Gallium-Containing Hydroxyapatite as a Promising Material for Photocatalytic Performance
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Synthesis of Ga-Containing Hydroxyapatite
Ca9.95Ga0.05(PO4)6(OH)2↓ + 18H2O(l) + 12NH3(g)
2.3. Characterization
2.4. Photocatalytic Experiments
3. Results and Discussion
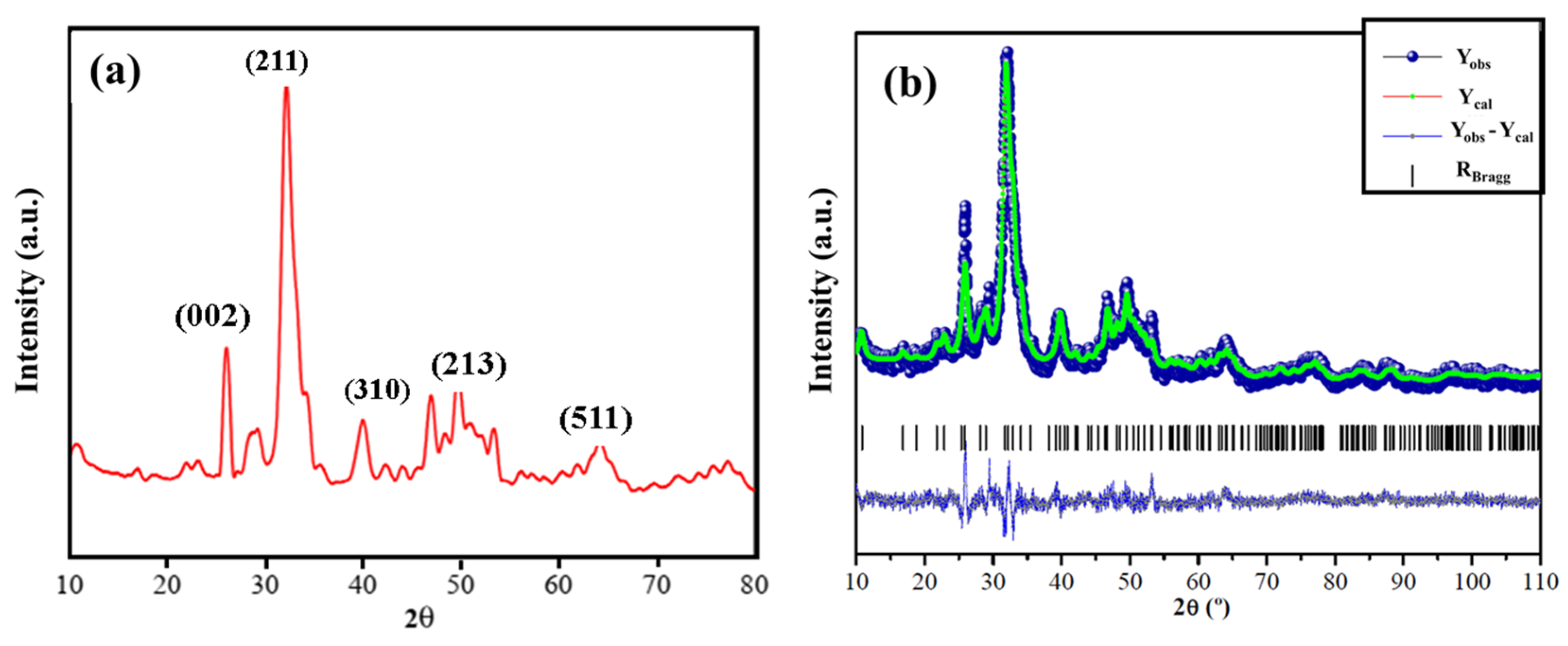
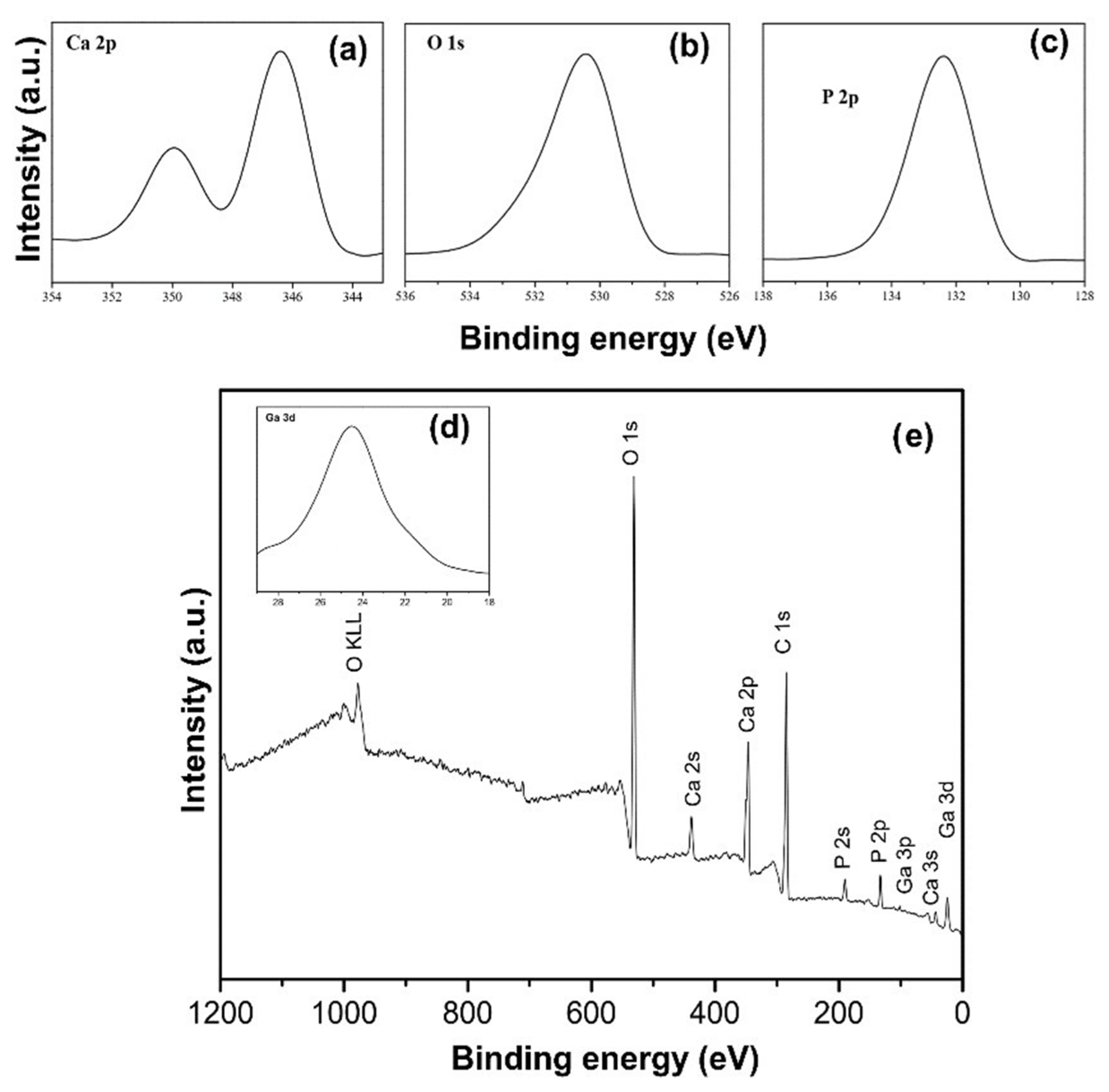
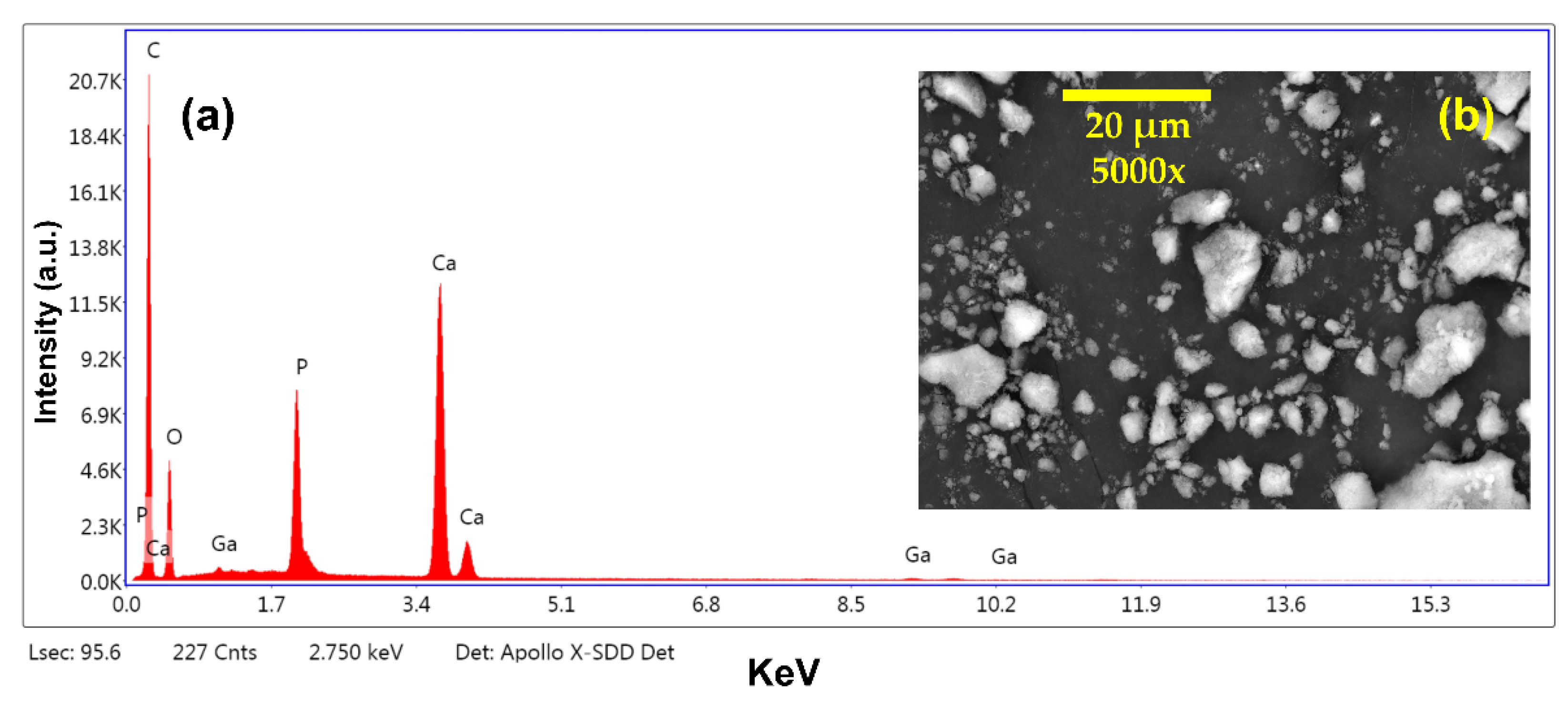
3.1. Structural Characterization
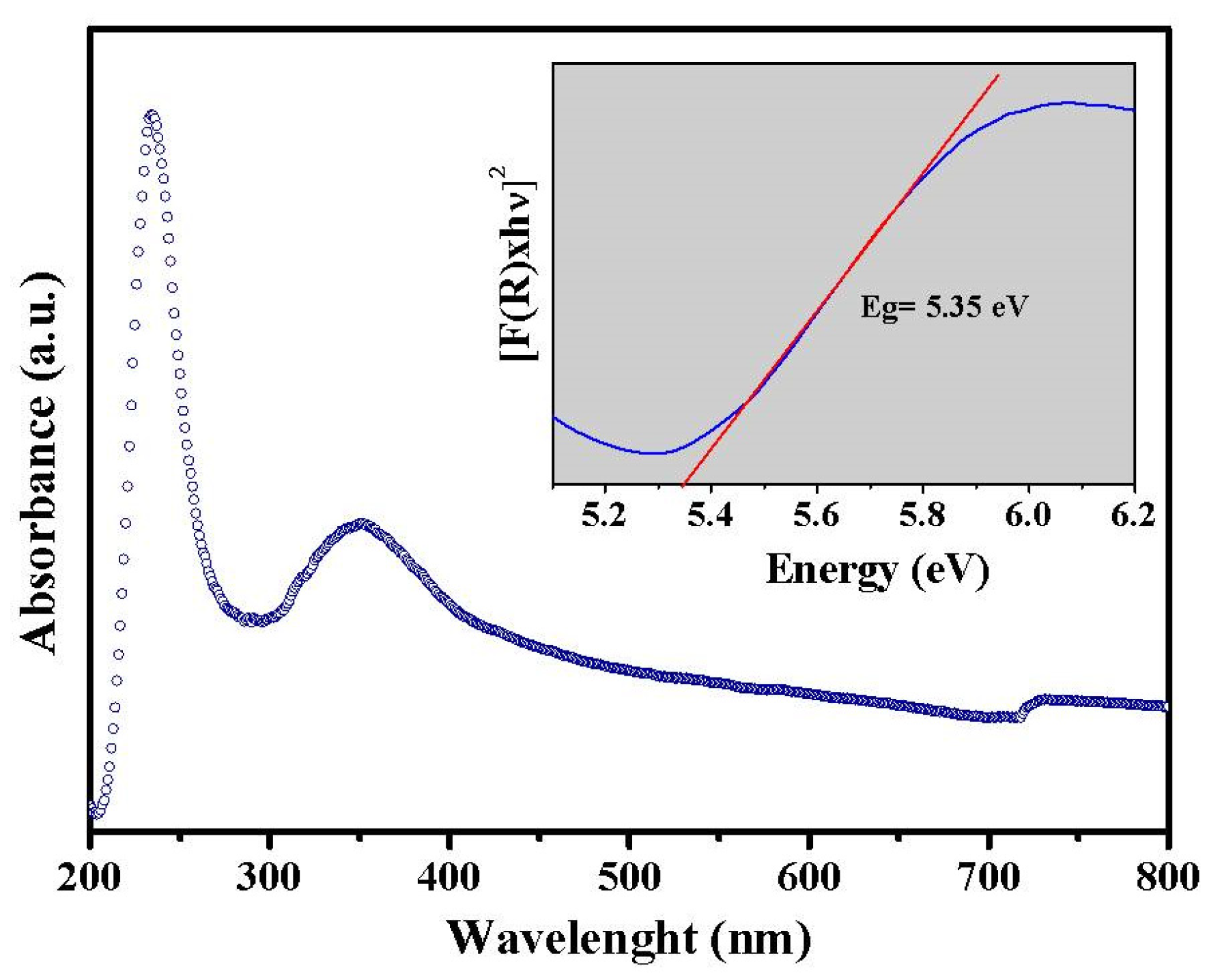
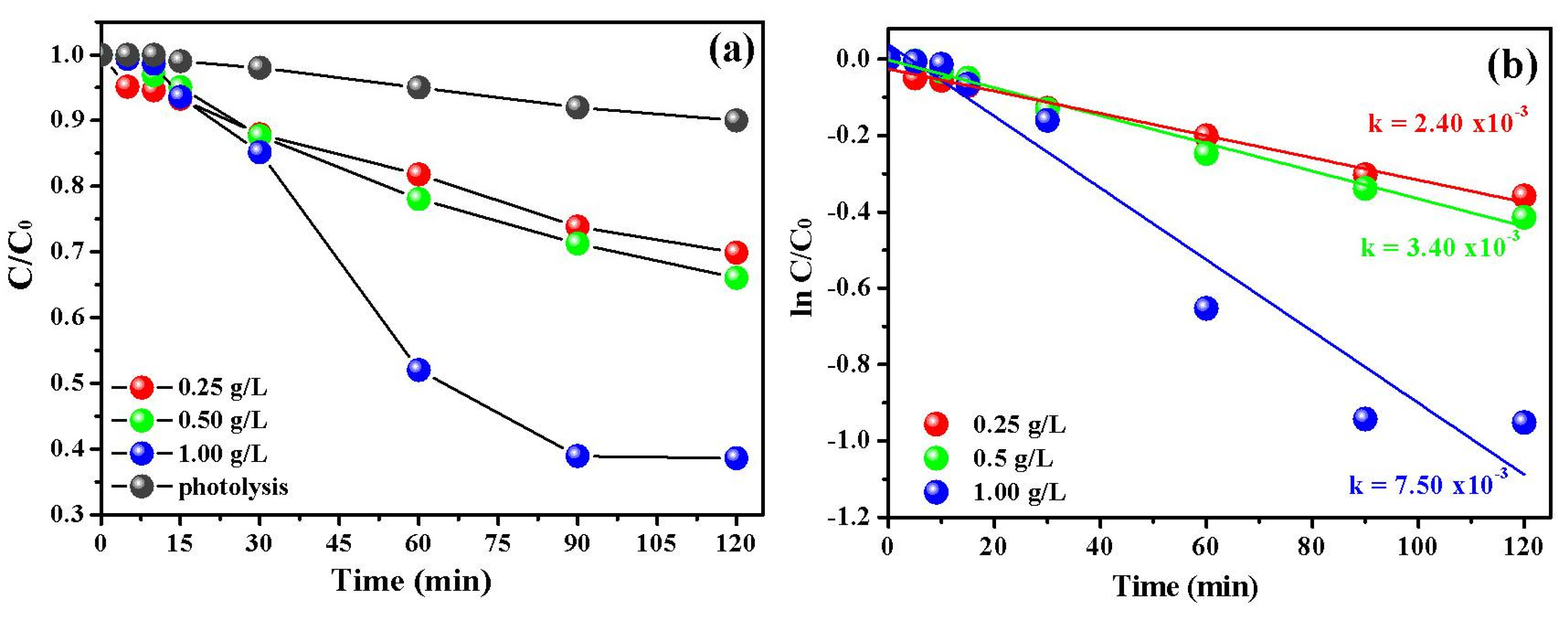
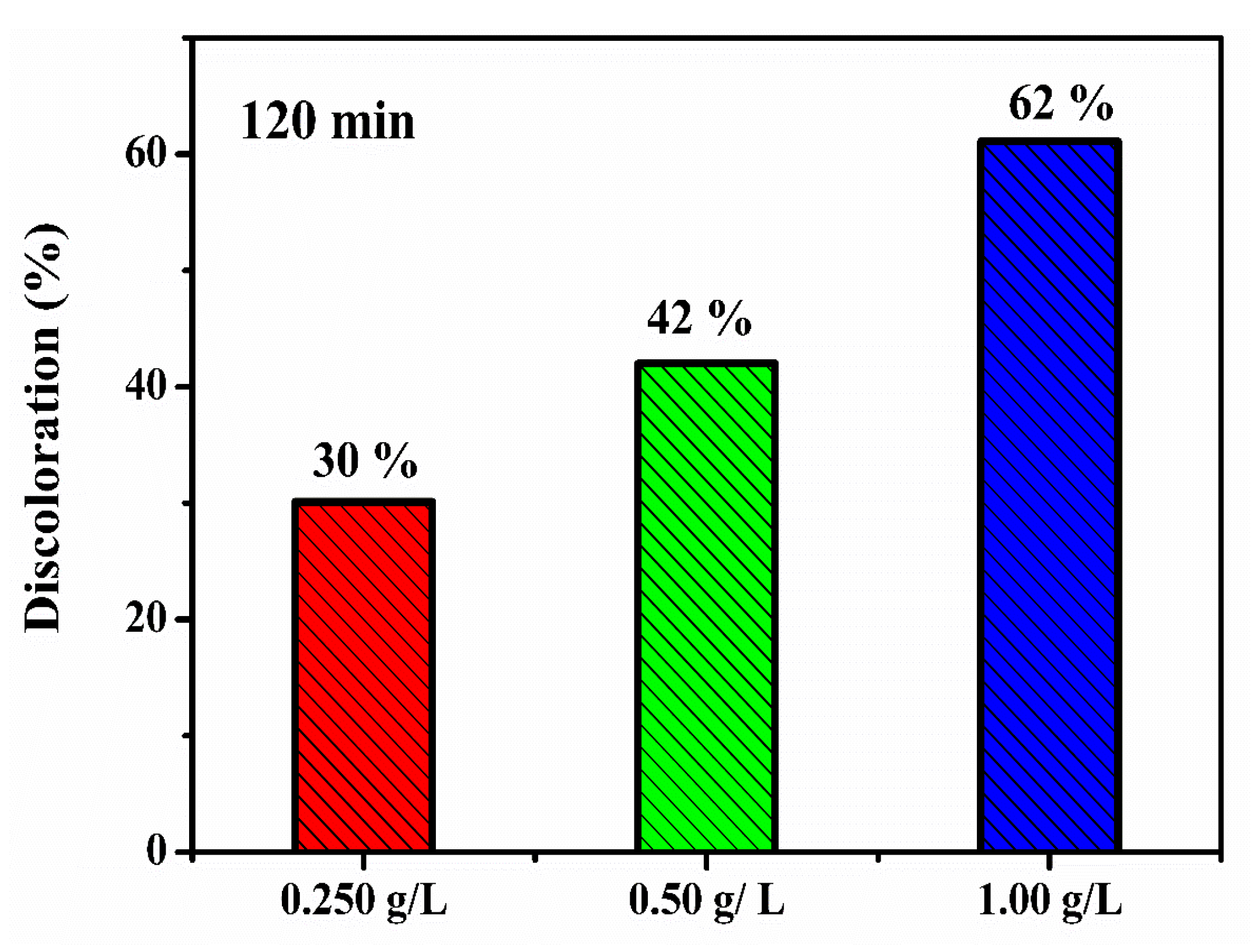
3.2. Photocatalytic Investigation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Al-Mamun, M.R.; Kader, S.; Islam, M.S.; Khan, M.Z.H. Photocatalytic activity improvement and application of UV-TiO2 photocatalysis in textile wastewater treatment: A review. J. Environ. Chem. Eng. 2019, 7, 103248. [Google Scholar] [CrossRef]
- Ge, M.; Hu, Z.; Wei, J.; He, Q.; He, Z. Recent advances in persulfate-assisted TiO2-based photocatalysis for wastewater treatment: Performances, mechanism and perspectives. J. Alloys Compd. 2021, 888, 161625. [Google Scholar] [CrossRef]
- Malato, S.; Blanco, J.; Vidal, A.; Richter, C. Photocatalysis with solar energy at a pilot-plant scale: An overview. Appl. Catal. B Environ. 2002, 37, 1–15. [Google Scholar] [CrossRef]
- Wang, J.L.; Xu, L.J. Advanced oxidation processes for wastewater treatment: Formation of hydroxyl radical and application. Crit. Rev. Environ. Sci. Technol. 2012, 42, 251–325. [Google Scholar] [CrossRef]
- Araujo, F.P.; Trigueiro, P.; Honório, L.M.C.; Oliveira, D.M.; Almeida, L.C.; Garcia, R.P.; Lobo, A.O.; Cantanhêde, W.; Silva-Filho, E.C.; Osajima, J.A. Eco-friendly synthesis and photocatalytic application of flowers-like ZnO structures using Arabic and Karaya Gums. Int. J. Biol. Macromol. 2020, 165, 2813–2822. [Google Scholar] [CrossRef] [PubMed]
- Qian, R.; Zong, H.; Schneider, J.; Zhou, G.; Zhao, T.; Li, Y.; Yang, J.; Bahnemann, D.W.; Pan, J.H. Charge carrier trapping, recombination and transfer during TiO2 photocatalysis: An overview. Catal. Today 2018, 335, 78–90. [Google Scholar] [CrossRef]
