Study on the Activity of Metakaolin Produced by Traditional Rotary Kiln in China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Geopolymer Gel Preparation (20 °C, 50% Relative Humidity)
2.3. Samples Preparation
2.4. Chemical Detection
2.5. Immersion Test
3. Results
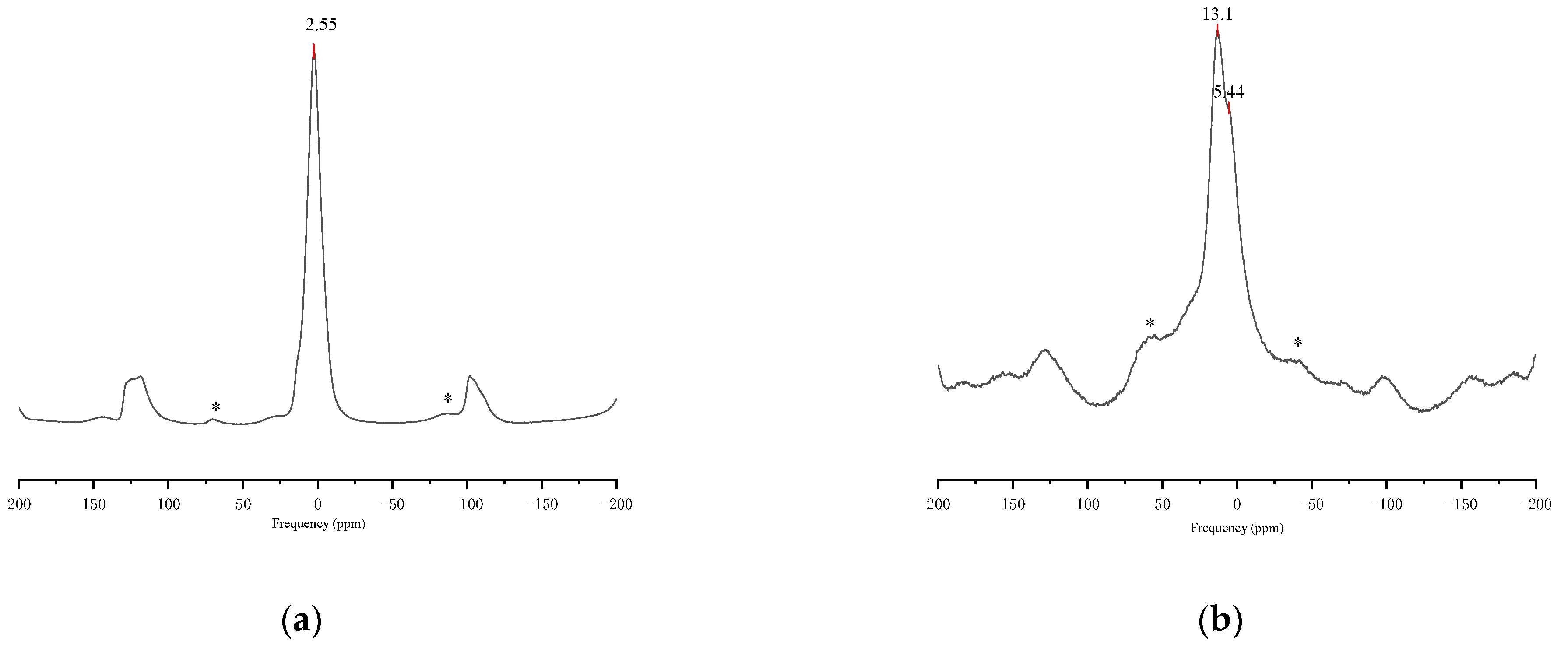
3.1. Coal-Based Metakaolin Activity
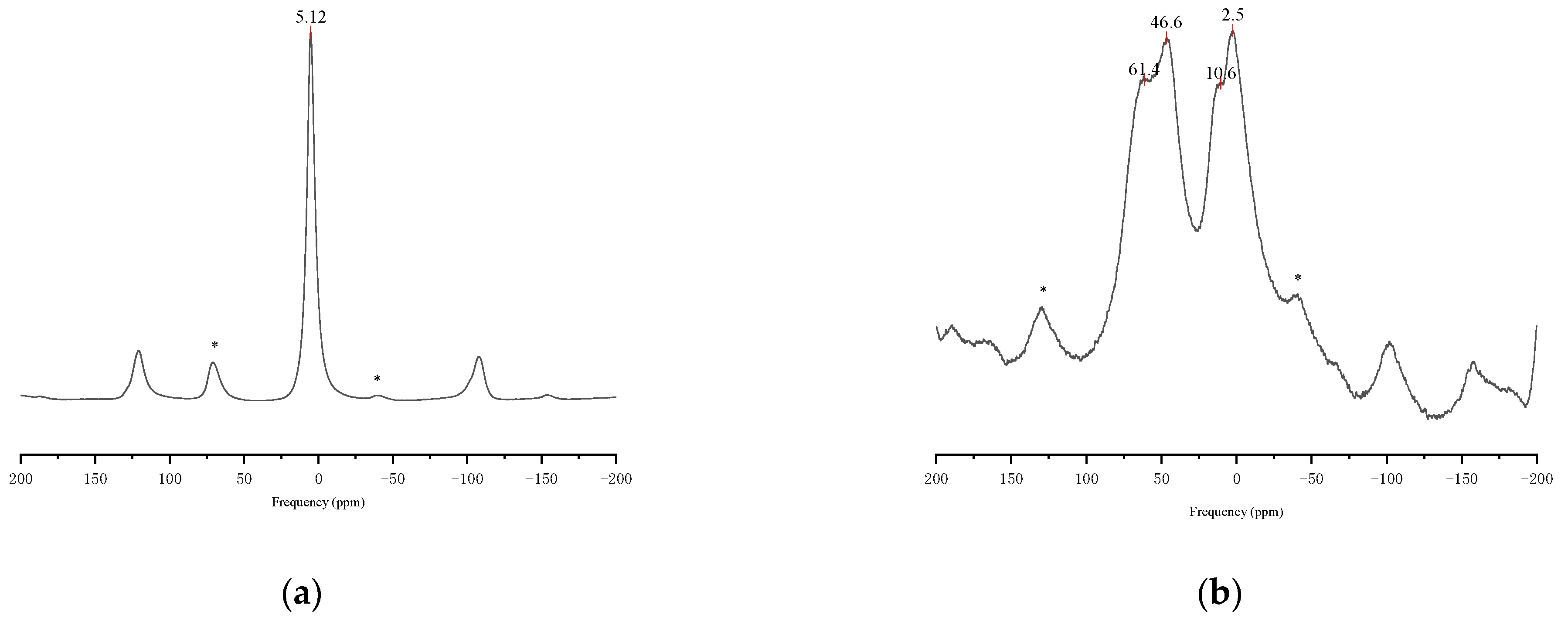
3.2. Ordinary Metakaolin Activity
4. Conclusions
- (1)
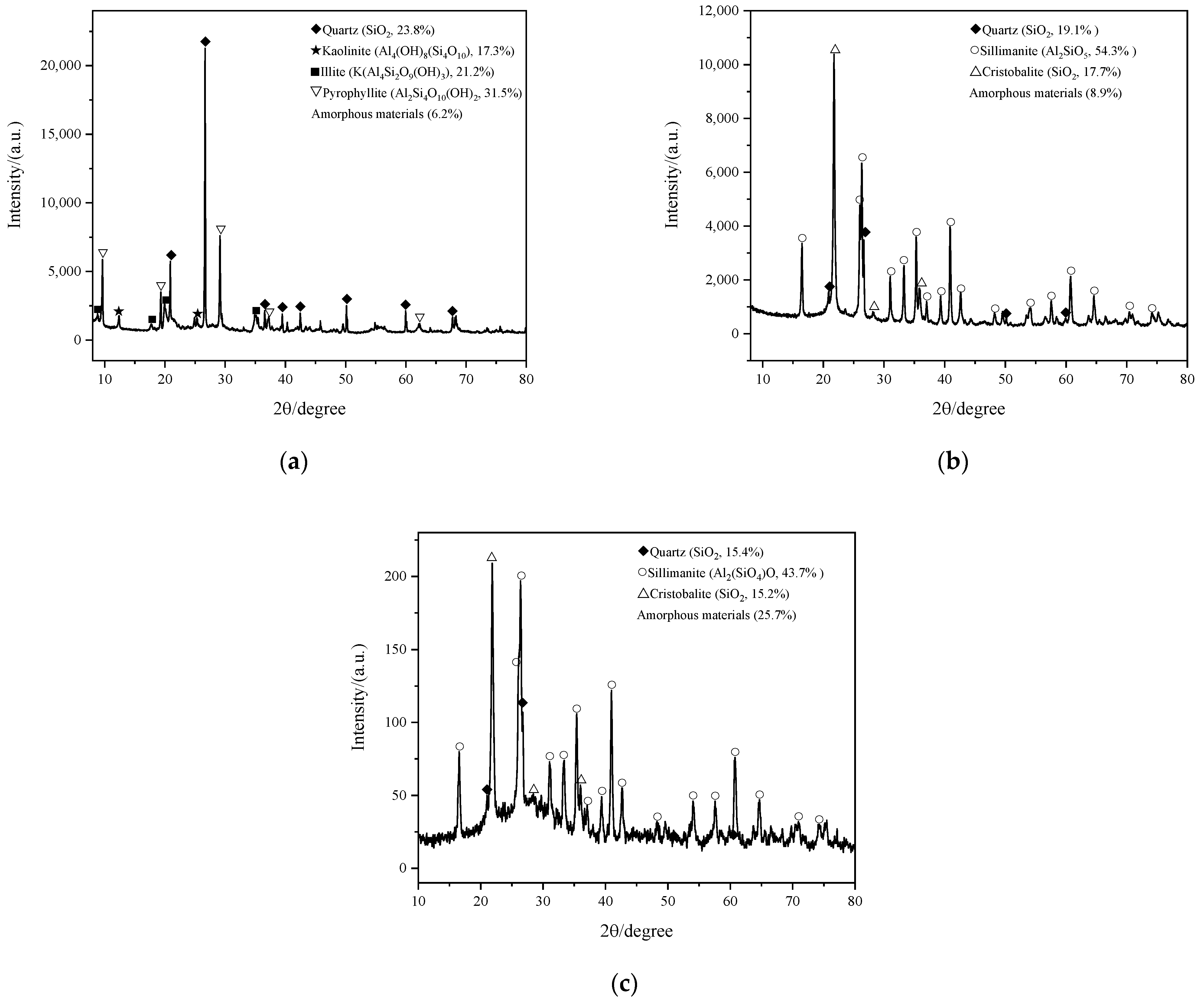
- The kaolinite accounted for a large proportion of coal-based kaolin, and the product of coal-based kaolin calcined in a rotary kiln was mainly amorphous aluminum silicate. Due to the over calcination of kaolin, aluminum in amorphous aluminum silicate mainly existed in the form of Al (VI). Some quartz and corundum were also formed in this calcination process, and they did not participate in the reaction of metakaolin and alkali activators to form geopolymer. When the mixture of coal-based metakaolin and alkali activator hardens after reacting, these crystals and geopolymer are connected by intermolecular forces, and upon encountering water, the crystals and geopolymer separated as intermolecular forces broke.
- (2)
- Low kaolinite purity and over calcination were two negative effects on the activity of ordinary metakaolin. The pyrophyllite and illite in ordinary metakaolin were difficult to produce amorphous substances that could be used as geopolymer raw materials in a traditional rotary kiln. Their calcination led to excessive sillimanite and cristobalite, which were difficult to react in an alkali solution at room temperature; over calcination further reduced the content of amorphous substances in the reaction products. The geopolymer content in the hardened mixture after the reaction of ordinary metakaolin and alkali activator was very small. When encountering water, the whole mixture was quickly destroyed with the break of intermolecular force.