Asymmetry of Sagittal Otolith Shape Based on Inner Ear Side Tested on Mediterranean Red Mullet (Mullus barbatus Linnaeus, 1758): Comparative Analysis of 2D and 3D Otolith Shape Data
Abstract
1. Introduction
2. Materials and Methods
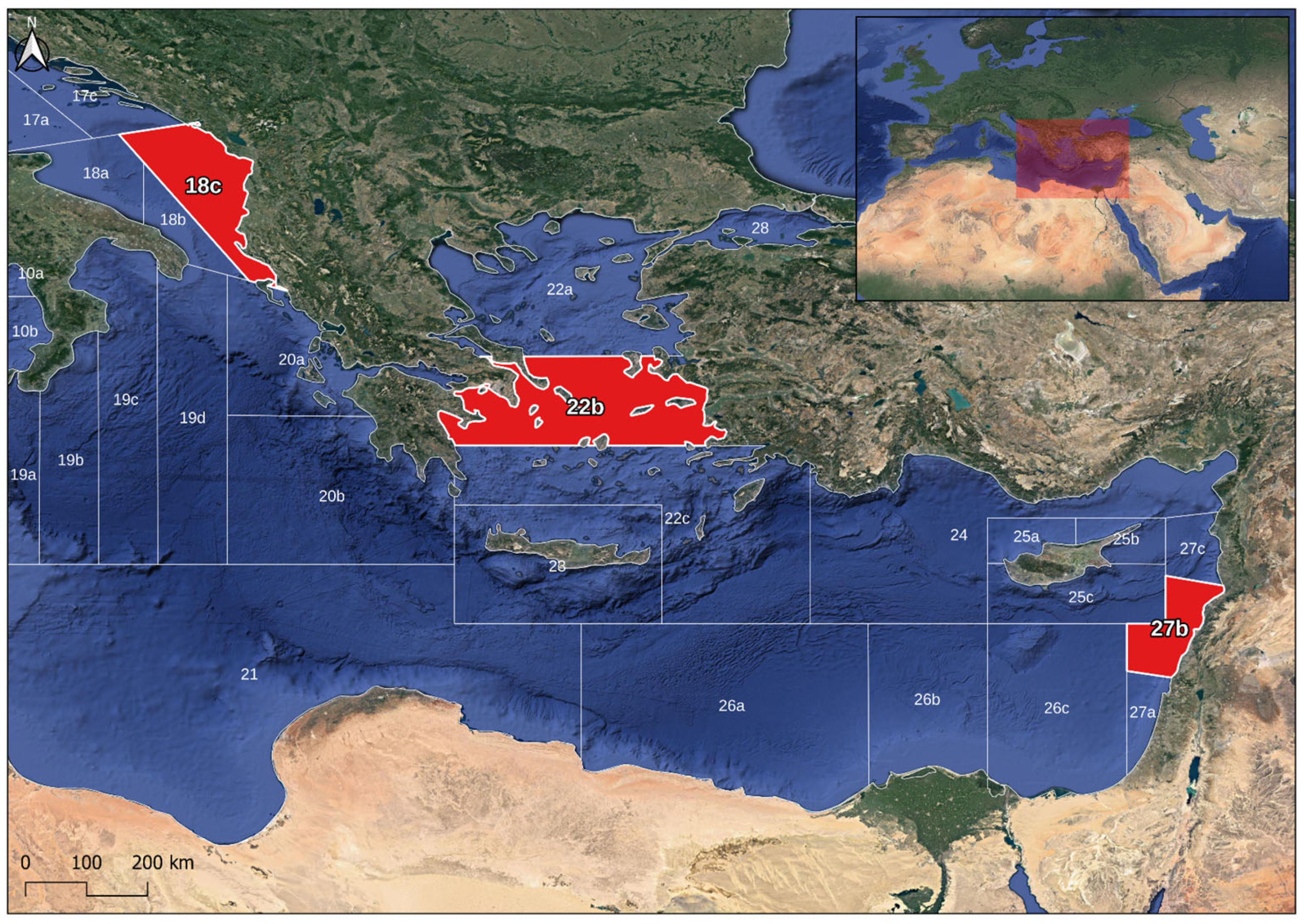
2.1. Sampling
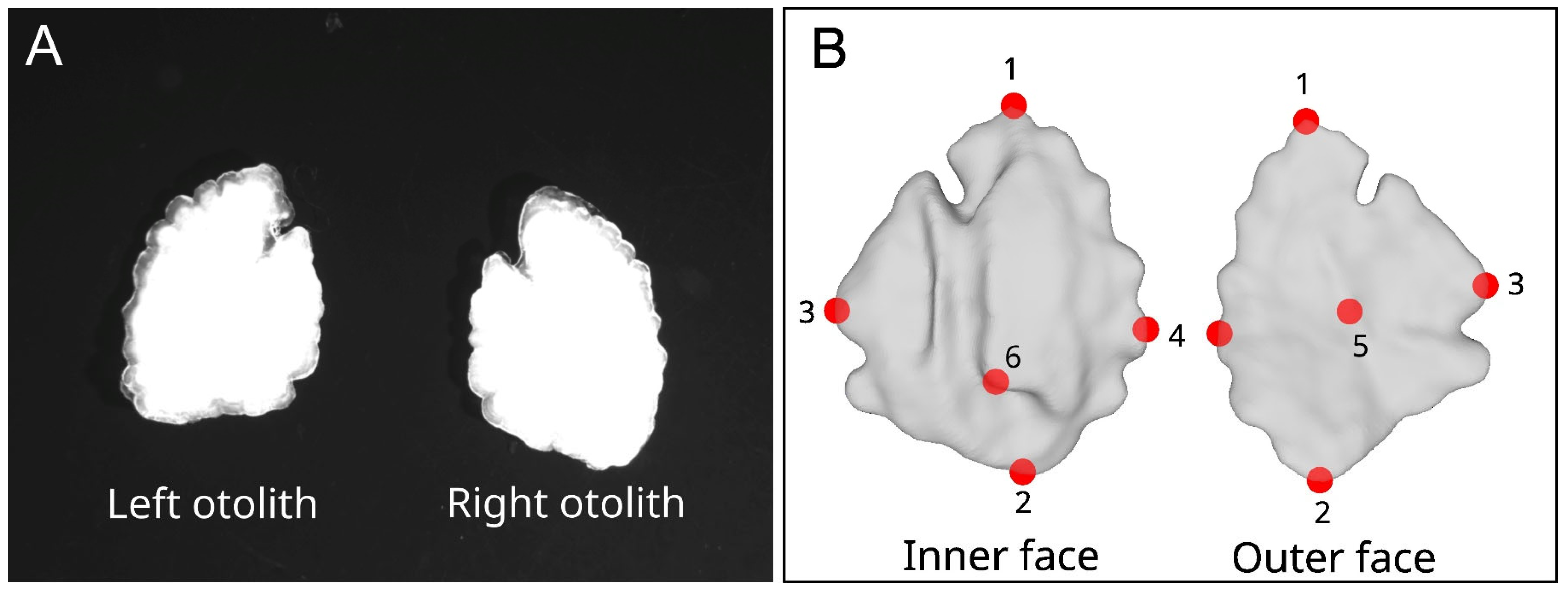
2.2. Two-Dimensional Outlines and Three-Dimensional Surfaces
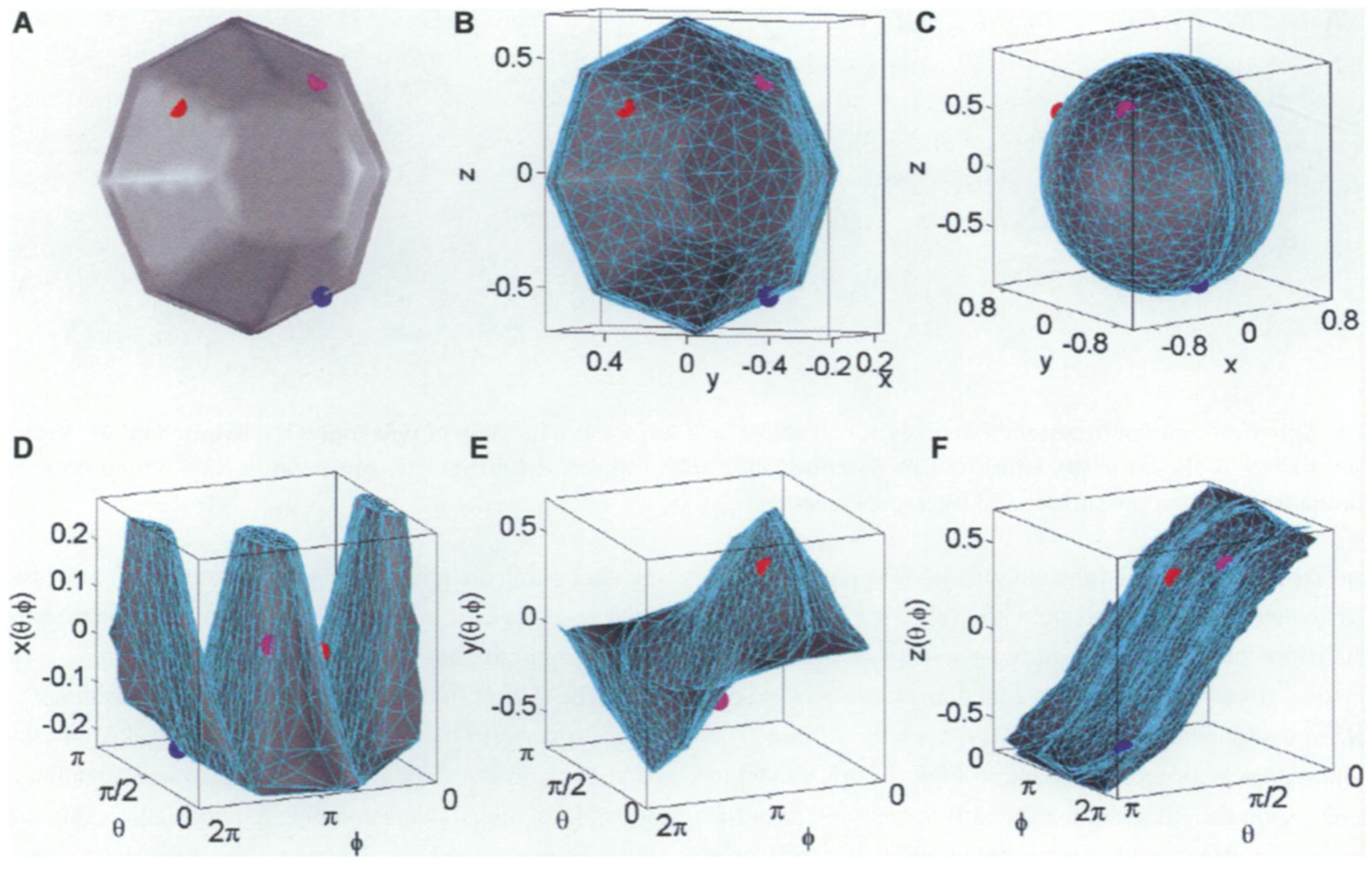
2.3. Two-Dimensional and Three-Dimensional Extraction of Shape Information
2.4. Statistical Analysis
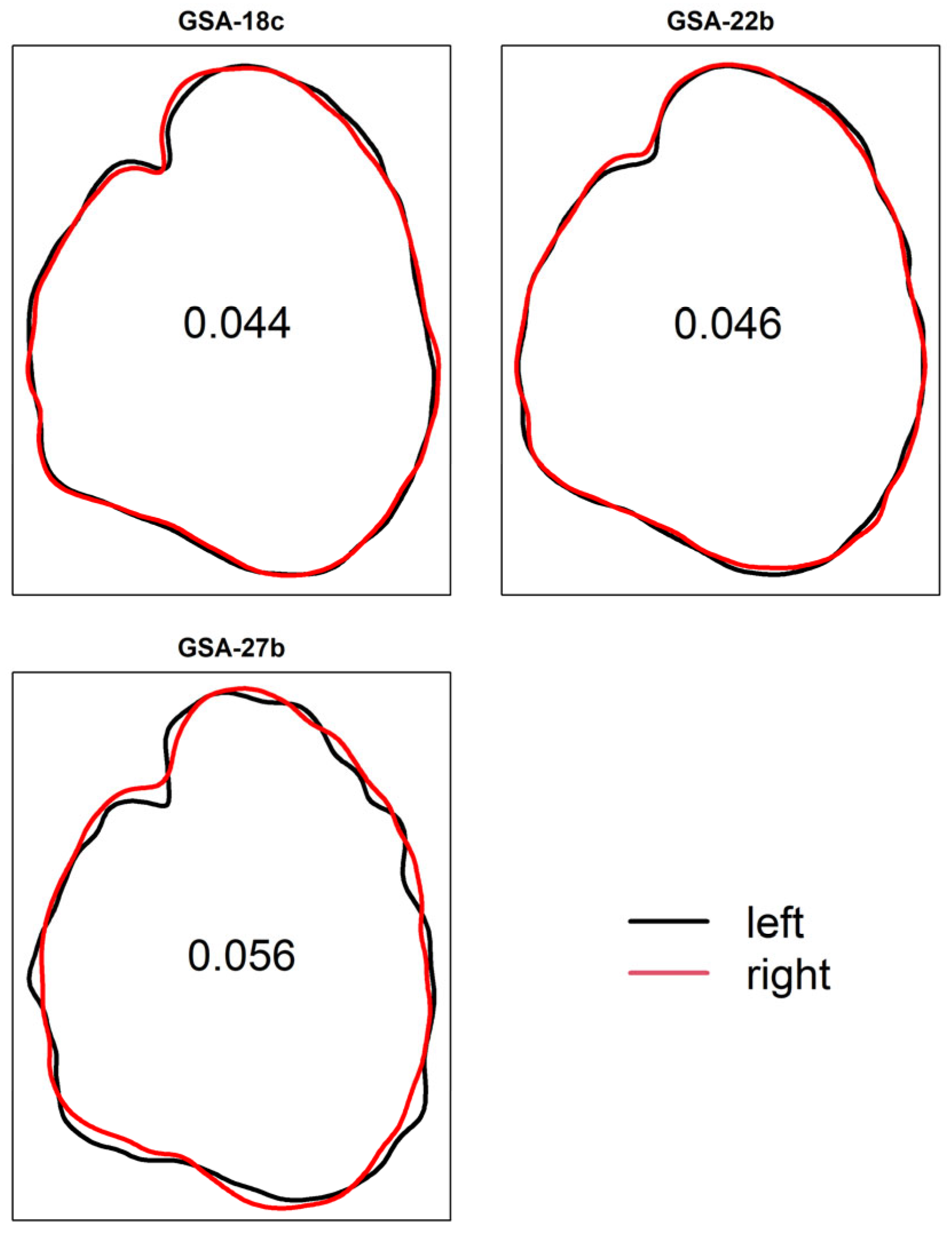
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. The State of World Fisheries and Aquaculture 2020; Sustainability in Action; FAO: Rome, Italy, 2020; ISBN 978-92-5-132755-5. [Google Scholar]
- Wagner, G.P.; Booth, G.; Bagheri-Chaichian, H. A Population Genetic Theory of Canalization. Evolution 1997, 51, 329–347. [Google Scholar] [CrossRef] [PubMed]
- Cadrin, S.X. Defining Spatial Structure for Fishery Stock Assessment. Fish. Res. 2020, 221, 105397. [Google Scholar] [CrossRef]
- Cadrin, S.X.; Maunder, M.N.; Punt, A.E. Spatial Structure: Theory, Estimation and Application in Stock “Assessment Models”. Fish. Res. 2020, 229, 105608. [Google Scholar] [CrossRef]
- Biseau, A. Diagnostic 2021 sur les Ressources Halieutiques Débarquées par la Pêche Française (Métropolitaine); “Ifremer Ref. RBE/EDERU/RS/2022/1 2022”; Ifremer: Brest, France, 2022; 40p. [Google Scholar]
- Cadrin, S.; Kerr, L.; Mariani, S. Stock Identification Methods: Applications in Fishery Science, 2nd ed.; Elsevier Academic Press: Cambridge, MA, USA, 2014; ISBN 978-0-12-397258-3. [Google Scholar]
