Mineralogy and Geochemistry of Late Permian Coals within the Tongzi Coalfield in Guizhou Province, Southwest China
Abstract
1. Introduction
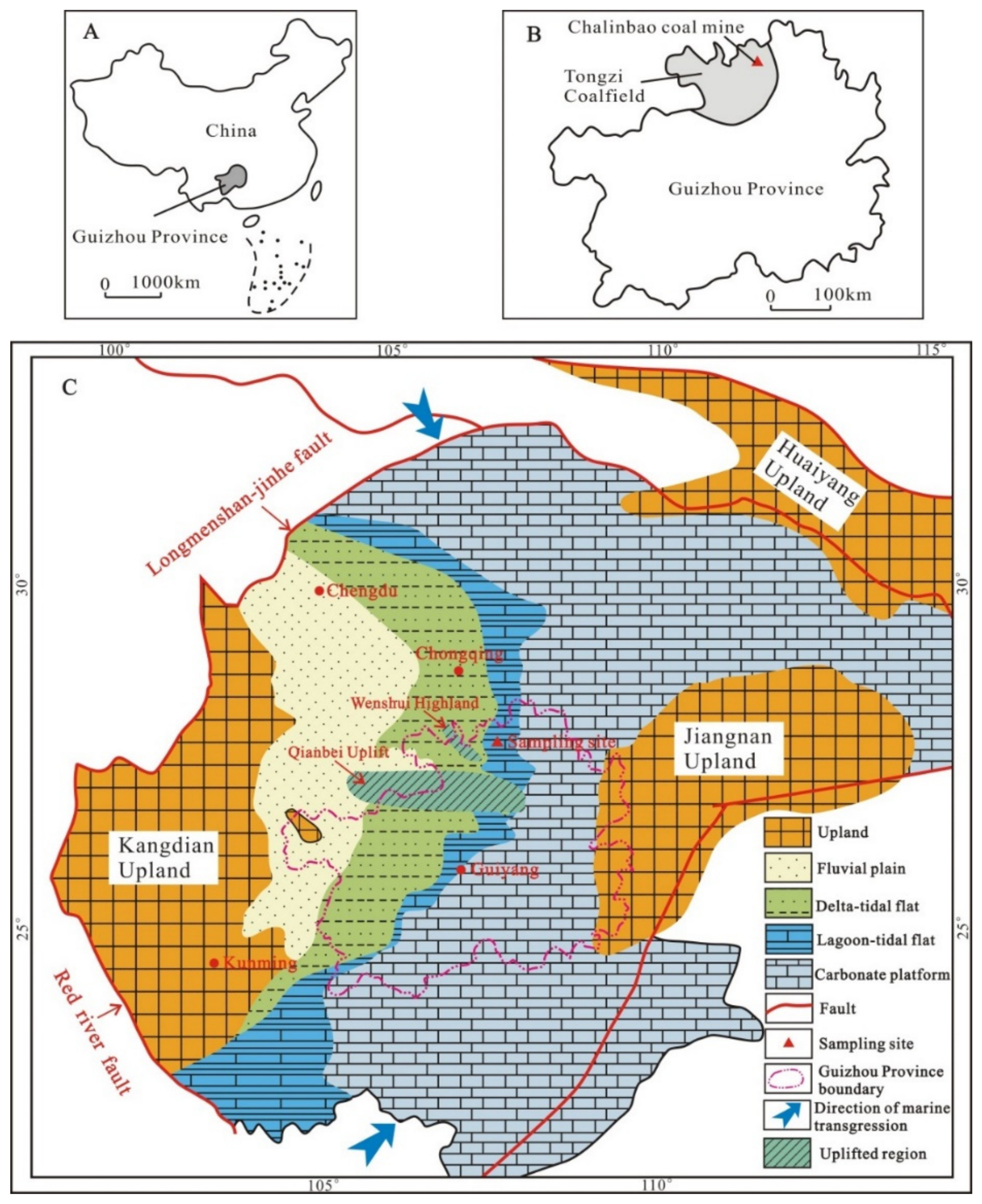
2. Geological Setting
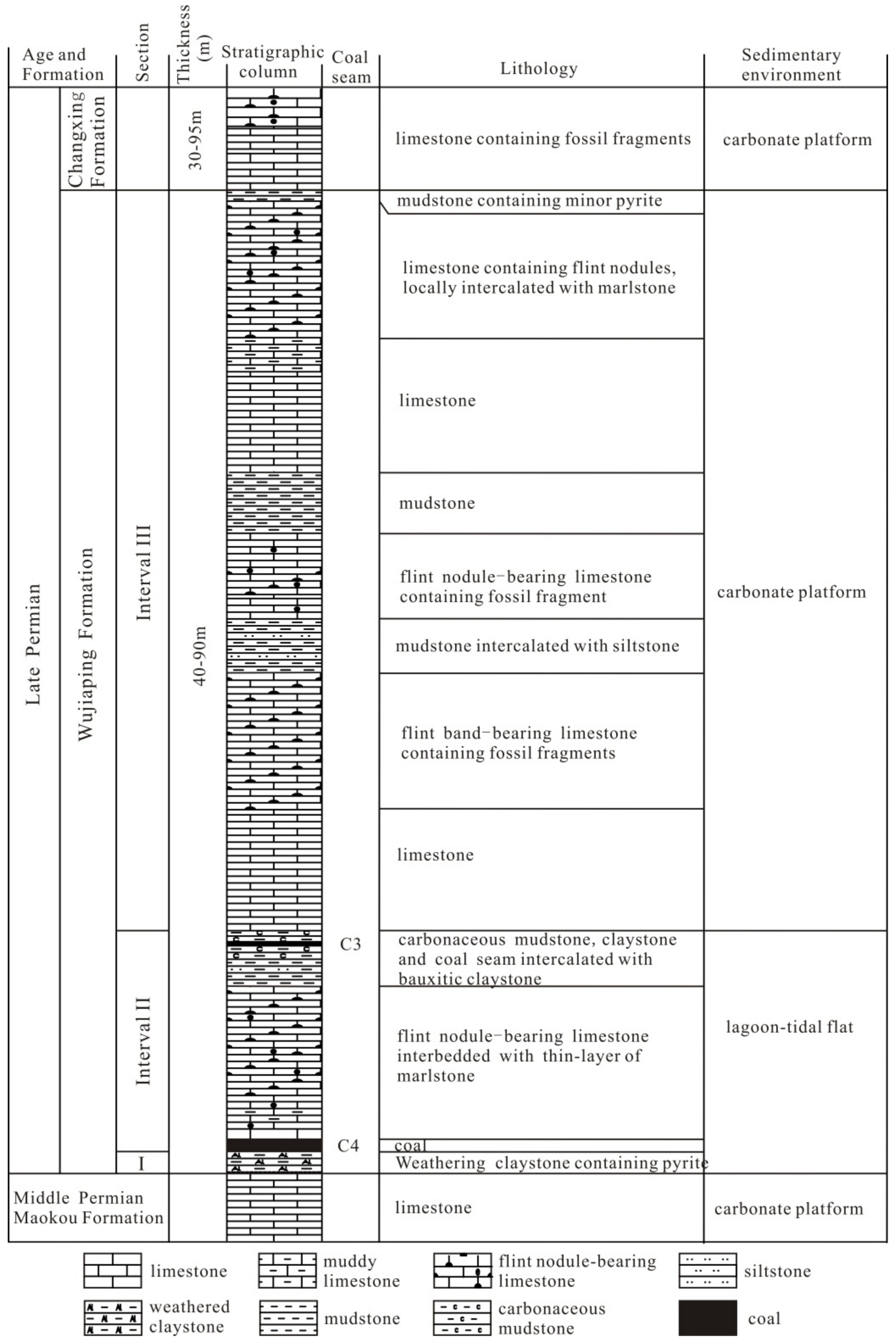
2.1. Coal-Bearing Strata
2.2. Sediment-Source Region
3. Methodology
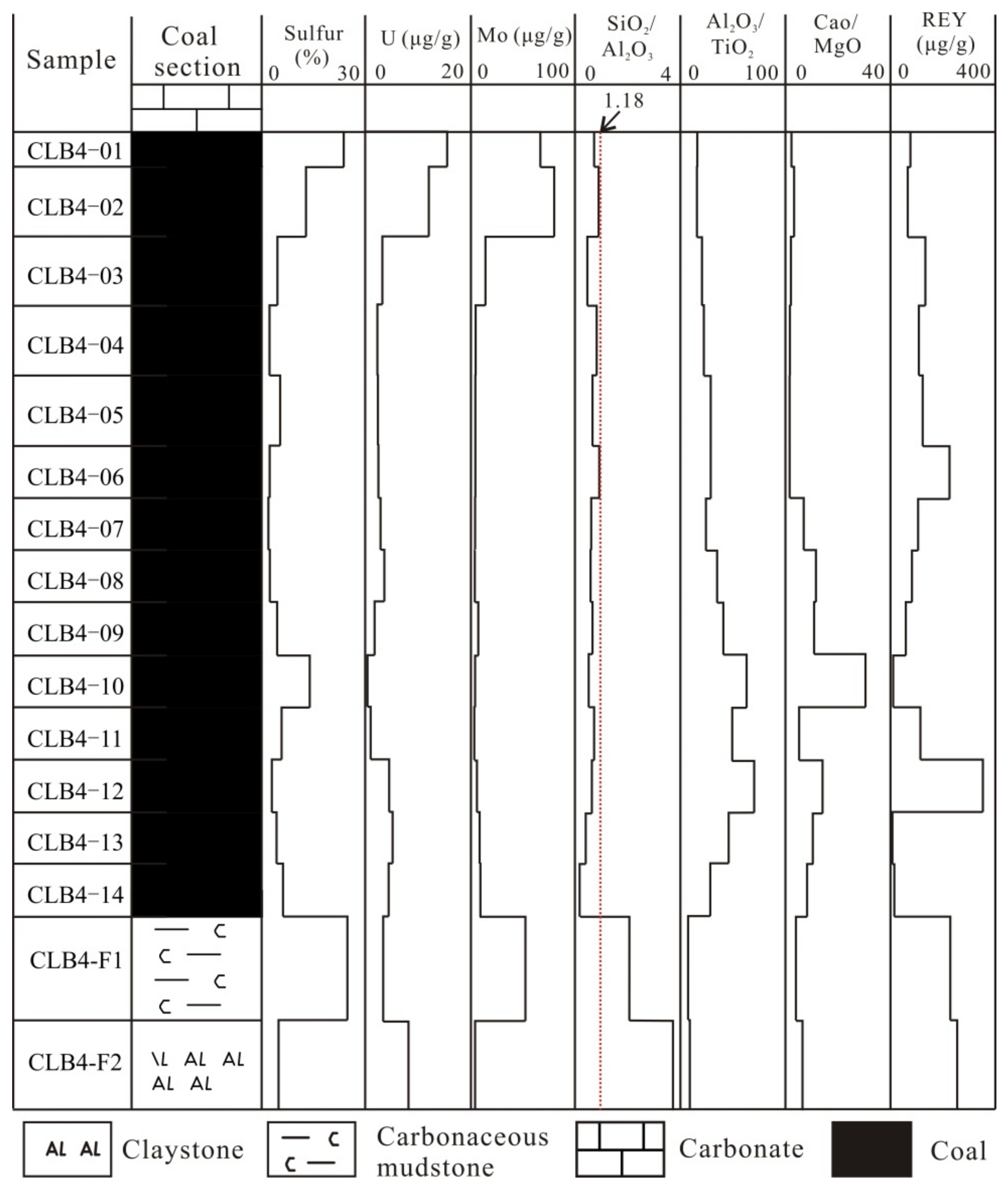
4. Results
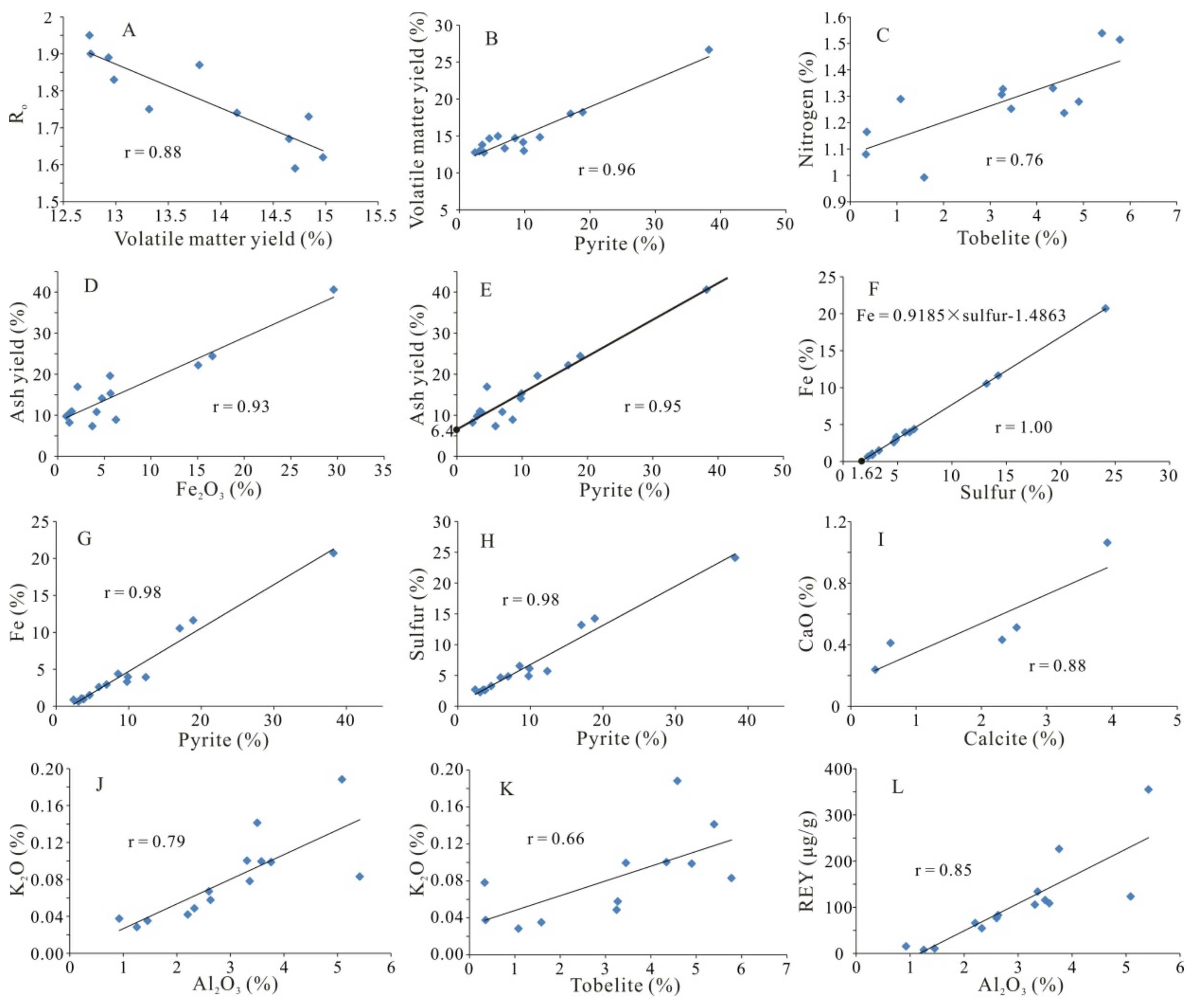
4.1. Coal Chemistry and Coal Rank
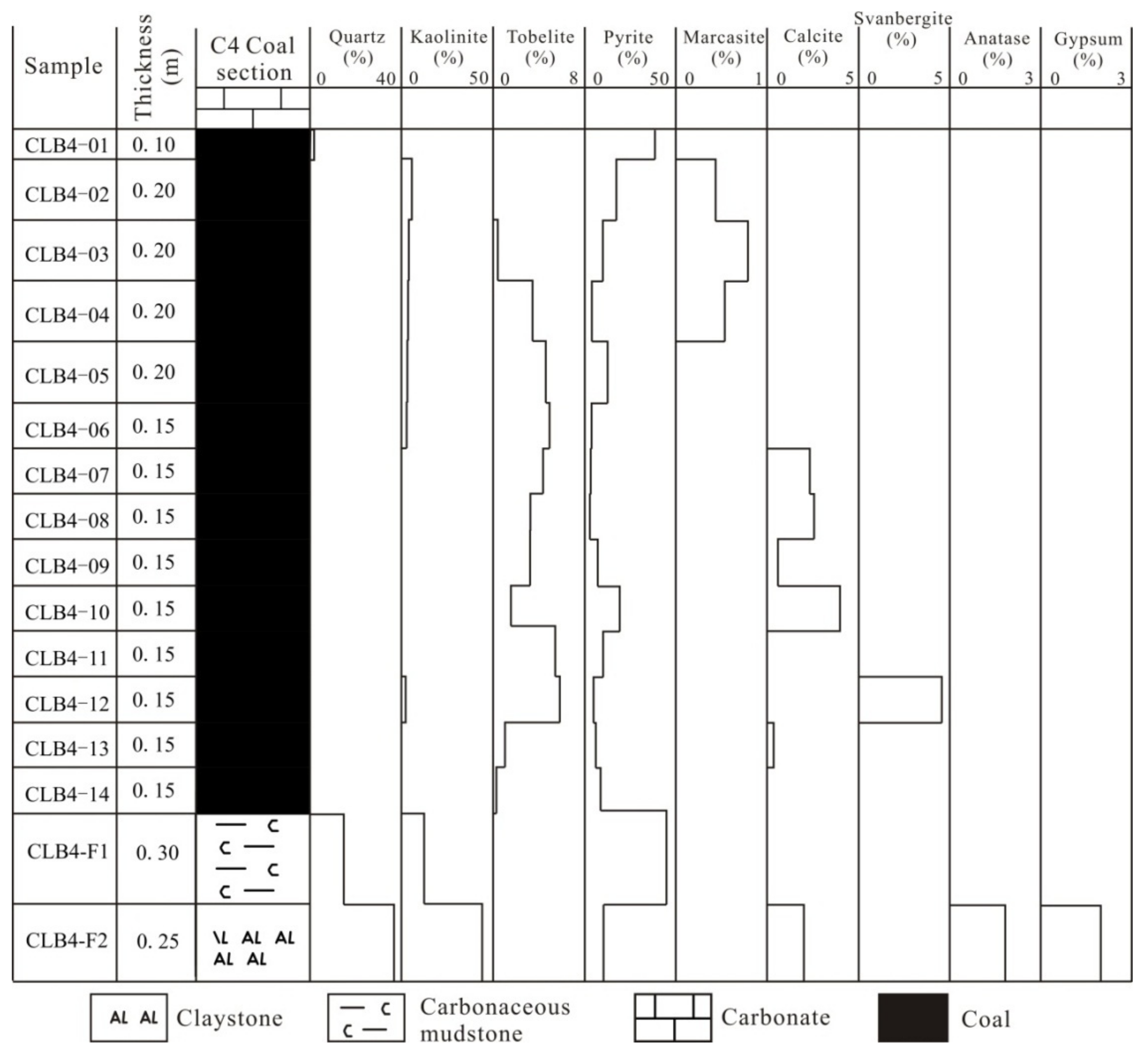
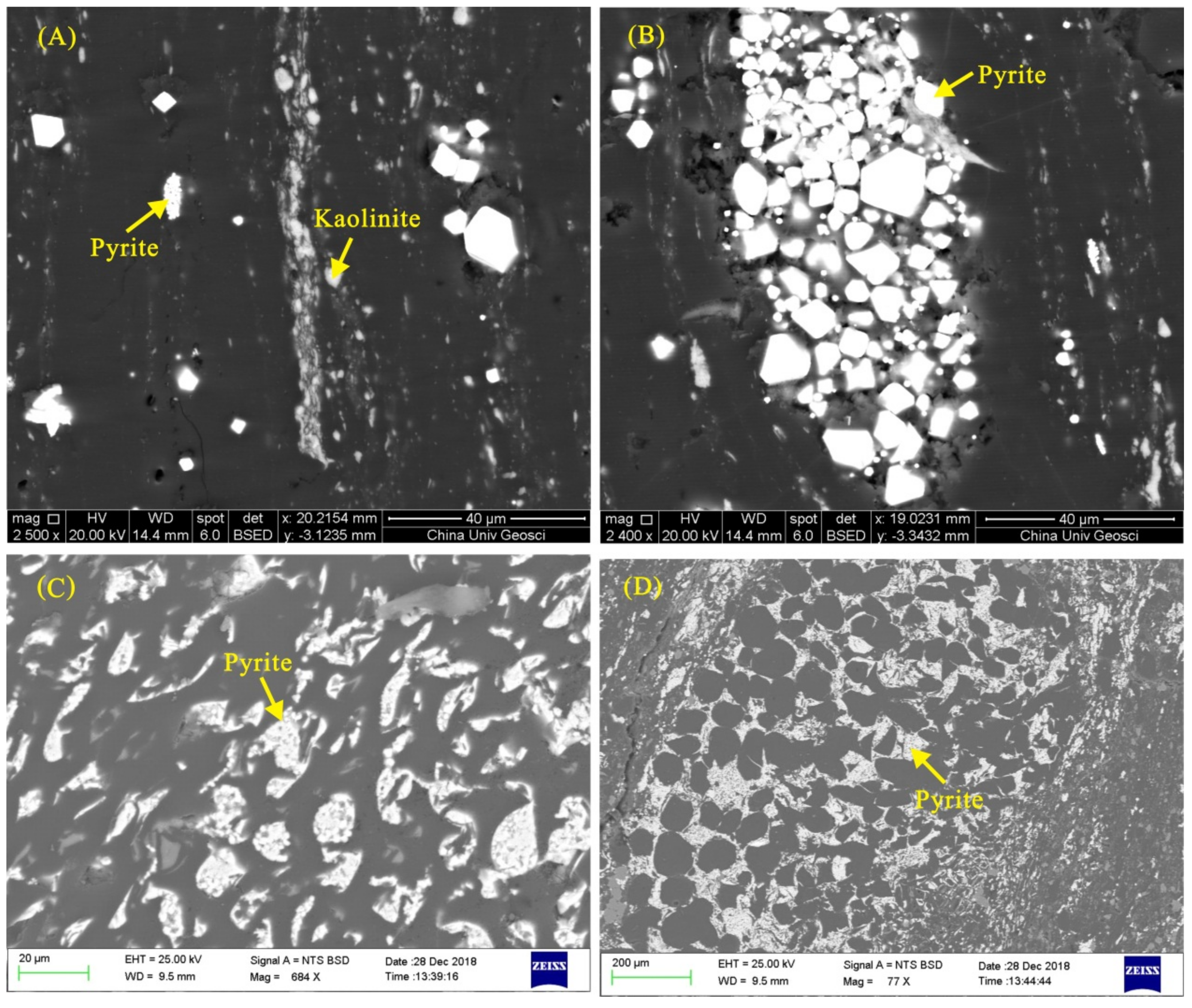
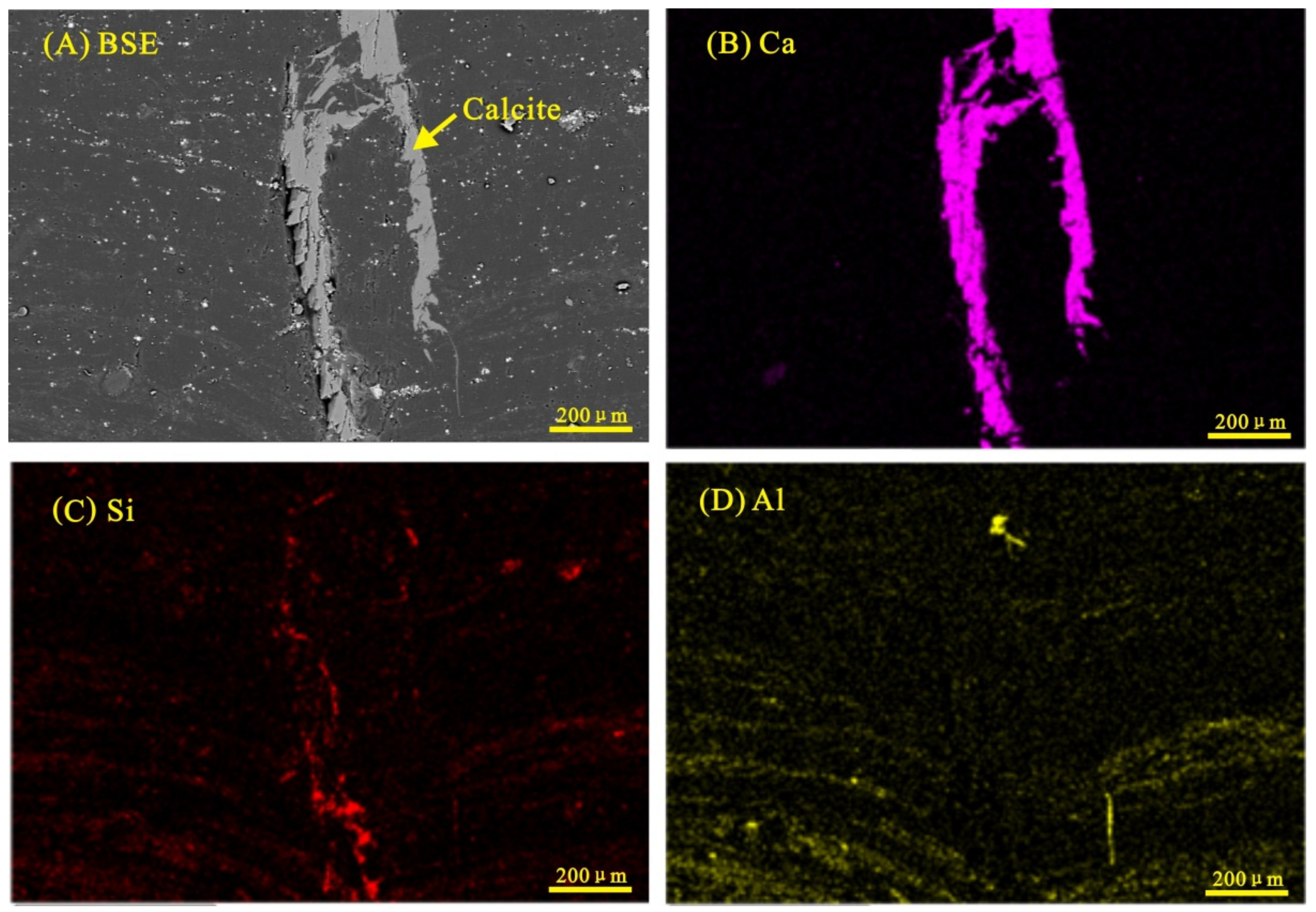
4.2. Minerals in the Coal and Floor Samples
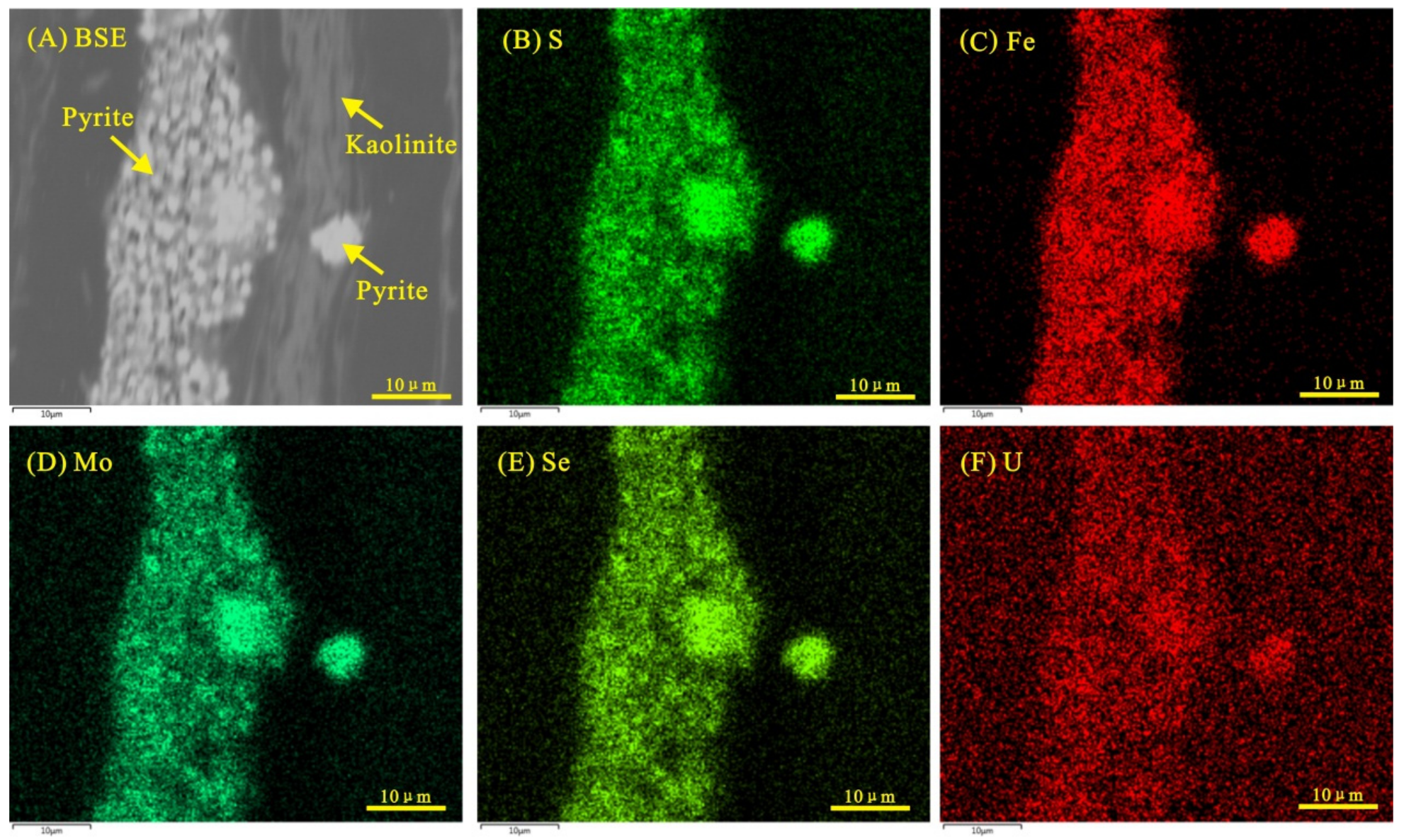
4.2.1. Pyrite
4.2.2. Kaolinite
4.2.3. Tobelite
4.2.4. Quartz
4.2.5. Sulfate Minerals
4.2.6. Calcite
4.2.7. Al-Oxyhydroxide Minerals
4.3. Geochemistry
4.3.1. Major-Element Oxides
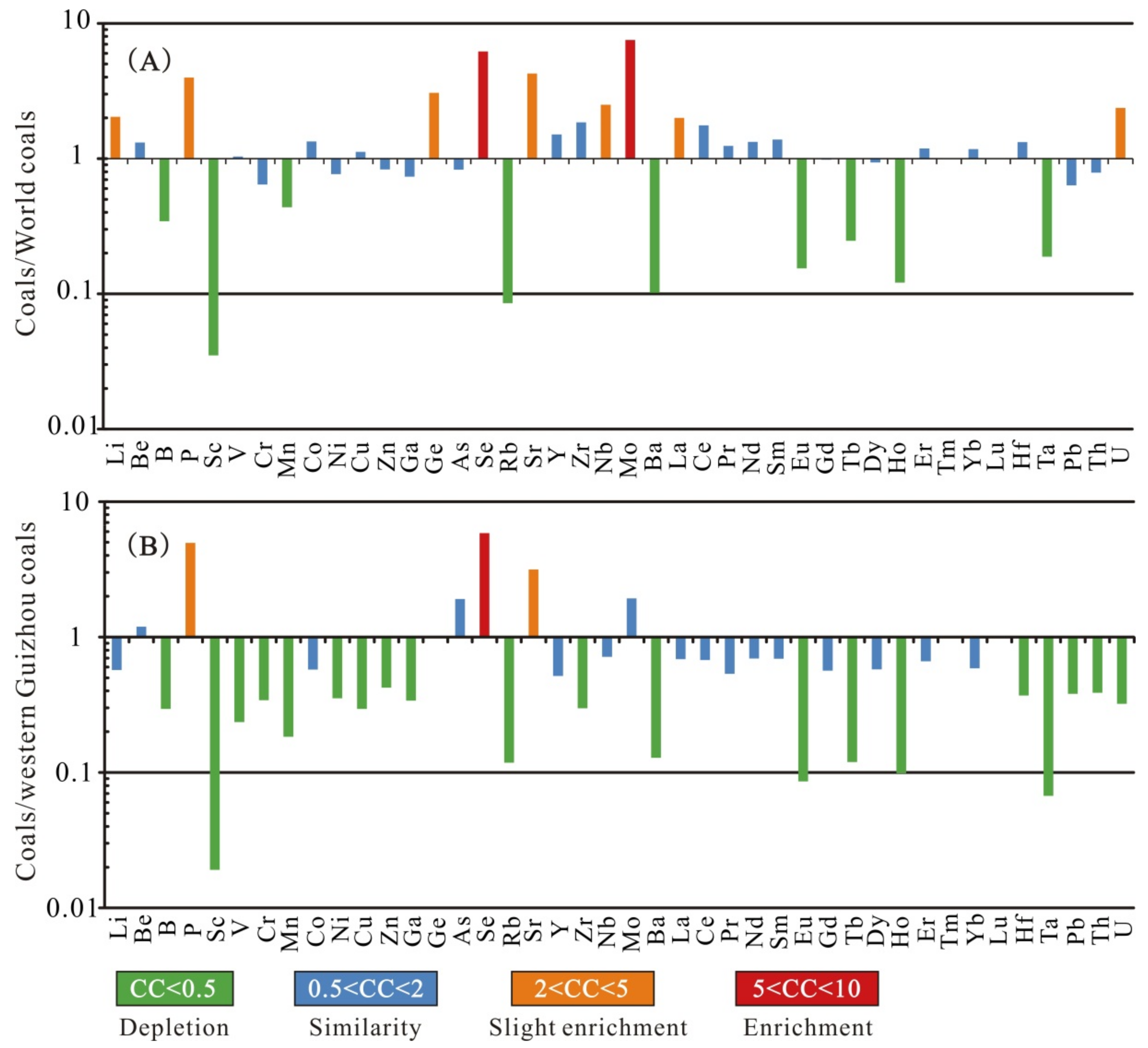
4.3.2. Trace Elements
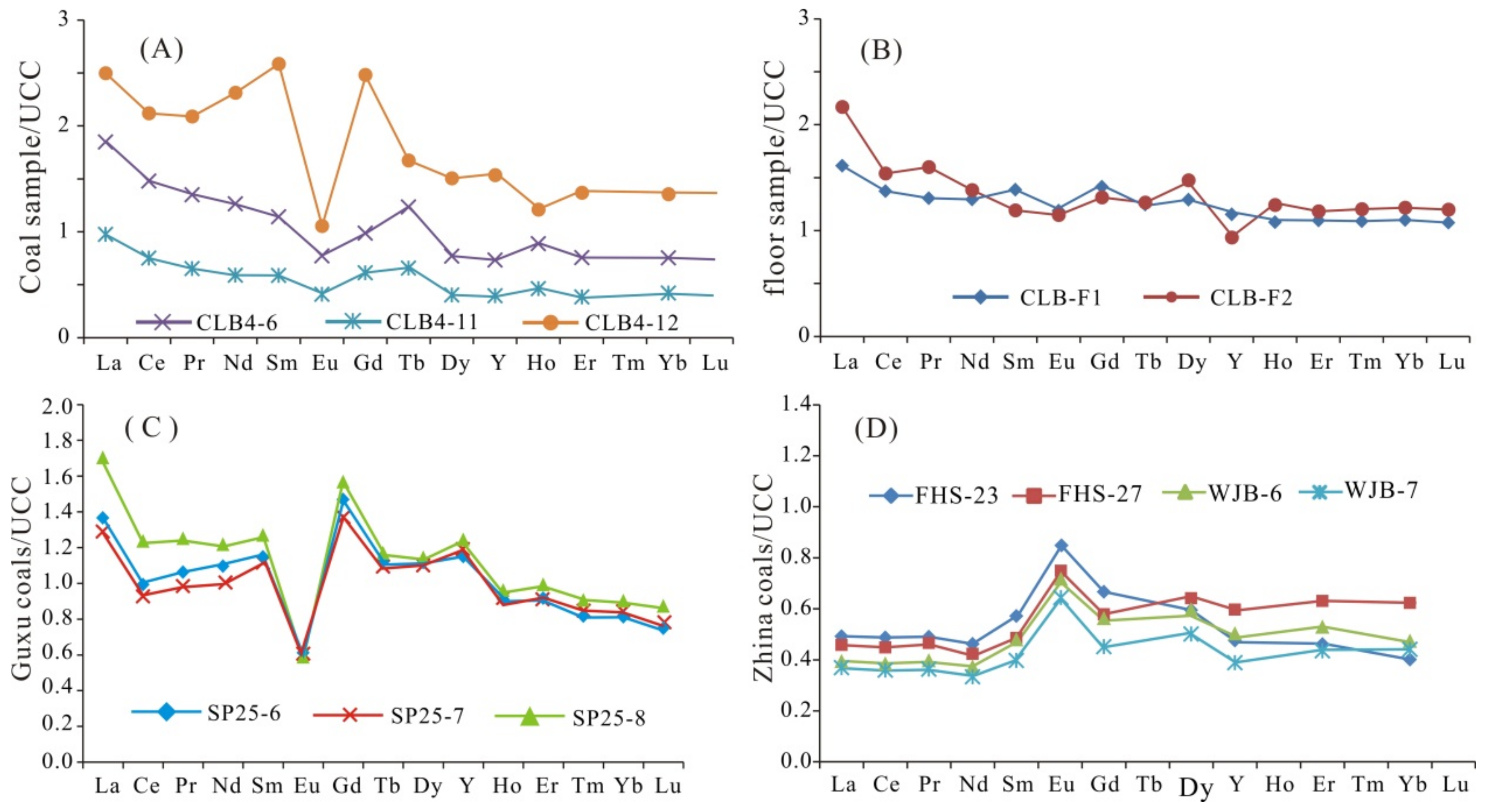
4.3.3. Rare Earth Elements and Yttrium (REY)
5. Discussion
5.1. Marine Influence
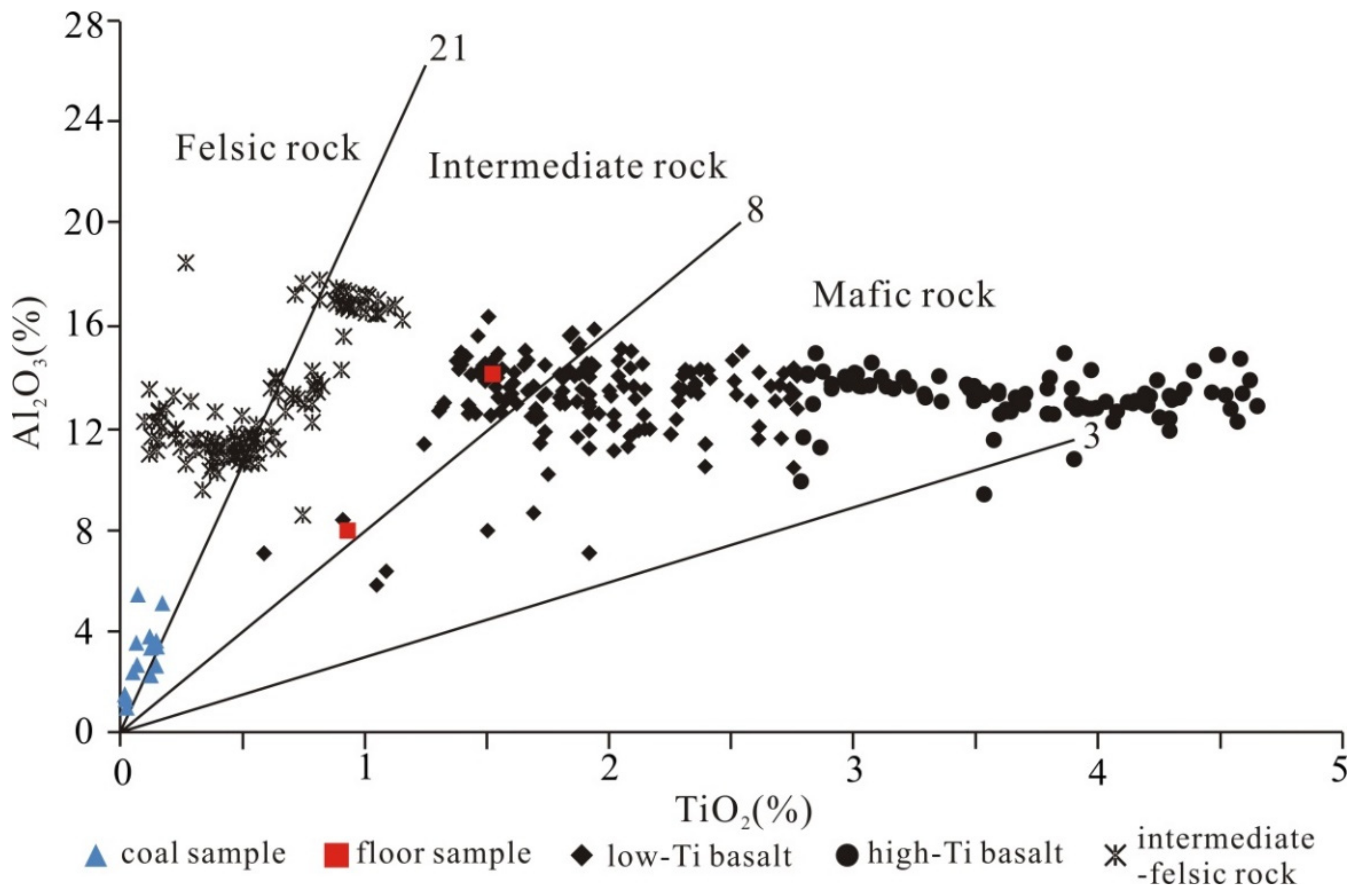
5.2. Sediment-Source Region
5.3. Origins of Minerals in the Coal and Floor Samples
5.3.1. Pyrite
5.3.2. Tobelite
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dai, S.; Ren, D.; Chou, C.-L.; Finkelman, R.B.; Seredin, V.V.; Zhou, Y. Geochemistry of trace elements in Chinese coals: A review of abundances, genetic types, impacts on human health, and industrial utilization. Int. J. Coal Geol. 2012, 94, 3–21. [Google Scholar] [CrossRef]
- Finkelman, R.B.; Dai, S.; French, D. The importance of minerals in coal as the hosts of chemical elements: A review. Int. J. Coal Geol. 2019, 212. [Google Scholar] [CrossRef]
- Ward, C.R. Analysis, origin and significance of mineral matter in coal: An updated review. Int. J. Coal Geol. 2016, 165, 1–27. [Google Scholar] [CrossRef]
- Spiro, B.F.; Liu, J.; Dai, S.; Zeng, R.; Large, D.; French, D. Marine derived 87Sr/86Sr in coal, a new key to geochronology and palaeoenvironment: Elucidation of the India-Eurasia and China-Indochina collisions in Yunnan, China. Int. J. Coal Geol. 2019, 215, 103304. [Google Scholar] [CrossRef]
- Dai, S.; Chekryzhov, I.Y.; Seredin, V.V.; Nechaev, V.P.; Graham, I.T.; Hower, J.C.; Ward, C.R.; Ren, D.; Wang, X. Metalliferous coal deposits in East Asia (Primorye of Russia and South China): A review of geodynamic controls and styles of mineralization. Gondwana Res. 2016, 29, 60–82. [Google Scholar] [CrossRef]
- China Coal Geology Bureau. Sedimentary Environments and Coal Accumulation of Late Permian Coal Formation in Western Guizhou, Southern Sichuan, and Eastern Yunnan, China; Chongqing University Press: Chongqing, China, 1996; pp. 156–216, (In Chinese with English Abstract). [Google Scholar]
- Shao, L.; Zhang, C.; Yan, Z.; Dong, D.; Gao, C.; Li, Y.; Xu, X.; Liang, W.; Yi, T.; Xu, X.; et al. Sequence-palaeogeography and coal accumulation of the Late Permian in South China. J. Palaeogeogr. 2016, 18, 905–919, (In Chinese with English Abstract). [Google Scholar]
- Song, X.Y.; Zhou, M.F.; Tao, Y.; Xiao, J.F. Controls on the metal compositions of magmatic sulfide deposits in the Emeishan large igneous province, SW China. Chem. Geol. 2008, 253, 38–49. [Google Scholar] [CrossRef]
- Xiao, L.; Xu, Y.G.; Mei, H.J.; Zheng, Y.F.; He, B.; Pirajno, F. Distinct mantle sources of low-Ti and high-Ti basalts from the western Emeishan large igneous province, SW China: Implications for plume–lithosphere interaction. Earth Planet. Sci. Lett. 2004, 228, 525–546. [Google Scholar] [CrossRef]
- Xu, Y.G.; Chung, S.L.; Shao, H.; He, B. Silicic magmas from the Emeishan large igneous province, Southwest China: Petrogenesis and their link with the end-Guadalupian biological crisis. Lithos 2010, 119, 47–60. [Google Scholar] [CrossRef]
- Li, B.; Zhuang, X.; Li, J.; Querol, X.; Font, O.; Moreno, N. Geological controls on mineralogy and geochemistry of the Late Permian coals in the Liulong Mine of the Liuzhi Coalfield, Guizhou Province, Southwest China. Int. J. Coal Geol. 2016, 154, 1–15. [Google Scholar] [CrossRef]
- Li, B.; Zhuang, X.; Li, J.; Querol, X.; Font, O.; Moreno, N. Enrichment and distribution of elements in the Late Permian coals from the Zhina Coalfield, Guizhou Province, Southwest China. Int. J. Coal Geol. 2017, 171, 111–129. [Google Scholar] [CrossRef]
