Vanadium Mineralization in the Kola Region, Fennoscandian Shield
Abstract
1. Introduction
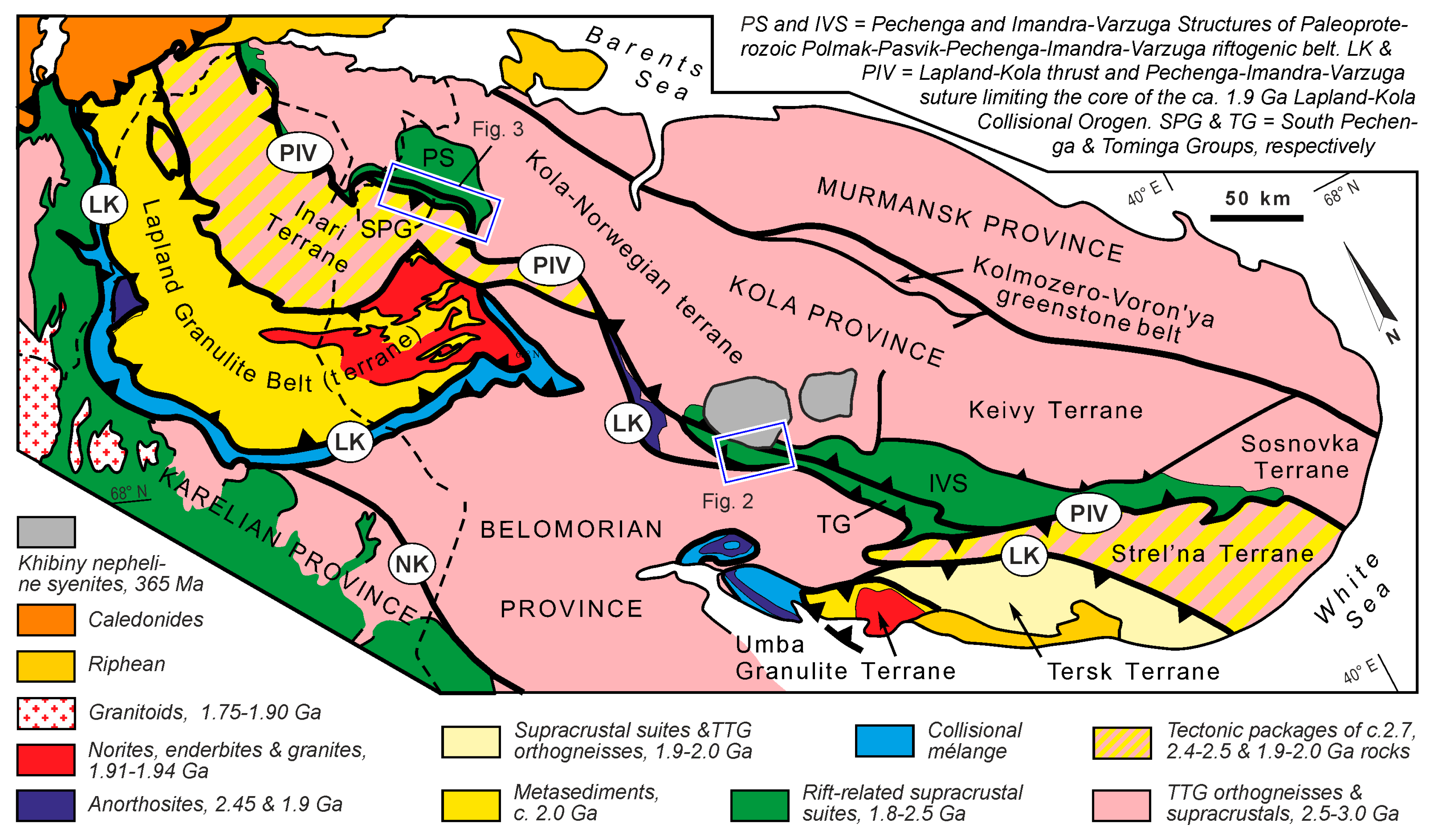
2. Geological Setting
2.1. Imandra-Varzuga Structure
2.2. Pechenga Structure
3. Methods
4. Vanadium Mineralization in the Kola Region
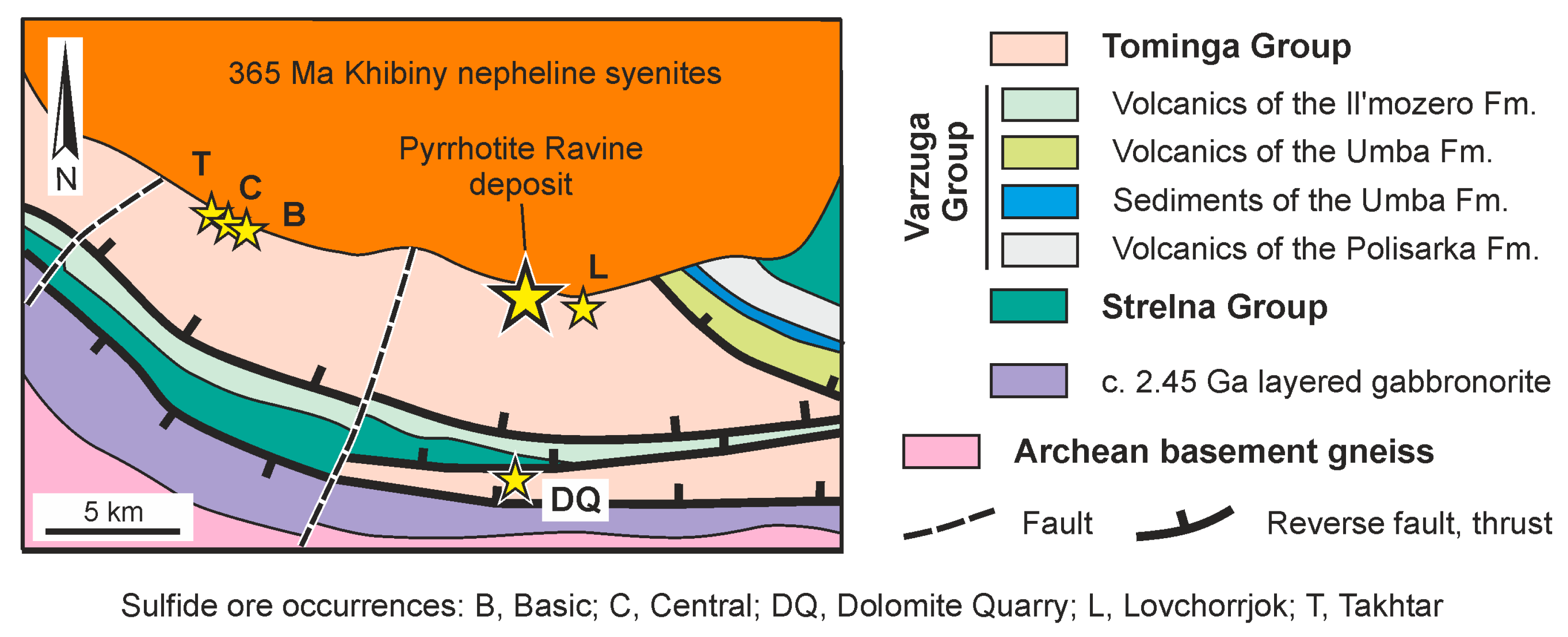
4.1. Pyrrhotite Ravine Sulfide Ore Deposit
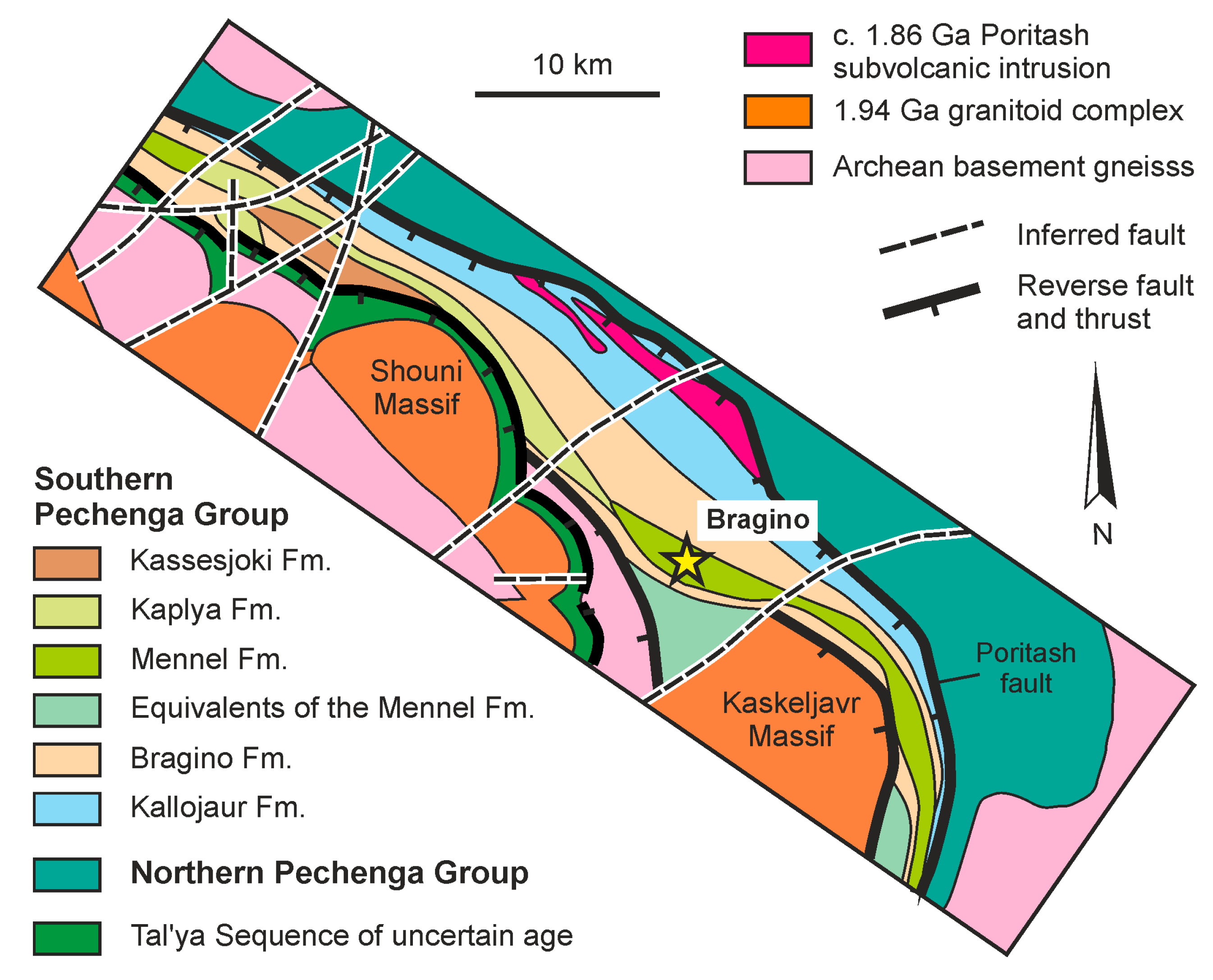
4.2. Bragino Sulfide Ore Occurrence
4.3. Vanadium Mineralization not Related to Sulfide Ores
5. Vanadium Mineralization Throughout the World
5.1. Vihanti, Finland
5.2. Outokumpu, Finland
5.3. Sätra, Sweden
5.4. Rampura Agucha, India
5.5. Biggejavri, Norway
5.6. Deadhorse Creek, Canada
6. Discussion
6.1. Structural Position and Age of the Sulfide Ores in Pechenga–Varzuga
6.2. Comparative Analysis of Vanadium Mineralization in Kola and Other Regions
6.3. Source of Vanadium
7. Conclusions
- Two types of vanadium mineralization which is rare in nature have been discovered in the Kola region. The mineralization includes both vanadium and vanadium-bearing minerals. Both mineralization types are located in supracrustal rocks of the Paleoproterozoic Pechenga-Varzuga riftogenic belt.
