Simultaneous Sequestration of Co2+ and Mn2+ by Fungal Manganese Oxide through Asbolane Formation
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
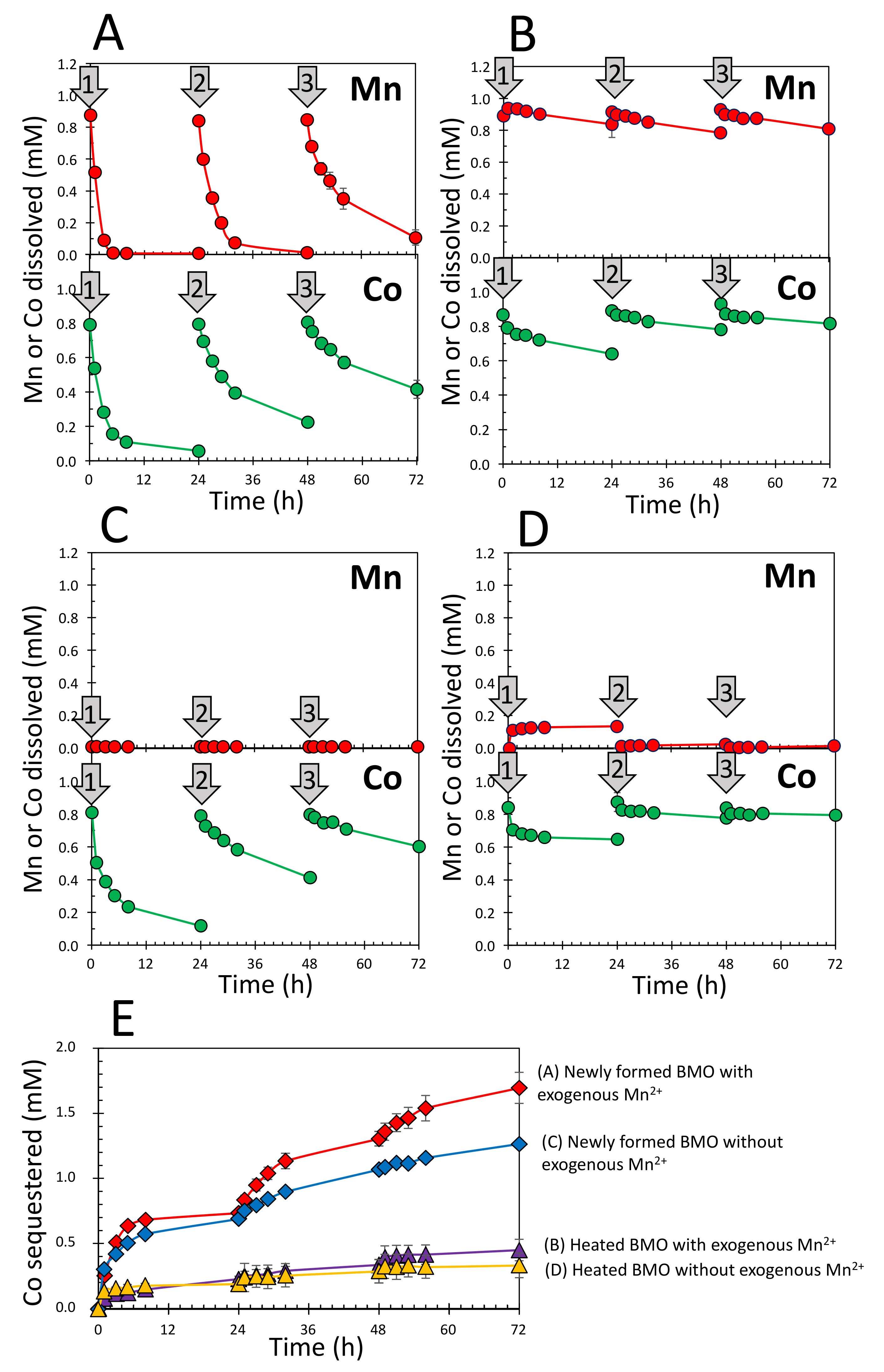
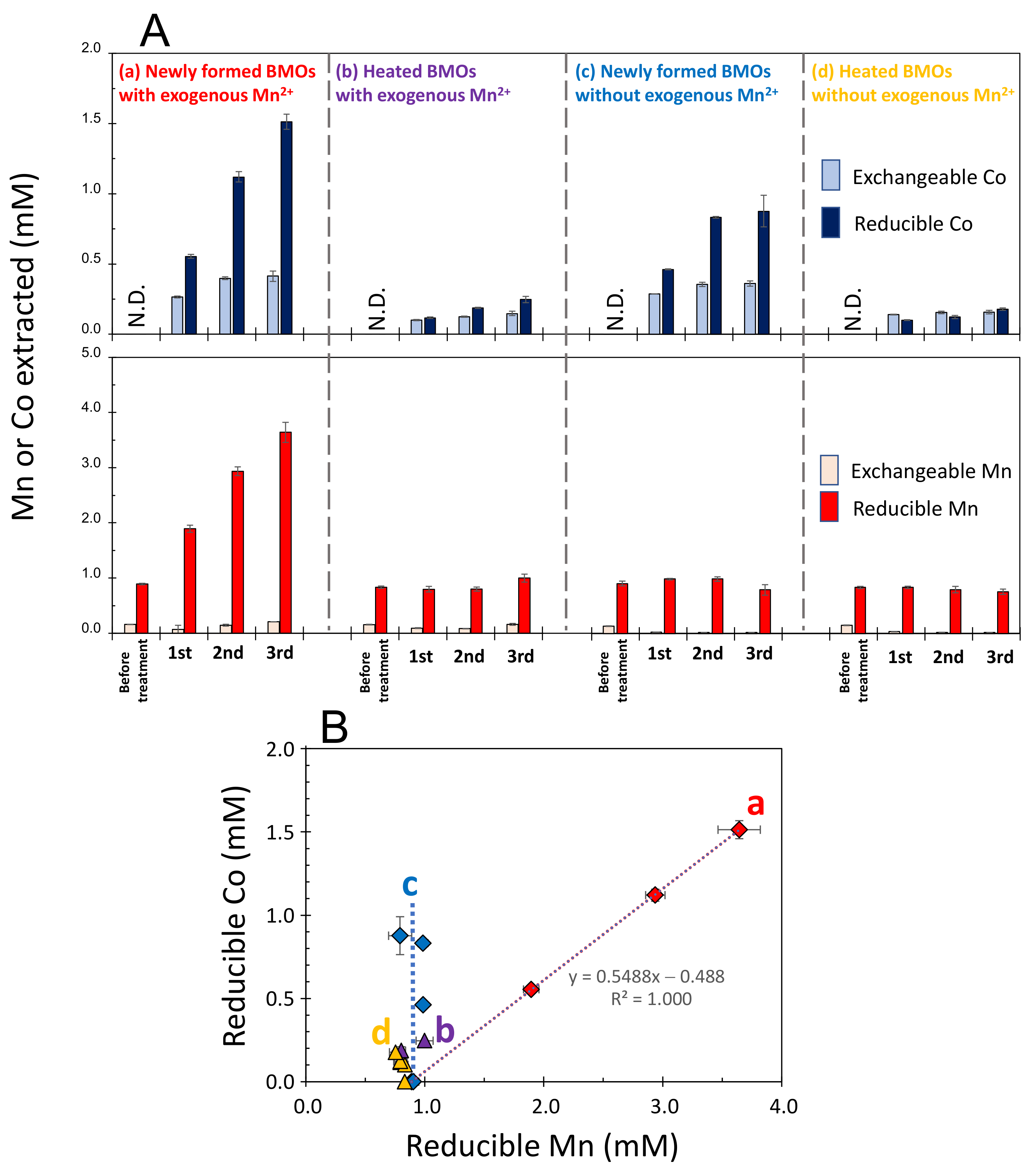
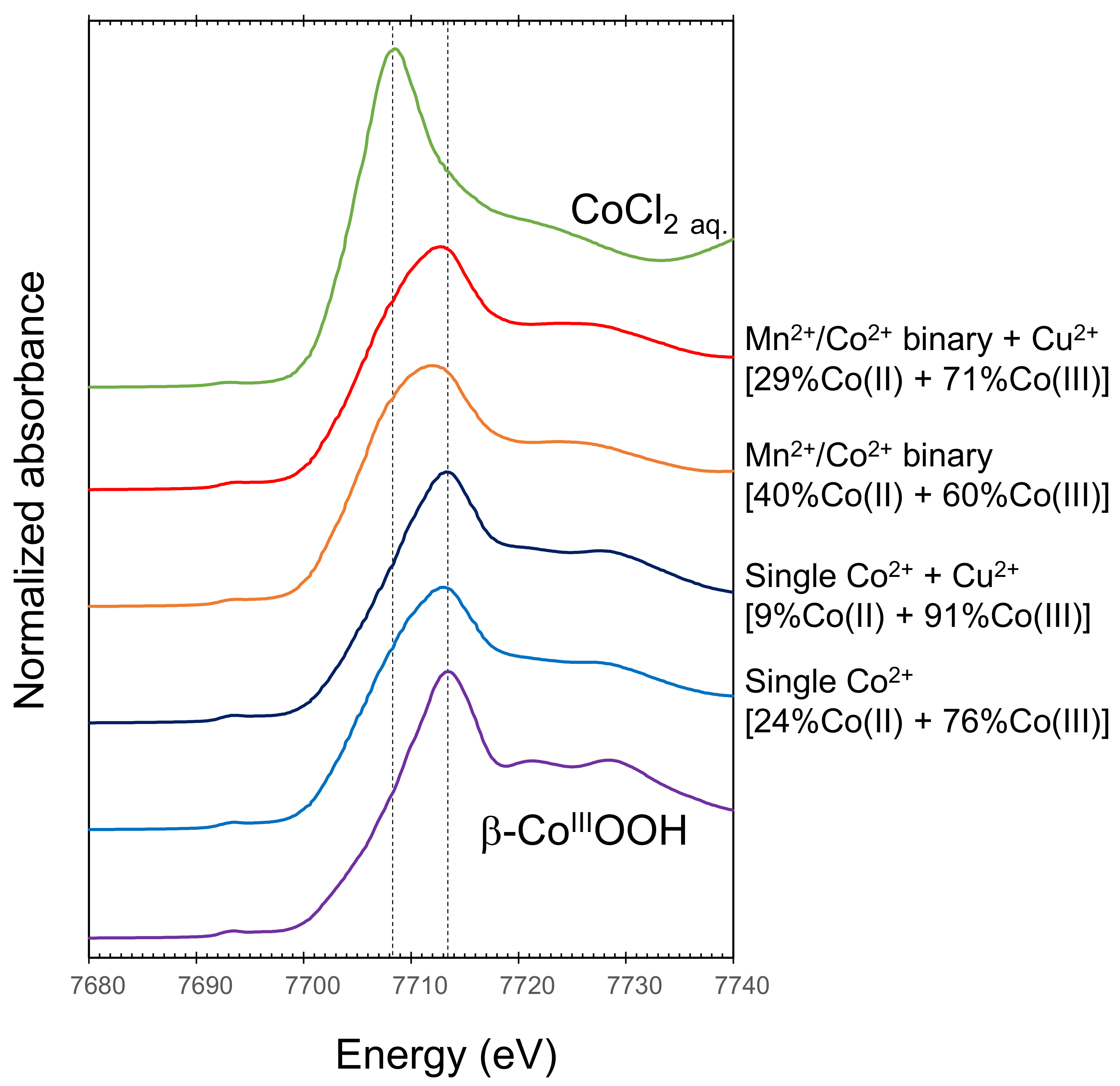
3.1. Sequestration of Co2+ by Newly Formed and Heated BMOs with/without Exogenous Mn2+
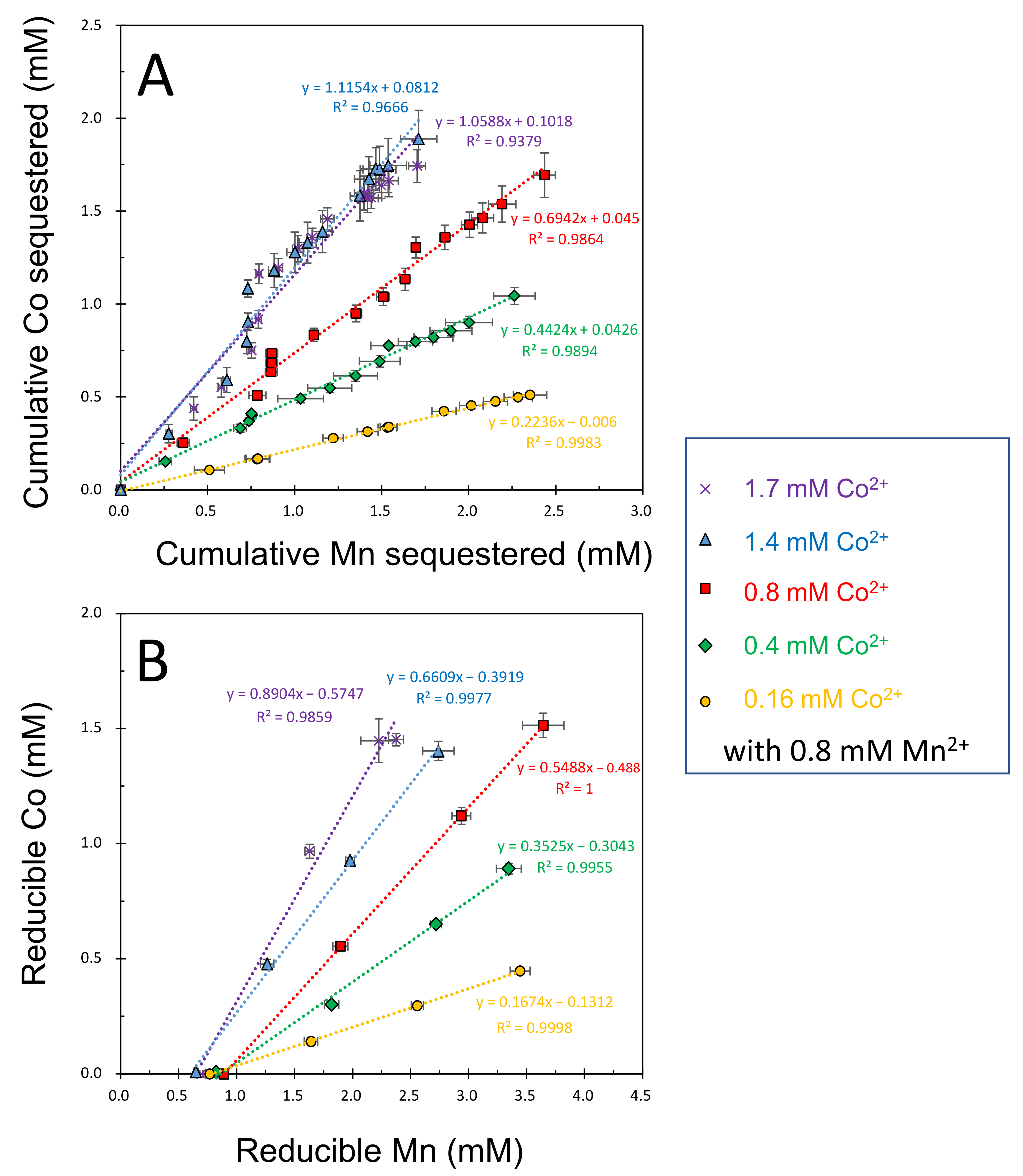
3.2. Mineralogical Composition of Solid Phases Formed in Mn2+/Co2+ Binary Systems
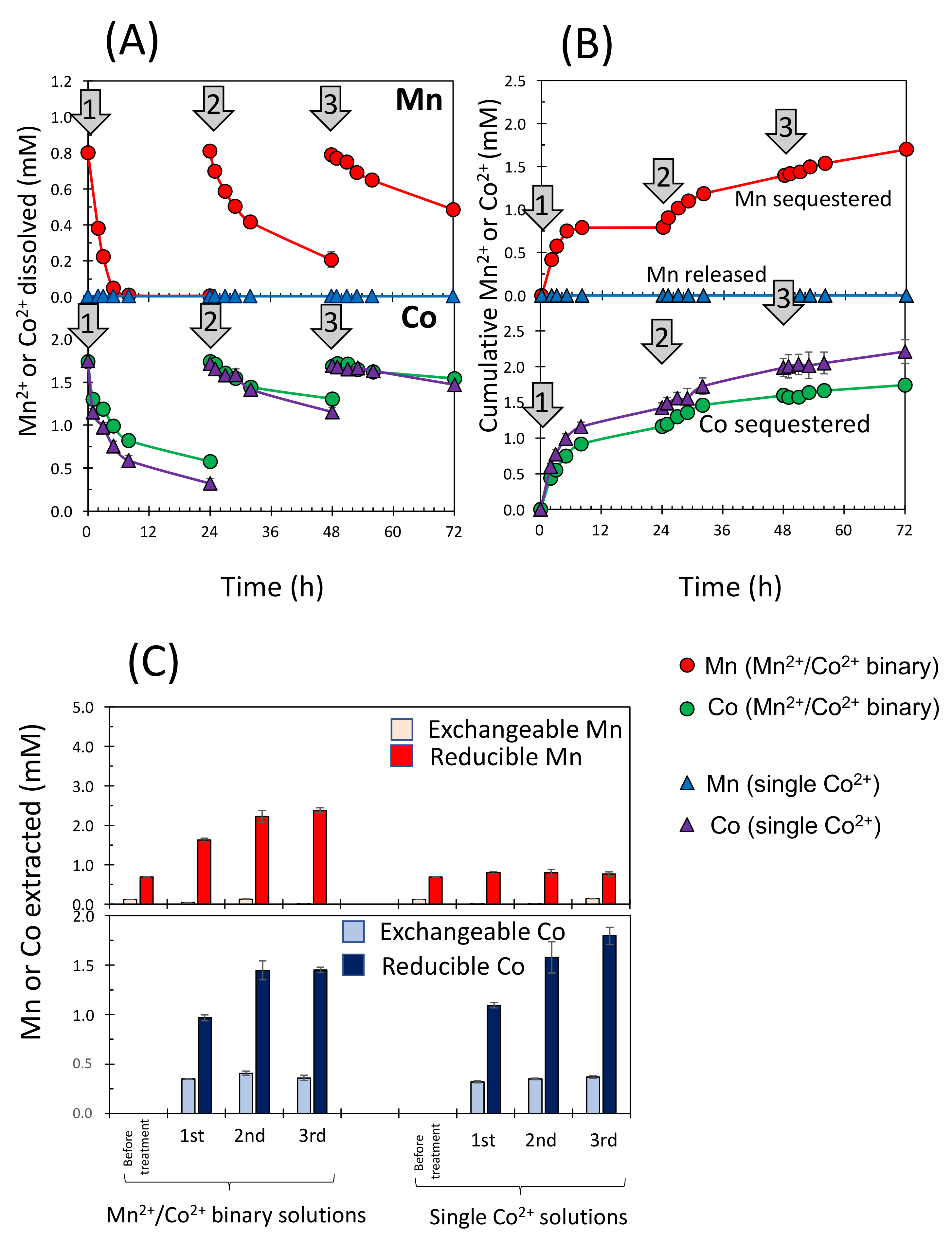
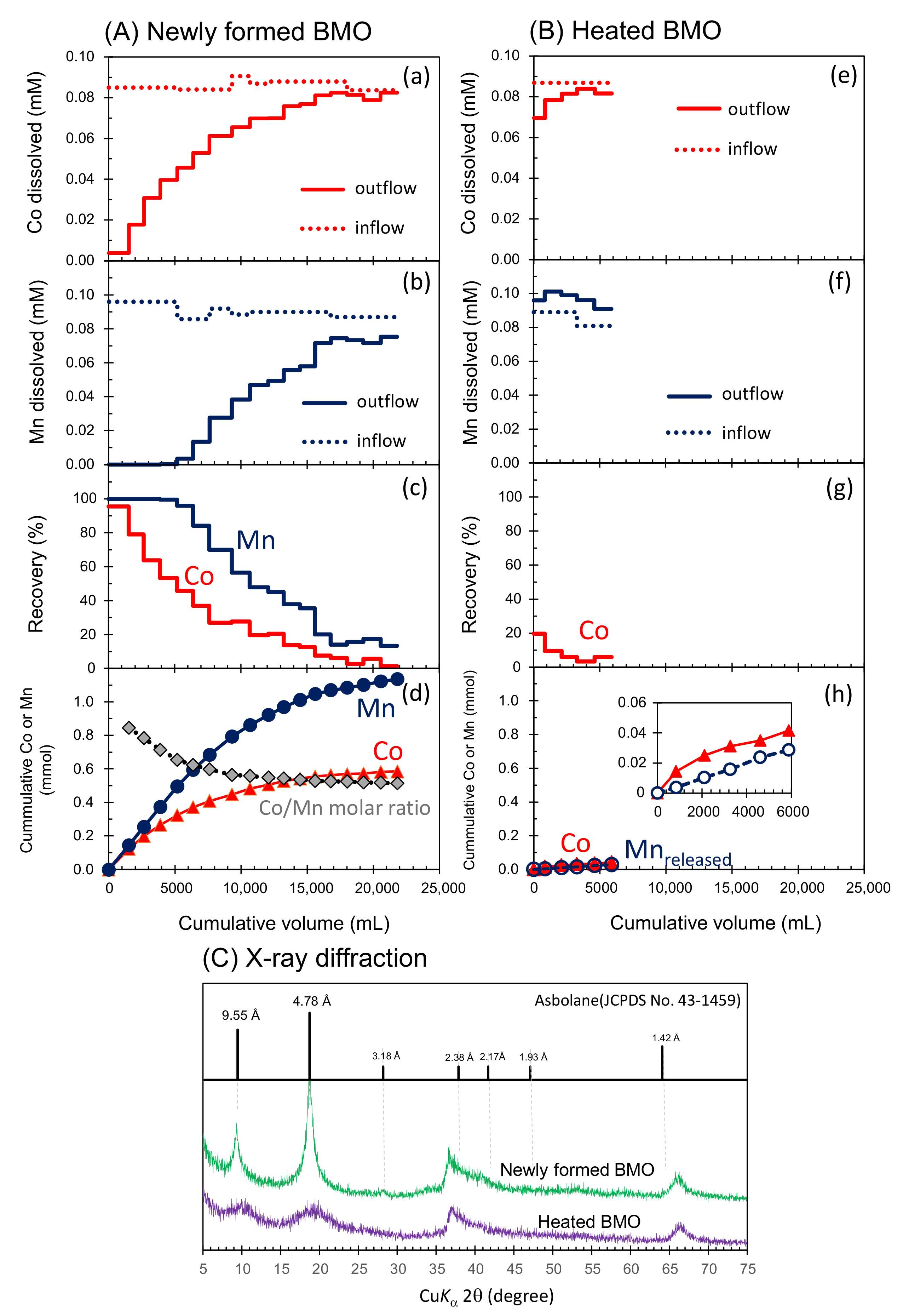
3.3. Simultaneous Sequestration of Co2+ and Mn2+ in the Column Experiments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Horn, S.; Gunn, A.G.; Petavratzi, E.; Shaw, R.A.; Eilu, P.; Törmänen, T.; Bjerkgård, T.; Sandstad, J.S.; Jonsson, E.; Kountourelis, S.; et al. Cobalt resources in Europe and the potential for new discoveries. Ore Geol. Rev. 2021, 130, 103915. [Google Scholar] [CrossRef]
- Leyssens, L.; Vinck, B.; Van Der Straeten, C.; Wuyts, F.; Maes, L. Cobalt toxicity in humans―A review of the potential sources and systemic health effects. Toxicology 2017, 387, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Mahey, S.; Kumar, R.; Sharma, M.; Bhardwaj, R. A critical review on toxicity of cobalt and its bioremediation strategies. SN Appl. Sci. 2020, 2, 1279. [Google Scholar] [CrossRef]
