Emerging CO2-Mineralization Technologies for Co-Utilization of Industrial Solid Waste and Carbon Resources in China
Abstract
1. Introduction
2. Materials and Methods
2.1. Large-Scale ISW in China
2.2. Process Intensification
2.3. Valuable Chemicals Produced
3. Current Status of Pertinent Policies and Research and Development Activities
3.1. China’s Policies of Carbon Caputre, Utilization, and Storage
3.2. Demonstration Projects of CO2 Mineralization Via ISW in China
4. Current Knowledge Gaps
4.1. The Importance of Assessment of CO2 Mineralization Technologies
4.2. Understanding of the Sustainability of CO2 Mineralization Technologies
4.2.1. Technical Feasibility
4.2.2. Economic Feasibility
4.2.3. Environment Impacts and Risks
4.3. Cooperation between Different Stakeholders
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
- Cao, W.; Yang, Q. The properties of the carbonated brick made of steel slag-slaked lime mixture.
- Chen, Q.; Ding, W.; Sun, H.; Peng, T. Mineral carbonation of yellow phosphorus slag and characterization of carbonated product.
- Ding, W.; Chen, Q.; Sun, H.; Peng, T. Modified mineral carbonation of phosphogypsum for CO2 sequestration.
- Gan, Z.; Cui, Z.; Yue, H.; Tang, S.; Liu, C.; Li, C.; Liang, B.; Xie, H. An efficient methodology for utilization of K-feldspar and phosphogypsum with reduced energy consumption and CO2 emissions.
- Gao, J.; Li, C.; Liu, W.; Hu, J.; Wang, L.; Liu, Q.; Liang, B.; Yue, H.; Zhang, G.; Luo, D.; et al. Process simulation and energy integration in the mineral carbonation of blast furnace slag.
- Hu, J.; Liu, W.; Wang, L.; Liu, Q.; Chen, F.; Yue, H.; Liang, B.; Lu, L.; Wang, Y.; Zhang, G.; et al. Indirect mineral carbonation of blast furnace slag with (NH4)(2)SO4 as a recyclable extractant.
- Ji, L.; Yu, H.; Wang, X.; Grigore, M.; French, D.; Gozukara, Y.; Yu, J.; Zeng, M. CO2 sequestration by direct mineralisation using fly ash from Chinese Shenfu coal.
- Ji, L.; Yu, H.; Yu, B.; Jiang, K.; Grigore, M.; Wang, X.; Zhao, S.; Li, K. Integrated absorption–mineralisation for energy-efficient CO2 sequestration: Reaction mechanism and feasibility of using fly ash as a feedstock.
- Ji, L.; Yu, H.; Zhang, R.; French, D.; Grigore, M.; Yu, B.; Wang, X.; Yu, J.; Zhao, S. Effects of fly ash properties on carbonation efficiency in CO2 mineralisation.
- Jiang, J.; Tian, S.; Zhang, C. Influence of SO2 in incineration flue gas on the sequestration of CO2 by municipal solid waste incinerator fly ash.
- Liu, W.; Su, S.; Xu, K.; Chen, Q.; Xu, J.; Sun, Z.; Wang, Y.; Hu, S.; Wang, X.; Xue, Y.; et al. CO2 sequestration by direct gas-solid carbonation of fly ash with steam addition.
- Luo, C.; Wu, K.; Yue, H.; Liu, Y.; Zhu, Y.; Jiang, W.; Lu, H.; Liang, B. DBU-based CO2 absorption–mineralization system: Reaction process, feasibility and process intensification.
- Ni, P.; Xiong, Z.; Tian, C.; Li, H.; Zhao, Y.; Zhang, J.; Zheng, C. Influence of carbonation under oxy-fuel combustion flue gas on the leachability of heavy metals in MSWI fly ash.
- Shangguan, W.; Song, J.; Yue, H.; Tang, S.; Liu, C.; Li, C.; Liang, B.; Xie, H. An efficient milling-assisted technology for K-feldspar processing, industrial waste treatment and CO2 mineralization.
- Shen, W.; Liu, Y.; Wu, M.; Zhang, D.; Du, X.; Zhao, D.; Xu, G.; Zhang, B.; Xiong, X. Ecological carbonated steel slag pervious concrete prepared as a key material of sponge city.
- Tan, W.; Gu, S.; Xia, W.; Li, Y.; Zhang, Z. Feature changes of mercury during the carbonation of FGD gypsum from different sources.