- The first vanadium mineralization type is characteristic of sulfide ores and was studied in the Pyrrhotite Ravine deposit and the Bragino occurrence in the Imandra-Varzuga and Pechenga structures, respectively. These sulfide ores are hosted by island-arc-related basic metavolcanics of the Tominga Group (Imandra-Varzuga) and the South Pechenga Group (Pechenga). The rocks were metamorphosed under upper greenschist/lower amphibolite-facies conditions.
- The first type of vanadium mineralization is localized in shear zones of the transpressional (late collisional) stage of the Paleoproterozoic Lapland–Kola collisional orogeny and was developed 1.90–1.86 Ga ago. This mineralization originated from a metamorphic recrystallization of sulfide ores that were originally enriched in vanadium and are thought to have formed during hydrothermal alteration of marine basalt. An additional input of vanadium is suggested to have been provided by metasomatizing fluids that circulated in the shear zones.
- A unique feature of the first vanadium mineralization type is that it is accompanied by chromium and scandium mineralization. Scandium is believed to have been brought simultaneously with vanadium by metasomatizing fluids introduced in Paleoproterozoic shear zones. Scandium mineralization is not mentioned in sulfide ores that bear vanadium mineralization in other regions of the world.
- The second type of vanadium mineralization is reported from the western part of the Imandra-Varzuga structure. It is localized in xenoliths of the Imandra-Varzuga basic metavolcanics in the 365 Ma Khibiny alkaline massif [47,48] and mineralized rocks are similar to fenites alkaline metasomatic rocks at a contact between island-arc-related basic metavolcanics and dolomites of the Tominga Group [49]. The formation of this vanadium mineralization type is related to a contact metamorphism and alkaline metasomatism produced by the giant Khibiny alkaline intrusion.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Emsley, J. The Elements, 2nd ed.; Clarendon Press: Wotton-under-Edge, UK, 1991; p. 260. ISBN 0198555687. [Google Scholar]
- Voytekhovsky, Y.L.; Neradovsky, Y.N.; Grishin, N.N.; Rakitina, E.Y.; Kasikov, A.G. The Kolvitsa ore deposit (geology, chemical and mineral composition of ores). Proc. Murm. State Tech. Univ. 2014, 17, 271–278. (In Russian) [Google Scholar]
- Cawthorn, R.G. The Bushveld Complex, South Africa. In Intrusions; Charlier, B., Namur, O., Latypov, R., Tegner, C., Eds.; Springer Science+Business Media: Dordrecht, The Netherlands, 2015; pp. 517–587. [Google Scholar]
- Fischer, L.A.; Yuan, Q. Fe-Ti-V-(P) resources in the upper zone of the Bushveld complex, South Africa. Pap. Proc. R. Soc. Tasman. 2016, 150, 15–22. [Google Scholar] [CrossRef]
- Yao, Y.; Viljoen, M.J.; Viljoen, R.P.; Wilson, A.H.; Zhong, H.; Liu, B.G.; Ying, H.L.; Tu, G.Z.; Luo, N. Geological characteristics of PGE-bearing layered intrusions in Southwest Sichuan province, China. In Economic Geology Research Unit. Information Circular; University of the Witwatersrand: Johannesburg, South Africa, 2001; p. 17. ISBN 1-86838-302-4. [Google Scholar]
- Zhou, M.F.; Wang, C.Y.; Pang, K.N.; Shellnutt, G.J.; Ma, Y. Origin of giant Fe-Ti-V oxide deposits in layered gabbroic intrusions, Pan-Xi district, Sichuan Province, SW China. In Mineral Deposit Research: Meeting the Global Challenge, Proceedings of the Eighth Biennial SGA Meeting, Beijing, China, 18–21 August 2005; Mao, J., Bierlein, F.P., Eds.; Springer: Berlin/Heidelberg, Germeny, 2005; pp. 511–513. [Google Scholar]
- Radtke, A.S. Coulsonite, FeV2O4, a spinel-type mineral from Lovelock, Nevada. Am. Mineral. 1962, 47, 1284–1291. [Google Scholar]
- Harris, C.; Hlongwane, W.; Gule, N.; Scheepers, R. Origin of tanzanite and associated gemstone mineralization at Merelani, Tanzania. S. Afr. J. Geol. 2014, 117, 15–30. [Google Scholar] [CrossRef]
- Reznitsky, L.Z.; Skliarov, E.V.; Ushchapovskaia, Z.F. Natalyite Na(V,Cr)Si2O6—A new chromium-vanadium pyroxene from Slyudianka. Proc. Russ. Mineral. Soc. 1985, 114, 630–635. (In Russian) [Google Scholar]
- Reznitsky, L.Z.; Sklyarov, E.V.; Ushchapovskaya, Z.F. Magnesiocoulsonite MgV2O4—A new mineral species in the spinel group. Proc. Russ. Mineral. Soc. 1995, 124, 91–98. (In Russian) [Google Scholar]
