Abstract
Due to the progressive fall of the ore grades and the increasingly refractory composition of minerals, concentrating plants have increased which has led to an increase in the generation of tailings. Tailings, especially those obtained in the past, have remaining copper and other valuable species in quantities that can potentially be recovered, such as gold, silver, vanadium, and rare earth elements which transforms this abundant waste into a potential source of precious or strategic metals for secondary mining. One of the techniques of solid–liquid separation that processes solutions with low concentrations of metals corresponds to adsorption, and more recently biosorption, which is based on the use of biological matrices that do not constitute an environmental liability after application. Biosorption occurs as a consequence of the wide variety of active functional groups present in different types of biomass. Bacterial, fungal, plant, and algal biomasses have been described as biosorbents, mainly for the treatment of diluted and simple solutions. This work aims to recover copper from leached tailings using biomass of the red algae Gracilaria chilensis as a biosorbent. The tailing samples were taken from an abandoned deposit, in the north of Chile, and after an acid leaching copper was biosorbed, kinetics of adsorption and the equilibrium isotherms were studied, applying the Freundlich and Langmuir models. Operational parameters such as adsorbent dose, pH, and initial metal concentration were studied.
1. Introduction
It is well-known that Chile is a country that supports much of its economy in mining activity, representing a contribution of 13% to its GDP. Among the minerals in which Chile occupies a leading position are copper, lithium, iodine, silver, and molybdenum. In the case of copper, 31% of the metal traded in the world comes from Chile (5,700,000 Mt), and half of this amount is obtained from the region of Antofagasta, which makes it a strategic region for country development [1,2].
In the current context, the copper ore grades are increasingly low, and in more refractory forms, therefore, mining operations have increased the amount of processed material, producing larger volumes of unprocessed low-grade ores that are below cut grade. Additionally, the installation of new concentrating plants or expansion of the existing ones has increased the generation of tailings. According to the latest cadastre, there are 740 tailings deposits in Chile and 51 in Antofagasta (Figure 1), of which 72% are inactive or abandoned and with an estimated amount of 520 Mt [3].
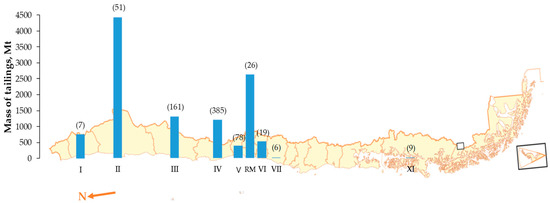
Figure 1.
Discarded tailings in Chile are significantly predominant in the northern. The numbers in parenthesis denote the quantity of tailing deposits in each region of Chile [3].
The prosperity of Chile, as in every mining-based country, is closely linked to the availability of exploited resources, their demand, and international prices. Thus, a decrease of the so-called copper price super-cycle results in a considerable drop in the national incomes, and therefore creates opportunity to develop new technologies that improve yields and reduce production costs, as well as evaluate the processing of new mineral resources as the wastes (tailings and low-grade ores) produced extensively by the mining operations. Although tailings are depleted in the element extracted during the flotation, they still have many other valuable species that potentially can be recovered, transforming this discarded resource into an interesting secondary raw material. In Chile, the processing of tailings to extract minerals is incipient, but with an emerging potential, since chemical and mineralogical analyses of some tailing deposits have detected the presence of valuable metals in quantities that would eventually allow their extraction, with attractive prices for their processing, such as copper, zinc, vanadium, strontium, silver, and even rare earths as lanthanum and yttrium [4,5].
Regarding macroalgae, they represent a diverse group of eukaryotic, photosynthetic marine organisms widely distributed throughout the world’s oceans that are classified into the following three divisions based on their pigmentation: brown (Phaeophyta), red (Rhodophyta), and green (Chlorophyta). The color is given by the predominance of one pigment over the others; thus, for example in the case of the brown algae the presence of fucoxanthin, a pigment that protects from sunlight, masks the color of chlorophylls a and c, betacarotenes, and other xanthophylls. As reserve substances, seaweeds have high molecular weight polysaccharides, being the principal laminarins, alginates, fucanoids, carrageenans, and agars. The walls of the macroalgae cells are composed of cellulose and alginic acid [6].
Seaweeds have been used as food since ancient times, mainly by China, Japan, and the Republic of Korea. Additionally, other relevant uses of seaweeds are the extraction of the phycocolloids, i.e., alginate, agar, and carrageenan, which are used as thickening and gelling agents that have a broad spectrum of industrial applications in pharmaceutics, cosmetics, food industry, and agriculture [6]. Approximately 1 Mt of fresh seaweed is now annually harvested to extract 55,000 t of these phycocolloids valued at USD $585 million. Particularly, the production of alginate (USD $213 million) and agar (USD $132 million) is made from extracts of brown and red algae, respectively. Chile is one of the first agar producers in the world, supplying 10% of the global biomass for agar and alginates [7,8].
The global catches of seaweeds for phycocolloids production are led by Chile, with 51.3% [9]. The extensive Chilean coast provides a wide diversity of brown and red algae species, with the following most relevant in order of economic importance: Gracilaria chilensis (Pelillo), Lessonia nigrescens (Black Huiro), Lessonia traberculata (Huiro palo), and Macrocystis pyrifera (Huiro) [10]. According to FAO [11], Chile was the world’s largest producer of seaweed outside Asia. In 2016, the total of brown and red algae collected in Chile was about 260,000 t [12].
Although the use of activated carbon as adsorbent has been demonstrated to be the best method for the recovery of metals, its high cost and difficult regeneration results in an unpractical application for the treatment of large volumes or diluted effluents, in this context, biosorption emerges as a cost-effective and efficient alternative for removal of heavy metals from wastewaters. Living and dead biomass of microorganisms, fungi, micro and macroalgae, and plant extracts have been used as biosorbent for the recovery of metals from industrial effluents such as lead, cadmium, zinc, copper, chromium, gold, and silver [13,14].
Biosorption is a rapid and reversible ion-binding process because it is independent of cellular metabolism, therefore, dead biomass can be used [15]. Some of the advantages of biosorption are the possibility to store the adsorbents for an extended period of time, no toxicity effects by high metal concentrations, no nutrient limitations, the metals captured can be readily desorbed, and the application can be performed over a wide range of pH (3 to 9) and temperature (4 to 90 °C) [16,17,18]. Other advantages of this process are that it is a clean and simple alternative for the recovery of metals in non-concentrated solutions, there are low capital investment costs, and these biological materials are renewable, inexpensive, and can be obtained from agriculture, aquaculture, or industrial waste [19,20].
