Carbonatite-Related REE Deposits: An Overview
Abstract
1. Introduction
2. Types and Basic Geological Characteristics of Carbonatite-Related REE Deposits
2.1. Types of Carbonatite-Related REE Deposits
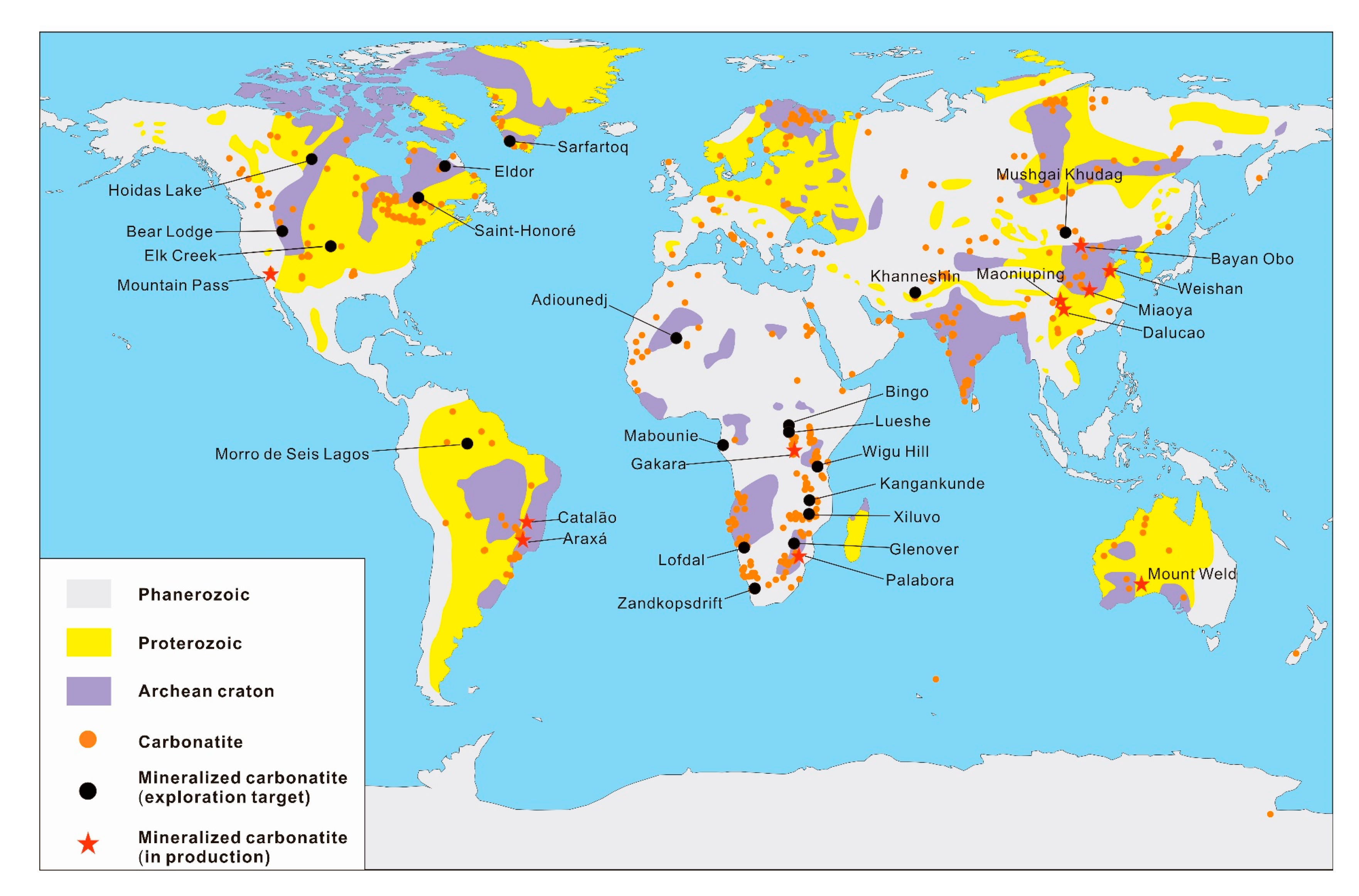
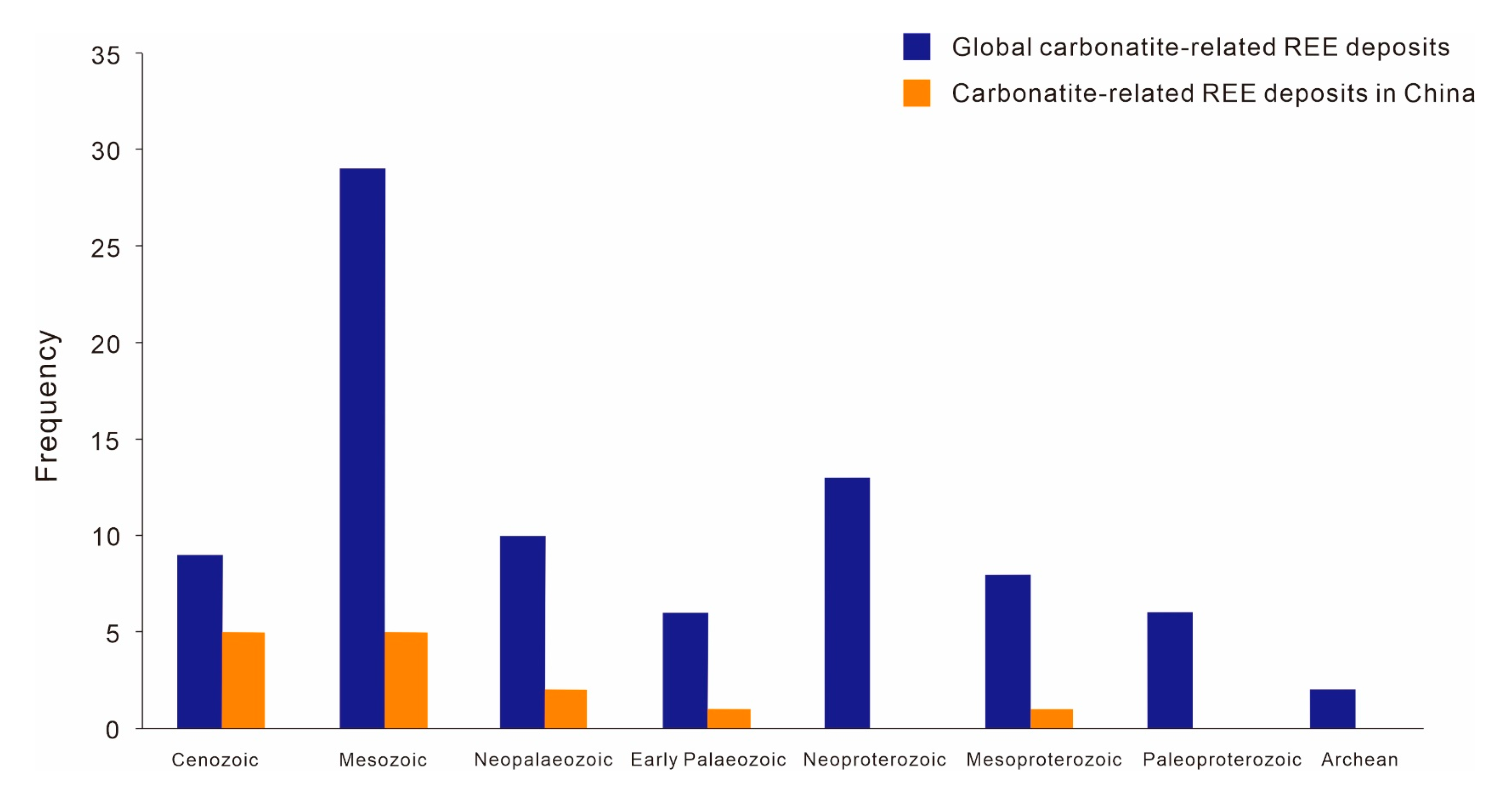
2.2. Spatial and Temporal Distribution
2.3. Ore and Orebody Characteristics
2.4. Geochemical Characteristics
2.5. Alteration Characteristics
3. Typical Carbonatite-Related REE Deposits