- Reznitsky, L.Z.; Sklyarov, E.V.; Armbruster, T.; Ushchapovskaya, Z.F.; Galuskin, E.V.; Polekhovsky, Y.S.; Barash, I.G. The new mineral oxyvanite V3O5 and the oxyvanite-berdesinskiite V2TiO5 isomorphic join in metamorphic rocks of Sludyanka complex (South Baikal region). Proc. Russ. Mineral. Soc. 2009, 138, 70–81. (In Russian) [Google Scholar]
- Kompanchenko, A.A.; Voloshin, A.V.; Bazai, A.V.; Polekhovsky, Yu.S. Evolution of a chromium-vanadium mineralization in massive sulfide ores at the Bragino occurrence in the South Pechenga structural zone (Kola region): An example of the spinel group minerals. Proc. Russ. Mineral. Soc. 2017, 146, 44–59. (In Russian) [Google Scholar]
- Karpov, S.M.; Voloshin, A.V.; Savchenko, Ye.E.; Selivanova, E.A. Vanadium-bearing minerals in ores of the Pyrrhotite Ravine massive sulfide deposit (Khibiny region, Kola peninsula). Proc. Russ. Mineral. Soc. 2013, 142, 83–99. (In Russian) [Google Scholar]
- Karpov, S.M.; Voloshin, A.V.; Kompanchenko, A.A.; Savchenko, Y.E.; Bazai, A.V. Crichtonite group minerals in massive sulfide ores and ore metasomatites of the Proterozoic structures of the Kola region. Proc. Russ. Mineral. Soc. 2016, 145, 39–56. (In Russian) [Google Scholar]
- Long, J.V.P.; Vourelainen, Y.; Kuovo, O. Karelianite, a new vanadium mineral. Am. Mineral. 1963, 48, 33–41. [Google Scholar]
- Sergeeva, N.E.; Eremin, N.I.; Dergachev, A.L. Vanadium mineralization in ore of the Vihanti massive sulfide base-metal deposit, Finland. Dokl. Earth Sci. 2011, 436, 210–212. [Google Scholar] [CrossRef]
- Zakrzewski, M.A.; Burke, E.A.J.; Lustenhouwer, W.J. Vourelainenite, a new spinel, and associated minerals from the Sätra (Doverstorp) pyrite deposit, central Sweden. Can. Mineral. 1982, 20, 281–290. [Google Scholar]
- Höller, W.; Stumpfl, E.F. Cr-V oxides from the Rampura Agucha Pb-Zn-(Ag) deposit, Rajasthan, India. Can. Mineral. 1995, 33, 745–752. [Google Scholar]
- Balagansky, V.V.; Glaznev, V.N.; Osipenko, L.G. Early Proterozoic evolution of the northeastern Baltic Shield: Terrane analysis. Geotectonics 1998, 32, 81–93. [Google Scholar]
- Daly, J.S.; Balagansky, V.V.; Timmerman, M.J.; Whitehouse, M.J. The Lapland-Kola Orogen: Palaeoproterozoic collision and accretion of the northern Fennoscandian lithosphere. In European Lithosphere Dynamics; Gee, D.G., Stephenson, R.A., Eds.; Memoir 32; Geological Society: London, UK, 2006; pp. 579–598. [Google Scholar]
- Glebovitsky, V.A. (Ed.) Early Precambrian of the Baltic Shield; Nauka: Saint-Petersburg, Russia, 2005; p. 711. ISBN 5-02-024950-5. (In Russian) [Google Scholar]
- Melezhik, V.A.; Sturt, B.A. General geology and evolutionary history of the early Proterozoic Polmak-Pasvik-Pechenga-Imandra/Varzuga-Ust’Ponoy Greenstone Belt in the north-eastern Baltic Shield. Earth Sci. Rev. 1994, 36, 205–241. [Google Scholar] [CrossRef]
- Melezhik, V.A.; Prave, A.R.; Hanski, E.J.; Fallick, A.E.; Lepland, A.; Kump, L.R.; Srauss, H. (Eds.) The Palaeoproterozoic of Fennoscandia as Context for the Fennoscandian Arctic Russia—Drilling Early Earth Project. In Reading the Archive of Earth’s Oxygenation; Springer: Heidelberg, Germany, 2013; p. 490. [Google Scholar]
- Mints, M.V.; Dokukina, K.A.; Konilov, A.N.; Philippova, I.B.; Zlobin, V.L.; Babayants, P.S.; Belousova, E.A.; Blokh, V.I.; Bogina, M.M.; Bush, D.A.; et al. East European Craton: Early Precambrian History and 3D Models of Deep Crustal Structure; Geological Society of America Special Paper 510; Geological Society of America: Boulder, CO, USA, 2015; p. 433. [Google Scholar]
- Rundqvist, D.V.; Mitrofanov, F.P. (Eds.) Precambrian Geology of the USSR. In Developments in Precambrian Geology 9; Elsevier: Amsterdam, The Netherlands, 1993; p. 528. ISBN 978-0-444-89380-2. [Google Scholar]
- Zagorodny, V.G.; Predovsky, A.A.; Basalaev, A.A.; Batieva, I.D.; Borisov, A.E.; Vetrin, V.R.; Voloshina, Z.M.; Dokuchaeva, V.S.; Zhangurov, A.A.; Kozlova, N.E.; et al. Imandra-Varzuga Zone of the Karelides (Geology, Geochemistry, History of Development); Nauka: Leningrad, Russia, 1982; p. 280. (In Russian) [Google Scholar]
- Petrov, V.P.; Belyaev, O.A.; Voloshina, Z.M.; Balagansky, V.V.; Glazunkov, A.N.; Pozhilenko, V.I. Endogenic Regimes of Early Precambrian Metamorphism; Nauka: Leningrad, Russia, 1990; p. 184. ISBN 5-02-024410-4. (In Russian) [Google Scholar]
- Skuf’in, P.K.; Bayanova, T.B.; Mitrofanov, F.P. Isotope age of subvolcanic graditoid rocks of the early Proterozoic Panarechka volcanotectonic structure, Kola Peninsula. Dokl. Earth Sci. 2006, 409, 774–778. [Google Scholar] [CrossRef]
