Fluctuating Asymmetry: Methods, Theory, and Applications
Abstract
1. Introduction
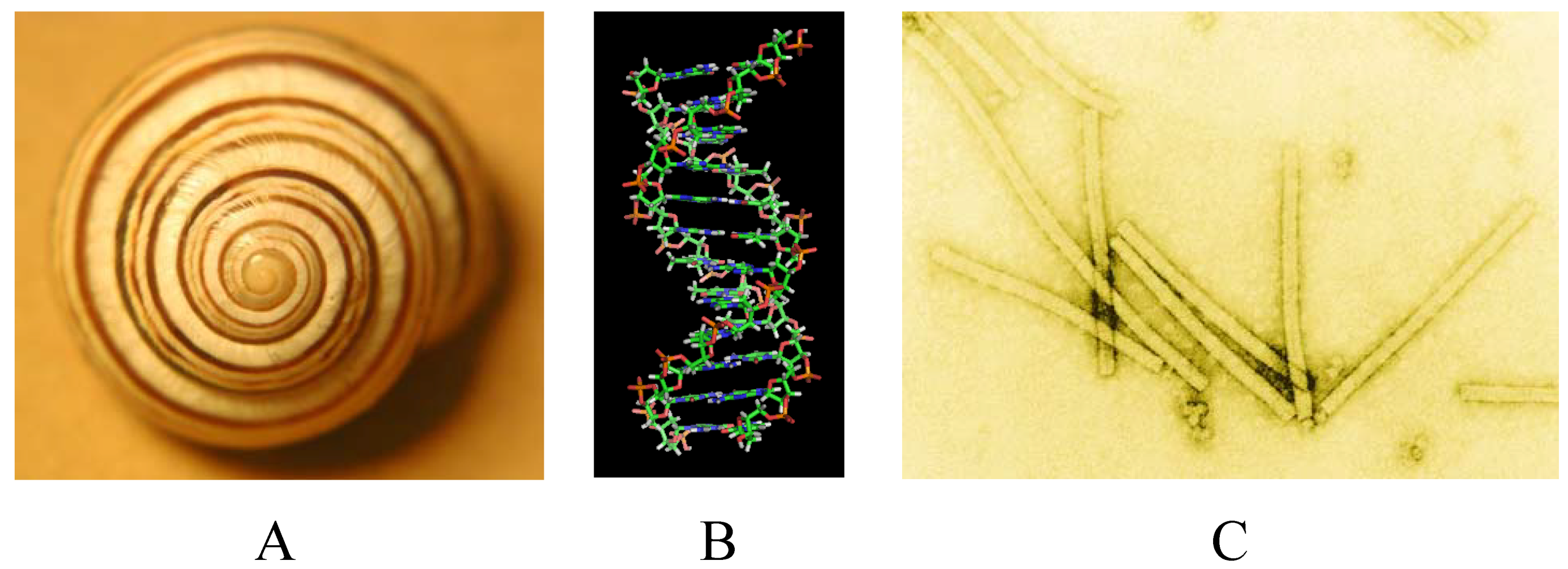
2. Symmetries
2.1. Types of Symmetry
2.2. Symmetry Groups
2.3. Types of Objects
3. Measuring Deviations from Perfect Symmetry
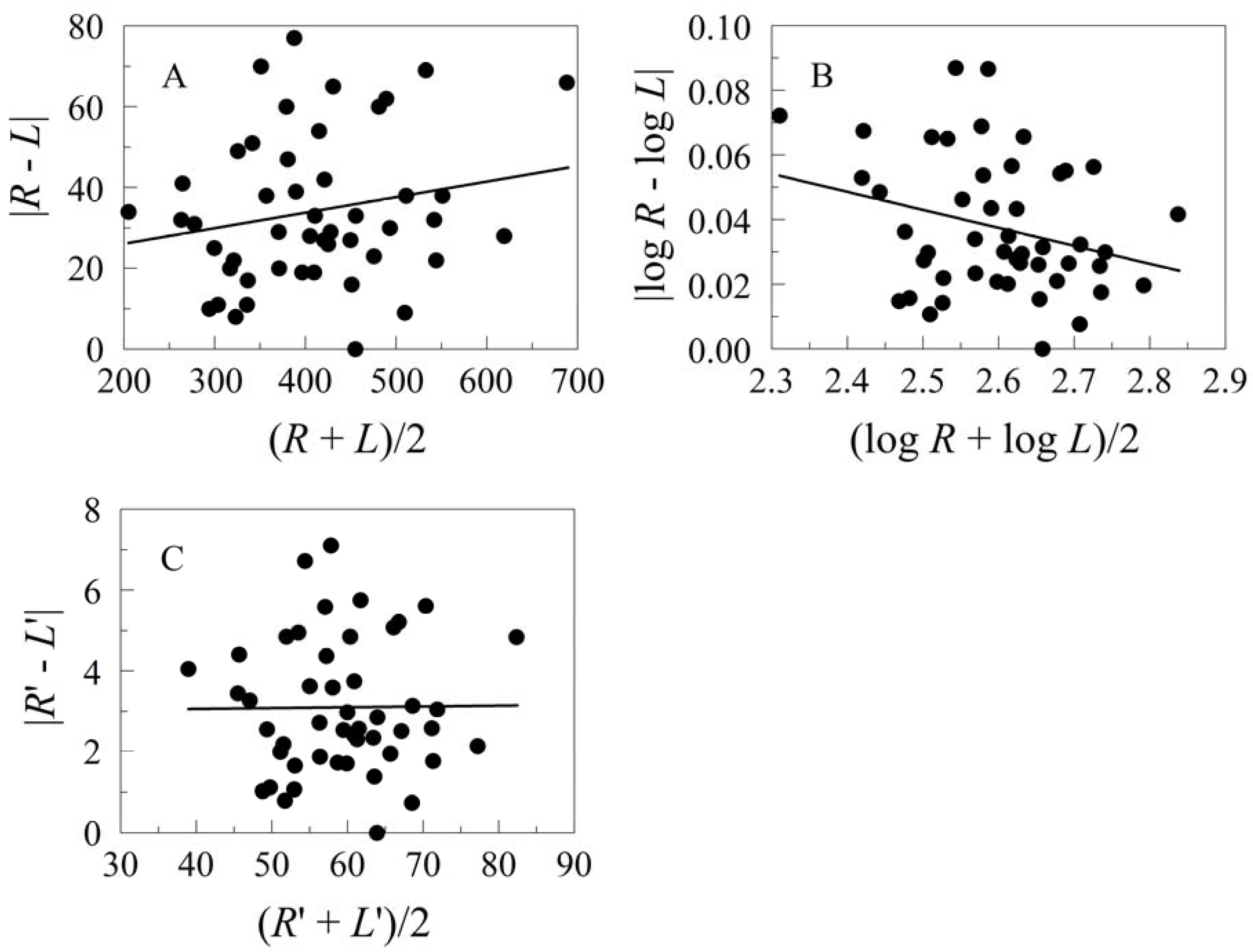
3.1. Measures of Dispersion
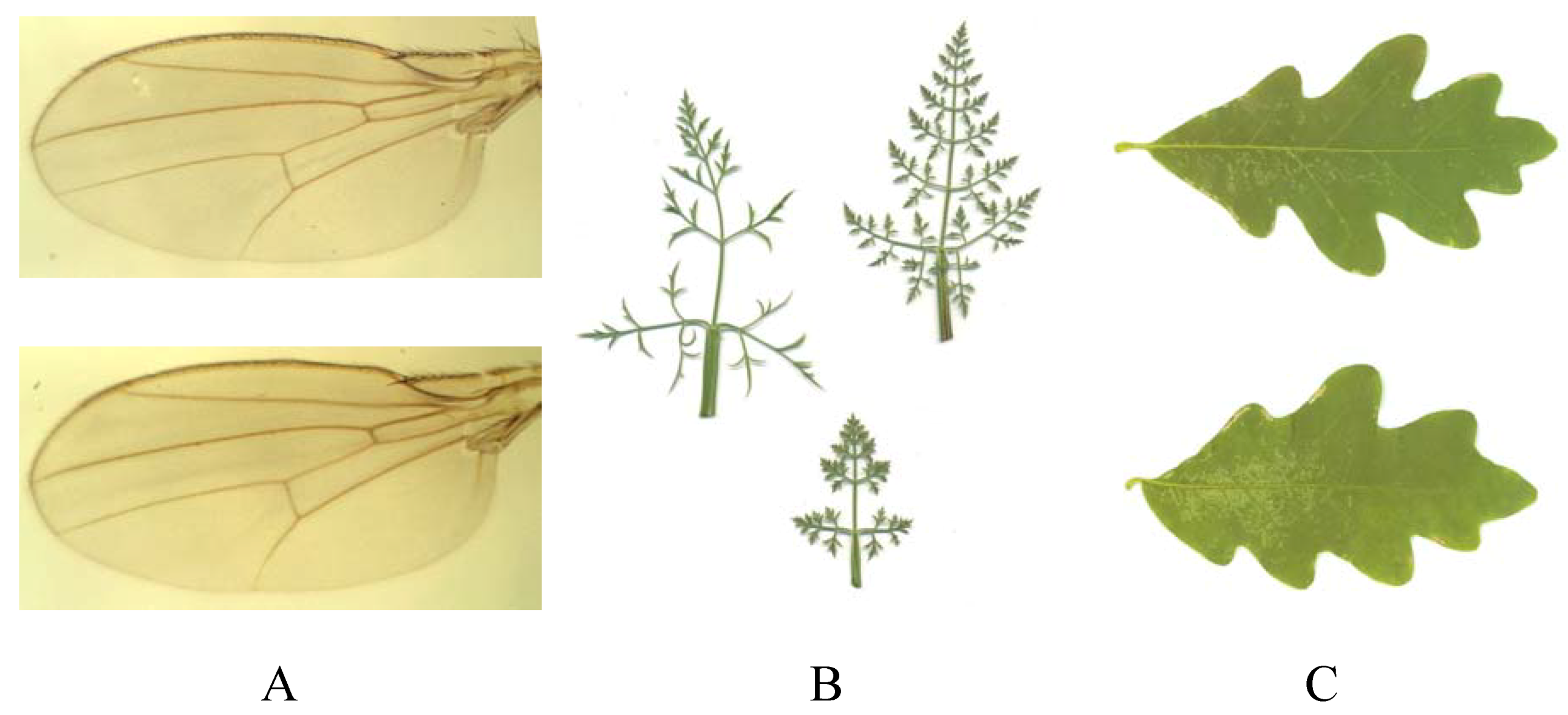
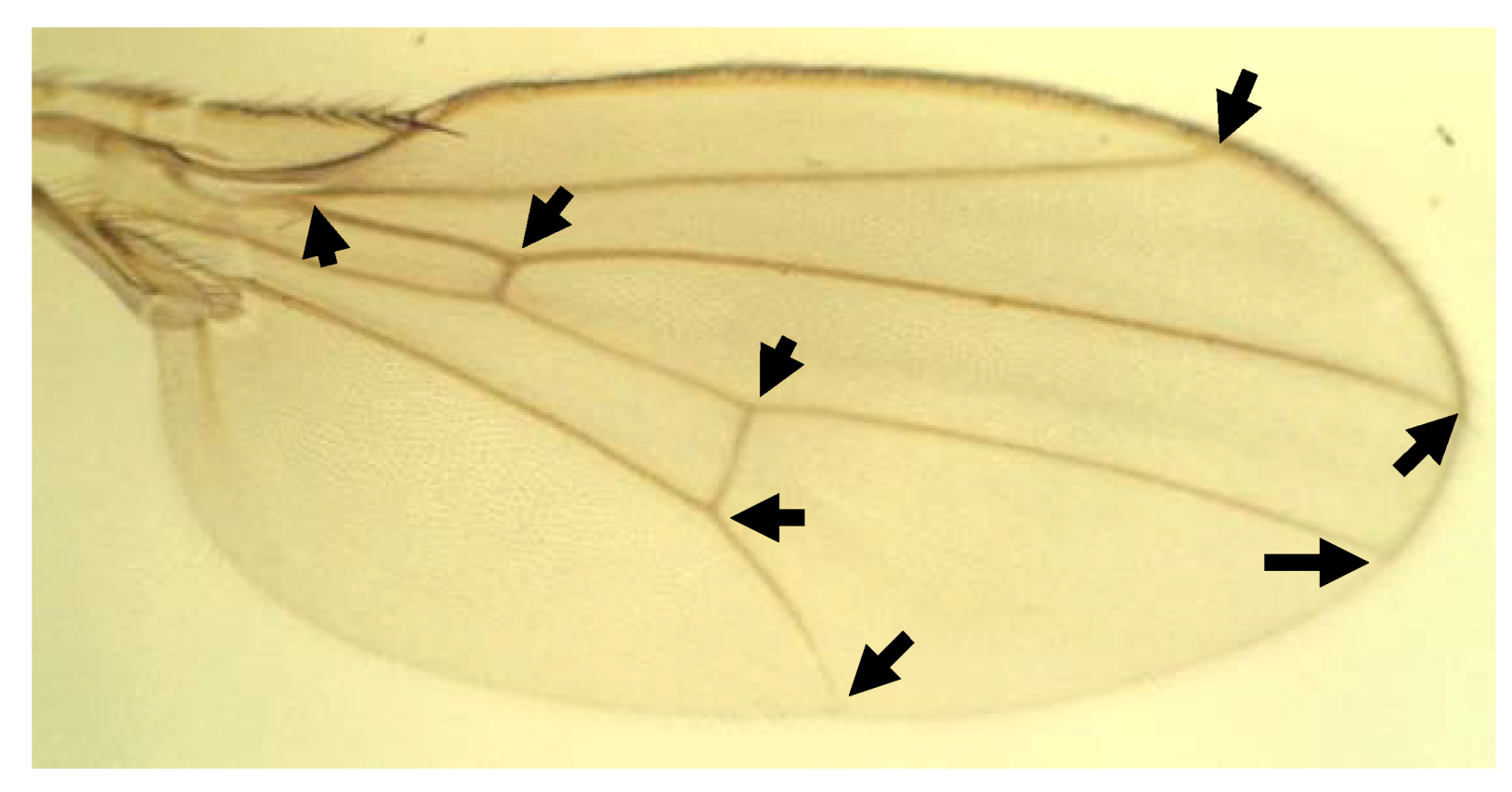
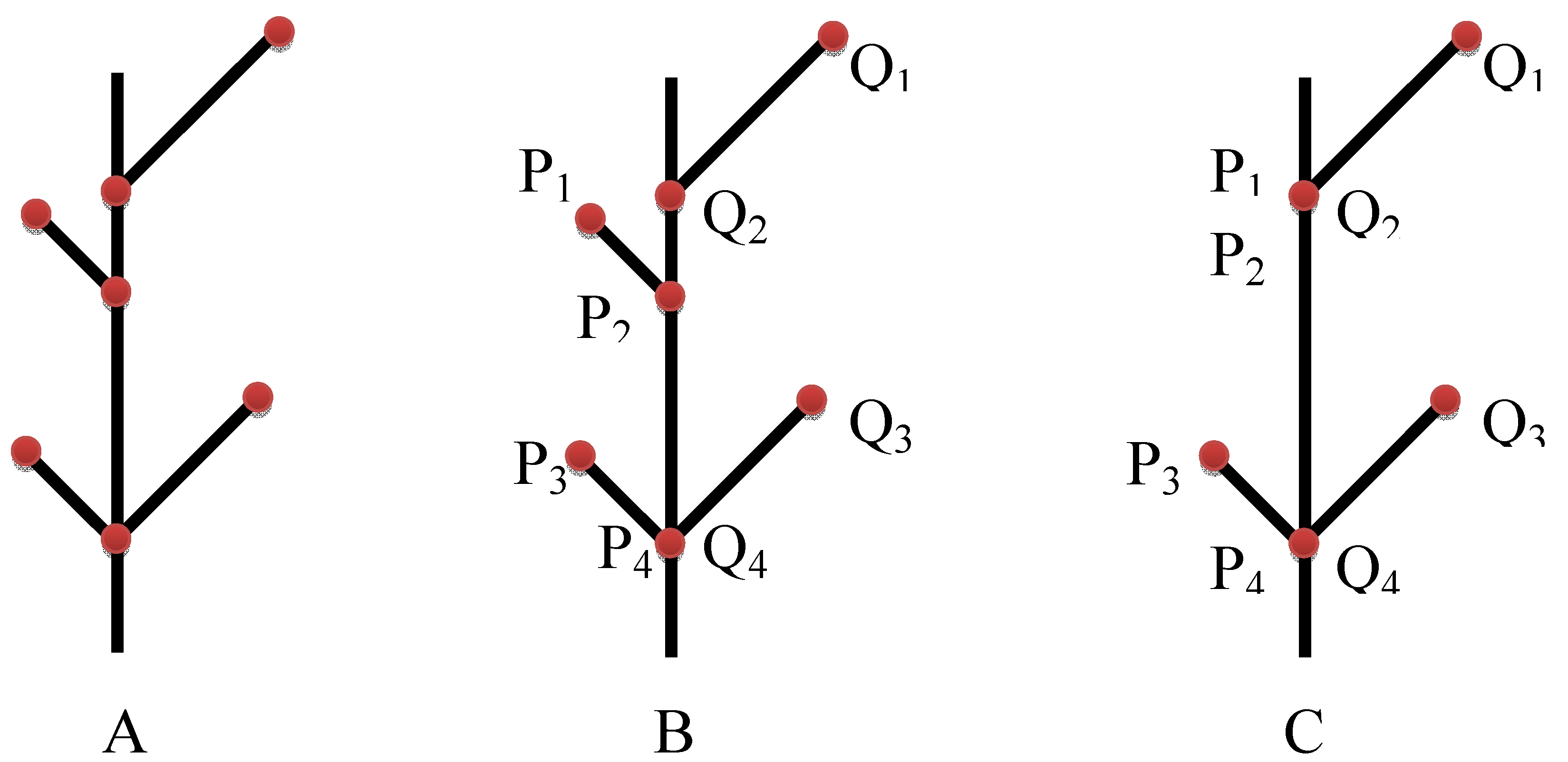
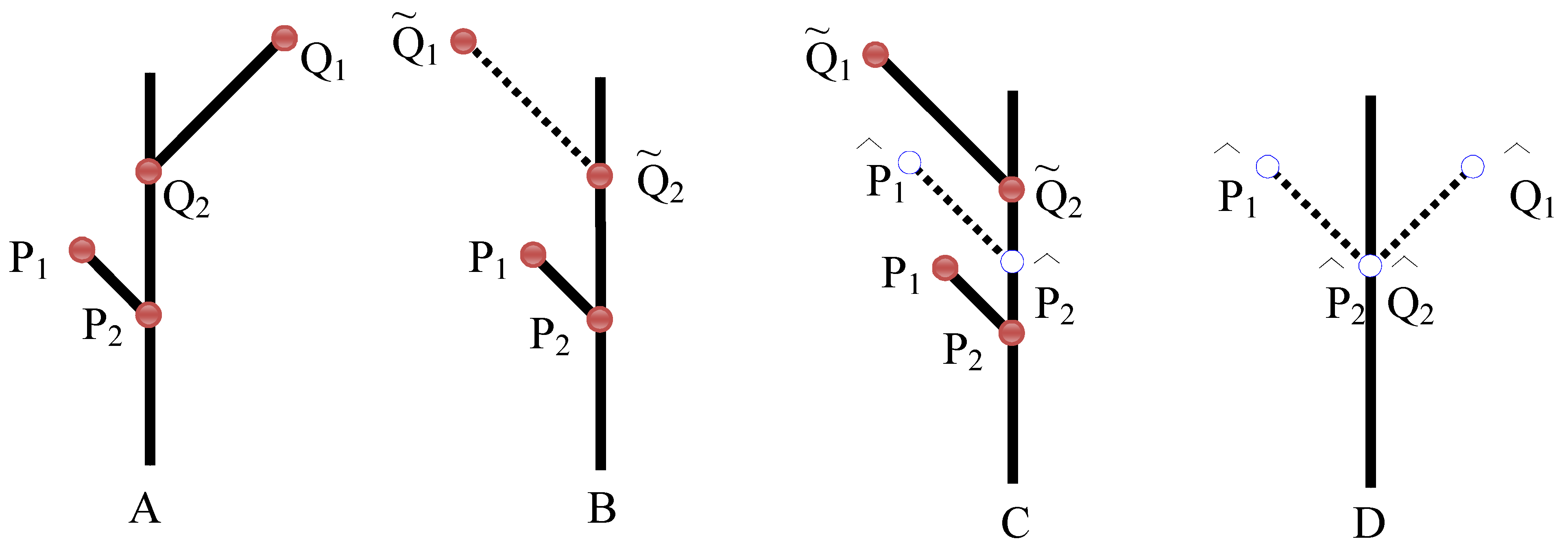
3.2. Landmark Methods for Shape Asymmetry
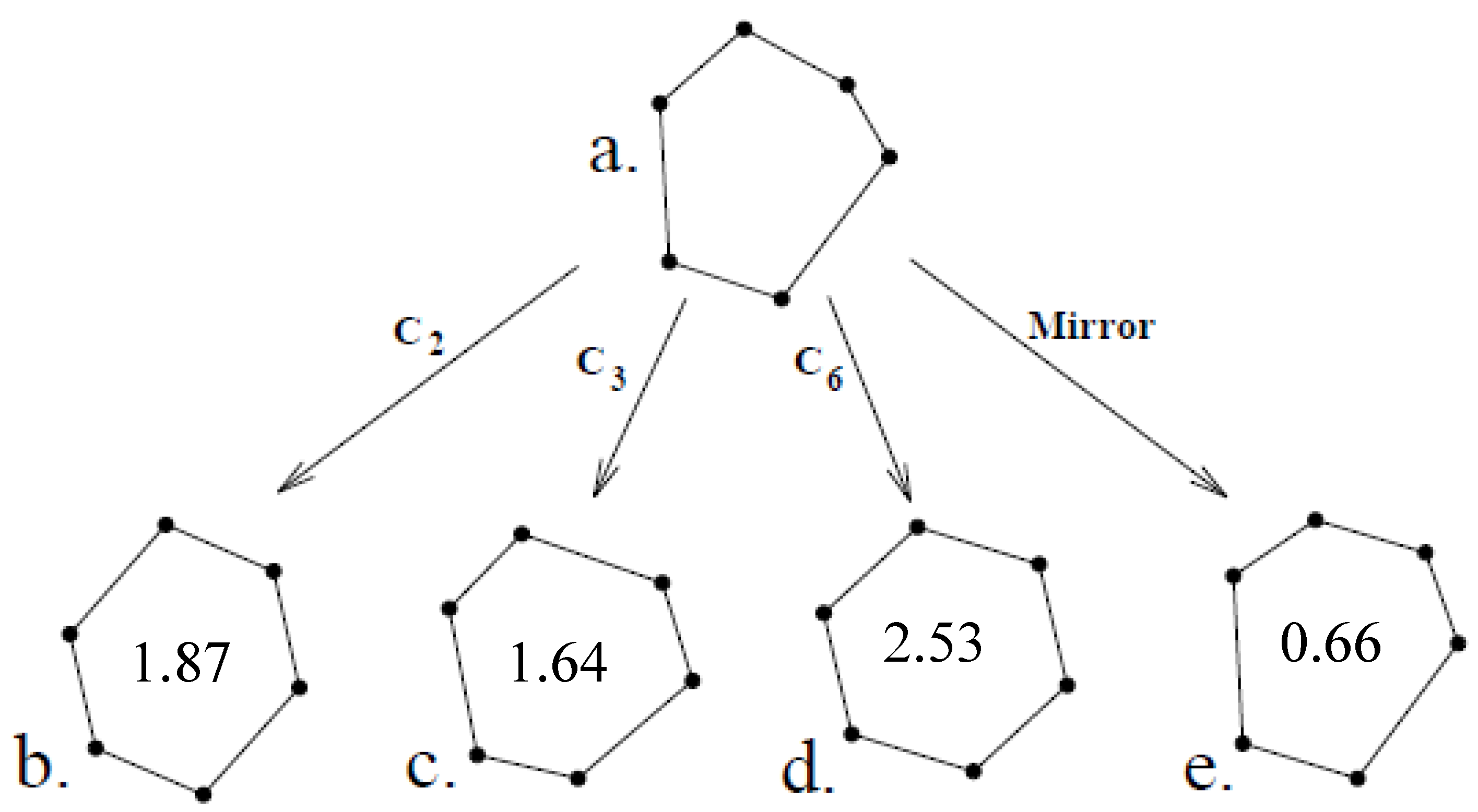
3.3. Continuous Symmetry Measures (CSM)
3.3.1. CSM in Physics and Chemistry
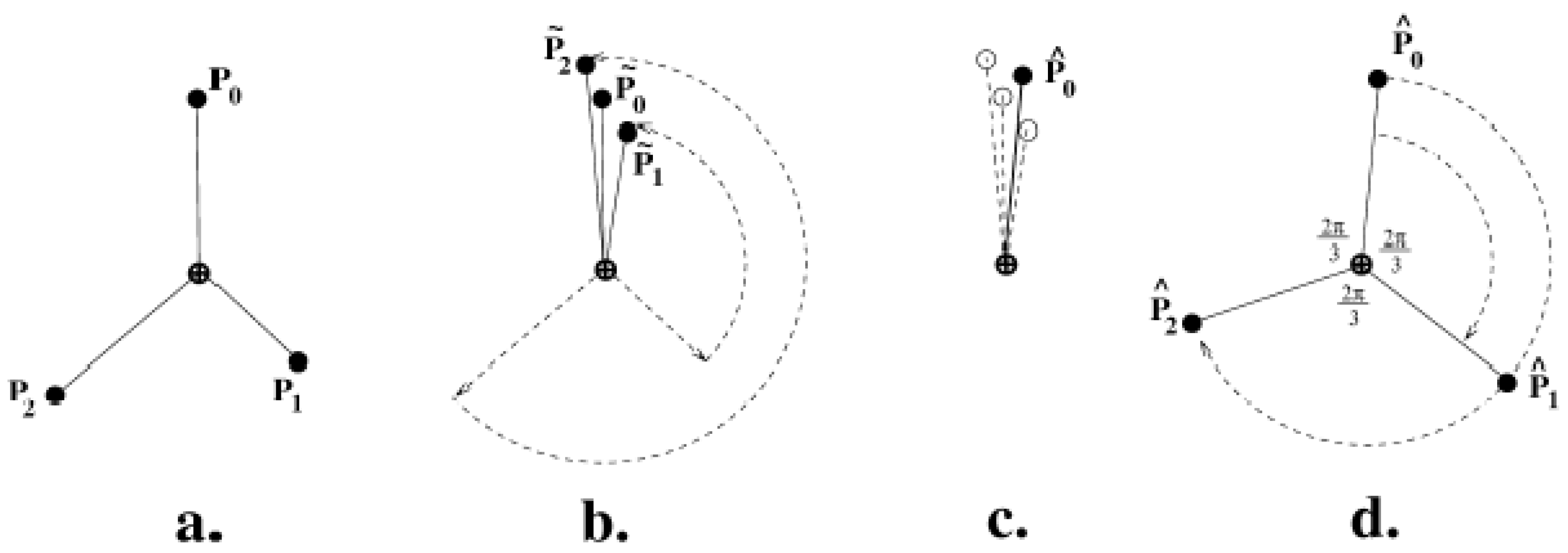
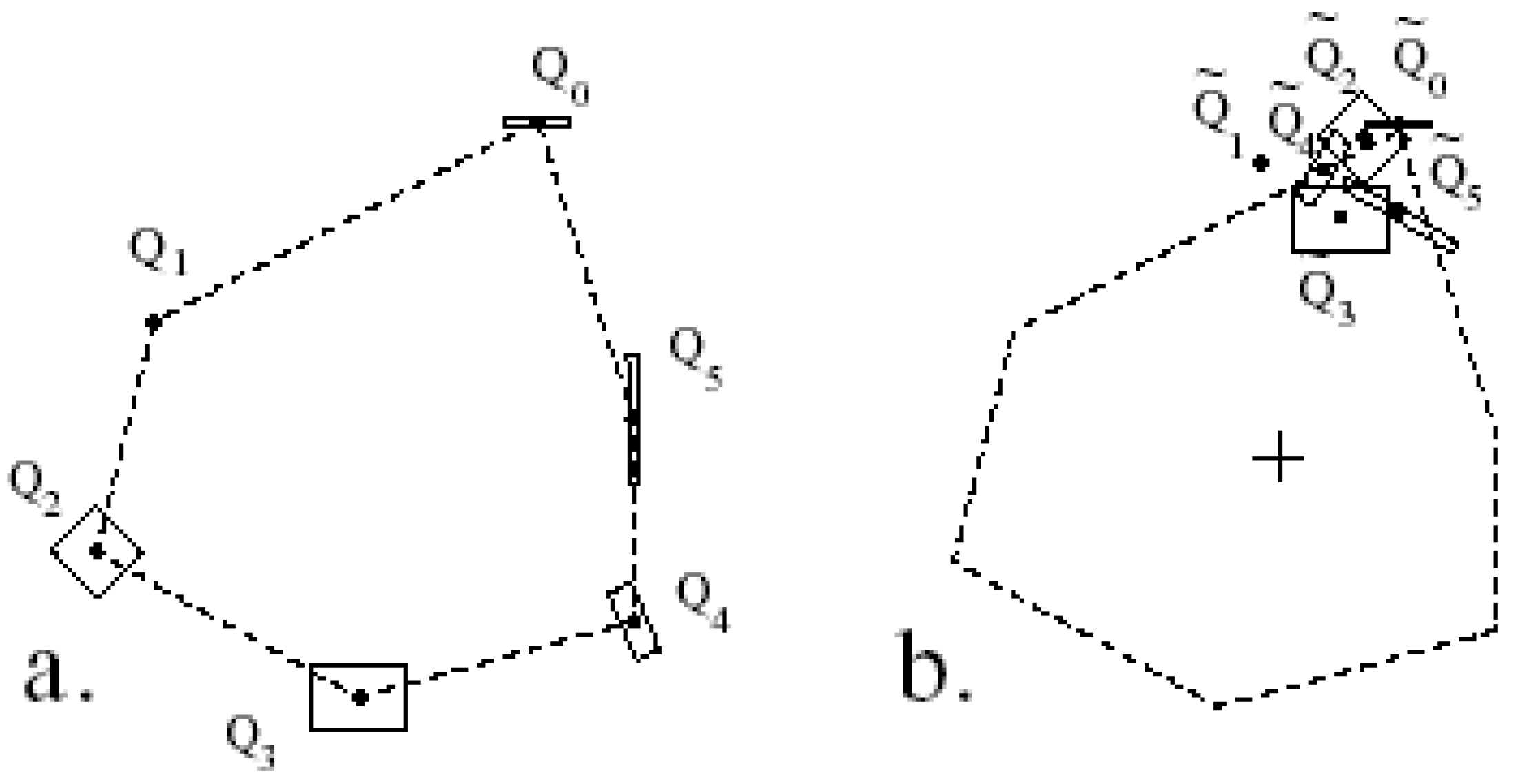
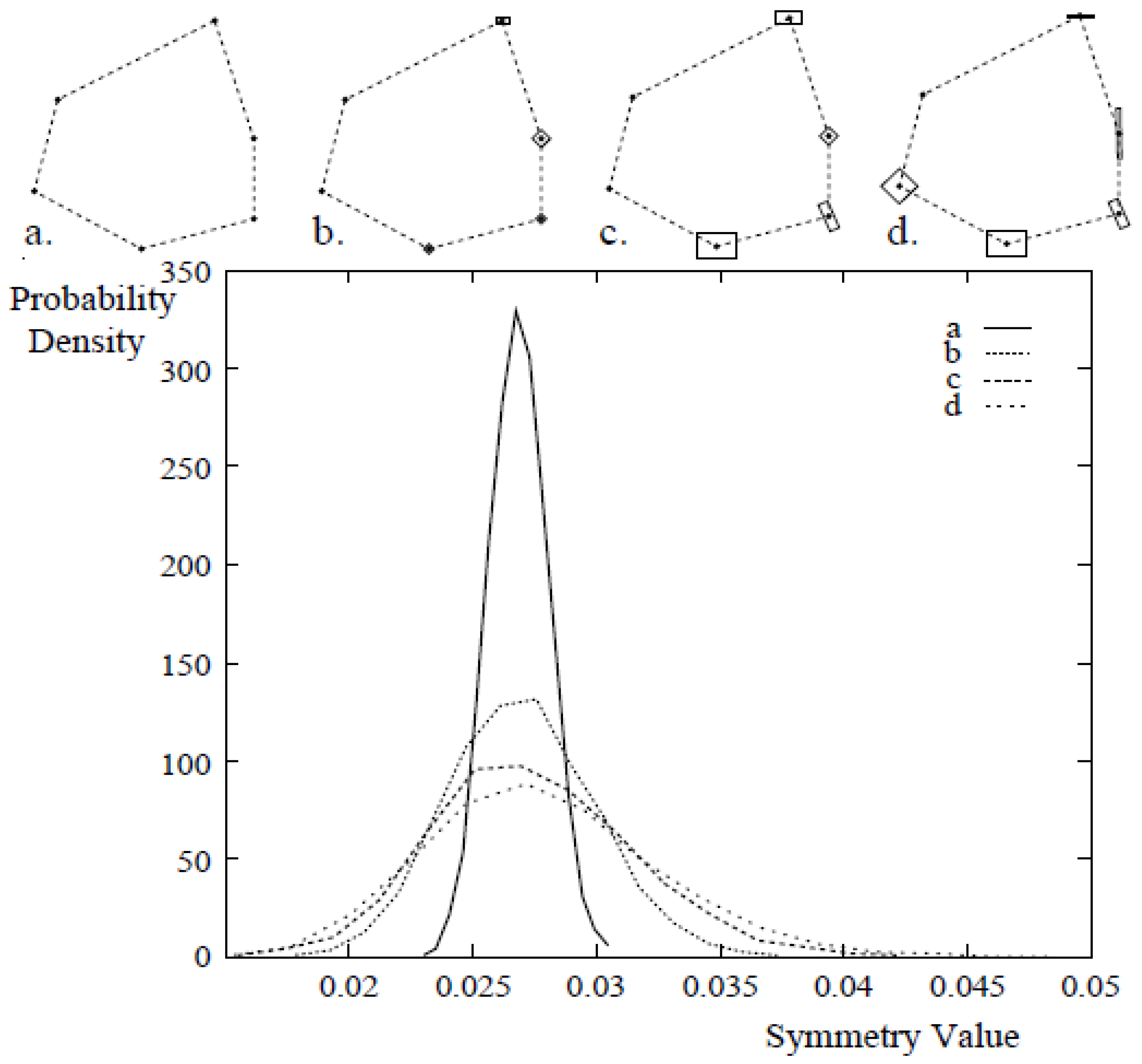
3.3.2. Measuring Asymmetry in the Absence of Landmarks
3.3.3. Analysis of Asymmetry Using Anchor Points
3.3.4. CSM-Developmental Stability Measurement
4. Measurement Error
4.1. Mixed-Model ANOVA
4.2. Fuzzy Analysis for Quantifying Measurement Error
5. Error Models and Size Scaling
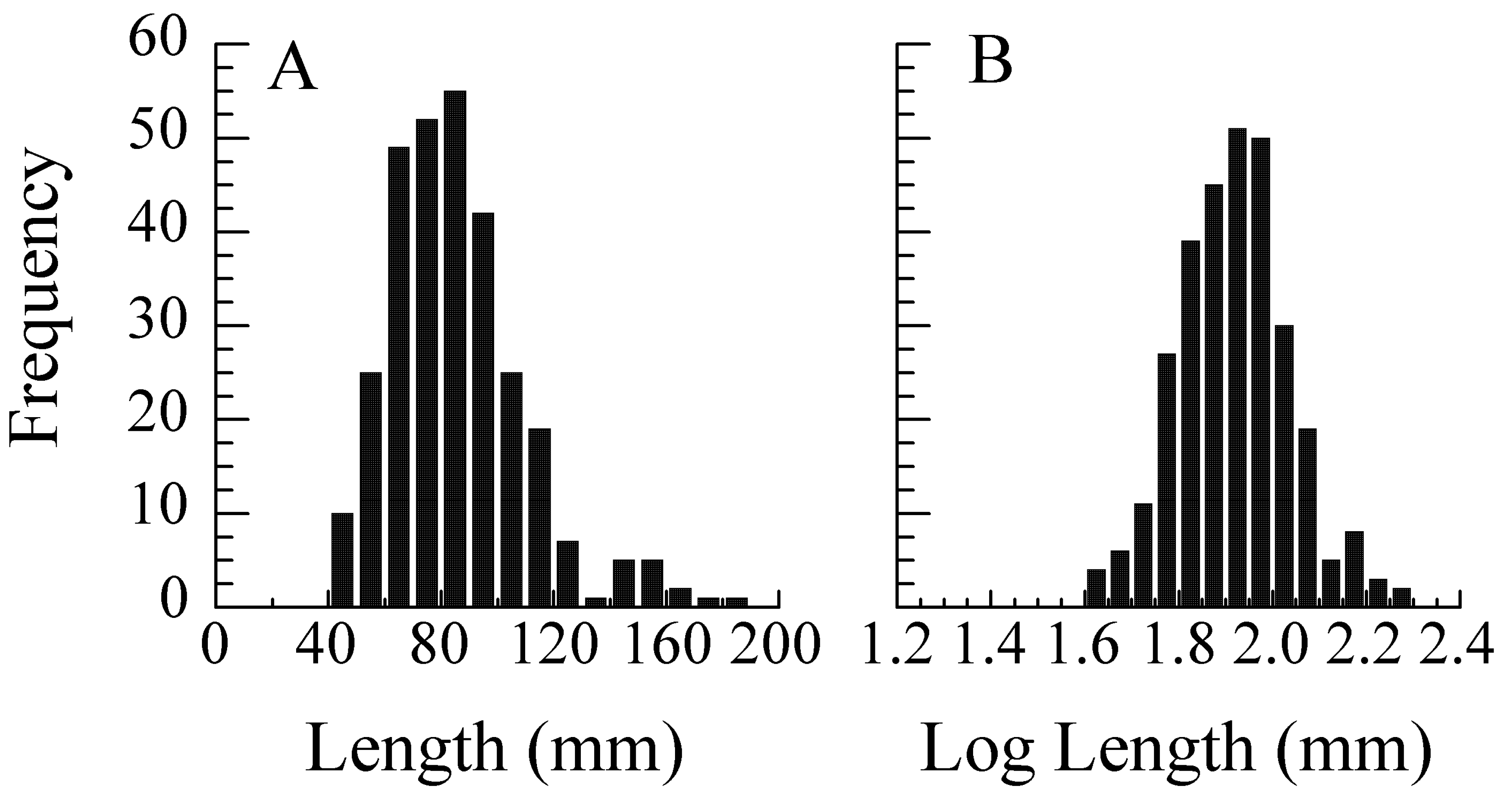
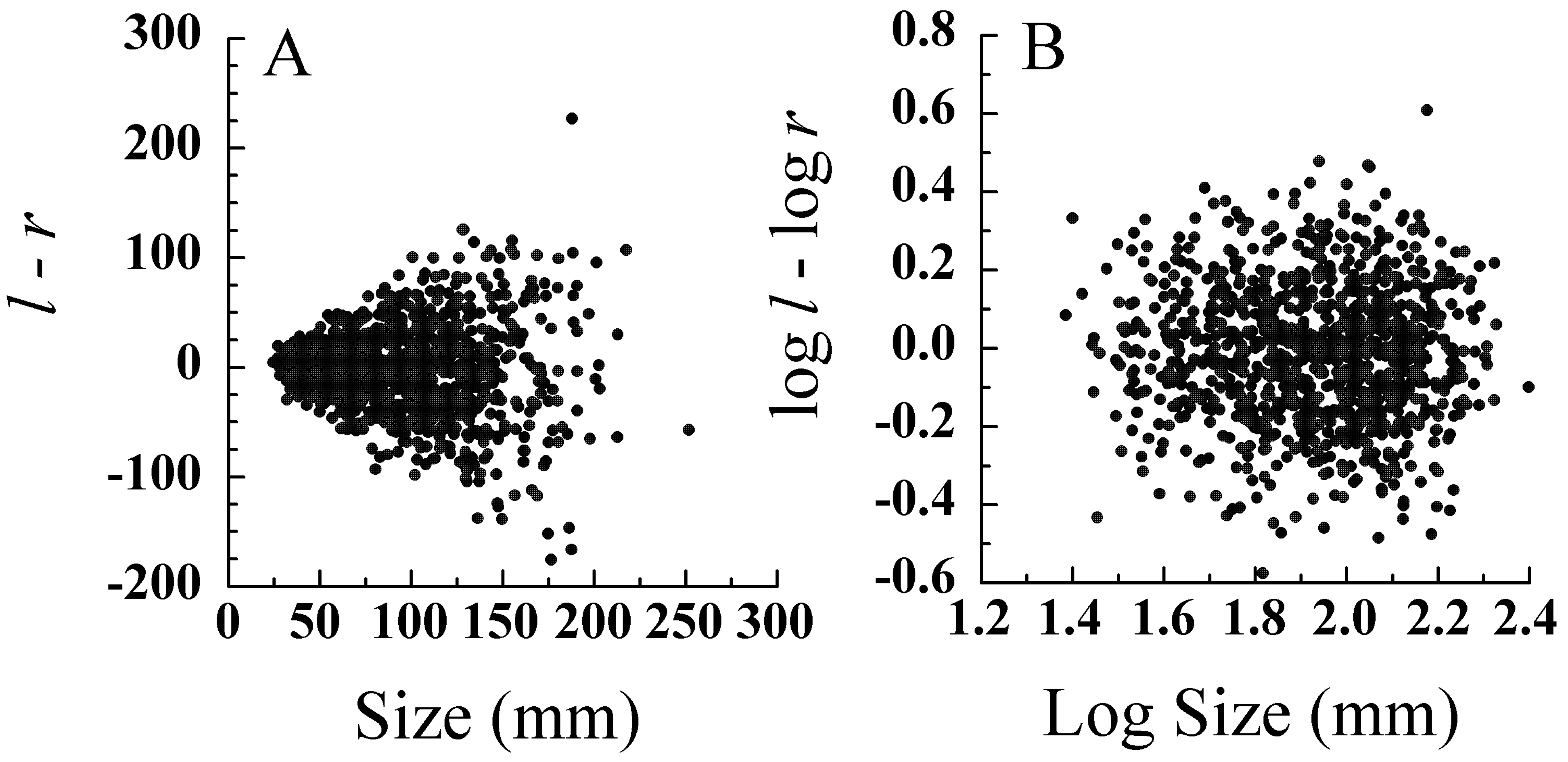
5.1. Error Models and Probability Distributions
5.2. Transformations for Size Scaling
5.3. Recommended Approaches
6. Developmental Homeostasis, Canalization, and Developmental Stability
7. Random Developmental Variation
7.1. Nature, Nurture, and Noise
7.2. Deterministic Chaos
7.3. Population and Individual Fluctuating Asymmetry
8. Origins of Developmental Homeostasis
8.1. Adaptation, Coadaptation, and Heterozygosity
8.2. Distributed Robustness
8.3. Environmental Stress
9. Applications
9.1. Stress
9.2. Fitness
9.3. Developmental Integration
9.4. Diverse Fluctuating Asymmetries
9.4.1. Fluctuating Rotational, Dihedral, and Radial Asymmetries
9.4.2. Fluctuating Translational Asymmetry
9.4.3. Fluctuating Helical Asymmetry
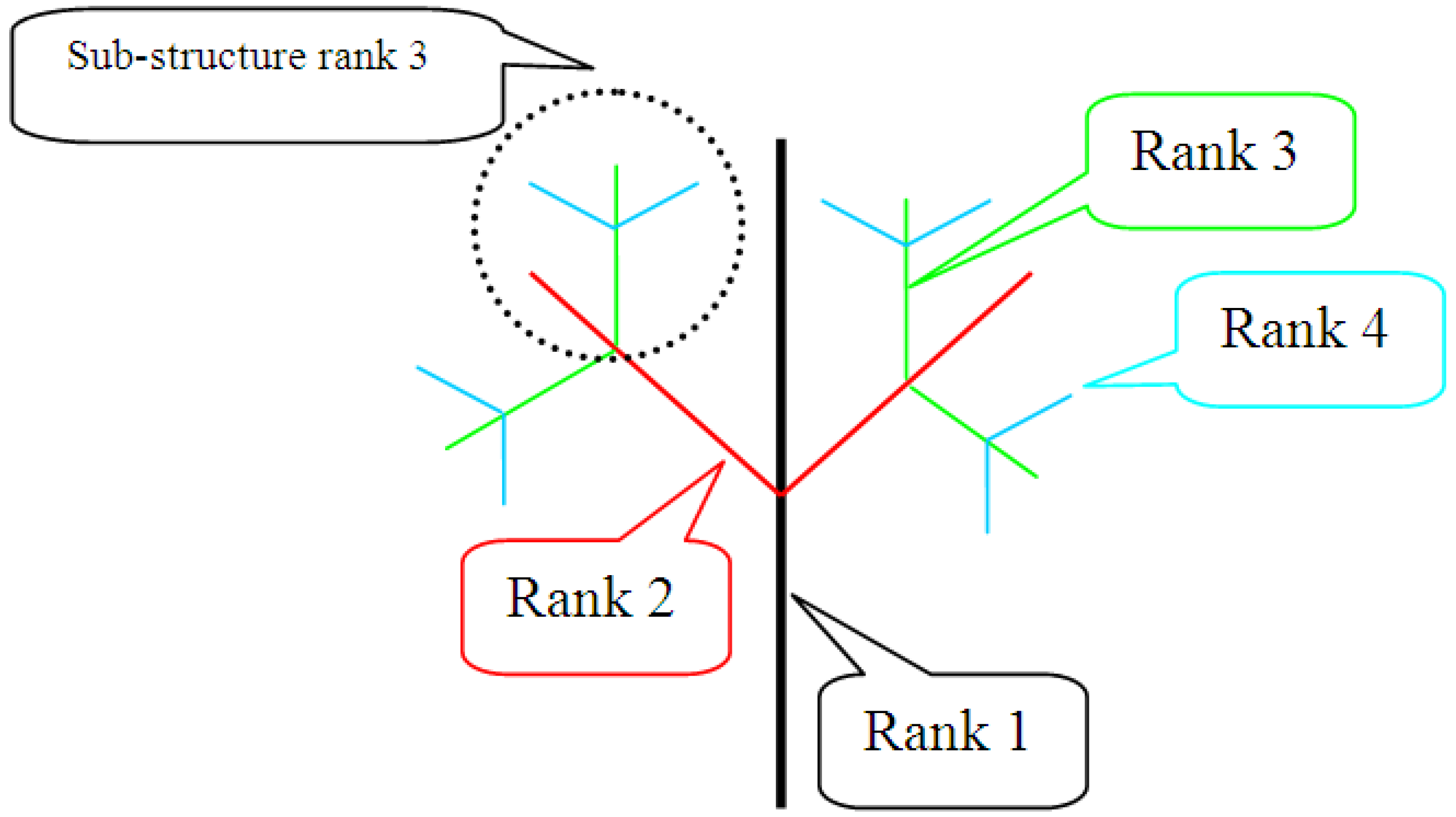
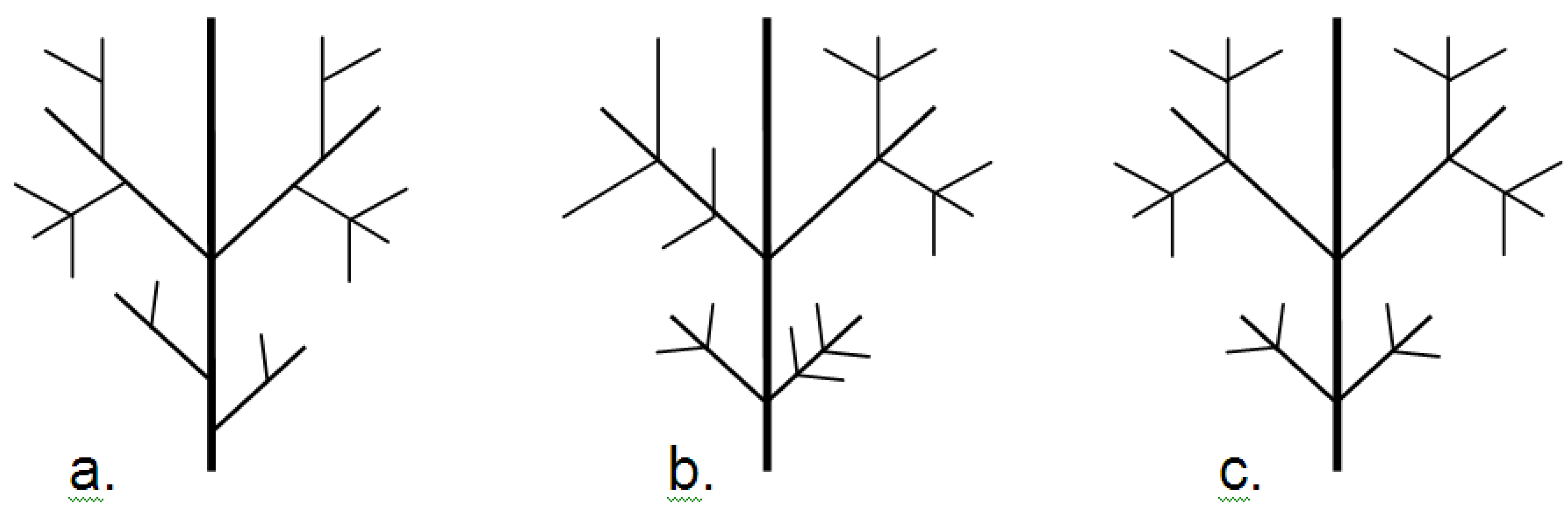
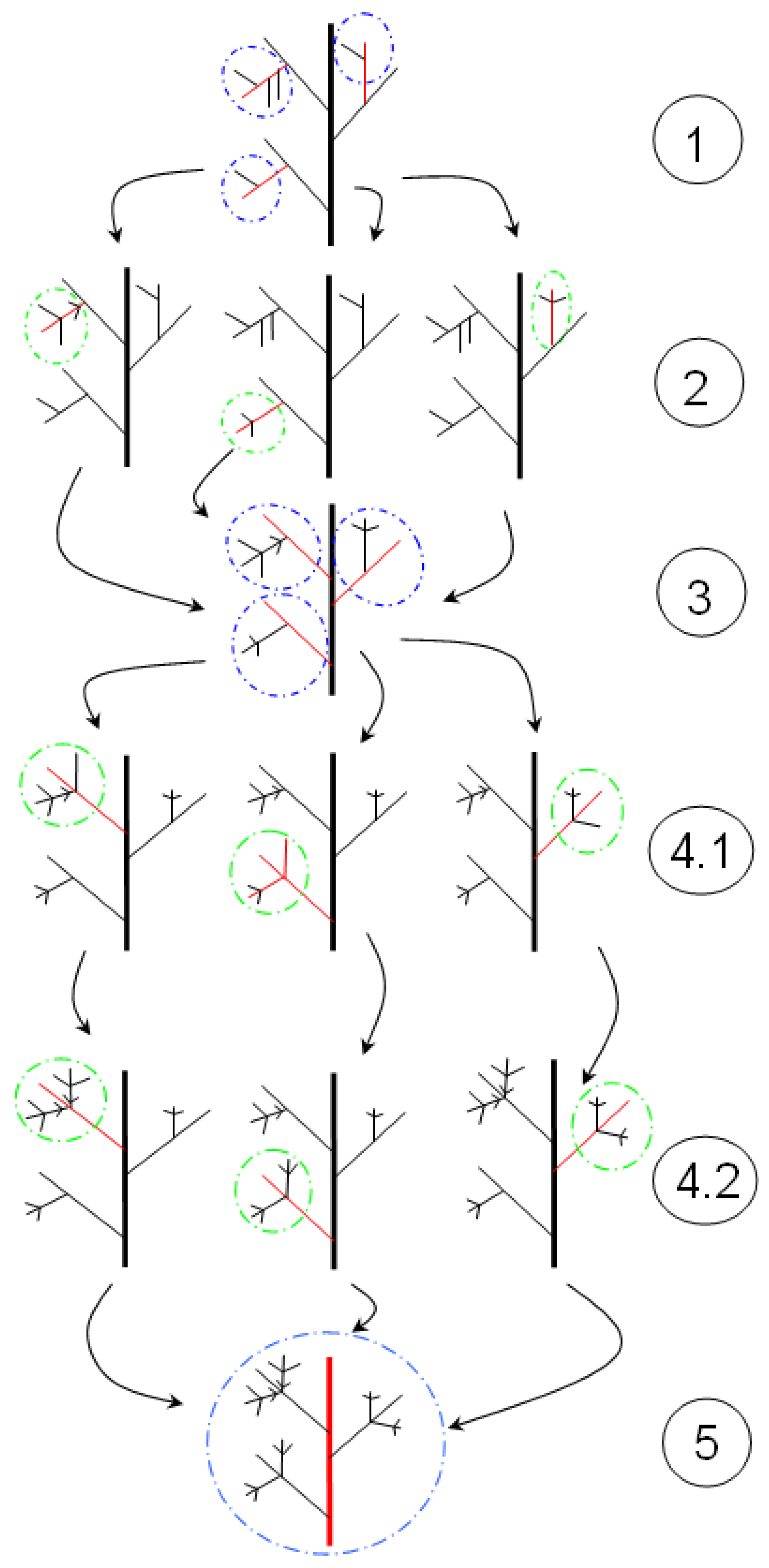
9.4.4. Fractal Dimension
9.5. Fluctuating Asymmetry and Developmental Instability of Diverse Taxonomic Groups
9.5.1. Viruses
9.5.2. Archaea and Eubacteria
9.5.3. Fungi
9.5.4. Plants
9.5.5. Animals
9.6. Fossil Material
9.7. Molecular Robustness
9.8. Specific Disciplines
9.8.1. Ecotoxicology
9.8.2. Conservation Biology
9.8.3. Anthropology and Evolutionary Psychology
9.8.4. Medicine and Public Health
9.8.5. Agriculture and Aquaculture
10. Fluctuating Asymmetry: Problems and Solutions
10.1. Inconsistencies among Studies of Fluctuating Asymmetry
10.2. The “Evolution Canyon” Microsites
10.3. Ongoing Research on Fluctuating Asymmetry at the “Evolution Canyon” Microsites
11. Conclusions
Acknowledgements
References
- Weyl, H. Symmetry; Princeton University Press: Princeton, NJ, USA, 1952. [Google Scholar]
- Abraham, E.R. The fractal branching of an arborescent sponge. Mar. Biol. 2001, 138, 503–510. [Google Scholar] [CrossRef]
- Astaurov, B.L. Analyse der erblichen Störungsfälle der bilateralen Symmetrie. Z. Indukt. Abstamm. Ver. 1930, 55, 183–262. [Google Scholar] [CrossRef]
- Ludwig, W. Das Rechts-Links Problem im Tierreich und beim Menschen; Springer: Berlin, Germany, 1932. [Google Scholar]
- Mather, K. Genetical control of stability in development. Heredity 1953, 7, 297–336. [Google Scholar] [CrossRef]
- Van Valen, L. A study of fluctuating asymmetry. Evolution 1962, 16, 125–142. [Google Scholar] [CrossRef]
- Soulé, M. Phenetics of natural populations. II. Asymmetry and evolution in a lizard. Am. Nat. 1967, 101, 141–160. [Google Scholar] [CrossRef]
- Jones, J.S. An asymmetrical view of fitness. Nature 1987, 325, 298–299. [Google Scholar] [CrossRef]
- Zakharov, V.M.; Graham, J.H. (Eds.) Developmental Stability in Natural Populations; Finnish Zoological Publishing Board: Helsinki, Finland, 1992. [Google Scholar]
- Markow, T.A. (Ed.) Developmental Instability: Its Origins and Evolutionary Implications; Kluwer: Dordrecht, The Netherlands, 1994. [Google Scholar]
- Møller, A.P.; Swaddle, J.P. Asymmetry, Developmental Stability, and Evolution; Oxford University Press: Oxford, UK, 1997. [Google Scholar]
- Polak, M. (Ed.) Developmental Instability: Causes and Consequences; Oxford University Press: New York, NY, USA, 2003. [Google Scholar]
- Zakharov, V.M. Future prospects for population phenogenetics. Sov. Sci. Rev. F, Physiol. Gen. Biol. Rev. 1989, 4, 1–79. [Google Scholar]
- Zakharov, V.M. Population phenogenetics: analysis of developmental stability in natural populations. Acta Zool. Fenn. 1992, 191, 7–30. [Google Scholar]
- Babcock, L.E. Asymmetry in the fossil record. Eur. Rev. 2005, 13, 135–143. [Google Scholar] [CrossRef]
- Costas, E.; Lopez-Rodas, V. Copper sulphate and DCMU-herbicide treatments increase asymmetry between sister cells in the toxic cyanobacteria Microcystis aeruginosa: implications for detecting environmental stress. Water Res. 2006, 40, 2447–2451. [Google Scholar] [CrossRef] [PubMed]
- Steven, A.C.; Trus, B.L.; Booy, F.P.; Cheng, N.; Zlotnick, A.; Caston, J.R.; Conway, J.F. The making and breaking of symmetry in virus capsid assembly: glimpses of capsid biology from cryoelectron microscopy. FASEB J. 1997, 11, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Danforth, C.H. Resemblance and difference in twins. J. Hered. 1919, 10, 399–409. [Google Scholar] [CrossRef]