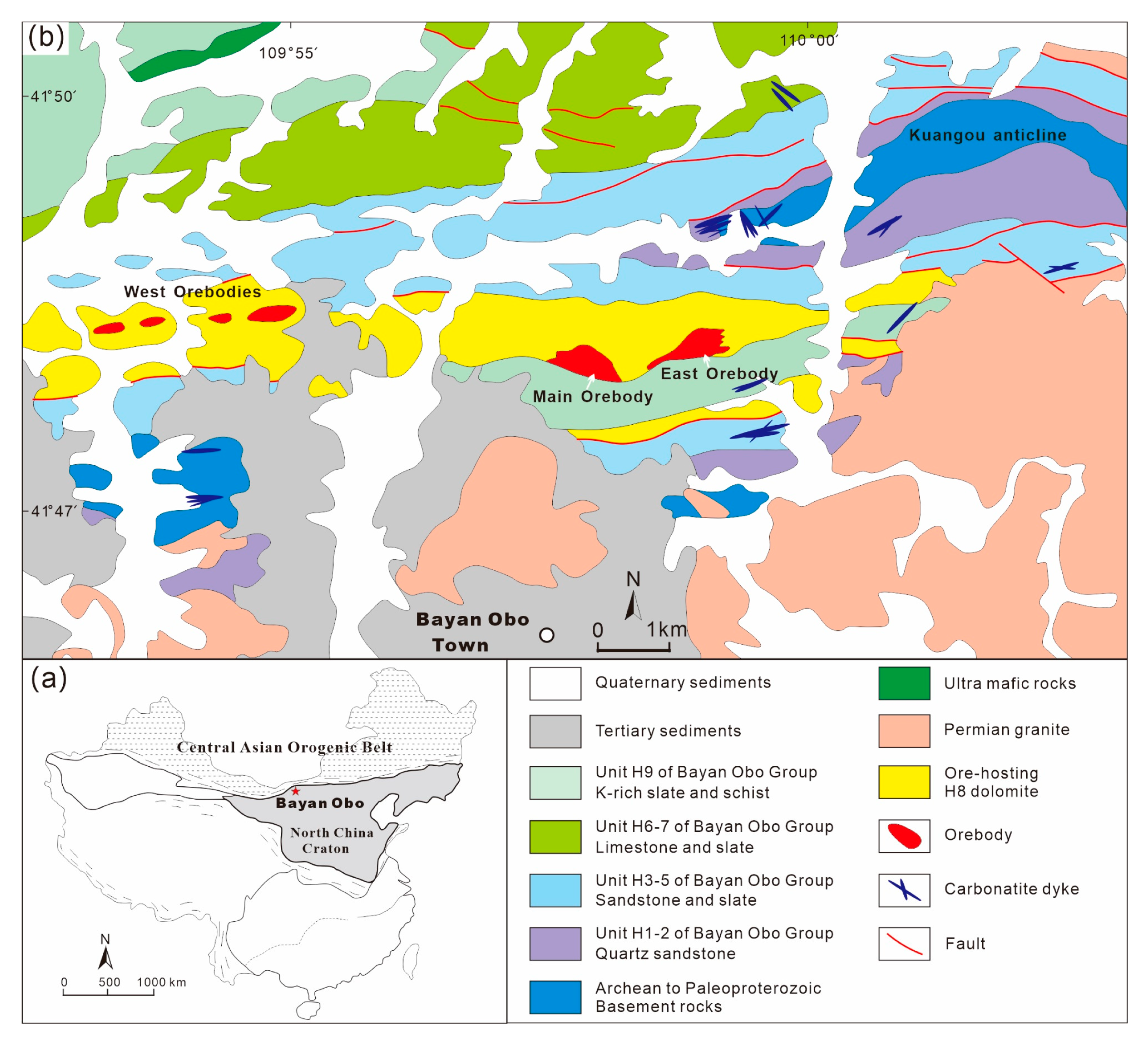
3.1. Bayan Obo REE-Nb-Fe Deposit, China
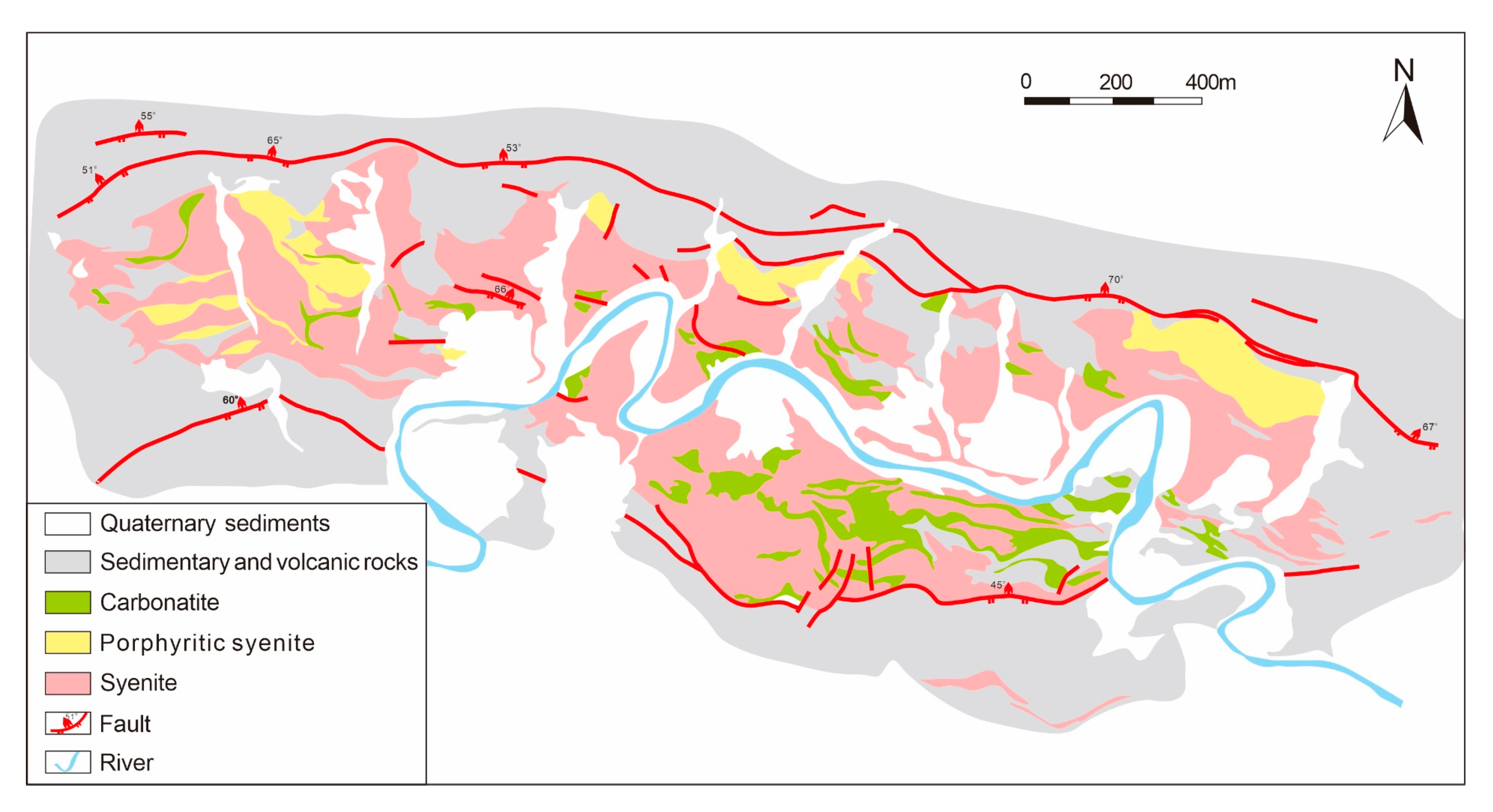
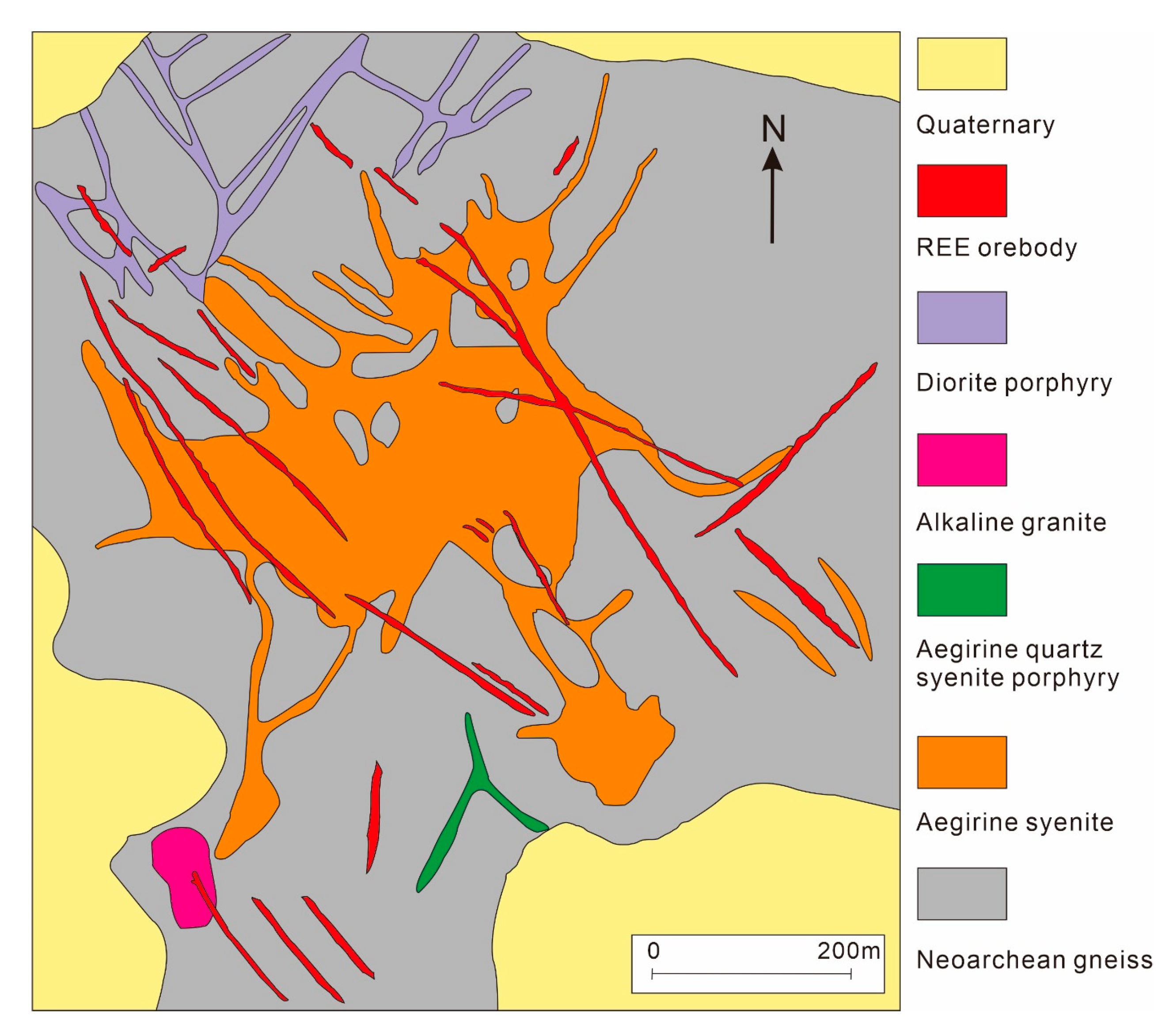
3.2. Miaoya Nb-REE Deposit, China
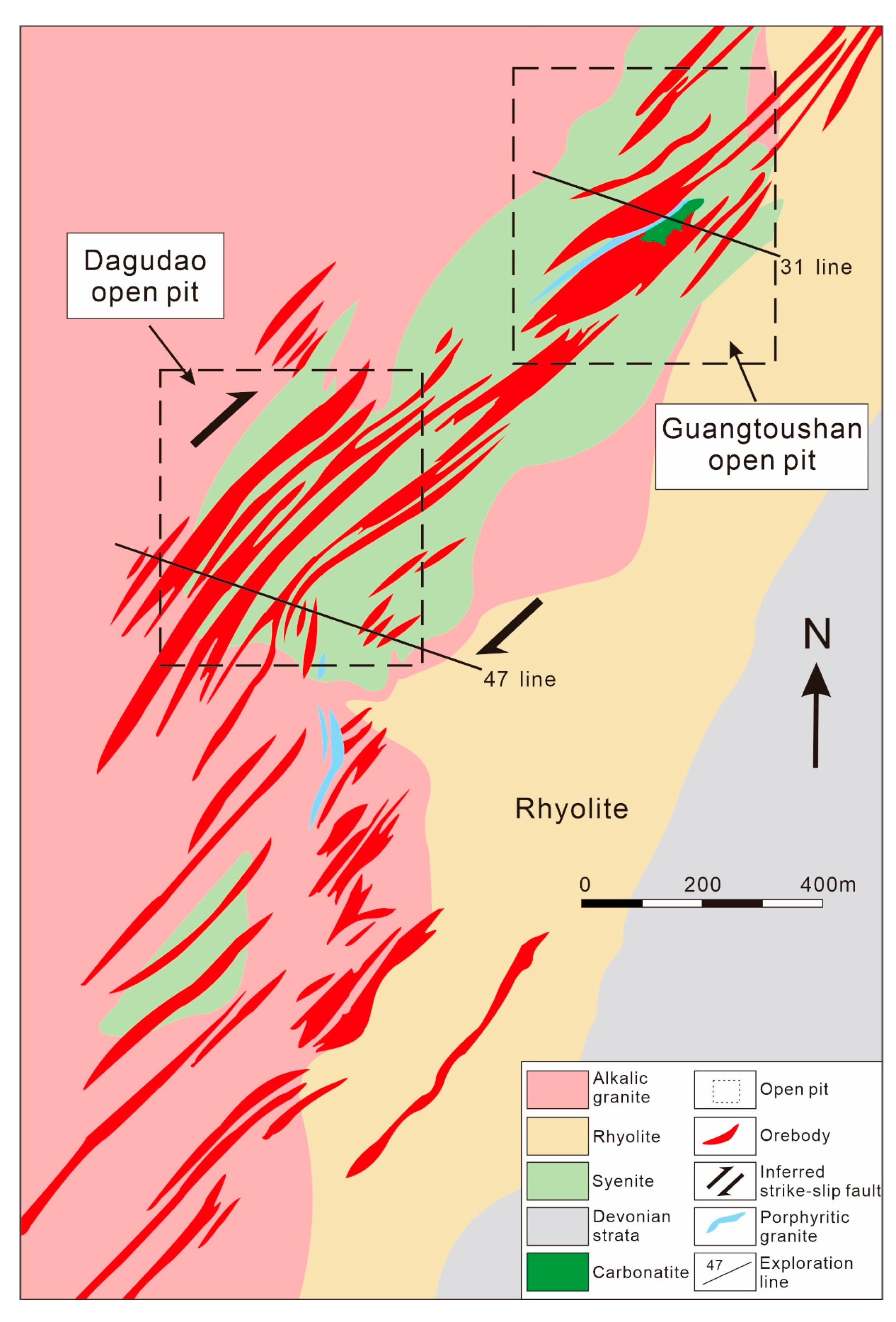
3.3. Maoniuping REE Deposit, China
3.4. Weishan REE Deposit, China
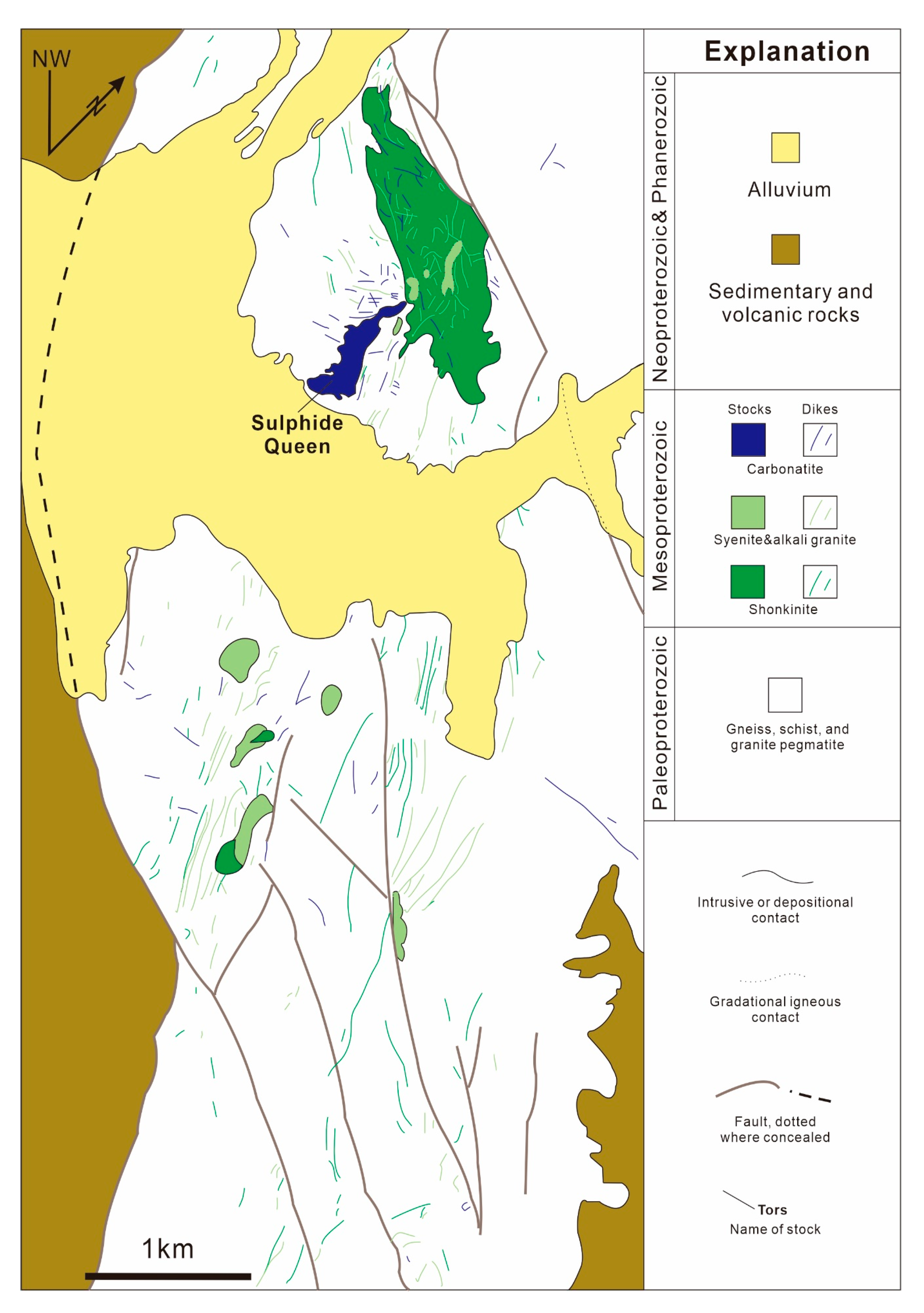
3.5. Mountain Pass REE Deposit, USA
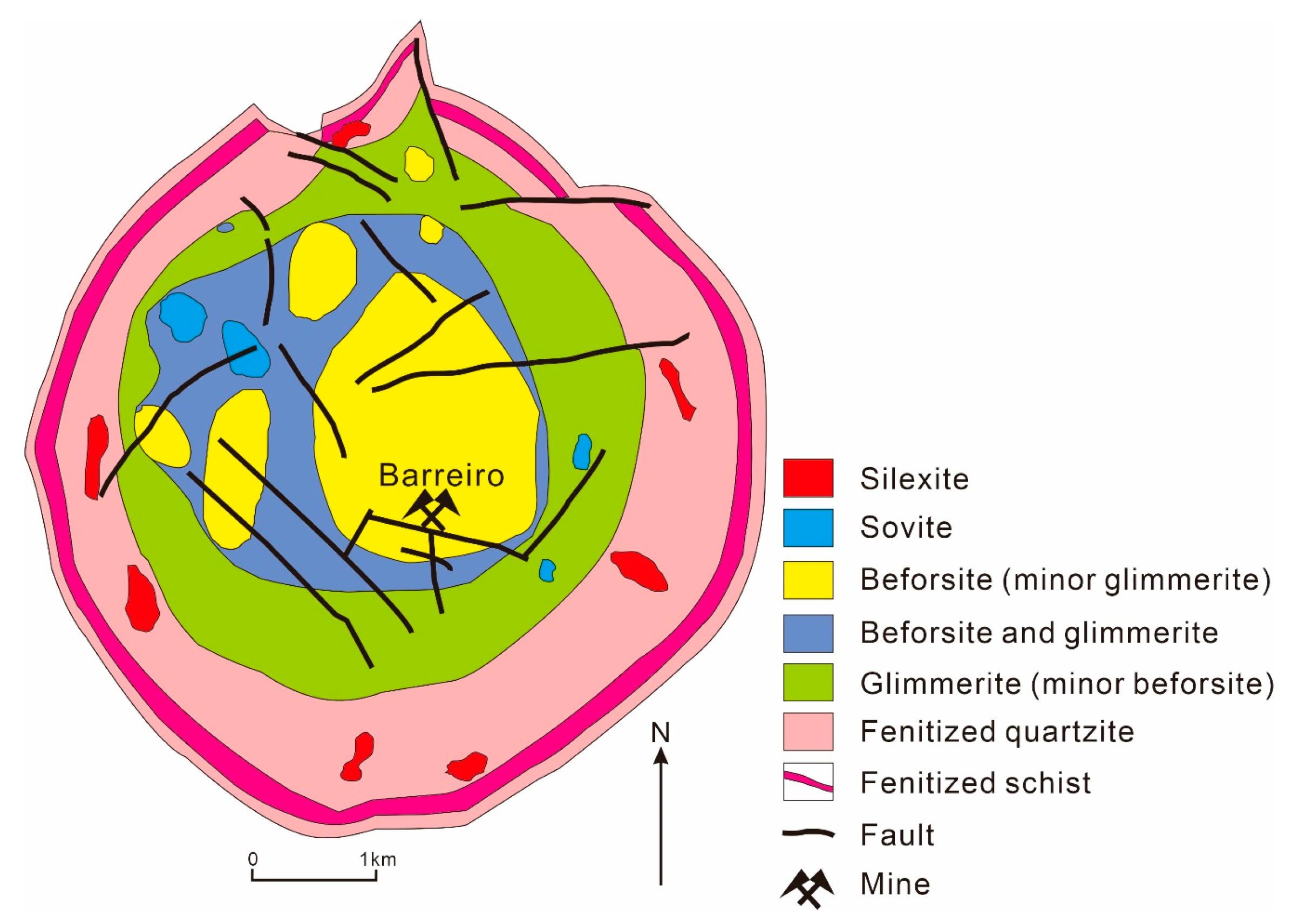
3.6. Araxá Nb-P-REE Deposit, Brazil