- Sheikh, I. Cobalt poisoning: A comprehensive review of the literature. Med. Toxicol. Clin. Forens. Med. 2016, 2, 2. [Google Scholar] [CrossRef]
- Yugo, M.; Soler, A. Outlook for battery raw materials (literature review). Concawe Rev. 2019, 28, 25–39. [Google Scholar]
- Kosiorek, M.; Wyszkowski, M. Effect of cobalt on the environment and living organisms―A review. Appl. Ecol. Environ. Res. 2019, 17, 11419–11449. [Google Scholar] [CrossRef]
- Moffett, J.W.; Ho, J. Oxidation of cobalt and manganese in seawater via a common microbially catalyzed pathway. Geochim. Cosmochim. Acta 1996, 60, 3415–3424. [Google Scholar] [CrossRef]
- Kay, J.T.; Conklin, B.; Fuller, C.C.; O’Day, P.A. Process of nickel and cobalt uptake by a manganese oxide forming sediment in Pinel Creek, globe mining distinct, Arizona. Environ. Sci. Technol. 2001, 35, 4719–4725. [Google Scholar] [CrossRef]
- Tebo, B.M.; Bargar, J.R.; Clement, B.G.; Dick, G.J.; Murray, K.J.; Parker, D.; Verity, R.; Webb, S.M. Biogenic manganese oxides: Properties and mechanisms of formation. Annu. Rev. Earth Planet. Sci. 2004, 32, 287–328. [Google Scholar] [CrossRef] [Green Version]
- Villalobos, M.; Bargar, J.; Sposito, G. Trace metal retention on biogenic manganese oxide nanoparticles. Elements 2005, 1, 223–226. [Google Scholar] [CrossRef]
- Murray, K.J.; Webb, S.M.; Bargar, J.R.; Tebo, B.M. Indirect oxidation of Co(II) in the presence of the marine Mn(II)-oxidizing bacterium Bacillus sp. strain SG-1. Appl. Environ. Microbiol. 2007, 73, 6905–6909. [Google Scholar] [CrossRef] [Green Version]