- Avigliano, E.; Maichak de Carvalho, B.; Leisen, M.; Romero, R.; Velasco, G.; Vianna, M.; Barra, F.; Volpedo, A.V. Otolith Edge Fingerprints as Approach for Stock Identification of Genidens barbus. Estuar. Coast. Shelf Sci. 2017, 194, 92–96. [Google Scholar] [CrossRef]
- Mahé, K. Sources de Variation de La Forme Des Otolithes: Implications Pour La Discrimination Des Stocks de Poissons. Ph.D. Thesis, Université du Littoral Côte d’Opale, Dunkirk, France, 2019. [Google Scholar]
- Begg, G.A.; Waldman, J.R. An Holistic Approach to Fish Stock Identification. Fish. Res. 1999, 43, 35–44. [Google Scholar] [CrossRef]
- Berg, F.; Østgaard, H.D.; Slotte, A.; Andersson, L.; Folkvord, A. A Combination of Genetic and Phenotypic Characterization of Spring- and Autumn-Spawning Herring Suggests Gene Flow between Populations. ICES J. Mar. Sci. 2021, 78, 694–703. [Google Scholar] [CrossRef]
- Sherman, K.; Sissenwine, M.; Christensen, V.; Duda, A.; Hempel, G.; Ibe, C.; Levin, S.; Lluch-Belda, D.; Matishov, G.; McGlade, J.; et al. A Global Movement toward an Ecosystem Approach to Management of Marine Resources. Mar. Ecol. Prog. Ser. 2005, 300, 275–279. [Google Scholar] [CrossRef]
- Spalding, M.D.; Fox, H.E.; Allen, G.R.; Davidson, N.; Ferdaña, Z.A.; Finlayson, M.; Halpern, B.S.; Jorge, M.A.; Lombana, A.; Lourie, S.A.; et al. Marine Ecoregions of the World: A Bioregionalization of Coastal and Shelf Areas. BioScience 2007, 57, 573–583. [Google Scholar] [CrossRef]
