Research on Behavior of Iron in the Zinc Sulfide Pressure Leaching Process
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Leaching Process
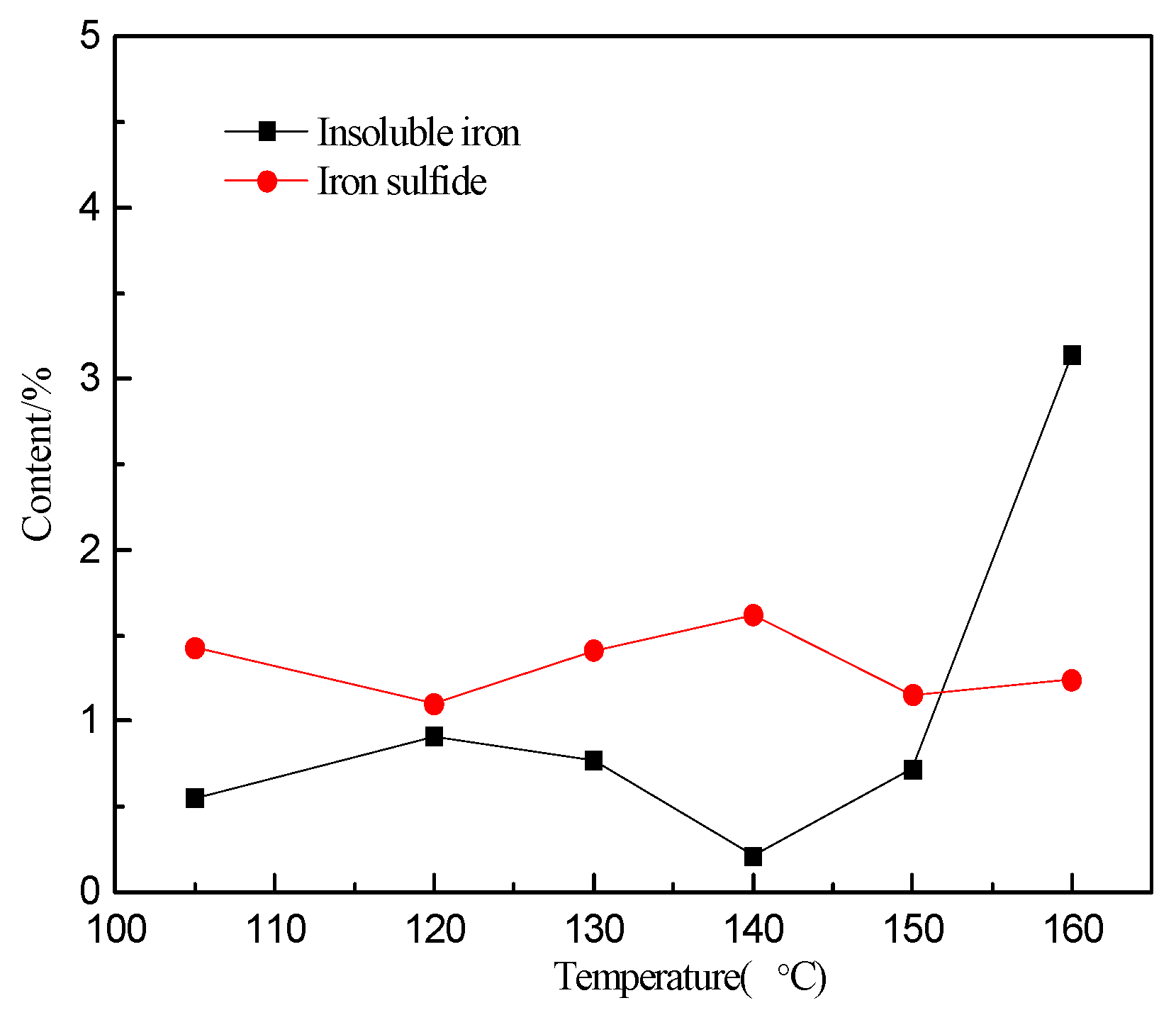
2.3. Calculation of Iron Deportment
2.4. Analysis and Detection
3. Results and Discussion
