and Its Mitigation; Springer: Berlin, Germany, 2013; ISBN 978-94-007-5364-8. [Google Scholar]
- Popp, J.; Harangi-Rákos, M.; Gabnai, Z.; Balogh, P.; Antal, G.; Bai, A. Biofuels and their co-products as livestock feed: Global economic and environmental implications. Molecules 2016, 21, 285. [Google Scholar] [CrossRef] [PubMed]
- Andrade, J.E.; Perez, A.; Sebastian, P.J.; Eapen, D. Review: A review of bio-diesel production processes. Biomass Bioenergy 2011, 35, 1008–1020. [Google Scholar] [CrossRef]
- Lotero, E.; Liu, Y.; Lopez, D.E.; Suwannakarn, K.; Bruce, D.A.; Goodwin, J.G. Synthesis of biodiesel via acid catalysis. Ind. Eng. Chem. 2005, 44, 5353–5363. [Google Scholar]
- Fukuda, H.; Kondo, A.; Noda, H. Biodiesel fuel production by transesterification of oils. J. Biosci. Bioeng. 2001, 92, 405–416. [Google Scholar] [CrossRef]
- Hoque, A.; Singh, A.; Chuan, Y.L. Biodiesel from low cost feedstocks: The effects of process parameters on the biodiesel yield. Biomass Bioenergy 2011, 35, 1582–1587. [Google Scholar] [CrossRef]
- Gaurav, A.; Flora, T.T.N.; Garry, L. A new green process for biodiesel production from waste oils via catalytic distillation using a solid acid catalyst–Modeling, economic and environmental analysis. Green Energy Environ. 2016, 1, 62–74. [Google Scholar] [CrossRef]
- Borges, M.E.; Diaz, L. Recent developments on heterogeneous catalysts for biodiesel production by oil esterification and transesterification reactions: A review. Renew. Sustain. Energy Rev. 2012, 16, 2839–2849. [Google Scholar] [CrossRef]
- Vieitez, I.; Da Silva, C.; Alckmin, I.; Borges, R.; Corazza, C.; Oliveira, V.; Grompone, A.; Jachmanián, I. Continuous catalyst-free methanolysis and ethanolysis of soybean oil under supercritical alcohol/water mixtures. Renew. Energy 2010, 35, 1976–1981. [Google Scholar] [CrossRef]
- Haas, J.; Foglia, A. Chapter 4: Alternate feedstocks and technologies for biodiesel production. In Biodiesel Handbook; Khothe, G., Van Gerpen, J., Eds.; AOCS Publishing: New York, NY, USA, 2005; ISBN 978-1-4398-2235-7. [Google Scholar]
- Shuler, M.L.; Kargi, F. Bioprocess Engineering: Basic Concepts, 2nd ed.; Prentice Hall: Upper Saddle River, NJ, USA, 2002; ISBN 0-13-081908-5. [Google Scholar]
- Walsh, B.P.; Og Cusack, D.; O’Sullivan, D.T.J. An industrial water management value system framework development. Sustain. Prod. Consum. 2016, 5, 82–93. [Google Scholar] [CrossRef]
- Hevia, M.A.G.; Pérez-Ramírez, J. Assessment of the low-temperature EnviNOx® variant for catalytic N2O abatement over steam-activated. Appl. Catal. B-Environ. 2008, 77, 248–254. [Google Scholar] [CrossRef]
- Al-Jammal, N.; Al-Hamamre, Z.; Alnaief, M. Manufacturing of zeolite based catalyst from zeolite tuff for biodiesel production from waste sunflower oil. Renew. Energy 2016, 93, 449–459. [Google Scholar] [CrossRef]
- Zahan, K.A.; Kano, M. Biodiesel production from palm oil, its by-products, and mill effluent: A review. Energies 2018, 11, 2132. [Google Scholar] [CrossRef]
- Biodiesel Production and Distribution U.S. Department of Energy–Energy Efficiency and Renewable Energy. Alternative Fuels Data Center. Available online: https://www.afdc.energy.gov/fuels/biodiesel_production.html (accessed on 2 June 2018).
- Al-Hamamre, Z.; Yamin, J. Parametric study of the alkali catalyzed transesterification of waste frying oil for biodiesel production. Energ. Convers. Manag. 2014, 79, 246–254. [Google Scholar] [CrossRef]
- Leung, C.; Guo, Y. Transesterification of neat and used frying oil: Optimization for biodiesel production. Fuel Process. Technol. 2006, 87, 883–890. [Google Scholar] [CrossRef]
- Heck, R.H.; Ferrauto, R.J. Catalytic Air Pollution Control. Commercial Technology; Van Nostrand Reinhold: New York, NY, USA, 1995. [Google Scholar]
- EPA. Available and Emerging Technologies for Reducing Greenhouse Gas Emissions from the Nitric Acid Production Industry, Sector Policies and Programs Division Office of Air Quality Planning and Standards; U.S. Environmental Protection Agency: Research Triangle Park, NC, USA, 2010. Available online: https://www.epa.gov/sites/production/files/2015-12/documents/nitricacid.pdf (accessed on 9 September 2018).
- Groves, M.; Sasonow, A. Uhde EnviNOx® Technology for NOX and N2O Abatement. A Contribution to Reducing Emissions from Nitric Acid Plants, Uhde GmbH, Dortmund, Germany. In Proceedings of the 5th International Symposium on Non-CO2 Greenhouse Gases (NCGG-5), Wageningen, The Netherlands, 30 June–3 July 2009; Available online: https://www.thyssenkrupp-industrial-solutions.com/media/download_1/nitrates/uhde_publications_pdf_en_15000012.pdf (accessed on 2 June 2018).
