Compositional and Textural Variations in Hainite-(Y) and Batievaite-(Y), Two Rinkite-Group Minerals from the Sakharjok Massif, Keivy Alkaline Province, NW Russia
Abstract
:1. Introduction
2. Materials and Methods
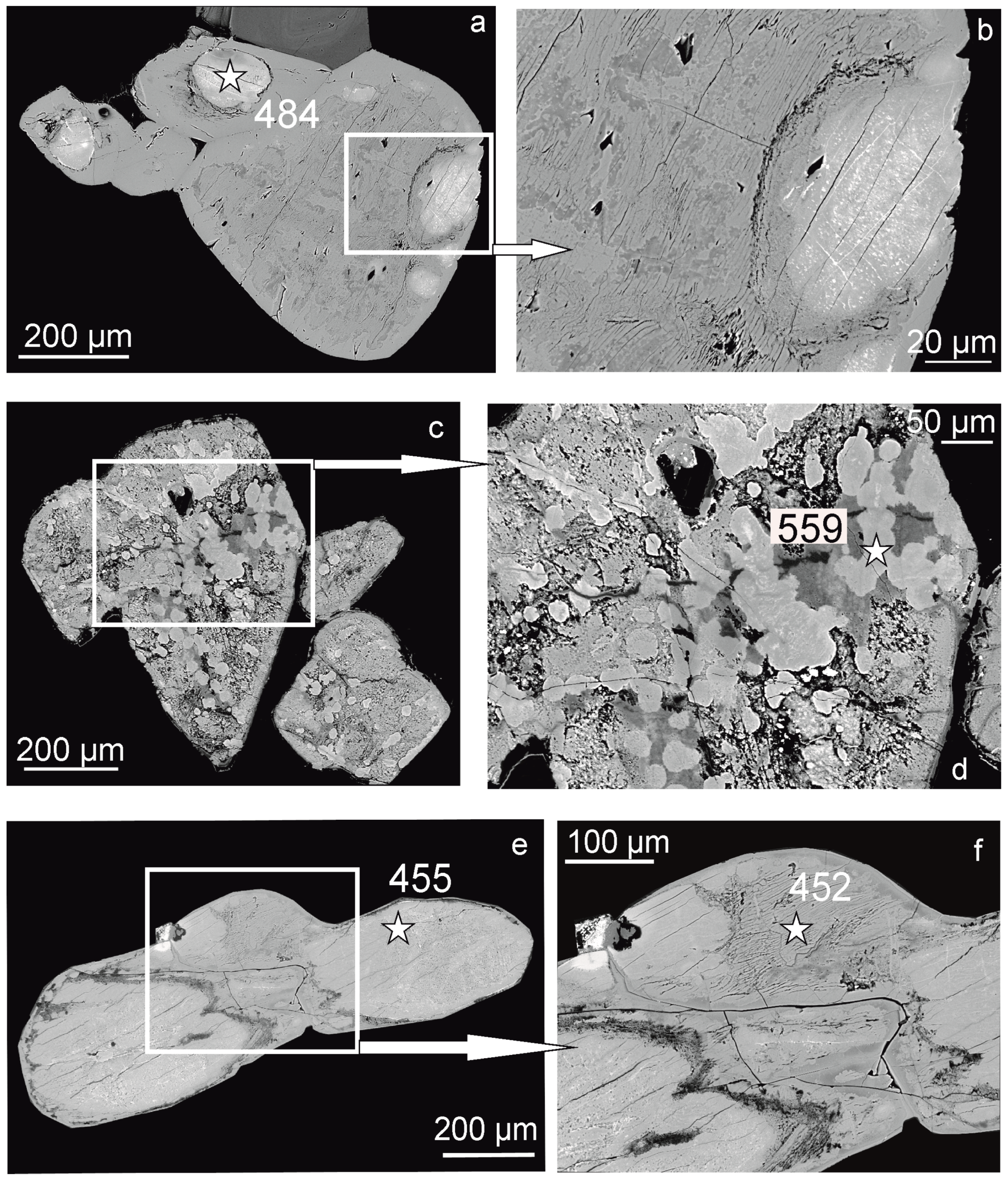
2.1. Occurrence
2.2. Chemical Composition
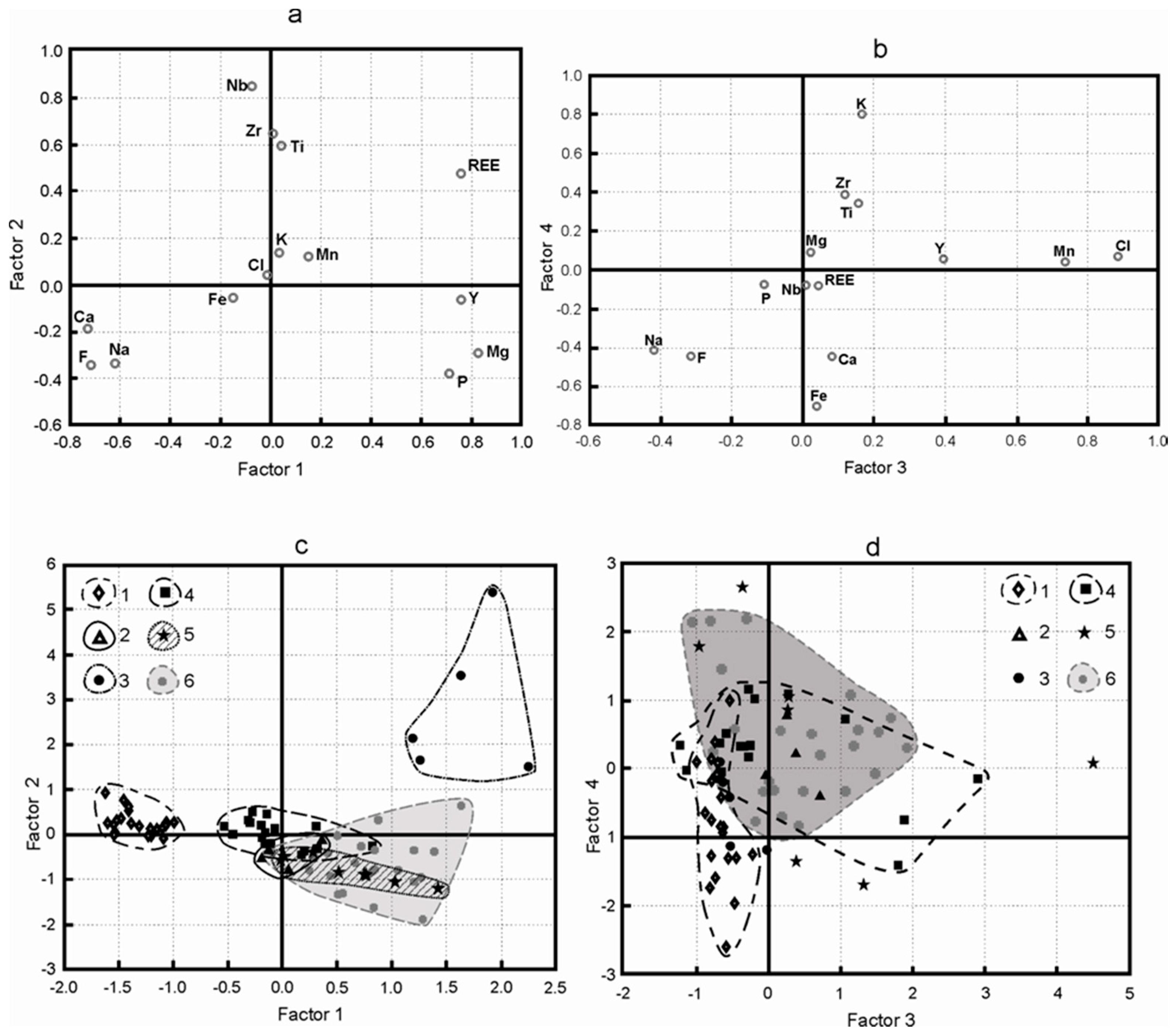
2.3. PCA
3. Results
3.1. Factor F1
3.2. Factor F2
3.3. Factor F3
3.4. Factor F4
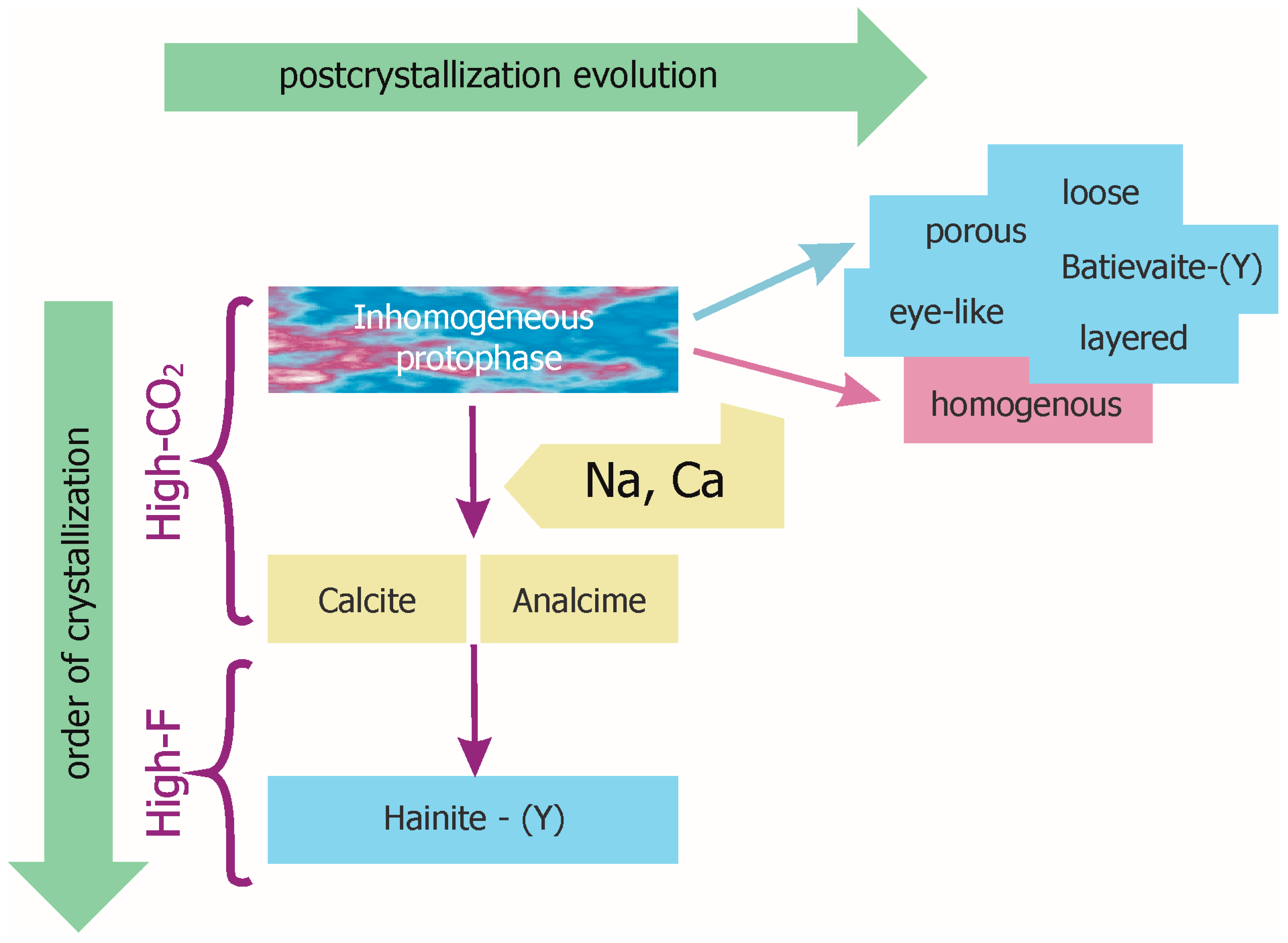
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lyalina, L.M.; Zolotarev, A.A., Jr.; Selivanova, E.A.; Savchenko, Y.E.; Zozulya, D.R.; Krivovichev, S.V.; Mikhailova, Y.A. Structural characterization and composition of Y-rich hainite from Sakharjok nepheline syenite pegmatite (Kola Peninsula, Russia). Miner. Pet. 2015, 109, 443–451. [Google Scholar] [CrossRef]
- Lyalina, L.M.; Zolotarev, A.A., Jr.; Selivanova, E.A.; Savchenko, Y.E.; Krivovichev, S.V.; Mikhailova, Y.A.; Kadyrova, G.I.; Zozulya, D.R. Batievaite-(Y), Y2Ca2Ti[Si2O7]2(OH)2(H2O)4, a new mineral from nepheline syenite pegmatite in the Sakharjok massif, Kola Peninsula, Russia. Miner. Pet. 2016, 110, 895–904. [Google Scholar] [CrossRef]
- Zolotarev, A.; Krivovichev, S.; Lyalina, L.; Selivanova, E. Crystal structure and chemistry of Na-deficient Y-dominant analogue of hainite/götzenite. Miner. Pet. 2016, 329, 895–904. [Google Scholar]
- Sokolova, E. From structure topology to chemical composition: I. Structural hierarchy and stereochemistry in titanium disilicate minerals. Can. Miner. 2006, 44, 1273–1330. [Google Scholar] [CrossRef]