- Palomares, M.L.D.; Froese, R.; Derrick, B.; Meeuwig, J.J.; Nöel, S.-L.; Tsui, G.; Woroniak, J.; Zeller, D.; Pauly, D. Fishery Biomass Trends of Exploited Fish Populations in Marine Ecoregions, Climatic Zones and Ocean Basins. Estuar. Coast. Shelf Sci. 2020, 243, 106896. [Google Scholar] [CrossRef]
- ICES. Stock Identification Methods Working Group (SIMWG). ICES Sci. Rep. 2022, 4, 66. [Google Scholar] [CrossRef]
- Cruz, T.A.; Thorpe, J.P.; Pullin, R.S.V. Enzyme Electrophoresis in Tilapia zillii: A Pattern for Determining Biochemical Genetic Markers for Use in Tilapia Stock Identification. Aquaculture 1982, 29, 311–329. [Google Scholar] [CrossRef]
- Beacham, T.D. Parentage-Based Tagging Combined with Genetic Stock Identification Is a Cost-Effective and Viable Replacement for Coded-Wire Tagging in Large-Scale Assessments of Canadian Salmon Fisheries. Fish. Res. 2021, 239, 105920. [Google Scholar] [CrossRef]
- Lester, R.J.G.; MacKenzie, K. The Use and Abuse of Parasites as Stock Markers for Fish. Fish. Res. 2009, 97, 1–2. [Google Scholar] [CrossRef]
- Pascual, S.; Abollo, E.; González, A.F. Biobanking and Genetic Markers for Parasites in Fish Stock Studies. Fish. Res. 2016, 173, 214–220. [Google Scholar] [CrossRef]
- Vasconcelos, J.; Hermida, M.; Saraiva, A.; González, J.A.; Gordo, L.S. The Use of Parasites as Biological Tags for Stock Identification of Blue Jack Mackerel, Trachurus Picturatus, in the North-Eastern Atlantic. Fish. Res. 2017, 193, 1–6. [Google Scholar] [CrossRef]