- Araujo, F.P.; Trigueiro, P.; Honório, L.M.C.; Furtini, M.B.; Oliveirab, D.M.; Almeidac, L.C.; Peña-Garcia, R.R.; Viana, B.C.; Silva-Filho, E.C.; Osajima, J.A. Novel green approach based on ZnO nanoparticles and polysaccharides for photocatalytic performance. Dalt. Trans. 2020, 49, 16394–16403. [Google Scholar] [CrossRef]
- Mohseni-Salehi, M.S.; Taheri-Nassaj, E.; Hosseini-Zori, M. Effect of dopant (Co, Ni) concentration and hydroxyapatite compositing on photocatalytic activity of titania towards dye degradation. J. Photochem. Photobiol. A Chem. 2018, 356, 57–70. [Google Scholar] [CrossRef]
- De Oliveira, W.V.; Morais, A.I.S.; Honorio, L.M.C.; Almeida, L.C.; Viana, B.C.; Furtini, M.B.; Silva-filho, E.C.; Osajima, J.A. TiO2 Immobilized on Fibrous Clay as Strategies to Photocatalytic Activity. Mater. Res. 2020, 23, 1–10. [Google Scholar] [CrossRef]
- Matos, J.; Ocares-Riquelme, J.; Poon, P.S.; Montaña, R.; García, X.; Campos, K.; Hernández-Garrido, J.C.; Titirici, M.M. C-doped anatase TiO2: Adsorption kinetics and photocatalytic degradation of methylene blue and phenol, and correlations with DFT estimations. J. Colloid Interface Sci. 2019, 547, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Honorio, L.M.C.; Trigueiro, P.A.; Viana, B.C.; Ribeiro, A.B.; Osajima, J.A. Nanostructured Materials for the Photocatalytic Degradation of Organic Pollutants in Water; Springer: Cham, Switzerland, 2019; ISBN 9783030337445. [Google Scholar]
- da Silva Lopes, J.; Rodrigues, W.V.; Oliveira, V.V.; Braga, A.D.; da Silva, R.T.; França, A.A.; da Paz, E.C.; Osajima, J.A.; da Silva Filho, E.C. Modification of kaolinite from Pará/Brazil region applied in the anionic dye photocatalytic discoloration. Appl. Clay Sci. 2019, 168, 295–303. [Google Scholar] [CrossRef]
- da Silva, O.G.; da Silva Filho, E.C.; da Fonseca, M.G.; Arakaki, L.N.H.; Airoldi, C. Hydroxyapatite organofunctionalized with silylating agents to heavy cation removal. J. Colloid Interface Sci. 2006, 302, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Szcześ, A.; Hołysz, L.; Chibowski, E. Synthesis of hydroxyapatite for biomedical applications. Adv. Colloid Interface Sci. 2017, 249, 321–330. [Google Scholar] [CrossRef]