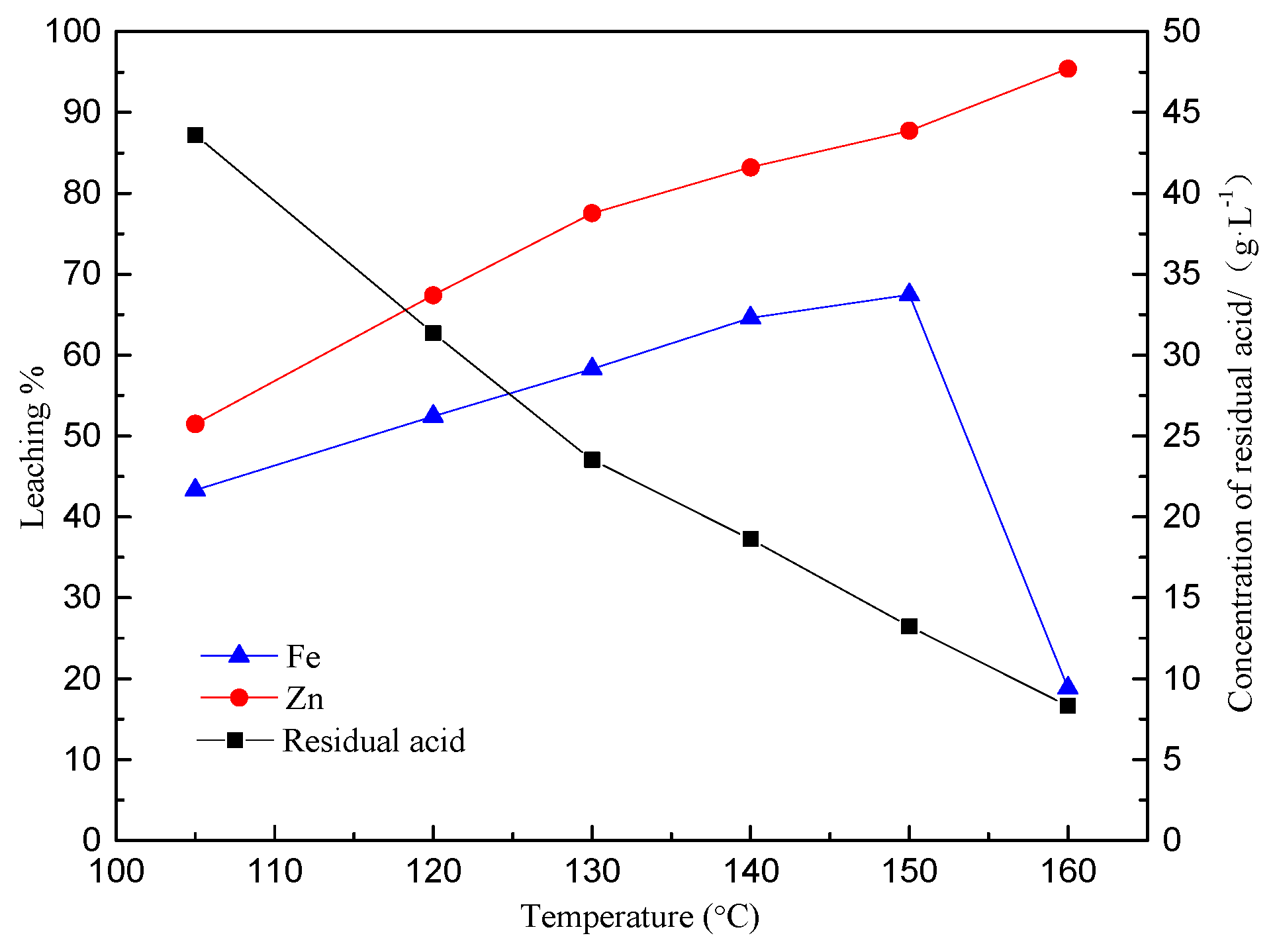
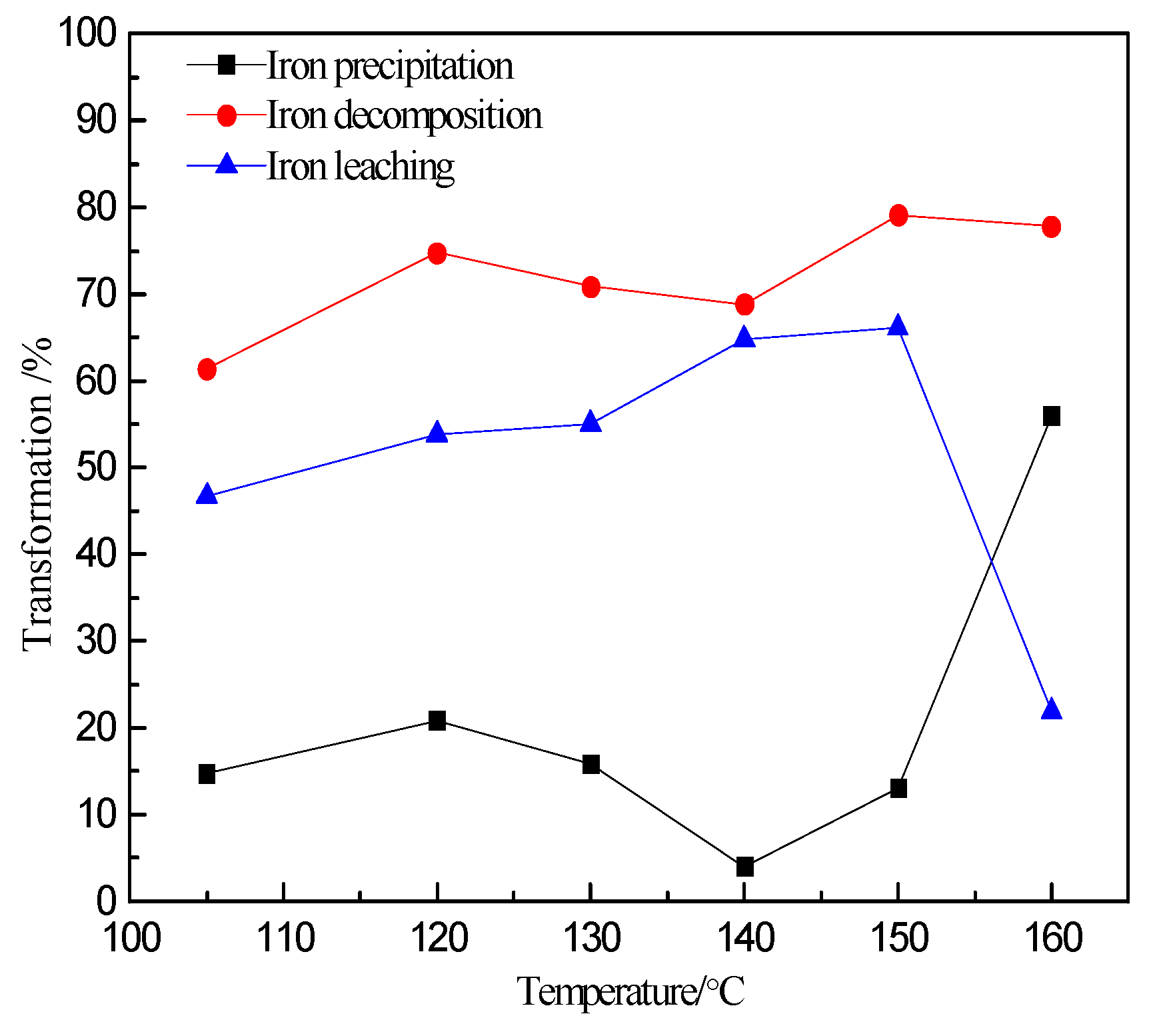
3.1. Effect of Temperature on Zinc and Iron Extraction
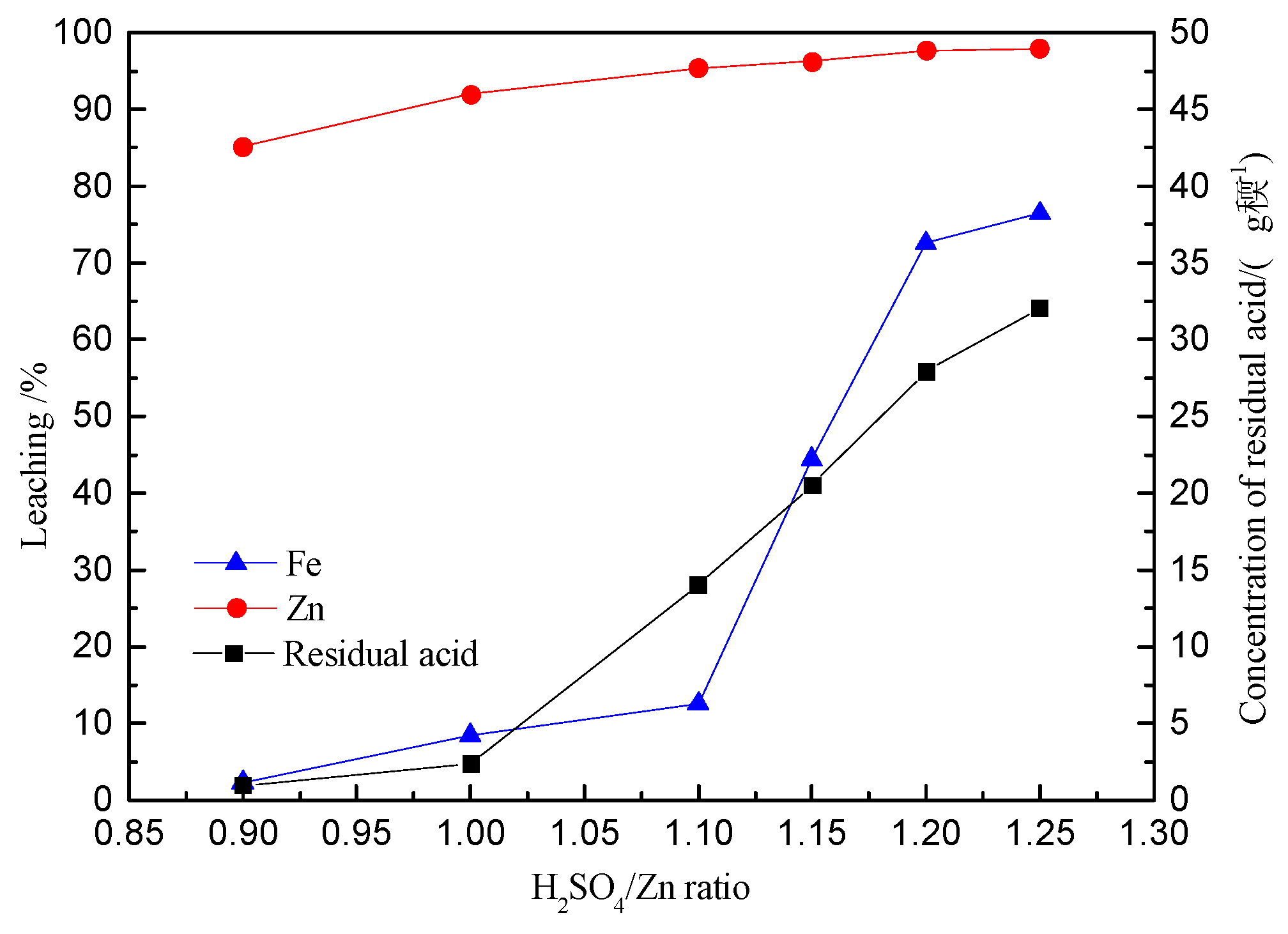
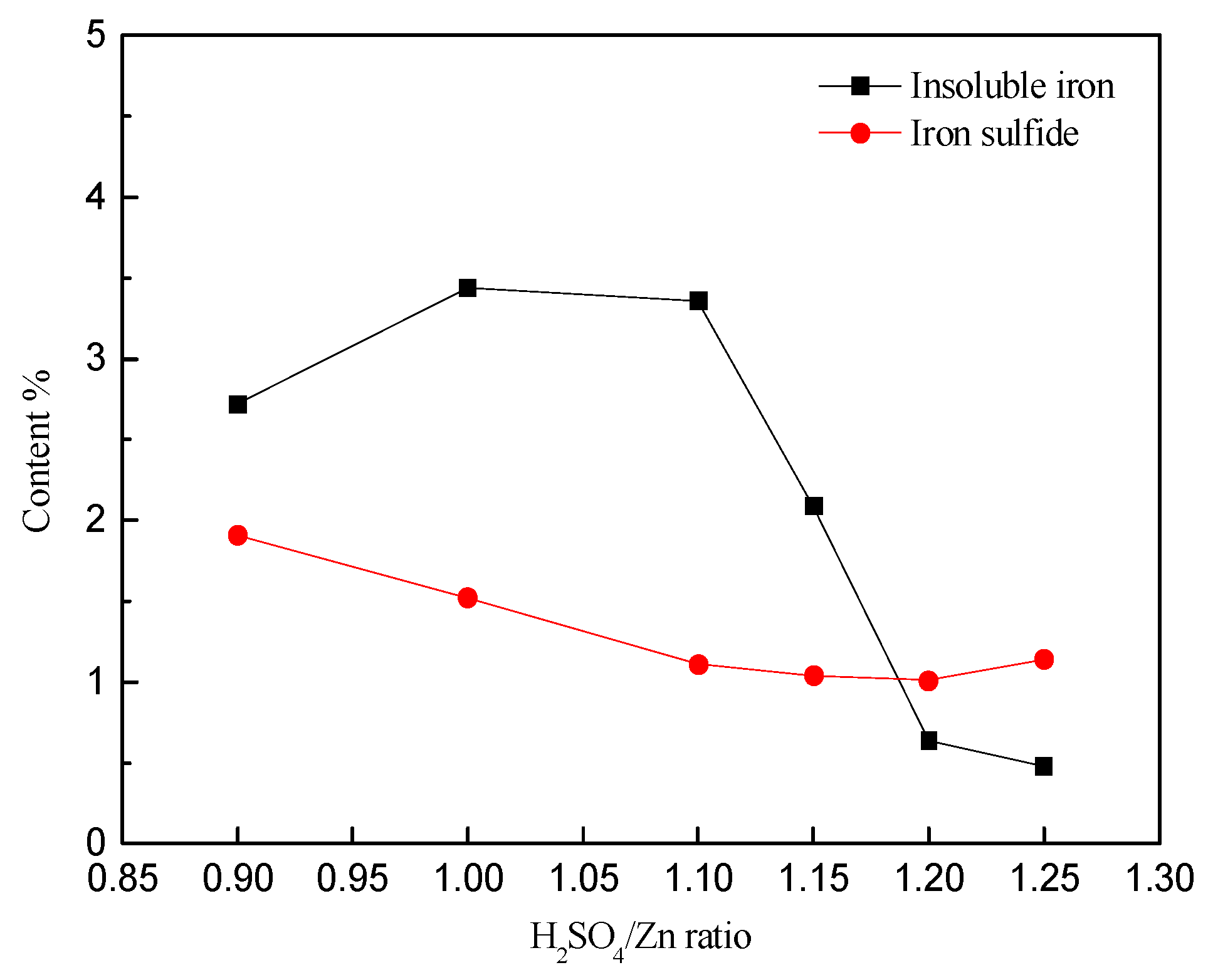
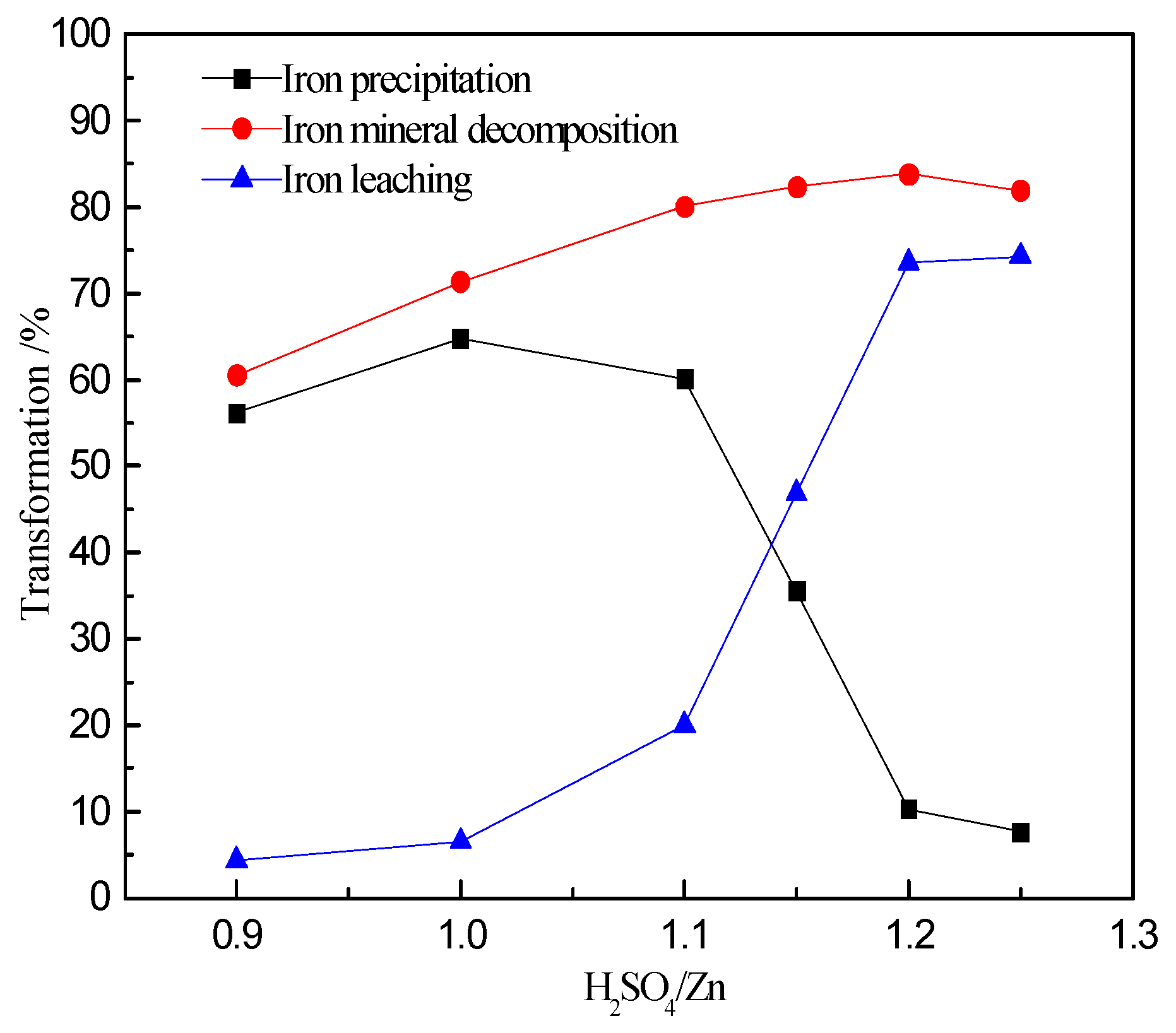