- Grahl-Nielsen, O. Chapter Twelve—Fatty Acid Profiles as Natural Marks for Stock Identification. In Stock Identification Methods, 2nd ed.; Cadrin, S.X., Kerr, L.A., Mariani, S., Eds.; Academic Press: San Diego, CA, USA, 2014; pp. 235–256. ISBN 978-0-12-397003-9. [Google Scholar]
- Morales-Nin, B.; Pérez-Mayol, S.; MacKenzie, K.; Catalán, I.A.; Palmer, M.; Kersaudy, T.; Mahé, K. European Hake (Merluccius merluccius) Stock Structure in the Mediterranean as Assessed by Otolith Shape and Microchemistry. Fish. Res. 2022, 254, 106419. [Google Scholar] [CrossRef]
- Wilson, R.R. Depth-Related Changes in Sagitta Morphology in Six Macrourid Fishes of the Pacific and Atlantic Oceans. Copeia 1985, 1985, 1011–1017. [Google Scholar] [CrossRef]
- Morales-Nin, B.Y.O. The Influence of Environmental Factors on Microstructure of Otoliths of Three Demersal Fish Species Caught off Namibia. S. Afr. J. Mar. Sci. 1987, 5, 255–262. [Google Scholar] [CrossRef]
- Mosegaard, H.; Svedäng, H.; Taberman, K. Uncoupling of Somatic and Otolith Growth Rates in Arctic Char (Salvelinus alpinus) as an Effect of Differences in Temperature Response. Can. J. Fish. Aquat. Sci. 1988, 45, 1514–1524. [Google Scholar] [CrossRef]
- Secor, D.H.; Dean, J.M. Somatic Growth Effects on the Otolith–Fish Size Relationship in Young Pond-Reared Striped Bass, Morone saxatilis. Can. J. Fish. Aquat. Sci. 1989, 46, 113–121. [Google Scholar] [CrossRef]
- Campana, S.E.; Casselman, J.M. Stock Discrimination Using Otolith Shape Analysis. Can. J. Fish. Aquat. Sci. 1993, 50, 1062–1083. [Google Scholar] [CrossRef]
