4.1. Melting Curve of KCl
The melting temperatures of KCl measured in this study are in general agreement with the literature data (
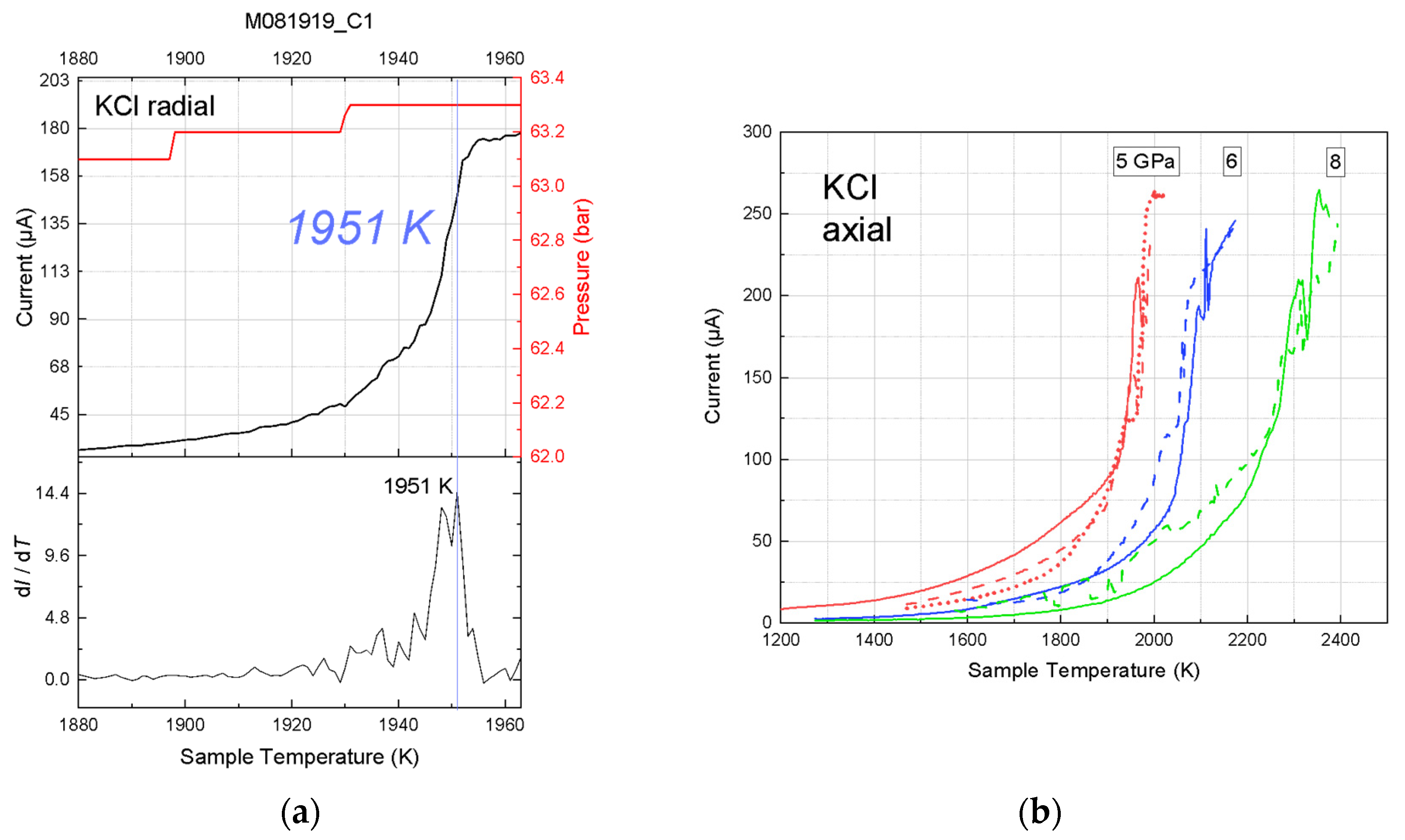
Figure 5). Where the pressure ranges overlap, the results of our radial-TC experiments agree with the existing data from piston-cylinder experiments [
3,
10], within uncertainties. Between 5 and 8 GPa, the results of our ionic conduction experiments are higher by 20–90 K, but marginally agree with the Simon–Glatzel melting curve [
11] fitted through all the literature data on KCl in the simple cubic B2 structure between 1.8 and 50 GPa, which can be described by
, where
T is in K,
P is in GPa, and the numbers in parentheses are uncertainties in the last digits.
