When and Why Did Brains Break Symmetry?
Abstract
:1. Asymmetry in the Brains of Vertebrates


2. Midline Crossing of Sensory Inputs and Motor Outputs
3. Asymmetry in the Brains of Invertebrates

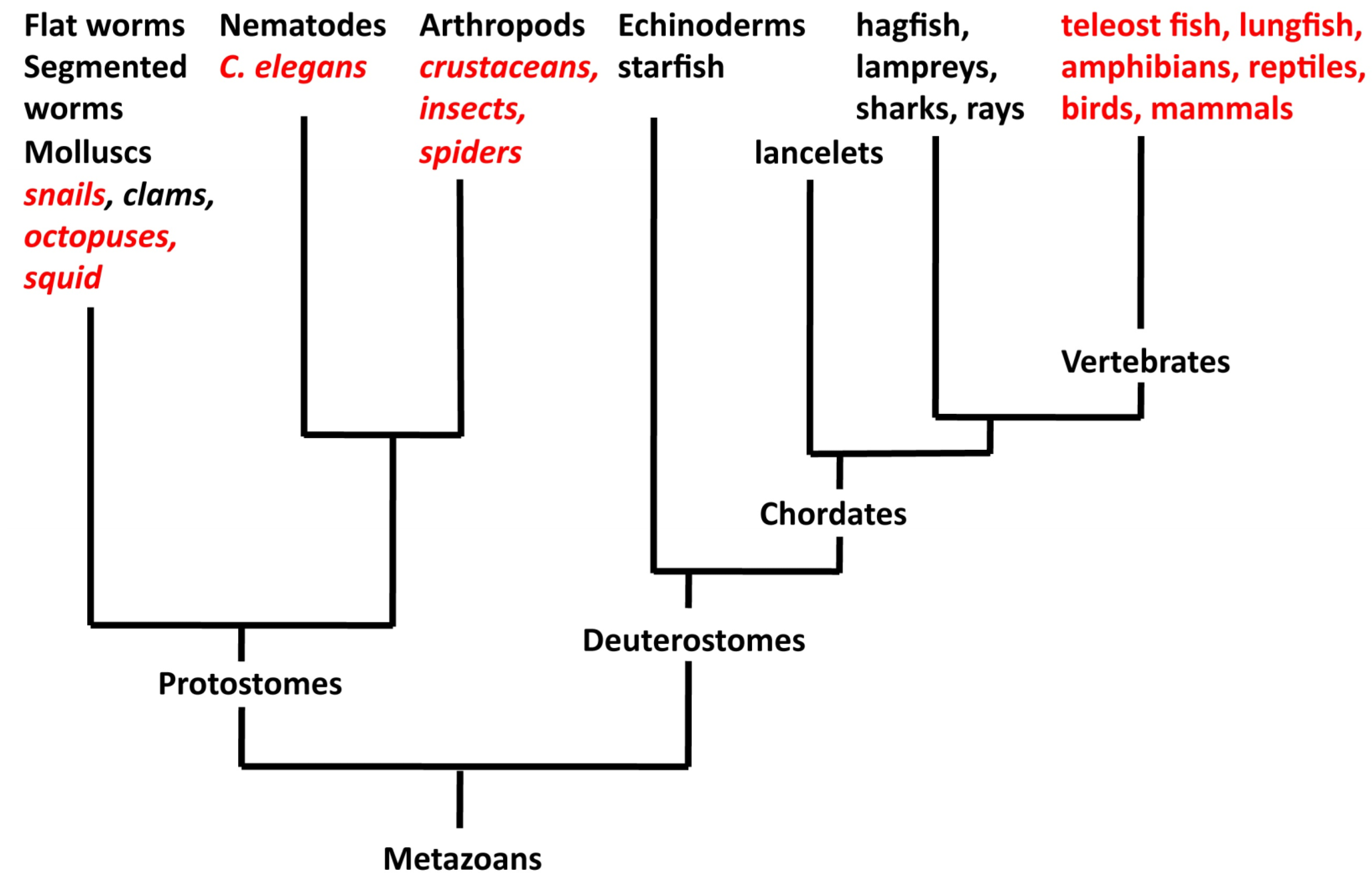
4. What Is the Relationship between Asymmetry in Invertebrates and Vertebrates?
5. Genes as the Foundation, Experience as the Decider
6. Advantages of Having an Asymmetrical Brain
7. Disadvantages in Having an Asymmetrical Brain
8. Why Lateralize Rather than Grow Larger?
9. Concluding Remarks
Author Contributions
Conflicts of Interest
References
- Geschwind, N.; Levitsky, W. Human brain: Left-right asymmetries in temporal speech region. Science 1968, 161, 186–187. [Google Scholar] [CrossRef] [PubMed]
- Toga, A.W.; Thompson, P.M. Mapping brain asymmetry. Nat. Rev. Neurosci. 2003, 4, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Rogers, L.J.; Vallortigara, G.; Andrew, R.J. Divided Brains: The Biology and Behaviour of Brain Asymmetries; Cambridge University Press: Cambridge, UK, 2013. [Google Scholar]
- Vallortigara, G.; Rogers, L.J.; Bisazza, A. Possible evolutionary origins of cognitive brain lateralization. Brain Res. Rev. 1999, 30, 164–175. [Google Scholar] [CrossRef]
- Vallortigara, G.; Chiandetti, C.; Sovrano, V.A. Brain asymmetry (animal). Wiley Interdiscip. Rev. Cogn. Sci. 2011, 2, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Rogers, L.J.; Sink, H.S. Transient asymmetry in the projections of the rostral thalamus to the visual hyperstriatum of the chicken, and reversal of its direction by light exposure. Exp. Brain Res. 1988, 70, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Dreosti, E.; Llopis, N.V.; Carl, M.; Yaksi, E.; Wilson, S.W. Left-right asymmetry is required for the habenulae to respond to both visual and olfactory stimuli. Curr. Biol. 2014, 24, 440–445. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, R.; Shinohara, Y.; Kato, Y.; Sugiyama, H.; Shigemoto, R.; Ito, I. Asymmetrical allocation of NMDA receptor ε2 subunits in hippocampal circuitry. Science 2003, 300, 990–994. [Google Scholar] [CrossRef] [PubMed]
- Shipton, O.A.; El-Gaby, M.; Apergis-Schoute, J.; Deisseroth, K.; Bannerman, D.M.; Paulsen, O.; Kohl, M.M. Left-right dissociation of hippocampal memory processes in mice. PNAS 2014, 111, 15238–15243. [Google Scholar] [CrossRef] [PubMed]
- Rogers, L.J. Development and functional lateralization in the avian brain. Brain Res. Bull. 2008, 76, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Vallortigara, G. Comparative neuropsychology of the dual brain: A stroll through left and right animals’ perceptual worlds. Brain Lang. 2000, 73, 189–219. [Google Scholar] [CrossRef] [PubMed]
- Rogers, L.J. The two hemispheres of the avian brain: Their differing roles in perceptual processing and the expression of behaviour. J. Ornithol. 2012, 153, S61–S74. [Google Scholar] [CrossRef]
- MacNeilage, P.; Rogers, L.J.; Vallortigara, G. Origins of the left and right brain. Sci. Am. 2009, 301, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Tommasi, L.; Vallortigara, G. Encoding of geometric and landmark information in the left and right hemispheres of the avian brain. Behav. Neurosci. 2001, 115, 602–613. [Google Scholar] [CrossRef] [PubMed]