- Sumner, F.B.; Huestis, R.R. Bilateral asymmetry and its relation to certain problems of genetics. Genetics 1921, 6, 445–485. [Google Scholar] [PubMed]
- Graham, J.H.; Freeman, D.C.; Emlen, J.M. Developmental stability: a sensitive indicator of populations under stress. In Environmental Toxicology and Risk Assessment; ASTM STP 1179; Landis, W.G., Hughes, J., Lewis, M.A., Eds.; American Society for Testing and Materials: Philadelphia, PA, USA, 1993; pp. 136–158. [Google Scholar]
- Klingenberg, C.P.; McIntyre, G.S. Geometric morphometrics of developmental instability: analyzing patterns of fluctuating asymmetry with procrustes methods. Evolution 1998, 52, 1363–1375. [Google Scholar] [CrossRef] [PubMed]
- Freeman, D.C.; Graham, J.H.; Emlen, J.M.; Tracy, M.; Hough, R.A.; Alados, C.L.; Escós, J. Plant developmental instability: new measures, applications, and regulation. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 367–386. [Google Scholar]
- Milner, D.; Hel-Or, H.; Keren, D.; Raz, S.; Nevo, E. Analyzing symmetry in biological systems. Proc. IEEE Int. Conf. Image Proc. 2005, 1, 361–364. [Google Scholar]
- Milner, D.; Raz, S.; Hel-Or, H.; Keren, D.; Nevo, E. A new measure of symmetry and its application to classification of bifurcating structures. Pattern Recogn. 2007, 40, 2237–2250. [Google Scholar] [CrossRef]
- Beloussov, L.V. The Dynamic Architecture of a Developing Organism; Kluwer: Dordrecht, The Netherlands, 1998. [Google Scholar]
- Zabrodsky, H.; Peleg, S.; Avnir, D. Symmetry as a continuous feature. IEEE T. Pattern Anal. 1995, 17, 1154–1166. [Google Scholar] [CrossRef]
- Beklemishev, V.N. Principles of Comparative Anatomy of Invertebrates; University of Chicago Press: Chicago, IL, USA, 1964. [Google Scholar]
- Grabowsky, G.L. Symmetry, locomotion, and the evolution of an anterior end: a lesson from sea urchins. Evolution 1994, 48, 1130–1146. [Google Scholar] [CrossRef]
- Thomas, A.L.R. The aerodynamic costs of asymmetry in the wings and tail of birds: asymmetric birds can’t fly round tight corners. Proc. R. Soc. Lond. B 1993, 254, 181–189. [Google Scholar]
- Brown, C.R.; Brown, M.B. Intense natural selection on body size and wing and tail asymmetry in Cliff Swallows during severe weather. Evolution 1998, 52, 1461–1475. [Google Scholar] [CrossRef]
- Finnerty, J.R. Did internal transport, rather than directed locomotion, favor the evolution of bilateral symmetry in animals? BioEssays 2005, 27, 1174–1180. [Google Scholar] [CrossRef] [PubMed]
- Davis, G.K.; Patel, N.H. The origin and evolution of segmentation. Trends Biochem. Sci. 1999, 24, M68–M72. [Google Scholar] [CrossRef]
- Couso, J.P. Segmentation, metamerism and the Cambrian explosion. Int. J. Dev. Biol. 2009, 53, 1305–1316. [Google Scholar] [CrossRef] [PubMed]
- Weibel, E.R. Fractal geometry: a design principle for living organisms. Am. J. Physiol. Lung Cell. Mol. Physiol. 1991, 261, L361–L369. [Google Scholar] [CrossRef] [PubMed]
- Enquist, B.J. Universal scaling in tree and vascular plant allometry: toward a general quantitative theory linking plant form and function from cells to ecosystems. Tree Physiol. 2002, 22, 1045–1064. [Google Scholar] [CrossRef] [PubMed]
- Farnsworth, K.D.; Niklas, K.J. Theories of optimization, form and function in branching architecture in plants. Funct. Ecol. 1995, 9, 355–363. [Google Scholar] [CrossRef]
- West, G.B.; Brown, J.H.; Enquist, B.J. The origin of universal scaling laws in biology. In Scaling in Biology; Brown, J.H., West, G.B., Eds.; Oxford University Press: New York, NY, USA, 2000. [Google Scholar]
- Banavar, J.R.; Maritan, A.; Rinaldo, A. Size and form in efficient transportation networks. Nature 1999, 399, 130–132. [Google Scholar] [CrossRef] [PubMed]
- Klingenberg, C.P.; Barluenga, M.; Meyer, A. Shape analysis of symmetric structures: quantifying variation among individuals and asymmetry. Evolution 2002, 56, 1909–1920. [Google Scholar] [CrossRef] [PubMed]
- Radwan, J.; Watson, P.J.; Farslow, J.; Thornhill, R. Procrustean analysis of fluctuating asymmetry in the bulb mite Rhizoglyphus robini Claparede (Astigmata: Acaridae). Biol. J. Linn. Soc. 2003, 80, 499–505. [Google Scholar] [CrossRef]
- Smith, D.R.; Crespi, B.J.; Bookstein, F.L. Fluctuating asymmetry in the honey bee, Apis mellifera: effects of ploidy and hybridization. J. Evol. Biol. 1997, 10, 551–574. [Google Scholar] [CrossRef]
- Auffray, J.-C.; Alibert, P.; Renaud, S.; Orth, A.; Bonhomme, F. Fluctuating asymmetry in Mus musculus subspecific hybridization: traditional and Procrustes comparative approach. In Advances in Morphometrics; Marcus, L.F., Corti, M., Loy, A., Naylor, G.J.P., Slice, D.E., Eds.; Plenum Press: New York, NY, USA, 1996; pp. 275–283. [Google Scholar]
- Møller, A.P.; Van Dongen, S. Ontogony of asymmetry and compensational growth in Elm Ulmus glabra leaves under different environmental conditions. Int. J. Plant. Sci. 2003, 164, 519–526. [Google Scholar] [CrossRef]
- Nagamitsu, T.; Kawahara, T.; Hotta, M. Phenotypic variation and leaf fluctuating asymmetry in isolated populations of an endangered dwarf birch Betula ovalifolia in Hokkaido, Japan. Plant Spec. Biol. 2004, 19, 13–21. [Google Scholar] [CrossRef]