- Sokolova, E.; Camara, F.C. The seidozerite supergroup of TS-block minerals: Nomenclature and classification, with change of the following names: Rinkite to rinkite-(Ce), mosandrite to mosandrite-(Ce), hainite to hainite-(Y) and innelite-1T to innelite-1A. Miner. Mag. 2017, 81, 1457–1484. [Google Scholar] [CrossRef]
- Pautov, L.A.; Agakhanov, A.A.; Karpenko, V.Y.; Uvarova, Y.A.; Sokolova, E.; Hawthorne, F.C. Rinkite-(Y), IMA 2017-043. CNMNC Newsletter No. 39, October 2017, page 1280. Miner. Mag. 2017, 81, 1279–1286. [Google Scholar]
- Cannillo, E.; Mazzi, F.; Rossi, G. Crystal structure of götzenite. Sov. Phys. Crystallogr. 1972, 16, 1026–1030. [Google Scholar]
- Christiansen, C.C.; Johnsen, O.; Makovicky, E. Crystal chemistry of the rosenbuschite group. Can. Miner. 2003, 41, 1203–1224. [Google Scholar] [CrossRef]
- Rønsbo, J.G.; Sørensen, H.; Roda-Robles, E.; Fontan, F.; Monchoux, P. Rinkite-nacareniobsite-(Ce) solid solution series and hainite from the Ilímaussaq alkaline complex: Occurrence and compositional variation. Bull. Geol. Soc. Den. 2014, 62, 1–15. [Google Scholar]
- Sharygin, V.V.; Stoppa, F.; Kolesov, B.A. Zr-Ti disilicates from the Pian di Celle volcano, Umbria, Italy. Eur. J. Miner. 1996, 8, 1199–1212. [Google Scholar] [CrossRef]
- Sokolova, E.; Hawthorne, F.C. From structure topology to chemical composition. XIV. Titaniumsilicates: Refinement of the crystal structure and revision of the chemical formula of mosandrite, (Ca3REE)[(H2O)2Ca0.5□0.5]Ti(Si2O7)2(OH)2(H2O)2, a Group-I mineral from the Saga mine, Morje, Porsgrunn, Norway. Miner. Mag. 2013, 77, 2753–2771. [Google Scholar]