- Valizadeh, S.; Rasoulifard, M.H.; Dorraji, M.S.S. Modified Fe3O4- hydroxyapatite nanocomposites as heterogeneous catalysts in three UV, Vis and Fenton like degradation systems. Appl. Surf. Sci. 2014, 319, 358–366. [Google Scholar] [CrossRef]
- Yao, Z.; Wang, X.; Hu, M.; Yao, Z.; Liu, X.; Ma, L.; He, Z.; Wang, X. Enhancement mechanism of hydroxyapatite for photocatalytic degradation of gaseous formaldehyde over TiO2/hydroxyapatite. J. Taiwan Inst. Chem. Eng. 2018, 85, 91–97. [Google Scholar]
- da Silva, O.G.; da Fonseca, M.G.; Arakaki, L.N.H. Silylated calcium phosphates and their new behavior for copper retention from aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2007, 301, 376–381. [Google Scholar] [CrossRef]
- Pereira, M.B.B.; Honório, L.M.C.; Lima-Júnior, C.G.; Silva Filho, E.C.; Gaslain, F.; Rigaud, B.; Fonseca, M.G.; Jaber, M. Modulating the structure of organofunctionalized hydroxyapatite/tripolyphosphate/chitosan spheres for dye removal. J. Environ. Chem. Eng. 2020, 8, 103980. [Google Scholar] [CrossRef]
- Ekka, B.; Nayak, S.R.; Achary, L.S.K.; Sarita; Kumar, A.; Mawatwal, S.; Dhiman, R.; Dash, P.; Patel, R.K. Synthesis of hydroxyapatite-zirconia nanocomposite through sonochemical route: A potential catalyst for degradation of phenolic compounds. J. Environ. Chem. Eng. 2018, 6, 6504–6515. [Google Scholar] [CrossRef]
- Shariffuddin, J.H.; Jones, M.I.; Patterson, D.A. Greener photocatalysts: Hydroxyapatite derived from waste mussel shells for the photocatalytic degradation of a model azo dye wastewater. Chem. Eng. Res. Des. 2013, 91, 1693–1704. [Google Scholar] [CrossRef]
- Manoj, M.; Mangalaraj, D.; Meena, P.; Yuan, A. Facile development and structural investigations of HAp and HAp/Ta nanostructures: Photocatalytic activity against Turq blue GL dye. Mater. Res. Express 2019, 7, 015012. [Google Scholar] [CrossRef]
- Liu, X.; Ma, J.; Yang, J. Visible-light-driven amorphous Fe(III)-substituted hydroxyapatite photocatalyst: Characterization and photocatalytic activity. Mater. Lett. 2014, 137, 256–259. [Google Scholar] [CrossRef]