Excluding the literature data at pressures above 20 GPa from diamond anvil cell (DAC) experiments [
2], the fit becomes
. This fit to the data from large volume apparatus alone diverge from the all-data fit at pressures above 10 GPa and differ by as much as 500 K at 25 GPa. The divergence confirms the notion that the Simon’s equation is empirical and cannot be used for extrapolating melting curves beyond the experimental pressure range, although it is widely used for interpolation because the formulation is simpler than other melting laws and requires no knowledge of the equation-of-state of the solid phase. Indeed, a previous study on NaCl found that the Simon’s law equation fit to data between 1 bar and 6.5 GPa overpredicts the melting temperature at 20 GPa by nearly 100 K [
4]. On the other hand, more recent DAC data on the melting curve of water questioned the accuracy of data from piston-cylinder experiments [
12]. Furthermore, the melting temperatures of iron determined in earlier DAC experiments were found to be overestimated by hundreds of degrees [
13]. Therefore, independent measurements at pressures below 10 GPa and new data at pressures above 10 GPa are needed to validate the melting curve of KCl fitted to all the existing data.
4.2. Melting Detection from Ionic Conduction
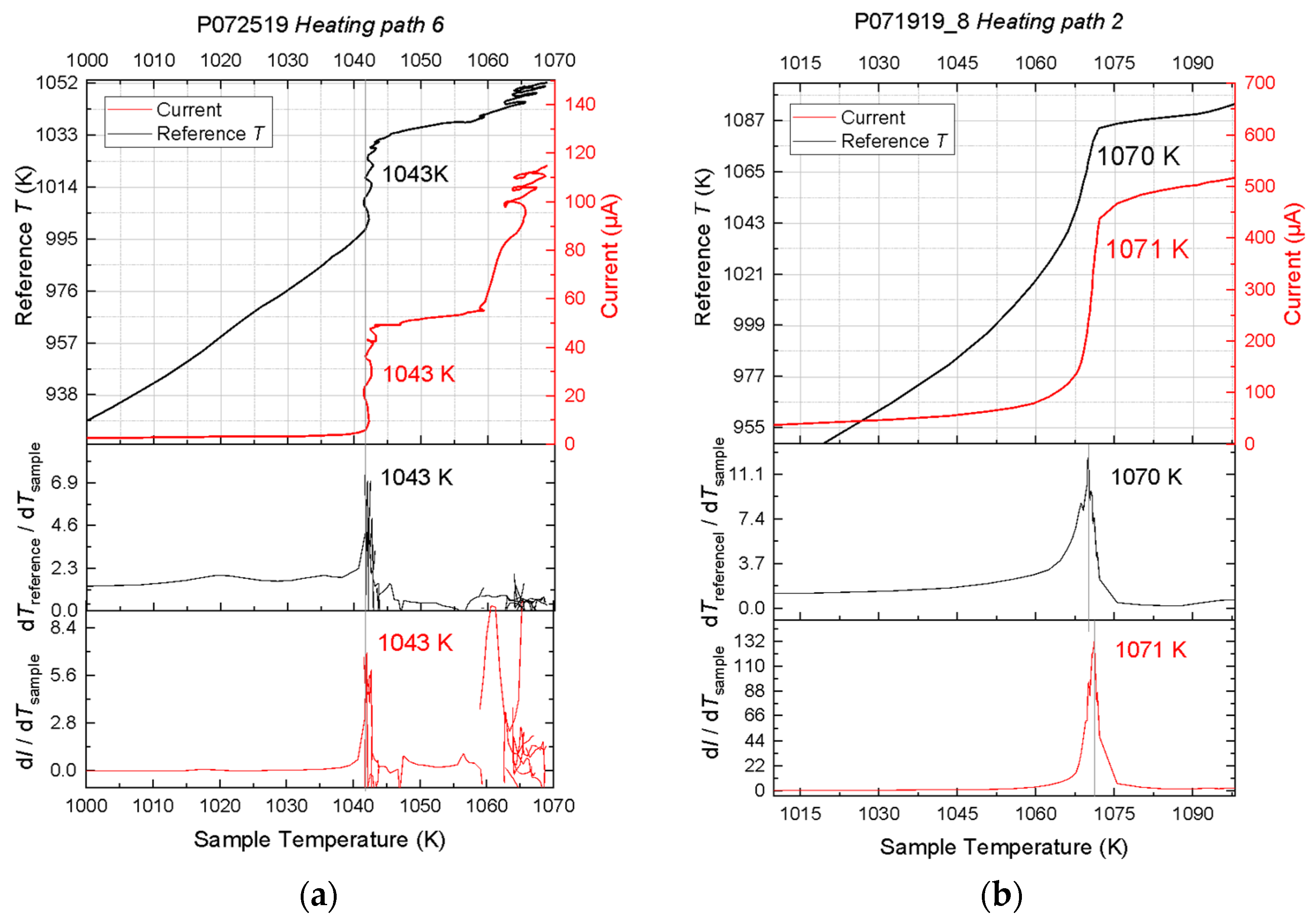