- Zhuang, X.; Querol, X.; Zeng, R.; Xu, W.; Alastuey, A.; López-Soler, A.; Plana, F. Mineralogy and geochemistry of coal from the Liupanshui mining district, Guizhou, south China. Int. J. Coal Geol. 2000, 45, 21–37. [Google Scholar] [CrossRef]
- Dai, S.; Ren, D.; Hou, X.; Shao, L. Geochemical and mineralogical anomalies of the late Permian coal in the Zhijin Coalfield of Southwest China and their volcanic origin. Int. J. Coal Geol. 2003, 55, 117–138. [Google Scholar] [CrossRef]
- Dai, S.; Li, D.; Ren, D.; Tang, Y.; Shao, L.; Shi, Z. Geochemistry of the late Permian No. 30 Coal Seam, Zhijin Coalfield of Southwest China: Influence of the siliceous low-temperature hydrothermal fluid. Appl. Geochem. 2004, 19, 1315–1330. [Google Scholar] [CrossRef]
- Dai, S.; Ren, D.; Tang, Y.; Yue, M.; Hao, L. Concentration and distribution of elements in Late Permian coals from western Guizhou Province, China. Int. J. Coal Geol. 2005, 61, 119–137. [Google Scholar] [CrossRef]
- Dai, S.; Chou, C.-L.; Yue, M.; Luo, K.; Ren, D. Mineralogy and geochemistry of a Late Permian coal in the Dafang Coalfield, Guizhou, China: Influence from siliceous and iron-rich calcic hydrothermal fluids. Int. J. Coal Geol. 2005, 61, 241–258. [Google Scholar] [CrossRef]
- Zhang, J.; Ren, D.; Zhu, Y.; Chou, C.-L.; Zeng, R.; Zheng, B. Mineral matter and potentially hazardous trace elements in coals from Qianxi Fault Depression Area in southwestern Guizhou, China. Int. J. Coal Geol. 2004, 57, 49–61. [Google Scholar] [CrossRef]
- Zhuang, X.; Su, S.; Xiao, M.; Li, J.; Alastuey, A.; Querol, X. Mineralogy and geochemistry of the Late Permian coals in the Huayingshan coal-bearing area, Sichuan Province, China. Int. J. Coal Geol. 2012, 94, 271–282. [Google Scholar] [CrossRef]
- Dai, S.; Wang, X.; Chen, W.; Li, D.; Chou, C.-L.; Zhou, Y.; Zhu, C.; Li, H.; Zhu, X.; Xing, Y.; et al. A high-pyrite semianthracite of Late Permian age in the Songzao Coalfield, southwestern China: Mineralogical and geochemical relations with underlying mafic tuffs. Int. J. Coal Geol. 2010, 83, 430–445. [Google Scholar] [CrossRef]
- Dai, S.; Liu, J.; Ward, C.R.; Hower, J.C.; French, D.; Jia, S.; Hood, M.M.; Garrison, T.M. Mineralogical and geochemical compositions of Late Permian coals and host rocks from the Guxu Coalfield, Sichuan Province, China, with emphasis on enrichment of rare metals. Int. J. Coal Geol. 2016, 166, 71–95. [Google Scholar] [CrossRef]
- Dai, S.; Xie, P.; Jia, S.; Ward, C.R.; Hower, J.C.; Yan, X.; French, D. Enrichment of U-Re-V-Cr-Se and rare earth elements in the late Permian coals of the Moxinpo Coalfield, Chongqing, China: Genetic implications from geochemical and mineralogical data. Ore Geol. Rev. 2017, 80, 1–17. [Google Scholar] [CrossRef]
- Liu, J.; Song, H.; Dai, S.; Nechaev, V.P.; Graham, I.T.; French, D.; Nechaeva, E.V. Mineralization of REE-Y-Nb-Ta-Zr-Hf in Wuchiapingian coals from the Liupanshui Coalfield, Guizhou, southwestern China: Geochemical evidence for terrigenous input. Ore Geol. Rev. 2019, 115, 103190. [Google Scholar] [CrossRef]
- He, B.; Xu, Y.G.; Huang, X.L.; Luo, Z.Y.; Shi, Y.R.; Yang, Q.J.; Yu, S.Y. Age and duration of the Emeishan flood volcanism, SW China: Geochemistry and SHRIMP zircon U–Pb dating of silicic ignimbrites, post-volcanic Xuanwei Formation and clay tuff at the Chaotian section. Earth Planet. Sci. Lett. 2007, 255, 306–323. [Google Scholar] [CrossRef]
- He, B.; Xu, Y.G.; Zhong, Y.T.; Guan, J.P. The Guadalupian–Lopingian boundary mudstones at Chaotian (SW China) are clastic rocks rather than acidic tuffs: Implication for a temporal coincidence between the end-Guadalupian mass extinction and the Emeishan volcanism. Lithos 2010, 119, 10–19. [Google Scholar] [CrossRef]
- He, B.; Xu, Y.G.; Chung, S.L.; Xiao, L.; Wang, Y. Sedimentary evidence for a rapid, kilometer-scale crustal doming prior to the eruption of the Emeishan flood basalts. Earth Planet. Sci. Lett. 2003, 213, 391–405. [Google Scholar] [CrossRef]
- Editing Committee of Stratigraphy (ECS). Permian in China; Geological Publishing House: Beijing, China, 2000; (In Chinese with English Abstract). [Google Scholar]