- Bouyarmane, H.; El Bekkali, C.; Labrag, J.; Es-saidi, I.; Bouhnik, O.; Abdelmoumen, H.; Laghzizil, A.; Nunzi, J.M.; Robert, D. Photocatalytic degradation of emerging antibiotic pollutants in waters by TiO2/Hydroxyapatite nanocomposite materials. Surfaces and Interfaces 2021, 24, 101155. [Google Scholar] [CrossRef]
- Al-Ahmed, Z.A.; Al-Radadi, N.S.; Ahmed, M.K.; Shoueir, K.; El-Kemary, M. Dye removal, antibacterial properties, and morphological behavior of hydroxyapatite doped with Pd ions. Arab. J. Chem. 2020, 13, 8626–8637. [Google Scholar] [CrossRef]
- Sousa, R.B.; Dametto, A.C.; Sabio, R.M.; de Carvalho, R.A.; Vieira, E.G.; do Amaral Oliveira, A.F.; Ribeiro, L.K.; Barud, H.S.; Silva-Filho, E.C. Cerium-doped calcium phosphates precipitated on bacterial cellulose platform by mineralization. Ceram. Int. 2020, 46, 26985–26990. [Google Scholar] [CrossRef]
- Basfer, N.M.; Mansour, S.F.; Ahmed, M.K. Physicochemical properties of hydroxyapatite modified with vanadium ions for degradation of methylene blue. J. Mol. Struct. 2021, 1240, 130562. [Google Scholar] [CrossRef]
- Pang, Y.; Kong, L.; Chen, D.; Yuvaraja, G.; Mehmood, S. Facilely synthesized cobalt doped hydroxyapatite as hydroxyl promoted peroxymonosulfate activator for degradation of Rhodamine B. J. Hazard. Mater. 2020, 384, 121447. [Google Scholar] [CrossRef] [PubMed]
- Zou, R.; Xu, T.; Lei, X.; Wu, Q.; Xue, S. Novel and efficient red phosphorus/hollow hydroxyapatite microsphere photocatalyst for fast removal of antibiotic pollutants. J. Phys. Chem. Solids 2020, 139, 109353. [Google Scholar] [CrossRef]
- Lei, X.; Xu, T.; Yao, W.; Wu, Q.; Zou, R. Hollow hydroxyapatite microspheres modified by CdS nanoparticles for efficiently photocatalytic degradation of tetracycline. J. Taiwan Inst. Chem. Eng. 2020, 106, 148–158. [Google Scholar] [CrossRef]
- Le Hoang, T.T.T.; Insin, N.; Sukpirom, N. Catalytic activity of silver nanoparticles anchored on layered double hydroxides and hydroxyapatite. Inorg. Chem. Commun. 2020, 121, 108199. [Google Scholar] [CrossRef]
