Notes on Contributions to the Science of Rare Earth Element Enrichment in Coal and Coal Combustion Byproducts
Abstract
:1. Introduction
2. Rare Earth Elements in Coals
- (1)
- Leaching of volcanic ash with subsequent concentration by organic matter;
- (2)
- Leaching of volcanic ash with subsequent incorporation into secondary minerals; and
- (3)
- Incorporation of volcanic minerals into the peat.
- (1)
- The highest LREE/HREE occurs in the tonstein and in the coal or illitic shale immediately underlying the tonstein;
- (2)
- The other lithotypes, in particular in the basal and uppermost lithotypes, have a lower LREE/HREE, suggesting concentration in secondary minerals.
3. Seredin and Dai Synthesis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- US Department of Energy Coal Production Statistics. Available online: http://www.eia.gov/coal/production/quarterly/ (accessed on 29 January 2016).
- US Department of Energy Rare Earth EDX Database. Available online: https://edx.netl.doe.gov/ree/ (accessed on 29 January 2016).
- Gschneidner, K.A., Jr. Rare Earths: The Fraternal Fifteen; United States Atomic Energy Commission. Division of Technical Information: Washington, DC, USA, 1964; p. 46. [Google Scholar]
- Dent, P.C. Rare Earth Future. Available online: http://www.magneticsmagazine.com/main/channels/materials-channels/rare-earth-future/ (accessed on 25 March 2016).
- Pecht, M.G.; Kaczmarek, R.E.; Song, X.; Hazelwood, D.A.; Kavetsky, R.A.; Anand, D.K. Rare Earth Materials: Insights and Concerns; CALCE EPSC Press: College Park, MD, USA, 2012; p. 194. [Google Scholar]
- U.S. Department of Energy. Critical Materials Strategy; U.S. Department of Energy: Washington, DC, USA, 2011; p. 197. Available online: http://energy.gov/sites/prod/files/DOE_CMS2011_FINAL_Full.pdf (accessed on 25 March 2016).
- European Commission. Report on Critical Raw Materials for the EU. In Report of the Ad Hoc Working Group on Defining Critical Raw Materials; European Commission: Bruxelles, Belgium, 2014; p. 41. Available online: http://ec.europa.eu/DocsRoom/documents/10010/attachments/1/translations/en/renditions/native (accessed on 25 March 2016).