- Kozlov, M.V.; Wilsey, B.J.; Koricheva, J.; Haukioja, E. Fluctuating asymmetry of birch leaves increases under pollution impact. J. Appl. Ecol. 1996, 33, 1489–1495. [Google Scholar] [CrossRef]
- Meissner, W.; Zolkos, K. What to divide leaves into ten parts for? The proposition of shape indices of leaf blade. Dendrobiology 2008, 60, 29–33. [Google Scholar]
- Sinclair, C.; Hoffmann, A.A. Monitoring salt stress in grapevines: are measures of plant trait variability useful? J. Appl. Ecol. 2003, 40, 928–937. [Google Scholar] [CrossRef]
- Palmer, A.R.; Strobeck, C. Fluctuating asymmetry: measurement, analysis, patterns. Annu. Rev. Ecol. Syst. 1986, 17, 391–421. [Google Scholar] [CrossRef]
- Palmer, A.R.; Strobeck, C. Fluctuating asymmetry analyses revisited. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 279–319. [Google Scholar]
- Graham, J.H.; Freeman, D.C.; Emlen, J.M. Antisymmetry, directional asymmetry, and dynamic morphogenesis. Genetica 1993, 89, 121–137. [Google Scholar] [CrossRef]
- Graham, J.H.; Emlen, J.M.; Freeman, D.C. Nonlinear dynamics and developmental instability. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 35–50. [Google Scholar]
- Graham, J.H.; Emlen, J.M.; Freeman, D.C.; Leamy, L.J.; Kieser, J. Directional asymmetry and the measurement of developmental instability. Biol. J. Linn. Soc. 1998, 64, 1–16. [Google Scholar] [CrossRef]
- Van Dongen, S.; Lens, L.; Molenberghs, G. Mixture analysis of asymmetry: modelling directional asymmetry, antisymmetry and heterogeneity in fluctuating asymmetry. Ecol. Lett. 1999, 2, 387–396. [Google Scholar]
- Swaddle, J.P.; Witter, M.S.; Cuthill, I.C. The analysis of fluctuating asymmetry. Anim. Behav. 1994, 48, 986–989. [Google Scholar] [CrossRef]
- Palmer, A.R. Fluctuating asymmetry analyses: a primer. In Developmental Instability: Its Origins and Evolutionary Implications; Markow, T.A., Ed.; Kluwer: Dordrecht, The Netherlands, 1994; pp. 335–364. [Google Scholar]
- Glasser, G.J. Variance formulas for the mean difference and coefficient of concentration. J. Am. Stat. Assoc. 1962, 57, 648–654. [Google Scholar] [CrossRef]
- Gorard, S. Revisiting a 90-year-old debate: the advantages of the mean deviation. Brit. J. Educ. Stud. 2005, 53, 417–430. [Google Scholar] [CrossRef]
- Levene, H. Robust tests for equality of variances. In Contributions to Probability and Statistics; Olkin, I., Ed.; Stanford University Press: Stanford, CA, USA, 1960; pp. 278–292. [Google Scholar]
- Palmer, A.R.; Strobeck, C. Fluctuating asymmetry as a measure of developmental stability: implications of non-normal distributions and power of statistical tests. Acta Zool. Fenn. 1992, 191, 57–72. [Google Scholar]
- Goldstein, D.G.; Taleb, N.N. We don’t quite know what we are talking about when we talk about volatility. J. Portfolio Manage. 2007, 33, 84–86. [Google Scholar] [CrossRef]
- Bookstein, F.L. Morphometric Tools for Landmark Data; Cambridge University Press: New York, NY, USA, 1991. [Google Scholar]
- Auffray, J.–C.; Debat, V.; Alibert, P. Shape asymmetry and developmental stability. In On Growth and Form: Spatio-temporal Pattern Formation in Biology; Chaplain, M.A.J., Singh, G.D., McLachlan, J.C., Eds.; John Wiley & Sons: London, UK, 1999; pp. 309–324. [Google Scholar]
- Mardia, K.V.; Bookstein, F.L.; Moreton, I.J. Statistical assessment of bilateral symmetry of shapes. Biometrika 2000, 87, 285–300. [Google Scholar] [CrossRef]
- Klingenberg, C.P.; Leamy, L.J. Quantitative genetics of geometric shape in the mouse mandible. Evolution 2001, 55, 2342–2352. [Google Scholar] [CrossRef]
- Zabrodsky, H.; Peleg, S.; Avnir, D. Continuous symmetry measures. J. Am. Chem. Soc. 1992, 114, 7843–7851. [Google Scholar] [CrossRef]
- Zabrodsky, H.; Peleg, S.; Avnir, D. Continuous symmetry for shapes. In Aspects of Visual Form Processing; Arcelli, C., Cordella, L., Sanniti di Baja, G., Eds.; World Scientific Publ.: Singapore, 1994; pp. 594–613. [Google Scholar]
- Rowland, H.A. The Physical Papers of Henry Augustus Rowland. Johns Hopkins Press: Baltimore, MD, USA, 1902; http://www.archive.org/details/physicalpapersof00rowlrich accessed on 18 March 2010.
- Zabrodsky, H.; Peleg, S.; Avnir, D. Continuous symmetry measures 2: Symmetry groups and the tetrahedron. J. Am. Chem. Soc. 1993, 115, 8278–8289. [Google Scholar] [CrossRef]
- Avnir, D.; Katzenelson, O.; Keinan, S.; Pinsky, M.; Pinto, Y.; Salomon, Y.; Hel-Or, H. The measurement of symmetry and chirality: conceptual aspects. In Concepts in Chemistry; Rouvray, D.H., Ed.; Research Studies Press: Somerset, UK, 1997; pp. 283–324. [Google Scholar]
- Zabrodsky, H.; Avnir, D. Continuous symmetry measures. 4. Chirality. J. Am. Chem. Soc. 1995, 117, 462–473. [Google Scholar] [CrossRef]
- Kanis, D.R.; Wong, J.S.; Marks, T.J.; Ratner, M.A.; Zabrodsky, H.; Keinan, S.; Avnir, D. Continuous symmetry analysis of hyperpolarizabilities. Characterization of second-order non-linear optical response of distorted benzene. J. Phys. Chem. 1995, 99, 11061–11066. [Google Scholar] [CrossRef]