Among the studied biosorbents, macroalgae are a promising group because the complex mucilaginous polysaccharides on their cell walls (alginates and fucoidans) have a high affinity for di- and trivalent cations (Figure 2) [21,22,23]. The most abundant functional groups related to the interaction with metals by macroalgae are carboxyl, amino, phosphate, hydroxyl, sulfhydryl, and sulfonate [24]. The use of seaweeds for biosorption has been mainly validated for wastewater treatment, preferably from diluted or pure solutions [25,26]. Plaza Cazón et al. [27] used Macrocystis pyrifera to remove zinc and cadmium from wastewater, demonstrating its good uptake capacity and similar to other macroalgae species such as Sargassum filipendula, Gymnogongrus torulosus, and Fucus vesiculosus, which are reported in previous works [28,29]. The same group evaluated the use of M. pyrifera and Undaria pinnatifida for the chromium and mercury biosorption from aqueous solutions, demonstrating that the carboxylic and amino groups are strongly involved in chromium binding, while amino and sulfhydryl for mercury uptake [30,31].
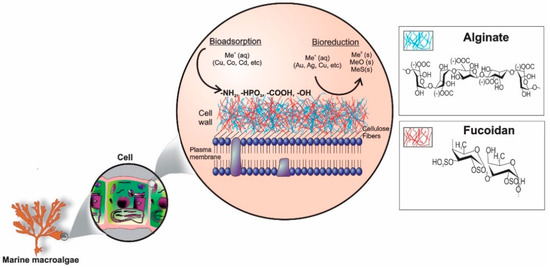
Figure 2.
Most abundant polysaccharides containing brown and red algae, alginate and fucoidan, which are related to the metal biosorption process.
An important aspect to consider is that biosorption is a surface process and the metal biosorbent interaction mechanism is physical by electrostatic interactions and Van der Waals forces, or chemical by ion exchange, proton shift, complexation, and chelation [32,33]. The factors that have an influence on the biosorption and that are related to the physicochemical properties of metals are concentration, molecular weight, ionic radius, and oxidation state. Moreover, other properties of the biosorbents and the operational conditions such as pH, temperature, and biosorbent concentration are also important the process performance. Among all the influencing factors, pH is one of the most important because it controls the ionic dissociation and speciation of sites and metals in the solution [34,35,36]. The wide variety of active functional groups in algae biomass would result in a diverse range of adsorption mechanisms, for this reason, different metals and quantities of metals can be obtained with different biosorbents [37].
It has also been described that biosorption leads to metal bioreduction, where some of the compounds forming the cell walls react with metal ions and convert them to their elemental form, producing tiny particles of metal, namely nanoparticles [38,39,40]. Metal nanoparticles have great scientific and technological interest because they have unique optoelectrical and physicochemical properties, due to their crystallographic and morphological characteristics. The use of nanoparticles is played an increasingly important role in the development of new key technologies applied to electronics, catalysis, drug release, cancer therapy, detection, and gas monitoring [41,42,43].
The valorization of contaminating industrial or mine waste by micro- and macroorganisms for the biosynthesis of metal nanoparticles is a research field that is not extensively discussed in the literature, but which proves to be emerging, according to the seeking for sustainable processes for effluent treatment and the generation of strategic nanomaterials required to develop technological products [44].
In this work, the biomass of the red algae Gracilaria chilensis was used as a biosorbent to recover copper from leached mining tailings. The tailing samples were taken from an abandoned deposit in the north of Chile and, after an acid leaching, copper was adsorbed, for which the kinetics of adsorption and the equilibrium isotherms were studied, applying the Freundlich and Langmuir models. Operational parameters such as adsorbent dose, pH, and initial metal concentration were also optimized.
It is important to note that although the biosorption of copper and other metals using different type of biomasses, including seaweeds, has been reported in several previous works, this does not address the biotechnological treatment of real mining or industrial effluents, which is a challenging aspect because the solutions are generally complex matrices with high content of multiple metallic species and high acidity or ionic strength, which affect the affinity and interaction behavior between metals and chemical groups on the cell walls. On the basis of this perspective, this work contributes to state-of-the-art metal biosorption from mining effluents.
2. Materials and Methods
2.1. Tailings and Biomass
Samples of tailings deposits located in the commune of Taltal, Antofagasta, Chile, were taken in two sampling campaigns. In the deposit, the first 15 cm of surface material was discarded to rule out potential noise in the composition. The granulometry of the tailings was characterized by sieving, using the 0.064 to 0.71 mm size fraction.
The samples were taken and homogenized, by using a Jis shovel. A representative section of each sample was obtained and, subsequently, chemically and mineralogically analyzed. The chemical analysis of the mineral was performed by inductively coupled plasma mass spectrometry (7800 ICP-MS, Agilent Technologies, Santa Clara, CA, USA) and the mineralogical content was obtained by X-ray diffraction (XRD, D500, Siemens, Munich, Germany).
In this investigation, biomass of the red alga species Gracilaria chilensis was used. The seaweed was collected from the coast of Mejillones, Antofagasta, Chile, from an experimental center of repopulation of this marine species. This alga, once collected, was sun-dried for two days and, subsequently, washed with distilled water to eliminate adhering remains. The samples were left to dry in a stove at 60 °C for 12 h.
For the characterization of the biomass, Fourier-transform infrared spectroscopy (FTIR) was used. The analysis showed the presence of ionizable functional groups (carboxyl, hydroxyl, and amino) capable of interacting with protons or metal ions. Furthermore, the band assignments of the FTIR for typical functional groups present in biomass are illustrated in Table 1.

Table 1.
Vibrational stretches of functional groups related to biosorption [45].
2.2. Mineral Leaching
For leaching of ore tailing, in a beaker, 500 mL of sulphuric acid 1 M was contacted with 16 g of tailings. The leaching was carried out for 4 h, on a mechanical stirrer at 500 rpm, and the obtained solution (namely PLS) was used to leach a new load of fresh tailing ore under the same conditions. Five leaching cycles of 4 h were carried out in order to increase the concentration of copper for the subsequent biosorption tests. Chemical analysis of copper and iron from solutions was performed by atomic absorption spectrometry (AAS, 240 AA, Agilent Technologies, Santa Clara, CA, USA).
In order to remove iron, which is the main interferent species in biosorption, the final PLS was treated by adding NaOH 2 M until reaching pH 4, i.e., the point where the iron precipitates as Fe(OH)3 and the copper remains in the solution, according to the equilibrium curves of pH vs. Eh (Figure 3). The solution was decanted for 10 h, and the liquid fraction was recovered for the biosorption experiments. Samples for chemical analysis of copper and iron were taken before and after the precipitation.
