Uranium (VI) Adsorbate Structures on Portlandite [Ca(OH)2] Type Surfaces Determined by Computational Modelling and X-ray Absorption Spectroscopy
Abstract
:1. Introduction
2. Materials and Methods—Computational and Experimental Details
2.1. Atomistic Models
2.2. Classical MD and PMF Simulations
2.3. Density Functional Theory Calculations
2.4. Experimental
2.4.1. Ex Situ Adsorption Experiments
2.4.2. In Situ Adsorption Experiments
2.4.3. reflEXAFS Measurements
3. Results
3.1. Bulk, Surface, and Uranium-Incorporated Portlandite
3.2. Speciation in Aqueous Solution
3.3. Sorption of Uranyl Hydroxides on Portlandite
3.4. Sorption on Defect Portlandite Surfaces
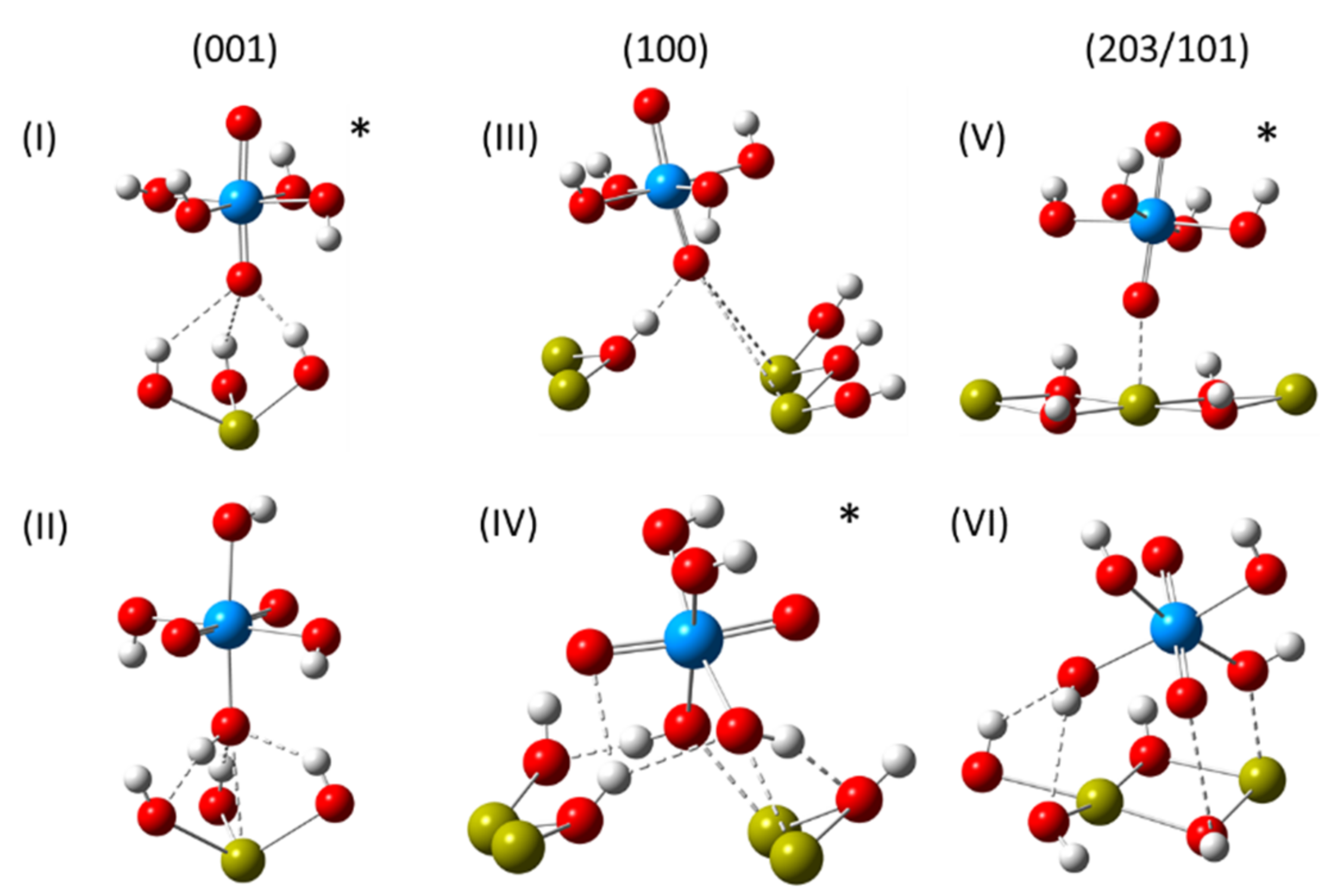
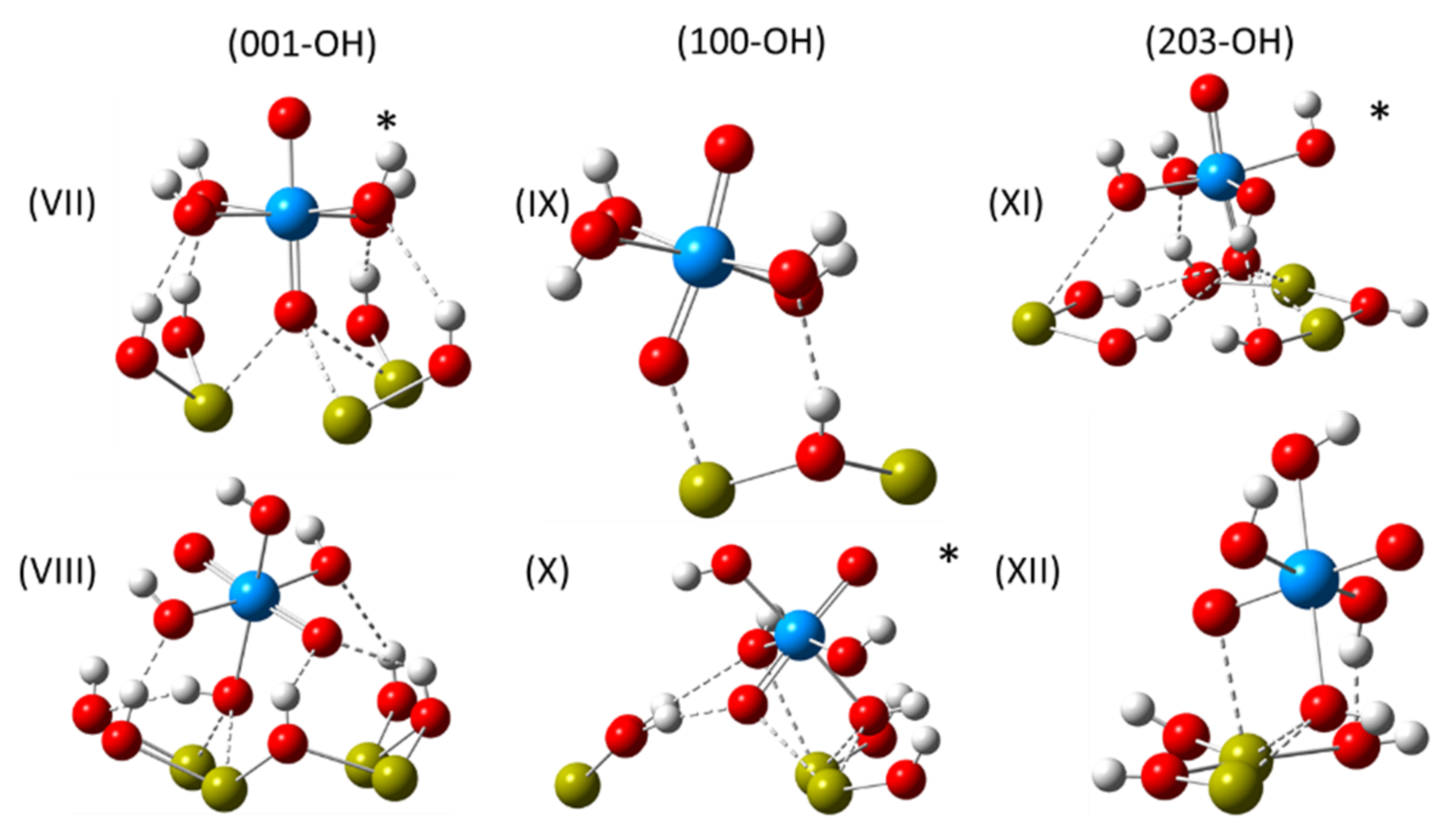
3.5. DFT Study of the Surface Complexes
- (001, ⊥)*—uranyl oxygen coordinates to the hydrogen atom of a surface hydroxyl group at a distance of 1.80 Å;
- (001, ߠ)—uranyl hydroxide forms two hydrogen bonds with the surface of 1.83 Å and 1.87 Å;
- (100, ⊥)—uranyl oxygen coordinates to the three exposed calcium ions with O-Ca distances of 2.60 Å, 2.93 Å and 3.12 Å;
- (100, ߠ)*—hydroxyl from the uranyl complex substitutes for the missing surface hydroxyl group with O-Ca distances of 2.58 Å, 2.64 Å and 3.21 Å. For comparison, the hydroxyl O-Ca distance on the clean (001) surface is only 2.39 Å;