The concurrent experiments at 1 bar demonstrate that ionic conduction measurements can be used to determine the melting temperature of ionic compounds. Where high-quality data were acquired at 1 bar, we observed simultaneous occurrences of latent heat ledge and peak rate of current rise with temperature, thus firmly establishing the criterion of melting detection from ionic conduction observations. The validity of this criterion is further supported by confirmation from the sinking of Pt sphere, movement of melt, and growth of large crystals from melt upon cooling.
The recorded current-temperature curves are not always of high quality, even at the ambient pressure. Random errors originated from several sources, including melt motion, cell deformation, and pressure drift with temperature. At 1 bar, the migration of melt along the wires or container wall altered the contact location and area of the electrodes, thus producing noise in the current reading. In the high-pressure experiments, components of the high-pressure assembly had diverse ranges of thermoelastic properties and porosities, and therefore responded to compression and heating differently. The resulting stress and deformation produced noise in the current and temperature signals. Furthermore, the current-temperature curve could have been broadened by pressure drift at high temperature. In some of our axial placement experiments, the current-temperature curves were not smooth and the rate of current rise with temperature fluctuated. As a result, the peak in the derivative plot was not as sharp and prominent as expected. Nevertheless, these data gave reasonable constraints on the melting temperature, which increased as a function of pressure, as expected.
