Chemical, Microstructural and Morphological Characterisation of Dentine Caries Simulation by pH-Cycling
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design and Sample Preparation
2.2. Artificial Caries Induction (pHc)
2.3. Attenuated Total Reflectance Infrared Spectrometry Analyses
2.4. Raman Spectrometry Analyses
2.5. Two Dimensional X-ray Diffraction Analyses
2.6. Scanning Electron Microscopy
2.7. Masson’s Trichrome Staining
2.8. Statistical Analyses
3. Results
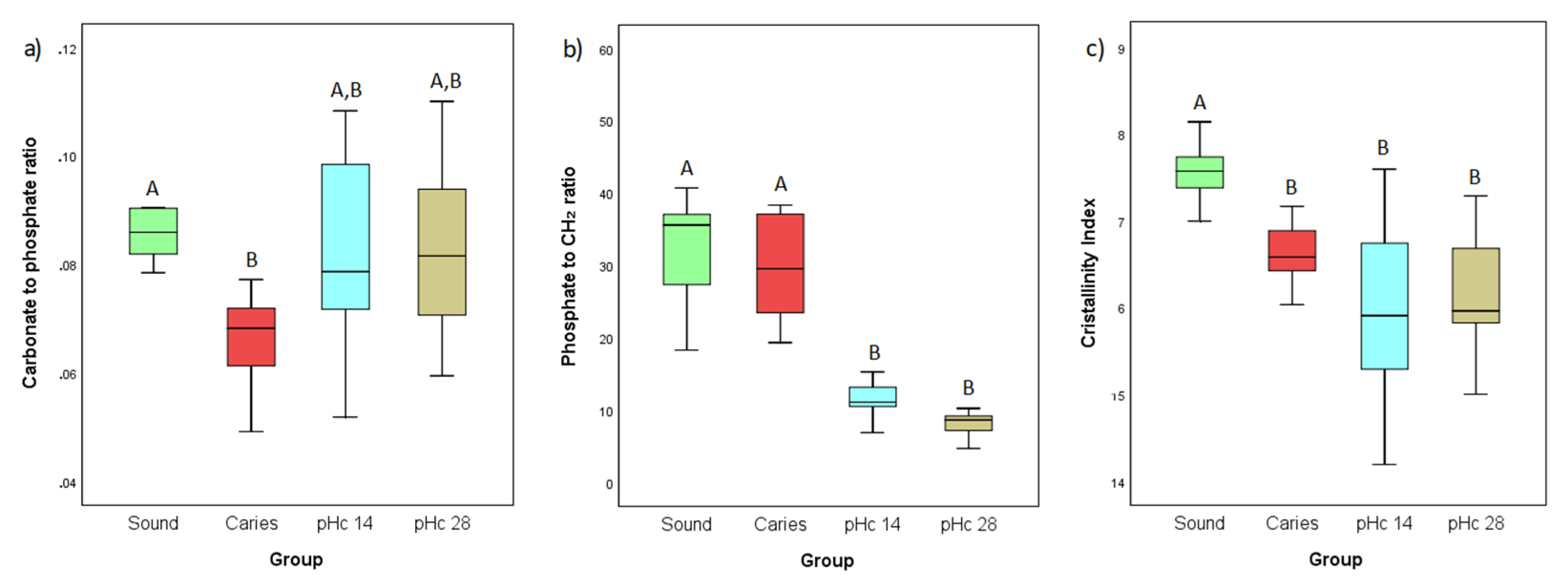
3.1. ATR-FTIR Analyses
3.2. Raman Analyses
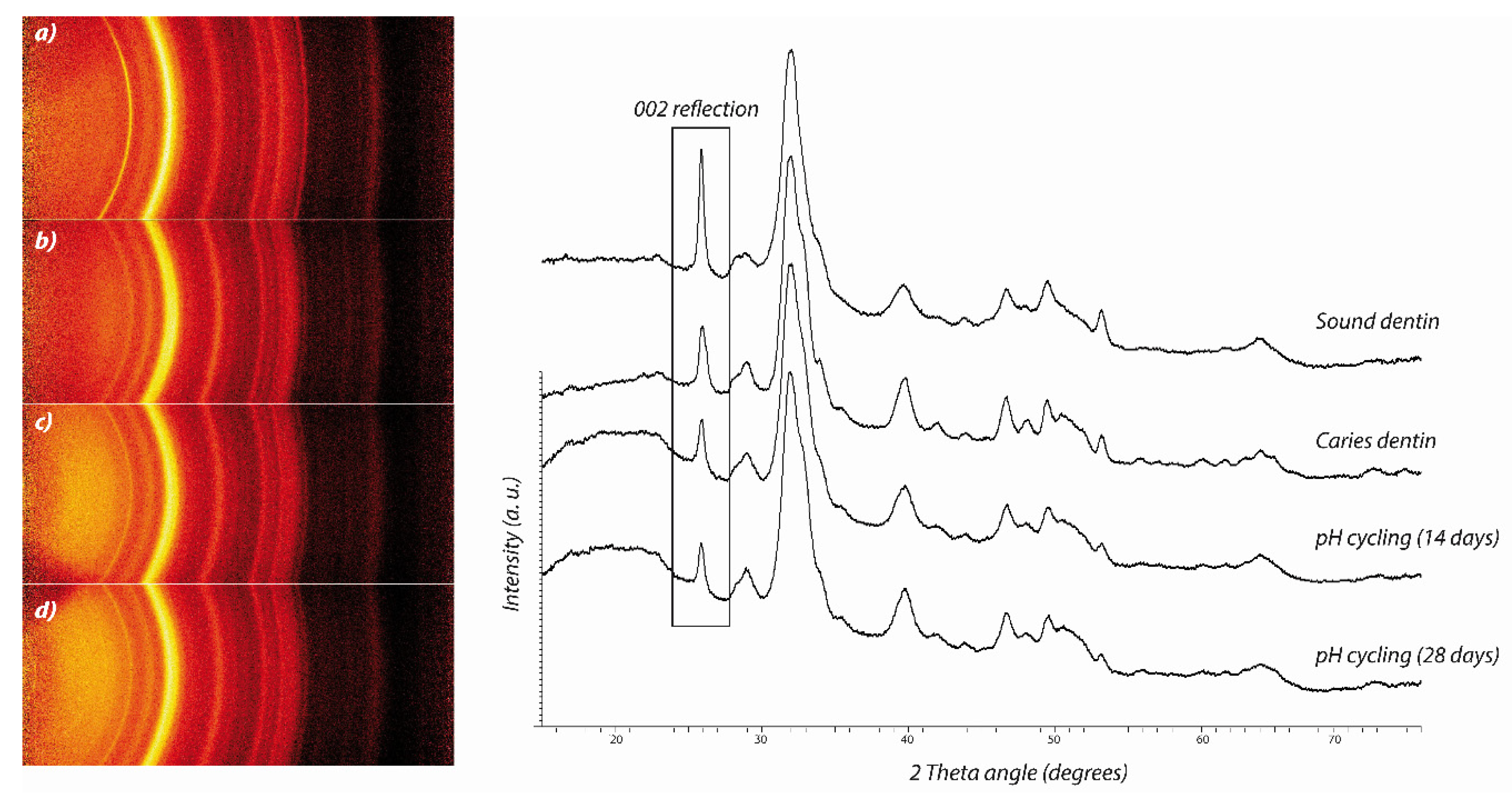
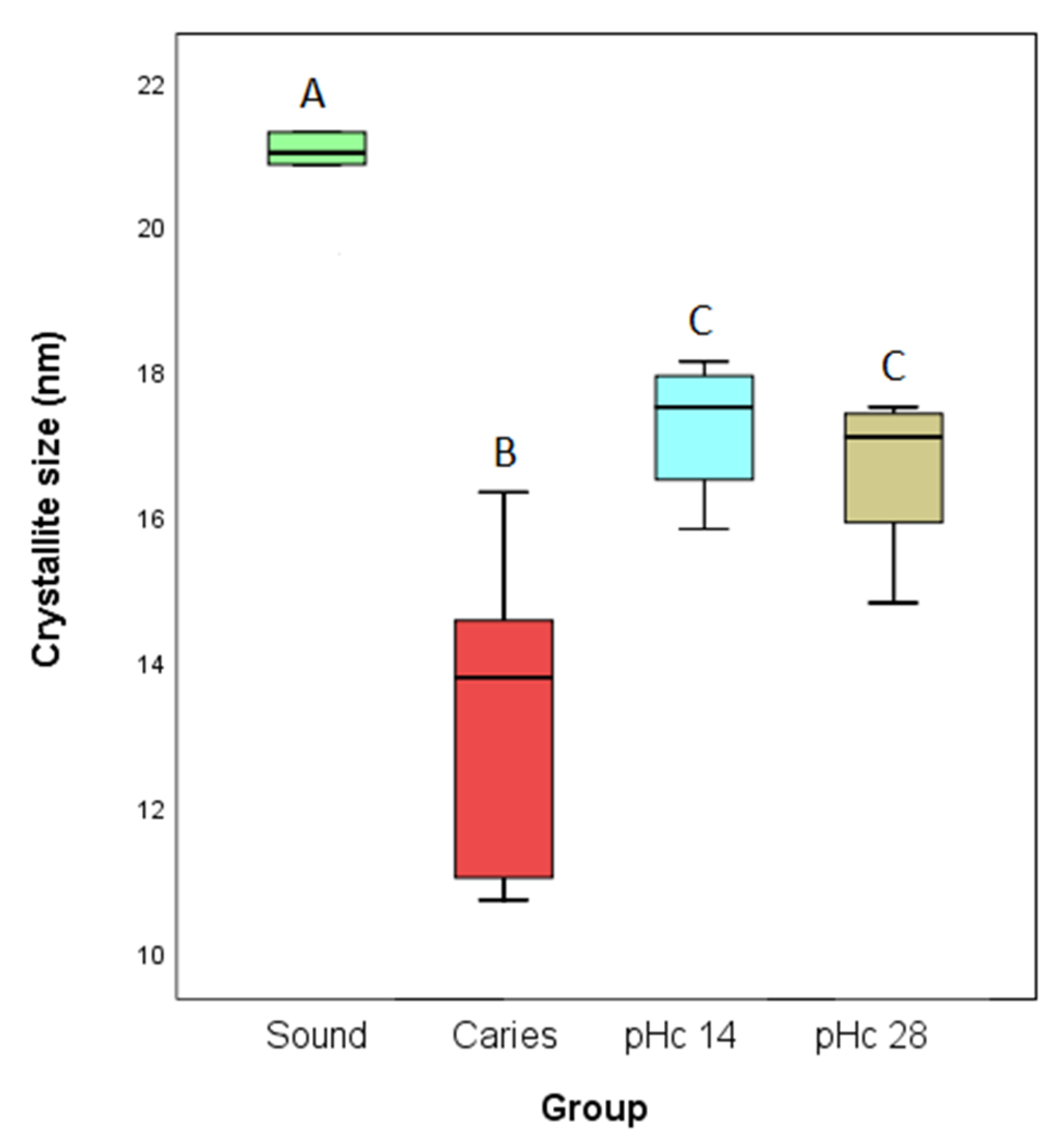
3.3. X-ray Diffraction Analyses
3.4. Scanning Electron Microscopy Analyses
3.5. Masson’s Trichrome
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nelson, S.J. Introduction to Dental Anatomy. In Wheeler’s Dental Anatomy, Physiology, and Occlusion; Elsevier, Saunders: St. Louis, MO, USA, 2015; pp. 1–19. [Google Scholar]
- De La Dure-Molla, M.; Fournier, B.P.; Manzanares, M.C.; Acevedo, A.C.; Hennekam, R.C.; Friedlander, L.; Boy-Lefèvre, M.-L.; Kerner, S.; Toupenay, S.; Garrec, P.; et al. Elements of morphology: Standard terminology for the teeth and classifying genetic dental disorders. Am. J. Med. Genet. Part A 2019, 179, 1913–1981. [Google Scholar] [CrossRef] [Green Version]
- Curzon, M.E.J.; Featherstone, J.D.B. Chemical composition of enamel. In CRC Handbook of Experimental Aspects of Oral Biochemistry; Lazari, E., Levy, B., Eds.; CRC Press: Boca Raton, FL, USA, 1983; pp. 123–134. [Google Scholar]
- Carlson, S.J. Vertebrate dental structures. In Skeletal Biomineralization: Patterns, Processes and Evolutionary Trends; Carter, J.G., Ed.; Van Nostrand: New York, NY, USA, 1990; Volume 1, pp. 531–556. [Google Scholar]
- LeGeros, R.Z.; LeGeros, J.P. Phosphate minerals in human tissues. In Phosphate Minerals; Nriagu, J.O., Moore, P.B., Eds.; Springer: Berlin/Heidelberg, Germany, 1984; pp. 351–385. [Google Scholar]
- Nanci, A. Structure of the oral tissues. In Ten Cate’s Oral Histology: Development, Structure and Function; Nanci, A., Ed.; Elsevier: St. Louis, MO, USA, 2018; pp. 18–45. [Google Scholar]
- Selwitz, R.H.; Ismail, A.I.; Pitts, N.B. Dental caries. Lancet 2007, 369, 51–59. [Google Scholar] [CrossRef]
- Machiulskiene, V.; Campus, G.; Carvalho, J.C.; Dige, I.; Ekstrand, K.R.; Jablonski-Momeni, A.; Maltz, M.; Manton, D.J.; Martignon, S.; Martinez-Mier, E.A.; et al. Terminology of dental caries and dental caries management: Consensus report of a workshop organized by ORCA and cariology research group of IADR. Caries Res. 2020, 54, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Pitts, N.B.; Zero, D.T.; Marsh, P.D.; Ekstrand, K.; Weintraub, J.A.; Ramos-Gomez, F.; Tagami, J.; Twetman, S.; Tsakos, G.; Ismail, A. Dental caries. Nat. Rev. Dis. Primers 2017, 3, 17030. [Google Scholar] [CrossRef] [Green Version]