- Tian, T.; Yan, Y.; Hu, Z.; Xu, Y.; Chen, Y.; Shi, J. Utilization of original phosphogypsum for the preparation of foam concrete.
- Wang, L.; Chen, Q.; Jamro, I.A.; Li, R.; Li, Y.; Li, S.; Luan, J. Geochemical modeling and assessment of leaching from carbonated municipal solid waste incinerator (MSWI) fly ash.
- Wang, L.; Liu, W.; Hu, J.; Liu, Q.; Yue, H.; Liang, B.; Zhang, G.; Luo, D.; Xie, H.; Li, C. Indirect mineral carbonation of titanium-bearing blast furnace slag coupled with recovery of TiO2 and Al2O3.
- Wang, P.; Mao, X.; Chen, S.-E. CO2 sequestration characteristics in the cementitious material based on gangue backfilling mining method.
- Xie, H.; Tang, L.; Wang, Y.; Liu, T.; Hou, Z.; Wang, J.; Wang, T.; Jiang, W.; Were, P. Feedstocks study on CO2 mineralization technology.
- Xiong, Y.; Aldahri, T.; Liu, W.; Chu, G.; Zhang, G.; Luo, D.; Yue, H.; Liang, B.; Li, C. Simultaneous preparation of TiO2 and ammonium alum, and microporous SiO2 during the mineral carbonation of titanium-bearing blast furnace slag.
- Yi, H.; Xu, G.; Cheng, H.; Wang, J.; Wan, Y.; Chen, H. An overview of utilization of steel slag.
- Zhang, H.; Dong, L.; Li, H.; Fujita, T.; Ohnishi, S.; Tang, Q. Analysis of low-carbon industrial symbiosis technology for carbon mitigation in a Chinese iron/steel industrial park.
- Zhao, Q.; Liu, C.; Gao, T.; Gao, L.; Saxen, H.; Zevenhoven, R. Remediation of stainless steel slag with MnO for CO2 mineralization.
- Zhao, Q.; Liu, K.; Sun, L.; Liu, C.; Jiang, M.; Saxen, H.; Zevenhoven, R. Towards carbon sequestration using stainless steel slag via phase modification and co-extraction of calcium and magnesium.
- Chen, Q.; Ding, W.; Sun, H.; Peng, T.; Ma, G. Utilization of Phosphogypsum to Prepare High-Purity CaCO3 in the NH4Cl–NH4OH–CO2 System.
- Chen, Q.J.; Ding, W.J.; Sun, H.J.; Peng, T.J.; Ma, G.H. Indirect mineral carbonation of phosphogypsum for CO2 sequestration.
References
- Seifritz, W. CO2 disposal by means of silicates. Nature 1990, 345, 486. [Google Scholar] [CrossRef]
- Intergovernmental Panel on Climate Change (IPCC). Special Report on Carbon Dioxide Capture and Storage; Cambridge University Press: Cambridge, UK, 2005; ISBN 92-9169-1190-4.
- Romanov, V.; Soong, Y.; Carney, C.; Rush, G.E.; Nielsen, B.; O’Connor, W. Mineralization of Carbon Dioxide: A Literature Review. Chembioeng. Rev. 2015, 2, 231–256. [Google Scholar] [CrossRef]
- Lackner, K.S.; Wendt, C.H.; Butt, D.P.; Joyce, E.L.; Sharp, D.H. Carbon dioxide disposal in carbonate minerals. Energy 1995, 20, 1153–1170. [Google Scholar] [CrossRef]
- Moazzem, S.; Rasul, M.G.; Khan, M.M.K. Energy recovery opportunities from mineral carbonation process in coal fired power plant. Appl. Therm. Eng. 2013, 51, 281–291. [Google Scholar] [CrossRef]
- Teir, S.; Kuusik, R.; Fogelholm, C.-J.; Zevenhoven, R. Production of magnesium carbonates from serpentinite for long-term storage of CO2. Int. J. Miner. Process. 2007, 85, 1–15. [Google Scholar] [CrossRef]
- Nduagu, E.; Fagerlund, J.; Zevenhoven, R. Contribution of iron to the energetics of CO2 sequestration in Mg–silicates-based rock. Energy Convers. Manag. 2012, 55, 178–186. [Google Scholar] [CrossRef]
- Kremer, D.; Wotruba, H. Separation of Products from Mineral Sequestration of CO2 with Primary and Secondary Raw Materials. Minerals 2020, 10, 1098. [Google Scholar] [CrossRef]