- Lai, S.; She, Y.; Zhan, W.; Guo, Y.; Guo, Y.; Wang, L.; Lu, G. Performance of Fe-ZSM-5 for selective catalytic reduction of NOx with NH3: Effect of the atmosphere during the preparation of catalysts. J. Mol. Catal. A-Chem. 2016, 424, 232–240. [Google Scholar] [CrossRef]
- Naber, J.E.; deJong, K.P.; Stork, W.H.J.; Kuipers, H.P.C.E.; Post, M.F.M. Industrial applications of zeolite catalysis. In Zeolites and Related Microporous Materials: State of the Art; Weitkamp, J., Karge, H.G., Pfeifer, H., Hölderich, W., Eds.; Elsevier: Amsterdam, The Netherlands, 1994; Volume 84, pp. 2197–2219. [Google Scholar]
- Fülöp, T.; Dobos, L.; Császár, V. Nitric acid production at Nitrogénművek Company, Hungary. Magy. Kem. Lapja 2013, 6, 178–183. [Google Scholar]
- EnviNOx®–Setting Emission, Standards for Nitric Acid Plants. ThyssenKrupp Uhde, Dortmund, Germany. Available online: www.digitalrefining.com/data/literature/file/386302294.pdf (accessed on 1 October 2018).
- Goering, C.E.; Fry, B. Engine durability screening test of a diesel oil/soy oil/alcohol microemulsion fuel. J. Am. Oil Chem. Soc. 1984, 61, 1627–1632. [Google Scholar] [CrossRef]
- Ayoola, A.A.; Hymore, K.F.; Omonhinmin, C.A. Optimization of biodiesel production from selected waste oils using response surface methodology. Biotechnology 2017, 16, 1–9. [Google Scholar]
- Dorodo, W. An alkali-catalyzed transesterification process for high free fatty acid waste oils. Trans. ASAE 2002, 45, 525–529. [Google Scholar]
- Eevera, T.; Rajendran, K.; Saradha, S. Biodiesel production process optimization and characterization to assess the suitability of the product for varied environmental conditions. Renew. Energy 2009, 34, 762–765. [Google Scholar] [CrossRef]
- Xie, W.; Huang, X.; Li, H. Soybean oil methyl esters preparation using NaX zeolites loaded with KOH as a heterogeneous catalyst. Bioresource Technol. 2007, 98, 936–939. [Google Scholar] [CrossRef] [PubMed]
- Intarapong, P.; Luengnaruemitchai, A.; Jai-In, S. Transesterification of palm oil over KOH/NaY zeolite in a packed-bed reactor. IJRER 2011, 4, 271–280. [Google Scholar]
- CO2 European Emission Allowances, Markets Insider. Available online: http://markets.businessinsider.com/commodities/co2-emissionsrechte (accessed on 22 April 2018).
- Liquid Petroleum Products–Fatty Acid Methyl Esters (FAME) for Use in Diesel Engines and Heating Applications–Requirements and Test Methods; German Version EN 14214:2012 + A1:2014. Beuth Verlag GmbH. Available online: https://www.beuth.de/en/standard/din-en-14214/197713876 (accessed on 2 June 2018).
- American Society for Testing and Materials (ASTM) D6751-02. Standard Specification for Biodiesel Fuel (B100) Blend Stock for Distillate Fuels; ASTM International: West Conshohocken, PA, USA, 2002. [Google Scholar]

| Sample | SBET, m2/g | Smicro, m2/g | V, cm3/g | Vmicro, cm2/g | Dav, nm |
|---|---|---|---|---|---|
| V2O5/TiO2 fresh | 39 | 2.0 | 0.2149 | 0.00062 | 18.2 |
| V2O5/TiO2 used | 35 | 2.4 | 0.2101 | 0.00080 | 20.0 |
| Fe-ZSM-5 fresh | 311 | 194.7 | 0.1737 | 0.09050 | 6.3 |
| Fe-ZSM-5 used | 312 | 195.2 | 0.1744 | 0.09127 | 6.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Juzsakova, T.; Al-Jammal, N.; Cretescu, I.; Sebestyén, V.; Le Phuoc, C.; Domokos, E.; Rédey, Á.; Stan, C.D. Case Studies for Clean Technology Development in the Chemical Industry Using Zeolite Based Catalysts. Minerals 2018, 8, 462. https://doi.org/10.3390/min8100462
Juzsakova T, Al-Jammal N, Cretescu I, Sebestyén V, Le Phuoc C, Domokos E, Rédey Á, Stan CD. Case Studies for Clean Technology Development in the Chemical Industry Using Zeolite Based Catalysts. Minerals. 2018; 8(10):462. https://doi.org/10.3390/min8100462
Chicago/Turabian StyleJuzsakova, Tatjána, Noor Al-Jammal, Igor Cretescu, Viktor Sebestyén, Cuong Le Phuoc, Endre Domokos, Ákos Rédey, and Catalina Daniela Stan. 2018. "Case Studies for Clean Technology Development in the Chemical Industry Using Zeolite Based Catalysts" Minerals 8, no. 10: 462. https://doi.org/10.3390/min8100462
APA StyleJuzsakova, T., Al-Jammal, N., Cretescu, I., Sebestyén, V., Le Phuoc, C., Domokos, E., Rédey, Á., & Stan, C. D. (2018). Case Studies for Clean Technology Development in the Chemical Industry Using Zeolite Based Catalysts. Minerals, 8(10), 462. https://doi.org/10.3390/min8100462