- Alvarez, S.; Alemany, P.; Casanova, D.; Cirera, J.; Llunell, M.; Avnir, D. Shape maps and polyhedral interconversion paths in transition metal chemistry. Coordin. Chem. Rev. 2005, 249, 1693–1708. [Google Scholar] [CrossRef]
- Pinsky, M.; Avnir, D. Continuous symmetry measures. 5. The classical polyhedra. Inorg. Chem. 1998, 37, 5575–5582. [Google Scholar] [CrossRef] [PubMed]
- Katzenelson, O.; Hel-Or, H.; Avnir, D. Chirality of large supramolecular structures. Chem. Eur. J. 1996, 2, 174–181. [Google Scholar] [CrossRef]
- Yogev-Einot, D.; Avnir, D. The temperature-dependent optical activity of quartz: from Le Châtelier to chirality measures. Tetrahedron-Asymmetr. 2006, 17, 2723–2725. [Google Scholar] [CrossRef]
- Steinberg, A.; Karni, M.; Avnir, D. Continuous symmetry analysis of NMR chemical shielding anisotropy. Chem. Eur. J. 2006, 12, 8534–8538. [Google Scholar] [CrossRef] [PubMed]
- Pinto, Y.; Fowler, P.W.; Mitchell, D.; Avnir, D. Continuous chirality analysis of model stone—Wales rearrangements in fullerenes. J. Phys. Chem. 1998, 102, 5776–5784. [Google Scholar] [CrossRef]
- Yogev-Einot, D.; Avnir, D. Pressure and temperature effects on the degree of symmetry and chirality of the molecular building blocks of low quartz. Acta Crystallogr. 2004, B60, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Keinan, S.; Avnir, D. Quantitative symmetry in structure-activity correlations: the near C2 symmetry of inhibitor/hiv-protease complexes. J. Am. Chem. Soc. 2000, 122, 4378–4384. [Google Scholar] [CrossRef]
- Keinan, S.; Avnir, D. Continuous symmetry analysis of tetrahedral/planar distortions. Copper chlorides and other AB4 species. Inorg. Chem. 2001, 40, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Frid, A.; Raz, S.; Hel-Or, H. Complete symmetry of multilevel bifurcating structures: merging global and local symmetry. Pattern Recogn. to be submitted for publication.
- Graham, J.H.; Shimizu, K.; Emlen, J.M.; Freeman, D.C.; Merkel, J. Growth models and the expected distribution of fluctuating asymmetry. Biol. J. Linn. Soc. 2003, 80, 57–65. [Google Scholar] [CrossRef]
- Leamy, L. Morphometric studies in inbred and hybrid house mice. V. Directional and fluctuating asymmetry. Am. Nat. 1984, 123, 579–593. [Google Scholar] [CrossRef]
- Samuels, M.L.; Casella, G.; McCabe, G.P. Interpreting blocks and random factors. J. Am. Stat. Assoc. 1991, 86, 798–808. [Google Scholar] [CrossRef]
- Samuels, M.L.; Casella, G.; McCabe, G.P. Interpreting blocks and random factors: rejoinder. J. Am. Stat. Assoc. 1991, 86, 818–821. [Google Scholar] [CrossRef]
- Harville, D.A. Interpreting blocks and random factors: comment. J. Am. Stat. Assoc. 1991, 86, 812–815. [Google Scholar] [CrossRef]
- Hocking, R.R. Interpreting blocks and random factors: comment. J. Am. Stat. Assoc. 1991, 86, 811. [Google Scholar] [CrossRef]
- Speed, T.P. Interpreting blocks and random factors: comment. J. Am. Stat. Assoc. 1991, 86, 808–811. [Google Scholar] [CrossRef]
- Kozhara, A.V. On the ratio of components of phenotypic variances of bilateral characters in populations of some fishes. Genetika 1989, 25, 1508–1513. (in Russian). [Google Scholar]
- Kozhara, A.V. Phenotypic variance of bilateral characters as an indicator of genetic and environmental conditions in bream Abramis brama (L.) (Pisces, Cyprinidae) population. J. Appl. Ichthyol. 1994, 10, 167–181. [Google Scholar] [CrossRef]
- Bennington, C.C.; Thayne, W.V. Use and misuse of mixed model analysis of variance in ecological studies. Ecology 1994, 75, 717–722. [Google Scholar] [CrossRef]
- Kempthorne, O. Fixed and mixed models in the analysis of variance. Biometrics 1975, 31, 473–486. [Google Scholar] [CrossRef]
- Yoccoz, N.G. Use, overuse, and misuse of significance tests in evolutionary biology and ecology. Bull. Ecol. Soc. Am. 1991, 72, 106–111. [Google Scholar]
- Zabrodsky, H.; Peleg, S.; Avnir, D. Symmetry of fuzzy data. Proc. CVPR IEEE 1994, 1, 499–504. [Google Scholar]
- Mosimann, J.E.; Campbell, G. Applications in biology: simple growth models. In Lognormal Distributions: Theory and Applications; Crow, E.L., Shimizu, K., Eds.; Marcel Dekker: New York, NY, USA, 1988; pp. 287–302. [Google Scholar]
- Aitchison, J.; Brown, J.A.C. The Lognormal Distribution; Cambridge University Press: Cambridge, UK, 1963. [Google Scholar]
- Crow, E.L.; Shimizu, K. (Eds.) Lognormal Distributions: Theory and Applications; Marcel Dekker: New York, NY, USA, 1988. [Google Scholar]
- Van Dongen, S.; Møller, A.P. On the distribution of developmental errors: comparing the normal, gamma, and log-normal distribution. Biol. J. Linn. Soc. 2007, 92, 197–210. [Google Scholar] [CrossRef]
- Cowart, N.M.; Graham, J.H. Within- and among-individual variation in fluctuating asymmetry of leaves in the fig (Ficus carica L.). Int. J. Plant Sci. 1999, 160, 116–121. [Google Scholar] [CrossRef]
- Freeman, D.C.; Brown, M.L.; Dobson, M.; Jordan, Y.; Kizy, A.; Micallef, C.; Hancock, L.C.; Graham, J.H.; Emlen, J.M. Developmental instability: measures of resistance and resilience using pumpkin (Cucurbita pepo). Biol. J. Linn. Soc. 2003, 78, 27–41. [Google Scholar] [CrossRef]
- Raz, S.; Graham, J.H.; Hel-Or, H.; Nevo, E. Developmental instability of vascular plants in contrasting microclimates at “Evolution Canyon.”. To be submitted for publication.