- Sasaki, K.; Matsuda, M.; Urata, T.; Hirajima, T.; Konno, H. Sorption of Co2+ ions on the biogenic Mn oxide produced by a Mn-oxidizing fungus, Paraconiothyrium sp. WL-2. Mater. Trans. 2008, 49, 605–611. [Google Scholar] [CrossRef] [Green Version]
- Sasaki, K.; Kaseyama, T.; Hirajima, T. Selective sorption of Co2+ over Ni2+ using biogenic manganese oxides. Mater. Trans. 2009, 50, 2643–2648. [Google Scholar] [CrossRef] [Green Version]
- Yu, Q.; Sasaki, K.; Tanaka, K.; Ohnuki, T.; Hirajima, T. Structural factors of biogenic birnessite produced by fungus Paraconiothyrium sp. WL-2 strain affecting sorption of Co2+. Chem. Geol. 2012, 310–311, 106–113. [Google Scholar] [CrossRef]
- Tanaka, K.; Yu, Q.; Sasaki, K.; Ohnuki, T. Cobalt(II) oxidation by biogenic Mn oxide produced by Pseudomonas sp. strain NGY-1. Geomicrobiol. J. 2013, 30, 874–885. [Google Scholar] [CrossRef]
- Chang, J.; Tani, Y.; Naitou, H.; Miyata, N.; Seyama, H.; Tanaka, K. Cobalt(II) sequestration on fungal biogenic manganese oxide enhanced by manganese(II) oxidase activity. Appl. Geochem. 2013, 37, 170–178. [Google Scholar] [CrossRef]
- Inthorn, D.; Tani, Y.; Chang, J.; Naitou, H.; Miyata, N. Magnetically modified fungal Mn oxides with high sequestration efficiency for simultaneously removing multiple heavy metal ions from wastewater. J. Environ. Chem. Eng. 2014, 2, 1635–1641. [Google Scholar] [CrossRef]