- Konopleva, N.G.; Ivanyuk, G.Y.; Pakhomovsky, Y.A.; Yakovenchuk, V.N.; Mikhailova, Y.A.; Selivanova, E.A. Typochemistry of Rinkite and Products of Its Alteration in the Khibiny Alkaline Pluton, Kola Peninsula. Geol. Ore Depos. 2015, 57, 614–625. [Google Scholar] [CrossRef]
- Selivanova, E.A.; Lyalina, L.M. Minerals of rinkite and rosenbuschite groups from Kola Peninsula. Regionalnaya Geologiya, Mineralogiya I Poleznyie Iskopaemyie Kolskogo Poluostrova; Trudy XIII Vserossiyskaya Fersmanovskaya nauchnaya sessiya, Apatity, Russia, 4–5 April 2016; Voytekhovsky, Y.L., Ed.; K & M: Apatity, Russia, 2016; pp. 97–101. (In Russian) [Google Scholar]
- Lyalina, L.M.; Zozulya, D.; Selivanova, E.; Savchenko, Y. Genetic relationship between batievaite-(Y) and hainite-(Y) from Sakharjok nepheline syenite pegmatite, Keivy alkaline province, NW Russia. In Proceedings of the Conference on Accessory Minerals—2017, Vienna, Austria, 13–17 September 2017; pp. 71–72. [Google Scholar]
- Khomyakov, A.P.; Yushkin, N.P. The principle of inheritance in crystallogenesis. Dokl. Akad. Nauk SSSR 1981, 256, 1229–1233. [Google Scholar]
- Slepnev, Y.S. The minerals of the rinkite group. Izv. Akad. Nauk. USSR Ser. Geol. 1957, 3, 63–75. [Google Scholar]
- Lykova, I.S.; Pekov, I.V.; Zubkova, N.V.; Chukanov, N.V.; Yapaskurt, V.O.; Chervonnaya, N.A.; Zolotarev, A.A. Crystal chemistry of cation-exchanged forms of epistolite-group minerals, Part, I. Ag- and Cu-exchanged lomonosovite and Ag-exchanged murmanite. Eur. J. Miner. 2015, 27, 535–549. [Google Scholar] [CrossRef]
- Lykova, I.S.; Pekov, I.V.; Zubkova, N.V.; Yapaskurt, V.O.; Chervonnaya, N.A.; Zolotarev, A.A.; Giester, G. Crystal chemistry of cation-exchanged forms of epistolite-group minerals. Part II. Vigrishinite and Zn-exchanged murmanite. Eur. J. Miner. 2015, 27, 669–682. [Google Scholar] [CrossRef]