- (203/101, ⊥)—uranyl oxygen atom coordinates to the hydrogen atom of a surface hydroxyl group at a distance of 1.75 Å;
- (203/101, ߠ)—one uranyl hydroxide group coordinates to an exposed calcium ion and is hydrogen bonded to a surface hydroxyl group with distances of 2.41 Å and 1.83 Å, respectively; a second uranyl hydroxide group also hydrogen bonds to another surface hydroxyl group at a distance of 1.78 Å;
- (001-OH, ⊥)*—uranyl oxygen coordinates to an exposed calcium ion at a distance of 2.40 Å;
- (001-OH, ߠ)—a hydroxyl group from the uranyl complex substitutes for the missing surface hydroxyl group with O-Ca distances of 2.46 Å and 2.60 Å;
- (100-OH, ⊥)—uranyl oxygen coordinates to an exposed calcium atom at a distance of 2.40 Å;
- (100-OH, ߠ)*—two uranyl hydroxide groups coordinate to two exposed calcium ions at distances of 2.40 Å and 2.60 Å;
- (203-OH, ⊥)*—uranyl oxygen atom coordinates to two exposed calcium ions at distances of 2.49 Å and 2.65 Å, replacing the missing hydroxyl group;
- (203-OH, ߠ)—a hydroxyl group from the uranyl complex substitutes for the missing surface hydroxyl group with O-Ca distances of 2.56 Å and 2.66 Å, while another hydroxyl group from the uranyl complex hydrogen bonds to a surface hydroxyl group at a distance of 1.75 Å.