- Chang, J.; Tani, Y.; Naitou, H.; Miyata, N.; Seyama, H. Fungal Mn oxides supporting Mn(II) oxidase activity as effective Mn(II) sequestering materials. Environ. Technol. 2013, 34, 2781–2787. [Google Scholar] [CrossRef]
- Dehaine, Q.; Tijsseling, L.T.; Glass, H.J.; Törmänen, T.; Butcher, A.R. Geometallurgy of cobalt ores: A review. Miner. Eng. 2021, 160, 106656. [Google Scholar] [CrossRef]
- Watanabe, J.; Tani, Y.; Chang, J.; Miyata, N.; Naitou, H.; Seyama, H. As(III) oxidation kinetics of biogenic manganese oxides formed by Acremonium strictum strain KR21-2. Chem. Geol. 2013, 347, 227–232. [Google Scholar] [CrossRef]
- Zheng, H.; Tani, Y.; Naitou, H.; Miyata, N.; Tojo, F. Oxidative Ce3+ sequestration by fungal manganese oxides with an associated Mn(II) oxidase activity. Appl. Geochem. 2016, 71, 110–122. [Google Scholar] [CrossRef]
- Suzuki, R.; Tani, Y.; Naitou, H.; Miyata, N.; Tanaka, K. Sequestration and oxidation of Cr(III) by fungal Mn oxides with Mn(II) oxidizing activity. Catalysts 2020, 10, 44. [Google Scholar] [CrossRef] [Green Version]
- Chang, J.; Tani, Y.; Naitou, H.; Miyata, N.; Tojo, F.; Seyama, H. Zn(II) sequestration by fungal biogenic manganese oxide through enzymatic and abiotic processes. Chem. Geol. 2014, 383, 155–163. [Google Scholar] [CrossRef]
- Tani, Y.; Kakinuma, S.; Chang, J.; Tanaka, K.; Miyata, N. Preferential elimination of Ba2+ through irreversible biogenic manganese oxide sequestration. Minerals 2021, 11, 53. [Google Scholar] [CrossRef]