- Skucha-Nowak, M.; Gibas, M.; Tanasiewicz, M.; Twardawa, H.; Szklarski, T. Natural and controlled demineralization for study purposes in minimally invasive dentistry. Adv. Clin. Exp. Med. 2015, 24, 891–898. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, L.; Hilton, J.F.; Habelitz, S.; Marshall, S.J.; Marshall, G.W. Dentin caries activity status related to hardness and elasticity. Eur. J. Oral Sci. 2003, 111, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Lenzi, T.L.; Soares, F.Z.; Tedesco, T.K.; de Oliveira Rocha, R. Is it possible to induce artificial caries-affected dentin using the same protocol to primary and permanent teeth? J. Contemp. Dent. Pract. 2015, 16, 638–642. [Google Scholar] [CrossRef]
- Scholtanus, J.D.; Purwanta, K.; Dogan, N.; Kleverlaan, C.J.; Feilzer, A.J. Microtensile bond strength of three simplified adhesive systems to caries-affected dentin. J. Adhes. Dent. 2010, 12, 273–278. [Google Scholar] [CrossRef]
- Shibata, S.; Vieira, L.C.C.; Baratieri, L.N.; Fu, J.; Hoshika, S.; Matsuda, Y.; Sano, H. Evaluation of microtensile bond strength of self-etching adhesives on normal and caries-affected dentin. Dent. Mater. J. 2016, 35, 166–173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwendicke, F.; Dörfer, C.E.; Paris, S. Incomplete caries removal: A systematic review and meta-analysis. J. Dent. Res. 2013, 92, 306–314. [Google Scholar] [CrossRef]
- Ehrlich, H.; Koutsoukos, P.G.; Demadis, K.D.; Pokrovsky, O.S. Principles of demineralization: Modern strategies for the isolation of organic frameworks. Part I. Common definitions and history. Micron 2008, 39, 1062–1091. [Google Scholar] [CrossRef] [PubMed]
- Erhardt, M.C.G.; Rodrigues, J.A.; Valentino, T.A.; Ritter, A.V.; Pimenta, L.A.F. In vitro μTBS of one-bottle adhesive systems: Sound versus artificially-created caries-affected dentin. J. Biomed. Mater. Res.-Part B Appl. Biomater. 2008, 86, 181–187. [Google Scholar] [CrossRef]
- Marquezan, M.; Corrêa, F.N.P.; Sanabe, M.E.; Rodrigues Filho, L.E.; Hebling, J.; Guedes-Pinto, A.C.; Mendes, F.M. Artificial methods of dentine caries induction: A hardness and morphological comparative study. Arch. Oral Biol. 2009, 54, 1111–1117. [Google Scholar] [CrossRef]
- Schwendicke, F.; Eggers, K.; Meyer-Lueckel, H.; Dörfer, C.; Kovalev, A.; Gorb, S.; Paris, S. In vitro induction of residual caries lesions in dentin: Comparative mineral loss and nano-hardness analysis. Caries Res. 2015, 49, 259–265. [Google Scholar] [CrossRef]