- Tai, C.Y.; Chen, W.R.; Shih, S.-M. Factors affecting wollastonite carbonation under CO2 supercritical conditions. Aiche J. 2006, 52, 292–299. [Google Scholar] [CrossRef]
- Ayub, S.A.; Tsegab, H.; Rahmani, O.; Beiranvand Pour, A. Potential for CO2 Mineral Carbonation in the Paleogene Segamat Basalt of Malaysia. Minerals 2020, 10, 1045. [Google Scholar] [CrossRef]
- Xie, H.; Tang, L.; Wang, Y.; Liu, T.; Hou, Z.; Wang, J.; Wang, T.; Jiang, W.; Were, P. Feedstocks study on CO2 mineralization technology. Environ. Earth Sci. 2016, 75. [Google Scholar] [CrossRef]
- Reynolds, B.; Reddy, K.J.; Argyle, M.D. Field Application of Accelerated Mineral Carbonation. Minerals 2014, 4, 191. [Google Scholar] [CrossRef]
- Bauer, M.; Gassen, N.; Stanjek, H.; Peiffer, S. Carbonation of lignite fly ash at ambient T and P in a semi-dry reaction system for CO2 sequestration. Appl. Geochem. 2011, 26, 1502–1512. [Google Scholar] [CrossRef]
- Eloneva, S.; Puheloinen, E.-M.; Kanerva, J.; Ekroos, A.; Zevenhoven, R.; Fogelholm, C.-J. Co-utilisation of CO2 and steelmaking slags for production of pure CaCO3—Legislative issues. J. Clean. Prod. 2010, 18, 1833–1839. [Google Scholar] [CrossRef]
- Chang, E.E.; Pan, S.Y.; Chen, Y.H.; Tan, C.S.; Chiang, P.C. Accelerated carbonation of steelmaking slags in a high-gravity rotating packed bed. J. Hazard. Mater. 2012, 227–228, 97–106. [Google Scholar] [CrossRef]
- Liu, R.; Wang, X.; Gao, S. CO2 capture and mineralization using carbide slag doped fly ash. Greenhouse Gases. Sci. Technol. 2019, 10. [Google Scholar] [CrossRef]
- Gan, Z.; Cui, Z.; Yue, H.; Tang, S.; Liu, C.; Li, C.; Liang, B.; Xie, H. An efficient methodology for utilization of K-feldspar and phosphogypsum with reduced energy consumption and CO2 emissions. Chin. J. Chem. Eng. 2016, 24, 1541–1551. [Google Scholar] [CrossRef]
- Ding, W.; Chen, Q.; Sun, H.; Peng, T. Modified mineral carbonation of phosphogypsum for CO2 sequestration. J. CO2 Util. 2019, 34, 507–515. [Google Scholar] [CrossRef]
- Power, I.M.; McCutcheon, J.; Harrison, A.L.; Wilson, S.A.; Dipple, G.M.; Kelly, S.; Southam, C.; Southam, G. Strategizing Carbon-Neutral Mines: A Case for Pilot Projects. Minerals 2014, 4, 399. [Google Scholar] [CrossRef]
- Yang, H.; Xu, Z.; Fan, M.; Gupta, R.; Slimane, R.B.; Bland, A.E.; Wright, I. Progress in carbon dioxide separation and capture: A review. J. Environ. Sci. 2008, 20, 14–27. [Google Scholar] [CrossRef]
- Bobicki, E.R.; Liu, Q.; Xu, Z.; Zeng, H. Carbon capture and storage using alkaline industrial wastes. Prog. Energy Combust. Sci. 2012, 38, 302–320. [Google Scholar] [CrossRef]
- Ministry of Ecology and Environment of the People’s Republic of China. 2019 China Ecological Environment Bulletin; 2020. Available online: http://www.mee.gov.cn/hjzl/sthjzk/ (accessed on 30 January 2021).
- Qi, Y.; Stern, N.; Wu, T.; Lu, J.; Green, F. China’s post-coal growth. Nat. Geosci. 2016, 9, 564–566. [Google Scholar] [CrossRef]
- Friedlingstein, P.; O’Sullivan, M.; Jones, M.W.; Andrew, R.M.; Hauck, J.; Olsen, A.; Peters, G.P.; Peters, W.; Pongratz, J.; Sitch, S.; et al. Global Carbon Budget 2020. Earth Syst. Sci. Data 2020, 12, 3269–3340. [Google Scholar] [CrossRef]
- Ministry of Ecology and Environment of the People’s Republic of China. China Ecological Environment Statistics Bulletin 2016–2019; 2020. Available online: http://www.mee.gov.cn/hjzl/sthjzk/sthjtjnb/202012/P020201214580320276493.pdf (accessed on 7 February 2021).