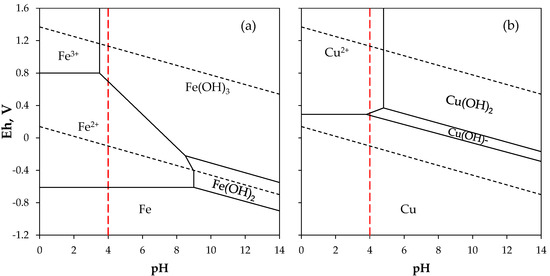
Figure 3.
Pourbaix diagram for (a) iron and (b) copper at 25 °C. Vertical lines at pH 4 denotes the precipitating condition used in the treatment of leached tailing (PLS) solution.
2.3. Batch Biosorption Procedure
To pretreat the biomass, 20 g of dried alga was suspended in 400 mL of CaCl2 0.2 M with stirring at 150 rpm for 4 h. A pH 5 was maintained with NaOH 0.1 M. The sample was washed several times with deionized water to remove excess calcium, filtered, and dried for 12 h at 60 °C.
In a first approach, kinetic study for biosorption process was conducted using CuSO4·5H2O solutions at defined copper concentrations of (2, 20, 40, 100, 200, and 400) mg/L, 1 g/L biomass, and pH 3. Subsequently, in a second stage, batch biosorption experiments for synthetic and actual leached tailing solutions were carried out using 50 mL of solution (200 mg/L for synthetic solution and 150 mg/L for PLS solution). The pH 1.5 was adjusted for each solution with NaOH and H2SO4 0.1 M. Biomass dosage of 0.5 g/L was added, and agitated at 160 rpm in an orbital shaker (IST-3075, Jeio Tech, Seoul, South Korea)for 8 h, at room temperature. Finally, the solution was filtered, and chemically analyzed by AAS for copper. The biosorption of metal, (mmol/g), was calculated from the mass balance of the sorption system (Equation (1)):
where V is the volume of the solution (L), W is the amount of dry biomass (g), Ci and Cf are the initial and final concentrations of the metal (mmol/L), respectively.
A kinetic study of biosorption was performed in order to determine the time required by the system to reach equilibrium and the speed of union of the ions to the active external and internal centers of the biosorbent. Different physical and chemical models are used to study the biosorption equilibrium. These models relate the amount of metal adsorbed per unit mass of the adsorbent with the remaining concentration of the metal in the solution (). In this work, the Freundlich and Langmuir expressions were used due to their simplicity, predictive capacity, and goodness-of-fit.
On the one hand, the Freundlich model (Equation (2)) assumes that it adsorbs a single layer of solute onto the surface of the adsorbent, establishing adjacent interactions between the molecules that are adsorbed and a heterogeneous distribution of the adsorption sites due to the diversity of the binding sites, as well as the nature of the adsorbed metal ions:
where (L/g) and n are the Freundlich constants related to the adsorption capacity and the adsorption intensity of the biosorbent by the metal, respectively. On the other hand, the Langmuir model (Equation (3)), supposes a single layer of adsorption with a homogeneous distribution of both the adsorption sites and the adsorption energies without interaction between the adsorbed molecules:
where qmax indicates the maximum adsorption capacity of the metal ion by the biosorbent (mmol/g) and b indicates the affinity of the biosorbent for the metal (L/mmol).
In order to represent the biosorption results by the adsorption models, the equilibrium parameters , , and were fitted by the least squared method. The differences between and were considered as denoted in Equation (4). Where is the experimental metal uptake, is the modeled uptake (by Freundlich and Langmuir expressions), and corresponds to the mean of experimental uptake.
2.4. Desorption
The biomass used in the biosorption experiments was filtered and added to an Erlenmeyer flask with 50 mL of desorbing agent (hydrochloric 0.1 M and sulphuric acid 0.05 M). Desorption was conducted for 90 and 180 min at 160 rpm in an orbital shaker. Finally, the treated biomass was washed with distilled water and calcium chloride to reactivate the biomass and reuse for another biosorption cycle.
The performance of the reused biomass in each adsorption and desorption cycle was evaluated, calculating an effectivity rate () by Equation (5):
where and are the amount of metal ion in solution after desorption and the amount of metal initially biosorbed by biomass (mg or mmol), respectively.
3. Results and Discussions
3.1. Characteristic of Tailing Sample
From the chemical analysis, the composition of main metal species is shown in Table 2, where metals were classified according to their abundance, in major and minor elements. Major metals are expressed in % and the most abundant metals are aluminum and iron. Minor metals, expressed in mg/kg, are mainly represented by copper and manganese. The tailing has a copper concentration above 1500 mg/kg, which is considered high for residual waste because the tailing used came from an old deposit and was produced by less efficient processes of ore flotation.

Table 2.
Chemical composition of main metals in the ore tailing.
The mineralogical analysis suggests that the most abundant species in the tailing samples correspond to quartz and other silicates, and copper is present in low quantity as oxidized species (Table 3). The XRD spectra were added in Figure S1, in the Supplementary Material.

Table 3.
Mineral composition of the ore tailing.
The particle size distribution is given in Figure 4, where it can be observed that around 50% of the mass is particles smaller than 94 μm, and 40% is smaller than 64 μm. This reflects that this tailing is a very fine material, and guided the decision to treat it, subsequently, by agitated leaching.
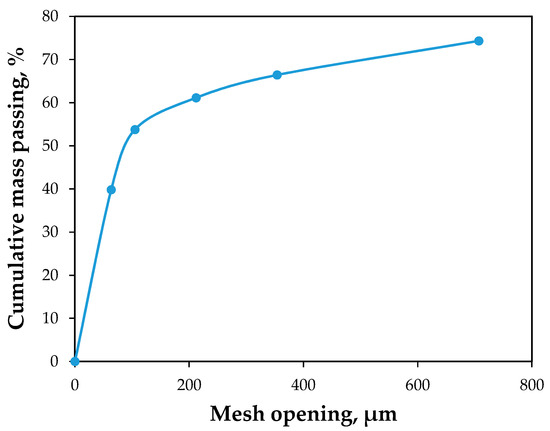
Figure 4.
Particle size distribution of ore tailing.
3.2. Leaching and Treatment of Tailings
The mineralogical profile of tailing suggests that its leaching can be performed using only sulfuric acid, due to copper is as an oxidized mineral (Chalcoalumite) and the ore is very fine, i.e., with a large surface area. The leaching was performed in a mechanically stirred vessel, at 500 rpm.
At the beginning, in the first 4 h leaching, the tailing contained 24.4 mg of copper and, after the treatment, the resulting concentration of copper was 45 mg/L (equivalent to 22.5 mg Cu), which means a recovery of 92%. To concentrate the copper content in the solution, five 4 h leaching cycles were performed, obtaining an increase in copper concentration of approximately 50 mg/L per cycle; at the final cycle, it was reached a concentration of 224 mg/L for copper and 602 mg/L for iron. There is an increase between the first and the fifth cycle of 5.4 and 4.1 times for copper and iron, respectively (Figure 5).