- Ramakrishnaiah, R.; Ur Rehman, G.U.; Basavarajappa, S.; Al Khuraif, A.A.; Durgesh, B.H.; Khan, A.S.; Ur Rehman, I. Applications of Raman spectroscopy in dentistry: Analysis of tooth structure. Appl. Spectrosc. Rev. 2015, 50, 332–350. [Google Scholar] [CrossRef]
- Wang, R.; Wang, Y. Fourier transform infrared spectroscopy in oral cancer diagnosis. Int. J. Mol. Sci. 2021, 22, 1206. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Boyes, V.; Festy, F.; Lynch, R.J.M.; Watson, T.F.; Banerjee, A. In-vitro subsurface remineralisation of artificial enamel white spot lesions pre-treated with chitosan. Dent. Mater. 2018, 34, 1154–1167. [Google Scholar] [CrossRef] [Green Version]
- Daood, U.; Swee Heng, C.; Neo Chiew Lian, J.; Fawzy, A.S. In vitro analysis of riboflavin-modified, experimental, two-step etch-and-rinse dentin adhesive: Fourier transform infrared spectroscopy and micro-Raman studies. Int. J. Oral Sci. 2015, 7, 110–124. [Google Scholar] [CrossRef]
- Guven, Y.; Tuna, E.B.; Dincol, M.E.; Aktoren, O. X-ray difrraction analysis of MTA-Plus, MTA-Angelus and DiaRoot BioAggregate. Eur. J. Dent. 2014, 8, 211–215. [Google Scholar] [PubMed]
- Zhao, I.S.; Mei, M.L.; Li, Q.L.; Lo, E.C.M.; Chu, C.H. Arresting simulated dentine caries with adjunctive application of silver nitrate solution and sodium fluoride varnish: An in vitro study. Int. Dent. J. 2017, 67, 206–214. [Google Scholar] [CrossRef] [Green Version]
- Barbosa-Martins, L.F.; de Sousa, J.P.; de Castilho, A.R.F.; Puppin-Rontani, J.; Davies, R.P.W.; Puppin-Rontani, R.M. Enhancing bond strength on demineralized dentin by pre-treatment with selective remineralising agents. J. Mech. Behav. Biomed. Mater. 2018, 81, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, R.; Mizutani, K.; Lin, T.; Kakizaki, S.; Mimata, A.; Watanabe, K.; Saito, N.; Meinzer, W.; Iwata, T.; Izumi, Y.; et al. Ex vivo evaluation of gingival ablation with various laser systems and electroscalpel. Photobiomodulation Photomed. Laser Surg. 2020, 38, 364–373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pabst, A.M.; Müller, W.E.G.; Ackermann, M. Three-dimensional scanning electron microscopy of maxillofacial biomaterials. Br. J. Oral Maxillofac. Surg. 2017, 55, 736–739. [Google Scholar] [CrossRef]
- Stape, T.H.S.; Tjäderhane, L.; Marques, M.R.; Aguiar, F.H.B.; Martins, L.R.M. Effect of dimethyl sulfoxide wet-bonding technique on hybrid layer quality and dentin bond strength. Dent. Mater. 2015, 31, 676–683. [Google Scholar] [CrossRef]
- Petersen, P.E.; Bourgeois, D.; Ogawa, H.; Estupinan-Day, S.; Ndiaye, C. The global burden of oral diseases and risks to oral health. Bull. World Health Organ. 2005, 83, 661–669. [Google Scholar]
