Marble Waste Valorization through Alkali Activation
Abstract
1. Introduction
2. Materials and Methods
3. Results
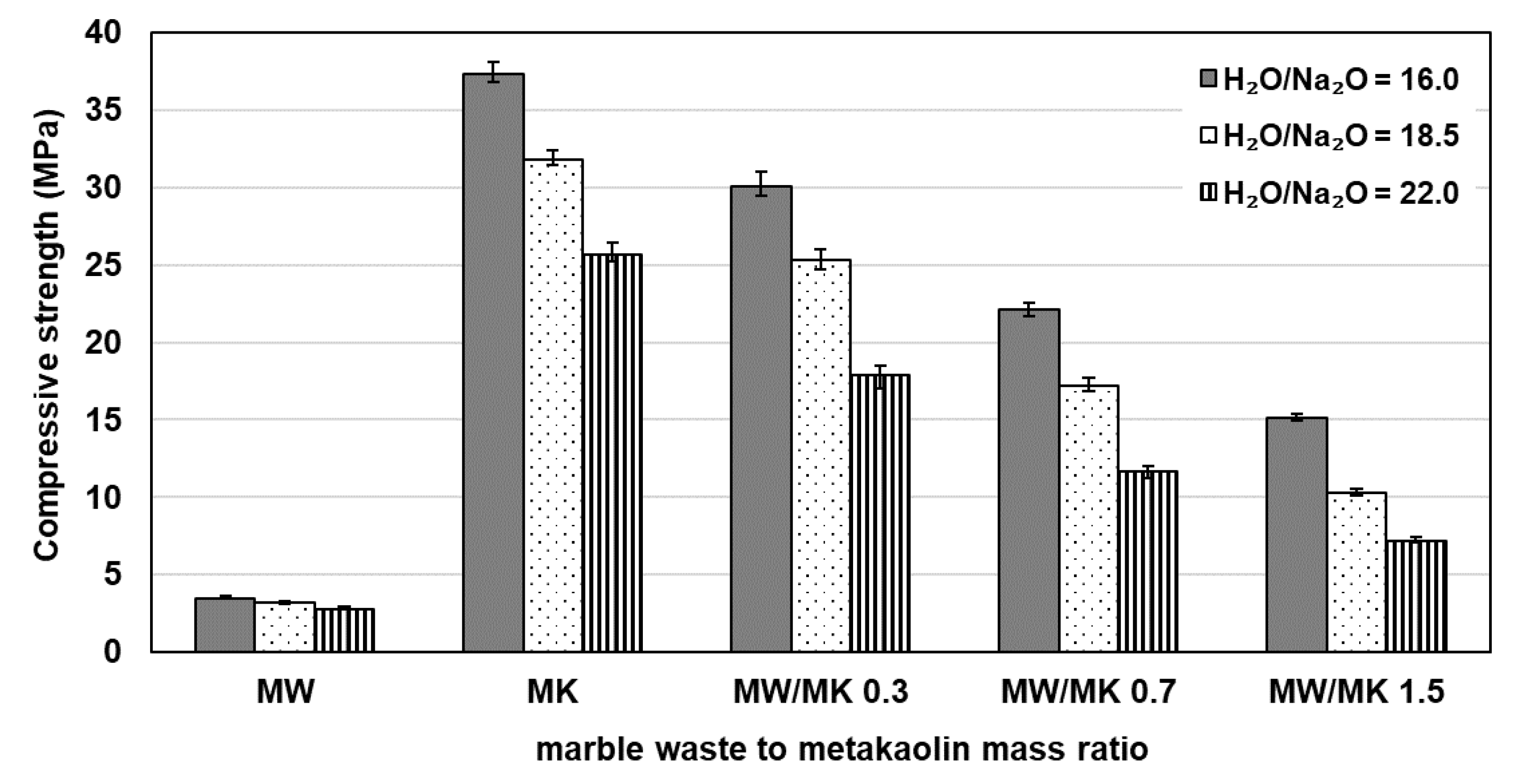
3.1. Effect of Marble Waste to Metakaolin Ratio
3.2. Effect of Curing Temperature
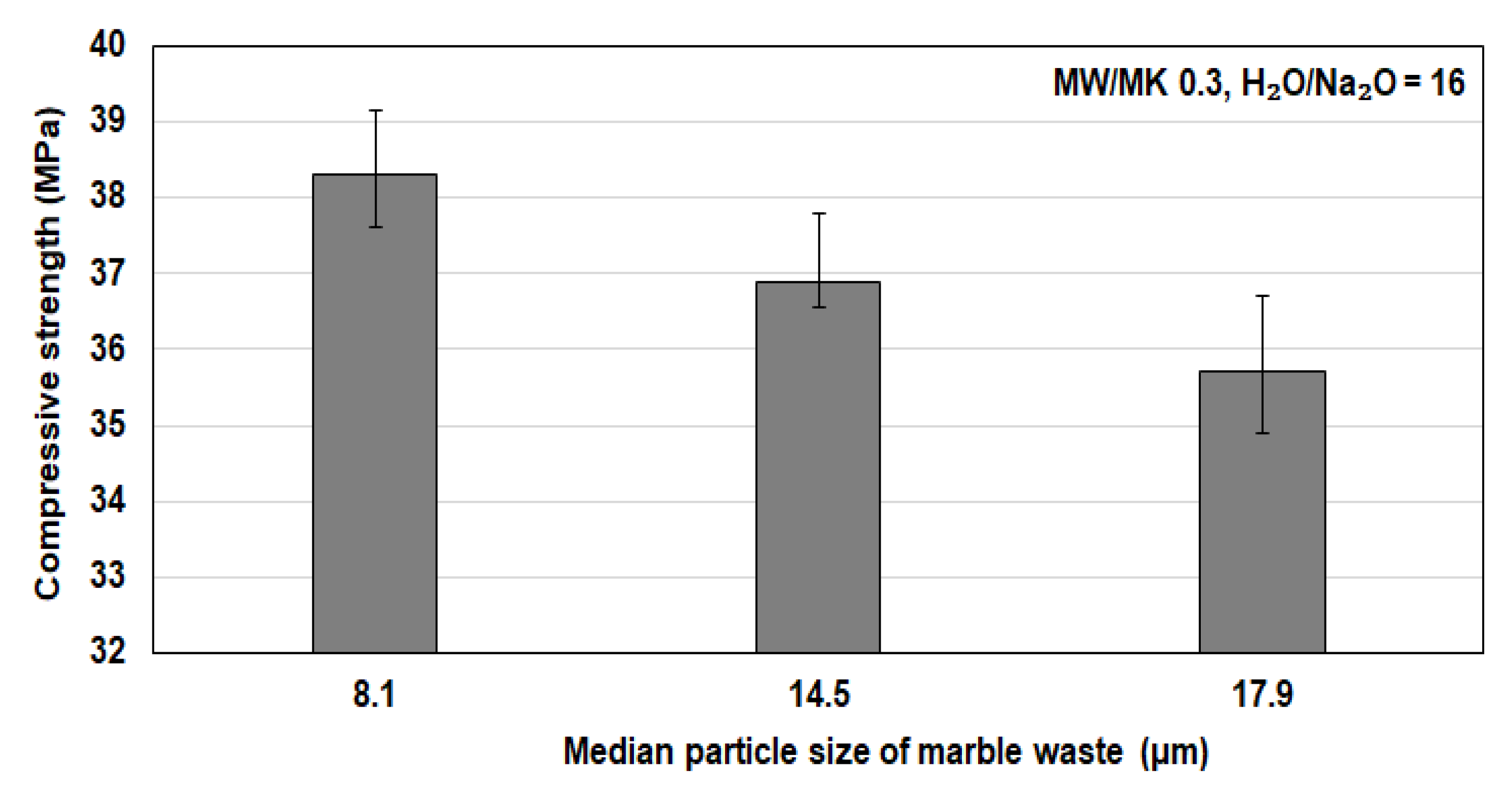
3.3. Effect of Marble Waste Particle Size on the Compressive Strength
3.4. Physical Properties of the Produced AAMs
3.5. Morphology of Selected AAMs
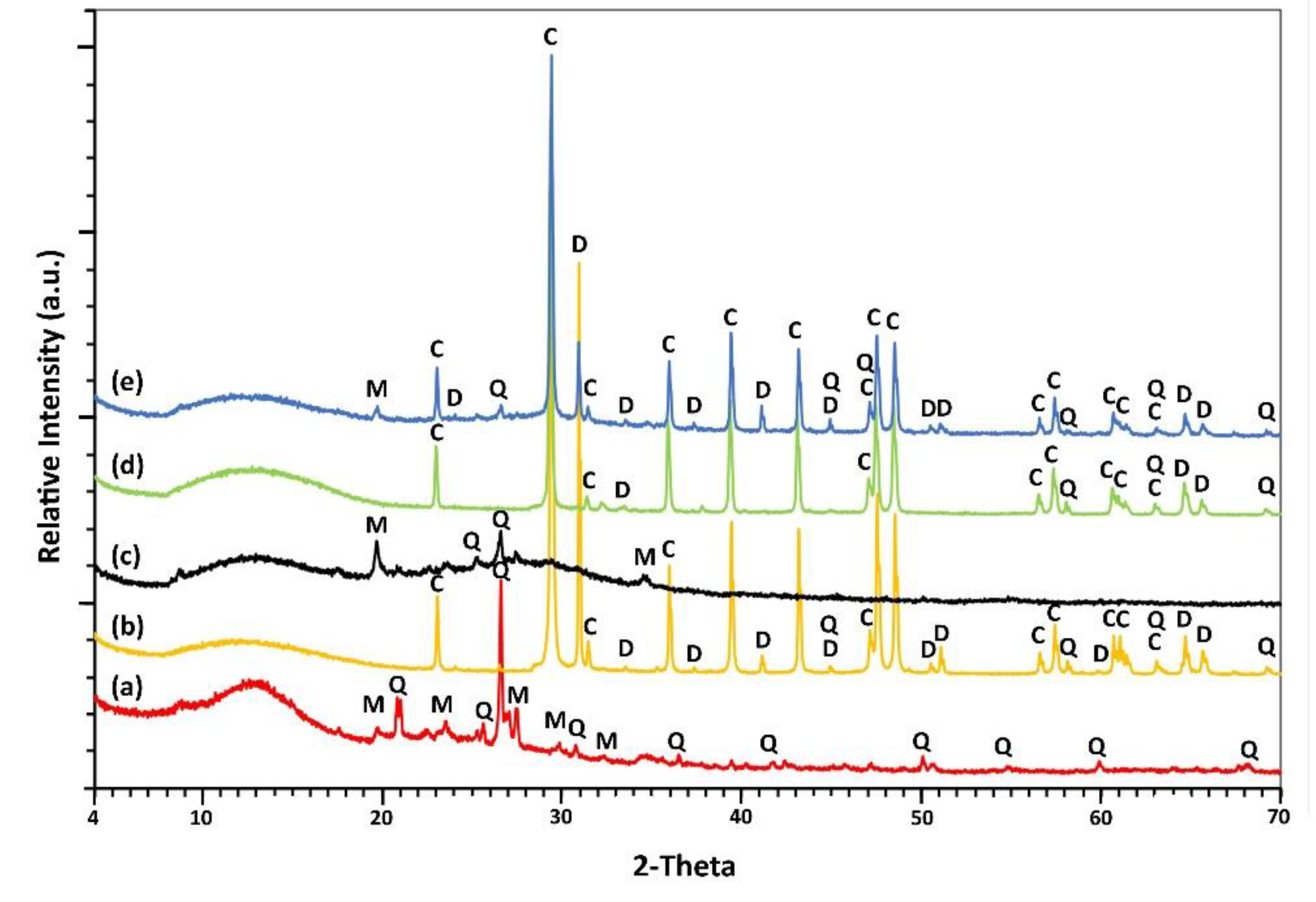
3.5.1. Mineralogical Analysis
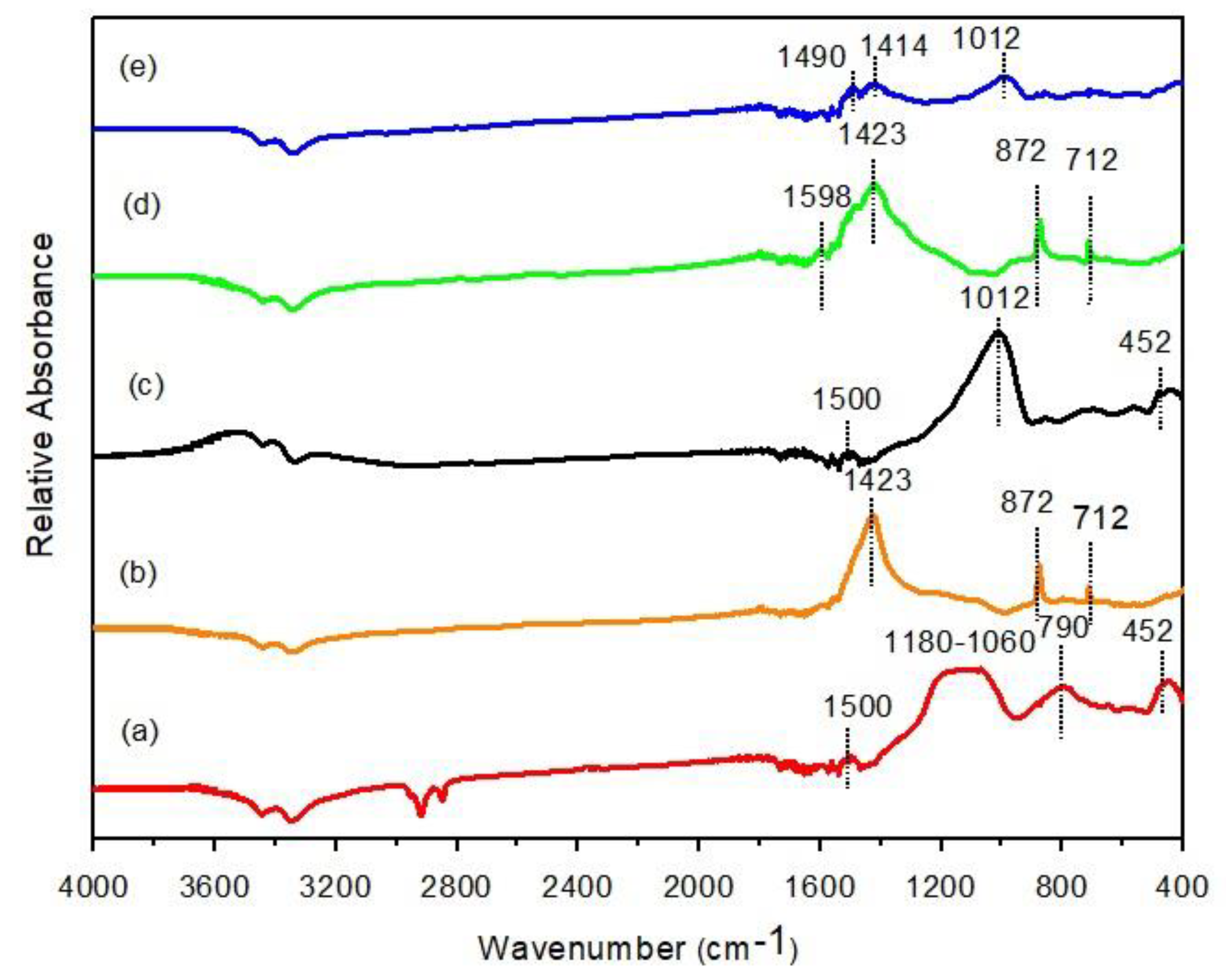
3.5.2. FTIR Analysis
3.5.3. SEM Analysis
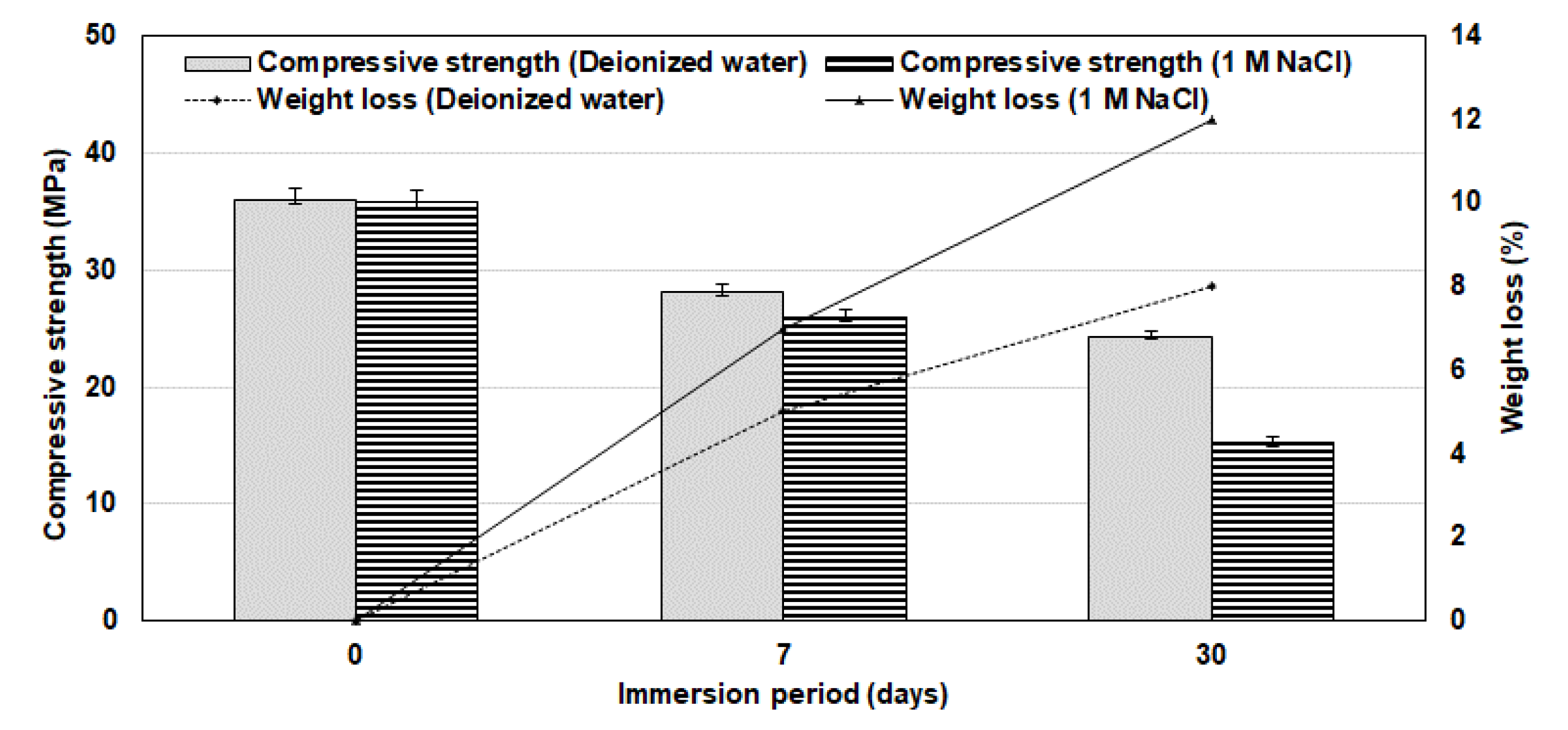
3.6. Structural Integrity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Demirel, B.; Alyamaç, K.E. Waste and Supplementary Cementitious Materials in Concrete: Characterization, Properties and Applications, 1st ed.; Woodhead Publishing: Cambridge/Sawston, UK, 2018; pp. 181–197. [Google Scholar]
- Kore, S.D.; Vyas, A.K. Impact of marble waste as coarse aggregate on properties of lean cement concrete. Case Stud. Constr. Mater. 2016, 4, 85–92. [Google Scholar] [CrossRef]