- Sujith, P.P.; Gonsalves, M.J.B.D. Ferromanganese oxide deposits: Geochemical and microbiological perspectives of interactions of cobalt and nickel. Ore Geol. Rev. 2021, 139, 104458. [Google Scholar] [CrossRef]
- Miyata, N.; Tani, Y.; Sakata, M.; Iwahori, K. Microbial manganese oxide formation and interaction with toxic metal ions. J. Biosci. Bioeng. 2007, 104, 1–8. [Google Scholar] [CrossRef]
- Grangeon, S.; Bataillard, P.; Coussy, S. The nature of manganese oxides in soils and their role as scavengers of trace elements: Implication for soil remediation. In Environmental Soil Remediation and Rehabilitation; van Hullebusch, E.D., Huguenot, D., Pechaud, Y., Simonnot, M.-O., Colombano, S., Eds.; Springer Nature: Cham, Switzerland, 2020; pp. 399–429. [Google Scholar] [CrossRef]
- Miyata, N.; Tani, Y.; Iwahori, K.; Soma, M. Enzymatic formation of manganese oxides by an Acremonium-like hyphomycete fungus, strain KR21-2. FEMS Microbiol. Ecol. 2004, 47, 101–109. [Google Scholar] [CrossRef] [Green Version]
- Miyata, N.; Tani, Y.; Maruo, K.; Tsuno, H.; Sakata, M.; Iwahori, K. Manganese(IV) oxide production by Acremonium sp. strain KR21-2 and extracellular Mn(II) oxidase activity. Appl. Environ. Microbiol. 