3.6. Experimental EXAFS Analysis
- 13.
- pH 12 in KOH, 48 h reaction time before ex situ data acquisition;
- 14.
- pH 11 with 2 mM CO32−, pre-prepared ex situ 48 h reaction time before data acquisition;
- 15.
- pH 12 in KOH, reacted in situ, sorption for approximately 2 h before data acquisition;
- 16.
- pH 12 in LiOH, reacted in situ, sorption for approximately 2 h before data acquisition.
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nuclear Decommissioning Authority (NDA). Geological Disposal: Generic Environmental Safety Case; Main Report NDA/RWMD/021; Nuclear Decommissioning Authority (NDA): Didcot, UK, October 2010.
- Wilson, J.; Watson, S.; Towler, G.; Limer, L. An Assessment of Key Controls for the Disposal of Uranium Wastes; Report QRS-1384P-R1; Nuclear Decommissioning Authority (NDA): Didcot, UK, May 2012.
- Gaona, X.; Kulik, D.A.; Macé, N.; Wieland, E. Aqueous-solid solution thermodynamic model of U(VI) uptake in C-S-H phases. Appl. Geochem. 2012, 27, 81–95. [Google Scholar] [CrossRef]
- El Shafei, G.M.S. The polarizing power of metal cations in (hydr) oxides. J. Colloid Interface Sci. 1996, 182, 249–253. [Google Scholar] [CrossRef]
- Galmarini, S.; Aimable, A.; Ruffray, N.; Bowen, P. Changes in portlandite morphology with solvent composition: Atomistic simulations and experiment. Cem. Concr. Res. 2011, 41, 1330–1338. [Google Scholar] [CrossRef] [Green Version]
- Galmarini, S.; Aimable, A.; Ruffray, N.; Bowen, P. Changes in portlandite morphology with solvent composition: Atomistic simulations and experiment. Cem. Concr. Res. 2014, 61–62, 71. [Google Scholar] [CrossRef]
- Reeder, R.J.; Nugent, M.; Tait, C.D.; Morris, D.E.; Heald, S.M.; Beck, K.M.; Hess, W.P.; Lanzirotti, A. Coprecipitation of uranium(VI) with calcite: XAFS, micro-XAS, and luminescence characterization. Geochim. Cosmochim. Acta 2001, 65, 3491–3503. [Google Scholar] [CrossRef]
- Kelly, S.D.; Rasbury, E.T.; Chattopadhyay, S.; Kropf, A.J.; Kemner, K.M. Evidence of a stable uranyl site in ancient organic-rich calcite. Environ. Sci. Technol. 2006, 40, 2262–2268. [Google Scholar] [CrossRef]
- Tits, J.; Geipel, G.; Macé, N.; Eilzer, M.; Wieland, E. Determination of uranium(VI) sorbed species in calcium silicate hydrate phases: A laser-induced luminescence spectroscopy and batch sorption study. J. Colloid Interface Sci. 2011, 359, 248–256. [Google Scholar] [CrossRef]
- Macé, N.; Wieland, E.; Dähn, R.; Tits, J.; Scheinost, A.C. EXAFS investigation on U(VI) immobilization in hardened cement paste: Influence of experimental conditions on speciation. Radiochim. Acta 2013, 101, 379–389. [Google Scholar] [CrossRef] [Green Version]
- Chardon, E.S.; Bosbach, D.; Bryan, N.D.; Lyon, I.C.; Marquardt, C.; Römer, J.; Schild, D.; Vaughan, D.J.; Wincott, P.L.; Wogelius, R.A.; et al. Reactions of the feldspar surface with metal ions: Sorption of Pb(II), U(VI) and Np(V), and surface analytical studies of reaction with Pb(II) and U(VI). Geochim. Cosmochim. Acta 2008, 72, 288–297. [Google Scholar] [CrossRef]
- Van Veelen, A.; Copping, R.; Law, G.T.W.; Smith, A.J.; Bargar, J.R.; Rogers, J.; Shuh, D.K.; Wogelius, R.A. Uranium uptake onto Magnox sludge minerals studied using EXAFS. Mineral. Mag. 2012, 76, 3095–3104. [Google Scholar] [CrossRef]
- Cao, Q.; Huang, F.; Zhuang, Z.; Lin, Z. A study of the potential application of nano-Mg(OH)(2) in adsorbing low concentrations of uranyl tricarbonate from water. Nanoscale 2012, 4, 2423–2430. [Google Scholar] [CrossRef]
- Baranek, P.; Lichanot, A.; Orlando, R.; Dovesi, R. Structural and vibrational properties of solid Mg(OH)(2) and Ca(OH)(2)—Performances of various hamiltonians. Chem. Phys. Lett. 2001, 340, 362–369. [Google Scholar] [CrossRef]