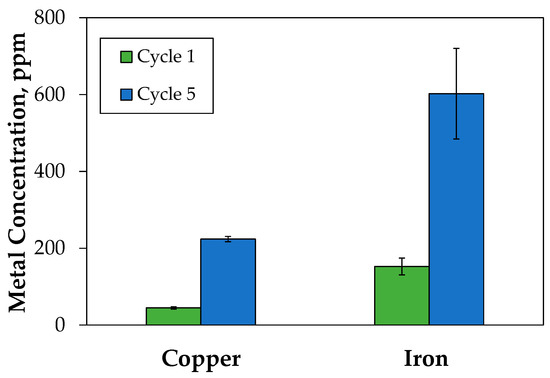
Figure 5.
Leaching of tailing with different dissolution cycles. Contact time per cycle of 4 h and 500 rpm.
Throughout the five cycles, the global recovery remained at 92%, which is good for the treatment of tailings without oxidizing agents. The level of recovery achieved was in the range of observations made by several authors for the leaching of this type of tailings, where the copper source was oxidized and not sulfured, where bioleaching had been a proved alternative [46]. In addition, the use of concentrated acid (1 M) resulted in high recoveries in short times.
With respect to iron precipitation, the removal of the iron excess was addressed by precipitation, adding NaOH to increase the pH to four. A significant removal was obtained, between 65% to 75%, but the losses of copper reached about 30%. Copper moved out, preferably by entrainment and occlusion in the precipitated fraction. The precipitating treatment decreased the iron concentration from 700 to 200 mg/L and copper levels from 200 to 150 mg/L (Figure 6). After the treatment, the compositions of copper and iron in the solution were similar.
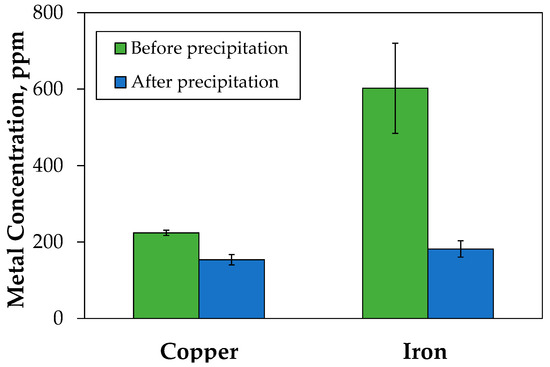
Figure 6.
Effect of precipitation treatment on the removal of iron from PLS. Solution pH 4 after precipitation.
3.3. Biosorption from Synthetic and Leached Tailing Solutions
3.3.1. FTIR
Metal biosorption depends especially on the components of the cell wall. In this context, FTIR was realized in order to characterize the chemical groups that are present in the Gracilaria chilensis biomass and those involved in the biosorption process. The biomass used for the biosorption experiment has chemical groups containing O–, N–, S–, and P–, which participated directly in the binding of metals, such as carboxyl, hydroxyl, and sulfate groups. The groups present in the algal cell wall are polysaccharides, fucoidan and alginate, which was also reported in other works [47,48].
Once detected, the functional groups in the biomass, the FTIR spectrogram of original biomass, were compared with the biomass used for biosorption, detecting changes associated with the influence of metal sorption (Figure 7). FTIR shows shifts of bands related to groups involved in the copper biosorption. The region between 3600 and 3200 cm−1 corresponds to hydroxyl stretches in several compounds, and amines and amides in amino acids. This band is broad because it has a high number and large density of hydrogen bonds. On the one hand, carbonyl stretching of carboxylic acids (C–O) appears near to 1700 cm−1 and the carboxylate stretching at 1650 to 1400 cm−1 (C=O). The carboxylic sites are deprotonated by the effect of Cu(II).
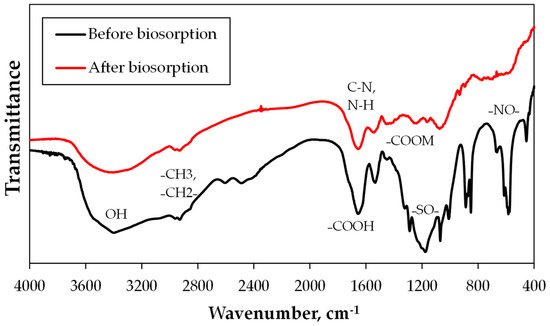
Figure 7.
Vibrational stretches of functional groups in the infrared spectroscopy by Fourier transform (FTIR) analysis before and after the synthetic biosorption.
On the other hand, the band at 1100 cm−1 is assigned to antisymmetric stretching in sulfonate from fucoidan. Primary and secondary amides are observed by the peaks at 3500 to 3200 cm−1 and 1650 to 1500 cm−1, respectively, which are related to amino acids and peptide bonds in proteins and glycoproteins.
The acid treatment of algae dissolves polysaccharide compounds, to a certain extent, in the outer layer of the cell wall, thus, creating additional binding sites (usually amino groups) which can be associated with the decreasing of the intensity of nitro group at −700 cm−1. In addition, the IR bands at around 600 to 650 cm−1 ascribed to the stretching vibration of the Cu–O bond in monoclinic CuO, which was also observed in other works related to bioreduction of copper oxide nanoparticles on the cell wall of different species [49,50].
3.3.2. Biosorption Kinetics and Desorption
In order to study the effect of contact time in the biosorption, the kinetic was studied for 120 min as the maximum time. The observed performance for synthetic and leached tailing solutions were compared, showing that copper uptake increases significantly with time, until approximately 1 h. After, the biosorption capacity is reduced due to the saturation of functional groups. The metal uptake, at the 120 min for synthetic and PLS solutions, obtained a maximum metal uptake of 0.41 and 0.35 mmol/g, respectively (Figure 8). The same response was observed by other authors for the copper recovery with the same genus of algal biomass, where the saturation equilibrium was reached after approximately 60 min, demonstrating that the biosorption is a fast process [51,52]. The presence of other ions in the PLS solution affects the copper biosorption because there could be competition between the multiple ions that compose the liquid matrix of the tailings and copper, which is reflected in lower values of copper uptake.
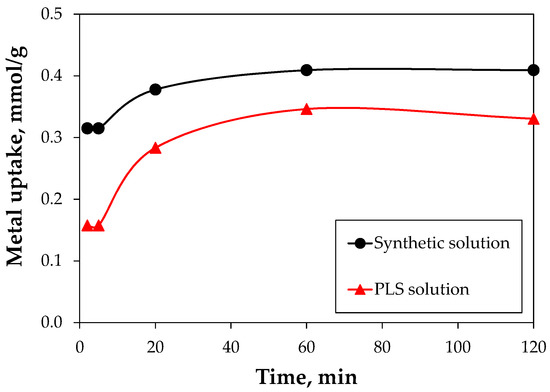
Figure 8.