- Mitrofanov, F.P.; Balashov, Y.A.; Balagansky, V.V. New geochronological data on lower Precambrian complexes of the Kola Peninsula. In Correlation of Lower Precambrian Formations of the Karelia-Kola Region, USSR, and Finland; Mitrofanov, F.P., Balagansky, V.V., Eds.; Kola Science Centre of the RAS: Apatity, Russia, 1991; pp. 12–16. [Google Scholar]
- Smol’kin, V.F.; Mitrofanov, F.P.; Avedisyan, A.A.; Balashov, Y.A.; Balagansky, V.V.; Borisov, A.E.; Borisova, V.V.; Voloshina, Z.M.; Kozlova, N.E.; Kravtsov, N.A.; et al. Magmatism, Sedimentogenesis and Geodynamics of the Pechenga Paleorift; Kola Science Centre of the Russian Academy of Sciences: Apatity, Russia, 1995; p. 256. (In Russian) [Google Scholar]
- Kozlovsky, E.A. (Ed.) The Superdeep Well in the Kola Peninsula; Springer: Berlin, Germany, 1987; p. 490. [Google Scholar]
- Zagorodny, V.G.; Radchenko, A.T. Tectonics of the Karelides of the Northeastern Baltic Shield; Nauka: Leningrad, Russia, 1988; p. 110. ISBN 5-02-024361-2. (In Russian) [Google Scholar]
- Hanski, E.J.; Huhma, H.; Melezhik, V.A. New isotopic and geochemical data from the Palaeoproterozoic Pechenga Greenstone Belt, NW Russia: Implication for basin development and duration of the volcanism. Precambrian Res. 2014, 245, 51–65. [Google Scholar] [CrossRef]
- Martin, A.P.; Condon, D.J.; Prave, A.R.; Melezhik, V.A.; Lepland, A.; Fallick, A.E. Dating the termination of the Palaeoproterozoic Lomagundi-Jatuli carbon isotopic event in the North Transfennoscandian Greenstone Belt. Precambrian Res. 2013, 224, 160–168. [Google Scholar] [CrossRef]
- Melezhik, V.A.; Huhma, H.; Condon, D.J.; Fallick, A.E.; Whitehouse, M.J. Temporal constraints on the Paleoproterozoic Lomagundi-Jatuli carbon isotopic event. Geology 2007, 35, 655–658. [Google Scholar] [CrossRef]
- Balashov, Y.A. Paleoproterozoic geochronology of the Pechenga-Varzuga structure, Kola Peninsula. Petrology 1996, 4, 3–25. [Google Scholar]
- Akhmedov, A.V.; Voronyaeva, L.V.; Pavlov, V.A.; Krupenik, V.A.; Kuznezov, V.A.; Sveshnikova, K.Yu. Gold potential of the South-Pechenga structural zone (Kola Peninsula): Occurrence types and prospects of discovery of economic gold content. Reg. Geol. Metallog. 2004, 20, 139–151. (In Russian) [Google Scholar]
- Skuf’in, P.K.; Elizarov, D.V.; Zhavkov, V.A. Geological and geochemical pecularities of volcanics of the the South Pechenga structural zone. Proc. Murm. State Tech. Univ. 2009, 12, 416–435. (In Russian) [Google Scholar]
- Skuf’in, P.K.; Bayanova, T.B.; Mitrofanov, F.P.; Apanasevich, E.A.; Levkovich, N.V. The absolute age of granitoids from the Shuoniyarvi pluton in the southern framework of the Pechenga structure, the Kola Peninsula. Dokl. Earth Sci. 2000, 370, 114–117. [Google Scholar]
- Vetrin, V.R.; Turkina, O.M.; Rodionov, N.V. U-Pb age and genesis of granitoids in the southern framing of the Pechenga structure, Baltic Shield. Dokl. Earth Sci. 2008, 219, 806–810. [Google Scholar] [CrossRef]
- Gorstka, V.N. The Contact Zone of the Khibiny Alkaline Massif (Geological and Petrographical Peculiarities, Chemistry and Petrology); Nauka: Leningrad, Russia, 1971; p. 99. (In Russian) [Google Scholar]
- Fedotov, Z.A. The Evolution of Proterozoic Volcanism of the Eastern Pechenga-Varzuga Belt (Petrogeochemical Aspects); Kola Science Centre of the RAS: Apatity, Russia, 1985; p. 120. (In Russian) [Google Scholar]
- Konstantov, S.V.; Sobolev, I.I.; Surovtseva, O.E. The South Contact of the Khibiny Massif. Aykuayvenchorr–Vudyavrchorr–Takhtarvumchorr Areas. Report on Geological Exploration of Iron Sulphides in 1931–1935; Scientific Archive of Geological Institute of the Kola Science Centre of the RAS: Apatity, Russia, 1935; p. 408. (In Russian) [Google Scholar]
- Whitney, D.L.; Evans, B.W. Abbreviations for names of rock-forming minerals. Am. Mineral. 2010, 95, 185–187. [Google Scholar] [CrossRef]
- Olerud, S. Davidite-loveringite in early Proterozoic albite felsites in Finnmark, north Norway. Mineral. Mag. 1988, 52, 400–402. [Google Scholar]
- Canet, C.; Alfonso, P.; Melgarejo, J.-C. V-rich minerals in contact-metamorphosed Silurian sedex deposit in the Poblet area, Southwestern Catalonia, Spain. Can. Mineral. 2003, 41, 561–579. [Google Scholar] [CrossRef]
- Potter, E.G.; Mitchell, R.H. Mineralogy of the Deadhorse Creek volcaniclastic breccia complex, northwestern Ontario, Canada. Contrib. Mineral. Petrol. 2005, 150, 212–229. [Google Scholar] [CrossRef]
- Skuf’in, P.K.; Theart, H.F.J. Geochemical and tectono-magmatic evolution of the volcano-sedimentary rocks of Pechenga and other greenstone fragments within the Kola Greenstone Belt, Russia. Precambrian Res. 2005, 141, 1–48. [Google Scholar] [CrossRef]