- Batieva, I.D.; Belkov, I.V. Sakharjokskii Peralkaline Massif, Component Rocks and Minerals; Kola Branch of USSR Academy of Sciences: Apatity, Russia, 1984; p. 133. (In Russian) [Google Scholar]
- Zozulya, D.R.; Lyalina, L.M.; Eby, N.; Savchenko, Y.E. Ore Geochemistry, Zircon Mineralogy, and Genesis of the Sakharjok Y–Zr Deposit, Kola Peninsula, Russia. Geol. Ore Depos. 2012, 54, 81–98. [Google Scholar] [CrossRef]
- Lyalina, L.M.; Savchenko, Y.E.; Selivanova, E.A.; Zozulya, D.R. Behoite and Mimetite from the Saharjok Alkaline Pluton, Kola Peninsula. Geol. Ore Depos. 2010, 52, 641–645. [Google Scholar] [CrossRef]

| Mineral | Ideal Structural Formula | ||||||
|---|---|---|---|---|---|---|---|
| 2AP | 2MH | 4MO | (Si2O7)n | 2(XOM) | 2(XOA) | ||
| Mosandrite-(Ce) | Ca2 | (CaREE) | (H2O)2Ca0.5□0.5 | Ti | (Si2O7)2 | (OH)2 | (H2O)2 |
| Rinkite-(Ce) | Ca2 | (CaREE) | Na(NaCa) | Ti | (Si2O7)2 | (OF) | F2 |
| Rinkite-(Y) | Ca2 | (CaY) | Na(NaCa) | Ti | (Si2O7)2 | (OF) | F2 |
| Nacareniobsite-(Ce) | (Ca, REE)2 | (Ca, REE)2 | Na3 | Nb | (Si2O7)2 | (OF) | F2 |
| Seidozerite | Na2 | Zr2 | Na2Mn | Ti | (Si2O7)2 | O2 | F2 |
| Grenmarite | Na2 | Zr2 | Na2Mn | Zr | (Si2O7)2 | O2 | F2 |
| Rosenbuschite | Ca4 | Ca2Zr2 | Na2Na4 | TiZr | (Si2O7)4 | O2F2 | F4 |
| Kochite | Ca2 | MnZr | Na3 | Ti | (Si2O7)2 | OF | F2 |
| Gotzenite | Ca2 | Ca2 | NaCa2 | Ti | (Si2O7)2 | (OF) | F2 |
| Hainite-(Y) | Ca2 | (CaY) | Na(NaCa) | Ti | (Si2O7)2 | (OF) | F2 |
| Batievaite-(Y) | Ca2 | Y2 | (H2O)2□ | Ti | (Si2O7)2 | (OH)2 | (H2O)2 |
| Fogoite-(Y) | Ca2 | Y2 | Na3 | Ti | (Si2O7)2 | (OF) | F2 |