- Mayfield, D.B.; Lewis, A.S. Environmental review of coal ash as a resource for rare earth and strategic elements. In Proceedings of the 2013 World of Coal Ash (WOCA) Conference, Lexington, KY, USA, 22–25 April 2013; pp. 22–25. Available online: http://www.flyash.info/2013/051-mayfield-2013.pdf (accessed on 25 March 2016).
- Seredin, V.V.; Dai, S. Coal deposits as a potential alternative source for lanthanides and yttrium. Int. J. Coal Geol. 2012, 94, 67–93. [Google Scholar] [CrossRef]
- Eskenazy, G. Rare-earth elements in some coal basins of Bulgaria. Geol. Balc. 1978, 8, 81–88. [Google Scholar]
- Eskenazy, G.M. Rare earth elements and yttrium in lithotypes of Bulgarian coals. Org. Geochem. 1987, 11, 83–89. [Google Scholar] [CrossRef]
- Eskenazy, G.M. Zirconium and hafnium in Bulgarian coals. Fuel 1987, 66, 1652–1657. [Google Scholar] [CrossRef]
- Eskenazy, G.M. Rare earth elements in a sampled coal from the Pirin Deposit, Bulgaria. Int. J. Coal Geol. 1987, 7, 301–314. [Google Scholar] [CrossRef]
- Eskenazy, G. Geochemistry of rare earth elements in Bulgarian coals. Annuaire de l’Universite´ de Sofia ‘‘St. Kliment Ohridski’’, Faculte’ de Geologie et Geographie Livre 1-Geologie 1995, 88, 39–65. [Google Scholar]
- Eskenazy, G.M. Aspects of the geochemistry of rare earth elements in coal: An experimental approach. Int. J. Coal Geol. 1999, 38, 285–295. [Google Scholar] [CrossRef]
- Eskenazy, G.M. Sorption of trace elements on xylain: An experimental study. Int. J. Coal Geol. 2015, 150–151, 166–169. [Google Scholar]
- Eskenazy, G.M.; Mincheva, E.I.; Rousseva, D.P. Trace elements in lignite lithotypes from the Elhovo coal basin. Comptes rendus del’Académie bulgare des Sciences 1986, 39, 99–101. [Google Scholar]
- Eskenazy, G.M.; Brinkin, K. Trace elements in the Karlovo coal deposit. Comptes rendus del’Académie bulgare des Sciences 1991, 44, 67–70. [Google Scholar]
- Hower, J.C.; Ruppert, L.F.; Eble, C.F. Lanthanide, Yttrium, and Zirconium anomalies in the Fire Clay coal bed, Eastern Kentucky. Int. J. Coal Geol. 1999, 39, 141–153. [Google Scholar] [CrossRef]
- Mardon, S.M.; Hower, J.C. Impact of coal properties on coal combustion by-product quality: Examples from a Kentucky power plant. Int. J. Coal Geol. 2004, 59, 153–169. [Google Scholar] [CrossRef]
