Thermal Stability and Resistance to Biodegradation of Humic Acid Adsorbed on Clay Minerals
Abstract
:1. Introduction
2. Materials and Methods
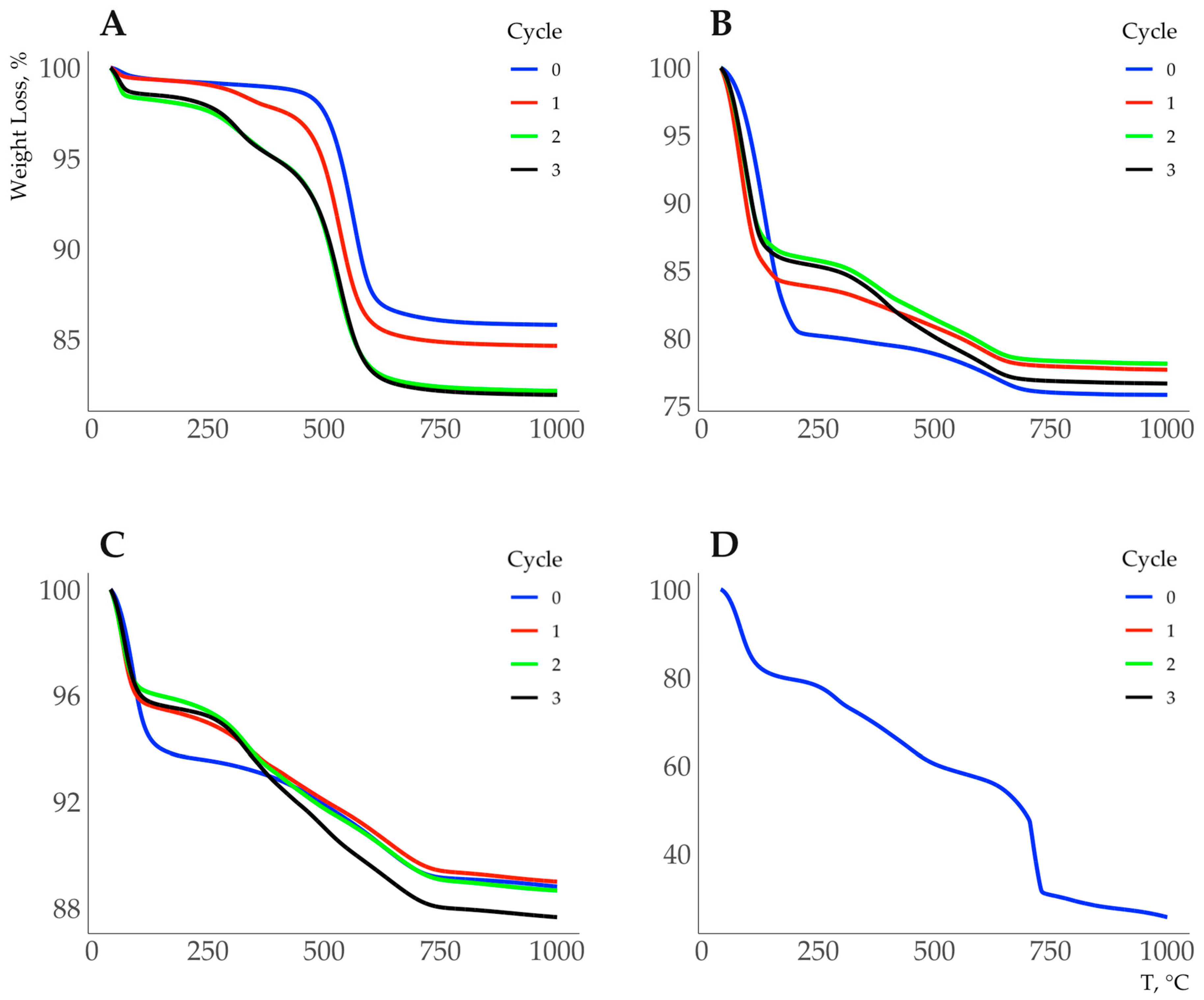
3. Results
Sorption of HA on Kaolinite, Muscovite and Bentonite
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Eusterhues, K.; Rumpel, C.; Kleber, M.; Kögel-Knabner, I. Stabilisation of Soil Organic Matter by Interactions with Minerals as Revealed by Mineral Dissolution and Oxidative Degradation. Org. Geochem. 2003, 34, 1591–1600. [Google Scholar] [CrossRef]
- Saidy, A.R.; Smernik, R.J.; Baldock, J.A.; Kaiser, K.; Sanderman, J. Microbial Degradation of Organic Carbon Sorbed to Phyllosilicate Clays with and without Hydrous Iron Oxide Coating. Eur. J. Soil Sci. 2015, 66, 83–94. [Google Scholar] [CrossRef]
- Lützow, M.V.; Kögel-Knabner, I.; Ekschmitt, K.; Matzner, E.; Guggenberger, G.; Marschner, B.; Flessa, H. Stabilization of Organic Matter in Temperate Soils: Mechanisms and Their Relevance under Different Soil Conditions—A Review. Eur. J. Soil Sci. 2006, 57, 426–445. [Google Scholar] [CrossRef]
- Chen, H.; Koopal, L.K.; Xiong, J.; Avena, M.; Tan, W. Mechanisms of Soil Humic Acid Adsorption onto Montmorillonite and Kaolinite. J. Colloid Interface Sci. 2017, 504, 457–467. [Google Scholar] [CrossRef]
- El-sayed, M.E.A.; Khalaf, M.M.R.; Gibson, D.; Rice, J.A. Assessment of Clay Mineral Selectivity for Adsorption of Aliphatic/Aromatic Humic Acid Fraction. Chem. Geol. 2019, 511, 21–27. [Google Scholar] [CrossRef]
- Chen, H.; Li, Q.; Wang, M.; Ji, D.; Tan, W. XPS and Two-Dimensional FTIR Correlation Analysis on the Binding Characteristics of Humic Acid onto Kaolinite Surface. Sci. Total Environ. 2020, 724, 138154. [Google Scholar] [CrossRef] [PubMed]
- Bougdah, N.; Messikh, N.; Bousba, S.; Magri, P.; Djazi, F.; Zaghdoudi, R. Adsorption of Humic Acid from Aqueous Solution on Different Modified Bentonites. Chem. Eng. Trans. 2017, 60, 223–228. [Google Scholar] [CrossRef]
- Al-Essa, K. Adsorption of Humic Acid onto Jordanian Kaolinite Clay: Effects of Humic Acid Concentration, PH, and Temperature. Sci. J. Chem. 2018, 6, 1. [Google Scholar] [CrossRef]
- Gouré-Doubi, H.; Martias, C.; Smith, A.; Villandier, N.; Sol, V.; Gloaguen, V.; Feuillade, G. Adsorption of Fulvic and Humic like Acids on Surfaces of Clays: Relation with SUVA Index and Acidity. Appl. Clay Sci. 2018, 154, 83–90. [Google Scholar] [CrossRef]
- Zhang, L.; Luo, L.; Zhang, S. Integrated Investigations on the Adsorption Mechanisms of Fulvic and Humic Acids on Three Clay Minerals. Colloids Surf. A Physicochem. Eng. Asp. 2012, 406, 84–90. [Google Scholar] [CrossRef]
- Elfarissi, F.; Pefferkorn, E. Kaolinite/Humic Acid Interaction in the Presence of Aluminium Ion. Colloids Surf. A Physicochem. Eng.Asp. 2000, 168, 1–12. [Google Scholar] [CrossRef]