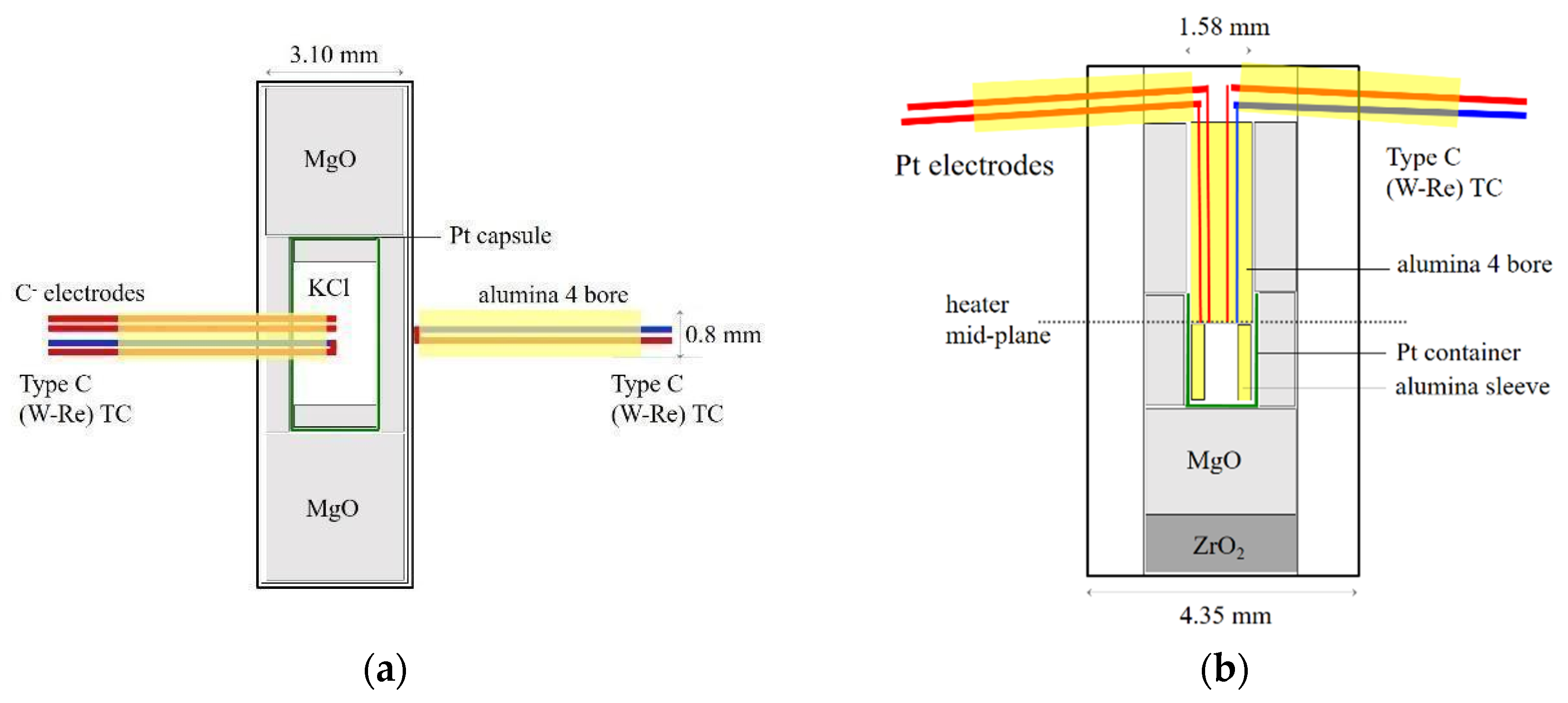
Compared with the symmetric configuration used in previous conduction experiments (e.g., Dong et al., 2019 [
6]), the single-side placement of thermocouple and electrodes reduce uncertainties in temperature measurements associated with temperature gradient. Furthermore, the placement of the thermocouple junction and electrode tips at the heater waist allows for detection of the first melt in the sample chamber. By limiting the amount of melt, this configuration minimizes heater deformation associated with melt movement, which may be significant before pore space is not fully closed. Heater deformation leads to perturbation of the temperature distribution and may cause shorting. With the thermocouple junction placed directly in the sample, the single-side placement has the additional advantage of accounting for the latent heat effect on the sample temperature. However, chemical contamination of the thermocouple could produce false signals. In particular, the W-Re based type C wires are known to be vulnerable to oxidation and they react with carbonate melts [
5]. The type S wires are more chemical inert, but they are less refractory and do not work at temperatures beyond ~2300 K.