| An. No.* | 452 | 484 | 455 | 475 | 559 | 470 | 477 | 469 | Variation |
|---|---|---|---|---|---|---|---|---|---|
| SiO2 | 32.39 | 32.97 | 28.63 | 29.46 | 31.62 | 30.96 | 30.54 | 30.70 | 24.60–33.03 |
| Al2O3 | 1.13 | 0.91 | 2.06 | 0.90 | 0.91 | 0.73 | 1.98 | 0.43 | 0.13–2.30 |
| TiO2 | 8.66 | 9.11 | 12.43 | 9.55 | 5.74 | 8.13 | 11.83 | 7.45 | 3.80–13.92 |
| ZrO2 | 1.48 | 1.92 | 1.61 | 3.04 | 1.09 | 2.41 | 3.60 | 1.91 | 0.94–5.18 |
| Nb2O5 | 1.63 | 1.84 | 1.60 | 2.37 | 1.02 | 2.18 | 3.03 | 1.71 | 0.35–4.78 |
| P2O5 | 0.00 | 0.00 | 0.00 | 0.00 | 0.19 | 0.00 | 0.00 | 0.00 | 0–0.50 |
| MnO | 0.27 | 0.33 | 2.34 | 1.72 | 1.19 | 0.58 | 0.63 | 0.15 | 0.14–8.92 |
| MgO | 0.11 | 0.00 | 0.27 | 0.00 | 0.45 | 0.00 | 0.00 | 0.06 | 0–0.45 |
| Fe2O3 | 0.19 | 0.14 | 0.28 | 0.08 | 0.22 | 0.15 | 0.21 | 0.22 | 0–0.66 |
| CaO | 24.64 | 24.20 | 21.86 | 22.72 | 16.29 | 26.16 | 14.67 | 27.95 | 12.28–34.73 |
| Na2O | 2.50 | 1.64 | 0.74 | 0.56 | 0.18 | 3.19 | 0.50 | 6.16 | 0.16–7.62 |
| K2O | 0.07 | 0.00 | 0.04 | 0.03 | 0.02 | 0.07 | 0.12 | 0.05 | 0–0.16 |
| Y2O3 | 10.87 | 11.07 | 11.23 | 12.71 | 15.35 | 10.96 | 10.41 | 9.46 | 7.61–18.51 |
| La2O3 | 0.32 | 0.44 | 0.60 | 0.31 | 1.01 | 0.17 | 0.59 | 0.00 | 0–1.58 |
| Ce2O3 | 0.28 | 0.23 | 0.56 | 0.47 | 2.77 | 0.24 | 0.64 | 0.00 | 0–3.18 |
| Pr2O3 | 0.00 | 0.00 | 0.00 | 0.00 | 0.21 | 0.00 | 0.21 | 0.00 | 0–0.57 |
| Nd2O3 | 0.00 | 0.00 | 0.12 | 0.00 | 0.54 | 0.14 | 0.29 | 0.00 | 0–1.04 |
| Sm2O3 | 0.00 | 0.00 | 0.00 | 0.00 | 0.11 | 0.00 | 0.00 | 0.00 | 0–0.26 |
| Gd2O3 | 0.00 | 0.00 | 0.20 | 0.00 | 0.39 | 0.00 | 0.00 | 0.00 | 0–0.40 |
| Tb2O3 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0–0.16 |