- Leung, B.; Forbes, M.R.; Houle, D. Fluctuating asymmetry as a bioindicator of stress: comparing efficacy of analyses involving multiple traits. Am. Nat. 2000, 155, 101–115. [Google Scholar] [CrossRef] [PubMed]
- Sherry, R.A.; Lord, E.M. Developmental stability in flowers of Clarkia tembloriensis (Onagraceae). J. Evol. Biol. 1996, 9, 911–930. [Google Scholar] [CrossRef]
- Karvonen, E.; Merilä, J.; Rintamäki, P.T.; Van Dongen, S. Geography of fluctuating asymmetry in the greenfinch, Carduelis chloris. Oikos 2003, 100, 507–516. [Google Scholar] [CrossRef]
- Van Dongen, S. Fluctuating asymmetry and developmental instability in evolutionary biology: past, present and future. J. Evol. Biol. 2006, 19, 1727–1743. [Google Scholar] [CrossRef]
- Lajus, D.L. Variation patterns of bilateral characters: variation among characters and among populations in the White Sea herring, Clupea pallasi marisalbi (Berg) (Clupeidae, Teleosti). Biol. J. Linn. Soc. 2001, 74, 237–253. [Google Scholar]
- Young, J.R. Removing bias for fluctuating asymmetry in meristic characters. J. Agr. Biol. Envir. St. 2007, 12, 485–497. [Google Scholar] [CrossRef]
- Van Dongen, S.; Lens, L. Modelling developmental instability as the joint action of noise and stability: a Bayesian approach. BMC Evolutionary Biology 2002, 2, 11. Accessed at http://www.biomedcentral.com/1471-2148/2/11. [CrossRef]
- Hamdoun, A.; Epel, D. Embryo stability and vulnerability in an always changing world. Proc. Natl. Acad. Sci. USA 2007, 104, 1745–1750. [Google Scholar] [CrossRef] [PubMed]
- Waddington, C.H. The Strategy of the Genes; George Allen Unwin: London, UK, 1957. [Google Scholar]
- Schmalhausen, I.I. Factors of Evolution; Blakiston Company: Philadelphia, PA, USA, 1949. [Google Scholar]
- De Visser, J.A.G.M.; Hermisson, J.; Wagner, G.P.; Meyers, L.A.; Bagheri-Chaichian, H.; Blanchard, J.L.; Chao, L.; Cheverud, J.M.; Elena, S.F.; Fontana, W.; Gibson, G.; Hansen, T.F.; Krakauer, D.; Lewontin, R.C.; Ofria, C.; Rice, S.H.; Von Dassow, G.; Wagner, A.; Whitlock, M.C. Perspective: evolution and detection of genetic robustness. Evolution 2003, 57, 1959–1972. [Google Scholar] [CrossRef] [PubMed]
- Nijhout, H.F.; Davidowitz, G. Developmental perspectives on phenotypic variation, canalization, and fluctuating asymmetry. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 3–13. [Google Scholar]
- Waddington, C.H. Experiments on canalizing selection. Genet. Res. 1960, 1, 140–150. [Google Scholar] [CrossRef]
- Holling, C.S. Resilience and stability of ecological systems. Annu. Rev. Ecol. Syst. 1973, 4, 1–23. [Google Scholar] [CrossRef]
- Nicolis, G.; Prigogine, I. Exploring Complexity: An Introduction; Freeman: New York, NY, USA, 1989. [Google Scholar]
- Lajus, D.L.; Graham, J.H.; Kozhara, A.V. Developmental instabilty and the stochastic component of total phenotypic variance. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 343–363. [Google Scholar]
- Falconer, D.S.; MacKay, T.F.C. Introduction to Quantitative Genetics, 4th ed; Longman: Essex, UK, 1996. [Google Scholar]
- Gärtner, K. A third component causing random variability beside environment and genotype. A reason for the limited success of a 30 year long effort to standardize laboratory animals? Lab Animal 1990, 24, 71–77. [Google Scholar] [CrossRef]
- Pinker, S. The Blank Slate: The Modern Denial of Human Nature; Penguin Putnam: New York, NY, USA, 2002. [Google Scholar]
- Lajus, D.L.; Alekseev, V.R. Phenotypic variation and developmental instability of life-history traits: a theory and a case study on within-population variation of resting eggs formation in Daphnia. J. Limnol. 2004, 63, 37–44. [Google Scholar] [CrossRef]
- McAdams, H.H.; Arkin, A. Stochastic mechanisms in gene expression. Proc. Natl. Acad. Sci. USA 1997, 94, 814–819. [Google Scholar] [CrossRef]
- McAdams, H.H.; Arkin, A. It’s a noisy business: genetic regulation at the nanomolar scale. Trends Genet. 1999, 15, 65–69. [Google Scholar] [CrossRef]
- Raser, J.M.; O’Shea, E.K. Noise in gene expression: origins, consequences, and control. Science 2005, 309, 2010–2013. [Google Scholar] [CrossRef] [PubMed]
- Elowitz, M.B.; Levine, A.J.; Siggia, E.D.; Swain, P.S. Stochastic gene expression in a single cell. Science 2002, 297, 1183–1186. [Google Scholar] [CrossRef] [PubMed]
- Cukierman, E.; Pankov, R.; Yamada, K.M. Cell interactions with three-dimensional matrices. Curr. Opin. Cell Biol. 2002, 14, 633–639. [Google Scholar] [CrossRef]
- Pankov, R.; Endo, Y.; Even-Ram, S.; Araki, M.; Clark, K.; Cukierman, E.; Matsumoto, K.; Yamada, K.M. A Rac switch regulates random versus directionally persistent cell migration. J. Cell Biol. 2005, 170, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Crank, J. The Mathematics of Diffusion; Oxford University Press: New York, NY, USA, 1980. [Google Scholar]
- Eldar, A.; Dorfman, R.; Weiss, D.; Ashe, H.; Shilo, B.-Z.; Barkai, N. Robustness of the BMP morphogen gradient in Drosophila embryonic patterning. Nature 2002, 419, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Kan, K.-J.; Ploeger, A.; Raijmakers, M.E.J.; Dolan, C.V.; van der Maas, H.L.J. Nonlinear epigenetic variance: review and simulations. Developmental Sci. 2010, 13, 11–27. [Google Scholar] [CrossRef]
- Bratsun, D.; Volfson, D.; Tsimring, L.S.; Hasty, J. Delay-induced stochastic oscillations in gene regulation. Proc. Natl. Acad. Sci. USA 2005, 102, 14593–14598. [Google Scholar] [CrossRef] [PubMed]
- Helsen, P.; Van Dongen, S. The normal distribution as appropriate model of developmental instability in Opuntia cacti flowers. J. Evol. Biol. 2009, 22, 1346–1353. [Google Scholar] [CrossRef]
- Cannon, W.B. The Wisdom of the Body; Norton: New York, NY, USA, 1932. [Google Scholar]
- Lerner, I.M. Genetic Homeostasis; Oliver & Boyd: Edinburgh, UK, 1954. [Google Scholar]
- Calow, P. Homeostasis and fitness. Am. Nat. 1982, 120, 416–419. [Google Scholar] [CrossRef]
- Woolf, C.M.; Markow, T.A. Genetic models of developmental homeostasis: historical perspectives. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 99–115. [Google Scholar]
- Dobzhansky, Th. Genetics of natural populations. XIX. Origin of heterosis through natural selection in populations of Drosophila pseudoobscura. Genetics 1950, 35, 288–302. [Google Scholar] [PubMed]
- Dobzhansky, Th.; Levene, H. Genetics of natural populations. XXIV. Developmental homeostasis in natural populations of Drosophila pseudoobscura. Genetics 1955, 40, 797–808. [Google Scholar] [PubMed]
- Clarke, G.M. The genetic basis of developmental stability. I. Relationships between stability, heterozygosity and genomic coadaptation. Genetica 1993, 89, 15–23. [Google Scholar] [CrossRef]
- Clarke, G.M. Developmental stability-fitness relationships in animals: some theoretical considerations. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 187–195. [Google Scholar]
- Zachos, F.E.; Hartl, G.B.; Suchentrunk, F. Fluctuating asymmetry and genetic variability in the roe deer (Capreolus capreolus): a test of the developmental stability hypothesis in mammals using neutral molecular markers. Heredity 2007, 98, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Pogson, G.H.; Zouros, E. Allozyme and RFLP heterozygosities as correlates of growth rate in the scallop Placopecten magellanicus: a test of the associative overdominance hypothesis. Genetics 1994, 137, 221–231. [Google Scholar] [PubMed]
- Thelen, G.C.; Allendorf, F.W. Heterozygosity-fitness correlations in rainbow trout: effects of allozyme loci or associative overdominance? Evolution 2001, 55, 1180–1187. [Google Scholar] [CrossRef] [PubMed]
- Myrand, B.; Tremblay, R.; Sévigny, J.-M. Selection against blue mussels (Mytilus edulis L.) homozygotes under various stressful conditions. J. Hered. 2002, 93, 238–248. [Google Scholar] [CrossRef] [PubMed]
- Parsons, P.A. Environments and evolution: interactions between stress, resource inadequacy and energetic efficiency. Biol. Rev. 2005, 80, 589–610. [Google Scholar] [CrossRef]
- Koehn, R.K.; Bayne, B.L. Towards a physiological and genetical understanding of the energetics of the stress response. Biol. J. Linn. Soc. 1989, 37, 157–171. [Google Scholar] [CrossRef]
- Levene, H. Genetic equilibrium when more than one ecological niche is available. Am. Nat. 1953, 87, 331–333. [Google Scholar] [CrossRef]
- Nevo, E. Genetic variation in natural populations: patterns and theory. Theor. Pop. Biol. 1978, 13, 121–177. [Google Scholar] [CrossRef]
- Nevo, E. Genetic diversity in nature: patterns and theory. Evol. Biol. 1988, 23, 217–247. [Google Scholar]
- Nevo, E. Molecular evolution and ecological stress at global, regional, and local scales: the Israeli perspective. J. Exp. Zool. 1998, 282, 95–119. [Google Scholar] [CrossRef]
- Nevo, E. Evolution in action across life at “Evolution Canyons”, Israel. Trends Evol. Biol. 2009, 1, e3. [Google Scholar] [CrossRef]
- Nevo, E.; Beiles, A. Genetic parallelism of protein polymorphism in nature: ecological test of the neutral theory of molecular evolution. Biol. J. Linn. Soc. 1988, 35, 229–245. [Google Scholar] [CrossRef]
- Nevo, E.; Beiles, A.; Ben-Shlomo, R. The evolutionary significance of genetic diversity: ecological, demographic and life history correlates. In Lecture Notes in Biomathematics, Vol. 53, Evolutionary Dynamics of Genetic Diversity; Mani, G.S., Ed.; Springer-Verlag: Berlin, Germany, 1984; pp. 13–213. [Google Scholar]
- Graham, J.H.; Felley, J.D. Genomic coadaptation and developmental stability within introgressed populations of Enneacanthus gloriosus and E. obesus (Pisces: Centrarchidae). Evolution 1985, 39, 104–114. [Google Scholar] [PubMed]
- Leary, R.F.; Allendorf, F.W.; Knudsen, K.L. Developmental instability and high meristic counts in interspecific hybrids of salmonid fishes. Evolution 1985, 39, 1318–1326. [Google Scholar] [CrossRef]
- Graham, J.H. Genomic coadaptation and developmental stability in hybrid zones. Acta Zool. Fenn. 1992, 191, 121–131. [Google Scholar]
- Alibert, P.; Auffray, J.-C. Genomic coadaptation, outbreeding depression, and developmental instability. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 116–134. [Google Scholar]
- Leamy, L.J.; Klingenberg, C.P. The genetics and evolution of fluctuating asymmetry. Annu. Rev. Ecol. Evol. Syst. 2005, 36, 1–21. [Google Scholar] [CrossRef]
- Nevo, E. Speciation in action and adaptation in subterranean mole rats: patterns and theory. Boll. Zool. 1985, 52, 65–95. [Google Scholar] [CrossRef]
- Wang, H.; McArthur, E.D.; Sanderson, S.C.; Graham, J.H.; Freeman, D.C. Narrow hybrid zone between two subspecies of big sagebrush (Artemisia tridentata: Asteraceae). IV. Reciprocal transplant experiments. Evolution 1997, 51, 95–102. [Google Scholar]
- Tracy, M.; Freeman, D.C.; Duda, J.J.; Miglia, K.J.; Graham, J.H.; Hough, A. Developmental instability: an appropriate indicator of plant fitness components. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 196–212. [Google Scholar]
- Pélabon, C.; Hansen, T.F.; Carlson, M.L.; Armbruster, W.S. Patterns of asymmetry in the twining vine Dalechampia scandens (Euphorbiaceae): ontogenetic and hierarchical perspectives. New Phytol. 2006, 170, 65–74. [Google Scholar]
- Vishalakshi, C.; Singh, B.N. Effect of directional selection for body size on fluctuating asymmetry in certain morphological traits in Drosophila ananassae. J. Biosci. 2009, 34, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Soulé, M. The epistasis cycle: a theory of marginal populations. Annu. Rev. Syst. Ecol. 1973, 4, 165–187. [Google Scholar] [CrossRef]
- Kat, P.W. The relationship between heterozygosity for enzyme loci and developmental homeostasis in peripheral populations of aquatic bivalves (Unionidae). Am. Nat. 1982, 119, 824–832. [Google Scholar] [CrossRef]
- White, T.A.; Searle, J.B. Mandible asymmetry and genetic diversity in island populations of the common shrew, Sorex araneus. J. Evol. Biol. 2008, 21, 636–641. [Google Scholar] [CrossRef] [PubMed]
- Johnson, W.; Gangestad, S.W.; Segal, N.L.; Bouchard, T.J., Jr. Heritability of fluctuating asymmetry in a human twin sample: the effect of trait aggregation. Am. J. Hum. Biol. 2008, 20, 651–658. [Google Scholar] [CrossRef] [PubMed]
- Fuller, R.C.; Houle, D. Inheritance of developmental instability. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: Oxford, UK, 2003; pp. 157–186. [Google Scholar]
- Van Dongen, S. What do we know about the heritability of developmental instability? Answers from a Bayesian model. Evolution 2007, 61, 1033–1042. [Google Scholar] [CrossRef]
- Leamy, L.J.; Routman, E.J.; Cheverud, J.M. An epistatic genetic basis for fluctuating asymmetry of mandible size in mice. Evolution 2002, 56, 642–653. [Google Scholar] [CrossRef] [PubMed]
- Milton, C.C.; Huynh, B.; Batterham, P.; Rutherford, S.L.; Hoffmann, A.A. Quantitative trait symmetry independent of Hsp90 buffering: distinct modes of genetic canalization and developmental stability. Proc. Natl. Acad. Sci. USA 2003, 100, 13396–13401. [Google Scholar] [CrossRef] [PubMed]
- Milton, C.C.; Batterham, P.; McKenzie, J.A.; Hoffmann, A.A. Effect of E(sev) and Su(Raf) Hsp83 mutants and trans-heterozygotes on bristle trait means and variation in Drosophila melanogaster. Genetics 2005, 171, 119–130. [Google Scholar] [CrossRef]
- Debat, V.; Milton, C.C.; Rutherford, S.; Klingenberg, C.P.; Hoffmann, A.A. Hsp90 and the quantitative variation of wing shape in Drosophila melanogaster. Evolution 2006, 60, 2529–2538. [Google Scholar] [CrossRef] [PubMed]
- Sangster, T.A.; Salathia, N.; Undurraga, S.; Milo, R.; Schellenberg, K.; Lindquist, S.; Queitsch, C. HSP90 affects the expression of genetic variation and developmental stability in quantitative traits. Proc. Natl. Acad. Sci. USA 2008, 105, 2963–2968. [Google Scholar] [CrossRef]
- Santos, M.; Iriarte, P.F.; Céspedes, W. Genetics and geometry of canalization and developmental stability in Drosophila subobscura. BMC Evol. Biol. 2005, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Polak, M.; Taylor, P.W. A primary role of developmental instability in sexual selection. Proc. R. Soc. Lond. B 2007, 274, 3133–3140. [Google Scholar] [CrossRef] [PubMed]
- Félix, M.-A.; Wagner, A. Robustness and evolution: concepts, insights, and challenges from a developmental model system. Heredity 2008, 100, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A. Distributed robustness versus redundancy as causes of mutational robustness. Bioessays 2005, 27, 176–188. [Google Scholar] [CrossRef] [PubMed]
- Barabási, A.-L.; Albert, R. Emergence of scaling in random networks. Science 1999, 286, 509–512. [Google Scholar] [PubMed]
- Jeong, H.; Tombor, B.; Albert, R.; Oltvai, Z.N.; Barabási, A.-L. The large-scale organization of metabolic networks. Nature 2000, 407, 651–654. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.; Mason, S.P.; Barabási, A.-L.; Oltvai, Z.N. Lethality and centrality in protein networks. Nature 2001, 411, 41–42. [Google Scholar] [CrossRef]