Copper biosorption kinetics of Gracilaria chilensis for synthetic and leached tailing (PLS) solution at pH 1.5 and biomass concentration of 0.5 g/L.
Among the studies of biosorption that have considered desorption which is important to assess biosorption potential application, multiple agents have been studied, with organic and inorganic acids showing the greatest capacity for desorption [30]. Once biosorption finished, the desorption was studied by changing the properties of the surface through acidification of the media. For this, H2SO4 and HCl were used as desorbing agents, for two desorption periods, 90 and 180 min. On the one hand, the results show that H2SO4 was the best desorbing agent because, for the 90 and 180 min of desorption period, the desorption-adsorption effectivity result was greater, between 1.5- and two-fold, as compared with HCl (Figure 9). On the other hand, the desorption curves for H2SO4 show that no significant differences were obtained between 90 and 180 min, suggesting that a short desorption time is adequate for subsequent biosorption–desorption cycles. In the case of HCl, the time of desorption is critical because the desorption effectivity increases almost two-fold. The best performance of sulfuric acid on hydrochloric acid has also been observed by other authors for chromium [53].
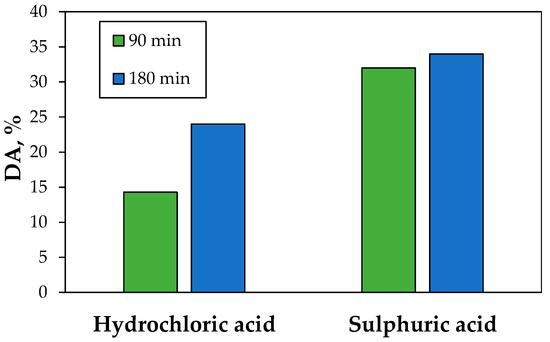
Figure 9.
Desorption/adsorption effectivity of copper by two desorbing agents, HCl and H2SO4, for two different desorption periods, 90 and 180 min.
3.3.3. Equilibrium Isotherms
The metal uptake was analyzed, and the experimental response was simulated by the Langmuir and Freundlich models. The uptake was controlled by the concentration at the equilibrium, which explains the gradual diminution in the variation of at high . Regarding the models, Langmuir, as a hyperbolic expression, appears to be more suitable for representing the whole biosorption, while Freundlich results in the best simulation at high concentrations, but with an overall fitting quality less than Langmuir one (Figure 10).
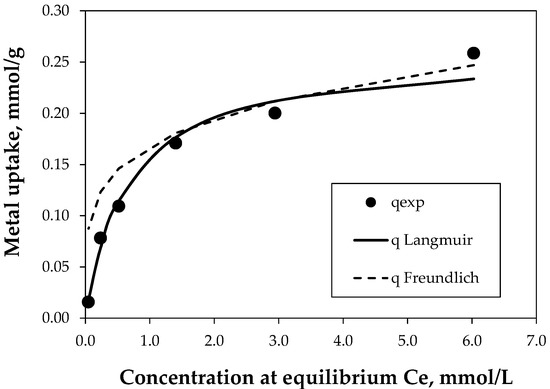
Figure 10.
Equilibrium isotherm of copper biosorption at different concentrations of synthetic solution for 10 h. Biomass concentration of 1 g/L and pH 3, denotes the experimental metal uptake, and represent the Langmuir- and Freundlich-simulated metal uptakes.
The fitting parameters of Langmuir expression were the maximum metal uptake () and the metal affinity (), which was determined by transforming the Langmuir equation into linear form, Equation (6):
In the case of the Freundlich model, the adsorption capacity () and the adsorption intensity () were determined by the log-linearization of the model, Equation (7):
The linear least-squares method and the linearly transformed equations were applied to correlate the sorption data, and the determination coefficient was used as an indicator of quality adjustment (Table 4). According to the results, the biosorption process was best represented by the Langmuir isotherm, where biosorption occurs at sites located on a surface and each retains a single molecule adsorbed to form a monolayer; the adsorption energy is the same in all the binding sites, and there is no interaction between other adsorbed species. The desired adsorption process occurrs when high values of and are obtained. A high value of indicates a high affinity that reflects a strong initial slope in the system. The Freundlich isotherm is an empirical equation; it assumes that the energy of adsorption on the surface is heterogeneous, that is to say, that the energy is not the same at all points on the surface of the adsorbent, in addition, there may be some interactions between the adsorbed molecules. The success of the model depends on the shape of the isotherm when the coefficient is greater than one, the isotherm is favorable.

Table 4.
Fitting of adsorption parameters used in Langmuir and Freundlich models.
The physical meaning of the parameters and the values reached suggest that G. chilensis is a good biosorbent for copper, with magnitude levels comparable with other biosorbents; the equilibrium parameters are in the range of the most common values obtained for nonlead biosorption processes, according to the literature which note high relative frequencies of and for copper of approximately 0.25 to 0.50 mmol/g (55%) and 5 to 10 L/mmol (70%), respectively [54].
One of the mathematical models frequently cited in the literature is the Langmuir model that provides a quantification of the biosorption phenomenon, such as the case for algae used as biosorbents. The maximum sorption uptake and the equilibrium constant or affinity degree of the biomass for the adsorbate can be derived from the Langmuir model. The equilibrium constant represents a fundamental tool in order to define the chemical equilibria in this type of systems. From the knowledge of those constants, it is possible to predict the behavior of different biomass with a given metal. Therefore, this could be a useful tool in order to make the right choice between different biosorbents for each occasion.
The parameters of Langmuir, and , obtained for the biosorption of copper by different types of algae, i.e., brown, red, and green, have been previously studied and are listed in Table 5 [26]. The brown algae present the highest sorption uptakes followed by red algae, especially those with carragenates in their cell walls (Chondrus crispus), and the lowest sorption uptakes correspond to green algae, which are poor in fucoids. The presence of alginates in cell walls of brown algae, similar to carragenates in red algae, is responsible for the binding of metal on the biomass. Alginate has more affinity to bivalent metals which is reflected in the qmax values, where brown algae have higher ones than other kinds of algae [55].

Table 5.
Values of Langmuir constants for copper biosorption by different macroalgae types [26].
According to the reference values, the alga of the present study, G. chilensis, showed a biosorption behavior within the range observed by similar macroalgae. A high-affinity coefficient for copper was reached but a limitation in the biosorption maximal capacity, denoting that the type of chemical groups on the biomass surface is adequate for a preferential copper uptaking but maybe in insufficient quantity.