- Ministry of Ecology and Environment of the People’s Republic of China. 2020 Annual Report on Prevention and Control of Solid Waste Pollution in China’s Large and Medium Cities; 2020. Available online: http://www.mee.gov.cn/hjzl/sthjzk/gtfwwrfz/ (accessed on 30 January 2021).
- Ahmaruzzaman, M. A review on the utilization of fly ash. Prog. Energy Combust. Sci. 2010, 36, 327–363. [Google Scholar] [CrossRef]
- Gomes, H.I.; Mayes, W.M.; Rogerson, M.; Stewart, D.I.; Burke, I.T. Alkaline residues and the environment: A review of impacts, management practices and opportunities. J. Clean. Prod. 2016, 112, 3571–3582. [Google Scholar] [CrossRef]
- Guo, J.; Bao, Y.; Wang, M. Steel slag in China: Treatment, recycling, and management. Waste Manag. 2018, 78, 318–330. [Google Scholar] [CrossRef]
- Steel Statistical Yearbook 2020 Concise Version. Available online: https://www.worldsteel.org/en/dam/jcr:5001dac8-0083-46f3-aadd-35aa357acbcc/SSY%25202020_concise%2520version.pdf (accessed on 27 January 2021).
- Shi, C. Steel Slag—Its Production, Processing, Characteristics, and Cementitious Properties. J. Mater. Civ. Eng. 2004, 16, 230–236. [Google Scholar] [CrossRef]
- Pan, S.-Y.; Chung, T.-C.; Ho, C.-C.; Hou, C.-J.; Chen, Y.-H.; Chiang, P.-C. CO2 Mineralization and Utilization using Steel Slag for Establishing a Waste-to-Resource Supply Chain. Sci. Rep. 2017, 7, 17227. [Google Scholar] [CrossRef]
- Değirmenci, N. Utilization of phosphogypsum as raw and calcined material in manufacturing of building products. Constr. Build. Mater. 2008, 22, 1857–1862. [Google Scholar] [CrossRef]