- Salva, O.R.; Regolin, L.; Mascalzoni, E.; Vallortigara, G. Cerebral and behavioural asymmetries in animal social recognition. Comp. Cogn. Behav. Rev. 2012, 7, 110–138. [Google Scholar] [CrossRef]
- Vallortigara, G.; Cozzutti, C.; Tommasi, L.; Rogers, L.J. How birds use their eyes: Opposite left-right specialisation for the lateral and frontal visual hemifield in the domestic chick. Curr. Biol. 2001, 11, 29–33. [Google Scholar] [CrossRef]
- Broad, K.D.; Mimmack, M.L.; Kendrick, K.M. Is right hemisphere specialization for face discrimination specific to humans? Europ. J. Neurosci. 2000, 12, 731–741. [Google Scholar] [CrossRef]
- Daisley, J.N.; Regolin, L.; Vallortigara, G. Logic in an asymmetrical (social) brain: Transitive inference in the young domestic chick. Soc. Neurosci. 2010, 5, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Austin, N.P.; Rogers, L.J. Asymmetry of flight and escape turning responses in horses. Laterality 2007, 12, 464–474. [Google Scholar] [CrossRef] [PubMed]
- Austin, N.A.; Rogers, L.J. Limb preferences and lateralization of aggression, reactivity and vigilance in feral horses, Equus caballus. Anim. Behav. 2012, 83, 239–247. [Google Scholar] [CrossRef]
- Letzkus, P.; Ribi, W.A.; Wood, J.T.; Zhu, H.; Zhang, S.W.; Srinivasan, M.V. Lateralization of olfaction in the honeybee Apis mellifera. Curr. Biol. 2006, 16, 1471–1476. [Google Scholar] [CrossRef] [PubMed]
- Rogers, L.J.; Vallortigara, G. From antenna to antenna: Lateral shift of olfactory memory in honeybees. PLoS One 2008, 3. [Google Scholar] [CrossRef] [PubMed]
- Frasnelli, E.; Vallortigara, G.; Rogers, L.J. Right-left antennal asymmetry of odour memory recall in three species of Australian stingless bees. Behav. Brain Res. 2011, 224, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Pascual, A.; Huang, K.-L.; Neveu, J.; Préat, T. Brain asymmetry and long-term memory. Nature 2004, 427, 605–606. [Google Scholar]
- Rogers, L.J.; Rigosi, E.; Frasnelli, E.; Vallortigara, G. A right antenna for social behaviour in honeybees. Nature Comm. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Frasnelli, E.; Vallortigara, G.; Rogers, L.J. Left-right asymmetries of behavioural and nervous system in invertebrates. Neurosci. Biobehav. Rev. 2012, 36, 1273–1291. [Google Scholar] [CrossRef] [PubMed]
- Frasnelli, E.; Iakovlev, I.; Reznikova, Z. Asymmetry in antennal contacts during trophallaxis. Behav. Brain Res. 2012, 232, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Ades, C.; Ramires, E.N. Asymmetry of leg use during prey handling in the spider Scytodes globula (Scytodidae). J. Insect Behav. 2002, 15, 563–570. [Google Scholar] [CrossRef]
- Heuts, B.A.; Lambrechts, D.Y.M. Positional biases in leg loss of spiders and harvestmen (Arachnida). Entomol. Ber. (Amst) 1999, 59, 13–20. [Google Scholar]
- Byrne, R.A.; Kuba, M.; Griebel, U. Lateral asymmetry of eye use in Octopus vulgaris. Anim. Behav. 2002, 64, 461–468. [Google Scholar] [CrossRef]
- Byrne, R.A.; Kuba, M.J.; Meisel, D.V. 2004 Lateralized eye use in Octopus vulgaris shows antisymmetrical distribution. Anim. Behav. 2004, 68, 1107–1114. [Google Scholar] [CrossRef]