- Xu, Y.G.; He, B.; Chung, S.L.; Menzies, M.A.; Frey, F.A. Geologic, geochemical, and geophysical consequences of plume involvement in the Emeishan flood-basalt province. Geology 2004, 32, 917–920. [Google Scholar] [CrossRef]
- Yang, R.; Bao, M.; Liao, L.; Wang, W.; Wei, H.; Wang, Q. Ancient weathering crust and its mineralization near the Middle-Upper Permian boundary in western Guizhou Province, China. Acta Miner. Sin. 2007, 27, 41–48, (In Chinese with English Abstract). [Google Scholar]
- Gao, C. Sequence-Palaeogeography and Coal-Accumulation of Late Permian in Chuan-Yu-Dian-Qian, China. Ph.D. Thesis, China University of Mining & Technology, Bejing, China, 2015; 172p. (In Chinese with English Abstract). [Google Scholar]
- Chung, S.L.; Jahn, B.M. Plume-lithosphere interaction in generation of the Emeishan flood basalts at the Permian-Triassic boundary. Geology 1995, 23, 889–892. [Google Scholar] [CrossRef]
- Shellnutt, J.G.; Zhou, M.F. Permian peralkaline, peraluminous and metaluminous A-type granites in the Panxi district, SW China: Their relationship to the Emeishan mantle plume. Chem. Geol. 2007, 243, 286–316. [Google Scholar] [CrossRef]
- Shellnutt, J.G.; Jahn, B.M. Formation of the Late Permian Panzhihua plutonic-hypabyssal-volcanic igneous complex: Implications for the genesis of Fe–Ti oxide deposits and A-type granites of SW China. Earth Planet. Sci. Lett. 2010, 289, 509–519. [Google Scholar] [CrossRef]
- Song, X.Y.; Zhou, M.F.; Keays, R.R.; Cao, Z.M.; Sun, M.; Qi, L. Geochemistry of the Emeishan flood basalts at Yangliuping, Sichuan, SW China: Implications for sulfide segregation. Contrib. Miner. Petrol. 2006, 152, 53–74. [Google Scholar] [CrossRef]
- Xiao, L.; Xu, Y.G.; Chung, S.L.; He, B.; Mei, H. Chemostratigraphic correlation of Upper Permian lavas from Yunnan Province, China: Extent of the Emeishan large igneous province. Int. Geol. Rev. 2003, 45, 753–766. [Google Scholar] [CrossRef]
- Xu, Y.; Chung, S.L.; Jahn, B.M.; Wu, G. Petrologic and geochemical constraints on the petrogenesis of Permian–Triassic Emeishan flood basalts in southwestern China. Lithos 2001, 58, 145–168. [Google Scholar] [CrossRef]
- ASTM Standard D3173-11. Test Method for Moisture in the Analysis Sample of Coal and Coke; ASTM International: West Conshohocken, PA, USA, 2011. [Google Scholar]
- ASTM Standard D3174-11. Annual Book of ASTM Standards. In Test Method for Ash in the Analysis Sample of Coal and Coke; ASTM International: West Conshohocken, PA, USA, 2011. [Google Scholar]
- ASTM Standard D3175-11. Test Method for Volatile Matter in the Analysis Sample of Coal and Coke; ASTM International: West Conshohocken, PA, USA, 2011. [Google Scholar]
- Li, B.; Zhuang, X.; Querol, X.; Moreno, N.; Córdoba, P.; Li, J.; Zhou, J.; Ma, X.; Liu, S.; Shangguan, Y. The mode of occurrence and origin of minerals in the Early Permian high-rank coals of the Jimunai depression, Xinjiang Uygur Autonomous Region, NW China. Int. J. Coal Geol. 2019, 205, 58–74. [Google Scholar] [CrossRef]
- Chung, F.H. Quantitative interpretation of X-ray diffraction patterns of mixtures: I. Matrix flushing method for quantitative multicomponent analysis. J. Appl. Crystalogr. 1974, 7, 519–525. [Google Scholar] [CrossRef]
- Querol, X.; Whateley, M.; Fernandez-Turiel, J.; Tuncali, E. Geological controls on the mineralogy and geochemistry of the Beypazari lignite, Central Anatolia, Turkey. Int. J. Coal Geol. 1997, 33, 255–271. [Google Scholar] [CrossRef]
- GB/T 1574-2007 (National Standard of P.R. China). Test Method for Analysis of Coal Ash; Standards Press of China: Beijing, China, 2007. (In Chinese) [Google Scholar]
- MT/T 850-2000 (National Standard of P.R. China). Classification for Mositure of Coal; Standards Press of China: Beijing, China, 2000. (In Chinese) [Google Scholar]
- GB/T 15224.1-2010 (National Standard of P.R. China). Classification for Quality of Coal; Standards Press of China: Beijing, China, 2010. Part 1: Ash Yield. (In Chinese) [Google Scholar]
- ASTM Standard D388-12. Standard Classification of Coals by Rank; ASTM International: West Conshohocken, PA, USA, 2012; Reapproved 2007. [Google Scholar]
- Chou, C.-L. Sulfur in coals: A review of geochemistry and origins. Int. J. Coal Geol. 2012, 100, 1–13. [Google Scholar] [CrossRef]