3.2. Effect of Acidity on Zinc and Iron Extractions
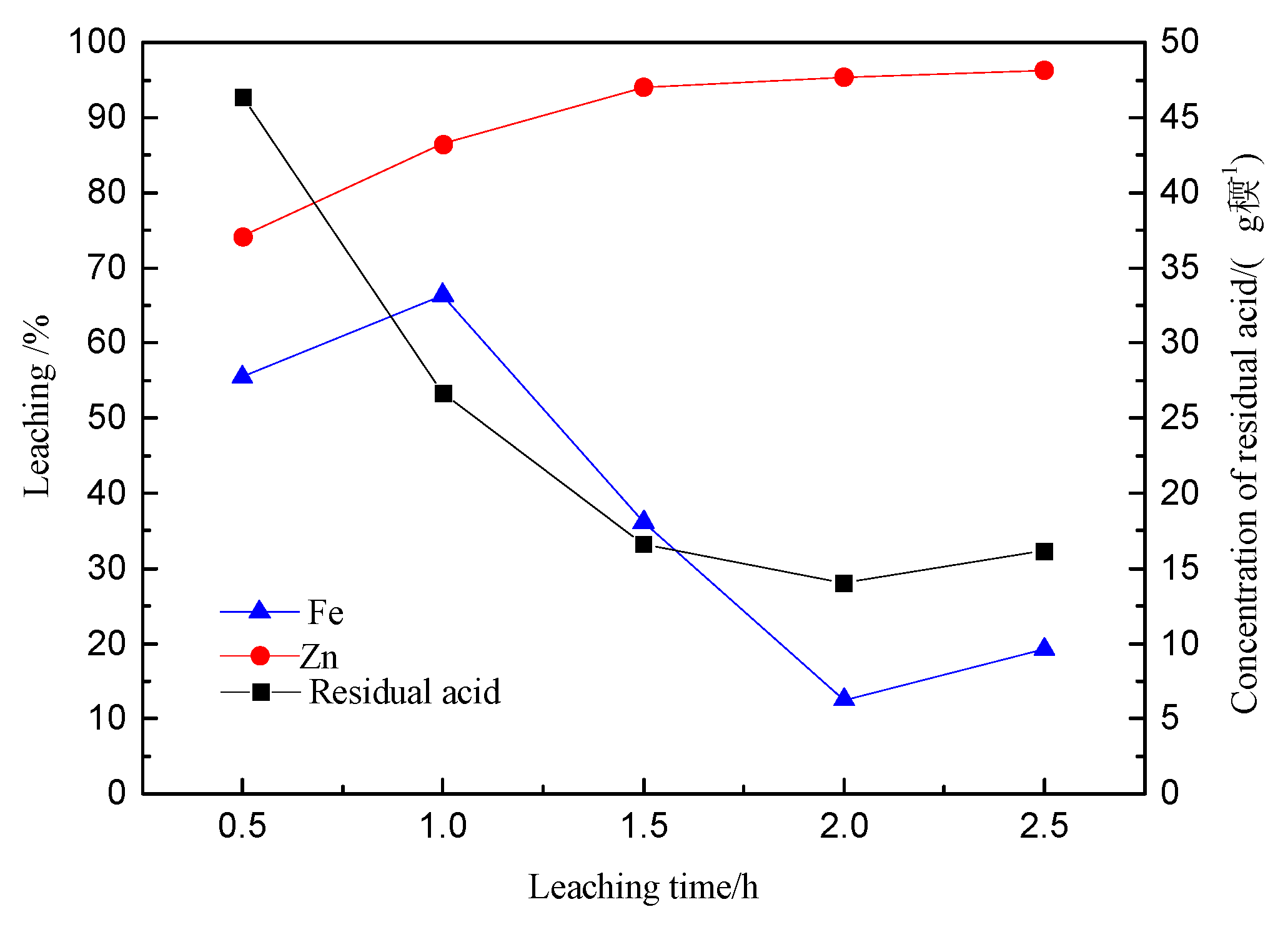
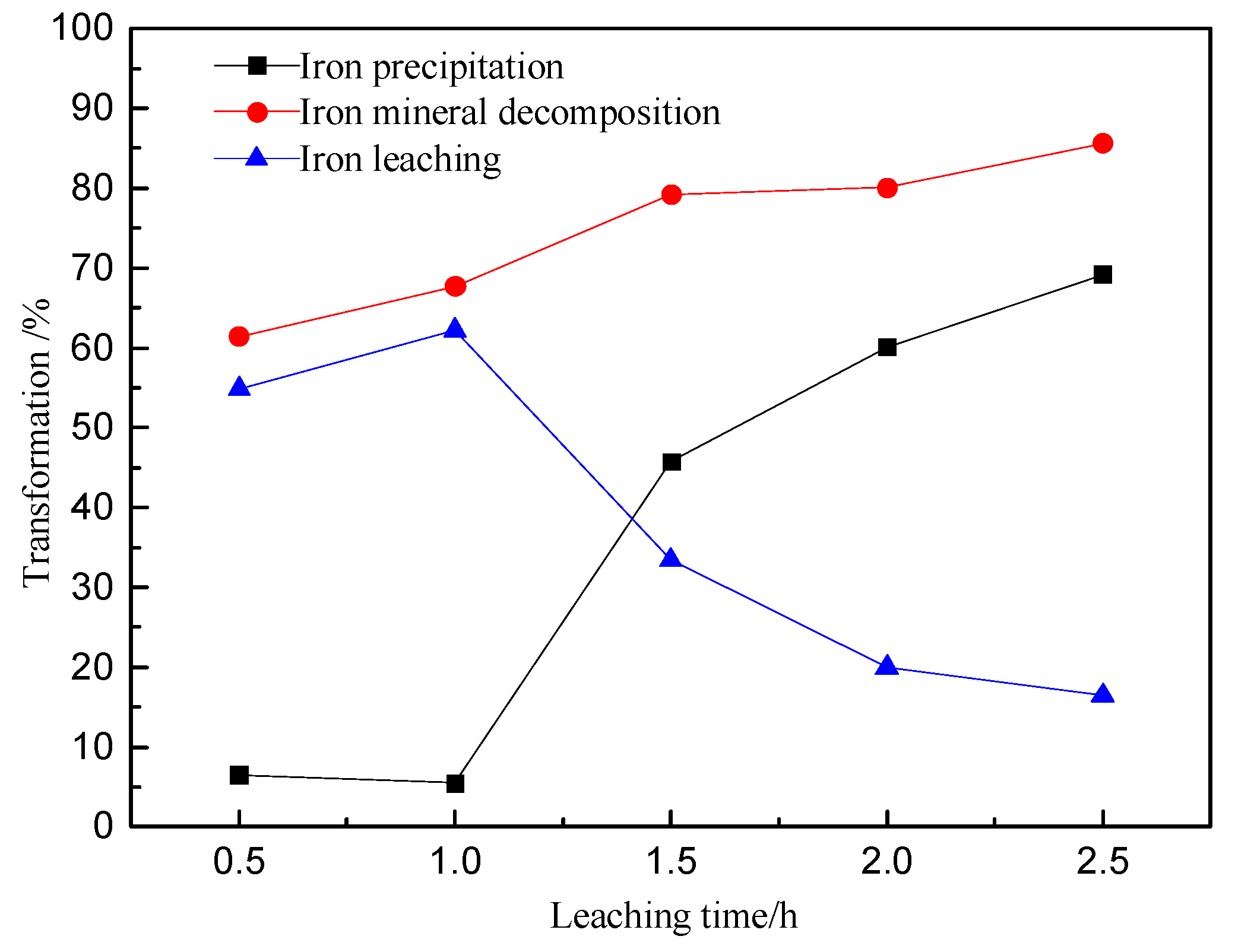
3.3. Effect of Leaching Time on Zinc and Iron Extractions
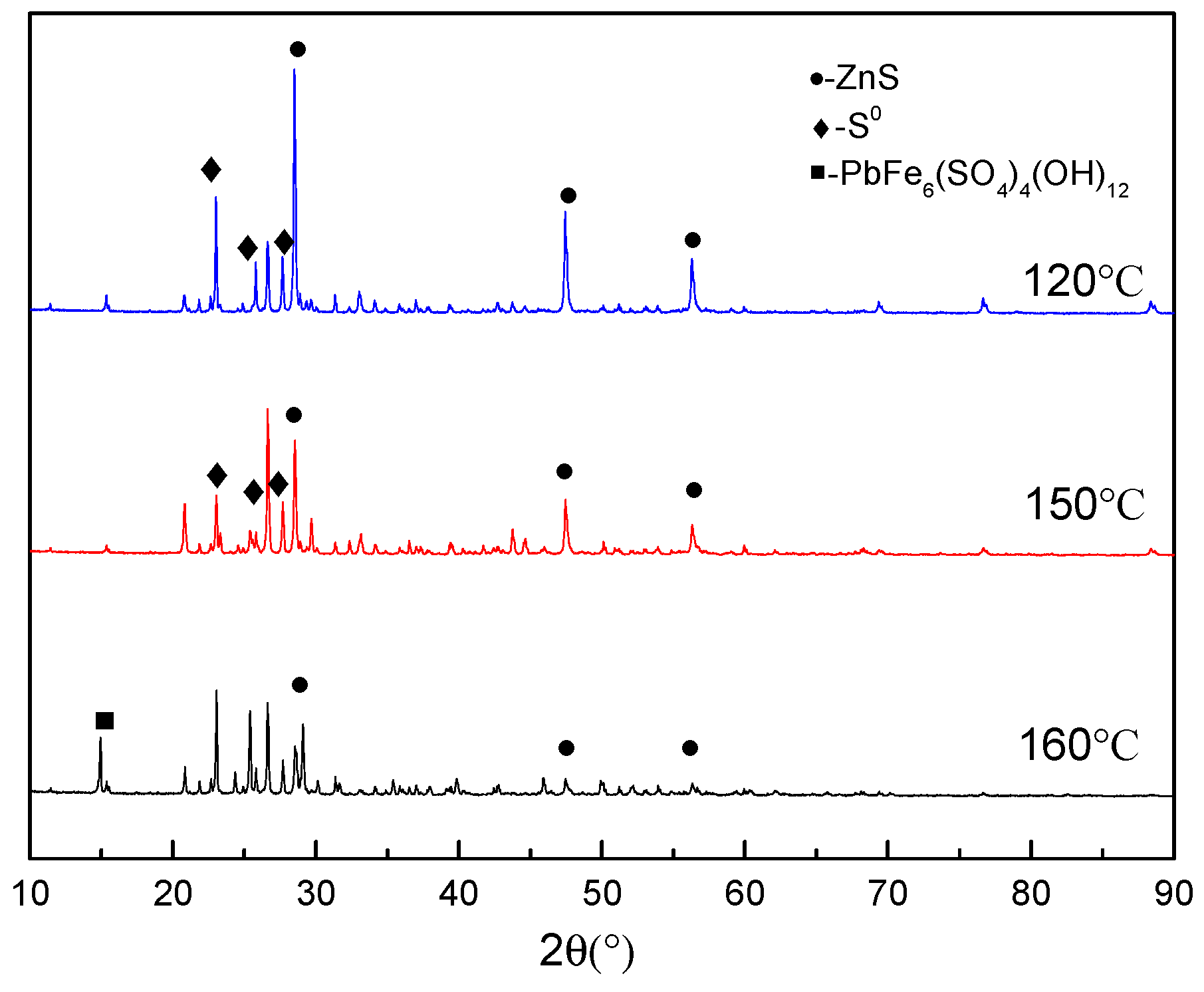