- Lee, W.H.; Lin, K.L.; Chang, T.H.; Ding, Y.C.; Cheng, T.W. Sustainable development and performance evaluation of marble-waste-based geopolymer concrete. Polymers 2020, 12, 1924. [Google Scholar] [CrossRef] [PubMed]
- Altun, N.E. Assessment of marble waste utilization as an alternative sorbent to limestone for SO2 control. Fuel Process. Technol. 2014, 128, 461–470. [Google Scholar] [CrossRef]
- Tekin, I. Properties of NaOH activated geopolymer with marble, travertine and volcanic tuff wastes. Constr. Build. Mater. 2016, 127, 607–617. [Google Scholar] [CrossRef]
- Thakur, A.K.; Pappu, A.; Thakur, V.K. Synthesis and characterization of new class of geopolymer hybrid composite materials from industrial wastes. J. Clean. Prod. 2019, 230, 11–20. [Google Scholar] [CrossRef]
- Hebhoub, H.; Aoun, H.; Belachia, M.; Houari, H.; Ghorbel, E. Use of waste marble aggregates in concrete. Constr. Build. Mater. 2011, 25, 1167–1171. [Google Scholar] [CrossRef]
- Saboya, F., Jr.; Xavier, G.C.; Alexandre, J. The use of the powder marble by-product to enhance the properties of brick ceramic. Constr. Build. Mater. 2007, 21, 1950–1960. [Google Scholar] [CrossRef]
- Khan, Z.; Umar, M.; Shahzada, S.; Arshad, A. Utilization of marble dust in fired clay bricks. J. Environ. Monit. 2017, 17, 4–10. [Google Scholar]