2006, 72, 6467–6473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tani, Y.; Miyata, N.; Ohashi, M.; Ohnuki, T.; Seyama, H.; Iwahori, K.; Soma, M. Interaction of inorganic arsenic with biogenic manganese oxide produced by a Mn-oxidizing fungus, strain KR21-2. Environ. Sci. Technol. 2004, 38, 6618–6624. [Google Scholar] [CrossRef]
- Tojo, F.; Kitayama, A.; Miyata, N.; Okano, K.; Fukushima, J.; Suzuki, R.; Tani, Y. Molecular cloning and heterologous expression of manganese(II)-oxidizing enzyme from Acremonium strictum strain KR21-2. Catalysts 2020, 10, 686. [Google Scholar] [CrossRef]
- Tani, Y.; Ohashi, M.; Miyata, N.; Seyama, H.; Iwahori, K.; Soma, M. Sorption of Co(II), Ni(II) and Zn(II) ions on biogenic manganese oxide produced by a Mn-oxidizing fungus, strain KR21-2. J. Environ. Sci. Health Part A Toxic/Hazard. Subst. Environ. Eng. 2004, 39, 2641–2660. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Tani, Y.; Naitou, H.; Miyata, N.; Seyama, H.; Tanaka, K. Sequestration of Cd(II) and Ni(II) ions on fungal manganese oxides associated with Mn(II) oxidase activity. Appl. Geochem. 2014, 47, 198–208. [Google Scholar] [CrossRef]
- Zheng, H.; Tani, Y.; Naitou, H.; Miyata, N.; Tojo, F.; Seyama, H. Sequestration of La3+ by fungal manganese oxides and the effect of Mn(II) oxidase activity. J. Environ. Chem. Eng. 2017, 5, 735–743. [Google Scholar] [CrossRef]