- Ismail, A.I.; Sohn, W.; Tellez, M.; Amaya, A.; Sen, A.; Hasson, H.; Pitts, N.B. The International Caries Detection and Assessment System (ICDAS): An integrated system for measuring dental caries. Community Dent. Oral Epidemiol. 2007, 35, 170–178. [Google Scholar] [CrossRef] [Green Version]
- Braun, A.; Guiraud, L.M.J.C.; Frankenberger, R. Histological validation of ICDAS II and radiological assessment of occlusal carious lesions in permanent teeth. Odontology 2017, 105, 46–53. [Google Scholar] [CrossRef]
- Paschalis, E.P.; DiCarlo, E.; Betts, F.; Sherman, P.; Mendelsohn, R.; Boskey, A.L. FTIR microspectroscopic analysis of human osteonal bone. Calcif. Tissue Int. 1996, 59, 480–487. [Google Scholar] [CrossRef]
- Mata-Miranda, M.M.; Guerrero-Ruiz, M.; Gonzalez-Fuentes, J.R.; Hernandez-Toscano, C.M.; Garcia-Andino, J.R.; Sanchez-Brito, M.; Vazquez-Zapien, G.J. Characterization of the biological fingerprint and identification of associated parameters in stress fractures by FTIR spectroscopy. BioMed Res. Int. 2019, 2019, 1241452. [Google Scholar] [CrossRef] [Green Version]
- Imbert, L.; Gourion-Arsiquaud, S.; Villarreal-Ramirez, E.; Spevak, L.; Taleb, H.; van der Meulen, M.C.H.; Mendelsohn, R.; Boskey, A.L. Dynamic structure and composition of bone investigated by nanoscale infrared spectroscopy. PLoS ONE 2018, 13, e0202833. [Google Scholar] [CrossRef]
- Levallois, B.; Terrer, E.; Panayotov, Y.; Salehi, H.; Tassery, H.; Tramini, P.; Salehi, H.; Cuisinier, F. Molecular structural analysis of carious lesions using micro-Raman spectroscopy. Eur. J. Oral Sci. 2012, 120, 444–451. [Google Scholar] [CrossRef]
- Toledano, M.; Toledano-Osorio, M.; Guerado, E.; Caso, E.; Aguilera, F.S.; Osorio, R. Biochemical assessment of nanostructures in human trabecular bone: Proposal of a Raman microspectroscopy based measurements protocol. Injury 2018, 49, S11–S21. [Google Scholar] [CrossRef] [PubMed]
- Slimani, A.; Nouioua, F.; Desoutter, A.; Levallois, B.; Cuisinier, F.J.G.; Tassery, H.; Terrer, E.; Salehi, H. Confocal Raman mapping of collagen cross-link and crystallinity of human dentin–enamel junction. J. Biomed. Opt. 2017, 22, 086003. [Google Scholar] [CrossRef]
- Cullity, B.D.; Stock, S.R. Elements of X-ray Diffraction, 3rd ed.; Prentice-Hall: Upper Saddle River, NJ, USA, 2001. [Google Scholar]
- Paschalis, E.P. Fourier transform infrared imaging of bone. In Bone Research Protocols; Helfrich, M.H., Ralston, S.H., Eds.; Human Press: Totowa, NJ, USA, 2012; pp. 517–525. ISBN 9781617794155. [Google Scholar]
- Neto, A.S.; Fonseca, A.C.; Abrantes, J.C.C.; Coelho, J.F.J.; Ferreira, J.M.F. Surface functionalization of cuttlefish bone-derived biphasic calcium phosphate scaffolds with polymeric coatings. Mater. Sci. Eng. C 2019, 105, 110014. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, Y. Proanthocyanidins’ efficacy in stabilizing dentin collagen against enzymatic degradation: MALDI-TOF and FTIR analyses. J. Dent. 2013, 41, 535–542. [Google Scholar] [CrossRef] [Green Version]