- Zhao, L.; Ward, C.R.; French, D.; Graham, I.T.; Dai, S.; Yang, C.; Xie, P.; Zhang, S. Origin of a kaolinite-NH4-illite-pyrophyllite-chlorite assemblage in a marine-influenced anthracite and associated strata from the Jincheng Coalfield, Qinshui Basin, Northern China. Int. J. Coal Geol. 2018, 185, 61–78. [Google Scholar] [CrossRef]
- Wang, X.; Dai, S.; Chou, C.-L.; Zhang, M.; Wang, J.; Song, X.; Wang, W.; Jiang, Y.; Zhou, Y.; Ren, D. Mineralogy and geochemistry of Late Permian coals fromthe Taoshuping Mine, Yunnan Province, China: Evidences for the sources ofminerals. Int. J. Coal Geol. 2012, 96, 49–59. [Google Scholar] [CrossRef]
- Rao, C.P.; Gluskoter, H.J. Occurrence and Distribution of Minerals in Illinois Coals; Circular 476; Illinois State Geological Survey: Urbana, IL, USA, 1973; 56p. [Google Scholar]
- Ward, C.R. Minerals in bituminous coals of the Sydney Basin (Australia) and the Illinois Basin (USA). Int. J. Coal Geol. 1989, 13, 455–479. [Google Scholar] [CrossRef]
- Ketris, M.P.; Yudovich, Y.E. Estimations of Clarkes for carbonaceous biolithes: World average for trace element contents in black shales and coals. Int. J. Coal Geol. 2009, 78, 135–148. [Google Scholar] [CrossRef]
- Dai, S.; Seredin, V.V.; Ward, C.R.; Hower, J.C.; Xing, Y.; Zhang, W.; Song, W.; Wang, P. Enrichment of U–Se–Mo–Re–V in coals preserved within marine carbonate successions: Geochemical and mineralogical data from the Late Permian Guiding Coalfield, Guizhou, China. Mineralium Deposita 2015, 50, 159–186. [Google Scholar] [CrossRef]
- Seredin, V.V.; Dai, S. Coal deposits as a potential alternative source for lanthanides and yttrium. Int. J. Coal Geol. 2012, 94, 67–93. [Google Scholar] [CrossRef]
- Taylor, S.R.; McLennan, S.M. The Continental Crust: Its Composition and Evolution; Blackwell: Oxford, UK, 1985; 312p. [Google Scholar]
- Dai, S.; Zhang, W.; Ward, C.R.; Seredin, V.V.; Hower, J.C.; Li, X.; Song, W.; Wang, X.; Kang, H.; Zheng, L.; et al. Mineralogical and geochemical anomalies of late Permian coals from the Fusui Coalfield, Guangxi Province, southern China: Influences of terrigenous materials and hydrothermal fluids. Int. J. Coal Geol. 2013, 105, 60–84. [Google Scholar] [CrossRef]
- Murray, R.W.; Buchholtz ten Brink, M.R.; Jones, D.L.; Gerlach, D.C.; Russ III, G.P. Rare earth elements as indicators of different marine depositional environments in chert and shale. Geology 1990, 18, 268–271. [Google Scholar] [CrossRef]
- Bullock, L.A.; Parnell, J.; Perez, M.; Armstrong, J.G.; Feldmann, J.; Boyce, A.J. High selenium in the Carboniferous Coal Measures of Northumberland, North East England. Int. J. Coal Geol. 2018, 195, 61–74. [Google Scholar] [CrossRef]
- Çelik, Y.; Karayiğit, A.İ.; Querol, X.; Oskay, R.G.; Mastalerz, M.; Özer, M.S.K. Coal characteristics, palynology, and palaeoenvironmental interpretation of the Yeniköy coal of Late Oligocene age in the Thrace Basin (NW Turkey). Int. J. Coal Geol. 2017, 181, 103–123. [Google Scholar] [CrossRef]
- Marynowski, L.; Pisarzowska, A.; Derkowski, A.; Rakociński, M.; Szaniawski, R.; Środoń, J.; Cohen, A.S. Influence of palaeoweathering on trace metal concentrations and environmental proxies in black shales. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2017, 472, 177–191. [Google Scholar] [CrossRef]
- Dai, S.; Luo, Y.; Seredin, V.V.; Ward, C.R.; Hower, J.C.; Zhao, L.; Liu, S.; Tian, H.; Zou, J. Revisiting the late Permian coal fromthe Huayingshan, Sichuan, southwestern China: Enrichment and occurrence modes of minerals and trace elements. Int. J. Coal Geol. 2014, 122, 110–128. [Google Scholar] [CrossRef]
- Zhou, Y.; Bohor, B.F.; Ren, Y. Trace element geochemistry of altered volcanic ash layers (tonsteins) in Late Permian coal-bearing formations of eastern Yunnan and western Guizhou Province, China. Int. J. Coal Geol. 2000, 44, 305–324. [Google Scholar] [CrossRef]
- Hayashi, K.I.; Fujisawa, H.; Holland, H.D.; Ohmoto, H. Geochemistry of 1.9 Ga sedimentary rocks from northeastern Labrador, Canada. Geochim. Cosmochim. Acta 1997, 61, 4115–4137. [Google Scholar] [CrossRef]
- Young, G.M.; Nesbitt, H.W. Processes controlling the distribution of Ti and Al in weathering profiles, siliciclastic sediments and sedimentary rocks. J. Sediment. Res. 1998, 68, 448–455. [Google Scholar] [CrossRef]