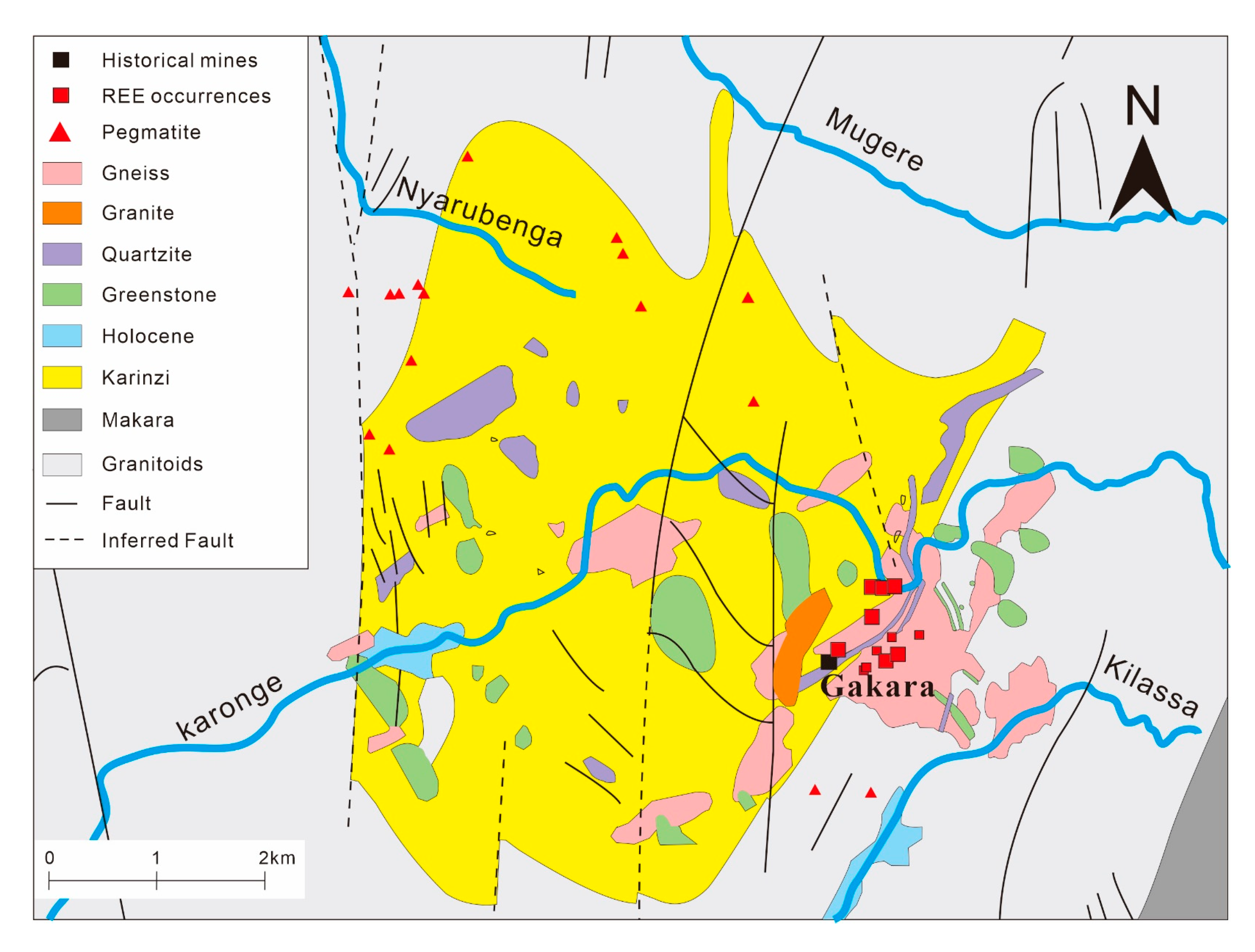
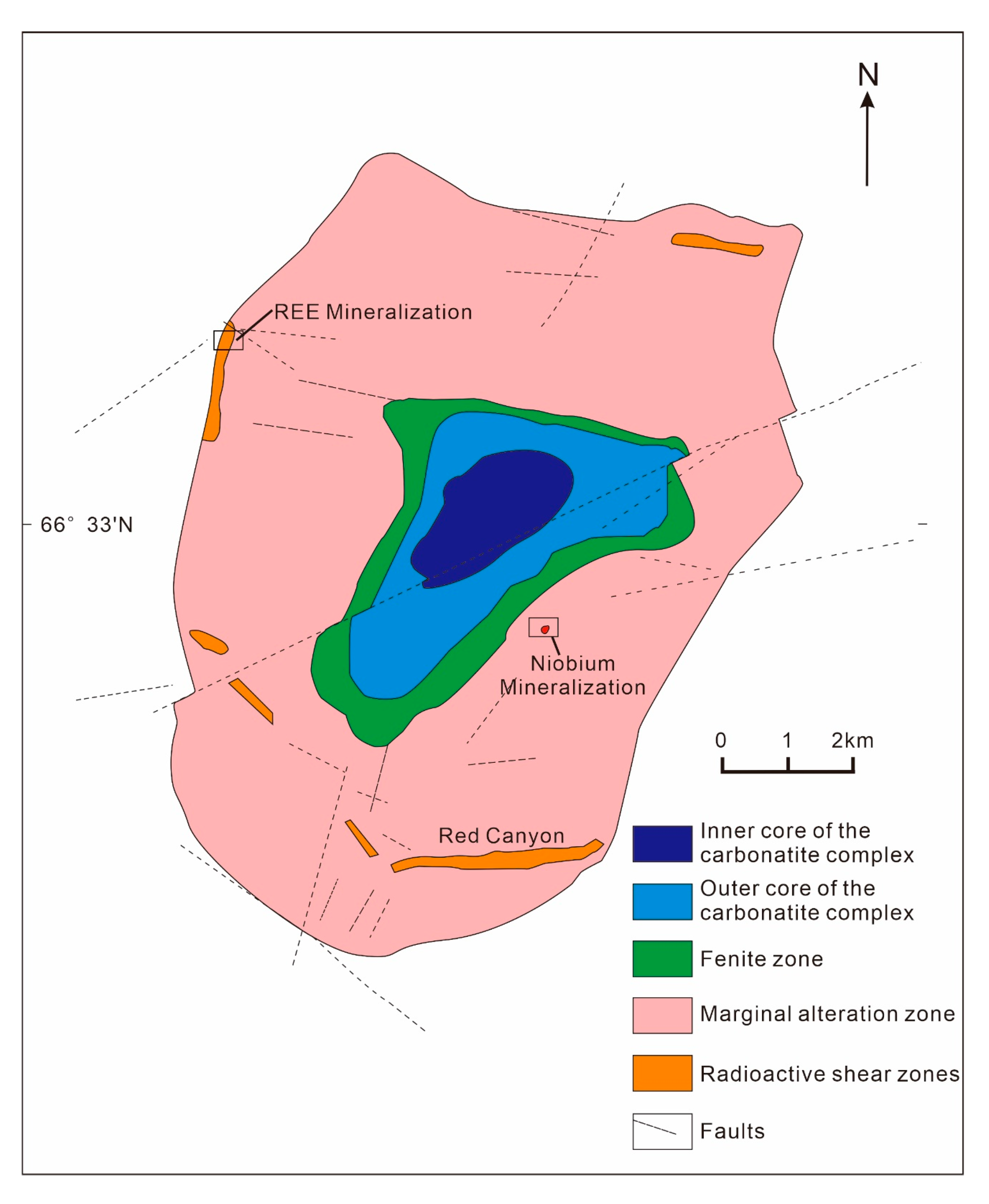
3.7. Gakara REE Deposit, Burundi
3.8. Sarfartoq REE Deposit, Greenland
4. Genesis of Carbonatite-Related REE Deposits
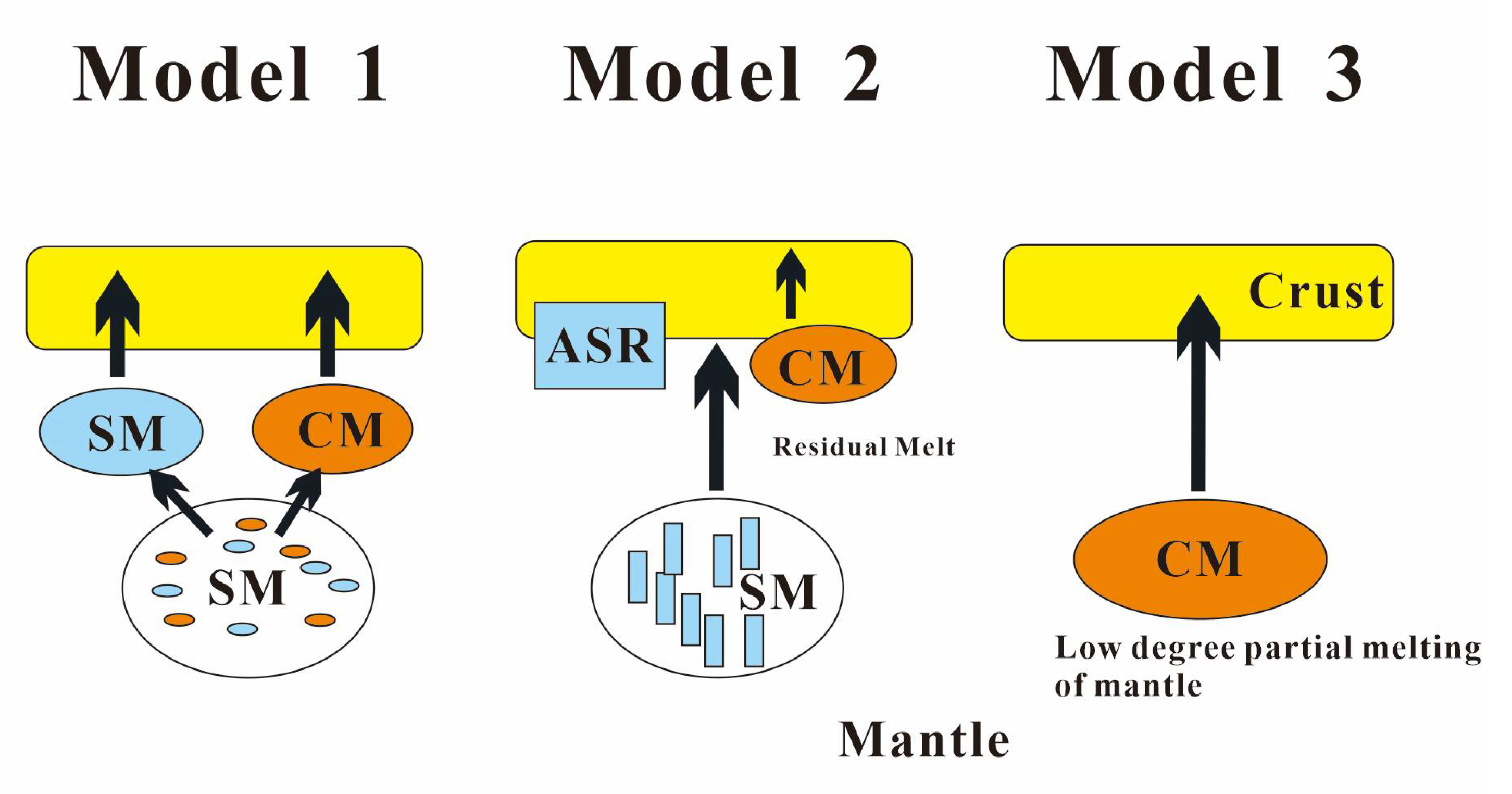
4.1. Origin of Carbonatites
4.2. Source of Ore-Forming Materials
4.3. Nature of Ore-Forming Fluids
4.4. Migration and Precipitation Mechanism
4.5. REE Enrichment Mechanism
4.5.1. Fractional Crystallization of Magma
4.5.2. Immiscibility of Melts and Hydrothermal Fluids
5. Conclusions and Remarks
- (1)
- At present, there is still a lack of reliable evidence to constrain the genesis of carbonatite. Experimental work needs to be designed to verify the influence of immiscibility on rock formation and REE enrichment. Meanwhile, isotope analysis, especially the non-traditional stable isotopes (such as Fe, Mg, Ba) can be carried out to examine the source of magma and its relationship with the mantle.