- Rosson, R.A.; Nealson, K.H. Manganese binding and oxidation by spores of a marine bacillus. J. Bacteriol. 1982, 151, 1027–1034. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Vrind, J.P.; de Vrind-de Jong, E.W.; de Voogt, J.W.; Westbroek, P.; Boogerd, F.C.; Rosson, R.A. Manganese oxidation by spores and spore coats of a marine Bacillus species. Appl. Environ. Microbiol. 1986, 52, 1096–1100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watanabe, J.; Tani, Y.; Miyata, N.; Seyama, H.; Mitsunobu, S.; Naitou, H. Concurrent sorption of As(V) and Mn(II) during biogenic manganese oxide formation. Chem. Geol. 2012, 306–307, 123–128. [Google Scholar] [CrossRef]
- Grangeon, S.; Lanson, B.; Miyata, N.; Tani, Y.; Manceau, A. Structure of nanocrystalline phyllomanganates produced by freshwater fungi. Am. Mineral. 2010, 95, 1608–1616. [Google Scholar] [CrossRef] [Green Version]
- Burlet, C.; Vanbrabant, Y. Study of the spectro-chemical signatures of cobalt-manganese layered oxides (asbolane-lithiophorite and their intermediates) by Raman spectroscopy. J. Raman Spectrosc. 2015, 46, 941–952. [Google Scholar] [CrossRef]
- Sánchez-España, J.; Yusta, I. Coprecipitation of Co2+, Ni2+ and Zn2+ with Mn(III/IV) oxides formed in metal-rich mine waters. Minerals 2019, 9, 226. [Google Scholar] [CrossRef] [Green Version]
- Roqué-Rosell, J.; Mosselmans, J.F.W.; Proenza, J.A.; Labrador, M.; Galí, S.; Atkinson, K.D.; Quinn, P.D. Sorption of Ni by “lithiophorite-asbolane” intermediates in Moa Bay lateritic deposits, eastern Cuba. Chem. Geol. 2010, 275, 9–18. [Google Scholar] [CrossRef]