- Kalinichev, A.G.; Kirkpatrick, R.J. Molecular dynamics modeling of chloride binding to the surfaces of calcium hydroxide, hydrated calcium aluminate, and calcium silicate phases. Chem. Mater. 2002, 14, 3539–3549. [Google Scholar]
- Hase, Y. Computational study of the solid-state vibrations and force field of magnesium and calcium hydroxides. J. Braz. Chem. Soc. 2006, 17, 741–745. [Google Scholar] [CrossRef]
- Laugesen, J.L. Density functional calculations of elastic properties of portlandite, Ca(OH)(2). Cem. Concr. Res. 2005, 35, 199–202. [Google Scholar] [CrossRef]
- Busing, W.R.; Levy, H.A. Neutron Diffraction Study of Calcium Hydroxide. J. Chem. Phys. 1957, 26, 563–568. [Google Scholar] [CrossRef]
- Petch, H.E.; Megaw, H.D. Crystal Structure of Brucite (MgOH2) and Portlandite (CaOH2) in Relation to Infrared Absorption. J. Opt. Soc. Am. 1954, 44, 744–745. [Google Scholar] [CrossRef]
- Desgranges, L.; Grebille, D.; Calvarin, G.; Chevrier, G.; Floquet, N.; Niepce, J.-C. Hydrogen thermal motion in calcium hydroxide—Ca (OH)2. Acta Crystallogr. Sect. B 1993, 49, 812–817. [Google Scholar] [CrossRef]
- Van Veelen, A.; Preedy, O.; Qi, J.; Law, G.T.W.; Morris, K.; Mosselmans, J.F.W.; Ryan, M.P.; Evans, N.D.M.; Wogelius, R.A. Uranium and technetium interactions with wustite [Fe1-xO] and portlandite [Ca(OH)(2)] surfaces under geological disposal facility conditions. Mineral. Mag. 2014, 78, 1097–1113. [Google Scholar] [CrossRef]
- Pointeau, I.; Landesman, C.; Giffaut, E.; Reiller, P. Reproducibility of the uptake of U(VI) onto degraded cement pastes and calcium silicate hydrate phases. Radiochim. Acta 2004, 92, 645–650. [Google Scholar] [CrossRef] [Green Version]
- Doudou, S.; Arumugam, K.; Vaughan, D.J.; Livens, F.R.; Burton, N.A. Investigation of ligand exchange reactions in aqueous uranyl carbonate complexes using computational approaches. Phys. Chem. Chem. Phys. 2011, 13, 11402–11411. [Google Scholar] [CrossRef]
- Doudou, S.; Vaughan, D.J.; Livens, F.R.; Burton, N.A. Atomistic Simulations of Calcium Uranyl(VI) Carbonate Adsorption on Calcite and Stepped-Calcite Surfaces. Environ. Sci. Technol. 2012, 46, 7587–7594. [Google Scholar] [CrossRef]
- Kerisit, S.; Parker, S. Free energy of adsorption of water and metal ions on the {1014} calcite surface. J. Am. Chem. Soc. 2004, 126, 10152–10161. [Google Scholar] [CrossRef]
- Spagnoli, D.; Cooke, D.J.; Kerisit, S.; Parker, S.C. Molecular dynamics simulations of the interaction between the surfaces of polar solids and aqueous solutions. J. Mater. Chem. 2006, 16, 1997–2006. [Google Scholar] [CrossRef]
- Greathouse, J.A.; Cygan, R.T. Molecular dynamics simulation of uranyl (VI) adsorption equilibria onto an external montmorillonite surface. Phys. Chem. Chem. Phys. 2005, 7, 3580–3586. [Google Scholar] [CrossRef]
- Greathouse, J.A.; O’Brien, R.J.; Bemis, G.; Pabalan, R.T. Molecular dynamics study of aqueous uranyl interactions with quartz (010). J. Phys. Chem. B 2002, 106, 1646–1655. [Google Scholar] [CrossRef]
- Kerisit, S.; Liu, C. Diffusion and Adsorption of Uranyl Carbonate Species in Nanosized Mineral Fractures. Environ. Sci. Technol. 2012, 46, 1632–1640. [Google Scholar] [CrossRef]
- Kerisit, S.; Liu, C. Molecular Dynamics Simulations of Uranyl and Uranyl Carbonate Adsorption at Aluminosilicate Surfaces. Environ. Sci. Technol. 2014, 48, 3899–3907. [Google Scholar] [CrossRef]
- Freeman, C.L.; Harding, J.H.; Cooke, D.J.; Elliott, J.A.; Lardge, J.S.; Duffy, D.M. New forcefields for modeling biomineralization processes. J. Phys. Chem. C 2007, 111, 11943–11951. [Google Scholar] [CrossRef]
- Abascal, J.L.F.; Vega, C. A general purpose model for the condensed phases of water: TIP4P/2005. J. Chem. Phys. 2005, 123, 234505. [Google Scholar] [CrossRef]
- Materials Studio v6.1.200; Accelrys Software Inc.: San Diego, CA, USA, 2012.