- Jozet-Alves, C.; Viblanc, V.; Romagny, S.; Dacher, M.; Healy, S.; Dickel, L. Ontogenetic study of side-turning preference in cuttlefish (Sepia officinalis). Anim. Behav. 2012, 83, 1313–1318. [Google Scholar] [CrossRef]
- Corballis, M.C. The trade-off between symmetry and asymmetry. Behav. Brain Sci. 2005, 28, 594–595. [Google Scholar] [CrossRef]
- Wentworth, S.L.; Muntz, W.R.A. Asymmetries in the sense organs and central nervous system of the squid Histioteuthis. J. Zool. 1989, 219, 607–619. [Google Scholar] [CrossRef]
- Boorman, C.J.; Shimeld, S.M. The evolution of left-right asymmetry in chordates. Bioessays 2002, 24, 1004–1011. [Google Scholar] [CrossRef] [PubMed]
- Boorman, C.J.; Shimeld, S.M. Pitx homeobox genes in Ciona and amphoxius show left-right asymmetry in a conserved chordate character and define the ascidian adenohypophysis. Evol. Dev. 2002, 4, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Soukup, V.; Yong, L.K.; Lu, T.-M.; Huang, S.-W.; Kozmik, Z.; Yu, K., Jr. The Nodal signaling pathway controls left-right asymmetric development in amphioxus. Evol. Dev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Hamada, H. Left-right patterning: Conserved and divergent mechanisms. Development 2012, 139, 3257–3262. [Google Scholar] [CrossRef] [PubMed]
- Grande, C.; Patel, N.H. Nodal signalling is involved in left-right asymmetry in snails. Nature 2009, 457, 1007–1011. [Google Scholar] [CrossRef] [PubMed]
- Warner, J.F.; McClay, D.R. Left-right asymmetry in the sea urchin. Genesis 2014, 52, 481–487. [Google Scholar] [CrossRef]
- Watanabe, H.; Schmidt, H.A.; Kuhn, A.; Höger, S.K.; Kocagöz, Y.; Laumann-Lipp, N.; Özbek, S.; Holstein, T.W. Nodal signalling determines biradial asymmetry in Hydra. Nature 2014, 515, 112–115. [Google Scholar] [CrossRef] [PubMed]
- Namigai, E.K.O.; Kenny, N.J.; Shimeld, S.M. Right across the tree of life: The evolution of left-right asymmetry in Bilateria. Genesis 2014, 52, 458–470. [Google Scholar] [CrossRef] [PubMed]
- Halpern, M.E.; Liang, J.O.; Gamse, J.T. Leaning to the left: Laterality in the zebrafish forebrain. Trends Neurosci. 2003, 26, 308–313. [Google Scholar] [CrossRef]
- Roussigne, M.; Blader, P.; Wilson, S.W. Breaking symmetry: The zebrafish as a model for understanding left-right asymmetry in the developing brain. Dev. Neurobiol. 2012, 72-3, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Cotelis, J.-B.; Géminard, C.; Spéder, P.; Magali, S.; Petzoldt, A.G.; Noselli, S. Drosophila left/right asymmetry establishment is controlled by the Hox gene abdominal-B. Dev. Cell 2013, 24, 89–97. [Google Scholar]
- Géminard, C.; Gonzales-Morales, N.; Coutelis, J.-B.; Noselli, S. The myosin ID pathway and left-right asymmetry in Drosophila. Genesis 2014, 52, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Halpern, M.E.; Hobert, O.; Wright, C.V.E. Left-right asymmetry: Advances and enigmas. Genesis 2014, 52, 451–454. [Google Scholar] [CrossRef] [PubMed]