- Kandil, A.-H.T.; Cheira, M.F.; Gado, H.S.; Soliman, M.H.; Akl, H.M. Ammonium sulfate preparation from phosphogypsum waste. J. Radiat. Res. Appl. Sci. 2017, 10, 24–33. [Google Scholar] [CrossRef]
- Oge, M.; Ozkan, D.; Celik, M.B.; Sabri Gok, M.; Cahit Karaoglanli, A. An Overview of Utilization of Blast Furnace and Steelmaking Slag in Various Applications. Mater. Today Proc. 2019, 11, 516–525. [Google Scholar] [CrossRef]
- Ma, X.; Li, Y.; Qian, Y.; Wang, Z. A Carbide Slag-Based, Ca12Al14O33-Stabilized Sorbent Prepared by the Hydrothermal Template Method Enabling Efficient CO2 Capture. Energies 2019, 12, 2617. [Google Scholar] [CrossRef]
- Wang, N.; Mao, M.; Mao, G.; Yin, J.; He, R.; Zhou, H.; Li, N.; Liu, Q.; Zhi, K. Investigation on carbide slag catalytic effect of Mongolian bituminous coal steam gasification process. Chemosphere 2021, 264, 128500. [Google Scholar] [CrossRef]
- Ibrahim, M.H.; El-Naas, M.H.; Benamor, A.; Al-Sobhi, S.S.; Zhang, Z. Carbon Mineralization by Reaction with Steel-Making Waste: A Review. Processes 2019, 7, 115. [Google Scholar] [CrossRef]
- Yongfeng, N. Three Waste Treatment Engineering Technical Manual; Solid Waste Volume; Chemical Industry Press Co., Ltd.: Beijing, China, 2000; ISBN 7-5025-2718-4. [Google Scholar]
- Zhang, D.; Gao, H.; Xiao, C. Recycling Technology of Smelting Waste Residue; Chemical Industry Press Co., Ltd.: Beijing, China, 2018; ISBN 978-7-122-29560-6. [Google Scholar]
- Mingxia, D.; Jinming, W.; Faqin, D.; Zhaojia, W.; Feihua, Y.; Kaibin, F.; Yu, L. Technological Mineralogical Characteristics of Phosphogypsum. Non-Met. Mines 2020, 43, 52–55. (In Chinese) [Google Scholar]
- Xie, H.; Yue, H.; Zhu, J.; Liang, B.; Li, C.; Wang, Y.; Xie, L.; Zhou, X. Scientific and Engineering Progress in CO2 Mineralization Using Industrial Waste and Natural Minerals. Engineering 2015, 1, 150–157. [Google Scholar] [CrossRef]