- Barkai, N.; Leibler, S. Robustness in simple biochemical networks. Nature 1997, 387, 913–917. [Google Scholar] [CrossRef]
- Bhalla, U.S.; Iyengar, R. Emergent properties of networks of biological signaling pathways. Science 1999, 283, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Albert, R.; Jeong, H.; Barabási, A.-L. Error and attack tolerance of complex networks. Nature 2000, 406, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Newman, M.E.J.; Barabási, A.-L.; Watts, D.J. The Structure and Dynamics of Networks; Princeton University Press: Princeton, NJ, USA, 2006. [Google Scholar]
- Ravasz, E.; Somera, A.L.; Mongru, D.A.; Oltvai, Z.N.; Barabási, A.-L. Hierarchical organization of modularity in metabolic networks. Science 2002, 297, 1551–1555. [Google Scholar] [CrossRef] [PubMed]
- Levy, S.F.; Siegal, M.L. Network hubs buffer environmental variation in Saccharomyces cerevisiae. PLoS Biol. 2008, 6, e264. [Google Scholar] [CrossRef] [PubMed]
- Babbitt, G.A. Inbreeding reduces power-law scaling in the distribution of fluctuating asymmetry: an explanation of the basis of developmental instability. Heredity 2006, 97, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Babbitt, G.A. How accurate is the phenotype? – An analysis of developmental noise in a cotton aphid clone. BMC Dev. Biol. 2008, 8, 19. [Google Scholar] [CrossRef]
- Babbitt, G.A.; Kiltie, R.; Bolker, B. Are fluctuating asymmetry studies adequately sampled? Implications of a new model for size distribution. Am. Nat. 2006, 167, 230–245. [Google Scholar] [CrossRef]
- Alekseeva, T.A.; Zinichev, V.V.; Zotin, A.I. Energy criteria of reliability and stability of development. Acta Zool. Fenn. 1992, 191, 159–165. [Google Scholar]
- Hoffmann, A.A.; Parsons, P.A. Evolutionary Genetics and Environmental Stress; Oxford University Press: Oxford, UK, 1991. [Google Scholar]
- Parsons, P.A. Fluctuating asymmetry: an epigenetic measure of stress. Biol. Rev. 1990, 65, 131–145. [Google Scholar] [CrossRef]
- Parsons, P.A. Fluctuating asymmetry: a biological monitor of environmental and genomic stress. Heredity 1992, 68, 361–364. [Google Scholar] [CrossRef]
- Grime, J.P. Plant Strategies and Vegetation Processes; John Wiley & Sons: Chichester, UK, 1979. [Google Scholar]
- Escós, J.M.; Alados, C.L.; Pugnaire, F.I.; Puigdefábregas, J.; Emlen, J.M. Stress resistance strategy in arid land shrub: interaction between developmental instability and fractal dimension. J. Arid Environ. 2000, 45, 325–336. [Google Scholar] [CrossRef]
- Morgan, M.B.; Edge, S.E.; Snell, T.W. Profiling differential gene expression of corals along a transect of waters adjacent to the Bermuda municipal dump. Mar. Pollut. Bull. 2005, 51, 524–533. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, J.G.; Kristensen, T.N.; Loeschcke, V. The evolutionary and ecological role of heat shock proteins. Ecol. Lett. 2003, 6, 1025–1037. [Google Scholar] [CrossRef]
- Kawasaki, S.; Borchert, C.; Deyholos, M.; Wang, H.; Brazille, S.; Kawai, K.; Galbraith, D.; Bohnert, H.J. Gene expression profiles during the initial phase of salt stress in rice. Plant Cell 2001, 13, 889–905. [Google Scholar] [CrossRef] [PubMed]
- Bjorksten, T.; David, P.; Pomiankowski, A.; Fowler, K. Fluctuating asymmetry of sexual and nonsexual traits in stalk-eyed flies: a poor indicator of developmental stress and genetic quality. J. Evol. Biol. 2000, 13, 89–97. [Google Scholar] [CrossRef]
- Cárcamo, H.A.; Floate, K.D.; Lee, B.L.; Beres, B.L.; Clarke, F.R. Developmental instability in a stem-mining sawfly: can fluctuating asymmetry detect plant host stress in a model system? Oecologia 2008, 156, 505–513. [Google Scholar] [CrossRef]
- West-Eberhard, M.J. Developmental Plasticity and Evolution; Oxford University Press: New York, NY, USA, 2003. [Google Scholar]
- Losick, R.; Desplan, C. Stochasticity and cell fate. Science 2008, 320, 65–68. [Google Scholar] [CrossRef]
- Elena, S.F.; Sanjuán, R. Virus evolution: insights from an experimental approach. Annu. Rev. Ecol. Evol. Syst. 2007, 38, 27–52. [Google Scholar] [CrossRef]
- Black-Samuelsson, S.; Andersson, S. The effect of nutrient stress on developmental instability in leaves of Acer platanoides (Aceraceae) and Betula pendula (Betulaceae). Am. J. Bot. 2003, 90, 1107–1112. [Google Scholar] [CrossRef]
- Fair, J.M.; Breshears, D.D. Drought stress and fluctuating asymmetry in Quercus undulata leaves: confounding effects of absolute and relative amounts of stress? J. Arid Environ. 2005, 62, 235–249. [Google Scholar] [CrossRef]
- Zhu, J.-K. Salt and drought stress signal transduction in plants. Annu. Rev. Plant Biol. 2002, 53, 247–273. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.C. Natural selection, the costs of reproduction and a refinement of Lack’s principle. Am. Nat. 1966, 100, 687–690. [Google Scholar] [CrossRef]
- Stearns, S.C. The Evolution of Life Histories; Oxford University Press: New York, NY, USA, 1992. [Google Scholar]
- Charnov, E.L. The optimal balance between growth rate and survival in mammals. Evol. Ecol. Res. 2004, 6, 307–313. [Google Scholar] [CrossRef]
- Obeso, J.R. Tansley Review No. 139. The costs of reproduction in plants. New Phytol. 2002, 155, 321–348. [Google Scholar] [CrossRef]
- Freeman, D.C.; Graham, J.H.; Emlen, J.M. Developmental stability in plants: symmetries, stress and epigenesis. Genetica 1993, 89, 97–119. [Google Scholar] [CrossRef]
- Lappalainen, J.H.; Martel, J.; Lempa, K.; Wilsey, B.; Ossipov, V. Effects of resource availability on carbon allocation and developmental instability in cloned birch seedings. Int. J. Plant Sci. 2000, 161, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Milligan, J.R.; Krebs, R.A.; Mal, T.K. Separating developmental and environmental effects on fluctuating asymmetry in Lythrum salicaria and Penthorum sedoides. Int. J. Plant Sci. 2008, 169, 625–630. [Google Scholar] [CrossRef]
- Lovelock, C.E.; Ball, M.C.; Martin, K.C.; Feller, I.C. Nutrient enrichment increases mortality of mangroves. PLoS One 2009, 4, e5600. [Google Scholar] [CrossRef] [PubMed]
- Anne, P.; Mawri, F.; Gladstone, S.; Freeman, D.C. Is fluctuating asymmetry a reliable biomonitor of stress? A test using life history parameters in soybean. Int. J. Plant Sci. 1998, 159, 559–565. [Google Scholar] [CrossRef]
- Pélabon, C.; Hansen, T.F.; Carter, A.J.R.; Houle, D. Response of fluctuating and directional asymmetry to selection on wing shape in Drosophila melanogaster. J. Evol. Biol. 2006, 19, 764–776. [Google Scholar] [CrossRef]
- Martel, J.; Lempa, K.; Haukioja, E. Effects of stress and rapid growth on fluctuating asymmetry and insect damage in birch leaves. Oikos 1999, 8, 208–216. [Google Scholar] [CrossRef]
- Wells, J.C.; Hallal, P.C.; Manning, J.T.; Victora, C.G. A trade-off between early growth rate and fluctuating asymmetry in Brazilian boys. Ann. Hum. Biol. 2006, 33, 112–124. [Google Scholar] [CrossRef] [PubMed]
- Nevo, E.; Bolshakova, M.A.; Martyn, G.I.; Musatenko, L.I.; Sytnik, K.M.; Pavlíček, T.; Beharav, A. Drought and light anatomical adaptive leaf strategies in three woody species caused by microclimatic selection at “Evolution Canyon”, Israel. Isr. J. Plant Sci. 2000, 48, 33–46. [Google Scholar]
- Auffray, J.-C.; Renaud, S.; Alibert, P.; Nevo, E. Developmental stability and adaptive radiation in Spalax ehrenbergi superspecies in the Near East. J. Evol. Biol. 1999, 12, 207–221. [Google Scholar] [CrossRef]
- Hódar, J.A. Leaf fluctuating asymmetry of Holm oak in response to drought under contrasting climatic conditions. J. Arid Environ. 2002, 52, 233–243. [Google Scholar] [CrossRef]
- Souza, G.M.; Viana, J.O.F.; Oliveira, R.F. Asymmetrical leaves induced by water deficit show asymmetric photosynthesis in common bean. Braz. J. Plant Physiol. 2005, 17, 223–227. [Google Scholar] [CrossRef]
- Hochwender, C.G.; Fritz, R.S. Fluctuating asymmetry in a Salix hybrid system: the importance of genetic versus environmental causes. Evolution 1999, 53, 408–416. [Google Scholar] [CrossRef] [PubMed]
- Midgley, G.F.; Wand, S.J.E.; Musil, C.F. Repeated exposure to enhanced UV-B radiation in successive generations increases developmental instability (leaf fluctuating asymmetry) in a desert annual. Plant Cell Environ. 1998, 21, 437–442. [Google Scholar] [CrossRef]
- Breuker, C.J.; Brakefield, P.M. Heat shock in the developmentally sensitive period of butterfly eyespots fails to increase fluctuating asymmetry. Evol. Dev. 2003, 5, 231–239. [Google Scholar] [CrossRef]
- Hosken, D.J.; Blanckenhorn, W.U.; Ward, P.I. Developmental stability in yellow dung flies (Scathophaga stercoraria): fluctuating asymmetry, heterozygosity and environmental stress. J. Evol. Biol. 2000, 13, 919–926. [Google Scholar] [CrossRef]
- Hagen, S.B.; Ims, R.A.; Yoccoz, N.G.; Sørlibråten, O. Fluctuating asymmetry as an indicator of elevation stress and distribution limits in mountain birch (Betula pubescens). Plant Ecol. 2008, 195, 157–163. [Google Scholar] [CrossRef]
- Swaddle, J.P.; Witter, M.S. Food, feathers and fluctuating asymmetry. Proc. R. Soc. Lond. B 1994, 255, 147–152. [Google Scholar]
- Campero, M.; De Block, M.; Ollevier, F.; Stoks, R. Metamorphosis offsets the link between larval stress, adult asymmetry and individual quality. Funct. Ecol. 2008, 22, 271–277. [Google Scholar] [CrossRef]
- Hopton, M.E.; Cameron, G.N.; Cramer, M.J.; Polak, M.; Uetz, G.W. Live animal radiography to measure developmental instability in populations of small mammals after a natural disaster. Ecol. Indic. 2009, 9, 883–891. [Google Scholar] [CrossRef]
- Pertoldi, C.; Kristensen, T.N.; Andersen, D.H.; Loeschcke, V. Developmental instability as an estimator of genetic stress. Heredity 2006, 96, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Vøllestad, L.A.; Hindar, K.; Møller, A.P. Meta-analysis of fluctuating asymmetry in relation to heterozygosity. Heredity 1999, 83, 206–218. [Google Scholar] [CrossRef]
- Pélabon, C.; Carlson, M.L.; Hansen, T.F.; Armbruster, W.S. Effects of crossing distance on offspring fitness and developmental stability in Dalechampia scandens (Euphorbiaceae). Am. J. Bot. 2005, 92, 842–851. [Google Scholar] [CrossRef]
- Handy, S.M.; McBreen, K.; Cruzan, M.B. Patterns of fitness and fluctuating asymmetry across a broad hybrid zone. Int. J. Plant Sci. 2004, 165, 973–981. [Google Scholar] [CrossRef]
- Kark, S.; Safriel, U.N.; Tabarroni, C.; Randi, E. Relationship between heterozygosity and asymmetry: a test across the distribution range. Heredity 2001, 86, 119–127. [Google Scholar] [CrossRef][Green Version]
- Woodruff, R. (Bowling Green State University, Bowling Green, OH). Personal communication, 2009.
- Stige, L.C.; Slagsvold, T.; Vøllestad, L.A. Individual fluctuating asymmetry in Pied Flycatchers (Ficedula hypoleuca) persists across moults, but is not heritable and not related to fitness. Evol. Ecol. Res. 2005, 7, 381–406. [Google Scholar]
- Bartoš, L.; Bahbouh, R.; Vach, M. Repeatability of size and fluctuating asymmetry of antler characteristics in red deer (Cervus elaphus) during ontogeny. Biol. J. Linn. Soc. 2007, 91, 215–226. [Google Scholar] [CrossRef]
- Mateos, C.; Alarcos, S.; Carranza, J.; Sánchez-Prieto, C.B.; Valencia, J. Fluctuating asymmetry of red deer antlers negatively relates to individual condition and proximity to prime age. Anim. Behav. 2008, 75, 1629–1640. [Google Scholar] [CrossRef]
- Whitlock, M.C. The heritability of fluctuating asymmetry and the genetic control of developmental stability. Proc. R. Soc. Lond. B 1996, 263, 849–853. [Google Scholar]
- Whitlock, M.C. The repeatability of fluctuating asymmetry: a revision and extension. Proc. R. Soc. Lond. B 1998, 265, 1429–1431. [Google Scholar] [CrossRef]
- Van Dongen, S. How repeatable is the estimation of developmental stability by fluctuating asymmetry? Proc. R. Soc. Lond. B 1998, 265, 1423–1427. [Google Scholar] [CrossRef]
- Gangestad, W.; Thornhill, R. Fluctuating asymmetry, developmental instability, and fitness: toward model-based interpretation. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 62–80. [Google Scholar]
- Andalo, C.; Bazin, A.; Shykoff, J.A. Is there a genetic basis for fluctuating asymmetry in the plant Lotus corniculatus grown in different environmental conditions? Int. J. Plant Sci. 2000, 161, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Lens, L.; Van Dongen, S.; Kark, S.; Matthysen, E. Fluctuating asymmetry as an indicator of fitness: can we bridge the gap between studies? Biol. Rev. 2002, 77, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Leamy, L. Morphological integration of fluctuating asymmetry in the mouse mandible. Genetica 1993, 89, 139–153. [Google Scholar] [CrossRef]
- Klingenberg, C.P.; Zaklan, S.D. Morphological integration between developmental compartments in the Drosophila wing. Evolution 2000, 54, 1273–1285. [Google Scholar] [CrossRef]
- Hallgrímsson, B.; Willmore, K.; Hall, B.K. Canalization, developmental stability, and morphological integration in primate limbs. Yearb. Phys. Anthropol. 2002, 45, 131–158. [Google Scholar] [CrossRef]
- Klingenberg, C.P. Developmental instability as a research tool: using patterns of fluctuating asymmetry to infer the developmental origins of morphological integration. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 427–442. [Google Scholar]
- Klingenberg, C.P. Developmental constraints, modules and evolvability. In Variation; Hallgrímsson, B., Hall, B.K., Eds.; Academic Press: San Diego, CA, USA, 2005; pp. 219–247. [Google Scholar]
- Klingenberg, C.P. Morphological integration and developmental modularity. Annu. Rev. Ecol. Evol. Syst. 2008, 39, 115–132. [Google Scholar] [CrossRef]
- Klingenberg, C.P. Morphometric integration and modularity in configurations of landmarks: tools for evaluating a priori hypotheses. Evol. Dev. 2009, 11, 405–421. [Google Scholar] [CrossRef] [PubMed]
- Badyaev, A.V.; Foresman, K.R.; Young, R.L. Evolution of morphological integration: developmental accommodation of stress-induced variation. Am. Nat. 2005, 166, 382–395. [Google Scholar] [PubMed]
- Webster, M.; Zelditch, M.L. Integration and regulation of developmental systems in trilobites. In Advances in Trilobite Research, Rábano, I., Gozalo, R., García-Bellido, D., Eds.; Instituto Geológico y Minero de España: Madrid, Spain, 2008; pp. 427–433. [Google Scholar]
- Møller, A.P.; Eriksson, M. Patterns of fluctuating asymmetry in flowers: implications for sexual selection in plants. J. Evol. Biol. 1994, 7, 97–113. [Google Scholar] [CrossRef]
- Raz, S.; Gishkan, I.; Lefel, Y.; Hel-Or, H.; Graham, J.H.; Nevo, E. Analyzing fluctuating radial asymmetry. To be submitted for publication.