- Smith, W.; Forester, T.R. DL_POLY_2.0: A general-purpose parallel molecular dynamics simulation package. J. Mol. Graph. 1996, 14, 136–141. [Google Scholar] [CrossRef]
- Bonomi, M.; Branduardi, D.; Bussi, G.; Camilloni, C.; Provasi, D.; Raiteri, P.; Donadio, D.; Marinelli, F.; Pietrucci, F.; Broglia, R.A.; et al. PLUMED: A portable plugin for free-energy calculations with molecular dynamics. Comput. Phys. Commun. 2009, 180, 1961–1972. [Google Scholar] [CrossRef] [Green Version]
- Hoover, W.G. Canonical dynamics: Equilibrium phase-space distributions. Phys. Rev. A 1985, 31, 1695–1697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Williams, C.D.; Burton, N.A.; Travis, K.P.; Harding, J.H. The Development of a Classical Force Field to Determine the Selectivity of an Aqueous Fe3+-EDA Complex for TcO4- and SO42-. J. Chem. Theory Comput. 2014, 10, 3345–3353. [Google Scholar] [CrossRef]
- Kumar, S.; Rosenberg, J.M.; Bouzida, D.; Swendsen, R.H.; Kollman, P.A. Multidimensional free-energy calculations using the weighted histogram analysis method. J. Comput. Chem. 1995, 16, 1339–1350. [Google Scholar] [CrossRef]
- Dimelow, R.J.; Bryce, R.A.; Masters, A.J.; Hillier, I.H.; Burton, N.A. Exploring reaction pathways with transition path and umbrella sampling: Application to methyl maltoside. J. Chem. Phys. 2006, 124, 114113. [Google Scholar] [CrossRef]
- CP2K Developer Group 2000–2015. Available online: https://www.cp2k.org/ (accessed on 5 November 2021).
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef] [Green Version]
- Goedecker, S.; Teter, M.; Hutter, J. Separable dual-space Gaussian pseudopotentials. Phys. Rev. B 1996, 54, 1703–1710. [Google Scholar] [CrossRef] [Green Version]
- Hartwigsen, C.; Goedecker, S.; Hutter, J. Relativistic separable dual-space Gaussian pseudopotentials from H to Rn. Phys. Rev. B 1998, 58, 3641–3662. [Google Scholar] [CrossRef] [Green Version]
- Krack, M. Pseudopotentials for H to Kr optimized for gradient-corrected exchange-correlation functionals. Theor. Chem. Acc. 2005, 114, 145–152. [Google Scholar] [CrossRef] [Green Version]
- Lippert, G.; Hutter, J.; Parrinello, M. A hybrid Gaussian and plane wave density functional scheme. Mol. Phys. 1997, 92, 477–487. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 09; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Zhao, Y.; Truhlar, D. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: Two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor. Chem. Acc. 2008, 120, 215–241. [Google Scholar]
- Küchle, W.; Dolg, M.; Stoll, H.; Preuss, H. Ab initio pseudopotentials for Hg through Rn: I. Parameter sets and atomic calculations. Mol. Phys. 1991, 74, 1245–1263. [Google Scholar] [CrossRef]
- Shamov, G.A.; Schreckenbach, G. Theoretical study of the oxygen exchange in uranyl hydroxide. An old riddle solved? J. Am. Chem. Soc. 2008, 130, 13735–13744. [Google Scholar] [CrossRef]
- Moll, H.; Reich, T.; Szabó, Z. The hydrolysis of dioxouranium(VI) investigated using EXAFS and O-17-NMR. Radiochim. Acta 2000, 88, 411. [Google Scholar] [CrossRef]
- Parkhurst, D.L.; Appelo, C.A.J. User’s guide to PHREEQC (Version 2): A computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations. Water-Resour. Investig. Rep. 1999, 99, 312. [Google Scholar]
- Newville, M.; Ravel, B.; Haskel, D.; Rehr, J.J.; Stern, E.A.; Yacoby, Y. Analysis of multiple-scattering XAFS data using theoretical standards. Phys. B Condens. Matter 1995, 208–209, 154–156. [Google Scholar] [CrossRef]
- Guillaumont, R.; Fanghänel, T.; Fuger, J.; Grenthe, I.; Neck, V.; Palmer, D.A.; Rand, M.H. Update on the Chemical Thermodynamics of Uranium, Neptunium, Plutonium, Americium and Technetium; Elsevier: Amsterdam, The Netherlands, 2003. [Google Scholar]
- The Geochemist’s Workbench 2021; Rockware: Golden, CO, USA; Available online: https://www.gwb.com/ (accessed on 5 November 2021).