- Galetakis, M.; Soultana, A. A review on the utilization of quarry and ornamental stone industry fine by-products in the construction sector. Constr. Build. Mater. 2016, 102, 769–781. [Google Scholar] [CrossRef]
- Davidovits, J. Geopolymers—Inorganic polymeric new materials. J. Therm. Anal. Calorim. 1991, 37, 1633–1656. [Google Scholar] [CrossRef]
- Komnitsas, K.; Zaharaki, D.; Perdikatsis, V. Effect of synthesis parameters on the compressive strength of low-calcium ferronickel slag inorganic polymers. J. Hazard. Mater. 2009, 161, 760–768. [Google Scholar] [CrossRef] [PubMed]
- Komnitsas, K. Potential of geopolymer technology towards green buildings and sustainable cities. Procedia Eng. 2011, 21, 1023–1032. [Google Scholar] [CrossRef]
- Luukkonen, T.; Abdollahnejad, Z.; Yliniemi, J.; Kinnunen, P.; Illikainen, M. One-part alkali-activated materials: A review. Cem. Concr. Res. 2018, 103, 21–34. [Google Scholar] [CrossRef]
- Komnitsas, K.; Bartzas, G.; Karmali, V.; Petrakis, E.; Kurylak, W.; Pietek, G.; Kanasiewicz, J. Assessment of alkali activation potential of a Polish ferronickel slag. Sustainability 2019, 11, 1863. [Google Scholar] [CrossRef]