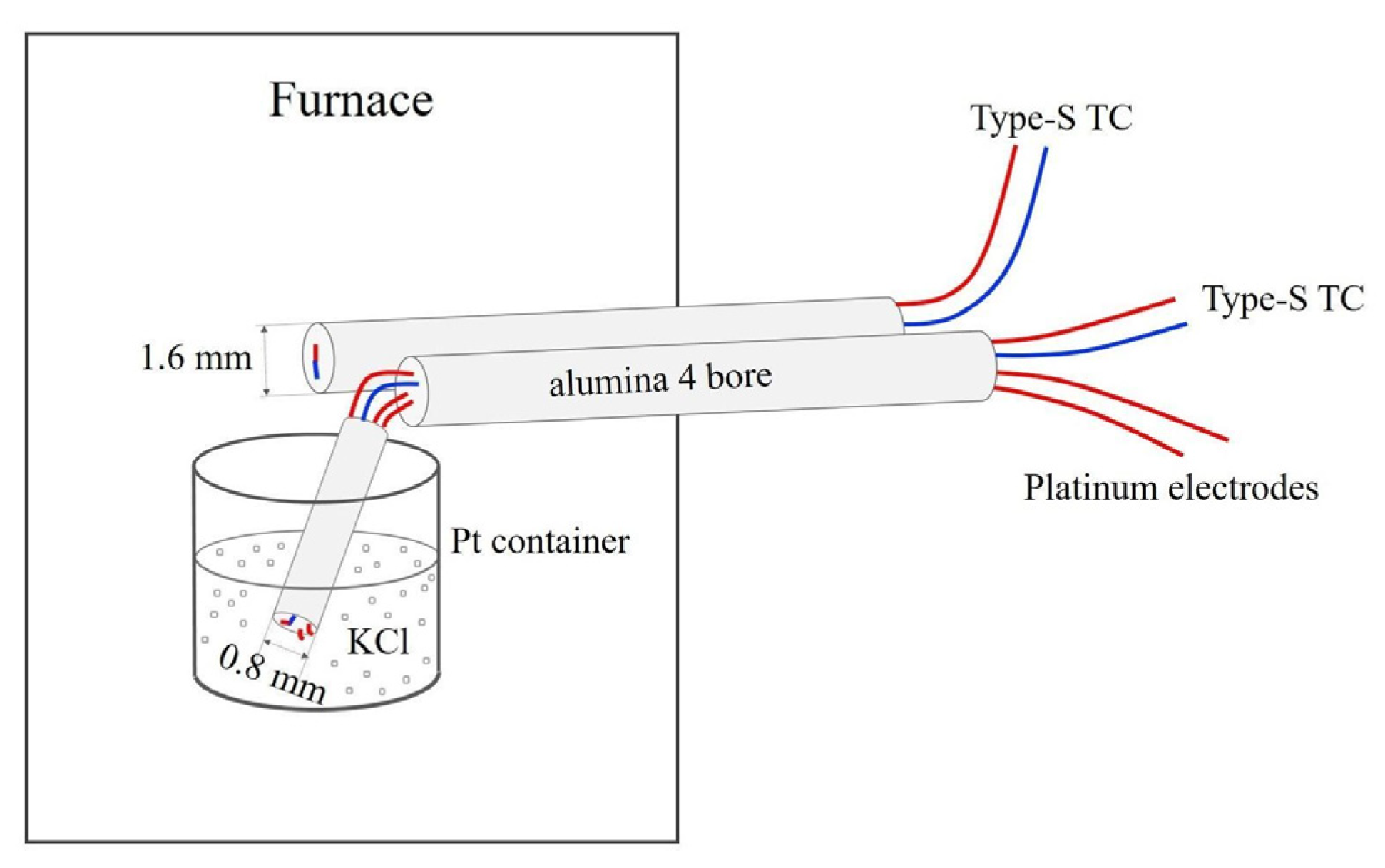
The axial and radial placement of thermocouple and electrodes each has its advantages and disadvantages. Installing the thermocouple requires similar levels of skills in the axial and radial placements, but the axial placement involves cutting two deep grooves in the octahedron and filling extra space with cement after installing the thermocouple, whereas the radial placement only involves drilling one bore through a fin of the octahedron. The radial placement ensures that the thermocouple junction and electrode tips are located at the hottest part of the heater to detect the first melt. Perforation in the heater causes larger temperature gradient and hot spots because each wire from the hot zone to the exterior acts as a heat sink that perturbs the thermal field. Moreover, melt may leak out from the perforation at the heater equator. However, the four-bore holder in the radial placement has a diameter of 0.80 mm so that the thermocouple junction and the electrode tips are less than 0.2 mm apart. This configuration allows reliable melting detection even in the presence of relatively large temperature gradients. Such a small holder can only fit 5 mil wires. The thinner wires lose less heat but are more fragile and break easily at a few GPa. In this study, we placed the second thermocouple just outside the heater in an attempt to observe the latent heat associated with melting in the radial experiments. These two wires can be left out in the radial placement experiment.