- Kienzler, B.; Luckscheiter, B.; Wilhelm, S. Waste form corrosion modeling: Comparison with experimental results. Waste Manag. 2001, 21, 741–752. [Google Scholar] [CrossRef]
- Langmuir, D. Aqueous Enviromental Geochemistry; Prentice-Hall Inc.: Upper Saddle River, NJ, USA, 1997. [Google Scholar]
- Vallet, V.; Wahlgren, U.; Schimmelpfennig, B.; Moll, H.; Szabó, Z.; Grenthe, I. Solvent effects on uranium(VI) fluoride and hydroxide complexes studied by EXAFS and quantum chemistry. Inorg. Chem. 2001, 40, 3516–3525. [Google Scholar] [CrossRef]
- Szabó, Z.; Grenthe, I. On the Mechanism of Oxygen Exchange Between Uranyl(VI) Oxygen and Water in Strongly Alkaline Solution as Studied by O-17 NMR Magnetization Transfer. Inorg. Chem. 2010, 49, 4928–4933. [Google Scholar] [CrossRef]
- Farkas, I.; Bányai, I.; Szabó, Z.; Wahlgren, U.; Grenthe, I. Rates and mechanisms of water exchange of UO22+(aq) and UO2(oxalate)F(H2O)(2)(-): A variable-temperature O-17 and F-19 NMR study. Inorg. Chem. 2000, 39, 799–805. [Google Scholar] [CrossRef]
- Wahlgren, U.; Moll, H.; Grenthe, I.; Schimmelpfennig, B.; Maron, L.; Vallet, V.; Gropen, O. Structure of uranium(VI) in strong alkaline solutions. A combined theoretical and experimental investigation. J. Phys. Chem. A 1999, 103, 8257–8264. [Google Scholar] [CrossRef]
- Schreckenbach, G.; Hay, P.J.; Martin, R.L. Theoretical study of stable trans and cis isomers in [UO2(OH)(4)](2-) using relativistic density functional theory. Inorg. Chem. 1998, 37, 4442–4451. [Google Scholar] [CrossRef]
- Sonnenberg, J.L.; Hay, P.J.; Martin, R.L.; Bursten, B.E. Theoretical investigations of uranyl-ligand bonding: Four- and five-coordinate uranyl cyanide, isocyanide, carbonyl, and hydroxide complexes. Inorg. Chem. 2005, 44, 2255–2262. [Google Scholar] [CrossRef]
- Clark, D.L.; Conradson, S.D.; Donohoe, R.J.; Keogh, D.W.; Morris, D.E.; Palmer, P.D.; Rogers, R.D.; Tait, C.D. Chemical speciation of the uranyl ion under highly alkaline conditions. Synthesis, structures, and oxo ligand exchange dynamics. Inorg. Chem. 1999, 38, 1456–1466. [Google Scholar] [CrossRef]
- Nordstrom, D.K.; Plummer, L.N.; Langmuir, D.; Busenberg, E.; May, H.M.; Jones, B.F.; Parkhurst, D.L. Revised Chemical Equilibrium Data for Major Water—Mineral Reactions and Their Limitations. In Chemical Modeling of Aqueous Systems II; American Chemical Society: Washington, DC, USA, 1990; Chapter 31; Volume 416, pp. 398–413. [Google Scholar]
- Bots, P.; Morris, K.; Hibberd, R.; Law, G.T.W.; Mosselmans, J.F.W.; Brown, A.P.; Doutch, J.; Smith, A.J.; Shaw, S. Uranium(VI) Colloidal Nanoparticles in Conditions Relevant to Radioactive Waste Disposal. Langmuir 2014, 30, 14396–14405. [Google Scholar] [CrossRef]
- Thompson, H.A.; Brown, G.E., Jr.; Parks, G.A. XAFS spectroscopic study of uranyl coordination in solids and aqueous solution. Am. Mineral. 1997, 82, 483–496. [Google Scholar] [CrossRef]
- Catalano, J.G.; Brown, G.E., Jr. Analysis of uranyl-bearing phases by EXAFS spectroscopy: Interferences, multiple scattering, accuracy of structural parameters, and spectral differences. Am. Mineral. 2004, 89, 1004–1021. [Google Scholar] [CrossRef]