- Mohajerani, A.; Suter, D.; Jeffrey-Bailey, T.; Song, T.; Arulrajah, A.; Horpibulsuk, S.; Law, D. Recycling waste materials in geopolymer concrete. Clean Technol. Environ. Policy 2019, 21, 493–515. [Google Scholar] [CrossRef]
- Komnitsas, K.; Yurramendi, L.; Bartzas, G.; Karmali, V.; Petrakis, E. Factors affecting co-valorization of fayalitic and ferronickel slags for the production of alkali activated materials. Sci. Total Environ. 2020, 721, 137753. [Google Scholar] [CrossRef]
- Wang, Q.; Ding, Z.Y.; Da, J.; Ran, K.; Sui, Z.T. Factors influencing bonding strength of geopolymer-aggregate interfacial transition zone. Adv. Mat. Res. 2011, 224, 1–7. [Google Scholar] [CrossRef]
- Colangelo, F.; Roviello, G.; Ricciotti, L.; Ferrándiz-Mas, V.; Messina, F.; Ferone, C.; Cheeseman, C.R. Mechanical and thermal properties of lightweight geopolymer composites. Cem. Concr. Compos. 2018, 86, 266–272. [Google Scholar] [CrossRef]
- Coppola, B.; Palmero, P.; Montanaro, L.; Tulliani, J.M. Alkali-activation of marble sludge: Influence of curing conditions and waste glass addition. J. Eur. Ceram. Soc. 2020, 40, 3776–3787. [Google Scholar] [CrossRef]