- Kloster, N.; Avena, M. Interaction of Humic Acids with Soil Minerals: Adsorption and Surface Aggregation Induced by Ca2+. Environ. Chem. 2015, 12, 731–738. [Google Scholar] [CrossRef]
- Zhu, X.; He, J.; Su, S.; Zhang, X.; Wang, F. Concept Model of the Formation Process of Humic Acid-Kaolin Complexes Deduced by Trichloroethylene Sorption Experiments and Various Characterizations. Chemosphere 2016, 151, 116–123. [Google Scholar] [CrossRef]
- Chotzen, R.A.; Polubesova, T.; Chefetz, B.; Mishael, Y.G. Adsorption of Soil-Derived Humic Acid by Seven Clay Minerals: A Systematic Study. Clays Clay Miner. 2016, 64, 628–638. [Google Scholar] [CrossRef]
- Lehmann, J.; Kleber, M. The Contentious Nature of Soil Organic Matter. Nature 2015, 528, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Kleber, M.; Sollins, P.; Sutton, R. A Conceptual Model of Organo-Mineral Interactions in Soils: Self-Assembly of Organic Molecular Fragments into Zonal Structures on Mineral Surfaces. Biogeochemistry 2007, 85, 9–24. [Google Scholar] [CrossRef]
- Barré, P.; Fernandez-Ugalde, O.; Virto, I.; Velde, B.; Chenu, C. Impact of Phyllosilicate Mineralogy on Organic Carbon Stabilization in Soils: Incomplete Knowledge and Exciting Prospects. Geoderma 2014, 235–236, 382–395. [Google Scholar] [CrossRef]
- Dell’abate, M.T.; Benedetti, A.; Brookes, P.C. Hyphenated Techniques of Thermal Analysis for Characterisation of Soil Humic Substances. J. Sep. Sci. 2003, 26, 433–440. [Google Scholar] [CrossRef]
- Mao, J.; Fang, X.; Schmidt-Rohr, K.; Carmo, A.M.; Hundal, L.S.; Thompson, M.L. Molecular-Scale Heterogeneity of Humic Acid in Particle-Size Fractions of Two Iowa Soils. Geoderma 2007, 140, 17–29. [Google Scholar] [CrossRef]
- Feng, W.; Plante, A.F.; Aufdenkampe, A.K.; Six, J. Soil Organic Matter Stability in Organo-Mineral Complexes as a Function of Increasing C Loading. Soil Biol. Biochem. 2014, 69, 398–405. [Google Scholar] [CrossRef]
- Gregorich, E.G.; Gillespie, A.W.; Beare, M.H.; Curtin, D.; Sanei, H.; Yanni, S.F. Evaluating Biodegradability of Soil Organic Matter by Its Thermal Stability and Chemical Composition. Soil Biol. Biochem. 2015, 91, 182–191. [Google Scholar] [CrossRef]
- Plante, A.F.; Fernández, J.M.; Leifeld, J. Application of Thermal Analysis Techniques in Soil Science. Geoderma 2009, 153, 1–10. [Google Scholar] [CrossRef]
- Fernández, J.M.; Peltre, C.; Craine, J.M.; Plante, A.F. Improved Characterization of Soil Organic Matter by Thermal Analysis Using CO2/H2O Evolved Gas Analysis. Environ. Sci. Technol. 2012, 46, 8921–8927. [Google Scholar] [CrossRef]