| Dy2O3 | 0.57 | 0.44 | 0.50 | 0.38 | 0.68 | 0.53 | 0.49 | 0.43 | 0.23–0.93 |
| Ho2O3 | 0.00 | 0.00 | 0.00 | 0.00 | 0.41 | 0.00 | 0.00 | 0.00 | 0–0.46 |
| Er2O3 | 0.71 | 0.90 | 0.94 | 0.94 | 1.27 | 1.10 | 1.37 | 0.94 | 0.71–1.61 |
| Tm2O3 | 0.11 | 0.36 | 0.19 | 0.34 | 0.22 | 0.24 | 0.35 | 0.25 | 0.11–0.46 |
| Yb2O3 | 2.38 | 2.10 | 2.00 | 2.48 | 2.14 | 2.94 | 3.35 | 2.70 | 1.83–4.02 |
| Lu2O3 | 0.37 | 0.52 | 0.36 | 0.61 | 0.27 | 0.44 | 0.62 | 0.39 | 0.21–0.80 |
| F | 3.27 | 3.46 | 1.81 | 1.85 | 0.00 | 3.31 | 1.10 | 5.95 | 0–6.80 |
| Cl | 0.05 | 0.00 | 0.22 | 0.15 | 0.00 | 0.10 | 0.00 | 0.00 | 0–0.35 |
| Sum | 92.00 | 92.58 | 90.57 | 90.65 | 84.27 | 94.72 | 86.50 | 96.90 | |
| apfu based on Si + Al = 4 | |||||||||
| Si | 3.84 | 3.87 | 3.69 | 3.86 | 3.87 | 3.89 | 3.72 | 3.94 | 3.67–3.98 |
| Al | 0.16 | 0.13 | 0.31 | 0.14 | 0.13 | 0.11 | 0.28 | 0.06 | 0.02–0.33 |
| Ti | 0.77 | 0.80 | 1.20 | 0.94 | 0.53 | 0.77 | 1.08 | 0.72 | 0.34–1.58 |
| Zr | 0.09 | 0.11 | 0.10 | 0.19 | 0.07 | 0.15 | 0.21 | 0.12 | 0.06–0.37 |
| Nb | 0.09 | 0.10 | 0.09 | 0.14 | 0.06 | 0.12 | 0.17 | 0.10 | 0.02–0.31 |
| P | 0.00 | 0.00 | 0.00 | 0.00 | 0.02 | 0.00 | 0.00 | 0.00 | 0–0.05 |
| Mn | 0.03 | 0.03 | 0.25 | 0.19 | 0.12 | 0.06 | 0.06 | 0.02 | 0.01–1.07 |
| Mg | 0.02 | 0.00 | 0.05 | 0.00 | 0.08 | 0.00 | 0.00 | 0.01 | 0–0.08 |
| Fe | 0.02 | 0.01 | 0.03 | 0.01 | 0.02 | 0.01 | 0.02 | 0.02 | 0–0.07 |
| Ca | 3.13 | 3.05 | 3.02 | 3.19 | 2.13 | 3.52 | 1.91 | 3.84 | 1.54–4.75 |
| Na | 0.57 | 0.37 | 0.18 | 0.14 | 0.04 | 0.78 | 0.12 | 1.53 | 0.04–1.86 |
| K | 0.01 | 0.00 | 0.01 | 0.00 | 0.00 | 0.01 | 0.02 | 0.01 | 0–0.02 |
| Y | 0.69 | 0.69 | 0.77 | 0.89 | 1.00 | 0.73 | 0.67 | 0.65 | 0.57–1.21 |