A successful conduction experiment requires the survival of the thermocouple and electrode wires and separation of the electrodes. In the KCl experiments, we used the W-Re-based type C wires, which are much stronger than the type S wires and therefore less likely to break or short, but they are more chemically reactive and cannot be used for carbonates such as CaCO
3 [
5]. The Pt-Rh-based type S thermocouples are soft and prone to failure due to breaking or shorting. In the axial configuration, space is available to accommodate wires with 8 mil or even 10 mil diameters. If correctly placed, these wires are sufficiently thick to survive compression to 20 GPa, but the thicker electrodes short more easily. Shorting may occur upon compression, when the Pt container and malleable wires deform under stress. Chance of shorting increases at high temperature, when the wires soften and flow more readily. The movement of melt may also lead to shorting.
The ionic conduction method has a number of advantages over the differential thermal analysis (DTA) method. At 1 bar, the latent heat ledge may be missing if the sample is heated up too fast or too slow, but the steep rise in ionic current is not affected by the heating rate. The latent heat effect was not observed in the high-pressure experiments, possibly because the sample mass was too small, and the signal was overwhelmed by the thermal response of the surrounding materials in the high-pressure assembly. Thermopile amplification used for differential scanning calorimetry studies at high pressures [
14,
15] may allow us to observe the latent heat ledge associated with melting. Alternatively, we can apply the ionic conduction method to detect melting.
Compared with the ionic conduction method, applying the Pt sphere maker method at high pressures is labor-intensive. Theoretically the melting point at a given pressure can be bracketed by two experiments, but in reality, many more experiments are needed to locate the melting temperature, even if all are successful. Duplicate experiments may produce contradictory results because of uncertainties in sample placement in a temperature gradient. With limited width and length of the sample, Pt spheres could easily get stuck to the container wall or trapped in the thin layer of melt, and therefore produce no results.
Admittedly, ion conduction measurement at high pressure is challenging because it requires well controlled sample geometry. All the parts need to be aligned and closely fitted to ensure success. Our current success rate is about one third for users with average skills. However, this type of experiment is highly efficient when successful. Our study showed that melting produces a steep rise in ionic current with temperature, but we cannot exclude the possibility of an abrupt current rise resulting from other changes such as decomposition or chemical reaction. For these reasons, it is important to examine the experiment product for chemical changes, and to validate the melting points from conduction experiments by sphere marker experiments at selected pressures.
4.3. Application of KCl in High Pressure Research
The proposed melting curve of KCl informs its potential use as a pressure calibrant for large-volume press or material for pressure transmission, thermal insulation, and sample containment in high pressure research.