- Rodriguez-Navarro, A.B.; Romanek, C.S.; Alvarez-Lloret, P.; Gaines, K.F. Effect of in ovo exposure to PCBs and Hg on clapper rail bone mineral chemistry from a contaminated salt marsh in coastal Georgia. Environ. Sci. Technol. 2006, 40, 4936–4942. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Magne, D.; Weiss, P.; Bouler, J.M.; Laboux, O.; Daculsi, G. Study of the maturation of the organic (type I collagen) and mineral (nonstoichiometric apatite) constituents of a calcified tissue (dentin) as a function of location: A fourier transform infrared microspectroscopic investigation. J. Bone Miner. Res. 2001, 16, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Paschalis, E.P.; Mendelsohn, R.; Boskey, A.L. Infrared assessment of bone quality: A review. Clin. Orthop. Relat. Res. 2011, 469, 2170–2178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Curylofo-Zotti, F.A.; Tanta, G.S.; Zucoloto, M.L.; Souza-Gabriel, A.E.; Corona, S.A.M. Selective removal of carious lesion with Er:YAG laser followed by dentin biomodification with chitosan. Lasers Med. Sci. 2017, 32, 1595–1603. [Google Scholar] [CrossRef]
- Velo, M.M.D.A.C.; Magalhães, A.C.; Shiota, A.; Farha, A.L.H.; Grizzo, L.T.; Honório, H.M.; Wang, L. Profile of high-fluoride toothpastes combined or not with fuctionalized tri-calcium phosphate on root dentin caries control: An in vitro study. Am. J. Dent. 2018, 31, 290–296. [Google Scholar]
- Lenzi, T.L.; Soares, F.Z.M.; Raggio, D.P.; Pereira, G.K.R.; Rocha, R.D.O. Dry-bonding etch-and-rinse strategy improves bond longevity of a universal adhesive to sound and artificially-induced caries-affected primary dentin. J. Adhes. Dent. 2016, 18, 475–482. [Google Scholar] [CrossRef]
- Nicoloso, G.F.; Antoniazzi, B.F.; Lenzi, T.L.; Soares, F.Z.M.; Rocha, R.D.O. The bonding performance of a universal adhesive to artificially-created caries-affected dentin. J. Adhes. Dent. 2017, 19, 317–321. [Google Scholar] [CrossRef] [PubMed]
- White, D.J. The comparative sensitivity of intra-oral, in vitro, and animal models in the “profile” evaluation of topical fluorides. J. Dent. Res. 1992, 71, 884–894. [Google Scholar] [CrossRef]
- Buzalaf, M.A.R.; Hannas, A.R.; Magalhães, A.C.; Rios, D.; Honório, H.M.; Delbem, A.C.B. pH-cycling models for in vitro evaluation of the efficacy of fluoridated dentifrices for caries control: Strengths and limitations. J. Appl. Oral Sci. 2010, 18, 316–334. [Google Scholar] [CrossRef]