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bauer, A.; Velde, B.; Berger, G. Kaolinite transformation in high molar KOH solutions. Appl. Geochem. 1998, 13, 619–629. [Google Scholar] [CrossRef]
- Davidovits, J.; Jean-Jacques, L. Procede D’agglomeration De Substances Minerales Comprimables Sous Forme De Poudre, Particules Ou Fibres. French Patent FR 2, 324, 427, 15 April 1974. [Google Scholar]
- Kakali, G.; Perraki, T.; Tsivilis, S.; Badogiannis, E. Thermal treatment of kaolin: The effect of mineralogy on the pozzolanic activity. Appl. Clay Sci. 2001, 20, 73–80. [Google Scholar] [CrossRef]
- Rocha, J.; Klinowski, J. Si-29 and Al-27 Magic-Angle-Spinning Nmr-Studies of the Thermal Transformation of Kaolinite. Phys. Chem. Miner 1990, 17, 179–186. [Google Scholar] [CrossRef]
- De Rossi, A.; Simao, L.; Ribeiro, M.J.; Novais, R.M.; Labrincha, J.A.; Hotza, D.; Moreira, R.F.P.M. In-situ synthesis of zeolites by geopolymerization of biomass fly ash and metakaolin. Mater. Lett. 2019, 236, 644–648. [Google Scholar] [CrossRef]
- Khale, D.; Chaudhary, R. Mechanism of geopolymerization and factors influencing its development: A review. J. Mater. Sci. 2007, 42, 729–746. [Google Scholar] [CrossRef]
- Ayeni, O.; Onwualu, A.P.; Boakye, E. Characterization and mechanical performance of metakaolin-based geopolymer for sustainable building applications. Constr. Build. Mater. 2021, 272, 121938. [Google Scholar] [CrossRef]
- Luukkonen, T.; Sarkkinen, M.; Kemppainen, K.; Ramo, J.; Lassi, U. Metakaolin geopolymer characterization and application for ammonium removal from model solutions and landfill leachate. Appl. Clay Sci. 2016, 119, 266–276. [Google Scholar] [CrossRef]