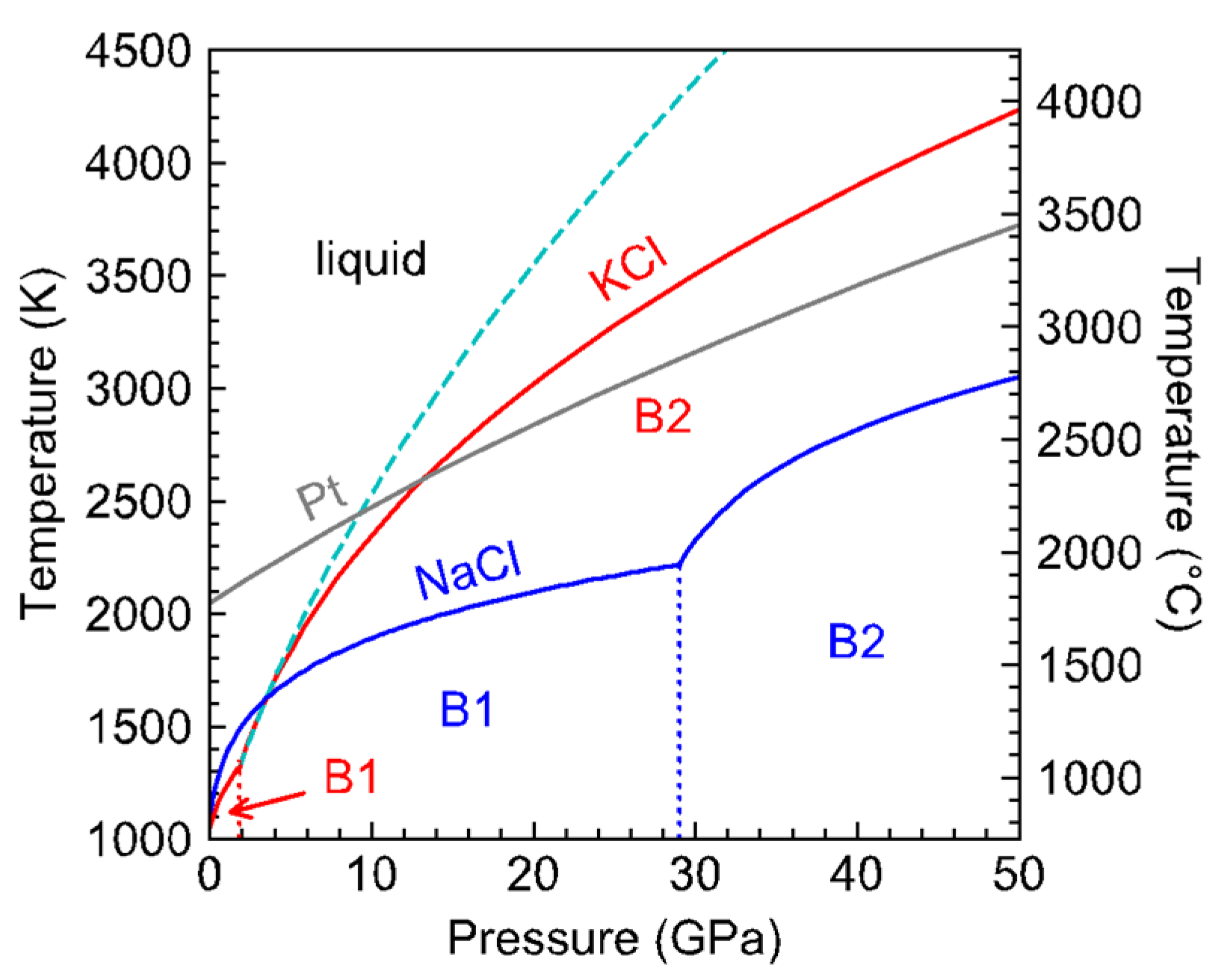
The melting curve of NaCl has been used to calibrate the relationship between press load and sample pressure in piston-cylinder and large-volume press. Such calibration revealed notable deviation from pressure calibration established at 1473 K in experiments using the 646-pressure medium [
7]. At 7 GPa and ~2000 K, the thermal pressure reached as much as 2 GPa, which can be explained by the low thermal conduction of zirconia-based 646 ceramics. While NaCl works well as a pressure calibrant at pressures up to ~7 GPa, it loses resolution at higher pressures, where its melting curve becomes relatively flat (
Figure 6). Between 1 bar and ~9 GPa, the melting point of KCl increases with pressure at an average rate of ~150 K/GPa. With such a large melting slope, KCl may be used as a high-resolution pressure calibrant at moderate pressures and high temperatures. Note that the reference melting curve of KCl carries an uncertainty of ±100 K and the measured melting temperature bears a comparable uncertainty of ±100 K with the current design of ionic conduction experiment including the uncertainties in temperature measurements, the peak width of d
I/d
T, and reproducibility among multiple heating cycles (
Table 2). As a result, the uncertainty in pressure calibration is at least ±0.7 GPa, which can be improved when the KCl melting curve is more tightly constrained and a better design of ionic conduction experiment enables more accurate and precise melting detection.