- (2)
- In view of the unclear REE partitioning behaviors in a magmatic–hydrothermal system, experimental petrology work on this aspect will enhance the understanding of the distribution of REEs in different systems, such as between carbonatite melts rich in F, S, P and silicate melts, between carbonates (its symbiotic precipitates) and carbonatite melts, between carbonatite melts and fluid or gas phase. This development will provide a better understanding of REE enrichment mechanisms.
- (3)
- Research on carbonatite-related REE deposits should expand to both REE fertile and barren carbonatites. Although carbonatite is absent in some REE deposits, the formation of those deposits is still closely related to carbonatite magma (such as the Nolans Bore P-REE-Th deposit, Australia) [131,132,133]. By using the method of “comparative metallogeny”, a comparative study on the metallogenic characteristics of carbonatites under different or the same tectonic background should be strengthened. In combination with better knowledge of the REE partitioning behavior and the sequence of mineral formation in high temperature and pressure experiments, a critical understanding of the factors controlling the fertility of carbonatites will eventually be achieved.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Loges, A.; Migdisov, A.A.; Wagner, T.; Williams-Jones, A.E.; Markl, G. An experimental study of the aqueous solubility and speciation of Y(III) fluoride at temperatures up to 250 °C. Geochim. Cosmochim. Acta 2013, 123, 403–415. [Google Scholar] [CrossRef]
- Binnemans, K.; Jones, P.T.; Blanpain, B.; Van Gerven, T.; Yang, Y.; Walton, A.; Buchert, M. Recycling of rare earths: A critical review. J. Clean. Prod. 2013, 51, 1–22. [Google Scholar] [CrossRef]
- Jha, M.K.; Kumari, A.; Panda, R.; Rajesh Kumar, J.; Yoo, K.; Lee, J.Y. Review on hydrometallurgical recovery of rare earth metals. Hydrometallurgy 2016, 161, 77. [Google Scholar] [CrossRef]
- Jordens, A.; Cheng, Y.P.; Waters, K.E. A review of the beneficiation of rare earth element bearing minerals. Miner. Eng. 2013, 41, 97–114. [Google Scholar] [CrossRef]
- Chakhmouradian, A.R.; Wall, F. Rare Earth Elements: Minerals, Mines, Magnets (and More). Elements 2012, 8, 333–340. [Google Scholar] [CrossRef]
- Kumari, A.; Panda, R.; Jha, M.K.; Kumar, J.R.; Lee, J.Y. Process development to recover rare earth metals from monazite mineral: A review. Miner. Eng. 2015, 79, 102–115. [Google Scholar] [CrossRef]
- Zhang, S.J.; Zhang, L.W.; Zhang, Y.W.; Shang, L.; Li, J.B. Summarize on rare earth mineral resources and their distribution at home and abroad. Inorg. Chem. Ind. 2020, 52, 9–16, (In Chinese with English abstract). [Google Scholar]
- Li, Z.; Hu, J.Z. World rare earth resources survey and development utilization trend. Mod. Min. 2017, 33, 97–105, (In Chinese with English abstract). [Google Scholar]
- Li, F.Q.; Dai, T.; Wang, G.S. A review on recycling and reuse of rare earth metals. Conserv. Util. Min. Resour. 2019, 39, 84–89, (In Chinese with English abstract). [Google Scholar]
- Wang, D.H.; Zhao, Z.; Yu, Y.; Wang, C.H.; Dai, J.J.; Sun, Y.; Zhao, T.; Li, J.K.; Huang, F.; Chen, Z.Y.; et al. A review of the achievements in the survey and study of ion-absorption type REE deposits in China. Acta Geosci. Sin. 2017, 38, 317–325, (In Chinese with English abstract). [Google Scholar]
- Fu, T.Y.; Li, B.H.; Dong, X.Y.; Xu, L. Analysis on the distribution, classification and characteristics of rare earth deposits in China. J. Henan Sci. Technol. 2015, 14, 124–126, (In Chinese with English abstract). [Google Scholar]
- Simandl, G.J.; Paradis, S. Carbonatites: Related ore deposits, resources, footprint, and exploration methods. Appl. Earth Sci. 2018, 127, 123–152. [Google Scholar] [CrossRef]
- Sun, J.; Zhu, X.K.; Chen, Y.L.; Fang, N.; Li, S.Z. Is the Bayan Obo ore deposit a micrite mound? A comparison with the Sailinhudong micrite mound. Int. Geol. Rev. 2014, 56, 1720–1731. [Google Scholar] [CrossRef]
- Liu, Q.P.; Zhao, Y.Y.; Liu, C.H. REE resources potential in Greenland and the availability evaluation in favor of China. Geol. Bull. China 2019, 38, 1386–1395, (In Chinese with English abstract). [Google Scholar]
- Schonwandt, H.K.; Barnes, G.B.; Ulrich, T. Chapter 5—A Description of the World-Class Rare Earth Element Deposit, Tanbreez, South Greenland. In Rare Earths Industry: Technological, Economic, and Environmental Implications; Elsevier: Amsterdam, The Netherlands, 2016; pp. 73–85. [Google Scholar]
- Streckeisen, A. Classification and nomenclature of volcanic rocks, lamprophyres, carbonatites and melilitic rocks IUGS Subcommission on the Systematics of Igneous Rocks. Geol. Rundsch. 1980, 69, 194–207. [Google Scholar] [CrossRef]
- Gou, R.T.; Zeng, P.S.; Liu, S.W.; Ma, J.; Wang, J.J.; Dai, Y.J.; Wang, Z.Q. Distribution characteristics of carbonatites of the world and its metallogenic significance. Acta Geol. Sin. 2019, 93, 2348–2361, (In Chinese with English abstract). [Google Scholar]
- Chakhmouradian, A.R.; Cooper, M.A.; Medici, L.; Abdu, Y.A.; Shelukhina, Y.S. Anzaite-(Ce), a new rare-earth mineral and structure type from the Afrikanda silicocarbonatite, Kola Peninsula, Russia. Mineral. Mag. 2018, 79, 1231–1244. [Google Scholar] [CrossRef]
- Mattsson, H.B.; Balashova, A.; Almqvist, B.S.G.; Bosshard-Stadlin, S.A.; Weidendorfer, D. Magnetic mineralogy and rock magnetic properties of silicate and carbonatite rocks from Oldoinyo Lengai volcano (Tanzania). J. Afr. Earth Sci. 2018, 142, 193–206. [Google Scholar] [CrossRef]
- Yang, K.F.; Fan, H.R.; Pirajno, F.; Li, X.C. The Bayan Obo (China) giant REE accumulation conundrum elucidated by intense magmatic differentiation of carbonatite. Geology 2019, 47, 1198–1202. [Google Scholar] [CrossRef]
- Yang, Z.M.; Woolley, A. Carbonatites in China: A review. J. Asian Earth Sci. 2006, 27, 559–575. [Google Scholar] [CrossRef]
- Linnen, R.L.; Samson, I.M.; Williams-Jones, A.E.; Chakhmouradian, A.R. Geochemistry of the Rare-Earth Element, Nb, Ta, Hf, and Zr Deposits. In Treatise on Geochemistry; Elsevier: Amsterdam, The Netherlands, 2014; pp. 543–568. [Google Scholar]
- Song, W.L.; Xu, C.; Wang, L.J.; Wu, M.; Zeng, L.; Wang, L.Z.; Feng, M. Review of the metallogenesis of the endogenetic rare earth elements deposits related to carbonatite-alkaline complex. Acta Sci. Nat. Univ. Pekin. 2013, 49, 725–740, (In Chinese with English abstract). [Google Scholar]
- Weng, Z.H.; Jowitt, S.M.; Mudd, G.M.; Haque, N. A Detailed Assessment of Global Rare Earth Element Resources: Opportunities and Challenges. Econ. Geol. 2015, 110, 1925–1952. [Google Scholar] [CrossRef]
- Kanazawa, Y.; Kamitani, M. Rare earth minerals and resources in the world. J. Alloy. Compd. 2006, 408–412, 1339–1343. [Google Scholar] [CrossRef]
- Woolley, A.R.; Kjarsgaard, B.A. Carbonatite occurrences of the world: Map and database. Geol. Surv. Can. Open File 2008, 5796, 1–21. [Google Scholar]
- Liu, Y.; Hou, Z.Q. A synthesis of mineralization styles with an integrated genetic model of carbonatite-syenite-hosted REE deposits in the Cenozoic Mianning-Dechang REE metallogenic belt, the eastern Tibetan Plateau, southwestern China. J. Asian Earth Sci. 2017, 137, 35–79. [Google Scholar] [CrossRef]
- Ying, Y.C.; Chen, W.; Lu, J.; Jiang, S.Y.; Yang, Y.H. In situ U–Th–Pb ages of the Miaoya carbonatite complex in the South Qinling orogenic belt, central China. Lithos 2017, 290–291, 159–171. [Google Scholar] [CrossRef]
- Verplanck, P.L.; Mariano, A.N.; Mariano, A., Jr. Rare Earth Element Ore Geology of Carbonatites. In Rare Earth and Critical Elements in Ore Deposits; Verplanck, P.L., Hitzman, M.W., Eds.; Soc Economic Geologists, Inc.: Littleton, CO, USA, 2016; pp. 5–32. [Google Scholar]
- Xie, Y.L.; Hou, Z.Q.; Goldfarb, R.J.; Guo, X.; Wang, L. Rare Earth Element Deposits in China. In Rare Earth and Critical Elements in Ore Deposits; Verplanck, P.L., Hitzman, M.W., Eds.; Soc Economic Geologists, Inc.: Littleton, CO, USA, 2016; pp. 115–136. [Google Scholar]
- Lai, X.D.; Yang, X.Y. U-Pb Ages and Hf Isotope of Zircons from a Carbonatite Dyke in the Bayan Obo Fe-REE Deposit in Inner Mongolia: Its Geological Significance. Acta Geol. Sin. Engl. Ed. 2019, 93, 1783–1796. [Google Scholar] [CrossRef]
- Xie, Y.L.; Li, Y.X.; Cooke, D.; Kamenetsky, V.; Chang, Z.S.; Danyushevsky, L.; Dominy, S.; Ryan, C.; Laird, J. Geochemical characteristics of carbonatite fluids at the Maoniuping REE deposit, Western Sichuan, China. In Let’s Talk Ore Deposits. In Proceedings of the Eleventh Biennial SGA Meeting, Antofagasta, Chile, 26–29 September 2011; Volume 1, pp. 196–198. [Google Scholar]