| La | 0.01 | 0.03 | 0.03 | 0.02 | 0.05 | 0.01 | 0.03 | 0.00 | 0–0.08 |
| Ce | 0.01 | 0.03 | 0.03 | 0.02 | 0.12 | 0.01 | 0.03 | 0.00 | 0–0.15 |
| Pr | 0.00 | 0.00 | 0.00 | 0.00 | 0.01 | 0.00 | 0.01 | 0.00 | 0–0.03 |
| Nd | 0.00 | 0.01 | 0.01 | 0.00 | 0.02 | 0.01 | 0.01 | 0.00 | 0–0.05 |
| Sm | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0–0.01 |
| Gd | 0.00 | 0.01 | 0.01 | 0.00 | 0.02 | 0.00 | 0.00 | 0.00 | 0–0.02 |
| Tb | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0–0.01 |
| Dy | 0.02 | 0.02 | 0.02 | 0.02 | 0.03 | 0.02 | 0.02 | 0.02 | 0.01–0.04 |
| Ho | 0.00 | 0.00 | 0.00 | 0.00 | 0.02 | 0.00 | 0.00 | 0.00 | 0–0.02 |
| Er | 0.03 | 0.04 | 0.04 | 0.04 | 0.05 | 0.04 | 0.05 | 0.04 | 0.03–0.07 |
| Tm | 0.00 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.00–0.02 |
| Yb | 0.09 | 0.08 | 0.08 | 0.10 | 0.08 | 0.11 | 0.12 | 0.11 | 0.07–0.19 |
| Lu | 0.01 | 0.01 | 0.01 | 0.02 | 0.01 | 0.02 | 0.02 | 0.02 | 0.01–0.03 |
| Sum cations | 9.59 | 9.40 | 9.94 | 9.92 | 8.49 | 10.38 | 8.56 | 11.22 | |
| F | 1.23 | 1.29 | 0.74 | 0.77 | 0.00 | 1.32 | 0.42 | 2.41 | 0–2.73 |
| Cl | 0.01 | 0.00 | 0.05 | 0.03 | 0.00 | 0.02 | 0.00 | 0.00 | 0–0.08 |
| Variable | Factor Loadings | |||
|---|---|---|---|---|
| Factor 1 | Factor 2 | Factor 3 | Factor 4 | |
| REE | 0.756320 | 0.476891 | 0.041060 | −0.082863 |
| Y | 0.754054 | −0.058515 | 0.394748 | 0.054088 |
| Na | −0.624196 | −0.337260 | −0.420135 | −0.412386 |
| K | 0.029323 | 0.141516 | 0.164027 | 0.802367 |
| Mg | 0.822040 | −0.288034 | 0.016398 | 0.091280 |
| Ca | −0.734975 | −0.183786 | 0.079822 | −0.440946 |
| Ti | 0.038583 | 0.595748 | 0.154522 | 0.348134 |
| Zr | 0.007376 | 0.643992 | 0.115776 | 0.387322 |