Previous studies showed that pressure calibration of multi-anvil experiments is affected by temperature and heating cycle. Leinenweber et al. (2012) [
8] found that the COMPRES 10/5 assembly experienced a pressure maximum during heating. At 15 to 20 GPa, the sample pressure varied by as much as 1.5 GPa between room temperature and 1900 K. More significantly, at 20–30 GPa, the COMPRES 8/3 assembly lost 5 to 10 GPa during heating between room temperature and 2300–2600 K [
8]. On the other hand, the sample pressure increased by 2 GPa between 1500 and 2300 K at a press load of 1400 ton, after a temperature cycle at 680 ton [
16]. Such large pressure variations with temperature and heating cycles suggest that some of the discrepancies in the measured melting temperatures between multiple heating cycles may originate from pressure change at a fixed press load, in addition to other sources such as melt movement and heater deformation. The reproducibility of the measured melting temperatures between repeated cycles and the agreement between our results and literature data on the melting curve of KCl suggests that in this study the deviations of sample pressures from the pressure calibrations are within the estimated uncertainties.
We propose that the ionic conduction method may be used to quantify the effects of temperature and heating cycle on pressure calibration. Although in its current state this method still bears relatively large uncertainties and may have limited resolutions at high pressures, it offers a potential solution when synchrotron radiation is not available or opening an X-ray window in the cell is not desirable.
The divergence between the melting curves of NaCl and KCl is interesting (
Figure 6). At 1 bar, they are both in the B1 face-centered cubic structure. As expected from the systematic relationship between the melting temperature and ionic radii [
20], KCl melts at a lower temperature than NaCl and transforms into the simple cubic B2 structure at a lower pressure than NaCl. The B1–B2 transition is associated with a volume reduction, which may have led to steepening of the melting slope, as observed in the melting curve of SiO
2 upon the transition from high-quartz to coesite and from coesite to stishovite [
21].
While NaCl remains in the B1 structure with small increase of melting temperature with pressure, the melting curve of KCl crosses that of NaCl near 3 GPa. Between 3 and 9 GPa, the melting point of KCl continues to rise steeply to cross that of Pt near 9 GPa (
Figure 6). The highly refractory nature of KCl at moderate pressures may provide value for high-pressure research. In diamond-anvil cell experiments NaCl and KCl are widely used as pressure medium because they are transparent and have low shear moduli. They also serve as thermal insulating layer for laser-heating experiments. If the sample of interest is a melt, then a refractory medium or insulator may be preferred for stability.
Finding a suitable container is also important for high temperature experiments on materials synthesis phase equilibria and chemical reactions. For sample containment, KCl possesses a unique combination of characteristics: It is water-soluble, relatively soft, and electrically insulating in the solid state, and highly refractory at moderate pressure, therefore it is a promising option along with commonly used container materials such as graphite, MgO, BN, and various metals.