- Marimuthu, S.; Antonisamy, A.J.; Malayandi, S.; Rajendran, K.; Tsai, P.C.; Pugazhendhi, A.; Ponnusamy, V.K. Silver nanoparticles in dye effluent treatment: A review on synthesis, treatment methods, mechanisms, photocatalytic degradation, toxic effects and mitigation of toxicity. J. Photochem. Photobiol. B Biol. 2020, 205, 111823. [Google Scholar] [CrossRef]
- Pajor, K.; Pajchel, Ł.; Zgadzaj, A.; Piotrowska, U.; Kolmas, J. Modifications of Hydroxyapatite by Gallium and Silver Ions—Physicochemical Characterization, Cytotoxicity and Antibacterial Evaluation. Int. J. Mol. Sci. 2020, 21, 5006. [Google Scholar] [CrossRef]
- Vieira, E.; Silva, M.; Maia-Filho, A.; Ferreira, D.; Figuerêdo-Silva, J.; Rovaris, K.; Fialho, A.C.; Leite-Oliveira, A.; Menezes de Oliveira, A.L.; da Fonseca, M.G.; et al. Effect of Cerium-Containing Hydroxyapatite in Bone Repair in Female Rats with Osteoporosis Induced by Ovariectomy. Minerals 2021, 11, 377. [Google Scholar] [CrossRef]
- Person, A.; Bocherens, H.; Mariotti, A.; Renard, M. Diagenetic evolution and experimental heating of bone phosphate. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1996, 126, 135–149. [Google Scholar] [CrossRef]
- Rietveld, H.M. A profile refinement method for nuclear and magnetic structures. J. Appl. Crystallogr. 1969, 2, 65–71. [Google Scholar] [CrossRef]
- Kim, T.-R.; Kim, M.-S.; Goh, T.S.; Lee, J.S.; Kim, Y.H.; Yoon, S.-Y.; Lee, C.-S. Evaluation of Structural and Mechanical Properties of Porous Artificial Bone Scaffolds Fabricated via Advanced TBA-Based Freeze-Gel Casting Technique. Appl. Sci. 2019, 9, 1965. [Google Scholar] [CrossRef]
- Ercan, I.; Kaygili, O.; Kayed, T.; Bulut, N.; Tombuloğlu, H.; İnce, T.; Al Ahmari, F.; Kebiroglu, H.; Ates, T.; Almofleh, A.; et al. Structural, spectroscopic, dielectric, and magnetic properties of Fe/Cu co-doped hydroxyapatites prepared by a wet-chemical method. Phys. B Condens. Matter 2022, 625, 413486. [Google Scholar] [CrossRef]