- Li, Q.; Chen, Z.A.; Zhang, J.T.; Liu, L.C.; Li, X.C.; Jia, L. Positioning and revision of CCUS technology development in China. Int. J. Greenh. Gas Control 2016, 46, 282–293. [Google Scholar] [CrossRef]
- Jiang, K.; Ashworth, P.; Zhang, S.; Liang, X.; Sun, Y.; Angus, D. China’s carbon capture, utilization and storage (CCUS) policy: A critical review. Renew. Sustain. Energy Rev. 2020, 119, 109601. [Google Scholar] [CrossRef]
- LI, B.; Bao, W.; Zheng, Y.; Zheng, K.; Tan, W. Pretreatment technology for phosphogypsum purification. Phosphate Compd. Fertil. 2018, 33, 28–31. (In Chinese) [Google Scholar]
- Guo, R.; Chen, Q.; Huang, H.; Hu, X.; Wang, T. Carbonation curing of industrial solid waste-based aerated concretes. Greenh. Gases Sci. Technol. 2019, 9, 433–443. [Google Scholar] [CrossRef]
- Ministry of Science and Technology of the People’s Republic of China. Roadmap for Carbon Capture, Utilization and Storage Technology Development in China; China Science Publishing and Media Ltd. (CSPM): Beijing, China, 2019; ISBN 978-7-030-62018-7.
- Yang, L.; Xu, M.; Yang, Y.; Fan, J.; Zhang, X. Comparison of subsidy schemes for carbon capture utilization and storage (CCUS) investment based on real option approach: Evidence from China. Appl. Energy 2019, 255, 113828. [Google Scholar] [CrossRef]
- Yu, S.; Horing, J.; Liu, Q.; Dahowski, R.; Davidson, C.; Edmonds, J.; Liu, B.; McJeon, H.; McLeod, J.; Patel, P.; et al. CCUS in China’s mitigation strategy: Insights from integrated assessment modeling. Int. J. Greenh. Gas Control 2019, 84, 204–218. [Google Scholar] [CrossRef]