- Matar, S.F.; Demazeau, G. Electronic band structure of CaUO4 from first principles. J. Solid State Chem. 2009, 182, 2678–2684. [Google Scholar] [CrossRef]
- Loopstra, B.O.; Rietveld, H.M. Structure of Some Alkaline-Earth Metal Uranates. Acta Crystallogr. Sect. B 1969, 25, 787–791. [Google Scholar] [CrossRef]
- King, R.B. Some aspects of structure and bonding in binary and ternary uranium(VI) oxides. Chem. Mater. 2002, 14, 3628–3635. [Google Scholar] [CrossRef]
- Deliens, M.; Piret, P. Metastudtite, UO4. 2H2O, a new mineral from Shinkolobwe, Shaba, Zaire. Am. Mineral. 1983, 68, 456–458. [Google Scholar]






| Model Structure | Functional | R(U-Oax) | R(U-OHeq) | R(U-U) | R(Ca-U) |
|---|---|---|---|---|---|
| [UO2(OH)4]2− | PBE | 1.861 | 2.298 | - | - |
| PBE0 | 1.810 | 2.286 | - | - | |
| B3LYP | 1.825 | 2.306 | - | - | |
| M06 | 1.801 | 2.305 | - | - | |
| PBE/PCM | 1.853 | 2.277 | - | - | |
| EXAFS a | 1.83 | 2.26 | - | - | |
| [(UO2)2(OH)6]2− | PBE | 1.825, 1.837 | 2.416–2.430 b 2.228–2.233 | 3.983 | - |
| CaUO2(OH)4 | PBE | 1.805, 1.897 | 2.502, 2.506, 2.155, 2.158 | - | 3.141 |
| U(VI)-substituted portlandite c | PBE | 2.197 | 2.577 | - | 3.764 |
| UO2-substituted portlandite d | PBE | 1.865, 1.867 | 2.259, 2.263, 2.261, 2.262 | - | 3.751, 3.755, 3.739, 3.749 |
| Ca(OH)2 Surface | Aqueous Species | U-OHeq(UO2) 1 | U-OHeq(*) | U-H2Oeq | U-Ca | |||
|---|---|---|---|---|---|---|---|---|
| CN | R (/Å) | CN | R (/Å) | CN | R (/Å) | R (/Å) | ||
| (001) | UO22+ | 0 (0) | - | 0 (0) | - (2.33) | 5 (3) | 2.53 (2.58) | 6.38,6.73 (3.68, 4.23) |
| UO2(OH)+ | 1 (1) | 2.18 (2.28) | 0 (2) | - (2.28) | 4 (1) | 2.58 (2.58) | 6.33 (4.18, 4.52) | |
| UO2(OH)2 | 2 (2) | 2.23 (2.28) | 0 (2) | - (2.28) | 2 (0) | 2.58 (-) | 5.53, 5.83 (3.63, 4.28) | |
| UO2(OH)3− | 3 | 2.38 | 0 | - | 1 | 2.63 | 5.38, 6.13 | |
| UO2(OH)42− | 4 | 2.28 | 0 | - | 0 | - | 5.63, 6.03 | |
| UO2(OH)53− | 5 | 2.33 | 0 | - | 0 | - | 4.43, 5.82, 6.98 | |
| (100) | UO22+ | 0 | - | 4 | 2.23 | 0 | - | 4.18 |
| UO2(OH)+ | 1 | 2.23 | 3 | 2.23 | 1 | 2.58 | 3.68, 4.23 | |
| UO2(OH)2 | 2 | 2.23 | 1 | 2.23 | 1 | 2.58 | 3.63, 4.68 | |
| UO2(OH)3− | 3 | 2.28 | 1 | 2.28 | 0 | - | 4.23 | |
| UO2(OH)42− | 3 | 2.28 | 1 | - | 0 | - | 4.13 | |
| UO2(OH)53− | 4 | 2.28 | 0 | - | 0 | - | 5.13 | |
| (203/101) | UO22+ | 0 | - | 4 | 2.33 | 0 | - | 3.68 |
| UO2(OH)+ | 1 | 2.28 | 3 | 2.28 | 0 | - | 4.33 | |
| UO2(OH)2 | 2 | 2.33 | 3 | 2.33 | 0 | - | 3.83 4.28 | |
| UO2(OH)3− | 3 | 2.28 | 1 | 2.28 | 0 | - | 3.78, 4.33 3.73, 4.13 | |
| UO2(OH)42− | 4 | 2.28 | 0 | - | 0 | - | 4.13, 5.63 | |
| UO2(OH)53− | 5 | 2.33 | 0 | - | 0 | - | 3.83, 4.23 | |
| Ca(OH)2 Surface | Aqueous Species | U-OHeq (≈) | U-OHeq (>) | U-Ca | ||
|---|---|---|---|---|---|---|
| CN | R (/Å) | CN | R (/Å) | R (/Å) | ||
| (001-OH) | UO2(OH)42− | 4 | 2.28 | 0 | - | 3.88, 4.43 |
| (100-OH) | UO2(OH)42− | 4 | 2.28 | 0 | - | 3.93 |
| (203-OH) | UO2(OH)42− | 4 | 2.28 | 0 | - | 4.13 |
| Ca(OH)2 Surface | Orientation | U-OHeq(UO2) 1 | U-OHeq(*) | U=O | U-Ca | ||
|---|---|---|---|---|---|---|---|