- Crowley, S.S.; Stanton, R.W.; Ryer, T.A. The effects of volcanic ash on the maceral and chemical composition of the C coal bed, Emery Coal Field, Utah. Org. Geochem. 1989, 14, 315–331. [Google Scholar] [CrossRef]
- Crowley, S.S.; Ruppert, L.F.; Belkin, H.F.; Stanton, R.W.; Moore, T.A. Factors affecting the geochemistry of a thick, subbituminous coal bed in the Powder River Basin: Volcanic, detrital, and peat-forming processes. Org. Geochem. 1993, 20, 843–854. [Google Scholar] [CrossRef]
- Seredin, V.V. Rare earth element-bearing coals from the Russian Far East deposits. Int. J. Coal Geol. 1996, 30, 101–129. [Google Scholar] [CrossRef]
- Dai, S.; Ren, D.; Li, S. Discovery of the superlarge gallium ore deposit in Junger, Inner Mongolia, North China. Chin. Sci. Bull. 2006, 51, 2243–2252. [Google Scholar] [CrossRef]
- Dai, S.; Ren, D.; Chou, C.L.; Li, S.; Jiang, Y. Mineralogy and geochemistry of the No. 6 coal (Pennsylvanian) in the Jungar Coalfield, Ordos Basin, China. Int. J. Coal Geol. 2006, 66, 253–270. [Google Scholar] [CrossRef]
- Dai, S.; Li, D.; Chou, C.L.; Zhao, L.; Zhang, Y.; Ren, D.; Ma, Y.; Sun, Y. Mineralogy and geochemistry of boehmite-rich coals: New insights from the Haerwusu Surface Mine, Jungar Coalfield, Inner Mongolia, China. Int. J. Coal Geol. 2008, 74, 185–202. [Google Scholar] [CrossRef]
- Dai, S.; Wang, X.; Chen, W.; Li, D.; Chou, C.L.; Zhou, Y.; Zhu, C.; Li, H.; Zhu, X.; Xing, Y.; et al. A high-pyrite semianthracite of Late Permian age in the Songzao Coalfield, southwestern China: Mineralogical and geochemical relations with underlying mafic tuffs. Int. J. Coal Geol. 2010, 83, 430–445. [Google Scholar] [CrossRef]
- Dai, S.; Zou, J.; Jiang, Y.; Ward, C.R.; Wang, X.; Li, T.; Xue, W.; Liu, S.; Tian, H.; Sun, X.; et al. Mineralogical and geochemical compositions of the Pennsylvanian coal in the Adaohai Mine, Daqingshan Coalfield, Inner Mongolia, China: Modes of occurrence and origin of diaspore, gorceixite, and ammonian illite. Int. J. Coal Geol. 2012, 94, 250–270. [Google Scholar] [CrossRef]