- Hillebrand, M.; Pflugmacher, S.; Hahn, A. Toxicological risk assessment in CO2 capture and storage technology. Int. J. Greenh. Gas Control 2016, 55, 118–143. [Google Scholar] [CrossRef]
- Yao, X.; Zhong, P.; Zhang, X.; Zhu, L. Business model design for the carbon capture utilization and storage (CCUS) project in China. Energy Policy 2018, 121, 519–533. [Google Scholar] [CrossRef]
- Miller, J. Building a better dialogue between energy research and policy. Nat. Energy 2019, 4, 816–818. [Google Scholar] [CrossRef]
- Li, Q.; Liu, G.; Cai, B.; Leamon, G.; Liu, L.-C.; Chen, Z.-A.; Li, X. Public awareness of the environmental impact and management of carbon dioxide capture, utilization and storage technology: The views of educated people in China. Clean Technol. Environ. Policy 2017, 19, 2041–2056. [Google Scholar] [CrossRef]

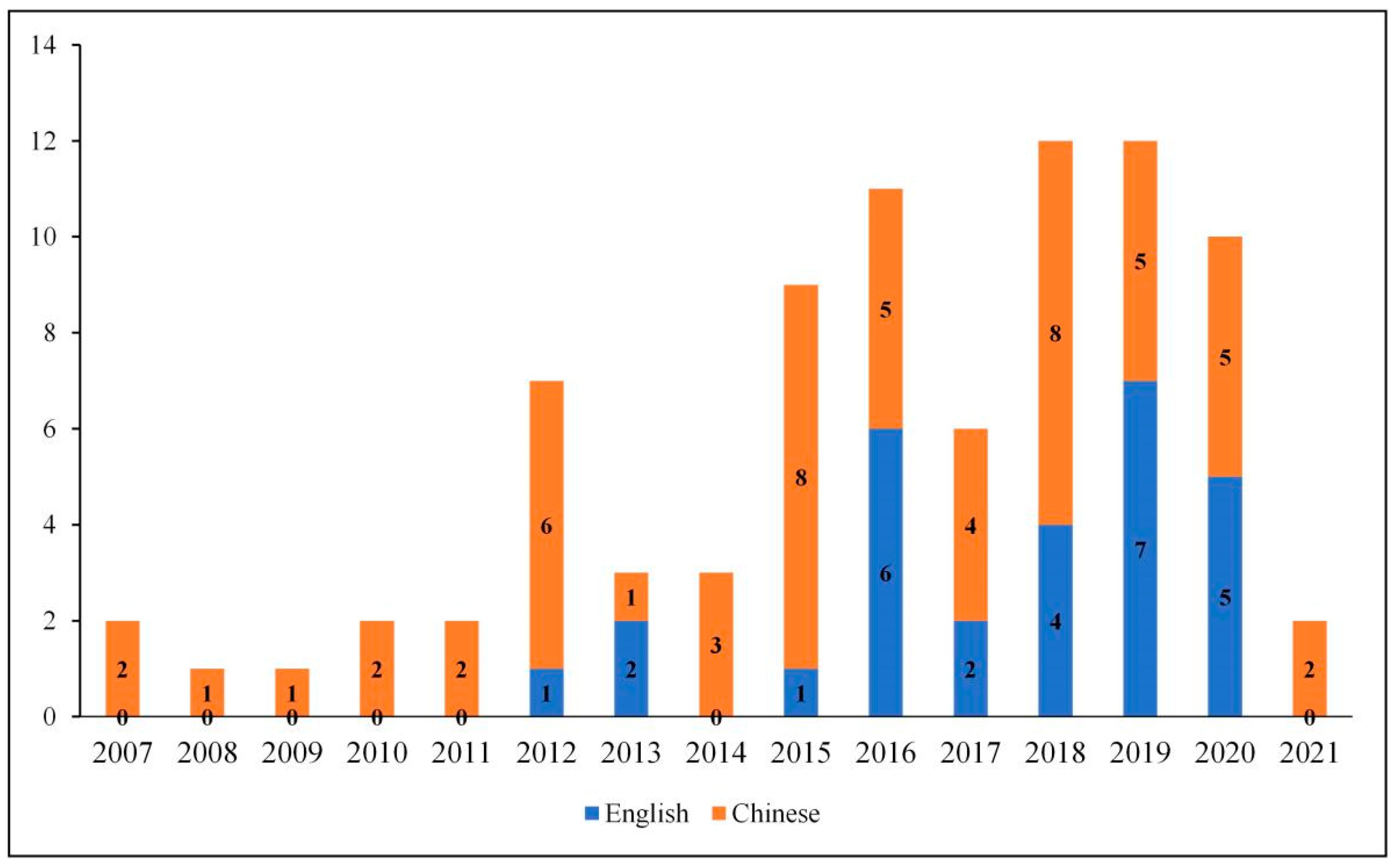
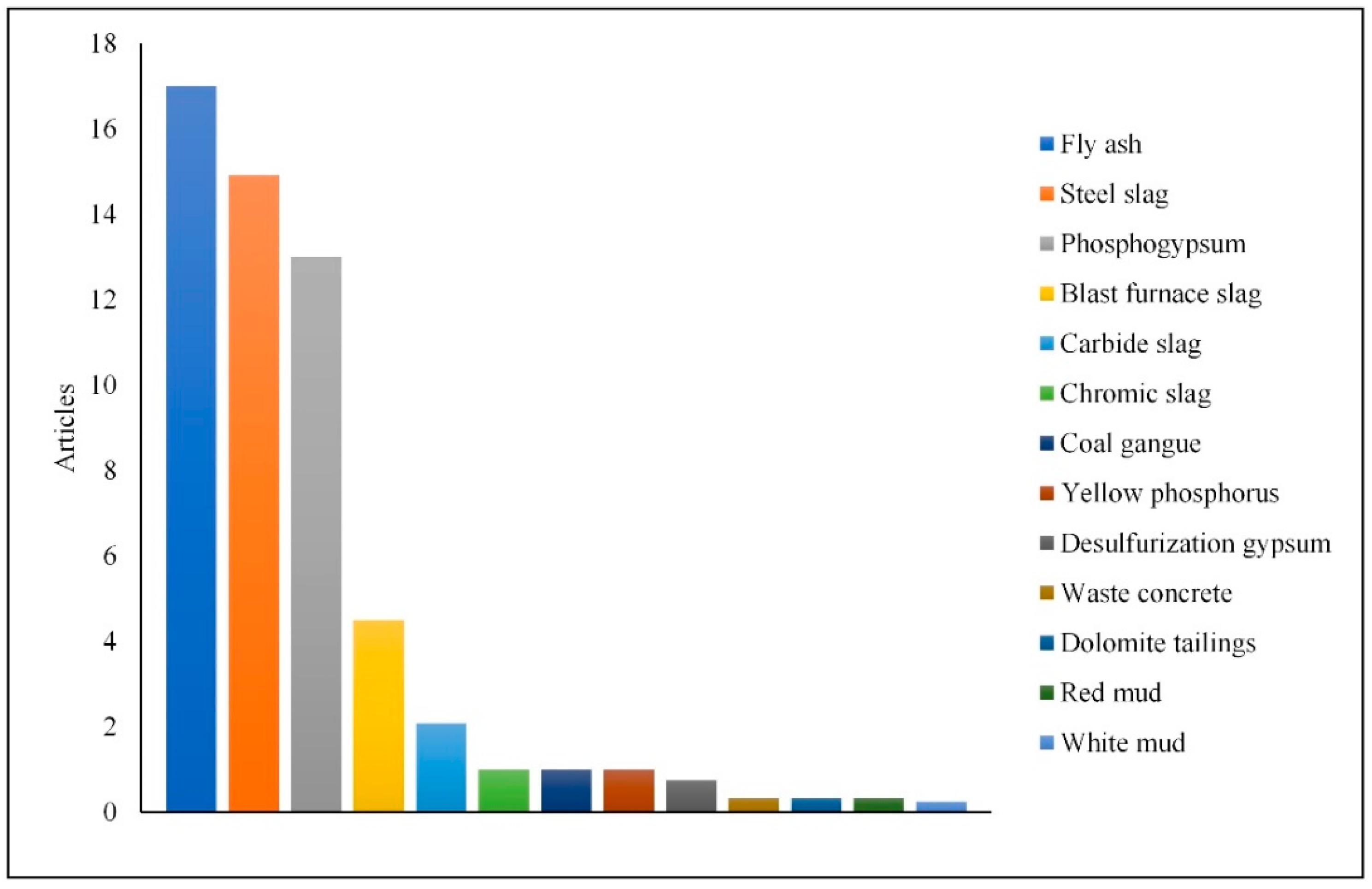

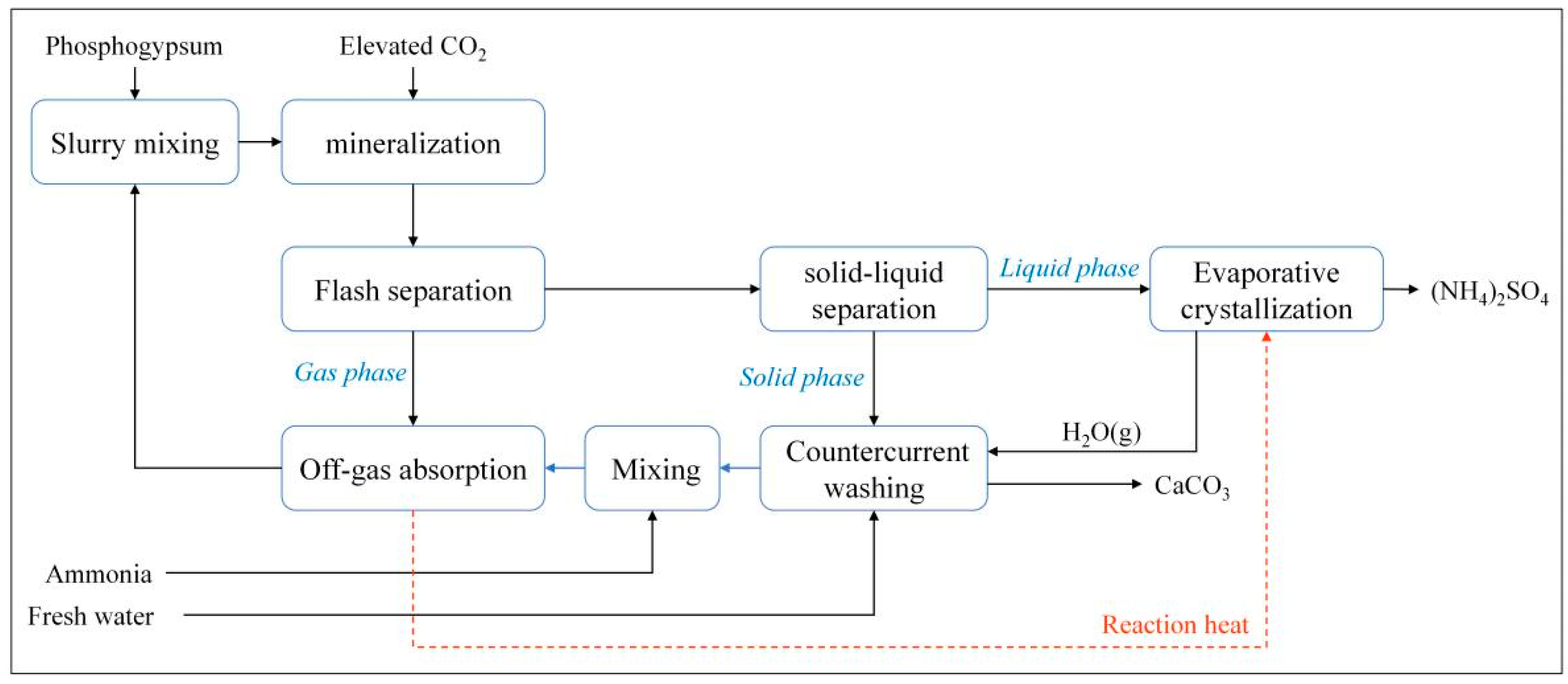

| Inclusive Criteria | Exclusion Criteria |
|---|---|
| Focusing on Chinese CO2 mineralization | Is not related to industrial solid waste (ISW) |
| Is a peer-reviewed journal article |
| Raw Materials | Alkalinity Content (%) 1 | Annual Production (million tons) | CO2 Fixation Efficiency (t CO2/t slag) | Theoretical Fixation Capacity (million tons) | |||
|---|---|---|---|---|---|---|---|
| CaO | MgO | Low | High | Low | High | ||
| Coal fly ash | 1.5–5.5 | 0.6–2.0 | 540 | 0.02 | 0.07 | 0.10 | 0.35 |
| Steel slag | 40–60 | 5–15 | 125 | 0.37 | 0.64 | 0.46 | 0.80 |
| Phosphogypsum | 25.54–42.87 | 0.1–0.16 | 75 | 0.20 | 0.34 | 0.15 | 0.25 |
| Blast furnace slag | 38–39 | 1–13 | 320 | 0.31 | 0.45 | 0.99 | 1.44 |