- Zhu, J.; Wang, L.X.; Peng, S.G.; Peng, L.H.; Wu, C.X.; Qiu, X.F. U-Pb zircon age, geochemical and isotopic characteristics of the Miaoya syenite and carbonatite complex, central China. Geol. J. 2017, 52, 938–954. [Google Scholar] [CrossRef]
- Wang, C.; Liu, J.C.; Zhang, H.D.; Zhang, X.Z.; Zhang, D.M.; Xi, Z.; Wang, Z.X.; Wang, Z.J. Geochronology and mineralogy of the Weishan carbonatite in Shandong province, eastern China. Geosci. Front. 2019, 10, 769–785. [Google Scholar] [CrossRef]
- Xu, C.; Campbell, I.H.; Allen, C.M.; Chen, Y.J.; Huang, Z.L.; Qi, L.; Zhang, G.S.; Yan, Z.F. U-Pb zircon age, geochemical and isotopic characteristics of carbonatite and syenite complexes from the Shaxiongdong, China. Lithos 2008, 105, 118–128. [Google Scholar] [CrossRef]
- Liu, S.; Fan, H.R.; Groves, D.I.; Yang, K.F.; Yang, Z.F.; Wang, Q.W. Multiphase carbonatite-related magmatic and metasomatic processes in the genesis of the ore-hosting dolomite in the giant Bayan Obo REE-Nb-Fe deposit. Lithos 2020, 354–355, 105359. [Google Scholar] [CrossRef]
- Yang, X.Y.; Lai, X.Y.; Pirajno, F.; Liu, Y.L.; Ling, M.X.; Sun, W.D. Genesis of the Bayan Obo Fe-REE-Nb formation in Inner Mongolia, North China Craton: A perspective review. Precambrian Res. 2017, 288, 39–71. [Google Scholar] [CrossRef]
- Song, W.L.; Xu, C.; Smith, M.P.; Chakhmouradian, A.R.; Brenna, M.; Kynický, J.; Chen, W.; Yang, Y.H.; Deng, M.; Tang, H.Y. Genesis of the world’s largest rare earth element deposit, Bayan Obo, China: Protracted mineralization evolution over ~1 b.y. Geology 2018, 46, 323–326. [Google Scholar] [CrossRef]
- Hu, L.; Li, Y.K.; Wu, Z.J.; Bai, Y.; Wang, A.J. Two metasomatic events recorded in apatite from the ore-hosting dolomite marble and implications for genesis of the giant Bayan Obo REE deposit, Inner Mongolia, Northern China. J. Asian Earth Sci. 2019, 172, 56–65. [Google Scholar] [CrossRef]
- Fan, H.R.; Yang, K.F.; Hu, F.F.; Liu, S.; Wang, K.Y. The giant Bayan Obo REE-Nb-Fe deposit, China: Controversy and ore genesis. Geosci. Front. 2016, 7, 335–344. [Google Scholar] [CrossRef]
- Su, J.H.; Zhao, X.F.; Li, X.C.; Hu, W.; Chen, M.; Xiong, Y.L. Geological and geochemical characteristics of the Miaoya syenite-carbonatite complex, Central China: Implications for the origin of REE-Nb-enriched carbonatite. Ore Geol. Rev. 2019, 113, 103101. [Google Scholar] [CrossRef]
- Ying, Y.C.; Chen, W.; Simonetti, A.; Jiang, S.Y.; Zhao, K.D. Significance of hydrothermal reworking for REE mineralization associated with carbonatite: Constraints from in situ trace element and C-Sr isotope study of calcite and apatite from the Miaoya carbonatite complex (China). Geochim. Cosmochim. Acta 2020, 280, 340–359. [Google Scholar] [CrossRef]
- Xu, C.; Kynicky, J.; Chakhmouradian, A.R.; Campbell, I.H.; Allen, C.M. Trace-element modeling of the magmatic evolution of rare-earth-rich carbonatite from the Miaoya deposit, Central China. Lithos 2010, 118, 145–155. [Google Scholar] [CrossRef]
- Jia, Y.H.; Liu, Y. REE Enrichment during Magmatic-Hydrothermal Processes in Carbonatite-Related REE Deposits: A Case Study of the Weishan REE Deposit, China. Minerals 2019, 10, 25. [Google Scholar] [CrossRef]
- Hou, Z.Q.; Tian, S.H.; Xie, Y.L.; Yang, Z.; Yuan, Z.S.; Yin, S.P.; Yi, L.S.; Fei, H.C.; Zou, T.R.; Bai, G.; et al. The Himalayan Mianning-Dechang REE belt associated with carbonatite-alkaline complexes, eastern Indo-Asian collision zone, SW China. Ore Geol. Rev. 2009, 36, 65–89. [Google Scholar] [CrossRef]
- Woolley, A.R. A discussion of carbonatite evolution and nomenclature, and the generation of sodic and potassic fenites. Mineral. Mag. 1982, 46, 13–17. [Google Scholar] [CrossRef]
- Liu, S.; Fan, H.R.; Yang, K.F.; Hu, F.F.; Rusk, B.; Liu, X.; Li, X.C.; Yang, Z.F.; Wang, Q.W.; Wang, K.Y. Fenitization in the giant Bayan Obo REE-Nb-Fe deposit: Implication for REE mineralization. Ore Geol. Rev. 2018, 94, 290–309. [Google Scholar] [CrossRef]
- Migdisov, A.A.; Williams-Jones, A.E. Hydrothermal transport and deposition of the rare earth elements by fluorine-bearing aqueous liquids. Miner. Depos. 2014, 49, 987–997. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Z.Y.; Yang, Z.S.; Sun, X.; Zhu, Z.M.; Zhang, Q.C. Mineralogical and geochemical studies of brecciated ores in the Dalucao REE deposit, Sichuan Province, southwestern China. Ore Geol. Rev. 2015, 70, 613–636. [Google Scholar] [CrossRef]
- Williams-Jones, A.E.; Migdisov, A.A.; Samson, I.M. Hydrothermal Mobilisation of the Rare Earth Elements—A Tale of “Ceria” and “Yttria”. Elements 2012, 8, 355–360. [Google Scholar] [CrossRef]
- Cooper, A.F.; Palin, J.M.; Collins, A.K. Fenitization of metabasic rocks by ferrocarbonatites at Haast River, New Zealand. Lithos 2016, 244, 109–121. [Google Scholar] [CrossRef]
- Currie, K.L.; Ferguson, J. A Study of Fenitization Around the Alkaline Carbonatite Complex at Callander Bay, Ontario, Canada. Can. J. Earth Sci. 1971, 8, 498–517. [Google Scholar] [CrossRef]
- Currie, K.L.; Ferguson, J. A Study of Fenitization in Mafic Rocks, with Special Reference to the Callander Bay Complex. Can. J. Earth Sci. 1972, 9, 1254–1261. [Google Scholar] [CrossRef]
- McKie, D. Fenitization. In Tuttle of Gittins J. (eds.) & Carbonatites; Interscience: New York, NY, USA, 1966; pp. 261–294. [Google Scholar]
- Le Bas, M.J. Fenites Associated with Carbonatites. Can. Mineral. 2008, 46, 915–932. [Google Scholar] [CrossRef]
- Yang, X.M.; Yang, X.Y.; Fan, H.R.; Guo, F.; Zhang, Z.F.; Zheng, Y.F. Petrological characteristics of fenites and their geological significance. Geol. Rev. 2000, 46, 481–490, (In Chinese with English abstract). [Google Scholar]
- Potter, N.J.; Kamenetsky, V.S.; Simonetti, A.; Goemann, K. Different types of liquid immiscibility in carbonatite magma: A case study of the Oldoinyo Lengai 1993 lava and melt inclusions. Chem. Geol. 2017, 455, 376–384. [Google Scholar] [CrossRef]
- Mitchell, R.H. Peralkaline nephelinite-natrocarbonatite immiscibility and carbonatite assimilation at Oldoinyo Lengai, Tanzania. Contrib. Mineral. Petrol. 2009, 158, 589–598. [Google Scholar] [CrossRef]
- Kresten, P.; Morogan, V. Fenitization at the Fen complex, southern Norway. Lithos 1986, 19, 27–42. [Google Scholar] [CrossRef]
- Kresten, P. The chemistry of fenitization: Examples from Fen, Norway. Chem. Geol. 1988, 68, 329–349. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, X.Q.; Nie, L.S.; Jennifer, M.M.; Liu, H.L.; Zhang, B.M.; Han, Z.X. Geochemical background and dispersion pattern of the world’s largest REE deposit of Bayan Obo, China. J. Geochem. Explor. 2020, 215, 106545. [Google Scholar] [CrossRef]
- Zhang, S.H.; Zhao, Y.; Liu, Y. A precise zircon Th-Pb age of carbonatite sills from the world’s largest Bayan Obo deposit: Implications for timing and genesis of REE-Nb mineralization. Precambrian Res. 2017, 291, 202–219. [Google Scholar] [CrossRef]
- Smith, M.P.; Campbell, L.S.; Kynicky, J. A review of the genesis of the world class Bayan Obo Fe-REE-Nb deposits, Inner Mongolia, China: Multistage processes and outstanding questions. Ore Geol. Rev. 2015, 64, 459–476. [Google Scholar] [CrossRef]
- Chen, W.; Liu, H.Y.; Lu, J.; Jiang, S.Y.; Simonetti, A.; Xu, C.; Zhang, W. The formation of the ore-bearing dolomite marble from the giant Bayan Obo REE-Nb-Fe deposit, Inner Mongolia: Insights from micron-scale geochemical data. Miner. Depos. 2019, 55, 131–146. [Google Scholar] [CrossRef]
- Yang, K.F.; Fan, H.R.; Santosh, M.; Hu, F.F.; Wang, K.Y. Mesoproterozoic carbonatitic magmatism in the Bayan Obo deposit, Inner Mongolia, North China: Constraints for the mechanism of super accumulation of rare earth elements. Ore Geol. Rev. 2011, 40, 122–131. [Google Scholar] [CrossRef]
- Deng, M.; Xu, C.; Song, W.L.; Tang, H.Y.; Liu, Y.; Zhang, Q.; Zhou, Y.; Feng, M.; Wei, C.W. REE mineralization in the Bayan Obo deposit, China: Evidence from mineral paragenesis. Ore Geol. Rev. 2017, 91, 100–109. [Google Scholar] [CrossRef]
- Sun, J.; Zhu, X.K.; Chen, Y.L.; Fang, N. Iron isotopic constraints on the genesis of Bayan Obo ore deposit, Inner Mongolia, China. Precambrian Res. 2013, 235, 88–106. [Google Scholar] [CrossRef]
- Zhang, W.; Chen, W.T.; Gao, J.F.; Chen, H.K.; Li, J.H. Two episodes of REE mineralization in the Qinling Orogenic Belt, Central China: In-situ U-Th-Pb dating of bastnäsite and monazite. Miner. Depos. 2019, 54, 1265–1280. [Google Scholar] [CrossRef]
- Li, S. Geochemical features and petrogenesis of Miaoya carbonatites, Hubei. Geochimica 1980, 1, 345–355, (In Chinese with English abstract). [Google Scholar]
- Zheng, X.; Liu, Y. Mechanisms of element precipitation in carbonatite-related rare-earth element deposits: Evidence from fluid inclusions in the Maoniuping deposit, Sichuan Province, southwestern China. Ore Geol. Rev. 2019, 107, 218–238. [Google Scholar] [CrossRef]
