Bending Improvement of CO2-Activated Materials through Crosslinking of Oligomers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Specimen Fabrication of CAM
2.2. Testing Procedures of CAM
3. Results and Discussion
3.1. Carbonation Properties of CAM
3.2. Further Improvement of the CAM Bending
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lippiatt, N.; Ling, T.-C.; Pan, S.-Y. Towards carbon-neutral construction materials: Carbonation of cement-based materials and the future perspective. J. Build. Eng. 2020, 28, 101062. [Google Scholar] [CrossRef]
- Zhang, D.; Ghouleh, Z.; Shao, Y. Review on carbonation curing of cement-based materials. J. CO2 Util. 2017, 21, 119–131. [Google Scholar] [CrossRef]
- Wang, D.; Xiong, C.; Li, W.; Chang, J. Growth of Calcium Carbonate Induced by Accelerated Carbonation of Tricalcium Silicate. ACS Sustain. Chem. Eng. 2020, 8, 14718–14731. [Google Scholar] [CrossRef]
- Ashraf, W.; Olek, J. Carbonation activated binders from pure calcium silicates: Reaction kinetics and performance controlling factors. Cem. Concr. Compos. 2018, 93, 85–98. [Google Scholar] [CrossRef]
- Zhao, S.; Liu, Z.; Mu, Y.; Wang, F.; He, Y. Effect of chitosan on the carbonation behavior of γ-C2S. Cem. Concr. Compos. 2020, 111, 103637. [Google Scholar] [CrossRef]
- Khan, R.I.; Ashraf, W.; Olek, J. Amino acids as performance-controlling additives in carbonation-activated cementitious materials. Cem. Concr. Res. 2021, 147, 106501. [Google Scholar] [CrossRef]
- Liu, Z.; Shao, C.; Jin, B.; Zhang, Z.; Zhao, Y.; Xu, X.; Tang, R. Crosslinking ionic oligomers as conformable precursors to calcium carbonate. Nature 2019, 574, 394–398. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, R.; Liu, Z.; Zhang, Z. A novel carbonate binder from waste hydrated cement paste for utilization of CO2. J. CO2 Util. 2019, 32, 276–280. [Google Scholar] [CrossRef]
- Li, L.; Cao, M.; Yin, H. Comparative roles between aragonite and calcite calcium carbonate whiskers in the hydration and strength of cement paste. Cem. Concr. Compos. 2019, 104, 103350. [Google Scholar] [CrossRef]
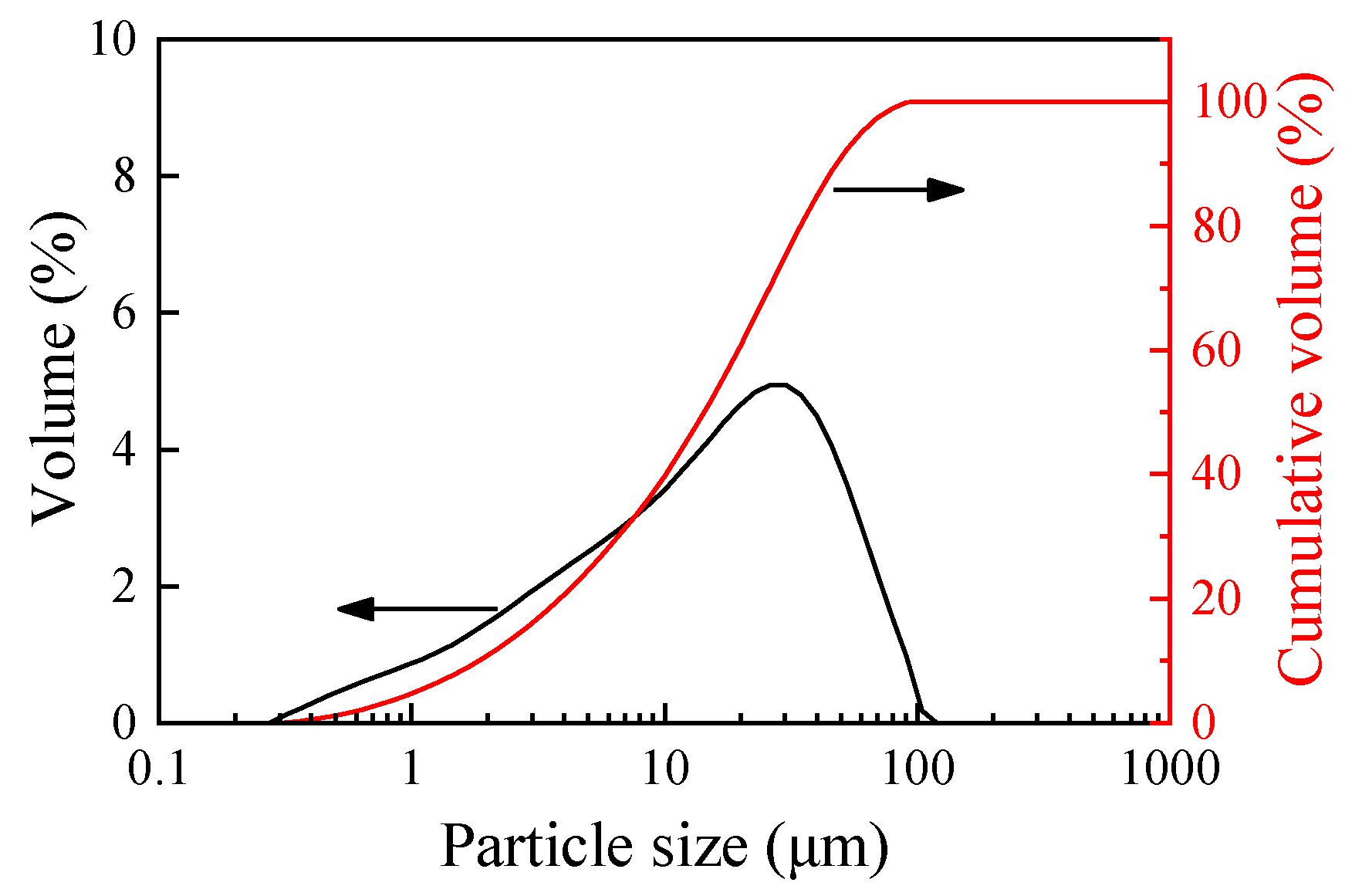
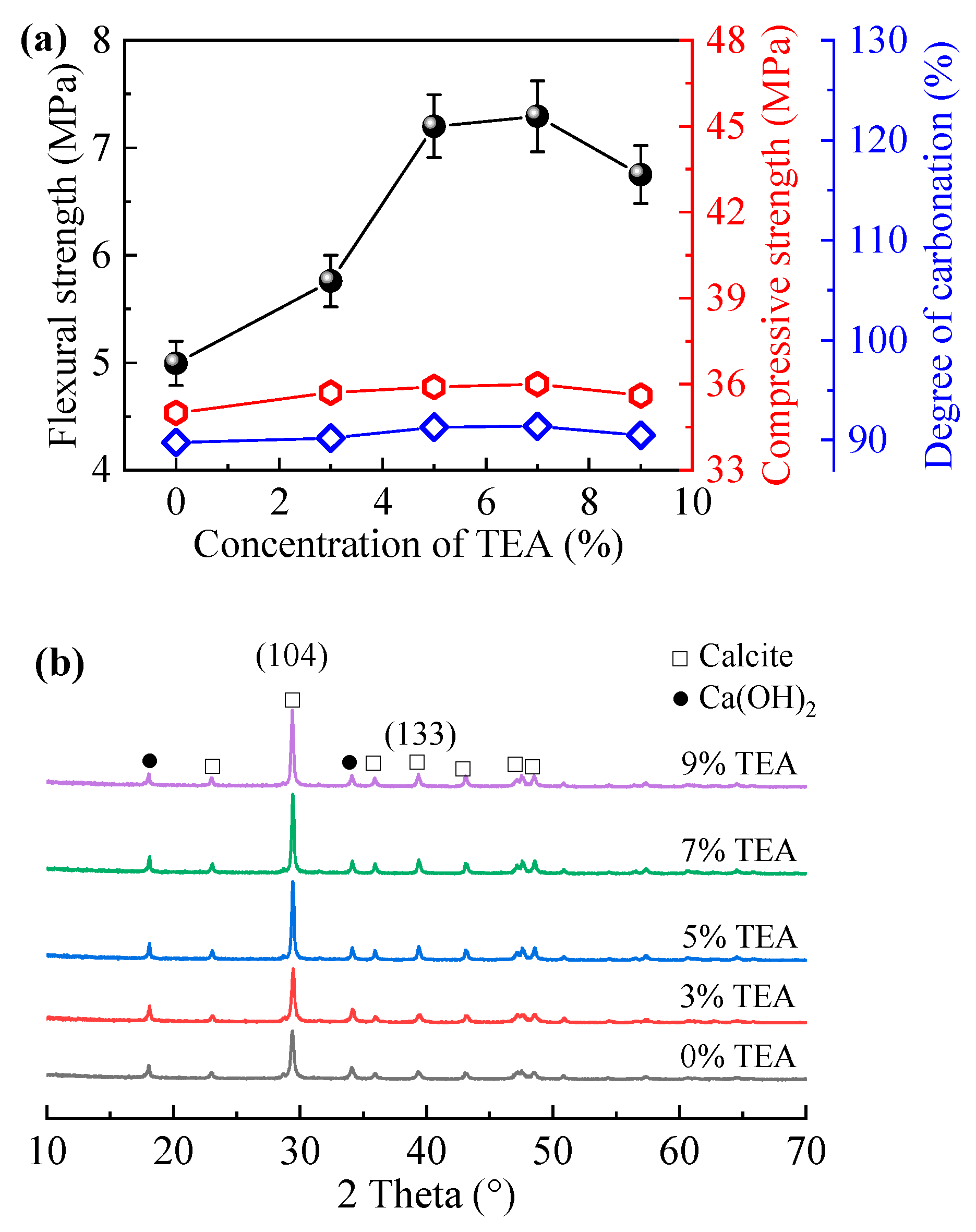

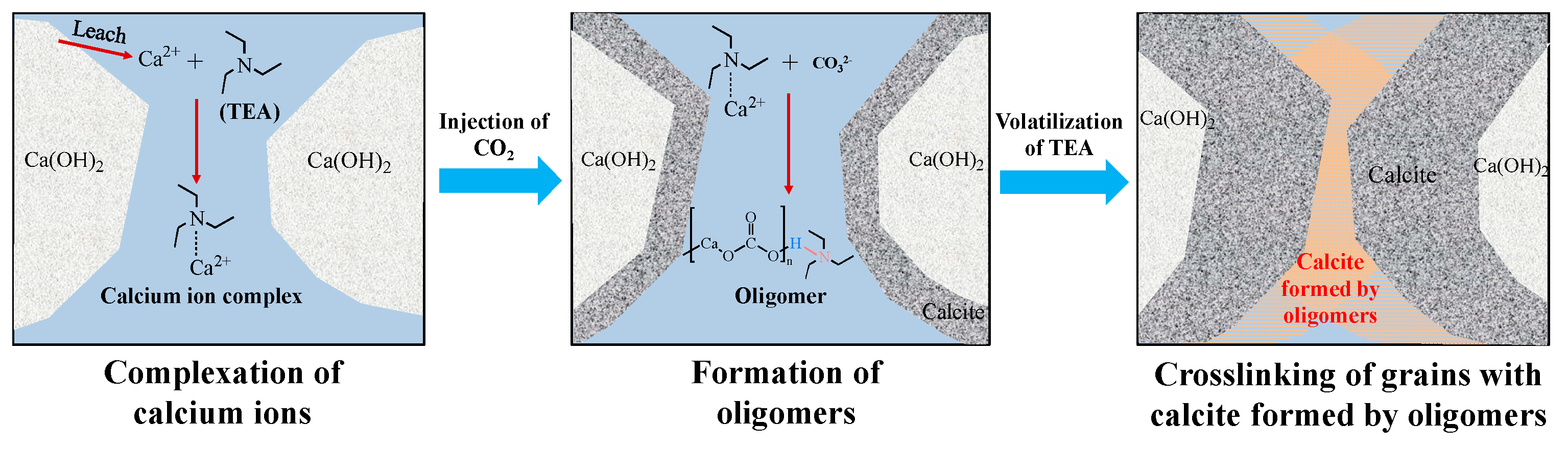
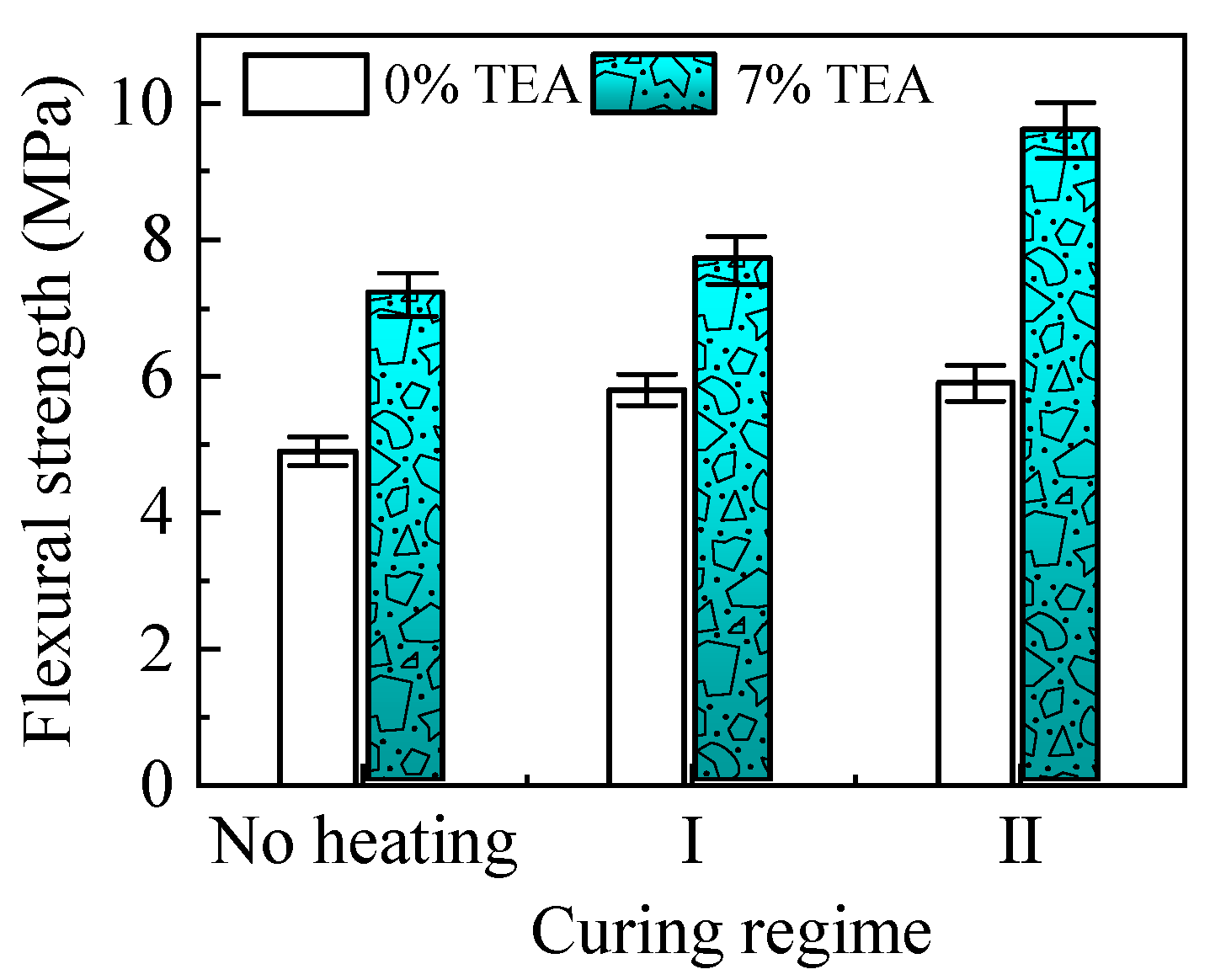
| Concentration of TEA, % | 0 | 3 | 5 | 7 | 9 |
| Ratios of Diffraction Peak Intensity | 5.2 | 5.5 | 6.1 | 6.2 | 5.7 |
| Grain Size, nm | 27.1 | 33.8 | 38.7 | 40.6 | 35.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Wang, Q.; Zhang, Z.; Lei, P. Bending Improvement of CO2-Activated Materials through Crosslinking of Oligomers. Minerals 2023, 13, 352. https://doi.org/10.3390/min13030352
Zhang Y, Wang Q, Zhang Z, Lei P. Bending Improvement of CO2-Activated Materials through Crosslinking of Oligomers. Minerals. 2023; 13(3):352. https://doi.org/10.3390/min13030352
Chicago/Turabian StyleZhang, Yunhua, Qing Wang, Zhipeng Zhang, and Pengxiang Lei. 2023. "Bending Improvement of CO2-Activated Materials through Crosslinking of Oligomers" Minerals 13, no. 3: 352. https://doi.org/10.3390/min13030352
APA StyleZhang, Y., Wang, Q., Zhang, Z., & Lei, P. (2023). Bending Improvement of CO2-Activated Materials through Crosslinking of Oligomers. Minerals, 13(3), 352. https://doi.org/10.3390/min13030352