- Bystrov, V.S.; Piccirillo, C.; Tobaldi, D.M.; Castro, P.M.L.; Coutinho, J.; Kopyl, S.; Pullar, R.C. Oxygen vacancies, the optical band gap (Eg) and photocatalysis of hydroxyapatite: Comparing modelling with measured data. Appl. Catal. B Environ. 2016, 196, 100–107. [Google Scholar] [CrossRef]
- Posner, A.S.; Perloff, A.; Diorio, A.F. Refinement of the hydroxyapatite structure. Acta Crystallogr. 1958, 11, 308–309. [Google Scholar] [CrossRef]
- Vieira, E.G.; Sousa, P.A.A.; Matos, J.M.E.; Santos, M.R.M.C. Síntese pelo método da coprecipitação e caracterização estrutural do tungstato de cálcio com estrutura tipo scheelita. Cerâmica 2013, 59, 417–425. [Google Scholar] [CrossRef][Green Version]
- Oliveira, C.; de Oliveira, A.L.M.; Chantelle, L.; Landers, R.; Medina-Carrasco, S.; Del Mar Orta, M.; Silva Filho, E.C.; Fonseca, M.G. Zinc (II) modified hydroxyapatites for tetracycline removal: Zn (II) doping or ZnO deposition and their influence in the adsorption. Polyhedron 2021, 194. [Google Scholar] [CrossRef]
- Ballardini, A.; Montesi, M.; Panseri, S.; Vandini, A.; Balboni, P.G.; Tampieri, A.; Sprio, S. New hydroxyapatite nanophases with enhanced osteogenic and anti-bacterial activity. J. Biomed. Mater. Res. Part A 2018, 106, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Kurtjak, M.; Vukomanović, M.; Krajnc, A.; Kramer, L.; Turk, B.; Suvorov, D. Designing Ga(iii)-containing hydroxyapatite with antibacterial activity. RSC Adv. 2016, 6, 112839–112852. [Google Scholar] [CrossRef]
- Melnikov, P.; Teixeira, A.R.; Malzac, A.; Coelho, M.d.B. Gallium-containing hydroxyapatite for potential use in orthopedics. Mater. Chem. Phys. 2009, 117, 86–90. [Google Scholar] [CrossRef]
- Neelgund, G.M.; Oki, A. Photocatalytic activity of hydroxyapatite deposited graphene nanosheets under illumination to sunlight. Mater. Res. Bull. 2022, 146, 111593. [Google Scholar] [CrossRef]
- Dreghici, D.B.; Butoi, B.; Predoi, D.; Iconaru, S.L.; Stoican, O.; Groza, A. Chitosan–Hydroxyapatite Composite Layers Generated in Radio Frequency Magnetron Sputtering Discharge: From Plasma to Structural and Morphological Analysis of Layers. Polymers 2020, 12, 3065. [Google Scholar] [CrossRef] [PubMed]