- Dai, S.; Jiang, Y.; Ward, C.R.; Gu, L.; Seredin, V.V.; Liu, H.; Zhou, D.; Wang, X.; Sun, Y.; Zou, J.; et al. Mineralogical and geochemical compositions of the coal in the Guanbanwusu Mine, Inner Mongolia, China: Further evidence for the existence of an Al (Ga and REE) ore deposit in the Jungar Coalfield. Int. J. Coal Geol. 2012, 98, 10–40. [Google Scholar] [CrossRef]
- Birk, D.; White, J.C. Rare earth elements in bituminous coals and underclays of the Sydney Basin, Nova Scotia: Element sites, distribution, mineralogy. Int. J. Coal Geol. 1991, 19, 219–251. [Google Scholar] [CrossRef]
- Kortenski, J.; Bakardjiev, S. Rare earth and radioactive elements in some coals from the Sofia, Svoge and Pemik Basins, Bulgaria. Int. J. Coal Geol. 1993, 22, 237–246. [Google Scholar] [CrossRef]
- Pollock, S.M.; Goodarzi, F.; Riediger, C.L. Mineralogical and elemental variation of coal from Alberta, Canada: An example from the No.2 seam, Genesee Mine. Int. J. Coal Geol. 2000, 43, 259–286. [Google Scholar] [CrossRef]
- Schatzel, S.J.; Stewart, B.W. Rare earth element sources and modification in the Lower Kittanning coal bed, Pennsylvania: Implications for the origin of coal mineral matter and rare earth element exposure in underground mines. Int. J. Coal Geol. 2003, 54, 223–251. [Google Scholar] [CrossRef]
- Aide, M.T.; Aide, C. Rare earth elements: Their importance in understanding soil genesis. ISRN Soil Sci. 2012, 2012. [Google Scholar] [CrossRef]
- Pédrot, M.; Dia, A.; Davranche, M. Dynamic stricture of humic substances: Rare earth elements as a fingerprint. J. Colloid Interface Sci. 2010, 345, 206–213. [Google Scholar] [CrossRef] [PubMed]
- Davranche, M.; Grybos, M.; Gruau, G.; Pédrot, M.; Dia, A.; Marsac, R. Rare earth element patterns: A tool for identifying trace metal sources during wetland soil reduction. Chem. Geol. 2011, 284, 127–137. [Google Scholar] [CrossRef]
- Finkelman, R.B. The origin, occurrence, and distribution of the inorganic constituents in low-rank coals. In Proceedings of the Basic Coal Science Workshop, Houston, TX, USA, 8–9 December 1981; Schobert, H.H., Ed.; Grand Forks Energy Technology Center: Grand Forks, ND, USA, 1981; pp. 70–90. [Google Scholar]