- Raade, G.; Balić-Žunić, T.; Stanley, C.J. Byrudite, (Be,□)(V3+,Ti)3O6, a new mineral from the Byrud emerald mine, South Norway. Mineral. Mag. 2015, 79, 261–268. [Google Scholar] [CrossRef]
- Mikhailova, Y.A.; Pakhomovsky, Y.A.; Menshikov, Y.P. Tausonite, baddeleyite and vuorelainenite from hornfels of mt. Kaskasnyunchorr (Khibiny massif). In Proceedings of the 1st Fersman Scientific Session; Geological Institute of the Kola Science Center: Apatity, Russia, 2004; pp. 28–29. (In Russian) [Google Scholar]
- Mikhailova, Y.A.; Konopleva, N.G.; Yakovenchuk, V.N.; Ivanyuk, G.Y.; Men’shikov, Y.P.; Pakhomovsky, Y.A. Corundum-group minerals in rocks of the Khibiny alkaline pluton, Kola Peninsula. Geol. Ore Depos. 2007, 49, 590–598. [Google Scholar] [CrossRef]
- Korchak, Y.A.; Pakhomovsky, Y.A.; Men’shikov, Y.P.; Ivanyuk, G.Y.; Yakovenchuk, V.N. Minerals of crichtonite group in hornfels of Khibiny massif. In Proceedings of the 5th Fersman Scientific Session; Geological Institute of the Kola Science Center: Apatity, Russia, 2008; pp. 264–265. (In Russian) [Google Scholar]
- Karpov, S.M.; Voloshin, A.V.; Telezhkin, A.A. The natalyite-aegirine series in alkaline metasomatites of the Imandra-Varzuga belt, Kola region. Proc. Russ. Mineral. Soc. 2018, 147. in press (In Russian) [Google Scholar]
- Rauhamäki, E.; Mäkelä, T.; Isomäki, O.-P. Geology of the Vihanti mine. In Precambrian ores of Finland. Guide to Excursions 078 A + C, Part 2 (Finland), Proceedings of the 26th International Geological Congress, Paris, France 7 July 1980; Häkli, T.A., Ed.; Geological Survey of Finland: Espoo, Finland, 1980; pp. 14–24. [Google Scholar]
- Rouhunkoski, P. On the geology and geochemistry of the Vihanti zinc ore deposit, Finland. Bull. Comm. Geol. Finl. 1968, 236, 121. [Google Scholar]
- Peltola, E. Origin of Precambrian Copper Sulfides of the Outokumpu Disctrict, Finland. Econ. Geol. 1978, 73, 461–477. [Google Scholar] [CrossRef]
- Peltonen, P. Ophiolites. In Precambrian Geology of Finland—Key to the Evolution of the Fennoscandian Shield; Lehtinen, M., Nurmi, P.A., Rämö, O.T., Eds.; Elsevier: Amsterdam, The Netherlands, 2005; pp. 237–278. [Google Scholar]
- Peltola, E. Geology of the Vuonos ore deposit. In Precambrian ores of Finland. Guide to excursions 078 A + C, Part 2 (Finland), Proceedings of the 26th International Geological Congress, Paris, France, 7 July 1980; Häkli, T.A., Ed.; Geological Survey of Finland: Espoo, Finland, 1980; pp. 33–41. [Google Scholar]
- Wikström, A. Beskrivning till berggrundskartan Katrineholm SV. Sver. Geol. Unders. Ser. Af. 1976, 116, 88. [Google Scholar]
- Allen, R.L.; Lunström, I.; Ripa, M.; Simeonov, A.; Christofferson, H. Facies analysis of a 1.9 Ga, contintental margin, back-arc, felsic caldera province with diverse Zn-Pb-Ag-(Cu-Au) sulfide and Feoxide deposits, Bergslagen region, Sweden. Econ. Geol. 1996, 91, 979–1008. [Google Scholar] [CrossRef]
- Höller, W.; Gandhi, S.M. Origin of tourmaline and oxide minerals from the metamorphosed Rampura Agucha Zn-Pb-(Ag) deposit, Rajasthan, India. Mineral. Petrol. 1997, 60, 99–110. [Google Scholar] [CrossRef]
- Deb, M.; Thorpe, R.L.; Cumming, G.L.; Wagner, P.A. Age, source and stratigraphic implications of Pb isotope data for conformable, sediment-hosted, base metal deposits in the Proterozoic Aravalli-Delhi orogenic belt, northwestern India. Precambrian Res. 1989, 43, 1–22. [Google Scholar] [CrossRef]
- Deb, M. Lithogeochemistry of rocks around Rampura Agucha massive zinc sulfide ore-body, NW India – implications for the evolution of a Proterozoic “Aulakogen”. In Metallogeny Related to Tectonics of the Proterozoic Mobile Belts; Sarkar, S.C., Ed.; Balkhema: Rotterdam, The Netherlands, 1992; pp. 1–35. [Google Scholar]
- Gandhi, S.M.; Paliwal, H.V.; Bhatnagar, S.N. Geology and ore reserve estimates of Rampura Agucha lead zinc deposit, Bhilwara District. J. Geol. Soc. India 1984, 25, 689–705. [Google Scholar]
- Henderson, H.C.; Viola, G.; Nasuti, A. A new tectonic model for the Palaeoproterozoic Kautokeino Greenstone Belt, northern Norway, based on high-resolution airborne magnetic data and field structural analysis and implications for mineral potential. Nor. J. Geol. 2015, 95, 339–363. [Google Scholar] [CrossRef]
- Platt, R.G.; Mitchell, R.H. Transition metal rutiles and titanates from the Deadhorse Creek diatreme complex, northwestern Ontario, Canada. Mineral. Mag. 1996, 60, 403–413. [Google Scholar] [CrossRef]