One of the criteria that should be considered when choosing a bioadsorbent for an industrial application is availability. In this sense, although G. chilensis has proven to be no better than brown algal biomass which generally involve species that are overexploited around the world, and therefore must be protected. Thus, a beneficial aspect is that G. chilensis is an algae that has been cultivated, and therefore can be produced in adapted marine farms.
On the one hand, a considerable number of bacteria, fungi, and algae have been investigated for their biosorbent properties in pure solutions. The efficiency of the process depends on the biomass chemical composition, which varies significantly for different species, even within the same genus or order [56]. On the basis of a compilation of different studies in fungi, bacteria, and algae used as biosorbents, the results show that fungi work at low concentrations and their metal uptake does not exceed 0.24 mmol/g. On the other hand, studies with bacterial biosorbents have a wider range of concentrations, and metal uptakes below 0.87 mmol/g, whereas algae show better biosorption by copper metals, working with large concentration values and obtaining, in some cases for brown algae, uptakes over 1.10 mmol/g (Figure 11). Data used to construct Figure 11 are listed in Table S1 of Supplementary Material.
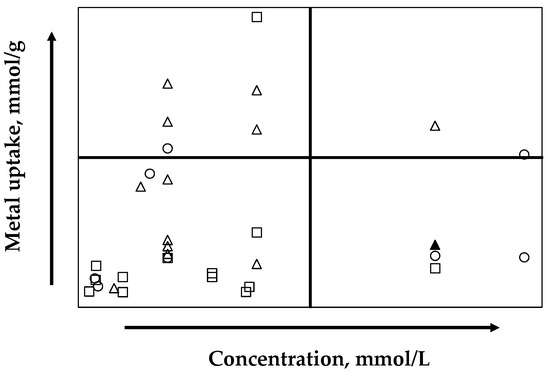
Figure 11.
Global comparison of copper uptake and working concentration of G. chilensis (▲) (this study) with other biomasses reported in the literature. Fungi (□); Bacteria (Ο); and Alga (Δ).
In recent years, there has been increasing interest in the use of new sorbent and very efficient biomaterials. At present, the various bibliographic revisions on the sorption uptake by different biomass do not assert which one is more effective due to the many determining factors involved. A comparison of these biosorbent suggests that algae, in general, are promising biosorbents for heavy metals. Furthermore, algae are natural products collected in large amounts, especially the marine and brown algae. In addition, most algae are innocuous and easily welcomed by the public and environmental agencies for their biotechnological use, since they do not generate toxic or non-removable byproducts [57].
An advantage of copper biosorption with algal biomass is the use of nonliving biomass that handles mining effluents, which have chemical characteristics that are unfavourable to cell growth (acidity, ionic strength, and heavy metal content). In addition, and particularly for G. chilensis, the algae can be cultured, and therefore are considered to be a source of reliable supply. In addition, the biomass acts as an ion exchanger, and therefore the process performs rapidly and the metal is desorbed readily. In terms of the generated residues, once the biosorption is performed, the depleted sorbent is biodegradable, which limits the pressure on the environment due to chemical reagents. The sludge and effluents obtained are potentially integrable to the mineral processes, since the materials used are compatible with any mineral beneficiation technique.
On the basis of a compariosn with the conventional methods to recover and remove metals from effluents, biosorption appears to be an economic alternative because it does not require expensive equipment and supplies and the process is not energetically intensive, such as is the case of ion exchange, electrowinning, reverse osmosis, electrodialysis, and electrocoagulation.
4. Conclusions
The red seaweed Gracilaria chilensis was used as biosorbent of copper in a batch system for synthetic and PLS solutions. Interactions between metal ions and functional groups on the biomass cell wall surface were identified by FTIR. The results propose that the chemical groups involved in the copper biosorption were carboxyl, hydroxyl, and amino.
Parameters influencing biosorption such as the initial metal concentration, the amount of biomass, and the pH of the solution were examined. The biosorption kinetic for synthetic and tailing PLS found that the optimal contact time for the biosorbent was 60 min. The uptake capacity of Cu(II) by the biomass was reduced by the presence of other metallic species in the solution. Experimental biosorption data was represented by the Langmuir and Freundlich isotherms. Langmuir reached the best fitting (R2 = 0.972), indicating that the copper biosorption on the G. chilensis biomass showed a monolayer adsorption trend on the active sites of the biomass surface. Equilibrium parameters were comparable with other reported values for the biosorption of other brown and red algae.
Two desorbent agents were tested, which showed that sulphuric acid was able to release more copper from the biomass in a period of 90 min. HCl was less efficient and require more time.
Finally, Gracilaria biomass has a good copper uptake, working with conditions of pH 1.5, biomass concentration of 0.5 g/L, and initial copper concentration of 200 mg/L. The copper concentration in the tested tailing PLS reached levels that allow its biosorption by five cycles of leaching. Subsequent studies about the optimal conditions of biosorption and the feasibility to recover copper from a continuous columnar system are projected as the next tasks to face.
Bioadsorption and bioreduction are promising techniques for obtaining sustainable products and services, integrating them into the decontamination and recovery of metallic species from industrial and mining effluents. This work is presented as the first research of this nature to deal with mining effluents under a biotechnological approach.
Supplementary Materials
The following are available online at https://www.mdpi.com/2075-163X/10/2/158/s1, Figure S1: XRD spectra of the ore tailing, Table S1: Detailed list of different biomasses used in the biosorption of copper. Parenthesis of algae denote the subclassification: R (red), B (Brown) and G (green).
Author Contributions
Methodology, S.C. and E.E.S.; formal analysis, S.C., E.E.S. and J.I.O.; investigation, S.C., E.E.S and J.I.O.; resources, J.I.O.; writing—original draft preparation, S.C. and J.I.O.; writing—review and editing, J.I.O.; supervision, J.I.O.; project administration, J.I.O. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by CONICYT, projects FONDECYT 11170616 and PIA ACM170005. The APC was funded by FONDECYT 11170616. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Acknowledgments
The authors wish to thank to CONICYT for its financial support through the projects FONDECYT 11170616 and PIA ACM170005.
Conflicts of Interest
The authors declare no conflict of interest.
References
- COCHILCO. Copper and Other Minerals Statistics; COCHILCO: Santiago, Chile, 2017. [Google Scholar]
- US Geological Survey (USGS). Mineral Commodities Summaries. Available online: https://pubs.er.usgs.gov/publication/70140094 (accessed on 10 February 2020).