| CN | R (/Å) | CN | R (/Å) | R (/Å) | R (/Å) | ||
| (001) | ⊥ | 4 | 2.316, 2.287, 2.244, 2.381 | 0 | - | 1.826, 1.866 | 5.418 |
| ߠ | 4 | 2.228, 2.204, 2.332, 2.326 | 0 | - | 1.859, 1.879 | 5.060 | |
| (100) | ⊥ | 4 | 2.180, 2.256, 2.307, 2.338 | 0 | - | 1.838, 1.893 | 5.800 |
| ߠ | 3 | 2.221, 2.262, 2.242 | 1 | 2.238 | 1.867, 1.887 | 4.054 | |
| (203/101) | ⊥ | 4 | 2.239, 2.277, 2.342, 2.306 | 0 | - | 1.836, 1.872 | 4.203 |
| ߠ | 2 | 2.258, 2.281 | 2 | 2.396, 2.618 | 1.853, 1.868 | 3.977 | |
| (001-OH) | ⊥ | 4 | 2.118, 2.255, 2.324, 2.368 | 0 | - | 1.866, 1.905 | 4.158 |
| ߠ | 3 | 2.262, 2.254, 2.341 | 1 | 2.343 | 1.820, 1.858 | 4.082 | |
| (100-OH) | ⊥ | 4 | 2.157, 2.242, 2.345, 2.348 | 0 | - | 1.869, 1.883 | 3.959 |
| ߠ | 3 | 2.178, 2.251, 2.256 | 1 | 2.302 | 1.857, 1.900 | 3.508, 3.871 | |
| (203-OH) | ⊥ | 4 | 2.240, 2.325, 2.341, 2.141 | 0 | - | 1.861, 1.906 | 3.740, 4.094 |
| ߠ | 3 | 2.222, 2.255, 2.284 | 1 | 2.371 | 1.864, 1.865 | 3.629, 4.441 | |
| Surface | Energy Difference/(kJ mol−1) |
|---|---|
| (001) | −38.7 |
| (001-OH) | −33.5 |
| (100) | 78.8 |
| (100-OH) | 144.1 |
| (203) | −82.2 |
| (203-OH) | −93.3 |
| Experiment | U-Oax | U-O | U-U | U-Ca(1) | U-Ca(2) | |
|---|---|---|---|---|---|---|
| Ex situ pH 12 in KOH | CN 1 | 1.7 | 4.5 | - | 1.0 | 1.0 |
| R (/Å) 2 | 1.87 (0.009) | 2.33 (0.01) | - | 3.08 (0.011) | 3.72 (0.008) | |
| Ex situ 2 mM CO32− pH 11 | CN | 2.0 | 5.0 | - | 2.0 | - |
| R (/Å) | 1.81 (0.004) | 2.29 (0.011) | - | 2.91 (0.006) | - | |
| In situ pH 12 in KOH | CN | 1.6 | 4.1 | 1.0 | 1.0 | - |
| R (/Å) | 1.86 (0.009) | 2.35 (0.008) | 3.83 (0.005) | 4.08 (0.009) | - | |
| In situ pH 12 in LiOH | CN | 2.4 | 4.1 | 1.7 | 2.1 | 4.0 |
| R (/Å) | 1.93 (0.01) | 2.28 (0.007) | 3.84 (0.016) | 3.59 (0.002) | 3.78 (0.008) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, C.A.; van Veelen, A.; Morris, K.; Mosselmans, J.F.W.; Wogelius, R.A.; Burton, N.A. Uranium (VI) Adsorbate Structures on Portlandite [Ca(OH)2] Type Surfaces Determined by Computational Modelling and X-ray Absorption Spectroscopy. Minerals 2021, 11, 1241. https://doi.org/10.3390/min11111241
Lee CA, van Veelen A, Morris K, Mosselmans JFW, Wogelius RA, Burton NA. Uranium (VI) Adsorbate Structures on Portlandite [Ca(OH)2] Type Surfaces Determined by Computational Modelling and X-ray Absorption Spectroscopy. Minerals. 2021; 11(11):1241. https://doi.org/10.3390/min11111241
Chicago/Turabian StyleLee, Christopher A., Arjen van Veelen, Katherine Morris, J. Fred W. Mosselmans, Roy A. Wogelius, and Neil A. Burton. 2021. "Uranium (VI) Adsorbate Structures on Portlandite [Ca(OH)2] Type Surfaces Determined by Computational Modelling and X-ray Absorption Spectroscopy" Minerals 11, no. 11: 1241. https://doi.org/10.3390/min11111241
APA StyleLee, C. A., van Veelen, A., Morris, K., Mosselmans, J. F. W., Wogelius, R. A., & Burton, N. A. (2021). Uranium (VI) Adsorbate Structures on Portlandite [Ca(OH)2] Type Surfaces Determined by Computational Modelling and X-ray Absorption Spectroscopy. Minerals, 11(11), 1241. https://doi.org/10.3390/min11111241