- Bridgwater, D.; Marker, M.; Mengel, F. The eastern extension of the early Proterozoic Torngat Orogenic Zone across the Atlantic. In Lithoprobe, Eastern Canadian Shield Onshore–Offshore Transect (ECSOOT); Wardle, R.J., Hall, J., Eds.; Report 27; Memorial University of Newfoundland: St. John’s, NL, Canada, 1992; pp. 76–91. [Google Scholar]
- Tuisku, P.; Huhma, H.; Whitehouse, M. Geochronology and geochemistry of the enderbite series in the Lapland Granulite Belt: Generation, tectonic setting, and correlation of the belt. Can. J. Earth Sci. 2012, 49, 1297–1315. [Google Scholar] [CrossRef]
- Cox, S.F. Deformational controls on the dynamic of fluid flow in mesothermal gold system. In Fractures, Fluid Flow and Mineralization; McCaffrey, K.J.W., Lonergan, L., Wilkinson, J.J., Eds.; Geological Society London Special Publication: London, UK, 1999; pp. 123–140. [Google Scholar]
- Ashworth, J.R.; Brown, M. An overview of diverse responses to diverse processes at high crustal temperatures. In High-Temperatuire Metamorphism and Crustal Anatexis; Ashworth, J.R., Brown, M., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1990; pp. 1–18. [Google Scholar]
- Balagansky, V.V.; Mudruk, S.V. On the age of a Paleoproterozoic collision in the southeastern Kola region, Baltic shield. In Geology and Geochronology of Rock-Forming and Ore-Forming Processes in Crystalline Shields; Mitrofanov, F.P., Bayanova, T.B., Eds.; Kola Science Centre of the RAS: Apatity, Russia, 2013; pp. 13–16. (In Russian) [Google Scholar]
- Goldfarb, R.J.; Groves, D.I.; Gardoll, S. Orogenic gold and geological time: A global synthesis. Ore Geol. Rev. 2001, 18, 1–75. [Google Scholar] [CrossRef]
- Scott, K.M.; Radford, N.W. Rutile compositions at the Big Bell Au deposit as a guide for exploration. Geochem. Explor. Environ. Anal. 2007, 7, 353–361. [Google Scholar] [CrossRef]
- Scott, K.M.; Radford, N.W.; Hough, R.M.; Reddy, S.M. Rutile compositions in the Kalgoorlie Goldfields and their implications for exploration. Aust. J. Earth Sci. 2011, 58, 803–812. [Google Scholar] [CrossRef]
- Urban, A.J.; Hoskins, B.F.; Grey, I.E. Characterization of V-Sb-W-bearing rutile from the Hemlo gold deposit, Ontario. Can. Mineral. 1992, 30, 319–326. [Google Scholar]
- Balagansky, V.V.; Belyaev, O.A. Gold-bearing shear zones in the Early Precambrian of the Kola Peninsula: A prognosis and the first results. In Petrography of the XXI Century. Petrology and Ore-Potential of the CIS Regions and the Baltic Shield; Mitrofanov, F.P., Fedotov, Z.A., Eds.; Kola Science Centre of the Russian Academy of Sciences: Apatity, Russia, 2005; Volume 3, pp. 37–38. (In Russian) [Google Scholar]
- Daly, J.S.; Balagansky, V.V.; Timmerman, M.J.; Whitehouse, M.J.; de Jong, K.; Guise, P.; Bogdanova, S.; Gorbatschev, R.; Bridgwater, D. Ion microprobe U-Pb zircon geochronology and isotopic evidence supporting a trans-crustal suture in the Lapland Kola Orogen, northern Fennoscandian Shield. Precambrian Res. 2001, 105, 289–314. [Google Scholar] [CrossRef]
- Molnár, F.; Middleton, A.; Stein, H.; O`Brien, H.; Lahaye, Y.; Huhma, H.; Pakkanen, L.; Johanson, B. Repeated syn- and post-orogenic gold mineralization events between 1.92 and 1.76 Ga along the Kiistala Shear Zone in the Central Lapland Greenstone Belt, northern Finland. Ore Geol. Rev. 2018, 101, 936–959. [Google Scholar] [CrossRef]
- Gill, S.B.; Piercey, S.J.; Layton-Matthews, D. Mineralogy and metal zoning of the Cambrian Zn-Pb-Cu-Ag-Au Lemarchant volcanogenic massive sulfide (VMS) deposit, Newfoundland. Can. Mineral. 2016, 54, 1307–1344. [Google Scholar] [CrossRef]
- Wagner, T.; Monecke, T. Germanium-bearing colusite from the Waterloo volcanic-rock-hosted massive sulfide deposit, Australia: Crystal chemistry and formation of colusite-group minerals. Can. Mineral. 2005, 43, 655–669. [Google Scholar] [CrossRef]
- Altermann, W.; Kazmierczak, J. Archean microfossils: A reappraisal of early life on Earth. Res. Microbiol. 2003, 154, 611–617. [Google Scholar] [CrossRef] [PubMed]
- Ohkouchi, N.; Kuroda, J.; Taira, A. The origin of the Cretaceous black shales in the surface ocean ecosystem and its triggers. Proc. Jpn. Acad. Ser. B 2015, 91, 273–291. [Google Scholar] [CrossRef] [PubMed]
- Yudovich, Y.E.; Ketris, M.P. Trace Elements in Black Shales; Nauka: Ekaterinburg, Russia, 1994; p. 304. [Google Scholar]