- Davidovits, R.; Davidovits, J.; Pélegris, C. Standardized Method in Testing Commercial Metakaolins for Geopolymer Formulations; Technical Paper #26-MK-Testing 2019; Geopolymer Institute Library, 16 rue Galilée, 02100 Saint-Quentin, France. Available online: www.geopolymer.org (accessed on 8 February 2022).
- Guan, X.; Chen, J.X.; Zhu, M.Y.; Gao, J. Performance of microwave-activated coal gangue powder as auxiliary cementitious material. J. Mater. Res. Technol. 2021, 14, 2799–2811. [Google Scholar] [CrossRef]
- Yang, Y.; Zeng, Q.L. Impact-slip experiments and systematic study of coal gangue “category” recognition technology part II: Improving effect of the proposed parallel voting system method on coal gangue “category” recognition accuracy based on impact-slip experiments. Powder Technol. 2022, 395, 893–904. [Google Scholar] [CrossRef]
- Duxson, P.; Provis, J.L.; Lukey, G.C.; Mallicoat, S.W.; Kriven, W.M.; van Deventer, J.S.J. Understanding the relationship between geopolymer composition, microstructure and mechanical properties. Colloid Surface A 2005, 269, 47–58. [Google Scholar] [CrossRef]
- Castillo, H.; Collado, H.; Droguett, T.; Sánchez, S.; Vesely, M.; Garrido, P.; Palma, S. Factors Affecting the Compressive Strength of Geopolymers: A Review. Minerals 2021, 11, 1317. [Google Scholar] [CrossRef]
- Nawaz, M.; Heitor, A.; Sivakumar, M. Geopolymers in construction-Recent developments. Constr. Build. Mater. 2020, 260, 120472. [Google Scholar] [CrossRef]
- Yugeswaran, S.; Vijay, M.; Suresh, K.; Ananthapadmanabhan, P.V.; Karoly, Z.; Szépvölgyi, J. Synthesis of mullite from sillimanite dissociation through transferred arc plasma torch. Int. J. Miner. Process. 2015, 99, 54–60. [Google Scholar] [CrossRef]