- Seredin, V.; Shpirt, M.; Vassyanovich, A. REE contents and distribution in humic matter of REE-rich coals. In Mineral Deposits: Processes to Processing; Stanley, C.J., Ed.; A.A. Balkema: Rotterdam, The Netherlands, 1999; pp. 267–269. [Google Scholar]
- Dai, S.; Zhou, Y.; Zhang, M.; Wang, X.; Wang, J.; Song, X.; Jiang, Y.; Luo, Y.; Song, Z.; Yang, Z.; et al. A new type of Nb (Ta)–Zr(Hf)–REE–Ga polymetallic deposit in the late Permian coal-bearing strata, eastern Yunnan, southwestern China: Possible economic significance and genetic implications. Int. J. Coal Geol. 2010, 83, 55–63. [Google Scholar] [CrossRef]
- Dai, S.; Zhao, L.; Peng, S.; Chou, C.L.; Wang, X.; Zhang, Y.; Li, D.; Sun, Y. Abundances and distribution of minerals and elements in high-alumina coal fly ash from the Jungar Power Plant, Inner Mongolia, China. Int. J. Coal Geol. 2010, 81, 320–332. [Google Scholar] [CrossRef]
- Seredin, V.V. From coal science to metal production and environmental protection: A new story of success. Int. J. Coal Geol. 2012, 90–91, 1–3. [Google Scholar] [CrossRef]
- Arbuzov, S.I.; Ershov, V.V.; Potseluev, A.A.; Rikhvanov, L.P. Rare Elements in Coals of the Kuznetsk Basin; Publisher House KPK: Kemerovo, Russia, 2000; p. 248. (In Russian) [Google Scholar]
- Arbuzov, S.I.; Mezhibor, A.M.; Spears, D.A.; Ilenok, S.S.; Shaldybin, M.V.; Belaya, E.V. Nature of tonsteins in the Azeisk deposit of the Irkutsk Coal Basin (Siberia, Russia). Int. J. Coal Geol. 2016, 153, 99–111. [Google Scholar] [CrossRef]
- Seredin, V.V. The first data on abnormal Niobium content in Russian coals. Dokl. Earth Sci. 1994, 335, 634–636. [Google Scholar]
- Seredin, V.V.; Finkelman, R.B. Metalliferous coals: A review of the main genetic and geochemical types. Int. J. Coal Geol. 2008, 76, 253–289. [Google Scholar] [CrossRef]
- Spears, D.A. The origin of tonsteins, an overview, and links with seatearths, fireclays and fragmental clay rocks. Int. J. Coal Geol. 2012, 94, 22–31. [Google Scholar] [CrossRef]
- Zhou, Y.; Bohor, B.F.; Ren, Y. Trace element geochemistry of altered volcanic ash layers (tonsteins) in late Permian coal-bearing formations of eastern Yunnan and western Guizhou Provinces, China. Int. J. Coal Geol. 2000, 44, 305–324. [Google Scholar] [CrossRef]