- Tracy, M.; Freeman, D.C.; Emlen, J.M.; Graham, J.H.; Hough, R.A. Developmental instability as a biomonitor of environmental stress: an illustration using plants and macroalgae. In Biomonitors and Biomarkers as Indicators of Environmental Change; Butterworth, F.M., Corkum, L.D., Guzmán-Rincón, J., Eds.; Plenum Press: New York, NY, USA, 1995; pp. 313–337. [Google Scholar]
- Paxman, G.J. Differentiation and stability in the development of Nicotiana rustica. Ann. Bot.-London 1956, 20, 331–347. [Google Scholar] [CrossRef]
- Zar, J.H. Biostatistical Analysis; Prentice-Hall: Englewood Cliffs, NJ, USA, 1984. [Google Scholar]
- Escós, J.; Alados, C.L.; Emlen, J.M. Impact of grazing on plant fractal architecture and fitness of a Mediterranean shrub Anthyllis cytisoides L. Funct. Ecol. 1997, 11, 66–78. [Google Scholar] [CrossRef]
- Alados, C.L.; Navarro, T.; Escós, J.; Cabezudo, B.; Emlen, J.M. Translational and fluctuating asymmetry as tools to detect stress in stress-adapted and nonadapted plants. Int. J. Plant Sci. 2001, 162, 607–616. [Google Scholar] [CrossRef]
- Emlen, J.M.; Freeman, D.C.; Graham, J.H. The adaptive basis of developmental instability: a hypothesis and its implications. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 51–61. [Google Scholar]
- Tan-Kristano, A.; Hoffmann, A.; Woods, R.; Batterham, P.; Cobbett, C.; Sinclair, C. Translational asymmetry as a sensitive indicator of cadmium stress in plants: a laboratory test with wild-type and mutant Arabidopsis thaliana. New Phytol. 2003, 159, 471–477. [Google Scholar] [CrossRef]
- Ambo-Rappe, R.; Lajus, D.L.; Schreider, M.J. Translational fluctuating asymmetry and leaf dimension in seagrass, Zostera capricorni Aschers in a gradient of heavy metals. Environ. Bioindic. 2007, 2, 99–116. [Google Scholar] [CrossRef]
- Ambo-Rappe, R.; Lajus, D.L.; Schreider, M.J. Increased heavy metal and nutrient contamination does not increase fluctuating asymmetry in the seagrass Halophila ovalis. Ecol. Indic. 2008, 8, 100–103. [Google Scholar] [CrossRef]
- Sinclair, C.; Hoffmann, A.A. Developmental stability as a potential tool in the early detection of salinity stress in wheat. Int. J. Plant Sci. 2003, 164, 325–331. [Google Scholar] [CrossRef]
- Mandelbrot, B.B. Fractals: Form, Chance, and Dimension; W. H. Freeman: San Francisco, CA, USA, 1977. [Google Scholar]
- Mandelbrot, B.B. The Fractal Geometry of Nature; W. H. Freeman: New York, NY, USA, 1982. [Google Scholar]
- Emlen, J.M.; Freeman, D.C.; Graham, J.H. Nonlinear growth dynamics and the origin of fluctuating asymmetry. Genetica 1993, 89, 77–96. [Google Scholar] [CrossRef]
- Goldberger, A.L.; Amaral, L.A.N.; Hausdorff, J.M.; Ivanov, P.C.; Peng, C.-K.; Stanley, H.E. Fractal dynamics in physiology: alterations with disease and aging. Proc. Natl. Acad. Sci. USA 2002, 99, 2466–2472. [Google Scholar] [CrossRef] [PubMed]
- Eghball, B.; Settimi, J.R.; Maranville, J.W. Fractal analysis for morphological description of corn roots under nitrogen stress. Agron. J. 1993, 85, 287–289. [Google Scholar] [CrossRef]
- Boxt, L.M.; Katz, J.; Liebovitch, L.S.; Jones, R.; Esser, P.D.; Reid, L. Fractal analysis of pulmonary arteries: the fractal dimension is lower in pulmonary hypertension. J. Thorac. Imag. 1994, 9, 8–13. [Google Scholar] [CrossRef]
- Alados, C.L.; Emlen, J.M.; Wachocki, B.; Freeman, D.C. Instability of development and fractal architecture in dryland plants as an index of grazing pressure. J. Arid Environ. 1998, 38, 63–76. [Google Scholar] [CrossRef]
- Alados, C.L.; Escós, J.; Emlen, J.M. Fluctuating asymmetry and fractal dimension of the sagittal suture as indicators of inbreeding depression in dama and dorcas gazelles. Can. J. Zool. 1995, 73, 1967–1974. [Google Scholar] [CrossRef]
- Escós, J.; Alados, C.L.; Emlen, J.M. Fractal structures and fractal functions as disease indicators. Oikos 1995, 74, 310–314. [Google Scholar] [CrossRef]
- Alados, C.L.; Escós, J.M.; Emlen, J.M. Fractal structure of sequential behaviour patterns: an indicator of stress. Anim. Behav. 1996, 51, 437–443. [Google Scholar] [CrossRef]
- Tan, C.O.; Cohen, M.A.; Eckberg, D.L.; Taylor, J.A. Fractal properties of human heart period variability: physiological and methodological implications. J. Physiol. 2009, 587, 3929–3941. [Google Scholar] [CrossRef]
- Di Ieva, A.; Grizzi, F.; Ceva-Grimaldi, G.; Russo, C.; Gaetani, P.; Aimar, E.; Levi, D.; Pisano, P.; Tancioni, F.; Nicola, G.; Tschabitscher, M.; Dioguardi, N.; Baena, R.R. Fractal dimension as a quantitator of the microvasculature of normal and adenomatous pituitary tissue. J. Anat. 2007, 211, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Schandorff, S. Developmental stability and skull lesions in the harbour seal (Phoca vitulina) in the 19th and 20th Centuries. Ann. Zool. Fenn. 1997, 34, 151–166. [Google Scholar]
- Górski, A.Z.; Skrzat, J. Error estimation of the fractal dimension measurements of cranial sutures. J. Anat. 2006, 208, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Mandelbrot, B.B. Intermittent turbulence in self-similar cascades: divergence of high moments and dimension of the carrier. J. Fluid. Mech. 1974, 62, 331–358. [Google Scholar] [CrossRef]
- Commoner, B.; Shearer, G.B.; Yamada, M. Linear biosynthesis of tobacco mosaic virus: changes in rod length during the course of infection. Proc. Natl. Acad. Sci. USA 1962, 48, 1788–1795. [Google Scholar] [CrossRef] [PubMed]
- Symington, J.; Commoner, B.; Yamada, M. Linear biosynthesis of tobacco mosaic virus: evidence that short virus rods are natural products of TMV biosynthesis. Proc. Natl. Acad. Sci USA 1962, 48, 1675–1683. [Google Scholar] [CrossRef] [PubMed]
- Galazka, A.; Milstien, J.; Zaffran, M. Thermostability of Vaccines; World Health Organization, Global Programme for Vaccines and Immunization: Geneva, Switzerland, 1998. [Google Scholar]
- Elena, S.F.; Carrasco, P.; Daròs, J.-A.; Sanjuán, R. Mechanisms of genetic robustness in RNA viruses. EMBO Rep. 2006, 7, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, J.A. Thinking about bacterial populations as multicellular organisms. Annu. Rev. Microbiol. 1998, 52, 81–104. [Google Scholar] [CrossRef]
- Ben-Jacob, E. Bacterial self-organization: co-enhancement of complexification and adaptability in a dynamic environment. Phil. Trans. R. Soc. Lond. A 2003, 361, 1283–1312. [Google Scholar] [CrossRef]
- Matsuyama, T.; Matsushita, M. Fractal morphogenesis by a bacterial cell population. Crit. Rev. Microbiol. 1993, 19, 117–135. [Google Scholar] [CrossRef]
- Singaravelan, N.; Grishkan, I.; Beharav, A.; Wakamatsu, K.; Ito, S.; Nevo, E. Adaptive melanin response of the soil fungus Aspergillus niger to UV radiation stress at ‘‘Evolution Canyon’’, Mount Carmel, Israel. PLoS One 2008, 3, e2993. [Google Scholar] [CrossRef] [PubMed]
- Bagchi, S.K.; Sharma, V.P.; Gupta, P.K. Developmental instability in leaves of Tectona grandis. Silvae Genet. 1989, 38, 1–6. [Google Scholar]
- Sakai, K.; Shimamoto, Y. Developmental instability in leaves and flowers of Nicotiana tabacum. Genetics 1965, 51, 801–813. [Google Scholar] [PubMed]
- Kozlov, M.V.; Niemelä, P. Difference in needle length—a new and objective indicator of pollution impact on scots pine (Pinus sylvestris). Water Air Soil Poll. 1999, 116, 365–370. [Google Scholar] [CrossRef]
- Palmer, R.A. Inferring relative levels of genetic variability in fossils: the link between heterozygosity and fluctuating asymmetry. Paleobiology 1986, 12, 1–5. [Google Scholar] [CrossRef]
- Smith, L.H. Asymmetry of early Paleozoic trilobites. Lethaia 1998, 31, 99–112. [Google Scholar] [CrossRef]
- Palmqvist, P.; Arribas, A.; Martinez-Navarro, B. Ecomorphological study of large canids from the lower Pleistocene of southeastern Spain. Lethaia 1999, 32, 75–88. [Google Scholar] [CrossRef]
- Szöllősi, G.J.; Derényi, I. Congruent evolution of genetic and environmental robustness in microRNA. Mol. Biol. Evol. 2009, 26, 867–874. [Google Scholar] [CrossRef] [PubMed]
- Borenstein, E.; Ruppin, E. Direct evolution of genetic robustness in microRNA. Proc. Natl. Acad. Sci. USA 2006, 103, 6593–6598. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, J.A.; Clarke, G.M. Diazinon resistance, fluctuating asymmetry and fitness in the Australian Sheep Blowfly, Lucilia cuprina. Genetics 1988, 140, 213–220. [Google Scholar]
- Cornelissen, T.; Stiling, P.; Drake, B. Elevated CO2 decreases leaf fluctuating asymmetry and herbivory by leaf miners on two oak species. Global Change Biol. 2003, 10, 27–36. [Google Scholar] [CrossRef]
- Graham, J.H.; Roe, K.; West, T.B. Effects of lead and benzene on developmental stability of Drosophila melanogaster. Ecotoxicology 1993, 2, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Polak, M.; Kroeger, D.E.; Cartwright, I.L.; Ponce deLeon, C. Genotype-specific responses of fluctuating asymmetry and of preadult survival to the effects of lead and temperature stress in Drosophila melanogaster. Environ. Pollut. 2004, 127, 145–155. [Google Scholar] [CrossRef]
- Pankakoski, E.; Koivisto, I.; Hyvärinen, H. Reduced developmental stability as an indicator of heavy metal pollution in the common shrew Sorex araneus. Acta Zool. Fenn. 1992, 191, 137–144. [Google Scholar]
- Estes, E.C.; Katholi, C.R.; Angus, R.A. Elevated fluctuating asymmetry in eastern mosquitofish (Gambusia holbrooki) from a river receiving paper mill effluent. Environ. Toxicol. Chem. 2006, 25, 1026–1033. [Google Scholar] [CrossRef] [PubMed]
- Hao, Z.; Xiangrong, W. Leaf developmental stability of Platanus acerifolia under urban environmental stress and its implication as an environmental indicator. Front. Biol. China 2006, 4, 411–417. [Google Scholar]
- Kozlov, M.V.; Niemelä, P.; Mälkönen, E. Effects of compensatory fertilization on pollution-induced stress in scots pine. Water Air Soil Poll. 2002, 134, 307–318. [Google Scholar] [CrossRef]
- Söderman, F.; Van Dongen, S.; Pakkasmaa, S.; Merilä, J. Environmental stress increases skeletal fluctuating asymmetry in the moor frog Rana arvalis. Oecologia 2007, 151, 593–604. [Google Scholar] [CrossRef]
- Allen, D.; Leamy, L.J. Effects of 2,3,4,8-tetrachloro-p-dioxin on directional and fluctuating asymmetry of mandible characters in mice. Ecotoxicology 2001, 10, 167–176. [Google Scholar] [CrossRef]
- Keller, J.M.; Allen, D.E.; Davis, C.R.; Leamy, L.J. 2,3,7,8-Tetrachlorodibenzo-p-dioxin affects fluctuating asymmetry of molar shape in mice, and an epistatic interaction of two genes for molar size. Heredity 2007, 98, 259–267. [Google Scholar] [CrossRef]
- Graham, J.H.; Fletcher, D.; Tigue, J.; McDonald, M. Growth and developmental stability of Drosophila melanogaster in low frequency magnetic fields. Bioelectromagnetics 2000, 21, 465–472. [Google Scholar] [CrossRef]
- Freeman, D.C.; Brown, M.; Duda, J.J.; Graham, J.H.; Emlen, J.H.; Krzysik, A.J.; Balbach, H.E.; Kovacic, D.A.; Zak, J.C. Photosynthesis and fluctuating asymmetry as indicators of plant response to soil disturbance in the fall-line sandhills of Georgia: a case study using Rhus copallinum and Ipomoea pandurata. Int. J. Plant Sci. 2004, 165, 805–816. [Google Scholar] [CrossRef]
- Freeman, D.C.; Brown, M.L.; Duda, J.J.; Graham, J.H.; Emlen, J.M.; Krzysik, A.J.; Balbach, H.; Kovacic, D.A.; Zak, J.C. Leaf fluctuating asymmetry, soil disturbance and plant stress: a multiple year comparison using two herbs, Ipomoea pandurata and Cnidoscolus stimulosus. Ecol. Indic. 2005, 5, 85–95. [Google Scholar] [CrossRef]
- Leary, R.F.; Allendorf, F.W. Fluctuating asymmetry as an indicator of stress: implications for conservation biology. Trends Ecol. Evol. 1989, 4, 214–217. [Google Scholar] [CrossRef]
- Lens, L.; Van Dongen, S.; Galbusera, P.; Schenck, T.; Matthysen, E.; Van de Casteele, T. Developmental instability and inbreeding in natural bird populations exposed to different levels of habitat disturbance. J. Evol. Biol. 2000, 13, 889–896. [Google Scholar] [CrossRef]
- Lens, L.; Van Dongen, S.; Matthysen, E. Fluctuating asymmetry as an early warning system in the critically endangered Taita Thrush. Conserv. Biol. 2002, 16, 479–487. [Google Scholar] [CrossRef]
- Teixeira, C.P.; Hirsch, A.; Perini, H.; Young, R.J. Marsupials from space: fluctuating asymmetry, geographical information systems and animal conservation. Proc. Biol. Sci. B 2006, 273, 1007–1012. [Google Scholar] [CrossRef]
- Perzigian, A.J. Fluctuating dental asymmetry: variation among skeletal populations. Am. J. Phys. Anthropol. 1977, 47, 81–88. [Google Scholar] [CrossRef]
- Teul, I.; Czerwiński, F.; Gawlikowska, A.; Konstanty-Kurkiewicz, V.; Sławiński, G. Asymmetry of the ovale and spinous foramina in mediaeval and contemporary skulls in radiological examinations. Folia Morphol. 2002, 61, 147–152. [Google Scholar]
- Gawlikowska, A.; Szczurowski, J.; Czerwiński, F.; Miklaszewska, D.; Adamiec, E.; Dzieciolowska, E. The fluctuating asymmetry of mediaeval and modern human skulls. Homo 2007, 58, 159–172. [Google Scholar] [CrossRef]
- Frederick, M.J.; Gallup, G.G., Jr. Fluctuating dental asymmetry in great apes, fossil hominins, and modern humans: implications for changing stressors during human evolution. Acta Psychologica Sinica 2007, 39, 489–494. [Google Scholar]
- DeLeon, V.B. Fluctuating asymmetry and stress in a Medieval Nubian population. Am. J. Phys. Anthropol. 2007, 132, 520–534. [Google Scholar] [CrossRef] [PubMed]
- Kegley, A.D.T.; Hemingway, J. Assessing fluctuating odontometric asymmetry among fossil hominin taxa through alternative measures of central tendency: effect of outliers and directional components on reported results. Homo 2007, 58, 33–52. [Google Scholar] [CrossRef] [PubMed]
- Kegley, A.D.T.; Hemingway, J. Surveying stress and developmental stability in Homo habilis via fluctuating odontometric asymmetry: assessing generic patterns and australopithecine differentiation. In Voyages in Science: Essays by South African Anatomists in Honour of Phillip V. Tobias’ 80th Birthday; Štrkalj, G., Pather, N., Kramer, B., Eds.; Content Solutions: Pretoria, South Africa, 2005; pp. 35–49. [Google Scholar]
- Brown, W.M.; Cronk, L.; Grochow, K.; Jacobson, A.; Liu, C.K.; Popović, Z.; Trivers, R. Dance reveals symmetry especially in young men. Nature 2005, 438, 1148–1150. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.S.; Hoffmann, H.L.; Mustanski, B.S. Fluctuating asymmetry and sexual orientation in men and women. Arch. Sex. Behav. 2008, 37, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Markow, T.A.; Wandler, K. Fluctuating dermatoglyphic asymmetry and the genetics of liability to schizophrenia. Psychiat. Res. 1986, 19, 323–328. [Google Scholar] [CrossRef]
- Mellor, C.S. Dermatoglyphic evidence of fluctuating asymmetry in schizophrenia. Brit. J. Psychiat. 1992, 160, 467–472. [Google Scholar] [CrossRef]
- Benderlioglu, Z.; Sciulli, P.W.; Nelson, R.J. Fluctuating asymmetry predicts human reactive aggression. Am. J. Hum. Biol. 2004, 16, 458–469. [Google Scholar] [CrossRef]
- Burton, C.; Stevenson, J.C.; Williams, D.C.; Everson, P.M.; Mahoney, E.R.; Trimble, J.E. Attention-deficit hyperactivity disorder (AD/HD) and fluctuating asymmetry (FA) in a college sample: an exploratory study. Am. J. Hum. Biol. 2003, 15, 601–619. [Google Scholar] [CrossRef]
- Stevenson, J.C.; Everson, P.M.; Williams, D.C.; Hipskind, G.; Mahoney, E.R.; Mehler, M.; Cawley, M.; Chamberlin, W.; Watts, L. Attention-deficit hyperactivity disorder and fluctuating asymmetry in another college sample. Am. J. Hum. Biol. 2006, 18, 402–414. [Google Scholar] [CrossRef]
- Saha, S.; Loesch, D.; Chant, D.; Welham, J.; El-Saadi, O.; Fañanás, L.; Mowry, B.; McGrath, J. Directional and fluctuating asymmetry in finger and a-b ridge counts in psychosis: a case-control study. BMC Psychiatry 2003, 3, 3. [Google Scholar] [CrossRef]
- Thornhill, R.; Møller, A.P. Developmental stability, disease and medicine. Biol. Rev. 1997, 72, 497–548. [Google Scholar] [CrossRef] [PubMed]
- Özener, B.; Fink, B. Facial symmetry in young girls and boys from a slum and a control area of Ankara, Turkey. Evol. Hum. Behav. submitted for publication.