- Goettsche, Z.S.; Ettinger, R.L.; Wefel, J.S.; Hogan, M.M.; Harless, J.D.; Qian, F. In vitro assessment of 3 dentifrices containing fluoride in preventing demineralization of overdenture abutments and root surfaces. J. Prosthet. Dent. 2014, 112, 1257–1264. [Google Scholar] [CrossRef] [PubMed]
- Marin, E.; Hiraishi, N.; Honma, T.; Boschetto, F.; Zanocco, M.; Zhu, W.; Adachi, T.; Kanamura, N.; Yamamoto, T.; Pezzotti, G. Raman spectroscopy for early detection and monitoring of dentin demineralization. Dent. Mater. 2020, 36, 1635–1644. [Google Scholar] [CrossRef]
- Enrich-Essvein, T.; Benavides-Reyes, C.; Álvarez-Lloret, P.; Bolaños-Carmona, M.V.; Rodríguez-Navarro, A.B.; González-López, S. Influence of de-remineralization process on chemical, microstructural, and mechanical properties of human and bovine dentin. Clin. Oral Investig. 2021, 25, 841–849. [Google Scholar] [CrossRef]
- Hong, L.; Ettinger, R.L.; Watkins, C.A.; Wefel, J.S. In vitro evaluation of fluoride varnish on overdenture abuments. J. Prosthet. Dent. 2003, 89, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Darling, L.A.; Ettinger, R.L.; Wefel, J.S.; Cooper, S.H. Prevention of demineralization by CO2 and Er, Cr:YSGG laser irradiation of ovedenture abutments. Am. J. Dent. 2006, 19, 227–230. [Google Scholar]
- Salehi, H.; Terrer, E.; Panayotov, I.; Levallois, B.; Jacquot, B.; Tassery, H.; Cuisinier, F. Functional mapping of human sound and carious enamel and dentin with Raman spectroscopy. J. Biophotonics 2013, 6, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Tjäderhane, L.; Larjava, H.; Sorsa, T.; Uitto, V.J.; Larmas, M.; Salo, T. The activation and function of host matrix metalloproteinases in dentin matrix breakdown in caries lesions. J. Dent. Res. 1998, 77, 1622–1629. [Google Scholar] [CrossRef]
- Chaussain-Miller, C.; Fioretti, F.; Goldberg, M.; Menashi, S. The role of matrix metalloproteinases (MMPs) in human caries. J. Dent. Res. 2006, 85, 22–32. [Google Scholar] [CrossRef]
- Tsuda, H.; Ruben, J.; Arends, J. Raman spectra of human dentin mineral. Eur. J. Oral Sci. 1996, 104, 123–131. [Google Scholar] [CrossRef]
- Farlay, D.; Panczer, G.; Rey, C.; Delmas, P.D.; Boivin, G. Mineral maturity and crystallinity index are distinct characteristics of bone mineral. J. Bone Miner. Metab. 2010, 28, 433–445. [Google Scholar] [CrossRef] [Green Version]
- Lin, S.Y.; Li, M.J.; Cheng, W.T. FT-IR and Raman vibrational microspectroscopies used for spectral biodiagnosis of human tissues. Spectroscopy 2007, 21, 1–30. [Google Scholar] [CrossRef]
- Rey, C.; Collins, B.; Goehl, T.; Dickson, I.R.; Glimcher, M.J. The carbonate environment in bone mineral: A resolution-enhanced Fourier transform infrared spectroscopy study. Calcif. Tissue Int. 1989, 45, 157–164. [Google Scholar] [CrossRef]