- Arbuzov, S.I.; Mashen’kin, V.S. Oxidation zone of coalfields as a promising source of noble and rare metals: A case of coalfields in Central Asia. In Problems and Outlook of Development of Mineral Resources and Fuel-Energetic Enterprises of Siberia; Tomsk Polytechnic University: Tomsk, Russia, 2007; pp. 26–31. (In Russian) [Google Scholar]
- Seredin, V.V. Metalliferous coals: Formation conditions and outlooks for development. In Coal Resources of Russia; Geoinformmark: Moscow, Russia, 2004; Volume 6, pp. 452–519. (In Russian) [Google Scholar]
- Seredin, V.V. A new method for primary evaluation of the outlook for rare earth element ores. Geol. Ore Depos. 2010, 52, 428–433. [Google Scholar] [CrossRef]
- Blissett, R.S.; Smalley, N.; Rowson, N.A. An investigation into six coal fly ashes from the United Kingdom and Poland to evaluate rare earth element content. Fuel 2014, 119, 236–239. [Google Scholar] [CrossRef]
- Saikia, B.K.; Ward, C.R.; Oliveira, M.L.S.; Hower, J.C.; Leao, F.D.; Johnston, M.N.; O’Bryan, A.; Sharma, A.; Baruah, B.P.; Silva, L.F.O. Geochemistry and nano-mineralogy of feed coals, mine overburden, and coal-derived fly ashes from Assam (North-east India): A multi-faceted analytical approach. Int. J. Coal Geol. 2015, 137, 19–37. [Google Scholar] [CrossRef]
- Chen, J.; Chen, P.; Yao, D.; Liu, Z.; Wu, Y.; Liu, W.; Hu, Y. Mineralogy and geochemistry of Late Permian coals from the Donglin Coal Mine in the Nantong coalfield in Chongqing, southwestern China. Int. J. Coal Geol. 2015, 149, 24–40. [Google Scholar] [CrossRef]
- Franus, W.; Wiatros-Motyka, M.M.; Wdowin, M. Coal fly ash as a resource for rare earth elements. Environ. Sci. Pollut. Res. 2015, 22, 9464–9474. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Ward, C.R.; French, D.; Graham, I.T. Major and Trace Element Geochemistry of Coals and Intra-Seam Claystones from the Songzao Coalfield, SW China. Minerals 2015, 5, 870–893. [Google Scholar] [CrossRef]
- Hower, J.C.; Eble, C.F.; O’Keefe, J.M.K.; Dai, S.; Wang, P.; Xie, P.; Liu, J.; Ward, C.R.; French, D. Petrology, Palynology, and Geochemistry of Gray Hawk Coal (Early Pennsylvanian, Langsettian) in Eastern Kentucky, USA. Minerals 2015, 5, 894–918. [Google Scholar] [CrossRef]