- Özener, B.; Duyar, İ.; Atamtürk, D. Body composition of young laborers: the results of a bioelectrical impedance analysis. Collegium Antropol. 2007, 31, 949–954. [Google Scholar]
- Van Dongen, S.; Wijnaendts, L.C.D.; Ten Broek, C.M.A.; Galis, F. Flutuating asymmetry does not consistently reflect severe developmental disorders in human fetuses. Evolution 2009, 63, 1832–1844. [Google Scholar] [CrossRef] [PubMed]
- Weeden, J.; Sabini, J. Physical attractiveness and health in Western societies: a review. Psychol. Bull. 2005, 131, 635–653. [Google Scholar] [CrossRef] [PubMed]
- Knierim, U.; Van Dongen, S.; Forkman, B.; Tuyttens, F.A.M.; Špinka, M.; Campo, J.L.; Weissengruber, G.E. Fluctuating asymmetry as an animal welfare indicator—a review of methodology and validity. Physiol. Behav. 2007, 92, 398–421. [Google Scholar] [CrossRef] [PubMed]
- Almeida, D.; Almodóvar, A.; Nicola, G.G.; Elvira, B. Fluctuating asymmetry, abnormalities and parasitism as indicators of environmental stress in cultured stocks of goldfish and carp. Aquaculture 2008, 279, 120–125. [Google Scholar] [CrossRef]
- Fréchette, M.; Goulletquer, P.; Daigle, G. Fluctuating asymmetry and mortality in cultured oysters (Crassostrea gigas) in Marennes-Oléron basin. Aquat. Living Resour. 2003, 16, 339–346. [Google Scholar] [CrossRef]
- Van Poucke, E.; Van Nuffel, A.; Van Dongen, S.; Sonck, B.; Lens, L.; Tuyttens, F.A.M. Experimental stress does not increase fluctuating asymmetry of broiler chickens at slaughter age. Poult. Sci. 2007, 86, 2110–2116. [Google Scholar] [CrossRef]
- Hoffmann, A.A.; Woods, R.E. Associating stress with developmental stability: problems and patterns. In Developmental Instability: Causes and Consequences; Polak, M., Ed.; Oxford University Press: New York, NY, USA, 2003; pp. 387–401. [Google Scholar]
- Leung, B.; Forbes, M.R. Fluctuating asymmetry in relation to stress and fitness: effects of trait type as revealed by meta-analysis. Ecoscience 1996, 3, 400–413. [Google Scholar] [CrossRef]
- Indrasamy, H.; Woods, R.E.; McKenzie, J.A.; Batterham, P. Fluctuating asymmetry for specific bristle characters in Notch mutants of Drosophila melanogaster. Genetica. 2000, 109, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Nevo, E. Asian, African and European biota meet at “Evolution Canyon” Israel: local tests of global biodiversity and genetic diversity patterns. Proc. Roy. Soc. Lond. B 1995, 262, 149–155. [Google Scholar]
- Nevo, E. Evolution in action across phylogeny caused by microclimatic stresses at “Evolution Canyon”. Theor. Pop. Biol. 1997, 52, 231–243. [Google Scholar] [CrossRef] [PubMed]
- Nevo, E. Evolution of genome-phenome diversity under environmental stress. Proc. Natl. Acad. Sci. USA 2001, 98, 6233–6240. [Google Scholar] [CrossRef] [PubMed]
- Nevo, E. "Evolution Canyon": a microcosm of life’s evolution focusing on adaptation and speciation. Isr. J. Ecol. Evol. 2006, 52, 485–506. [Google Scholar] [CrossRef]
- Nevo, E.; Raz, S.; Beiles, A. Biodiversity of reptiles at "Evolution Canyon", Lower Nahal Oren, Mount Carmel, Israel. Isr. J. Zool. 1996, 42, 395–402. [Google Scholar]
- Raz, S.; Retzkin, S.; Pavlíček, T.; Hoffman, A.; Kimchi, H.; Zehavi, D.; Beiles, A.; Nevo, E. Scorpion biodiversity and interslope divergence at “Evolution Canyon”, Lower Nahal Oren microsite, Mt. Carmel, Israel. PLoS ONE 2009, 4, e5214. [Google Scholar] [CrossRef] [PubMed]
- Pavlíček, T.; Sharon, D.; Kravchenko, V.; Saaroni, H.; Nevo, E. Microclimatic interslope differences underlying biodiversity contrasts in “Evolution Canyon”, Mt. Carmel, Israel. Isr. J. Earth Sci. 2003, 52, 1–9. [Google Scholar] [CrossRef]
- Auslander, M.; Nevo, E.; Inbar, M. The effects of slope orientation on plant growth, developmental instability and susceptibility to herbivores. J. Arid Environ. 2003, 55, 405–416. [Google Scholar] [CrossRef]
- Derzhavets, E.; Korol, A.; Nevo, E. Differences in fluctuating asymmetry in Drosophila melanogaster caused by microclimatic contrasts. Dros. Inf. Serv. 1997, 80, 73–75. [Google Scholar]
- Kennington, W.J.; Killeen, J.R.; Goldstein, D.B.; Partridge, L. Rapid laboratory evolution of adult wing area in Drosophila melanogaster in response to humidity. Evolution 2003, 57, 932–936. [Google Scholar] [CrossRef] [PubMed]





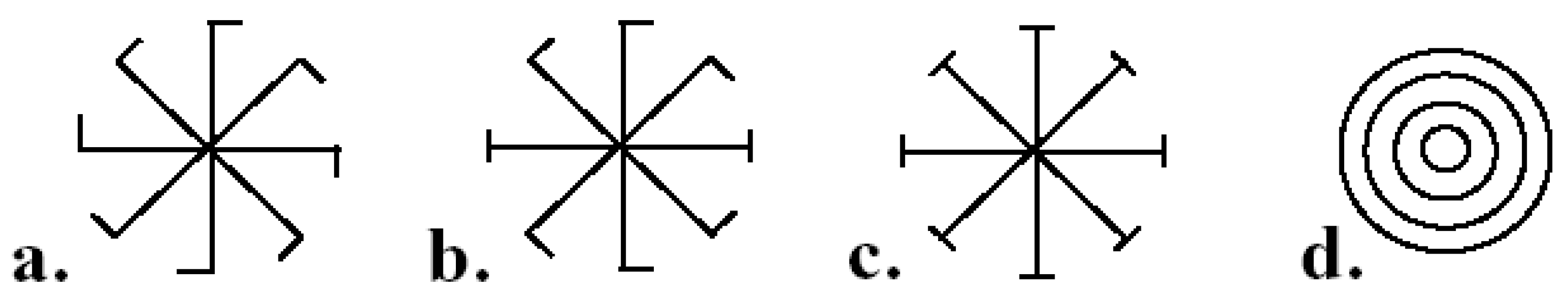
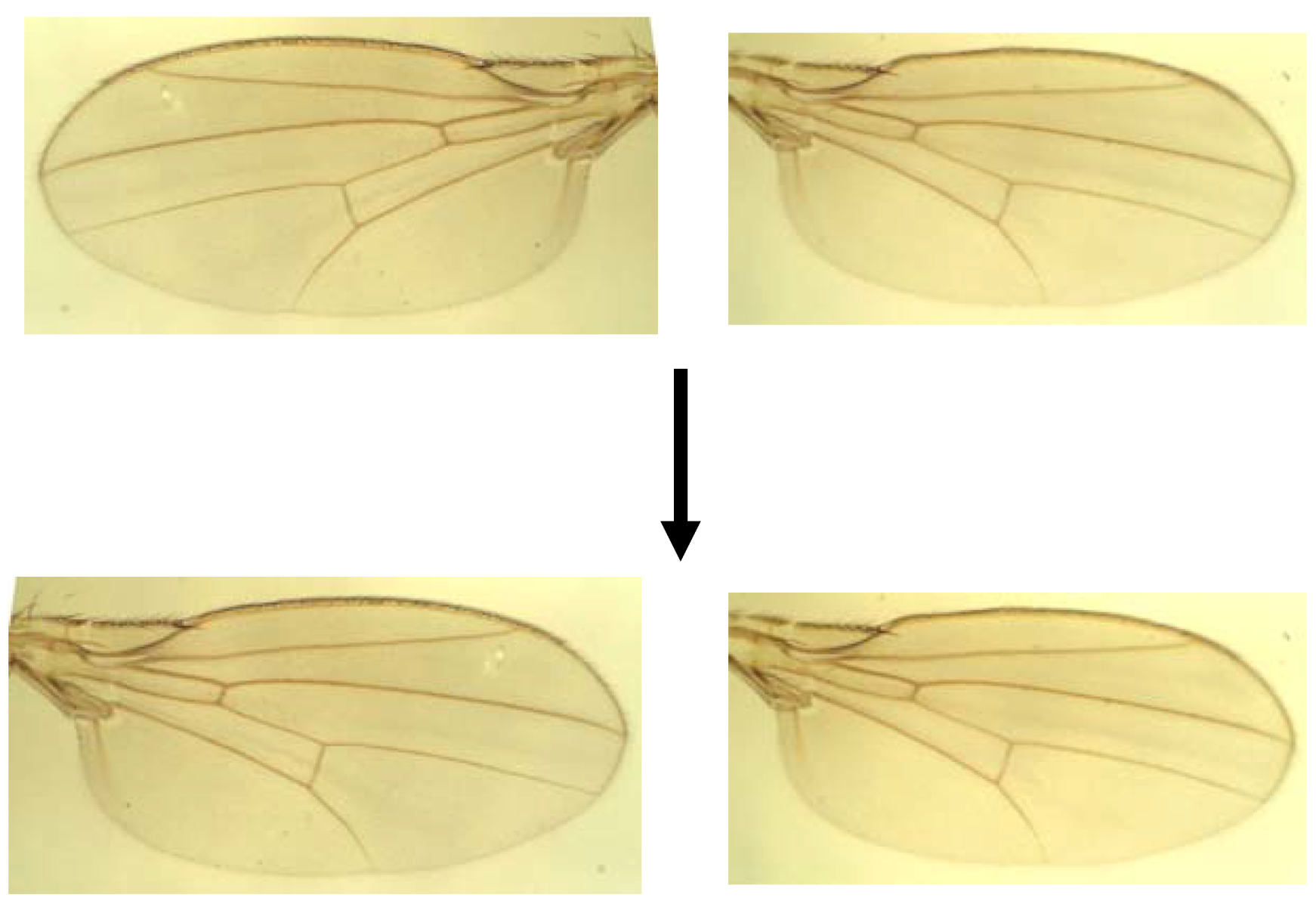
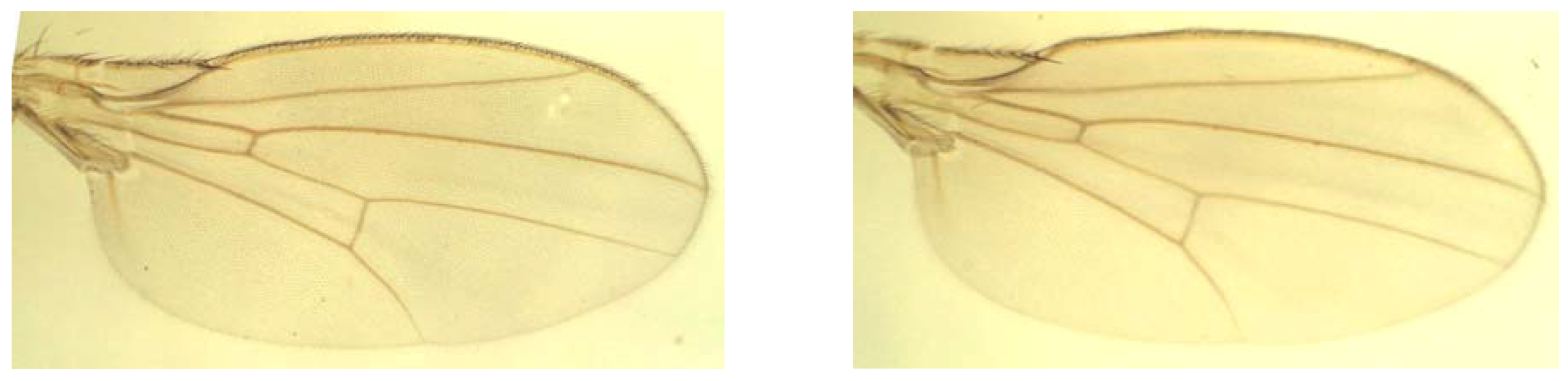

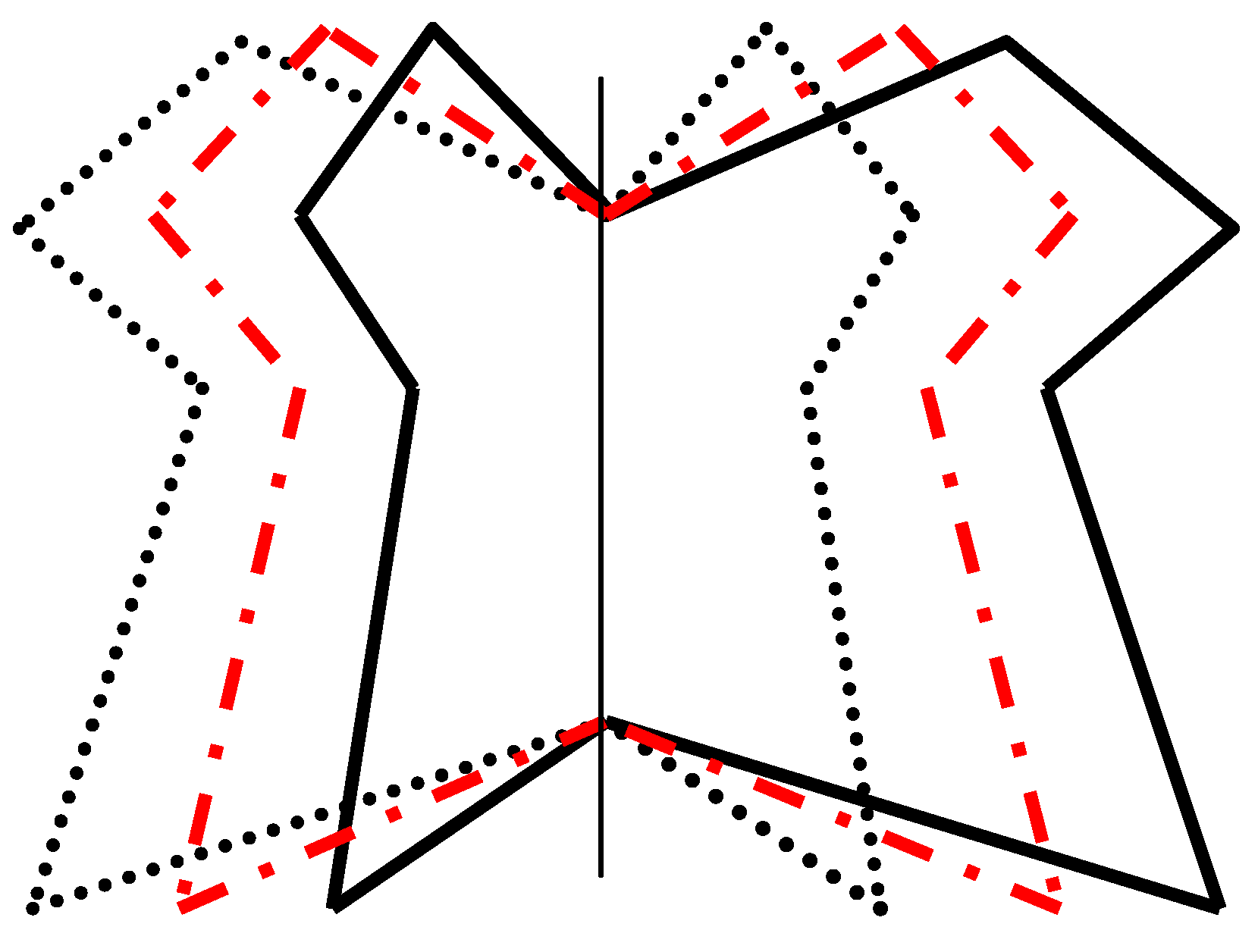


| Source | df | MS | Expected mean squares | Interpretation |
|---|---|---|---|---|
| Sides | 1 | MS S | σ 2m + R (σ 2S x I + N <σ 2s >) | Directional asymmetry |
| Individuals | N – 1 | MS I | σ 2m + R (σ 2S x I + 2σ 2I ) | Size/shape variation |
| Sides x Individuals | N – 1 | MS S x I | σ 2m + R σ 2S x I | FA and antisymmetry |
| Replicates (S x I) | N(R – 1) | MSerror | σ 2m | Measurement error |
| Transformation | Equation | References |
|---|---|---|
| Division by the mean | d or |d| divided by the trait mean (l + r)/2 | [48] |
| Log transform | log l – log r | [99,52,82,49] |
| Power transform | [(l λ – 1)/ λ] – [(r λ – 1)/ λ] for λ ≠ 0 | |
| log l – log r for λ = 0 | ||
| Half-normal transform | (|d + 0.00005)0.33 | [54] |
| Stressor | Taxa | Traits | Result1 | Reference |
|---|---|---|---|---|
| Water limitation/ | Quercus ilex, wet site | leaf | ↑FA | [222] |
| drought | Quercus ilex, dry site | leaf | 0 | [222] |
| Phaseolus vulgaris | leaf | ↑FA | [223] | |
| Salix sericea | leaf | 0 | [224] | |
| Salix eriocephala | leaf | 0 | [224] | |
| Flooding | Betula pubescens | leaf | 0 | [218] |
| High salinity | Glycine max | leaf | 0 | [216] |
| UV-B radiation | Dimorphotheca sinuate | leaf | ↑FA | [225] |
| Heat shock | Bicyclus anynana | eyespots | 0 | [226] |
| Scathophaga stercoraria | tibia and wing | ↑FA | [227] | |
| High elevation/cold | Betula pubescens | leaf | ↑FA | [228] |
| Food limitation | Sternus vulgaris | primary feathers | ↑FA | [229] |
| Scathophaga stercoraria | tibia and wing | 0 | [227] | |
| Coenagrion puella | femurs, wings | 02 | [230] | |
| Cyrtodiopsis dalmanni | eye stalks, wings | 0 | [200] | |
| Nutrient limitation | Acer platanoides | leaf | 0 | [205] |
| Betula pendula | leaf | 0 | [205] | |
| Nitrogen enrichment | Betula pubescens | leaf | ↑FA | [213] |
| Lythrum salicaria | leaf | ↑FA | [214] | |
| Penthorum sedoides | leaf | 0 | [214] | |
| Competition | Salix sericea | leaf | 0 | [224] |
| Salix eriocephala | leaf | 0 | [224] | |
| Insect attack | Betula pubescens | leaf | 0 | [228] |
| Grazing/browsing | Betula pubescens | leaf | ↑FA | [218] |
| Infection | Salix sericea | leaf | 0 | [224] |
| Salix eriocephala | leaf | 0 | [224] | |
| Natural disaster | Peromyscus leucopus | femur length | ↑FA | [231] |
| P. maniculatus | femur length | ↓FA | [231] |
| Stressor | Species | Traits | Result1 | Reference |
|---|---|---|---|---|
| Inbreeding | Scathophaga stercoraria | tibia and wing | 0 | [227] |
| Hybridization | Dalechampia scandens | leaf | 0 | [234] |
| Piriqueta caroliniana | leaf | ↑FA | [235] | |
| Salix spp. | leaf | ↑FA | [224] | |
| Enneacanthus spp. | meristic | ↑FA | [154] | |
| Directional selection | Drosophila melanogaster | wing shape | 0 | [217] |
| Heterozygosity | Alectoris chukar | toe length | 0 | [236] |
| Molecular chaperones | Drosophila melanogaster | bristles, wings | 0 | [171] |
| (Hsp90, Hsp83) | Drosophila melanogaster | wing shape | 0 and ↑FA2 | [173] |
| Arabidopsis thaliana | hypocotyl | ↑Vp 3 | [174] | |
| DNA damage | Dimorphotheca sinuate | leaf | ↑FA | [225] |
| Transposons | Drosophila melanogaster | bristles | 0 | [237] |
| Stressor | Taxa | Traits | Result1 | Reference |
|---|---|---|---|---|
| Heavy metals | Drosophila melanogaster | bristles | ↑FA | [301] |
| Drosophila melanogaster | bristles | 0 | [302] | |
| Sorex araneus | various | ↑FA | [303] | |
| Paper mill effluent | Gambusia holbrooki | various | ↑FA | [304] |
| Urban air pollution | Platanus | leaf | ↑FA | [305] |
| SO2 emmissions | Betula spp. | leaf | ↑FA2 | [45] |
| Pinus sylvestris | leaf | ↑FA | [293,306] | |
| Acidification | Rana arvalis | skeleton | ↑FA | [307] |
| Pesticides | Lucilia cuprina | bristle, wing | ↑FA3 | [299] |
| Elevated CO2Dioxin | Quercus spp.Mus musculus | leafmandible | ↓FA04 | [300][308] |
| teeth | ↑FA | [309] | ||
| Magnetic fields | Drosophila melanogaster | bristles | 0 | [310] |
| wing veins | ↑↓FA5 | [310] | ||
| phenodeviants | ↑↓FA5 | [310] | ||
| Military training | Rhus copallinum | leaf | ↓FA6 | [311] |
| Ipomoea pandurata | leaf | ↓FA6 | [311] | |
| Cnidoscolus stimulosus | leaf | ↑FA | [312] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Graham, J.H.; Raz, S.; Hel-Or, H.; Nevo, E. Fluctuating Asymmetry: Methods, Theory, and Applications. Symmetry 2010, 2, 466-540. https://doi.org/10.3390/sym2020466
Graham JH, Raz S, Hel-Or H, Nevo E. Fluctuating Asymmetry: Methods, Theory, and Applications. Symmetry. 2010; 2(2):466-540. https://doi.org/10.3390/sym2020466
Chicago/Turabian StyleGraham, John H., Shmuel Raz, Hagit Hel-Or, and Eviatar Nevo. 2010. "Fluctuating Asymmetry: Methods, Theory, and Applications" Symmetry 2, no. 2: 466-540. https://doi.org/10.3390/sym2020466
APA StyleGraham, J. H., Raz, S., Hel-Or, H., & Nevo, E. (2010). Fluctuating Asymmetry: Methods, Theory, and Applications. Symmetry, 2(2), 466-540. https://doi.org/10.3390/sym2020466