- Lippmaa, E.; Samoson, A.; Mgi, M. High-Resolution 27Al NMR of Aluminosilicates. J. Am. Chem. Soc. 1986, 108, 1730–1735. [Google Scholar] [CrossRef]
- Müller, D.; Gessner, W.; Behrens, H.; Scheler, G. Determination of the aluminum coordination in aluminum-oxygen compounds by solid-state high-resolution 27AI NMR. Chem. Phys. Lett. 1981, 79, 59–62. [Google Scholar] [CrossRef]
- Mackenzie, K.; Smith, M.E. Multinuclear Solid-State Nuclear Magnetic Resonance of Inorganic Materials; Pergamon: Amsterdam, The Netherlands, 2002. [Google Scholar]
- Davidovits, J. Geopolymer Chemistry and Applications, 5th ed.; Institut Géopolymère: Saint-Quentin, France, 2020. [Google Scholar]
- Liu, C.L.; Ma, S.H.; Ding, J.; Lu, Y.; Zheng, S.L.; Zhang, Y. Kinetics of decomposition of mullite and corundum in coal fly ash under highly alkaline condition. Trans. Nonferr. Met. Soc. 2019, 29, 868–875. [Google Scholar] [CrossRef]
- Duxson, P.; Lukey, G.C.; Separovic, F.; Van Deventer, J.S.J. Effect of Alkali Cations on Aluminum Incorporation in Geopolymeric. Gels 2005, 44, 832–839. [Google Scholar] [CrossRef]
- Bich, C.; Ambroise, J.; Pera, J. Influence of degree of dehydroxylation on the pozzolanic activity of metakaolin. Appl. Clay Sci. 2009, 44, 194–200. [Google Scholar] [CrossRef]








| Coal-Based Kaolin | Ordinary Kaolin | ||||||
|---|---|---|---|---|---|---|---|
| Element | Conc | Compound | Conc | Element | Conc | Compound | Conc |
| Si | 49.268% | SiO2 | 49.088% | Si | 57.432% | SiO2 | 52.815% |
| Al | 37.751% | Al2O3 | 44.144% | Al | 38.724% | Al2O3 | 45.355% |
| Fe | 6.500% | Fe2O3 | 3.136% | Ti | 1.308% | TiO2 | 0.704% |
| Ti | 3.581% | TiO2 | 2.181% | Fe | 1.060% | Fe2O3 | 0.473% |