- Begg, G.; Overholtz, W.; Munroe, N. The Use of Internal Otolith Morphometrics for Identification of Haddock (Melanogrammus aeglefinus) Stocks on Georges Bank. Fish. Bull. 2001, 99, 1–14. [Google Scholar]
- Lombarte, A.; Lleonart, J. Otolith Size Changes Related with Body Growth, Habitat Depth and Temperature. Environ. Biol. Fishes 1993, 37, 297–306. [Google Scholar] [CrossRef]
- Lombarte, A.; Torres, G.J.; Morales-Nin, B. Specific Merluccius otolith growth patterns related to phylogenetics and environmental factors. J. Mar. Biol. Assoc. U.K. 2003, 83, 277–281. [Google Scholar] [CrossRef]
- Cardinale, M.; Doering-Arjes, P.; Kastowsky, M.; Mosegaard, H. Effects of Sex, Stock, and Environment on the Shape of Known-Age Atlantic Cod (Gadus morhua) Otoliths. Can. J. Fish. Aquat. Sci. 2004, 61, 158–167. [Google Scholar] [CrossRef]
- Vignon, M.; Morat, F. Environmental and Genetic Determinant of Otolith Shape Revealed by a Non-Indigenous Tropical Fish. Mar. Ecol. Prog. Ser. 2010, 411, 231–241. [Google Scholar] [CrossRef]
- Vignon, M. Disentangling and Quantifying Sources of Otolith Shape Variation across Multiple Scales Using a New Hierarchical Partitioning Approach. Mar. Ecol. Prog. Ser. 2015, 534, 163–177. [Google Scholar] [CrossRef]
- Simoneau, M.; Casselman, J.M.; Fortin, R. Determining the Effect of Negative Allometry (Length/Height Relationship) on Variation in Otolith Shape in Lake Trout (Salvelinus namaycush), Using Fourier-Series Analysis. Can. J. Zool. 2000, 78, 1597–1603. [Google Scholar] [CrossRef]