- Simão, L.; Hotza, D.; Ribeiro, M.J.; Novais, R.M.; Montedo, O.R.K.; Raupp-Pereira, F. Development of new geopolymers based on stone cutting waste. Constr. Build. Mater. 2020, 257, 119525. [Google Scholar] [CrossRef]
- Petrakis, E.; Karmali, V.; Bartzas, G.; Komnitsas, K. Grinding kinetics of slag and effect of final particle size on the compressive strength of alkali activated materials. Minerals 2019, 9, 714. [Google Scholar] [CrossRef]
- Aughenbaugh, K.L.; Williamson, T.; Juenger, M.C.G. Critical evaluation of strength prediction methods for alkali-activated fly ash. Mater. Struct. 2015, 48, 607–620. [Google Scholar] [CrossRef]
- Clausi, M.; Tarantino, S.C.; Magnani, L.L.; Riccardi, M.P.; Tedeschi, C.; Zema, M. Metakaolin as a precursor of materials for applications in Cultural Heritage: Geopolymer-based mortars with ornamental stone aggregates. Appl. Clay Sci. 2016, 132, 589–599. [Google Scholar] [CrossRef]
- BS EN 1936:2006. Natural Stone Test Methods. Determination of Real Density and Apparent Density, and of Total and Open Porosity; British Standards Institution: London, UK, 2006; Available online: https://shop.bsigroup.com/ProductDetail/?pid=000000000030149240 (accessed on 8 April 2020).