| P | 0.709521 | −0.378585 | −0.111755 | −0.071313 |
| Nb | −0.074589 | 0.843153 | 0.008262 | −0.076845 |
| Mn | 0.145517 | 0.123806 | 0.734688 | 0.044299 |
| Fe | −0.150331 | −0.055727 | 0.037116 | −0.700456 |
| F | −0.715492 | −0.339113 | −0.316716 | −0.439336 |
| Cl | −0.012788 | 0.038252 | 0.883334 | 0.069680 |
| Expl.Var | 3.813428 | 2.240130 | 1.839141 | 1.999090 |
| Prp.Totl | 0.272388 | 0.160009 | 0.131367 | 0.142792 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Selivanova, E.A.; Lyalina, L.M.; Savchenko, Y.E. Compositional and Textural Variations in Hainite-(Y) and Batievaite-(Y), Two Rinkite-Group Minerals from the Sakharjok Massif, Keivy Alkaline Province, NW Russia. Minerals 2018, 8, 458. https://doi.org/10.3390/min8100458
Selivanova EA, Lyalina LM, Savchenko YE. Compositional and Textural Variations in Hainite-(Y) and Batievaite-(Y), Two Rinkite-Group Minerals from the Sakharjok Massif, Keivy Alkaline Province, NW Russia. Minerals. 2018; 8(10):458. https://doi.org/10.3390/min8100458
Chicago/Turabian StyleSelivanova, Ekaterina A., Lyudmila M. Lyalina, and Yevgeny E. Savchenko. 2018. "Compositional and Textural Variations in Hainite-(Y) and Batievaite-(Y), Two Rinkite-Group Minerals from the Sakharjok Massif, Keivy Alkaline Province, NW Russia" Minerals 8, no. 10: 458. https://doi.org/10.3390/min8100458
APA StyleSelivanova, E. A., Lyalina, L. M., & Savchenko, Y. E. (2018). Compositional and Textural Variations in Hainite-(Y) and Batievaite-(Y), Two Rinkite-Group Minerals from the Sakharjok Massif, Keivy Alkaline Province, NW Russia. Minerals, 8(10), 458. https://doi.org/10.3390/min8100458