- Monteiro, L.R.; Beneditto, A.P.M.D.; Guillermo, L.H.; Rivera, L.A. Allometric Changes and Shape Differentiation of Sagitta Otoliths in Sciaenid Fishes. Fish. Res. 2005, 74, 288–299. [Google Scholar] [CrossRef]
- D’Iglio, C.; Natale, S.; Albano, M.; Savoca, S.; Famulari, S.; Gervasi, C.; Lanteri, G.; Panarello, G.; Spanò, N.; Capillo, G. Otolith Analyses Highlight Morpho-Functional Differences of Three Species of Mullet (Mugilidae) from Transitional Water. Sustainability 2021, 14, 398. [Google Scholar] [CrossRef]
- Trojette, M.; Abderraouf, B.F.; Fatnassi, M.; Marsaoui, B.; Mahouachi, N.E.H.; Chalh, D.A.; Jean-pierre, Q.; Trabelsi, M. Stock Discrimination of Two Insular Populations of Diplodus annularis (Actinopterygii: Perciformes: Sparidae) along the Coast of Tunisia by Analysis of Otolith Shape. Acta Ichthyol. Piscat. 2015, 45, 363–372. [Google Scholar] [CrossRef]
- Casselman, J.M. Determination of Age and Growth. In The Biology of Fish Growth; Weatherley, A.H., Gill, H.S., Eds.; Academic Press: New York, NY, USA, 1987; pp. 209–242. [Google Scholar]
- Reibisch, J. Ueber die Eizahl bei Pleuronectes Platessa und die Altersbestimmung Dieser Form Aus den Otolithen. Nebst Bemerkung zu Vorstehender Arbeit von Dr. V. Hensen; Wiss. Meeresunt: Kiel, Germany, 1899; pp. 233–248. [Google Scholar]
- Campana, S.E.; Neilson, J.D. Microstructure of Fish Otoliths. Can. J. Fish. Aquat. Sci. 1985, 42, 1014–1032. [Google Scholar] [CrossRef]
- R Foundation for Statistical Computing. R Core Team R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2022; Available online: https://www.R-project.org (accessed on 10 May 2022).