- Vine, J.D.; Tourtelot, E.B. Geochemistry of black shale deposits—A summary report. Econ. Geol. 1970, 65, 253–272. [Google Scholar] [CrossRef]
- Butler, I.B.; Nesbitt, R.W. Trace element distribution in the chalcopyrite wall of a black smoker chimney: Insights from laser ablation inductively coupled plasma mass spectrometry (LA-ICP-MS). Earth Planet. Sci. Lett. 1999, 167, 335–345. [Google Scholar] [CrossRef]
- Zaykov, V.V.; Maslennikov, V.V.; Zaykova, E.V.; Herrington, R. Ore-Formation and Ore-Facies Analyses of Massive Sulphide Deposits of the Ural Paleoocean; Institute of Mineralogy, the Ural Branch of Russian Academy of Sciences: Miass, Russia, 2001; p. 315. (In Russian) [Google Scholar]
- Borisenko, L.F. Scandium: Main Features of its Geochemistry, Mineralogy, and Genetic Types of Ore Deposits; Academy of Sciences of the USSR: Moscow, Russia, 1961; p. 132. (in Russian) [Google Scholar]
- Vikentiev, I.V. Precious metal and telluride mineralogy of large volcanic-hosted massive sulfide deposits in the Ural. Mineral. Petrol. 2006, 87, 305–326. [Google Scholar] [CrossRef]
- Vikent’ev, I.V.; Yudovskaya, M.A.; Moloshag, V.P. Speciation of noble metals and conditions of their concentration in massive sulfide ores of the Urals. Geol. Ore Depos. 2006, 48, 77–107. [Google Scholar] [CrossRef]
- Pouret, O.; Dia, A. Vanadium. In Encyclopedia of Geochemistry; White, W.M., Ed.; Springer: Cham, Switzerland, 2016. [Google Scholar]
- Rudnick, R.L.; Gao, S. Composition of the continental crust. In Treatise on Geochemistry; Turekian, K.K., Holland, H.D., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; pp. 1–64. [Google Scholar]



| Mineral/Area | PR | Br | Vih | Otk | Sät | RA | Ctl | DC | Bgj |
|---|---|---|---|---|---|---|---|---|---|
| Oxides | |||||||||
| Berdesinskiite | − | − | + | − | − | − | + | − | − |
| Burydite | − | + | − | − | − | − | − | − | − |
| Chromite | − | + | − | + | − | − | + | − | + |
| Coulsonite | + | + | + | − | − | + | + | − | − |
| Crichtonite | + | + | − | − | − | − | − | + | − |
| Davidite-(Ce) | + | + | − | − | − | − | − | − | + |
| Davidite-(La) | + | + | − | − | − | − | − | − | + |
| Eskolaite | − | − | − | + | − | + | − | − | − |
| Ilmenite | + | + | − | − | − | − | − | + | − |
| Karelianite | + | − | + | + | + | + | − | − | − |
| Kyzylkumite | − | + | + | − | − | − | − | − | − |
| Lindsleyite | − | + | − | − | − | − | − | − | − |
| Loveringite | + | − | − | − | − | − | − | − | + |
| Nolanite | − | + | + | + | − | − | − | − | − |
| Rutile | + | + | + | − | − | + | − | + | − |
| Senaite | − | + | − | − | − | − | − | − | − |
| Shcherbinaite | − | − | + | − | − | − | − | − | − |
| Schreyerite | − | + | + | − | − | + | − | − | − |
| Tivanite | − | + | + | − | − | − | − | − | − |
| Silicates | |||||||||
| Aegirine | − | − | − | − | − | − | − | + | − |
| “Braginoite” | − | + | − | − | − | − | − | − | − |
| Chamosite | + | + | − | − | − | − | − | + | + |
| Clinochlore | + | + | − | + | − | − | − | − | − |
| Diopside | + | − | − | + | − | − | − | − | − |
| Fuchsite | − | − | − | + | − | − | − | − | − |
| Goldmanite | + | − | − | − | − | − | + | − | − |
| Jervisite | − | + | − | − | − | − | − | + | − |
| Mukhinite | + | − | − | − | − | − | − | − | − |
| Muscovite | + | + | − | − | − | − | − | − | − |
| Natalyite | − | − | − | − | − | − | − | + | − |
| Phlogopite | + | + | − | − | − | − | − | − | − |
| Roscoelite | − | + | − | − | − | − | − | − | − |
| Thortveitite | − | + | − | − | − | − | − | + | + |
| Uvarovite | − | − | − | + | − | − | − | − | − |
| “Vanadomukhinite” | + | − | − | − | − | − | − | − | − |
| “Vanadoallanite-(Ce)” | − | + | − | − | − | − | − | − | − |
| “Vanadoallanite-(Nd)” | − | + | − | − | − | − | − | − | − |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kompanchenko, A.A.; Voloshin, A.V.; Balagansky, V.V. Vanadium Mineralization in the Kola Region, Fennoscandian Shield. Minerals 2018, 8, 474. https://doi.org/10.3390/min8110474
Kompanchenko AA, Voloshin AV, Balagansky VV. Vanadium Mineralization in the Kola Region, Fennoscandian Shield. Minerals. 2018; 8(11):474. https://doi.org/10.3390/min8110474
Chicago/Turabian StyleKompanchenko, Alena A., Anatoly V. Voloshin, and Victor V. Balagansky. 2018. "Vanadium Mineralization in the Kola Region, Fennoscandian Shield" Minerals 8, no. 11: 474. https://doi.org/10.3390/min8110474
APA StyleKompanchenko, A. A., Voloshin, A. V., & Balagansky, V. V. (2018). Vanadium Mineralization in the Kola Region, Fennoscandian Shield. Minerals, 8(11), 474. https://doi.org/10.3390/min8110474