- Manceau, A.; LLorca, S.; Calas, G. Crystal chemistry of cobalt and nickel in lithiophorite and asbolane from New Caledonia. Geochim. Cosmochim. Acta 1987, 51, 105–113. [Google Scholar] [CrossRef]
- Post, J.E. Manganese oxide minerals: Crystal structures and economic and environmental significance. Proc. Natl. Acad. Sci. USA 1999, 96, 3447–3454. [Google Scholar] [CrossRef] [PubMed] [Green Version]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aoshima, M.; Tani, Y.; Fujita, R.; Tanaka, K.; Miyata, N.; Umezawa, K. Simultaneous Sequestration of Co2+ and Mn2+ by Fungal Manganese Oxide through Asbolane Formation. Minerals 2022, 12, 358. https://doi.org/10.3390/min12030358
Aoshima M, Tani Y, Fujita R, Tanaka K, Miyata N, Umezawa K. Simultaneous Sequestration of Co2+ and Mn2+ by Fungal Manganese Oxide through Asbolane Formation. Minerals. 2022; 12(3):358. https://doi.org/10.3390/min12030358
Chicago/Turabian StyleAoshima, Miku, Yukinori Tani, Rina Fujita, Kazuya Tanaka, Naoyuki Miyata, and Kazuhiro Umezawa. 2022. "Simultaneous Sequestration of Co2+ and Mn2+ by Fungal Manganese Oxide through Asbolane Formation" Minerals 12, no. 3: 358. https://doi.org/10.3390/min12030358
APA StyleAoshima, M., Tani, Y., Fujita, R., Tanaka, K., Miyata, N., & Umezawa, K. (2022). Simultaneous Sequestration of Co2+ and Mn2+ by Fungal Manganese Oxide through Asbolane Formation. Minerals, 12(3), 358. https://doi.org/10.3390/min12030358