| Carbide slag | 65–71 | 0.22–1.68 | 40 | 0.51 | 0.58 | 0.21 | 0.23 |
| Cooperative Units | Raw Material | Source of CO2 | Year | Products |
|---|---|---|---|---|
| Sinopec Group and Sichuan University | Phosphogypsum | Natural gas purification plant | 2013 | CaCO3, (NH4)2SO4 |
| Sinochem Fuling Chongqing Chemical Industry Co., Ltd. and Chinese Academy of Sciences | Phosphogypsum | Ammonia synthesis plant | 2017 | CaCO3, (NH4)2SO4 |
| Yuanchu Technology Corp. and Tsinghua University | Silicate ore, construction solid waste | Cement plant | 2018 | Light-weight CaCO3, Silica |
| Henan Qiangnai New Materials Co., Ltd. and Zhejiang University | Steel slag, fly ash | Chemical plant | 2020 | Concrete brick |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, J.; Liao, W.; Zhang, G. Emerging CO2-Mineralization Technologies for Co-Utilization of Industrial Solid Waste and Carbon Resources in China. Minerals 2021, 11, 274. https://doi.org/10.3390/min11030274
Meng J, Liao W, Zhang G. Emerging CO2-Mineralization Technologies for Co-Utilization of Industrial Solid Waste and Carbon Resources in China. Minerals. 2021; 11(3):274. https://doi.org/10.3390/min11030274
Chicago/Turabian StyleMeng, Junlin, Wenjie Liao, and Guoquan Zhang. 2021. "Emerging CO2-Mineralization Technologies for Co-Utilization of Industrial Solid Waste and Carbon Resources in China" Minerals 11, no. 3: 274. https://doi.org/10.3390/min11030274
APA StyleMeng, J., Liao, W., & Zhang, G. (2021). Emerging CO2-Mineralization Technologies for Co-Utilization of Industrial Solid Waste and Carbon Resources in China. Minerals, 11(3), 274. https://doi.org/10.3390/min11030274