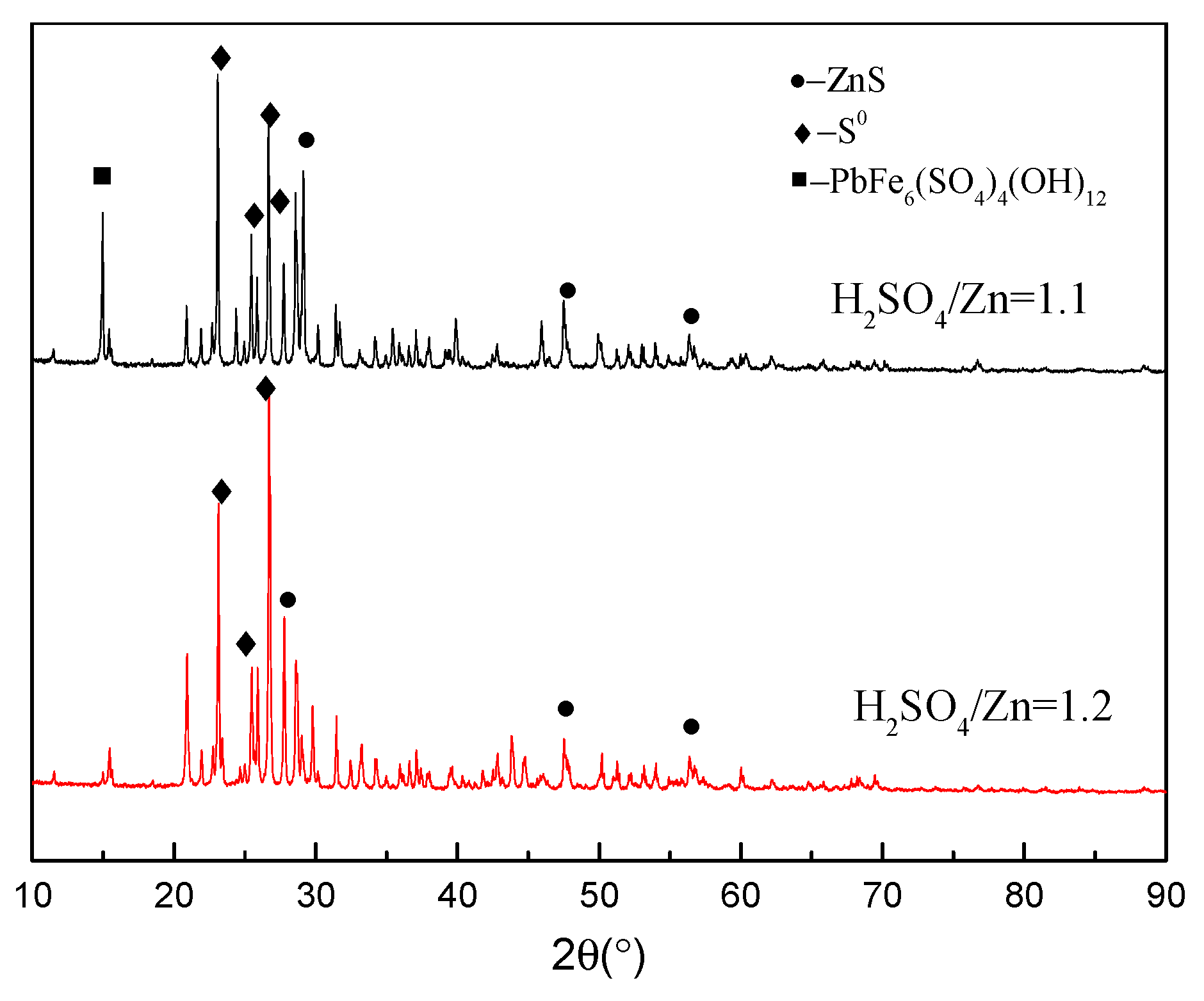
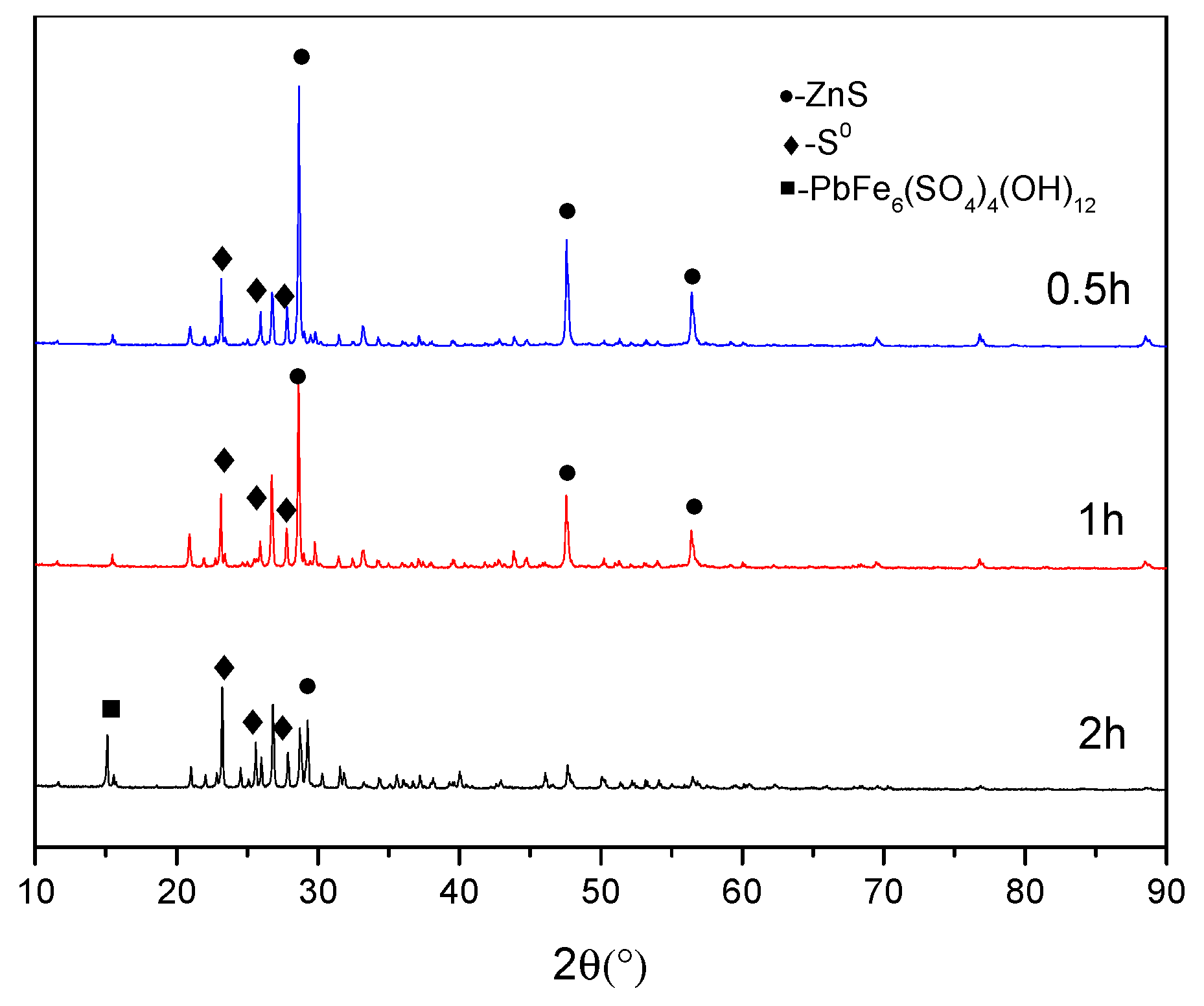
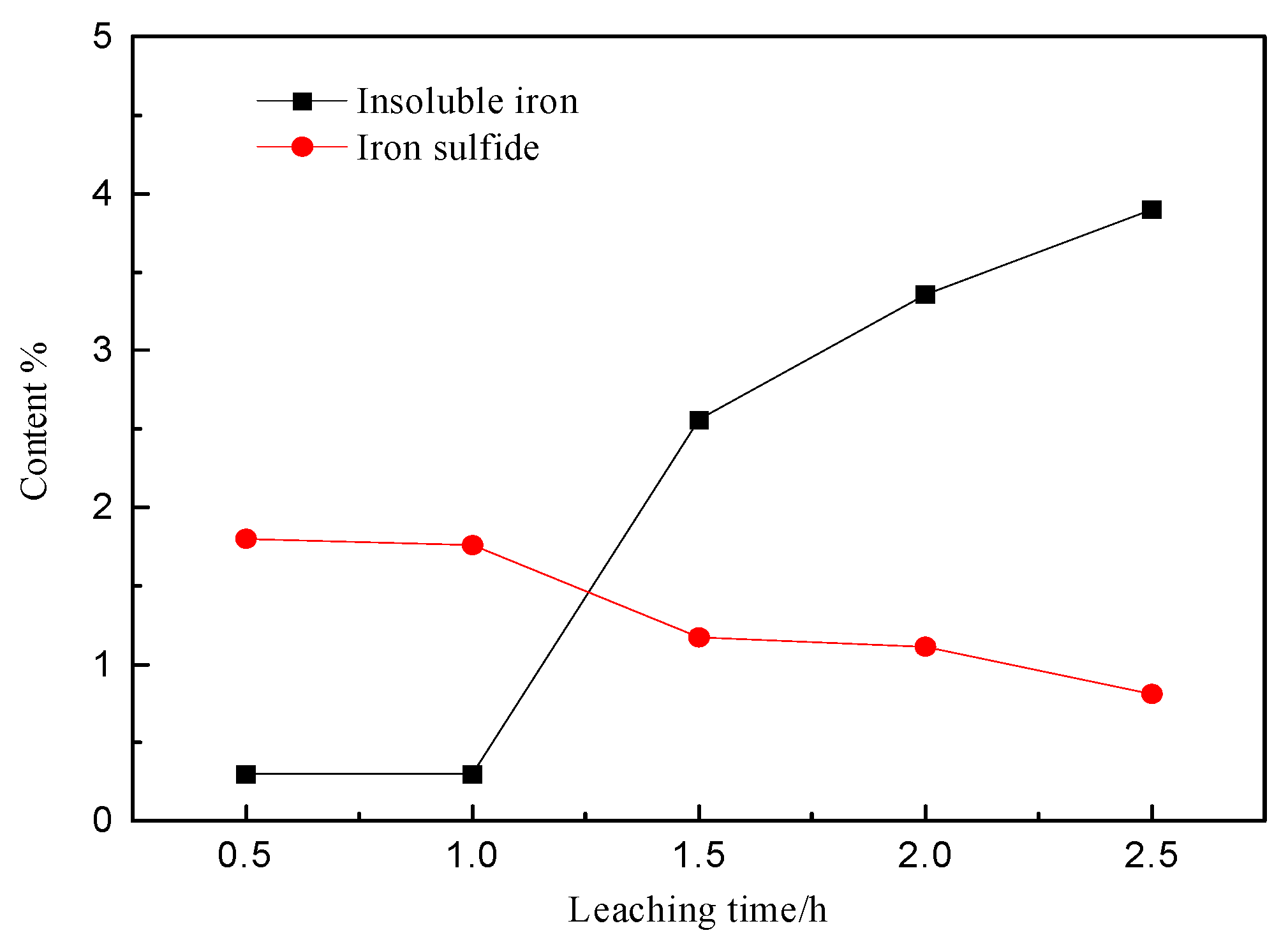
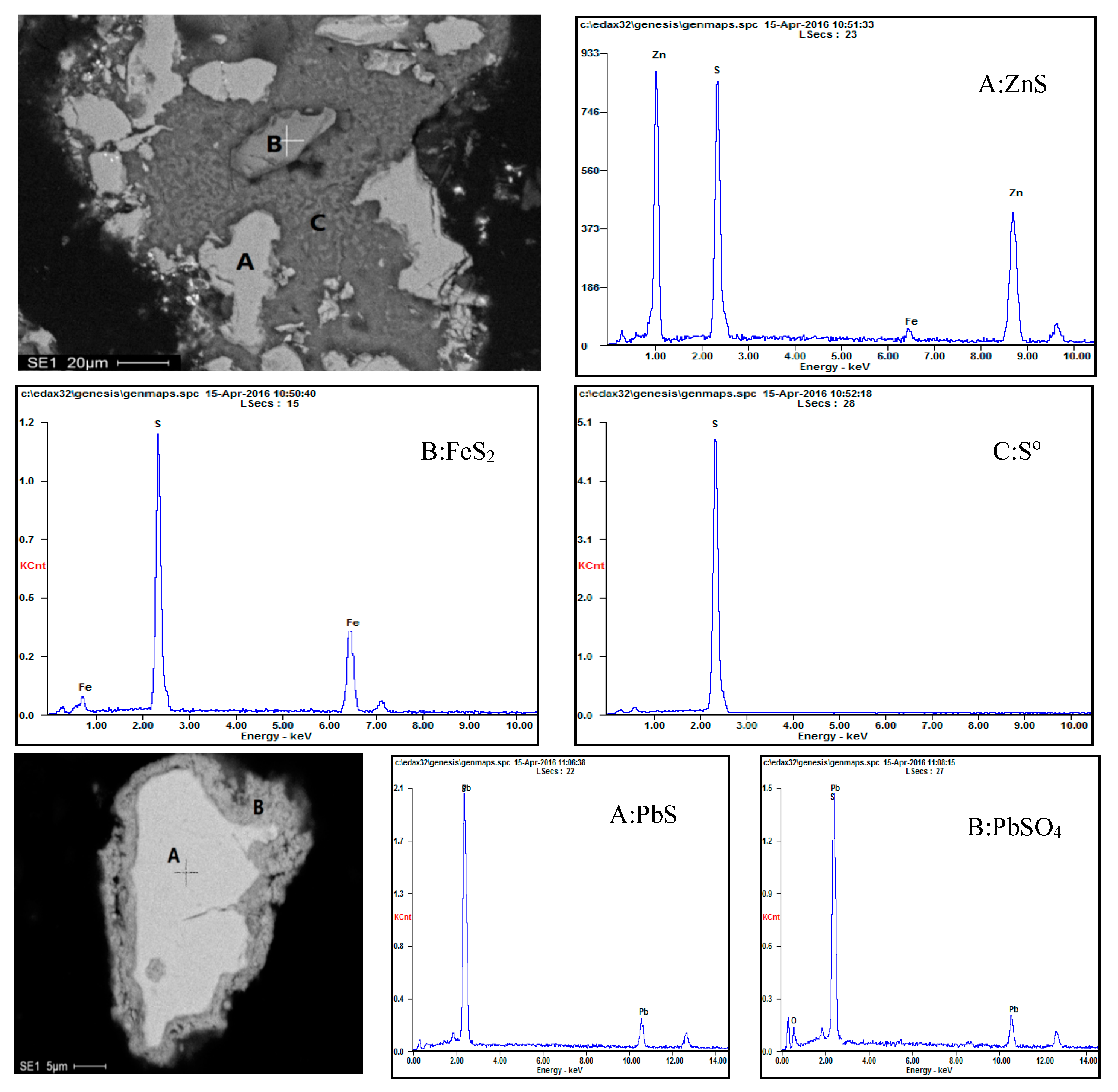
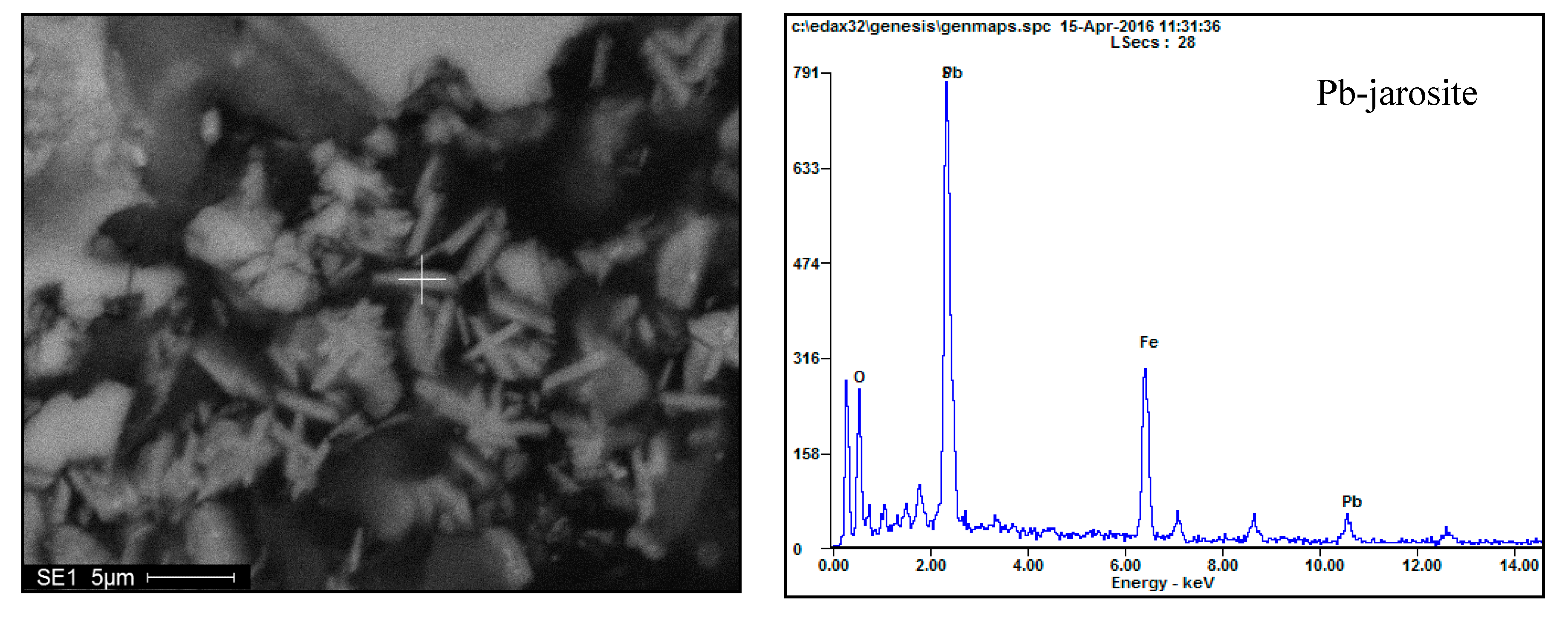
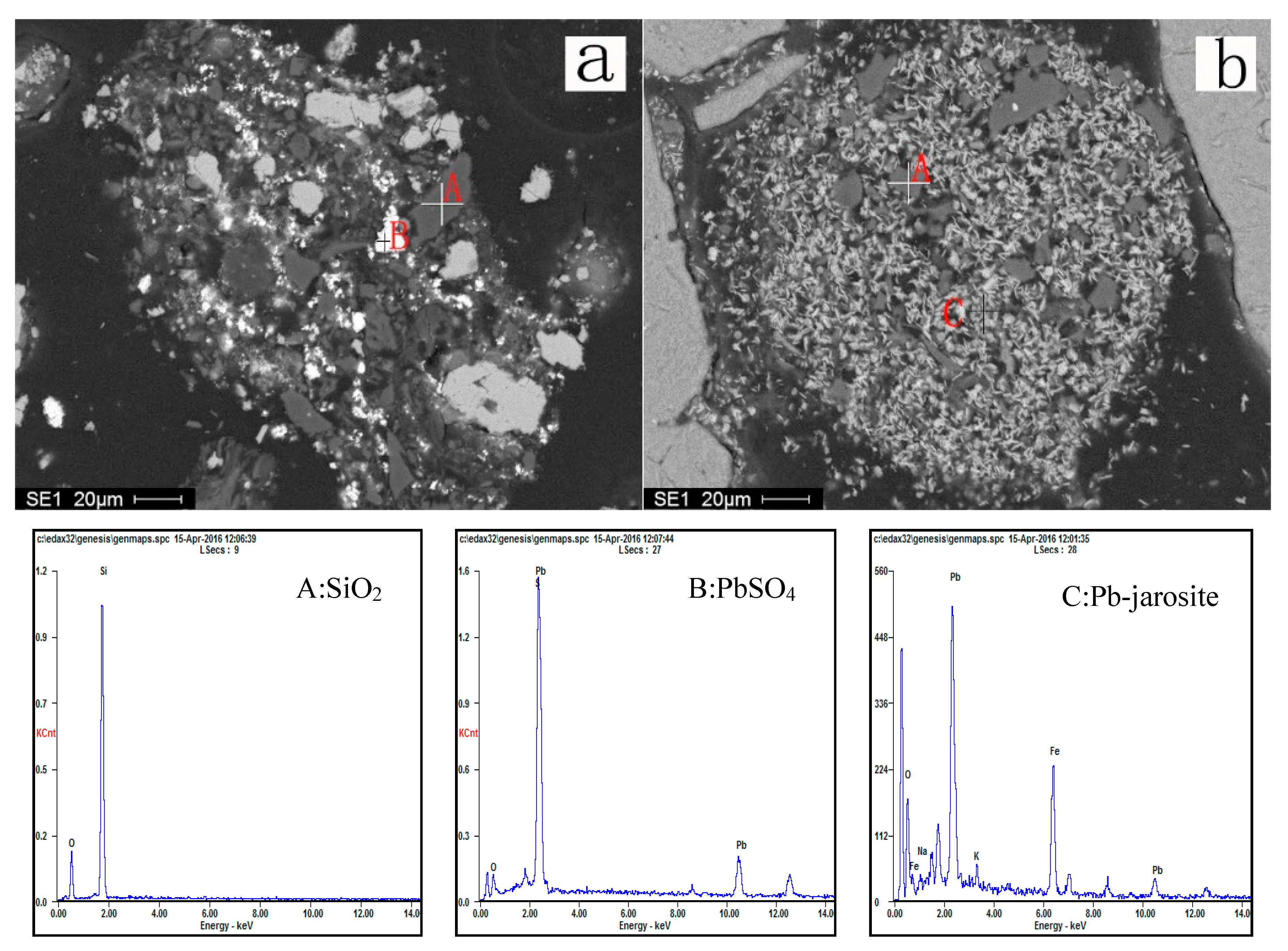
3.4. Phase Analysis of Residues
3.4.1. Morphology Analysis
3.4.2. EDS Analysis
4. Conclusions
- The iron extraction increased with the ratio of H2SO4/Zn within the range of 0.9:1 to 1.25:1; however, most rapidly in the range 1.0:1 to 1.2:1.
- The iron extraction displayed an initial increase that was followed by a decrease, with respect to the leaching time.
- The precipitation of iron increased sharply from approximately 15.0% to 55.9% when the temperature exceeds 150 °C.
- The precipitation of iron increased initially to a maximum of 64.8% (H2SO4/Zn = 1:1) and then decreased to its lowest value of 7.63% (H2SO4/Zn = 1.25:1).
- The precipitation of iron was maintained at about 6% (for leaching time ≤ 1 h). Subsequently, it increased quite markedly and reached the highest value of 69.2% (after leaching time = 2.5h).