- BS EN 13755:2008. Natural Stone Test Methods. Determination of Water Absorption at Atmospheric Pressure; British Standards Institution: London, UK, 2006; Available online: https://shop.bsigroup.com/ProductDetail/?pid=000000000030163234 (accessed on 8 April 2020).
- ASTM C1262–10. Standard Test Method for Evaluating the Freeze-Thaw Durability of Dry-Cast Segmental Retaining Wall Units and Related Concrete Units; ASTM International: West Conshohocken, PA, USA, 2010; Available online: https://www.astm.org/DATABASE.CART/HISTORICAL/C1262-10.htm (accessed on 6 April 2020).
- Bernal, S.A. Effect of the activator dose on the compressive strength and accelerated carbonation resistance of alkali silicate-activated slag/metakaolin blended materials. Constr. Build. Mater. 2015, 98, 217–226. [Google Scholar] [CrossRef]
- Samson, G.; Cyr, M.; Gao, X.X. Formulation and characterization of blended alkali-activated materials based on flash-calcined metakaolin, fly ash and GGBS. Constr. Build. Mater. 2017, 144, 50–64. [Google Scholar] [CrossRef]
- Jin, M.; Lian, F.; Xia, R.; Wang, Z. Formulation and durability of a geopolymer based on metakaolin/tannery sludge. Waste Manag. 2018, 79, 717–728. [Google Scholar] [CrossRef]
- Liang, G.; Zhu, H.; Zhang, Z.; Wu, Q.; Du, J. Investigation of the waterproof property of alkali-activated metakaolin geopolymer added with rice husk ash. J. Clean. Prod. 2019, 230, 603–612. [Google Scholar] [CrossRef]
- Perez-Cortes, P.; Escalante-Garcia, J.I. Alkali activated metakaolin with high limestone contents - Statistical modeling of strength and environmental and cost analyses. Cem. Concr. Compos. 2020, 106, 103450. [Google Scholar] [CrossRef]
- Sarkar, M.; Dana, K. Partial replacement of metakaolin with red ceramic waste in geopolymer. Ceram. Int. 2020. [Google Scholar] [CrossRef]
- Liu, J.; Hu, L.; Tang, L.; Ren, J. Utilisation of municipal solid waste incinerator (MSWI) fly ash with metakaolin for preparation of alkali-activated cementitious material. J. Hazard. Mater. 2021, 402, 123451. [Google Scholar] [CrossRef]
- Zaharaki, D.; Komnitsas, K.; Perdikatsis, V. Use of analytical techniques for identification of inorganic polymer gel composition. J. Mater. Sci. 2010, 45, 2715–2724. [Google Scholar] [CrossRef]
- Gado, R.A.; Hebda, M.; Łach, M.; Mikuła, J. Alkali activation of waste clay bricks: Influence of the silica modulus, SiO2/Na2O, H2O/Na2O molar ratio, and liquid/solid ratio. Materials 2020, 13, 383. [Google Scholar] [CrossRef] [PubMed]
- Soultana, A.; Valouma, A.; Bartzas, G.; Komnitsas, K. Properties of inorganic polymers produced from brick waste and metallurgical slag. Minerals 2019, 9, 551. [Google Scholar] [CrossRef]
- Bing-hui, M.; Zhu, H.; Xue-min, C.; Yan, H.; Si-yu, G. Effect of curing temperature on geopolymerization of metakaolin-based geopolymers. Appl. Clay Sci. 2014, 99, 144–148. [Google Scholar]
- Chen, L.; Wang, Z.; Wang, Y.; Feng, J. Preparation and properties of alkali activated metakaolin-based geopolymer. Materials 2016, 9, 767. [Google Scholar] [CrossRef] [PubMed]
- Mahmoodi, O.; Siad, H.; Lachemi, M.; Dadsetan, S.; Sahmaran, M. Development of normal and very high strength geopolymer binders based on concrete waste at ambient environment. J. Clean. Prod. 2020, 279, 123436. [Google Scholar] [CrossRef]
- Provis, J.L.; Arbi, K.; Bernal, S.A.; Bondar, D.; Buchwald, A.; Castel, A.; Chithiraputhiran, S.; Cyr, M.; Dehghan, A.; Dombrowski-Daube, K.; et al. RILEM TC 247-DTA round robin test: Mix design and reproducibility of compressive strength of alkali-activated concretes. Mater. Struct. 2019, 52, 99. [Google Scholar] [CrossRef]