- Liu, Y.; Chakhmouradian, A.R.; Hou, Z.Q.; Song, W.L.; Kynický, J. Development of REE mineralization in the giant Maoniuping deposit (Sichuan, China): Insights from mineralogy, fluid inclusions, and trace-element geochemistry. Miner. Depos. 2018, 54, 701–718. [Google Scholar] [CrossRef]
- Li, J.K.; Yuan, Z.X.; Bai, G.; Chen, Y.C.; Wang, D.H.; Ying, L.J.; Zhang, J. Ore-forming fluid evolvement and its controlling to REE(Ag) mineralizing in the Weishan deposit, Shandong. Jmineral Pet. 2009, 29, 60–68, (In Chinese with English abstract). [Google Scholar]
- Castor, S.B. The Mountain Pass Rare-Earth Carbonatite and Associated Ultrapotassic Rocks, California. Can. Mineral. 2008, 46, 779–806. [Google Scholar] [CrossRef]
- Mariano, A.N.; Mariano, A. Rare Earth Mining and Exploration in North America. Elements 2012, 8, 369–376. [Google Scholar] [CrossRef]
- Denton, K.M.; Ponce, D.A.; Peacock, J.R.; Miller, D.M. Geophysical characterization of a Proterozoic REE terrane at Mountain Pass, eastern Mojave Desert, California, USA. Geosphere 2019, 16, 456–471. [Google Scholar] [CrossRef]
- Jones, A.P.; Wyllie, P.J. Low-temperature glass quenched from a synthetic, rare earth carbonatite; implications for the origin of the Mountain Pass Deposit, California. Econ. Geol. 1983, 78, 1721–1723. [Google Scholar] [CrossRef]
- Poletti, J.E.; Cottle, J.M.; Hagen-Peter, G.A.; Lackey, J.S. Petrochronological Constraints on the Origin of the Mountain Pass Ultrapotassic and Carbonatite Intrusive Suite, California. J. Petrol. 2016, 57, 1555–1598. [Google Scholar] [CrossRef]
- Nascimento, M.; Lemos, F.; Guimarães, R.; Sousa, C.; Soares, P. Modeling of REE and Fe Extraction from a Concentrate from Araxá (Brazil). Minerals 2019, 9, 451. [Google Scholar] [CrossRef]
- Li, Y.K.; Chen, R.Y.; Ke, C.H.; Chen, J.; Hao, M.Z.; Li, R.P. The strategic and critical minerals associated with alkaline and alkaline-carbonatite complexes Brazil. Acta Geol. Sin. 2019, 93, 1422–1443, (In Chinese with English abstract). [Google Scholar]
- Neumann, R.; Medeiros, E.B. Comprehensive mineralogical and technological characterisation of the Araxá (SE Brazil) complex REE (Nb-P) ore, and the fate of its processing. Int. J. Miner. Process. 2015, 144, 1–10. [Google Scholar] [CrossRef]
- Buyse, F.; Dewaele, S.; Decrée, S.; Mees, F. Mineralogical and geochemical study of the rare earth element mineralization at Gakara (Burundi). Ore Geol. Rev. 2020, 124, 103659. [Google Scholar] [CrossRef]
- Ntiharirizwa, S.; Boulvais, P.; Poujol, M.; Branquet, Y.; Morelli, C.; Ntungwanayo, J.; Midende, G. Geology and U-Th-Pb Dating of the Gakara REE Deposit, Burundi. Minerals 2018, 8, 394. [Google Scholar] [CrossRef]
- Lehmann, B.; Nakai, S.i.; Höhndorf, A.; Brinckmann, J.; Dulski, P.; Hein, U.F.; Masuda, A. REE mineralization at Gakara, Burundi: Evidence for anomalous upper mantle in the western Rift Valley. Geochim. Cosmochim. Acta 1994, 58, 985–992. [Google Scholar] [CrossRef]
- Secher, K.; Larsen, L.M. Geology and mineralogy of the Sarfartôq carbonatite complex, southern West Greenland. Lithos 1980, 13, 199–212. [Google Scholar] [CrossRef]
- Goodenough, K.M.; Schilling, J.; Jonsson, E.; Kalvig, P.; Charles, N.; Tuduri, J.; Deady, E.A.; Sadeghi, M.; Schiellerup, H.; Müller, A.; et al. Europe’s rare earth element resource potential: An overview of REE metallogenetic provinces and their geodynamic setting. Ore Geol. Rev. 2016, 72, 838–856. [Google Scholar] [CrossRef]
- Bedini, E.; Rasmussen, T.M. Use of airborne hyperspectral and gamma-ray spectroscopy data for mineral exploration at the Sarfartoq carbonatite complex, southern West Greenland. Geosci. J. 2018, 22, 641–651. [Google Scholar] [CrossRef]
- Ye, H.M.; Zhang, X. Advances on the carbonatite research in recent years. Resour. Surv. Environ. 2015, 36, 21–27, (In Chinese with English abstract). [Google Scholar]
- Wallace, M.E.; Green, D.H. An experimental determination of primary carbonatite magma composition. Nature 1988, 335, 343–346. [Google Scholar] [CrossRef]
- Wyllie, P.J.; Lee, W.J. Model System Controls on Conditions for Formation of Magnesiocarbonatite and Calciocarbonatite Magma from the Mantle. J. Petrol. 1998, 39, 1885–1893. [Google Scholar] [CrossRef]
- Hamilton, D.L.; Freestone, I.C.; Dawson, J.B.; Donaldson, C.H. Origin of carbonatites by liquid immiscibility. Nature 1979, 279, 52–54. [Google Scholar] [CrossRef]
- Harmer, R. The Case for Primary, Mantle-derived Carbonatite Magma. J. Petrol. 1998, 39, 1895–1903. [Google Scholar] [CrossRef]
- Lee, W.J.; Wyllie, P.J. Processes of Crustal Carbonatite Formation by Liquid Immiscibility and Differentiation, Elucidated by Model Systems. J. Petrol. 1998, 39, 2005–2013. [Google Scholar] [CrossRef]
- Veksler, I.; Lentz, D. Parental magma of plutonic carbonatites, carbonate-silicate immiscibility and decarbonation reactions: Evidence from melt and fluid inclusions. Melt Incl. Plutonic Rocks 2006, 36, 123–150. [Google Scholar]
- Manthilake, M.A.G.M.; Sawada, Y.; Sakai, S. Genesis and evolution of Eppawala carbonatites, Sri Lanka. J. Asian Earth Sci. 2008, 32, 66–75. [Google Scholar] [CrossRef]
- Hulett, S.R.W.; Simonetti, A.; Rasbury, E.T.; Hemming, N.G. Recycling of subducted crustal components into carbonatite melts revealed by boron isotopes. Nat. Geosci. 2016, 9, 904–908. [Google Scholar] [CrossRef]
- Nelson, D.R.; Chivas, A.R.; Chappell, B.W.; McCulloch, M.T. Geochemical and isotopic systematics in carbonatites and implications for the evolution of ocean-island sources. Geochim. Cosmochim. Acta 1988, 52, 1–17. [Google Scholar] [CrossRef]
- Zhu, X.K.; Sun, J.; Pan, C.X. Sm-Nd isotopic constraints on rare-earth mineralization in the Bayan Obo ore deposit, Inner Mongolia, China. Ore Geol. Rev. 2015, 64, 543–553. [Google Scholar] [CrossRef]
- Ionov, D. Trace Element Composition of Mantle-derived Carbonates and Coexisting Phasesin Peridotite Xenoliths from Alkali Basalts. J. Petrol. 1998, 39, 1931–1941. [Google Scholar] [CrossRef]
- Veksler, I.V.; Petibon, C.; Jenner, G.A.; Dorfman, A.M.; Dingwell, D.B. Trace Element Partitioning in Immiscible Silicate-Carbonate Liquid Systems: An Initial Experimental Study Using a Centrifuge Autoclave. J. Petrol. 1998, 39, 2095–2104. [Google Scholar] [CrossRef]
- Veksler, I.V.; Dorfman, A.M.; Dulski, P.; Kamenetsky, V.S.; Danyushevsky, L.V.; Jeffries, T.; Dingwell, D.B. Partitioning of elements between silicate melt and immiscible fluoride, chloride, carbonate, phosphate and sulfate melts, with implications to the origin of natrocarbonatite. Geochim. Cosmochim. Acta 2012, 79, 20–40. [Google Scholar] [CrossRef]
- Foley, S.F.; Yaxley, G.M.; Rosenthal, A.; Buhre, S.; Kiseeva, E.S.; Rapp, R.P.; Jacob, D.E. The composition of near-solidus melts of peridotite in the presence of CO2 and H2O between 40 and 60 kbar. Lithos 2009, 112, 274–283. [Google Scholar] [CrossRef]
- Ionov, D.; Harmer, R.E. Trace element distribution in calcite-dolomite carbonatites from Spitskop: Inferences for differentiation of carbonatite magma and the origin of carbonates in mantle xenoliths. Earth Planet. Sci. Lett. 2002, 198, 495–510. [Google Scholar] [CrossRef]
- Banerjee, A.; Chakrabarti, R. A geochemical and Nd, Sr and stable Ca isotopic study of carbonatites and associated silicate rocks from the ~65 Ma old Ambadongar carbonatite complex and the Phenai Mata igneous complex, Gujarat, India: Implications for crustal contamination, carbonate recycling, hydrothermal alteration and source-mantle mineralogy. Lithos 2019, 326–327, 572–585. [Google Scholar]
- Hou, Z.Q.; Tian, S.H.; Yuan, Z.X.; Xie, Y.L.; Yin, S.P.; Yi, L.S.; Fei, H.C.; Yang, Z.M. The Himalayan collision zone carbonatites in western Sichuan, SW China: Petrogenesis, mantle source and tectonic implication. Earth Planet. Sci. Lett. 2006, 244, 234–250. [Google Scholar] [CrossRef]
- Kato, Y.; Fujinaga, K.; Nakamura, K.; Takaya, Y.; Kitamura, K.; Ohta, J.; Toda, R.; Nakashima, T.; Iwamori, H. Deep-sea mud in the Pacific Ocean as a potential resource for rare-earth elements. Nat. Geosci. 2011, 4, 535–539. [Google Scholar] [CrossRef]
- Mimura, K.; Nakamura, K.; Yasukawa, K.; Machida, S.; Ohta, J.; Fujinaga, K.; Kato, Y. Significant impacts of pelagic clay on average chemical composition of subducting sediments: New insights from discovery of extremely rare-earth elements and yttrium-rich mud at Ocean Drilling Program Site 1149 in the western North Pacific Ocean. J. Asian Earth Sci. 2019, 186, 104059. [Google Scholar] [CrossRef]