- Peltre, C.; Fernández, J.M.; Craine, J.M.; Plante, A.F. Relationships between Biological and Thermal Indices of Soil Organic Matter Stability Differ with Soil Organic Carbon Level. Soil Sci. Soc. Am. J. 2013, 77, 2020–2028. [Google Scholar] [CrossRef]
- Plante, A.F.; Fernández, J.M.; Haddix, M.L.; Steinweg, J.M.; Conant, R.T. Biological, Chemical and Thermal Indices of Soil Organic Matter Stability in Four Grassland Soils. Soil Biol. Biochem. 2011, 43, 1051–1058. [Google Scholar] [CrossRef]
- Karavanova, E.I. Dissolved Organic Matter: Fractional Composition and Sorbability by the Soil Solid Phase (Review of Literature). Eurasian Soil Sci. 2013, 46, 833–844. [Google Scholar] [CrossRef]
- Chechetko, E.S.; Tolpeshta, I.I.; Zavgorodnyaya, Y.A. Application of Dodecyltrimethylammonium-Modified Bentonite for Water Purification from Oil and Water-Soluble Oil Components. Mosc. Univ. Soil Sci. Bull. 2017, 72, 119–124. [Google Scholar] [CrossRef]
- Semenov, A.A.; Demin, V.V.; Biryukov, M.V.; Zavgorodnyaya, Y.A. Lokalizaciya Bioprotektornogo Dejstviya Guminovyh Veshchestv v Pochvah. Estestv. Tekhnicheskie Nauk. 2008, 4, 84–93. [Google Scholar]
- Barreto, M.S.C.; Ramlogan, M.; Oliveira, D.M.S.; Verburg, E.E.J.; Elzinga, E.J.; Rouff, A.A.; Jemo, M.; Alleoni, L.R.F. Thermal Stability of Soil Organic Carbon after Long-Term Manure Application across Land Uses and Tillage Systems in an Oxisol. Catena 2021, 200, 105164. [Google Scholar] [CrossRef]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis, 3rd ed.; Springer: New York, NY, USA, 2016; ISBN 978-3-319-24277-4. [Google Scholar]
- Francioso, O.; Montecchio, D. Diffuse Reflectance Fourier Transform Spectroscopy and Thermal Analysis Applied to Humic Substances Espectroscopía Por Transformada de Fourier de Reflectancia Difusa y Análisis Térmico Aplicados a Substancias Húmicas. Óptica pura y aplicada 2007, 40, 207–213, ISSN-e 2171-8814. [Google Scholar]
- Francioso, O.; Montecchio, D.; Gioacchini, P.; Ciavatta, C. Thermal Analysis (TG-DTA) and Isotopic Characterization (13C-15N) of Humic Acids from Different Origins. Appl. Geochem. 2005, 20, 537–544. [Google Scholar] [CrossRef]
- Ghosh, S.; Wang, Z.Y.; Kang, S.; Bhowmik, P.C.; Xing, B.S. Sorption and Fractionation of a Peat Derived Humic Acid by Kaolinite, Montmorillonite, and Goethite. Pedosphere 2009, 19, 21–30. [Google Scholar] [CrossRef]
- Dimirkou, A.; Ioannou, A.; Doula, M. Preparation, Characterization and Sorption Properties for Phosphates of Hematite, Bentonite and Bentonite-Hematite Systems. Adv. Colloid Interface Sci. 2002, 97, 37–61. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.-S. Measurment of Point of Zero Charge of Bentonite by Solubilization Technique and Its Dependence of Surface Potential on PH. Environ. Eng. Res. 2003, 8, 222–227. [Google Scholar] [CrossRef]