- Komnitsas, K.; Zaharaki, D.; Vlachou, A.; Bartzas, G.; Galetakis, M. Effect of synthesis parameters on the quality of construction and demolition wastes (CDW) geopolymers. Adv. Powder Technol. 2015, 26, 368–376. [Google Scholar] [CrossRef]
- Vogt, O.; Ukrainczyk, N.; Ballschmiede, C.; Koenders, E. Reactivity and Microstructure of Metakaolin Based Geopolymers: Effect of Fly Ash and Liquid/Solid Contents. Materials 2019, 12, 3485. [Google Scholar] [CrossRef]
- Tan, Q.W.; Li, N.; Xu, Z.H.; Chen, X.Y.; Peng, X.; Shuai, Q.; Yao, Z.Z. Comparative performance of cement and metakaolin based-geopolymer blocks for strontium immobilization. J. Ceram. Soc. Jpn. 2019, 127, 44–49. [Google Scholar] [CrossRef]
- Król, M.; Minkiewicz, J.; Mozgawa, W. IR spectroscopy studies of zeolites in geopolymeric materials derived from kaolinite. J. Mol. Struct. 2016, 1126, 200–206. [Google Scholar] [CrossRef]
- Zapata, P.A.; Palza, H.; Díaz, B.; Armijo, A.; Sepúlveda, F.; Ortiz, J.A.; Ramírez, M.P.; Oyarzún, C. Effect of CaCO₃ nanoparticles on the mechanical and photo-degradation properties of LDPE. Molecules 2018, 24, 126. [Google Scholar] [CrossRef] [PubMed]
- Beleuk à Moungam, L.; Mohamed, H.; Kamseu, E.; Billong, N.; Melo, U. Properties of geopolymers made from fired clay bricks wastes and rice husk ash (RHA)-Sodium hydroxide (NaOH) activator. Mater. Sci. Appl. 2017, 8, 537–552. [Google Scholar] [CrossRef]
- Zaharaki, D.; Galetakis, M.; Komnitsas, K. Valorization of construction and demolition (C&D) and industrial wastes through alkali activation. Constr. Build. Mater. 2016, 121, 686–693. [Google Scholar]
- Bignozzi, M.C.; Manzi, S.; Lancellotti, I.; Kamseu, E.; Barbieri, L.; Leonelli, C. Mix-design and characterization of alkali activated materials based on meta-kaolin and ladle slag. Appl. Clay Sci. 2013, 73, 78–85. [Google Scholar] [CrossRef]
- Li, Y.P.; Shen, L.; Mirmoghtadaei, R.; Ai, L. A design of experiment approach to study the effects of raw material on the performance of geopolymer concrete. Adv. Civ. Eng. Mater. 2017, 6, 526–549. [Google Scholar] [CrossRef]
- Valentini, L.; Silvia Contessi, S.; Dalconi, M.C.; Zorzi, F.; Enrico Garbin, E. Alkali-activated calcined smectite clay blended with waste calcium.carbonate as a low-carbon binder. J. Clean. Prod. 2018, 184, 41–49. [Google Scholar] [CrossRef]
- Konno, H.; Nanri, Y.; Kitamura, M. Crystallization of aragonite in the causticizing reaction. Powder Technol. 2002, 123, 33–39. [Google Scholar] [CrossRef]
- Aboulayt, A.; Riahi, M.; Ouazzani Touhami, M.; Hannache, H.; Gomina, M.; Moussa, R. 2017. Properties of metakaolin based geopolymer incorporating calcium carbonate. Adv. Powder Technol. 2017, 28, 2393–2401. [Google Scholar] [CrossRef]
- Cwirzen, A.; Provis, J.L.; Penttala, V.; Habermehl-Cwirzen, K. 2014. The effect of limestone on sodium hydroxide-activated metakaolin-based geopolymers. Construct. Build. Mater. 2014, 66, 53–62. [Google Scholar] [CrossRef]
- Tekin, I.; Gencel, O.; Gholampour, A.; Oren, O.H.; Koksal, F.; Ozbakkaloglu, T. Recycling zeolitic tuff and marble waste in the production of ecofriendly geopolymer concretes. J. Clean. Prod. 2020, 268, 122298. [Google Scholar] [CrossRef]
- Komnitsas, K. Co-valorization of marine sediments and construction & demolition wastes through alkali activation. J. Environ. Chem. Eng. 2016, 4, 4661–4669. [Google Scholar]
- Wang, Y.S.; Alrefaei, Y.; Dai, J.G. Silico-aluminophosphate and alkali-aluminosilicate geopolymers: A comparative review. Front. Mater. 2019, 6, 106. [Google Scholar] [CrossRef]