- SERNAGEOMIN. Catastro de Depósitos de Relaves en Chile; SERNAGEOMIN: Santiago, Chile, 2018. [Google Scholar]
- CORFO. Identificar Elementos de Valor en Residuos Mineros (Relaves) Y Evaluar Su Recuperación Como Productos Comerciales 13BPC3-19021; CORFO: Santiago, Chile, 2013. [Google Scholar]
- Rojas, S. Estudio de la existencia de tierras raras en tranques de relaves y ripios de las plantas de ENAMI. Undergraduate Thesis, Universidad de Atacama, Copiapó, Chile, 2003. [Google Scholar]
- Gómez-Ordóñez, E.; Jiménez-Escrig, A.; Rupérez, P. Dietary fibre and physicochemical properties of several edible seaweeds from the northwestern Spanish coast. Food Res. Int. 2010, 43, 2289–2294. [Google Scholar] [CrossRef]
- Bixler, H.J.; Porse, H. A decade of change in the seaweed hydrocolloids industry. J. Appl. Phycol. 2011, 23, 321–335. [Google Scholar] [CrossRef]
- Vásquez, J.A.; Piaget, N.; Vega, J.M.A. The Lessonia nigrescens fishery in northern Chile: How you harvest is more important than how much you harvest. J. Appl. Phycol. 2012, 24, 417–426. [Google Scholar] [CrossRef]
- Rebours, C.; Marinho-Soriano, E.; Zertuche-González, J.A.; Hayashi, L.; Vásquez, J.A.; Kradolfer, P.; Soriano, G.; Ugarte, R.; Abreu, M.H.; Bay-Larsen, I.; et al. Seaweeds: An opportunity for wealth and sustainable livelihood for coastal communities. J. Appl. Phycol. 2014, 26, 1939–1951. [Google Scholar] [CrossRef] [PubMed]
- Buschmann, A.H.; Prescott, S.; Potin, P.; Faugeron, S.; Vásquez, J.A.; Camus, C.; Infante, J.; Hernández-González, M.C.; Gutierrez, A.; Varela, D.A. The status of kelp exploitation and marine agronomy, with emphasis on Macrocystis pyrifera, in Chile. Adv. Bot. Res. 2004, 71, 161–188. [Google Scholar] [CrossRef]
- FAO. World Aquaculture 2010; FAO: Rome, Italy, 2011. [Google Scholar]
- SERNAPESCA. Informe de Estadísticas Oficiales, Enero-Noviembre 2016; SERNAPESCA: Valparaíso, Chile, 2016. [Google Scholar]
- Cui, J.; Zhang, L. Metallurgical recovery of metals from electronic waste: A review. J. Hazard. Mater. 2008, 158, 228–256. [Google Scholar] [CrossRef]
- Michalak, I.; Chojnacka, K.; Witek-Krowiak, A. State of the art for the biosorption process—A review. Appl. Biochem. Biotechnol. 2013, 170, 1389–1416. [Google Scholar] [CrossRef]
- Vijayaraghavan, K.; Yun, Y.-S. Bacterial biosorbents and biosorption. Biotechnol. Adv. 2008, 26, 266–291. [Google Scholar] [CrossRef]
- Baysal, Z.; Çinar, E.; Bulut, Y.; Alkan, H.; Dogru, M. Equilibrium and thermodynamic studies on biosorption of Pb(II) onto Candida albicans biomass. J. Hazard. Mater. 2009, 161, 62–67. [Google Scholar] [CrossRef]
- Sathishkumar, M.; Mahadevan, A.; Vijayaraghavan, K.; Pavagadhi, S.; Balasubramanian, R. Green recovery of gold through biosorption, biocrystallization, and pyro-crystallization. Ind. Eng. Chem. Res. 2010, 49, 7129–7135. [Google Scholar] [CrossRef]
- Selatnia, A.; Madani, A.; Bakhti, M.Z.; Kertous, L.; Mansouri, Y.; Yous, R. Biosorption of Ni2+ from aqueous solution by a NaOH-treated bacterial dead Streptomyces rimosus biomass. Miner. Eng. 2004, 17, 903–911. [Google Scholar] [CrossRef]
- Davis, T.A.; Volesky, B.; Mucci, A. A review of the biochemistry of heavy metal biosorption by brown algae. Water Res. 2003, 37, 4311–4330. [Google Scholar] [CrossRef]
- Kuyucak, N. Feasibility of Biosorbents Application. In Biosorption of Heavy Metals; Volesky, B., Ed.; CRC Press: Boca Raton, FL, USA, 1990. [Google Scholar]
- Figueira, M.M.; Volesky, B.; Mathieu, H.J. Instrumental analysis study of iron species biosorption by Sargassum biomass. Environ. Sci. Technol. 1999, 33, 1840–1846. [Google Scholar] [CrossRef]
- Fourest, E.; Canal, C.; Roux, J.-C. Improvement of heavy metal biosorption by mycelial dead biomasses (Rhizopus arrhizus, Mucor miehei and Penicillium chrysogenum): PH control and cationic activation. FEMS Microbiol. Rev. 1994, 14, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Khoo, K.-M.; Ting, Y.-P. Biosorption of gold by immobilized fungal biomass. Biochem. Eng. J. 2001, 8, 51–59. [Google Scholar] [CrossRef]
- Raize, O.; Argaman, Y.; Yannai, S. Mechanisms of biosorption of different heavy metals by brown marine macroalgae. Biotechnol. Bioeng. 2004, 87, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Mata, Y.N.; Torres, E.; Blázquez, M.L.; Ballester, A.; González, F.; Muñoz, J.A. Gold(III) biosorption and bioreduction with the brown alga Fucus vesiculosus. J. Hazard. Mater. 2009, 166, 612–618. [Google Scholar] [CrossRef]
- Romera, E.; González, F.; Ballester, A.; Blázquez, M.L.; Muñoz, J.A. Comparative study of biosorption of heavy metals using different types of algae. Bioresour. Technol. 2007, 98, 3344–3353. [Google Scholar] [CrossRef]
- Plaza Cazón, J.; Benítez, L.; Donati, E.; Viera, M. Biosorption of chromium(III) by two brown algae Macrocystis pyrifera and Undaria pinnatifida: Equilibrium and kinetic study. Eng. Life Sci. 2012, 12, 95–103. [Google Scholar] [CrossRef]
- Luna, A.S.; Costa, A.L.H.; da Costa, A.C.A.; Henriques, C.A. Competitive biosorption of cadmium(II) and zinc(II) ions from binary systems by Sargassum filipendula. Bioresour. Technol. 2010, 101, 5104–5111. [Google Scholar] [CrossRef]
- Mata, Y.N.; Blázquez, M.L.; Ballester, A.; González, F.; Muñoz, J.A. Characterization of the biosorption of cadmium, lead and copper with the brown alga Fucus vesiculosus. J. Hazard. Mater. 2008, 158, 316–323. [Google Scholar] [CrossRef]