- Zhang, W.; Rezaee, M.; Bhagavatula, A.; Li, Y.; Groppo, J.; Honaker, R. A review of the occurrence and promising recovery methods of rare earth elements from coal and coal by-products. Int. J. Coal Prep. Util. 2015, 35, 295–330. [Google Scholar] [CrossRef]
- Li, B.; Zhuang, X.; Li, J.; Querol, X.; Font, O.; Moreno, N. Geological controls on mineralogy and geochemistry of the Late Permian coals in the Liulong Mine of the Liuzhi Coalfield, Guizhou Province, Southwest China. Int. J. Coal Geol. 2016, 154–155, 1–15. [Google Scholar] [CrossRef]
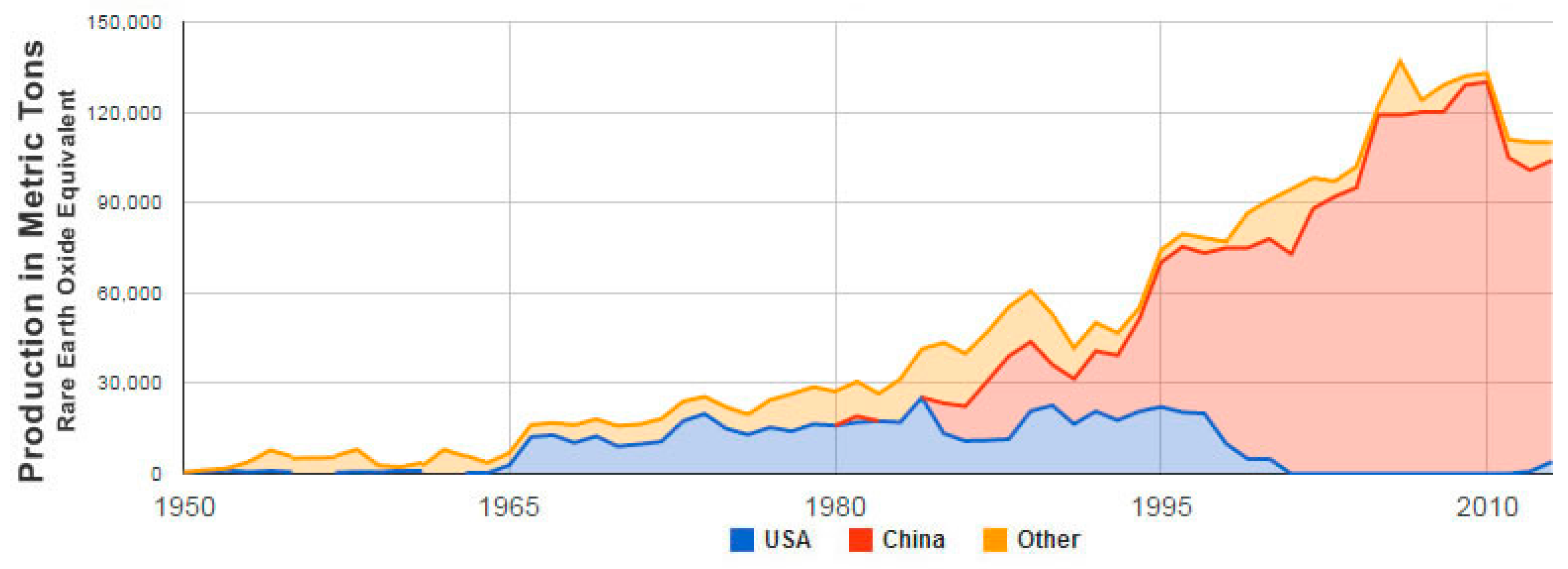
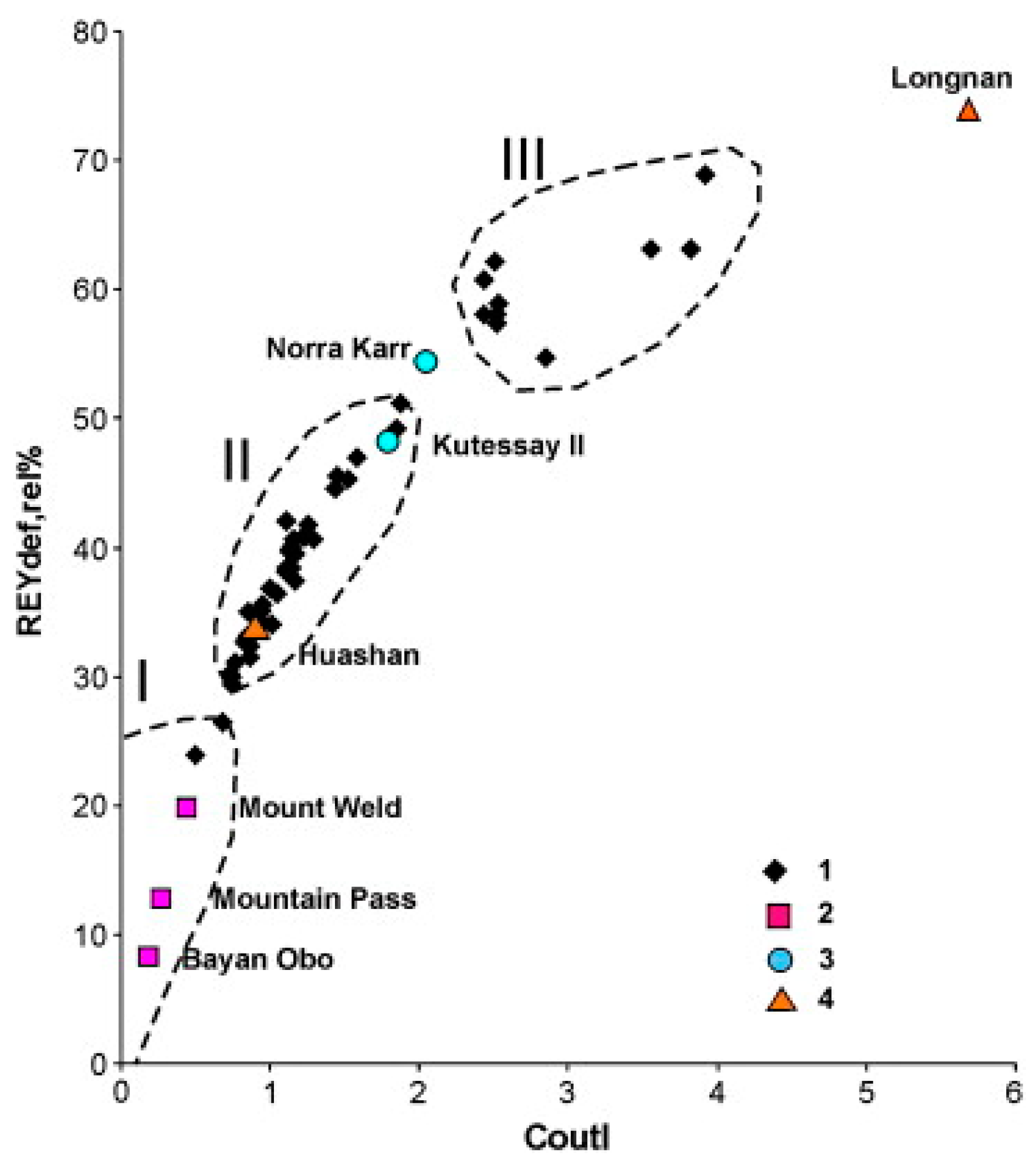
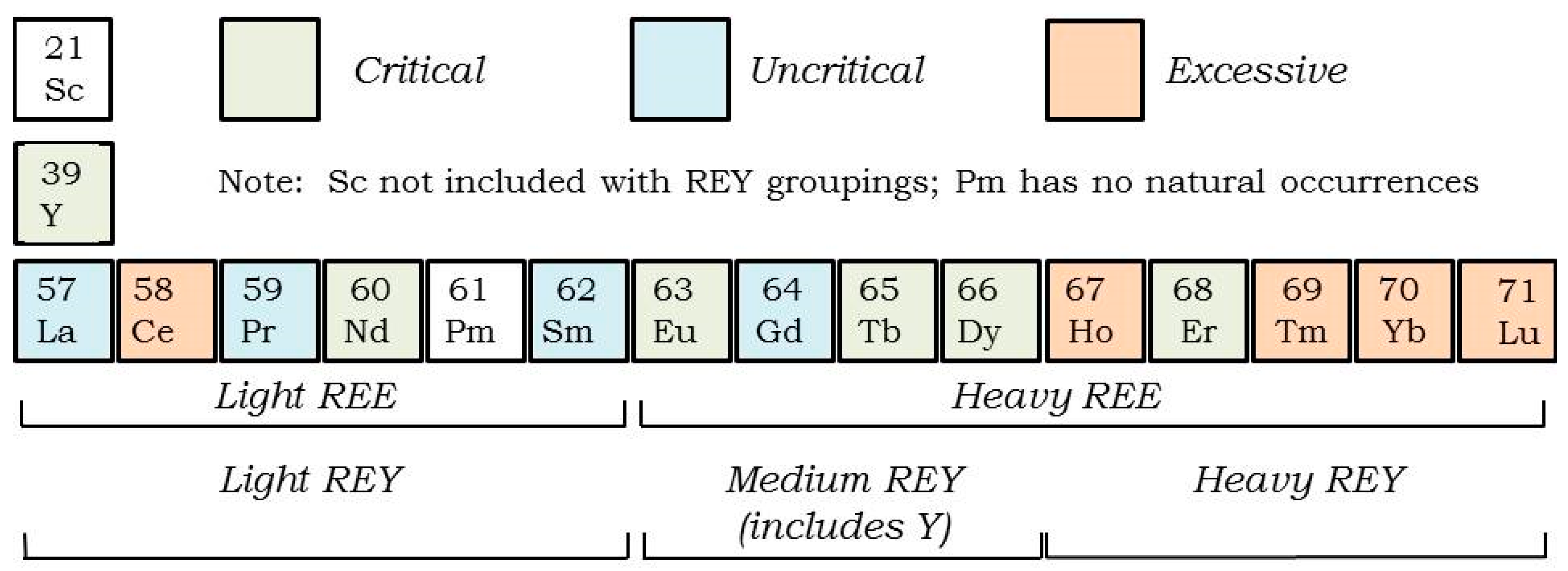
| Type | REO Content in Ash, % | Associated Elements | Typical Example |
|---|---|---|---|
| Terrigenous | 0.1–0.4 | Al, Ga, Ba, Sr, | Jungar, China [25,26] |
| Tuffaceous | 0.1–0.5 | Zr, Hf, Nb, Ta, Ga | Dean, USA [20] |
| Infiltrational | 0.1–1.2 | U, Mo, Se, Re | Aduunchulun, Mongolia [48] |
| Hydrothermal | 0.1–1.5 | As, Sb, Hg, Ag, Au, etc. | Rettikhovka, Russia [49] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hower, J.C.; Granite, E.J.; Mayfield, D.B.; Lewis, A.S.; Finkelman, R.B. Notes on Contributions to the Science of Rare Earth Element Enrichment in Coal and Coal Combustion Byproducts. Minerals 2016, 6, 32. https://doi.org/10.3390/min6020032
Hower JC, Granite EJ, Mayfield DB, Lewis AS, Finkelman RB. Notes on Contributions to the Science of Rare Earth Element Enrichment in Coal and Coal Combustion Byproducts. Minerals. 2016; 6(2):32. https://doi.org/10.3390/min6020032
Chicago/Turabian StyleHower, James C., Evan J. Granite, David B. Mayfield, Ari S. Lewis, and Robert B. Finkelman. 2016. "Notes on Contributions to the Science of Rare Earth Element Enrichment in Coal and Coal Combustion Byproducts" Minerals 6, no. 2: 32. https://doi.org/10.3390/min6020032
APA StyleHower, J. C., Granite, E. J., Mayfield, D. B., Lewis, A. S., & Finkelman, R. B. (2016). Notes on Contributions to the Science of Rare Earth Element Enrichment in Coal and Coal Combustion Byproducts. Minerals, 6(2), 32. https://doi.org/10.3390/min6020032