- Dai, S.; Li, T.; Seredin, V.V.; Ward, C.R.; Hower, J.C.; Zhou, Y.; Zhang, M.; Song, X.; Song, W. Origin of minerals and elements in the Late Permian coals, tonsteins, and host rocks of the Xinde Mine, Xuanwei, eastern Yunnan, China. Int. J. Coal Geol. 2014, 121, 53–78. [Google Scholar] [CrossRef]
- Dai, S.; Yang, J.; Ward, C.R.; Hower, J.C.; Liu, H.; Garrison, T.M.; O’Keefe, J.M. Geochemical and mineralogical evidence for a coal-hosted uranium deposit in the Yili Basin, Xinjiang, northwestern China. Ore Geol. Rev. 2015, 70, 1–30. [Google Scholar] [CrossRef]
- Dai, S.; Guo, W.; Nechaev, V.P.; French, D.; Ward, C.R.; Spiro, B.F.; Finkelman, R.B. Modes of occurrence and origin of mineral matter in the Palaeogene coal (No. 19-2) from the Hunchun Coalfield, Jilin Province, China. Int. J. Coal Geol. 2018, 189, 94–110. [Google Scholar] [CrossRef]
- Hower, J.C.; Eble, C.F.; Dai, S.; Belkin, H.E. Distribution of rare earth elements in eastern Kentucky coals: Indicators of multiple modes of enrichment. Int. J. Coal Geol. 2016, 160, 73–81. [Google Scholar] [CrossRef]
- Dai, S.; Graham, I.T.; Ward, C.R. A review of anomalous rare earth elements and yttrium in coal. Int. J. Coal Geol. 2016, 159, 82–95. [Google Scholar] [CrossRef]
- Rard, J.A. Chemistry and thermodynamics of europium and some if its simpler inorganic compounds and aqueous species. Chem. Rev. 1985, 85, 555–582. [Google Scholar] [CrossRef]
- Cullers, R.L.; Chaudhuri, S.; Arnold, B.; Lee, M.; Wolf, C.W., Jr. Rare earth distributions in clay minerals and in the clay-sized fraction of the Lower Permian Havensville and Eskridge shales of Kansas and Oklahoma. Geochim. Cosmochim. Acta 1975, 39, 1691–1703. [Google Scholar] [CrossRef]
- Cullers, R.L. Implications of elemental concentrations for provenance, redox conditions, and metamorphic studies of shales and limestones near Pueblo, CO, USA. Chem. Geol. 2002, 191, 305–327. [Google Scholar] [CrossRef]
- Nesbitt, H.W.; Wilson, R.E. Recent chemical weathering of basalts. Am. J. Sci. 1992, 292, 740–777. [Google Scholar] [CrossRef]
- Dai, S.; Ren, D.; Chou, C.L.; Li, S.; Jiang, Y. Mineralogy and geochemistry of the no. 6 coal (Pennsylvanian) in the Junger Coalfield, Ordos Basin, China. Int. J. Coal Geol. 2006, 66, 253–270. [Google Scholar] [CrossRef]
- Dai, S.; Li, D.; Chou, C.L.; Zhao, L.; Zhang, Y.; Ren, D.; Sun, Y. Mineralogy and geochemistry of boehmite-rich coals: New insights from the Haerwusu Surface Mine, Jungar Coalfield, Inner Mongolia, China. Int. J. Coal Geol. 2008, 74, 185–202. [Google Scholar] [CrossRef]
- Dai, S.; Zou, J.; Jiang, Y.; Ward, C.R.; Wang, X.; Li, T.; Xue, W.; Liu, S.; Tian, H.; Sun, X.; et al. Mineralogical and geochemical compositions of the Pennsylvanian coal in the Adaohai Mine, Daqingshan Coalfield, Inner Mongolia, China: Modes of occurrence and origin of diaspore, gorceixite, and ammonian illite. Int. J. Coal Geol. 2012, 94, 250–270. [Google Scholar] [CrossRef]
- Dai, S.; Jiang, Y.; Ward, C.R.; Gu, L.; Seredin, V.V.; Liu, H.; Zhou, D.; Wang, X.; Sun, Y.; Zou, J.; et al. Mineralogical and geochemical compositions of the coal in the Guanbanwusu Mine, Inner Mongolia, China: Further evidence for the existence of an Al (Ga and REE) ore deposit in the Jungar Coalfield. Int. J. Coal Geol. 2012, 98, 10–40. [Google Scholar] [CrossRef]
- Permana, A.K.; Ward, C.R.; Li, Z.; Gurba, L.W. Distribution and origin of minerals in high-rank coals of the South Walker Creek area, Bowen Basin, Australia. Int. J. Coal Geol. 2013, 116, 185–207. [Google Scholar] [CrossRef]
- Wang, X.; Dai, S.; Ren, D.; Yang, J. Mineralogy and geochemistry of Al-hydroxide/oxyhydroxide mineral-bearing coals of Late Paleozoic age from the Weibei coalfield, southeastern Ordos Basin, North China. Appl. Geochem. 2011, 26, 1086–1096. [Google Scholar] [CrossRef]
- Dai, S.; Ji, D.; Ward, C.R.; French, D.; Hower, J.C.; Yan, X.; Wei, Q. Mississippian anthracites in Guangxi Province, southern China: Petrological, mineralogical, and rare earth element evidence for high-temperature solutions. Int. J. Coal Geol. 2018, 197, 84–114. [Google Scholar] [CrossRef]
- Daniels, E.J.; Altaner, S.P. Clay mineral authigenesis in coal and shale from the Anthracite region, Pennsylvania. Am. Mineral. 1990, 75, 825–839. [Google Scholar]