- Plaza Cazón, J.; Bernardelli, C.; Viera, M.; Donati, E.; Guibal, E. Zinc and cadmium biosorption by untreated and calcium-treated Macrocystis pyrifera in a batch system. Bioresour. Technol. 2012, 116, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Plaza Cazón, J.; Viera, M.; Donati, E.; Guibal, E. Biosorption of mercury by Macrocystis pyrifera and Undaria pinnatifida: Influence of zinc, cadmium and nickel. J. Environ. Sci. 2011, 23, 1778–1786. [Google Scholar] [CrossRef]
- Crist, R.H.; Martin, J.R.; Crist, D.R. Interaction of Metal Ions with Acid Sites of Biosorbents Peat Moss and Vaucheria and Model Substances Alginic and Humic Acids. Environ. Sci. Technol. 1999, 33, 2252–2256. [Google Scholar] [CrossRef]
- Volesky, B. Biosorption of Heavy Metals; Nova Science Publishers: Boca Raton, FL, USA, 1990. [Google Scholar]
- Esposito, A.; Pagnanelli, F.; Vegliò, F. PH-related equilibria models for biosorption in single metal systems. Chem. Eng. Sci. 2002, 57, 307–313. [Google Scholar] [CrossRef]
- Gavrilescu, M. Removal of Heavy Metals from the Environment by Biosorption. Eng. Life Sci. 2004, 4, 219–232. [Google Scholar] [CrossRef]
- Yang, J.; Volesky, B. Modeling Uranium-Proton Ion Exchange in Biosorption. Environ. Sci. Technol. 1999, 33, 4079–4085. [Google Scholar] [CrossRef]
- Goyal, N.; Jain, S.; Banerjee, U. Comparative studies on the microbial adsorption of heavy metals. Adv. Environ. Res. 2003, 7, 7–311. [Google Scholar] [CrossRef]
- Gamez, G.; Dokken, K.; Herrera, I.; Parsons, J.G.; Tiemann, K.J.; Gardea-Torresdey, J.L. Chemical processes involved in Au (III) binding and bioreduction by alfalfa biomass. In Proceedings of the 2000 Conference on Hazardous Waste Research, Denver, CO, USA, 23–25 May 2000. [Google Scholar]
- Gericke, M.; Pinches, A. Biological synthesis of metal nanoparticles. Hydrometallurgy 2006, 83, 132–140. [Google Scholar] [CrossRef]
- Torres, E.; Mata, Y.N.; Blázquez, M.L.; Muñoz, J.A.; González, F.; Ballester, A. Gold and Silver Uptake and Nanoprecipitation on Calcium Alginate Beads. Langmuir 2005, 21, 7951–7958. [Google Scholar] [CrossRef]
- Castro, L.; Blázquez, M.L.; Muñoz, J.A.; González, F.; Ballester, A. Biological synthesis of metallic nanoparticles using algae. IET Nanobiotechnol. 2013, 7, 109–116. [Google Scholar] [CrossRef]
- Mack, C.; Wilhelmi, B.; Duncan, J.R.; Burgess, J.E. Biosorption of precious metals. Biotechnol. Adv. 2007, 25, 264–271. [Google Scholar] [CrossRef]
- Mandal, S.; Phadtare, S.; Sastry, M. Interfacing biology with nanoparticles. Curr. Appl. Phys. 2005, 5, 118–127. [Google Scholar] [CrossRef]
- Wong-Pinto, L.; Menzies, A.; Ordóñez, J.I. Bionanomining: Biotechnological synthesis of metal nanoparticles from mining waste—opportunity for sustainable management of mining environmental liabilities. Appl. Microbiol. Biotechnol. 2020. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Chen, J.P. A comprehensive review on biosorption of heavy metals by algal biomass: Materials, performances, chemistry, and modeling simulation tools. Bioresour. Technol. 2014, 160, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Falagán, C.; Grail, B.M.; Johnson, D.B. New approaches for extracting and recovering metals from mine tailings. Miner. Eng. 2017, 106, 71–78. [Google Scholar] [CrossRef]
- Lesmana, S.O.; Febriana, N.; Soetaredjo, F.E.; Sunarso, J.; Ismadji, S. Studies on potential applications of biomass for the separation of heavy metals from water and wastewater. Biochem. Eng. J. 2009, 44, 19–41. [Google Scholar] [CrossRef]
- Wang, J.; Chen, C. Biosorbents for heavy metals removal and their future. Biotechnol. Adv. 2009, 27, 195–226. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.R. Green synthesis, morphological and optical studies of CuO nanoparticles. J. Mol. Struct. 2017, 1150, 553–557. [Google Scholar] [CrossRef]
- Usman, M.; El Zowalaty, M.; Shameli, K.; Norhazlin, Z.; Salama, M.; Ibrahim, N.A. Synthesis, characterization, and antimicrobial properties of copper nanoparticles. Int. J. Nanomed. 2013, 8, 4467–4479. [Google Scholar] [CrossRef]
- Lavania-Baloo, I.N.; Salihi, I.U.; Zainoddin, J. The use of macroalgae (Gracilaria changii) as bio-adsorbent for Copper (II) removal. IOP Conf. Ser. Mater. Sci. Eng. 2017, 012031. [Google Scholar] [CrossRef]
- Teimouri, A.; Eslamian, S.; Shabankare, A. Removal of heavy metals from aqueous solution by red alga Gracilaria corticata as a new biosorbent. Trends Life Sci. 2016, 5, 236–243. [Google Scholar]
- Higuera, O.; Escalante, H.; Laverde, D. Reducción de cromo contenido en efluentes líquidos de la industria del cuero mediante un proceso de adsorción-desorción con algas marinas. Sci. Tech. 2005, 29, 115–120. [Google Scholar]
- Pagnanelli, F. Equilibrium, Kinetic and Dynamic Modelling of Biosorption Processes. In Microbial Biosorption of Metals; Kotrba, P., Mackova, M., Macek, T., Eds.; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Romera, E.; Ballester, A. Biosorption with Algae: A Statistical Review. Crit. Rev. Biotechnol. 2006, 223–235. [Google Scholar] [CrossRef] [PubMed]
- Gadd, G.M. Biosorption: Critical review of scientific rationale, environmental importance and significance for pollution treatment. J. Chem. Technol. Biotechnol. 2009, 84, 13–28. [Google Scholar] [CrossRef]
- Cuizano, N.; Navarro, A.E. Biosorción de metales pesados por algas marinas: Posible solución a la contaminación a bajas concentraciones. Química y Medio Ambient. 2008, 104, 120–125. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).