- Zatsepin, D.A.; Boukhvalov, D.W.; Zatsepin, A.F.; Kuznetsova, Y.A.; Gogova, D.; Shur, V.Y.; Esin, A.A. Atomic structure, electronic states, and optical properties of epitaxially grown β-Ga2O3 layers. Superlattices Microstruct. 2018, 120, 90–100. [Google Scholar] [CrossRef]
- Rajhi, F.Y.; Yahia, I.S.; Zahran, H.Y.; Kilany, M. Synthesis, structural, optical, dielectric properties, gamma radiation attenuation, and antimicrobial activity of V-doped hydroxyapatite nanorods. Mater. Today Commun. 2021, 26, 101981. [Google Scholar] [CrossRef]
- Vukomanović, M.; Žunič, V.; Otoničar, M.; Repnik, U.; Turk, B.; Škapin, S.D.; Suvorov, D. Hydroxyapatite/platinum bio-photocatalyst: A biomaterial approach to self-cleaning. J. Mater. Chem. 2012, 22, 10571–10580. [Google Scholar] [CrossRef]
- Amedlous, A.; Amadine, O.; Essamlali, Y.; Maati, H.; Semlal, N.; Zahouily, M. Copper Loaded Hydroxyapatite Nanoparticles as eco-friendly Fenton-like catalyst to Effectively Remove Organic Dyes. J. Environ. Chem. Eng. 2021, 9, 105501. [Google Scholar] [CrossRef]
- Piccirillo, C.; Pinto, R.A.; Tobaldi, D.M.; Pullar, R.C.; Labrincha, J.A.; Pintado, M.M.E.; Castro, P.M.L. Light induced antibacterial activity and photocatalytic properties of Ag/Ag3PO4 -based material of marine origin. J. Photochem. Photobiol. A Chem. 2015, 296, 40–47. [Google Scholar] [CrossRef]
- Piccirillo, C.; Dunnill, C.W.; Pullar, R.C.; Tobaldi, D.M.; Labrincha, J.A.; Parkin, I.P.; Pintado, M.M.; Castro, P.M.L. Calcium phosphate-based materials of natural origin showing photocatalytic activity. J. Mater. Chem. A 2013, 1, 6452. [Google Scholar] [CrossRef]
- Sathiyavimal, S.; Vasantharaj, S.; Shanmugavel, M.; Manikandan, E.; Nguyen-Tri, P.; Brindhadevi, K.; Pugazhendhi, A. Facile synthesis and characterization of hydroxyapatite from fish bones: Photocatalytic degradation of industrial dyes (crystal violet and Congo red). Prog. Org. Coat. 2020, 148, 105890. [Google Scholar] [CrossRef]
- Reeta Mary, I.; Leethiyal, R.; Sekar, P.; Mangalaraj, D.; Viswanathan, C.; Ponpandian, N. Self-assembly of nanostructured hydroxyapatite spheres for photodegradation of methylene blue dye. Mater. Today Proc. 2019, 18, 1729–1734. [Google Scholar] [CrossRef]