Author Contributions
Funding
Conflicts of Interest
References
- Liao, M.; Deng, T. Zinc and lead extraction from complex raw sulfides by sequential bioleaching and acidic brine leach. Miner. Eng. 2004, 17, 17–22. [Google Scholar] [CrossRef]
- Santos, S.M.C.; Machado, R.M.; Correia, M.J.N.; Reis, M.T.A.; Ismael, M.R.C.; Carvalho, J.M.R. Ferric sulphate/chloride leaching of zinc and minor elements from a sphalerite concentrate. Miner. Eng. 2010, 23, 606–615. [Google Scholar] [CrossRef]
- Souza, A.D.; Pina, P.S.; Leao, V.A. Bioleaching and chemical leaching as an integrated process in the zinc industry. Miner. Eng. 2007, 20, 591–599. [Google Scholar] [CrossRef]
- Souza, A.D.; Pina, P.S.; Leao, V.A.; Silva, C.A.; Siqueira, P.F. The leaching kinetics of a zinc sulphide concentrate in acid ferric sulphate. Hydrometallurgy 2007, 89, 72–81. [Google Scholar] [CrossRef]
- Sahu, S.; Sahu, K.; Pandey, B. Leaching of zinc sulfide concentrate from the ganesh-himal deposit of Nepal. Metall. Mater. Trans. B: Process Metall. Mater. Process. Sci. 2006, 37, 541–549. [Google Scholar]
- Owusu, G.; Dreisinger, D.B.; Peters, E. Effect of surfactants on zinc and iron dissolution rates during oxidative leaching of sphalerite. Hydrometallurgy 1995, 38, 315–324. [Google Scholar] [CrossRef]
- Han, J.; Liu, W.; Qin, W.; Peng, B.; Yang, K.; Zheng, Y. Recovery of zinc and iron from high iron-bearing zinc calcine by selective reduction roasting. Ind. Eng. Chem. 2015, 22, 272–279. [Google Scholar] [CrossRef]
- Nikkhou, F.; Xia, F.; Deditius, A.P. Variable surface passivation during direct leaching of sphalerite by ferric sulfate, ferric chloride, and ferric nitrate in a citrate medium. Hydrometallurgy 2019, 188, 201–215. [Google Scholar] [CrossRef]
- Ghassa, S.; Noaparast, M.; Shafaei, S.Z.; Abdollahi, H.; Gharabaghi, M.; Boruomand, Z. A study on the zinc sulfide dissolution kinetics with biological and chemical ferric reagents. Hydrometallurgy 2017, 171, 362–373. [Google Scholar] [CrossRef]
- Koerker, F.W.; Calderwood, H.N. The system ferric oxide-sulfur trioxide-water. J. Phys. Chem. 2002, 42, 1151–1155. [Google Scholar] [CrossRef]
- Hasegawa, F.; Tozawa, K.; Nishimura, T. Solubility of ferrous sulfate in aqueous solutions at high temperatures. J. Min. Mater. Process. Inst. Jpn. 1996, 112, 879–884. [Google Scholar]
- Umetsu, V.; Tozawa, K.; Sasaki, K. The hydrolysis of ferric sulphate solutions at elevated temperatures. Can. Metall. Quart. 1977, 16, 111–117. [Google Scholar] [CrossRef]
- Chen, Z.; He, X. Zinc concentrate oxygen pressure leaching technology. Hum. Nonferrous Met. 2002, 18, 26–28. [Google Scholar]
- Tang, J.; Zhou, X. Behavior of iron in the process of pressure leaching of zinc sulfide concentrate. Non Ferr. Met. (Smelt. Part) 1987, 3, 32–35. [Google Scholar]
- Posnjak, E.; Merwin, H.E. The system Fe2O3-SO3-H2O. J. Am. Chem. Soc. 1922, 44, 1965–1994. [Google Scholar] [CrossRef]
- Yue, M.; Sun, N.; Zou, X.; Shao, J.; Liu, J.; Wang, K.; Lu, Y. The discussion on hydrolysis precipitation of ferric oxide directly from ferric-ion rich zinc leachate. China Nonferrous Metall. 2012, 4, 80–85. [Google Scholar]
- Ruiz, M.C.; Zapata, J.; Padilla, R. Effect of variables on the quality of hematite precipitated from sulfate solutions. Hydrometallurgy 2007, 89, 32–39. [Google Scholar] [CrossRef]
- Dutrizac, J.E. Converting jarosite residues into compact hematite products. JOM 1990, 42, 36–39. [Google Scholar] [CrossRef]
- Ghassa, S.; Noaparast, M.; Shafaei, S.Z.; Abdollahi, H.; Gharabaghi, M.; Boruomand, Z. Medium-pressure test of zinc sulfide pressure leaching. Sulfuric Acid Ind. 1983, 4, 50–55. [Google Scholar]
- Xu, Z.; Jiang, Q.; Wang, C. Behavior of zinc, sulfur and iron during low temperature pressure leaching of iron sphalerite. Non Ferr. Met. (Smelt. Part) 2012, 7, 6–11. [Google Scholar]
- Beijing Research Institute of Mining and Metallurgy. Chemical Phase Analysis; Metallurgical Industry Press: Beijing, China, 1976. [Google Scholar]
- Qin, S.; Jiang, K.; Zhang, B.; Wang, H. Study on phase form and content of Sulfur and Iron in leaching residue of oxygen pressure leaching of sphalerites. Chin. J. Inorg. Anal. Chem. 2016, 6, 57–61. [Google Scholar]
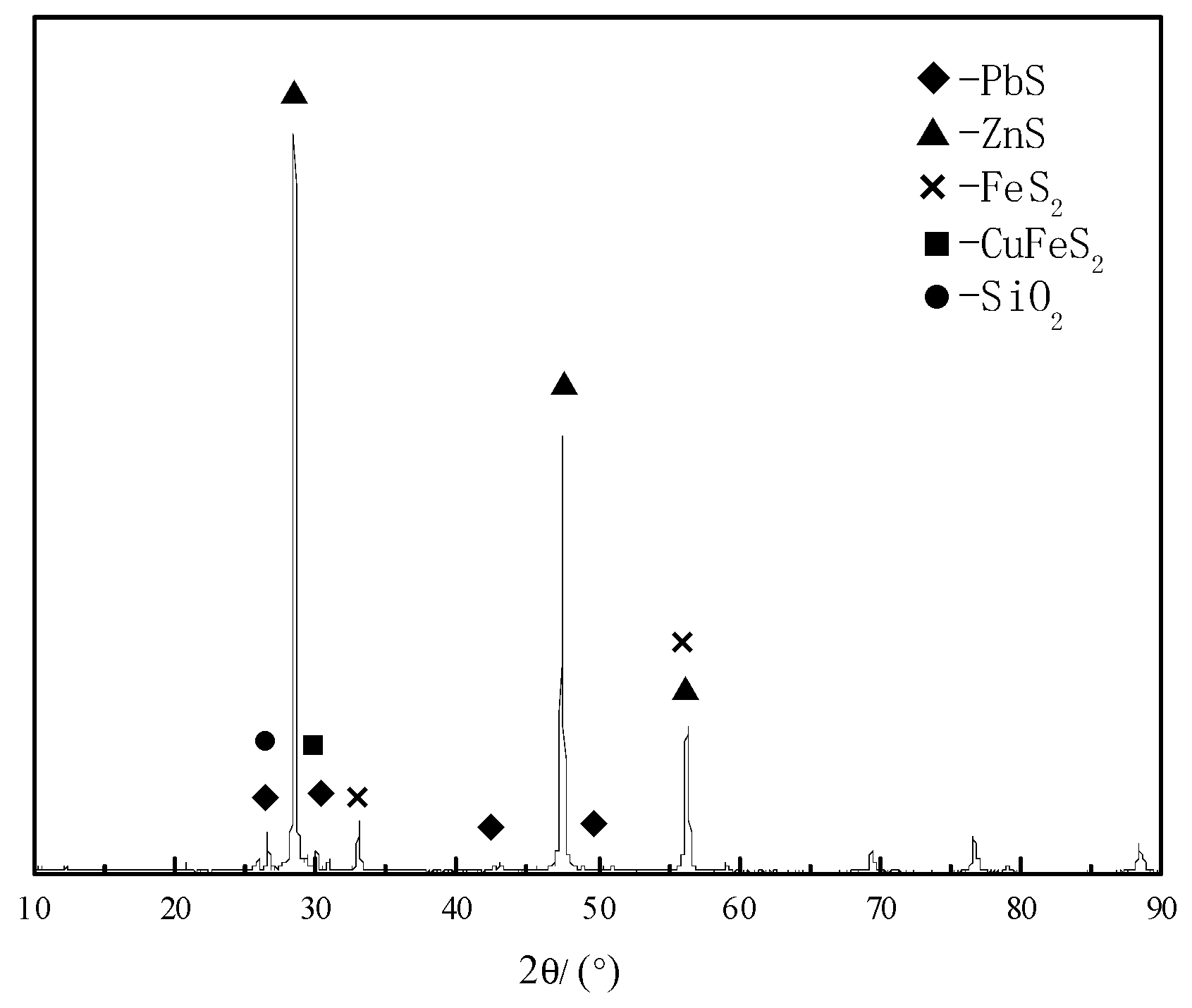






| Element | Zn | Fe | S | Cu | Pb | SiO2 | CaO | K2O | Na2O |
|---|---|---|---|---|---|---|---|---|---|
| Content/% | 55.5 | 2.4 | 29.3 | 0.64 | 1.6 | 5.4 | 0.71 | 0.10 | <0.10 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qin, S.-C.; Jiang, K.-X.; Wang, H.-B.; Zhang, B.-S.; Wang, Y.-F.; Zhang, X.-D. Research on Behavior of Iron in the Zinc Sulfide Pressure Leaching Process. Minerals 2020, 10, 224. https://doi.org/10.3390/min10030224
Qin S-C, Jiang K-X, Wang H-B, Zhang B-S, Wang Y-F, Zhang X-D. Research on Behavior of Iron in the Zinc Sulfide Pressure Leaching Process. Minerals. 2020; 10(3):224. https://doi.org/10.3390/min10030224
Chicago/Turabian StyleQin, Shu-Chen, Kai-Xi Jiang, Hai-Bei Wang, Bang-Sheng Zhang, Yu-Fang Wang, and Xue-Dong Zhang. 2020. "Research on Behavior of Iron in the Zinc Sulfide Pressure Leaching Process" Minerals 10, no. 3: 224. https://doi.org/10.3390/min10030224
APA StyleQin, S.-C., Jiang, K.-X., Wang, H.-B., Zhang, B.-S., Wang, Y.-F., & Zhang, X.-D. (2020). Research on Behavior of Iron in the Zinc Sulfide Pressure Leaching Process. Minerals, 10(3), 224. https://doi.org/10.3390/min10030224