- Boskey, A.L. Mineralization of bones and teeth. Elements 2007, 3, 385–391. [Google Scholar] [CrossRef]
- Dowker, S.E.P.; Anderson, P.; Elliot, J.C.; Gao, X.J. Crystal chemistry and dissolution of calcium phosphate in dental enamel. Mineral. Mag. 1999, 63, 791–800. [Google Scholar] [CrossRef]
- Danilchenko, S.N.; Moseke, C.; Sukhodub, L.F.; Sulkio-Cleff, B. X-ray diffraction studies of bone apatite under acid demineralization. Cryst. Res. Technol. 2004, 39, 71–77. [Google Scholar] [CrossRef]
- Berna, F.; Matthews, A.; Weiner, S. Solubilities of bone mineral from archaeological sites: The recrystallization window. J. Archaeol. Sci. 2004, 31, 867–882. [Google Scholar] [CrossRef]
- Dominguez-Gasca, N.; Benavides-Reyes, C.; Sánchez-Rodríguez, E.; Rodríguez-Navarro, A.B. Changes in avian cortical and medullary bone mineral composition and organization during acid-induced demineralization. Eur. J. Mineral. 2019, 31, 209–216. [Google Scholar] [CrossRef]
- Hiraishi, N.; Sono, R.; Islam, M.S.; Otsuki, M.; Tagami, J.; Takatsuka, T. Effect of hesperidin in vitro on root dentine collagen and demineralization. J. Dent. 2011, 39, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Sereda, G.; VanLaecken, A.; Turner, J.A. Monitoring demineralization and remineralization of human dentin by characterization of its structure with resonance-enhanced AFM-IR chemical mapping, nanoindentation, and SEM. Dent. Mater. 2019, 35, 617–626. [Google Scholar] [CrossRef]
- Erhardt, M.C.; Lobo, M.M.; Goulart, M.; Coelho-de-Souza, F.H.; Valentino, T.A.; Pisani-Proenca, J.; Conceicao, E.N.; Pimenta, L.A. Microtensile bond strength of etch-and-rinse and self-etch adhesives to artificially created carious dentin. Gen. Dent. 2014, 62, 56–61. [Google Scholar]
- Kucukyilmaz, E.; Savas, S.; Akcay, M.; Bolukbasi, B. Effect of silver diamine fluoride and ammonium hexafluorosilicate applications with and without Er: YAG laser irradiation on the microtensile bond strength in sound and caries-affected dentin. Lasers Surg. Med. 2016, 48, 62–69. [Google Scholar] [CrossRef]
- Fathy, S.M. Remineralization ability of two hydraulic calcium-silicate based dental pulp capping materials: Cell-independent model. J. Clin. Exp. Dent. 2019, 11, e360–e366. [Google Scholar] [CrossRef] [PubMed]
- Shivakumar, K.M.; Vidya, S.K.; Chandu, G.N. Dental caries vaccine. Indian J. Dent. Res. 2009, 20, 99–106. [Google Scholar] [CrossRef] [PubMed]

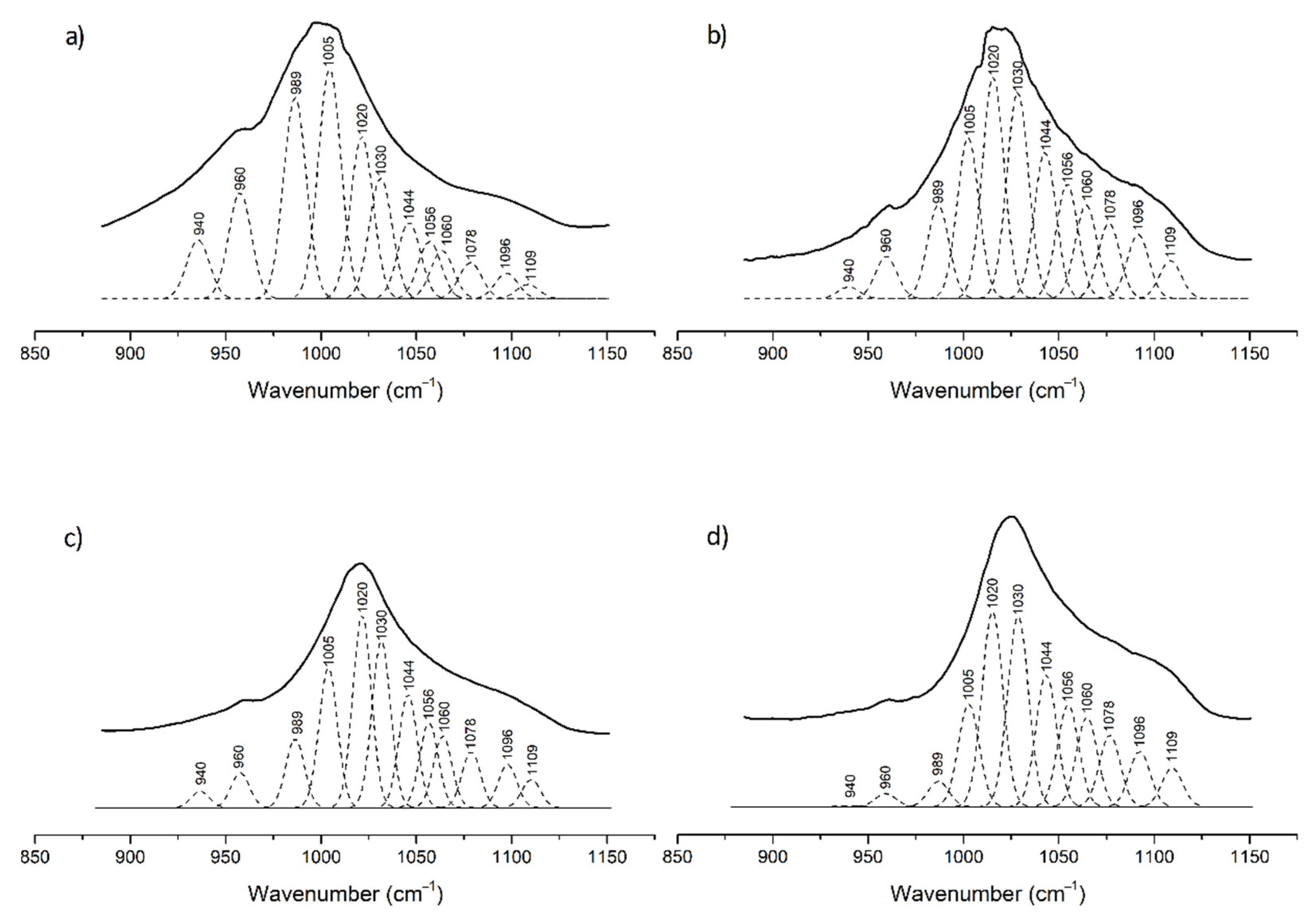



| Wavenumber (cm−1) | Sound (n = 10) | Caries (n = 10) | pHc 14 (n = 10) | pHc 28 (n = 10) |
|---|---|---|---|---|
| P1_CO3_a866 | 0.386 ± (0.073) A | 0.248 ± (0.072) B | 0.227 ± (0.047) B | 0.197 ± (0.075) B |