- Miranda, M.O.; Viana, B.C.; Honório, L.M.; Trigueiro, P.; Fonseca, M.G.; Franco, F.; Osajima, J.A.; Silva-Filho, E.C. Oxide-Clay Mineral as Photoactive Material for Dye Discoloration. Minerals 2020, 10, 132. [Google Scholar] [CrossRef]
- Chen, D.; Cheng, Y.; Zhou, N.; Chen, P.; Wang, Y.; Li, K.; Huo, S.; Cheng, P.; Peng, P.; Zhang, R.; et al. Photocatalytic degradation of organic pollutants using TiO2-based photocatalysts: A review. J. Clean. Prod. 2020, 268, 121725. [Google Scholar] [CrossRef]
- Reddy, M.P.; Venugopal, A.; Subrahmanyam, M. Hydroxyapatite photocatalytic degradation of calmagite (an azo dye) in aqueous suspension. Appl. Catal. B Environ. 2007, 69, 164–170. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pereira Rocha, R.L.; Silva, T.L.; Araujo, F.P.; Vieira, E.G.; Honório, L.M.; Furtini, M.B.; da Fonseca, M.G.; Silva-Filho, E.C.d.; Osajima, J.A. Gallium-Containing Hydroxyapatite as a Promising Material for Photocatalytic Performance. Minerals 2021, 11, 1347. https://doi.org/10.3390/min11121347
Pereira Rocha RL, Silva TL, Araujo FP, Vieira EG, Honório LM, Furtini MB, da Fonseca MG, Silva-Filho ECd, Osajima JA. Gallium-Containing Hydroxyapatite as a Promising Material for Photocatalytic Performance. Minerals. 2021; 11(12):1347. https://doi.org/10.3390/min11121347
Chicago/Turabian StylePereira Rocha, Rafael Lisandro, Thalena Lima Silva, Francisca Pereira Araujo, Ewerton Gomes Vieira, Luzia Maria Honório, Marcelo Barbosa Furtini, Maria Gardênnia da Fonseca, Edson Cavalcanti da Silva-Filho, and Josy Anteveli Osajima. 2021. "Gallium-Containing Hydroxyapatite as a Promising Material for Photocatalytic Performance" Minerals 11, no. 12: 1347. https://doi.org/10.3390/min11121347
APA StylePereira Rocha, R. L., Silva, T. L., Araujo, F. P., Vieira, E. G., Honório, L. M., Furtini, M. B., da Fonseca, M. G., Silva-Filho, E. C. d., & Osajima, J. A. (2021). Gallium-Containing Hydroxyapatite as a Promising Material for Photocatalytic Performance. Minerals, 11(12), 1347. https://doi.org/10.3390/min11121347