- Daniels, E.J.; Aronson, J.L.; Altaner, S.P.; Clauer, N. Late Permian age of NH4- bearing illite in anthracite from eastern Pennsylvania: Temporal limits on coalification in the central Appalachians. Geol. Soc. Am. Bull. 1994, 106, 760–766. [Google Scholar] [CrossRef]
- Ward, C.R.; Christie, P.J. Clays and other minerals in coal seams of the Moura-Baralaba area, Bowen Basin, Australia. Int. J. Coal Geol. 1994, 47, 31–49. [Google Scholar] [CrossRef]
- Daniels, E.J.; Altaner, S.P. Inorganic nitrogen in anthracite from eastern Pennsylvania, USA. Int. J. Coal Geol. 1993, 22, 21–35. [Google Scholar] [CrossRef]
- Sucha, V.; Kraus, I.; Madejova, J. Ammonium illite from anchimetamorphic shales associated with anthracite in the Zemplinicum of the Western Carpathians. Clay Miner. 1994, 29, 369–377. [Google Scholar] [CrossRef]
- Liu, Q.; Zhang, P.; Ding, S.; Lin, X.; Zheng, N. Ammoniumillite in Permo-Carboniferous coal-bearing strata, North China. Chin. Sci. Bull. 1996, 41, 717–719. (In Chinese) [Google Scholar]
- Liang, S.; Liu, Q.; Yu, C.; Song, H. Ammonium-bearing illite in tonsteins of Permo- Carboniferous coal accumulation area of Northern China. J. Hebei Inst. Archit. Sci. Technol. 2005, 22, 59–65, (In Chinese with English Abstract). [Google Scholar]
- Williams, L.B.; Ferrell, R.E., Jr.; Chinn, E.W.; Sassen, R. Fixed-ammonium in clays associated with crude oils. Appl. Geochem. 1989, 4, 605–616. [Google Scholar] [CrossRef]
- Williams, L.B.; Wilcoxon, B.R.; Ferrell, R.E.; Sassen, R. Diagenesis of ammonium during hydrocarbon maturation and migration, Wilcox Group, Louisiana, USA. Appl. Geochem. 1992, 7, 123–134. [Google Scholar] [CrossRef]
- Boudou, J.P.; Schimmelmann, A.; Ader, M.; Mastalerz, M.; Sebilo, M.; Gengembre, L. Organic nitrogen chemistry during low-grade metamorphism. Geochim. Cosmochim. Acta 2008, 72, 1199–1221. [Google Scholar] [CrossRef]
- Juster, T.C.; Brown, P.E.; Bailey, S.W. NH4-bearing illite in very low grade metamorphic rocks associated with coal, northeastern Pennsylvania. Am. Mineral. 1987, 72, 555–565. [Google Scholar]
- Williams, L.B.; Ferrell, R.E. Ammonium substitution in illite during maturation of organic matter. Clays Clay Miner. 1991, 39, 400–408. [Google Scholar] [CrossRef]
- Zheng, Q.; Liu, Q.; Shi, S. Mineralogy and geochemistry of ammonian illite in intra-seam partings in Permo-Carboniferous coal of the Qinshui Coalfield, North China. Int. J. Coal Geol. 2016, 153, 1–11. [Google Scholar] [CrossRef]
- Hao, G. Characteristics, origin and geological significance of ammonium illite in coal measures. Ph.D. Thesis, China University of Mining & Technology, Bejing, China, 2013; 120p. (In Chinese with English Abstract). [Google Scholar]
- Liu, Q.; Liu, L.; Shen, S.; Ding, S.; Zhen, L.; Cheng, H. Leaching experiments on nitrogen release from ammonium illite gangue. J. China Coal Soc. 2009, 34, 1022–1026, (In Chinese with English Abstract). [Google Scholar]
- Liu, Q.; Liu, L.; Ding, S. The water-solubility and exchangeability of NH4+ in ammonium illite. Acta Mineral. Sin. 2010, 3, 3, (In Chinese with English Abstract). [Google Scholar]



| Sample | Thickness | Mad | Ad | VMdaf | Cdaf | Hdaf | Ndaf | St,d | Ro |
|---|---|---|---|---|---|---|---|---|---|
| CLB4-1 | 0.10 | 1.51 | 40.6 | 26.7 | 70.0 | 4.13 | 0.64 | 24.1 | 1.81 |
| CLB4-2 | 0.20 | 1.80 | 22.2 | 18.0 | 71.9 | 4.23 | 0.90 | 13.2 | 1.75 |
| CLB4-3 | 0.20 | 1.35 | 14.1 | 14.2 | 83.2 | 4.53 | 1.08 | 4.9 | 1.74 |
| CLB4-4 | 0.20 | 1.30 | 10.7 | 12.8 | 87.9 | 4.73 | 1.25 | 2.6 | 1.95 |
| CLB4-5 | 0.20 | 1.17 | 19.6 | 14.8 | 81.5 | 4.76 | 1.24 | 5.7 | 1.73 |
| CLB4-6 | 0.15 | 0.74 | 10.9 | 13.8 | 87.0 | 4.59 | 1.28 | 2.7 | 1.87 |
| CLB4-7 | 0.15 | 0.91 | 9.8 | 12.9 | 87.8 | 4.65 | 1.33 | 2.3 | 1.89 |
| CLB4-8 | 0.15 | 1.18 | 8.2 | 12.8 | 87.9 | 4.82 | 1.33 | 2.7 | 1.90 |
| CLB4-9 | 0.15 | 1.43 | 10.8 | 13.3 | 86.4 | 4.67 | 1.31 | 4.8 | 1.75 |
| CLB4-10 | 0.15 | 1.16 | 24.4 | 18.2 | 74.1 | 3.76 | 0.99 | 14.2 | 1.70 |
| CLB4-11 | 0.15 | 1.43 | 15.3 | 13.0 | 84.7 | 4.74 | 1.54 | 6.1 | 1.63 |
| CLB4-12 | 0.15 | 1.35 | 16.9 | 14.7 | 86.3 | 4.80 | 1.51 | 3.3 | 1.67 |
| CLB4-13 | 0.15 | 1.57 | 7.4 | 15.0 | 87.0 | 4.88 | 1.29 | 4.7 | 1.62 |