- Hernández-Hernández, K.A.; Solache-Ríos, M.; Díaz-Nava, M.C. Removal of Brilliant Blue FCF from Aqueous Solutions Using an Unmodified and Iron-Modified Bentonite and the Thermodynamic Parameters of the Process. Water Air Soil Pollut. 2013, 224, 1562. [Google Scholar] [CrossRef]
- Zachara, J.M.; Smith, S.C. Edge Complexation Reactions of Cadmium on Specimen and Soil-Derived Smectite. Soil Sci. Soc. Am. J. 1994, 58, 762–769. [Google Scholar] [CrossRef]
- Liu, X.; Lu, X.; Sprik, M.; Cheng, J.; Meijer, E.J.; Wang, R. Acidity of Edge Surface Sites of Montmorillonite and Kaolinite. Geochim. Cosmochim. Acta 2013, 117, 180–190. [Google Scholar] [CrossRef]
- Kolchanova, K.; Tolpeshta, I.; Izosimova, Y. Adsorption of Fulvic Acid and Water Extractable Soil Organic Matter on Kaolinite and Muscovite. Agronomy 2021, 11, 2420. [Google Scholar] [CrossRef]
- Kaiser, K.; Guggenberger, G. Mineral Surfaces and Soil Organic Matter. Eur. J. Soil Sci. 2003, 54, 219–236. [Google Scholar] [CrossRef]








| Sample | Pore Volume, cm3/g | N, % * | C, % * | C/N * | S, m2/g | C, g/m2 | |
|---|---|---|---|---|---|---|---|
| Bentonite | 0.084 | 0.18 | 0.09 | 0.5 | 88.7 | 1.02 × 10−5 | |
| Kaolinite | 0.107 | 0.12 | 0.09 | 0.8 | 18.7 | 4.81× 10−5 | |
| Muscovite | 0.175 | 0.18 | 0.05 | 0.3 | 98.5 | 0.51 × 10−5 | |
| HA | ND | 0.91 | 40.35 | 44.3 | ND | ND | |
| Bentonite + HA | (1) | ND | 0.17 | 3.18 | 18.7 | ND | 0.35 × 10−3 |
| (2) | ND | 0.17 | 3.01 | 17.7 | |||
| Kaolinite + HA | (1) | ND | 0.08 | 2.11 | 26.4 | ND | 1.03 × 10−3 |
| (2) | ND | 0.06 | 1.75 | 29.2 | |||
| Muscovite + HA | (1) | ND | 0.10 | 1.76 | 17.6 | ND | 0.18 × 10−3 |
| (2) | ND | 0.12 | 1.71 | 14.3 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Danilin, I.; Tolpeshta, I.; Izosimova, Y.; Pozdnyakov, L.; Stepanov, A.; Salimgareeva, O. Thermal Stability and Resistance to Biodegradation of Humic Acid Adsorbed on Clay Minerals. Minerals 2023, 13, 1310. https://doi.org/10.3390/min13101310
Danilin I, Tolpeshta I, Izosimova Y, Pozdnyakov L, Stepanov A, Salimgareeva O. Thermal Stability and Resistance to Biodegradation of Humic Acid Adsorbed on Clay Minerals. Minerals. 2023; 13(10):1310. https://doi.org/10.3390/min13101310
Chicago/Turabian StyleDanilin, Igor, Inna Tolpeshta, Yulia Izosimova, Lev Pozdnyakov, Andrey Stepanov, and Olga Salimgareeva. 2023. "Thermal Stability and Resistance to Biodegradation of Humic Acid Adsorbed on Clay Minerals" Minerals 13, no. 10: 1310. https://doi.org/10.3390/min13101310
APA StyleDanilin, I., Tolpeshta, I., Izosimova, Y., Pozdnyakov, L., Stepanov, A., & Salimgareeva, O. (2023). Thermal Stability and Resistance to Biodegradation of Humic Acid Adsorbed on Clay Minerals. Minerals, 13(10), 1310. https://doi.org/10.3390/min13101310