- Hobert, O. Development of left/right asymmetry in the Caenorhabitis elegans nervous system: From zygote to postmitotic neuron. Genesis 2014, 52, 528–543. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, Y.-W.; Alqadah, A.; Chuang, C.-F. Asymmetric neural development in the Caenorhabditis elegans olfactory system. Genesis 2014, 52, 544–554. [Google Scholar] [CrossRef] [PubMed]
- Rogers, L.J. Light input and the reversal of functional lateralization in the chicken brain. Behav. Brain Res. 1990, 38, 211–221. [Google Scholar] [CrossRef]
- Manns, M.; Strökens, F. Functional and structural comparison of visual lateralization in birds—Similar but still different. Front. Psychol. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Budaev, S.; Andrew, R.J. Patterns of early embryonic light exposure determine behavioural asymmetries in zebrafish: A habenular hypothesis. Behav. Brain Res. 2009, 200, 91–94. [Google Scholar] [CrossRef] [PubMed]
- Andrew, R.J. Origins of asymmetry in the CNS. Semin. Cell Dev. Biol. 2009, 20, 485–490. [Google Scholar] [CrossRef] [PubMed]
- Chiandetti, C.; Galliussi, J.; Andrew, R.J.; Vallortigara, G. Early-light embryonic stimulation suggests a second route, via gene activation, to cerebral lateralization in vertebrates. Nature Comm. Sci. Rep. 2013, 3. [Google Scholar] [CrossRef] [PubMed]
- Rogers, L.J. Asymmetry of brain and behavior in animals, its development, function and human relevance. Genesis 2014, 52, 555–571. [Google Scholar] [CrossRef] [PubMed]
- Chung, W. Analysis of the asymmetric gene expression between the left and right hemispheres of Drosophila brain. J. Behav. Brain Sci. 2015, 5, 440–447. [Google Scholar] [CrossRef]
- Jozet-Alves, C.; Hébert, M. Embryonic exposure to predator odour modulates visual lateralization in cuttlefish. Proc. Roy. Soc. B. 2013, 208. [Google Scholar] [CrossRef] [PubMed]
- Bianki, V.L. The Mechanisms of Brain Lateralization; Gordon and Breach Science Publishers: Montreux, Switzerland, 1993. [Google Scholar]
- Rogers, L.J.; Zucca, P.; Vallortigara, G. Advantage of having a lateralized brain. Proc. Roy. Soc. Lond. B. 2004, 271, S420–S422. [Google Scholar] [CrossRef] [PubMed]
- Güntürkün, O.; Diekamp, B.; Manns, M.; Nottelmann, F.; Prior, H.; Schwarz, A.; Skiba, M. Asymmetry pays: Visual lateralization improves discrimination success in pigeons. Curr. Biol. 2000, 10, 1079–1081. [Google Scholar] [CrossRef]
- McGrew, W.C.; Marchant, L.F. Laterality of hand use pays off in foraging success for wild chimpanzees. Primates 1999, 40, 509–513. [Google Scholar] [CrossRef]
- Rigosi, E.; Haase, A.; Rath, L.; Anfora, G.; Vallortigara, G.; Szyszka, P. Asymmetric neural coding revealed by in vivo calcium imaging in the honey bee brain. Proc. R. Soc. B. 2015, 282. [Google Scholar] [CrossRef] [PubMed]
- Versace, E.; Vallortigara, G. Forelimb preferences in human beings and other species: multiple models for testing hypotheses on lateralization. Front. Psychol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, W.D.; Misiura, M.; Pope, S.M.; Latash, E.M. Behavioural and brain asymmetries in primates: A preliminary evaluations of two evolutionary hypotheses. Ann. N.Y. Acad. Sci. 2015. [Google Scholar] [CrossRef] [PubMed]
- Rogers, L.J.; Workman, L. Light exposure during incubation affects competitive behaviour in domestic chicks. Appl. Anim. Behav. Sci. 1989, 23, 187–198. [Google Scholar]