| CLB4-14 | 0.15 | 1.56 | 8.9 | 14.7 | 85.0 | 4.46 | 1.16 | 6.5 | 1.59 |
| CLB-F1 | 0.30 | nd | 72.7 | nd | nd | nd | nd | 24.4 | nd |
| CLB-F2 | 0.25 | nd | 100.0 | nd | nd | nd | nd | 5.1 | nd |
| Sample | Kaolinite | Quartz | Calcite | Pyrite | Marcasite | Tobelite | Svanbergite | Anatase | Gypsum |
|---|---|---|---|---|---|---|---|---|---|
| CLB4-1 | <dl | 2.0 | <dl | 38.2 | <dl | <dl | <dl | <dl | <dl |
| CLB4-2 | 4.4 | <dl | <dl | 17.0 | 0.4 | <dl | <dl | <dl | <dl |
| CLB4-3 | 3.2 | <dl | <dl | 9.8 | 0.8 | 0.3 | <dl | <dl | <dl |
| CLB4-4 | 2.9 | <dl | <dl | 3.8 | 0.5 | 3.5 | <dl | <dl | <dl |
| CLB4-5 | 2.6 | <dl | <dl | 12.4 | <dl | 4.6 | <dl | <dl | <dl |
| CLB4-6 | 2.5 | <dl | <dl | 3.5 | <dl | 4.9 | <dl | <dl | <dl |
| CLB4-7 | <dl | <dl | 2.3 | 3.1 | <dl | 4.3 | <dl | <dl | <dl |
| CLB4-8 | <dl | <dl | 2.5 | 2.4 | <dl | 3.3 | <dl | <dl | <dl |
| CLB4-9 | <dl | <dl | 0.6 | 7.0 | <dl | 3.2 | <dl | <dl | <dl |
| CLB4-10 | <dl | <dl | 3.9 | 18.9 | <dl | 1.6 | <dl | <dl | <dl |
| CLB4-11 | <dl | <dl | <dl | 9.9 | <dl | 5.4 | <dl | <dl | <dl |
| CLB4-12 | 1.6 | <dl | <dl | 4.6 | <dl | 5.8 | 4.6 | <dl | <dl |
| CLB4-13 | <dl | <dl | 0.4 | 5.9 | <dl | 1.1 | <dl | <dl | <dl |
| CLB4-14 | <dl | <dl | <dl | 8.5 | <dl | 0.4 | <dl | <dl | <dl |
| CLB-F1 | 12.4 | 14.8 | <dl | 45.6 | <dl | <dl | <dl | <dl | <dl |
| CLB-F2 | 44.0 | 39.4 | 2.1 | 10.6 | <dl | <dl | <dl | 1.9 | 2.0 |
| Sample | Weight (g) | Volume (mL) | NH4+ (mg/L) | NH4+ (μg/g) |
|---|---|---|---|---|
| CLB4-5+6 | 0.5002 | 25 | 0.283 | 14.14 |
| CLB4-5+6 | 0.5010 | 50 | 0.231 | 23.05 |
| CLB4-5+6 | 0.5093 | 25 | 0.306 | 15.02 |
| CLB4-5+6 | 0.5018 | 50 | 0.239 | 23.81 |
| CLB4-5+6 | 0.2504 | 50 | 0.178 | 35.54 |
| CLB4-2 | 0.5074 | 25 | 0.033 | 1.63 |
| CLB4-2 | 0.5031 | 50 | 0.020 | 1.99 |
| CLB4-2 | 0.5072 | 25 | 0.038 | 1.87 |
| CLB4-2 | 0.5030 | 50 | 0.021 | 2.11 |
| Sample | CLB 4-1 | CLB 4-2 | CLB 4-3 | CLB 4-4 | CLB 4-5 | CLB 4-6 | CLB 4-7 | CLB 4-8 | CLB 4-9 | CLB 4-10 | CLB 4-11 | CLB 4-12 | CLB 4-13 | CLB 4-14 | CLB -F1 | CLB -F2 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | 1.96 | 2.06 | 1.66 | 3.06 | 3.50 | 3.58 | 2.14 | 1.61 | 1.60 | 0.78 | 2.66 | 3.61 | 0.53 | 0.18 | 17.17 | 58.95 |
| TiO2 | 0.15 | 0.13 | 0.15 | 0.15 | 0.18 | 0.13 | 0.13 | 0.07 | 0.06 | 0.02 | 0.07 | 0.08 | 0.03 | 0.03 | 0.93 | 1.52 |
| Al2O3 | 2.60 | 2.21 | 3.36 | 3.58 | 5.09 | 3.76 | 3.31 | 2.63 | 2.33 | 1.46 | 3.50 | 5.42 | 1.25 | 0.92 | 7.96 | 16.18 |
| Fe2O3 | 29.59 | 15.06 | 4.74 | 1.36 | 5.62 | 1.51 | 0.93 | 1.26 | 4.19 | 16.60 | 5.67 | 2.14 | 3.72 | 6.26 | 35.29 | 9.79 |
| MgO | 0.04 | 0.03 | 0.06 | 0.06 | 0.09 | 0.07 | 0.06 | 0.05 | 0.04 | 0.04 | 0.07 | 0.05 | 0.02 | 0.03 | 0.13 | 0.20 |
| CaO | 0.08 | 0.09 | 0.10 | 0.09 | 0.07 | 0.09 | 0.43 | 0.51 | 0.41 | 1.06 | 0.34 | 0.72 | 0.24 | 0.20 | 0.48 | 1.30 |
| Na2O | 0.02 | 0.03 | 0.04 | 0.05 | 0.06 | 0.04 | 0.03 | 0.03 | 0.03 | 0.02 | 0.05 | 0.05 | 0.05 | 0.06 | 0.11 | 0.24 |
| K2O | 0.07 | 0.04 | 0.08 | 0.10 | 0.19 | 0.10 | 0.10 | 0.06 | 0.05 | 0.04 | 0.14 | 0.08 | 0.03 | 0.04 | 0.19 | 0.42 |
| Li | 17 | 13 | 35 | 35 | 52 | 48 | 46 | 40 | 25 | 10 | 30 | 40 | 5.5 | 2.2 | 49 | 76 |
| Be | <dl | 1.9 | 3.1 | 2.9 | 2.6 | 3.1 | 3.8 | 3.2 | 3.0 | 2.0 | 3.2 | 4.8 | 1.6 | 1.6 | 0.92 | 1.2 |
| P | 166 | 99 | 93 | 57 | 312 | 636 | 66 | 57 | 67 | 48 | 1536 | 9753 | 220 | 816 | 1092 | 131 |
| B | 2.0 | 6.0 | 1.0 | 13 | 15 | 7.3 | 17 | 29 | 18 | 7.0 | 32 | 41 | 22 | 16 | 28 | 19 |
| Sc | 2 | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | 8.2 | 9.7 |
| V | 106 | 39 | 24 | 22 | 19 | 16 | 21 | 29 | 29 | 10 | 18 | 26 | 27 | 21 | 144 | 237 |
| Cr | 20 | 9.2 | 13 | 12 | 10 | 8.3 | 10 | 11 | 10 | 2.6 | 6.9 | 12 | 15 | 13 | 97 | 175 |
| Mn | 58 | 23 | 8.8 | 9.0 | 9.0 | 9.5 | 45 | 36 | 47 | 106 | 26 | 26 | 21 | 10 | 697 | 20 |
| Co | 26 | 2.4 | 2.5 | 1.3 | 2.1 | 1.6 | 2.4 | 2.3 | 6.1 | 9.2 | 14 | 19 | 8.4 | 15 | 47 | 15 |
| Ni | 64 | 9.4 | 6.9 | 5.6 | 8.5 | 8.0 | 7.1 | 6.4 | 7.4 | 6.6 | 13 | 17 | 10 | 13 | 40 | 57 |
| Cu | 31 | 18 | 12 | 8.7 | 25 | 13 | 11 | 11 | 14 | 27 | 25 | 24 | 14 | 18 | 59 | 83 |
| Zn | 4.2 | 26 | 12 | 29 | 6.9 | 28 | 16 | 7.3 | 6.8 | 19 | 11 | 46 | 39 | 76 | 47 | 77 |
| Ga | 3.9 | 4.9 | 5.4 | 5.1 | 6.1 | 6.0 | 5.0 | 4.1 | 3.4 | 2.6 | 6.2 | 5.0 | 1.8 | 2.3 | 18 | 17 |