| Ca | 0.990% | CaO | 0.523% | K | 0.665% | K2O | 0.265% |
| K | 0.931% | K2O | 0.424% | Ca | 0.444% | CaO | 0.201% |
| Cl | 0.243% | Cl | 0.094% | Zr | 0.106% | ZrO2 | 0.044% |
| Zr | 0.212% | SO3 | 0.089% | Cl | 0.039% | SO3 | 0.034% |
| V | 0.114% | ZrO2 | 0.084% | S | 0.035% | V2O5 | 0.026% |
| S | 0.093% | V2O5 | 0.077% | V | 0.030% | Cl | 0.014% |
| Sr | 0.077% | P2O5 | 0.053% | Cr | 0.029% | CuO | 0.014% |
| Cr | 0.069% | Cr2O3 | 0.031% | Zn | 0.023% | ZnO | 0.009% |
| P | 0.067% | SrO | 0.028% | Sr | 0.021% | Cr2O3 | 0.008% |
| Ga | 0.024% | Ga2O3 | 0.010% | Pb | 0.019% | Ga2O3 | 0.008% |
| Nb | 0.019% | MnO | 0.008% | Ga | 0.018% | MnO | 0.007% |
| Th | 0.018% | NbO | 0.007% | Mn | 0.017% | SrO | 0.007% |
| Mn | 0.017% | Y2O3 | 0.006% | Cu | 0.013% | PbO | 0.007% |
| Y | 0.014% | ThO2 | 0.006% | Nb | 0.010% | NbO | 0.006% |
| Ni | 0.008% | NiO | 0.006% | Y | 0.007% | Y2O3 | 0.003% |
| Cu | 0.004% | CuO | 0.005% | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, R.; Fang, Z.; Jin, M.; Shang, Y. Study on the Activity of Metakaolin Produced by Traditional Rotary Kiln in China. Minerals 2022, 12, 365. https://doi.org/10.3390/min12030365
Cao R, Fang Z, Jin M, Shang Y. Study on the Activity of Metakaolin Produced by Traditional Rotary Kiln in China. Minerals. 2022; 12(3):365. https://doi.org/10.3390/min12030365
Chicago/Turabian StyleCao, Rongchuan, Zheng Fang, Man Jin, and Yu Shang. 2022. "Study on the Activity of Metakaolin Produced by Traditional Rotary Kiln in China" Minerals 12, no. 3: 365. https://doi.org/10.3390/min12030365
APA StyleCao, R., Fang, Z., Jin, M., & Shang, Y. (2022). Study on the Activity of Metakaolin Produced by Traditional Rotary Kiln in China. Minerals, 12(3), 365. https://doi.org/10.3390/min12030365