| P2_CO3_a871 | 0.474 ± (0.068) A | 0.458 ± (0.115) A | 0.457 ± (0.039) A | 0.449 ± (0.051) A |
| P3_CO3_a878 | 0.140 ± (0.029) A | 0.294 ± (0.083) B | 0.316 ± (0.052) B | 0.353 ± (0.068) B |
| P1_PO4_a940 | 0.080 ± (0.019) A | 0.006 ± (0.007) B | 0.012 ± (0.004) B | 0.009 ± (0.005) B |
| P2_PO4_a960 | 0.129 ± (0.014) A | 0.031 ± (0.019) B | 0.037 ± (0.008) B | 0.030 ± (0.009) B |
| P3_PO4_a996 | 0.205 ± (0.021) A | 0.056 ± (0.036) B | 0.062 ± (0.014) B | 0.050 ± (0.018) B |
| P4_PO4_a1005 | 0.192 ± (0.037) A | 0.146 ± (0.037) A.B | 0.144 ± (0.013) B | 0.134 ± (0.015) B |
| P5_PO4_a1020 | 0.131 ± (0.016) A | 0.223 ± (0.027) B | 0.227 ± (0.028) B | 0.231 ± (0.020) B |
| P6_PO4_a1030 | 0.085 ± (0.008) A | 0.186 ± (0.030) B | 0.176 ± (0.012) B | 0.183 ± (0.012) B |
| P7_PO4_a1044 | 0.047 ± (0.011) A | 0.099 ± (0.020) B | 0.094 ± (0.007) B | 0.100 ± (0.008) B |
| P8_PO4_a1056 | 0.031 ± (0.009) A | 0.060 ± (0.013) B | 0.059 ± (0.007) B | 0.060 ± (0.008) B |
| P9_PO4_a1060 | 0.028 ± (0.009) A | 0.052 ± (0.012) B | 0.054 ± (0.008) B | 0.056 ± (0.007) B |
| P10_PO4_a1078 | 0.032 ± (0.007) A | 0.052 ± (0.010) B | 0.058 ± (0.010) B | 0.061 ± (0.008) B |
| P11_PO4_a1096 | 0.026 ± (0.004) A | 0.053 ± (0.010) B | 0.047 ± (0.004) B | 0.050 ± (0.006) B |
| P12_PO4_a1109 | 0.017 ± (0.005) A | 0.037 ± (0.009) B | 0.031 ± (0.009) B | 0.036 ± (0.006) B |
| Group | Element | Weight (%) |
|---|---|---|
| Sound dentine | Ca | 27.82 |
| P | 14.10 | |
| Caries group | Ca | 17.40 |
| P | 8.87 | |
| pHc 14 | Ca | 23.08 |
| P | 11.68 | |
| pHc 28 | Ca | 22.78 |
| P | 11.36 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuluaga-Morales, J.S.; Bolaños-Carmona, M.V.; Cifuentes-Jiménez, C.C.; Álvarez-Lloret, P. Chemical, Microstructural and Morphological Characterisation of Dentine Caries Simulation by pH-Cycling. Minerals 2022, 12, 5. https://doi.org/10.3390/min12010005
Zuluaga-Morales JS, Bolaños-Carmona MV, Cifuentes-Jiménez CC, Álvarez-Lloret P. Chemical, Microstructural and Morphological Characterisation of Dentine Caries Simulation by pH-Cycling. Minerals. 2022; 12(1):5. https://doi.org/10.3390/min12010005
Chicago/Turabian StyleZuluaga-Morales, Juan Sebastián, María Victoria Bolaños-Carmona, Carolina Cecilia Cifuentes-Jiménez, and Pedro Álvarez-Lloret. 2022. "Chemical, Microstructural and Morphological Characterisation of Dentine Caries Simulation by pH-Cycling" Minerals 12, no. 1: 5. https://doi.org/10.3390/min12010005
APA StyleZuluaga-Morales, J. S., Bolaños-Carmona, M. V., Cifuentes-Jiménez, C. C., & Álvarez-Lloret, P. (2022). Chemical, Microstructural and Morphological Characterisation of Dentine Caries Simulation by pH-Cycling. Minerals, 12(1), 5. https://doi.org/10.3390/min12010005