| Ge | 1.2 | 3.2 | 3.0 | 4.1 | 4.5 | 1.7 | 1.7 | 6.2 | 7.2 | 3.4 | 11 | 17 | 22 | 18 | 1.2 | 1.0 |
| As | 18 | 17 | 4.6 | 2.7 | 11 | 2.4 | 1.9 | 2.0 | 3.7 | 18 | 4.9 | 6.5 | 6.9 | 5.3 | 24 | 9.3 |
| Se | 15 | 19 | 10 | 5.6 | 19 | 6.8 | 4.3 | 3.9 | 5.0 | 16 | 8.3 | 13 | 7.7 | 6.2 | 20 | 11 |
| Rb | 1.2 | 0.8 | 1.8 | 2.2 | 4.7 | 2.0 | 2.2 | <dl | <dl | <dl | 4.2 | 1.5 | <dl | 1.1 | 4.8 | 8.3 |
| Sr | 84 | 84 | 81 | 87 | 197 | 365 | 90 | 89 | 87 | 97 | 590 | 3947 | 83 | 76 | 104 | 104 |
| Y | 10 | 7.5 | 13 | 14 | 14 | 16 | 21 | 13 | 8.5 | 3.6 | 8.7 | 34 | 3.5 | 5.7 | 25 | 21 |
| Zr | 119 | 38 | 83 | 52 | 103 | 109 | 163 | 47 | 32 | 8.9 | 24 | 128 | 17 | 10 | 332 | 326 |
| Nb | 7.2 | 5.1 | 12 | 7.9 | 18 | 20 | 21 | 7.7 | 3.4 | 1.2 | 6.2 | 26 | 2.5 | 1.1 | 44 | 49 |
| Mo | 67 | 80 | 14 | 4.9 | 4.7 | 4.7 | 3.2 | 3.5 | 7.5 | 4.3 | 3.7 | 6.1 | 7.8 | 9.4 | 53 | 4.3 |
| Sn | 1.4 | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | 5.3 | 4.0 |
| Cs | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | 0.52 | 0.90 |
| Ba | 32 | 6.1 | 13 | 15 | 28 | 21 | 17 | 7.9 | 6.3 | 6.5 | 16 | 36 | 3.8 | 5.4 | 27 | 20 |
| La | 15 | 16 | 25 | 21 | 28 | 55 | 17 | 14 | 8 | 1.4 | 29 | 75 | 0.9 | 1.5 | 48 | 65 |
| Ce | 29 | 25 | 54 | 40 | 47 | 95 | 35 | 30 | 20 | 3.5 | 48 | 136 | 2.0 | 3.8 | 88 | 99 |
| Pr | 2.9 | 2.4 | 6.0 | 4.4 | 5.0 | 9.6 | 3.9 | 3.2 | 2.2 | <dl | 4.6 | 15 | <dl | <dl | 9.3 | 11 |
| Nd | 10 | 8.5 | 22 | 16 | 18 | 33 | 15 | 13 | 8.5 | 1.8 | 15 | 60 | 1.0 | 2.2 | 33.6 | 35.9 |
| Sm | 2.0 | 1.7 | 4.7 | 3.5 | 3.1 | 5.1 | 3.3 | 2.9 | 2.0 | <dl | 2.6 | 12 | <dl | <dl | 6.2 | 5.4 |
| Eu | <dl | <dl | <dl | <dl | <dl | 0.68 | <dl | <dl | <dl | <dl | 0.36 | 0.9 | <dl | <dl | 1.0 | 1.0 |
| Gd | 1.9 | 1.7 | 3.7 | 3.1 | 2.7 | 3.7 | 3.3 | 2.6 | 1.7 | <dl | 2.3 | 9.4 | <dl | 1.0 | 5.4 | 5.0 |
| Tb | <dl | <dl | <dl | <dl | <dl | 0.79 | <dl | <dl | <dl | <dl | 0.42 | 1.07 | <dl | <dl | 0.80 | 0.81 |
| Dy | 2.0 | 1.3 | 2.4 | 2.4 | 2.4 | 2.7 | 3.3 | 2.1 | 1.3 | <dl | 1.4 | 5.3 | <dl | 0.8 | 4.5 | 5.2 |
| Ho | <dl | <dl | <dl | <dl | <dl | 0.71 | <dl | <dl | <dl | <dl | 0.37 | 0.97 | <dl | <dl | 0.86 | 0.99 |
| Er | 1.3 | 0.83 | 1.5 | 1.4 | 1.6 | 1.7 | 2.2 | 1.2 | 0.89 | <dl | 0.88 | 3.1 | <dl | <dl | 2.5 | 2.7 |
| Tm | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | 0.36 | 0.40 |
| Yb | 1.5 | 0.82 | 1.4 | 1.3 | 1.6 | 1.7 | 2.2 | 1.2 | 0.92 | <dl | 0.92 | 3.0 | <dl | <dl | 2.4 | 2.7 |
| Lu | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | 0.34 | 0.38 |
| Hf | 1.7 | 1.0 | 2.3 | 1.5 | 2.8 | 2.9 | 4.3 | 1.3 | 0.90 | 0.25 | 0.62 | 3.5 | 0.46 | 0.29 | 7.7 | 9.1 |
| Ta | <dl | <dl | <dl | <dl | <dl | <dl | 0.79 | <dl | <dl | <dl | <dl | <dl | <dl | <dl | 2.6 | 2.8 |
| Tl | 0.78 | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | <dl | 0.15 | 0.10 |
| Pb | 8.3 | 13 | 6.8 | 2.6 | 12 | 4.6 | 2.2 | 2.1 | 3.7 | 14 | 2.8 | 4.1 | 2.4 | 1.9 | 19 | 21 |
| Th | 2.0 | 2.3 | 4.0 | 3.6 | 7.1 | 4.1 | 4.8 | 2.2 | 1.3 | <dl | 0.93 | 3.1 | <dl | <dl | 9.9 | 14 |
| U | 16 | 12 | 3.3 | 2.4 | 2.5 | 2.5 | 3.0 | 3.7 | 1.8 | 0.51 | 1.1 | 4.6 | 5.3 | 4.5 | 3.5 | 8.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, B.; Zhuang, X.; Querol, X.; Moreno, N.; Yang, L.; Shangguan, Y.; Li, J. Mineralogy and Geochemistry of Late Permian Coals within the Tongzi Coalfield in Guizhou Province, Southwest China. Minerals 2020, 10, 44. https://doi.org/10.3390/min10010044
Li B, Zhuang X, Querol X, Moreno N, Yang L, Shangguan Y, Li J. Mineralogy and Geochemistry of Late Permian Coals within the Tongzi Coalfield in Guizhou Province, Southwest China. Minerals. 2020; 10(1):44. https://doi.org/10.3390/min10010044
Chicago/Turabian StyleLi, Baoqing, Xinguo Zhuang, Xavier Querol, Natalia Moreno, Linjian Yang, Yunfei Shangguan, and Jing Li. 2020. "Mineralogy and Geochemistry of Late Permian Coals within the Tongzi Coalfield in Guizhou Province, Southwest China" Minerals 10, no. 1: 44. https://doi.org/10.3390/min10010044
APA StyleLi, B., Zhuang, X., Querol, X., Moreno, N., Yang, L., Shangguan, Y., & Li, J. (2020). Mineralogy and Geochemistry of Late Permian Coals within the Tongzi Coalfield in Guizhou Province, Southwest China. Minerals, 10(1), 44. https://doi.org/10.3390/min10010044