- Vallortigara, G. The evolutionary psychology of left and right: Costs and benefits of lateralization. Dev. Psychobiol. 2006, 48, 418–427. [Google Scholar] [CrossRef] [PubMed]
- Ghirlanda, S.; Vallortigara, G. The evolution of brain lateralization: A game theoretical analysis of population structure. Proc. Roy. Soc. B. 2004, 153, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Ghirlanda, S.; Frasnelli, E.; Vallortigara, G. Intraspecific competition and coordination in the evolution of lateralization. Phil. Trans. Roy. Soc. Lond. B. 2009, 364, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Raymond, M.; Pointier, D.; Dufour, A.-B.; Moller, A.P. Frequency-dependent maintenance of left handedness in humans. Proc. Roy. Soc. B. 1996, 263, 1627–1633. [Google Scholar] [CrossRef] [PubMed]
- Hori, M. Frequency-dependent natural selection in the handedness of scale-eating cichlid fish. Science 1993, 260, 216–219. [Google Scholar] [CrossRef] [PubMed]
- Bisazza, A.; Cantalupo, C.; Capocchiano, M.; Vallortigara, G. Population lateralization and social behaviour: A study of 16 species of fish. Laterality 2000, 3, 269–284. [Google Scholar]
- Vallortigara, G.; Rogers, L.J. Survival with an asymmetrical brain: Advantages and disadvantages of cerebral lateralization. Behav. Brain Sci. 2005, 28, 575–633. [Google Scholar] [CrossRef] [PubMed]
- Fonseca-Azevedo, K.; Herculano-Houzel, S. Metabolic constraint imposes tradeoff between body size and number of brain neurons in human evolution. Proc. Natl. Acad. Sci. USA 2012, 109, 18571–18576. [Google Scholar] [CrossRef] [PubMed]
- Wylie, D.R.W.; Gutiérrez-Ibánez, C.; Iwaniuk, A. Integrating brain, behavior, and phylogeny to understand the evolution of sensory systems in birds. Front. Neurosci. 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Kotrschal, A.; Rogell, B.; Bundsen, A.; Svensson, B.; Zajitschek, S.; Brännström, I.; Immler, S.; Maklakov, A.A.; Kolm, N. Artificial selection on relative brain size in the guppy reveals costs and benefits of evolving a larger brain. Curr. Biol. 2013, 23, 168–171. [Google Scholar] [CrossRef] [PubMed]
- Gotts, S.J.; Jo, H.J.; Wallace, G.L.; Saad, Z.S.; Cox, R.W.; Martin, A. Two distinct forms of functional lateralization in the human brain. PNAS 2013, E3435–E3444. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.F. Using both sides of your brain: The case for rapid interhemispheric switching. PLoS Biol. 2008, 6. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rogers, L.J.; Vallortigara, G. When and Why Did Brains Break Symmetry? Symmetry 2015, 7, 2181-2194. https://doi.org/10.3390/sym7042181
Rogers LJ, Vallortigara G. When and Why Did Brains Break Symmetry? Symmetry. 2015; 7(4):2181-2194. https://doi.org/10.3390/sym7042181
Chicago/Turabian StyleRogers, Lesley J., and Giorgio Vallortigara. 2015. "When and Why Did Brains Break Symmetry?" Symmetry 7, no. 4: 2181-2194. https://doi.org/10.3390/sym7042181
APA StyleRogers, L. J., & Vallortigara, G. (2015). When and Why Did Brains Break Symmetry? Symmetry, 7(4), 2181-2194. https://doi.org/10.3390/sym7042181