- Jaya, N.A.; Al Bakri Abdullah, M.M.; Li, L.Y.; Sandu, A.V.; Hussin, K.; Ming, L.Y. Durability of metakaolin geopolymers with various sodium silicate/sodium hydroxide ratios against seawater exposure. AIP Conf. Proc. 2017, 1887, 020063. [Google Scholar]
- Azarsa, P.; Gupta, R. Freeze-thaw performance characterization and leachability of potassium-based geopolymer concrete. J. Compos. Sci. 2020, 4, 45. [Google Scholar] [CrossRef]
- Garcia-Lodeiro, I.; Carcelen-Taboada, V.; Fernández-Jiménez, A.; Palomo, A. Manufacture of hybrid cements with fly ash and bottom ash from a municipal solid waste incinerator. Constr. Build. Mater. 2016, 105, 218–226. [Google Scholar] [CrossRef]




| Particle Size | Marble Waste | Metakaolin | ||
|---|---|---|---|---|
| Size 1 | Size 2 | Size 3 | ||
| d90 | 100.9 | 86.8 | 48.9 | 25.5 |
| d50 | 17.9 | 14.5 | 8.1 | 8.8 |
| d10 | 1.1 | 1.0 | 0.6 | 2.1 |
| Oxides | Marble Waste | Metakaolin |
|---|---|---|
| SiO2 | 0.9 | 55.9 |
| Al2O3 | 0.1 | 38.5 |
| CaO | 53.1 | 0.0 |
| Na2O | 0.7 | 0.6 |
| Fe2O3 | 0.5 | 0.5 |
| MgO | 1.5 | 0.0 |
| K2O | 0.0 | 2.1 |
| LOI * | 41.8 | 1.6 |
| Total | 98.6 | 99.2 |
| Marble Waste | Metakaolin | NaOH | H2O | Na2SiO3 | SiO2/Al2O3 | Na2O/SiO2 | H2O/Na2O | L/S |
|---|---|---|---|---|---|---|---|---|
| 60 | - | 6 | 15 | 19 | 15.3 | 1.1 | 14.9 | 0.53 |
| - | 54 | 6 | 17 | 23 | 2.4 | 1.0 | 16.0 | 0.66 |
| 13 | 49 | 3 | 16 | 19 | 2.9 | 0.8 | 22.0 | 0.54 |
| 13 | 49 | 4 | 15 | 19 | 2.9 | 0.9 | 18.5 | 0.51 |
| 13 | 49 | 5 | 14 | 19 | 2.9 | 1.0 | 16.0 | 0.49 |
| 25 | 37 | 3 | 16 | 19 | 3.0 | 0.8 | 22.0 | 0.54 |
| 25 | 37 | 4 | 15 | 19 | 3.0 | 0.9 | 18.5 | 0.51 |
| 25 | 37 | 5 | 14 | 19 | 3.0 | 1.0 | 16.0 | 0.49 |
| 37 | 25 | 3 | 16 | 19 | 3.2 | 0.8 | 22.0 | 0.54 |
| 37 | 25 | 4 | 15 | 19 | 3.2 | 0.9 | 18.5 | 0.51 |
| 37 | 25 | 5 | 14 | 19 | 3.2 | 1.0 | 16.0 | 0.49 |
| AAM Code | Porosity (%) | Density (kg/m3) | Water Absorption (%) | Compressive Strength (MPa) |
|---|---|---|---|---|
| MK | 20.6 | 1790 | 11.3 | 40.3 |
| MW | 33.9 | 1483 | 28.7 | 3.9 |
| MW/MK 0.3 | 25.1 | 1611 | 14.2 | 35.7 |
| Raw Materials | H2O/Na2O Molar Ratio | Curing Conditions | Compressive Strength (Mpa) | References |
|---|---|---|---|---|
| Marble waste, metakaolin, epoxy resin, expanded polystyrene | 11.0 | 40 °C, 24 h | 6.0 | [19] |
| Ornamental stones, metakaolin, standard siliceous sand | 10.0 | 20 °C, 90 % relative humidity, 28 days | 72.0 | [24] |
| Marble and volcanic stones cutting waste, metakaolin | 13.0 | Room temperature, 28 days | 28.6 | [21] |
| Marble waste, metakaolin | 16.0 | 40 °C, 24 h | 35.7 | This study |
| Temperature (°C) | Compressive Strength (MPa) | Weight Loss (%) |
|---|---|---|
| 40 | 35.7 | - |
| 200 | 16.2 | 24.6 |
| 400 | 7.5 | 28.4 |
| 600 | 1.6 | 33.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Komnitsas, K.; Soultana, A.; Bartzas, G. Marble Waste Valorization through Alkali Activation. Minerals 2021, 11, 46. https://doi.org/10.3390/min11010046
Komnitsas K, Soultana A, Bartzas G. Marble Waste Valorization through Alkali Activation. Minerals. 2021; 11(1):46. https://doi.org/10.3390/min11010046
Chicago/Turabian StyleKomnitsas, Konstantinos, Athanasia Soultana, and Georgios Bartzas. 2021. "Marble Waste Valorization through Alkali Activation" Minerals 11, no. 1: 46. https://doi.org/10.3390/min11010046
APA StyleKomnitsas, K., Soultana, A., & Bartzas, G. (2021). Marble Waste Valorization through Alkali Activation. Minerals, 11(1), 46. https://doi.org/10.3390/min11010046