- Yasukawa, K.; Liu, H.; Fujinaga, K.; Machida, S.; Haraguchi, S.; Ishii, T.; Nakamura, K.; Kato, Y. Geochemistry and mineralogy of REY-rich mud in the eastern Indian Ocean. J. Asian Earth Sci. 2014, 93, 25–36. [Google Scholar] [CrossRef]
- Ling, M.X.; Liu, Y.L.; Williams, I.S.; Teng, F.Z.; Yang, X.Y.; Ding, X.; Wei, G.J.; Xie, L.H.; Deng, W.F.; Sun, W.D. Formation of the world’s largest REE deposit through protracted fluxing of carbonatite by subduction-derived fluids. Sci. Rep. 2013, 3, 1776. [Google Scholar] [CrossRef]
- Ni, P.; Zhou, J.; Chi, Z.; Pan, J.-Y.; Li, S.-N.; Ding, J.-Y.; Han, L. Carbonatite dyke and related REE mineralization in the Bayan Obo REE ore field, North China: Evidence from geochemistry, C O isotopes and Rb Sr dating. J. Geochem. Explor. 2020, 215, 106560. [Google Scholar] [CrossRef]
- Wu, Y.B.; Zheng, Y.F. Tectonic evolution of a composite collision orogen: An overview on the Qinling-Tongbai-Hong’an-Dabie-Sulu orogenic belt in central China. Gondwana Res. 2013, 23, 1402–1428. [Google Scholar] [CrossRef]
- Xu, C.; Kynicky, J.; Chakhmouradian, A.R.; Li, X.H.; Song, W.L. A case example of the importance of multi-analytical approach in deciphering carbonatite petrogenesis in South Qinling orogen: Miaoya rare-metal deposit, central China. Lithos 2015, 227, 107–121. [Google Scholar] [CrossRef]
- Fan, H.R.; Xie, Y.H.; Wang, K.Y.; Yang, X.M. Carbonatitic Fluids and REE Mineralization. Earth Science Frontiers. 2001, 8, 289–295, (In Chinese with English abstract). [Google Scholar]
- Zhou, J.; Ni, P.; Ding, J.Y.; Zhu, X.T. Fluid inclusion study related to cabonatitic magmatic process. Geol. J. China Univ. 2003, 9, 293–301, (In Chinese with English abstract). [Google Scholar]
- Xie, Y.L.; Xu, J.H.; Chen, W.; He, J.P.; Hou, Z.Q.; Xu, W.Y. Characteristics of carbonatite fluid in the Maoniuping REE deposit, Mianning, China. In Mineral Deposit Research: Meeting the Global Challenge; Springer: Berlin/Heidelberg, Germany, 2005. [Google Scholar]
- Xie, Y.L.; Yin, S.P.; Xu, J.H.; Chen, W.; Yi, L.S. A Study on the fluids in carbonatite of the Mianning-Dechang REE metallogenic belt. Bull. Mineral. Petrol. Geochem. 2006, 25, 66–74, (In Chinese with English abstract). [Google Scholar]
- Shu, X.C.; Liu, Y. Fluid inclusion constraints on the hydrothermal evolution of the Dalucao Carbonatite-related REE deposit, Sichuan Province, China. Ore Geol. Rev. 2019, 107, 41–57. [Google Scholar] [CrossRef]
- Xie, Y.L.; Hou, Z.Q.; Yin, S.P.; Dominy, S.C.; Xu, J.H.; Tian, S.H.; Xu, W.Y. Continuous carbonatitic melt–fluid evolution of a REE mineralization system: Evidence from inclusions in the Maoniuping REE Deposit, Western Sichuan, China. Ore Geol. Rev. 2009, 36, 90–105. [Google Scholar] [CrossRef]
- Ting, W.P.; Burke, E.A.J.; Rankin, A.H.; Woolley, A.R. Characterisation and petrogenetic significance of CO2, H2O and CH4 fluid inclusions in apatite from the Sukulu carbonatite, Uganda. Eur. J. Mineral. 1994, 6, 787–803. [Google Scholar] [CrossRef]
- Kong, L.R.; Zhang, G.R. Ore Types and Characteristics of Melt (Fluid) Inclusions on the Maoniuping REE Deposit Mianning, Sichuan, China. Appl. Mech. Mater. 2014, 675–677, 1308–1311. [Google Scholar] [CrossRef]
- She, H.D.; Fan, H.R.; Hu, F.F.; Yang, K.F.; Yang, Z.F.; Wang, Q.W. Migration and precipitation of rare earth elements in the hydrothermal fluids. Acta Petrol. Sin. 2018, 34, 3567–3581, (In Chinese with English abstract). [Google Scholar]
- Migdisov, A.; Williams-Jones, A.E.; Brugger, J.; Caporuscio, F.A. Hydrothermal transport, deposition, and fractionation of the REE: Experimental data and thermodynamic calculations. Chem. Geol. 2016, 439, 13–42. [Google Scholar] [CrossRef]
- Migdisov, A.A.; Williams-Jones, A.E. An experimental study of the solubility and speciation of neodymium (III) fluoride in F-bearing aqueous solutions. Geochim. Cosmochim. Acta 2007, 71, 3056–3069. [Google Scholar] [CrossRef]
- Migdisov, A.A.; Williams-Jones, A.E. A spectrophotometric study of Nd(III), Sm(III) and Er(III) complexation in sulfate-bearing solutions at elevated temperatures. Geochim. Cosmochim. Acta 2008, 72, 5291–5303. [Google Scholar] [CrossRef]
- Migdisov, A.A.; Williams-Jones, A.E.; Wagner, T. An experimental study of the solubility and speciation of the Rare Earth Elements (III) in fluoride- and chloride-bearing aqueous solutions at temperatures up to 300°C. Geochim. Cosmochim. Acta 2009, 73, 7087–7109. [Google Scholar] [CrossRef]
- Debruyne, D.; Hulsbosch, N.; Muchez, P. Unraveling rare earth element signatures in hydrothermal carbonate minerals using a source–sink system. Ore Geol. Rev. 2016, 72, 232–252. [Google Scholar] [CrossRef]
- Louvel, M.; Bordage, A.; Testemale, D.; Zhou, L.; Mavrogenes, J. Hydrothermal controls on the genesis of REE deposits: Insights from an in situ XAS study of Yb solubility and speciation in high temperature fluids (T < 400 °C). Chem. Geol. 2015, 417, 228–237. [Google Scholar]
- Liu, Y.; Chen, C.; Shu, X.C.; Guo, D.X.; Li, Z.J.; Zhao, H.X.; Jia, Y.H. The formation model of the carbonatite-syenite complex REE deposits in the east of Tibetan Plateau: A case study of Dalucao REE deposit. Acta Petrol. Sin. 2017, 33, 1978–2000, (In Chinese with English abstract). [Google Scholar]
- Verplanck, P.L. The Role of Fluids in the Formation of Rare Earth Element Deposits. Procedia Earth Planet. Sci. 2017, 17, 758–761. [Google Scholar] [CrossRef]
- Chikanda, F.; Otake, T.; Ohtomo, Y.; Ito, A.; Yokoyama, T.D.; Sato, T. Magmatic-Hydrothermal Processes Associated with Rare Earth Element Enrichment in the Kangankunde Carbonatite Complex, Malawi. Minerals 2019, 9, 442. [Google Scholar] [CrossRef]
- Qin, C.J.; Qiu, Y.Z.; Wen, H.J.; Xu, C. Genesis of Maoniuping REE deposit, Sichuan: Evidence from inclusions. Acta Petrol. Sin. 2008, 24, 2155–2162, (In Chinese with English abstract). [Google Scholar]
- Anenburg, M.; Burnham, A.D.; Mavrogenes, J.A. REE Redistribution Textures in Altered Fluorapatite: Symplectites, Veins, and Phosphate-Silicate-Carbonate Assemblages from the Nolans Bore P-REE-Th Deposit, Northern Territory, Australia. Can. Mineral. 2018, 56, 331–354. [Google Scholar] [CrossRef]
- Anenburg, M.; Mavrogenes, J.A.; Bennett, V.C. The Fluorapatite P-REE-Th Vein Deposit at Nolans Bore: Genesis by Carbonatite Metasomatism. J. Petrol. 2020, 61, egaa003. [Google Scholar] [CrossRef]
- Anenburg, M.; Mavrogenes, J.A. Carbonatitic versus hydrothermal origin for fluorapatite REE-Th deposits: Experimental study of REE transport and crustal “antiskarn” metasomatism. Am. J. Sci. 2018, 318, 335–366. [Google Scholar] [CrossRef]

| Country | Deposit | Deposit-Type |
|---|---|---|
| China | Bayan Obo (REE-Nb-Fe) | Igneous carbonatite |
| China | Maoniuping (REE) | Carbonatite |
| China | HREE-enriched deposits in southern China | Weathered crust elution |
| Brazil | Araxá Catalão (REE) | Weathered Carbonatite |
| Brazil | Mrro do Ferro (Th-REE) | Carbonatite |
| Australia | Mount Weld (REE) | Weathered Carbonatite |
| United States | Mountain Pass (REE) | Carbonatite |
| Russia | Tomtor (REE) | Weathered Carbonatite |
| Russia | Lovozero (REE-Nb) | Alkaline igneous rock |
| India | Amba Dongar (REE) | Carbonatite |
| Vietnam | Mau Sai (REE) | Carbonatite |
| Burundi | Gakara (REE) | Carbonatite |
| Malawi | Kangankunde (REE) | Carbonatite |
| South Africa | Palabora (REE) | Carbonatite |
| South Africa | Steenkampskraal (REE-Th-Cu) | Alkaline igneous rock |
| Turkey | Aksu Diamas (REE) | Placer |
| Greenland | Tanbreez (REE) | Alkaline igneous rock |
| Sweden | Norra Kärr (REE) | Alkaline igneous rock |
| Classification | Major Elements |
|---|---|
| Calciocarbonatite | CaO/(CaO + MgO + FeO + Fe2O3 + MnO) > 0.8 |
| Magnesiocarbonatite | CaO/(CaO + MgO + FeO + Fe2O3 + MnO) < 0.8 and MgO > (FeO + Fe2O3 + MnO) |
| Ferrocarbonatite | CaO/(CaO + MgO + FeO + Fe2O3 + MnO) < 0.8 and MgO<(FeO + Fe2O3 + MnO) |
| Natrocarbonatite | High content of Na, K, Ca |
| Silicocarbonatite | SiO2 > 20% of the whole rock |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Z.-Y.; Fan, H.-R.; Zhou, L.; Yang, K.-F.; She, H.-D. Carbonatite-Related REE Deposits: An Overview. Minerals 2020, 10, 965. https://doi.org/10.3390/min10110965
Wang Z-Y, Fan H-R, Zhou L, Yang K-F, She H-D. Carbonatite-Related REE Deposits: An Overview. Minerals. 2020; 10(11):965. https://doi.org/10.3390/min10110965
Chicago/Turabian StyleWang, Zhen-Yu, Hong-Rui Fan, Lingli Zhou, Kui-Feng Yang, and Hai-Dong She. 2020. "Carbonatite-Related REE Deposits: An Overview" Minerals 10, no. 11: 965. https://doi.org/10.3390/min10110965
APA StyleWang, Z.-Y., Fan, H.-R., Zhou, L., Yang, K.-F., & She, H.-D. (2020). Carbonatite-Related REE Deposits: An Overview. Minerals, 10(11), 965. https://doi.org/10.3390/min10110965