- Bird, J.L.; Eppler, D.T.; Checkley, D.M., Jr. Comparisons of Herring Otoliths Using Fourier Series Shape Analysis. Can. J. Fish. Aquat. Sci. 1986, 43, 1228–1234. [Google Scholar] [CrossRef]
- Pawson, M.G. Using Otolith Weight to Age Fish. J. Fish Biol. 1990, 36, 521–531. [Google Scholar] [CrossRef]
- Worthington, D.O.; Doherty, P.J.; Fowler, A.J. Variation in the Relationship between Otolith Weight and Age: Implications for the Estimation of Age of Two Tropical Damselfish (Pomacentrus moluccensis and P. wardi). Can. J. Fish. Aquat. Sci. 1995, 52, 233–242. [Google Scholar] [CrossRef]
- Cardinale, M.; Arrhenius, F. Using Otolith Weight to Estimate the Age of Haddock (Melanogrammus aeglefinus): A Tree Model Application. J. Appl. Ichthyol. 2004, 20, 470–475. [Google Scholar] [CrossRef]
- Francis, R.I.C.C.; Harley, S.J.; Campana, S.E.; Doering-Arjes, P.; Francis, R.I.C.C.; Harley, S.J.; Campana, S.E.; Doering-Arjes, P. Use of Otolith Weight in Length-Mediated Estimation of Proportions at Age. Mar. Freshw. Res. 2005, 56, 735–743. [Google Scholar] [CrossRef]
- Sadighzadeh, Z.; Valinassab, T.; Vosugi, G.; Motallebi, A.A.; Fatemi, M.R.; Lombarte, A.; Tuset, V.M. Use of Otolith Shape for Stock Identification of John’s Snapper, Lutjanus johnii (Pisces: Lutjanidae), from the Persian Gulf and the Oman Sea. Fish. Res. 2014, 155, 59–63. [Google Scholar] [CrossRef]
- Dos Santos, J.B.Q.; Chiessi, C.M.; Crivellari, S.; de Vasconcelos Filho, J.E.; Pereira, N.S.; Freitas, M.O.; Ferreira, B.P. Identification of Western South Atlantic Stocks of the Lane Snapper (Lutjanus synagris) from an Otolith Based Multi-Proxy Approach. Fish. Res. 2022, 253, 106356. [Google Scholar] [CrossRef]
- Zitek, A.; Mayrhofer, B.; Oehm, J.; Irrgeher, J.; Prohaska, T. Affordable 3D Scanning of Small Otoliths for Improved Shape Analysis by Photogrammetry Techniques. In Proceedings of the 5th International Otolith Symposium 2014, Mallorca, Spain, 22 October 2014. [Google Scholar]
- Mapp, J.J.I.; Fisher, M.H.; Atwood, R.C.; Bell, G.D.; Greco, M.K.; Songer, S.; Hunter, E. Three-Dimensional Rendering of Otolith Growth Using Phase Contrast Synchrotron Tomography. J. Fish Biol. 2016, 88, 2075–2080. [Google Scholar] [CrossRef]
- Marti-Puig, P.; Danés, J.; Manjabacas, A.; Lombarte, A. New Parameterisation Method for Three-Dimensional Otolith Surface Images. Mar. Freshw. Res. 2016, 67, 1059. [Google Scholar] [CrossRef]
- Vasconcelos-Filho, J.E.; Thomsen, F.S.L.; Stosic, B.; Antonino, A.C.D.; Duarte, D.A.; Heck, R.J.; Lessa, R.P.T.; Santana, F.M.; Ferreira, B.P.; Duarte-Neto, P.J. Peeling the Otolith of Fish: Optimal Parameterization for Micro-CT Scanning. Front. Mar. Sci. 2019, 6, 728. [Google Scholar] [CrossRef]
- Quindazzi, M.J.; Summers, A.P.; Juanes, F. Efficiency Is Doing Things Right: High-Throughput, Automated, 3D Methods in the Modern Era of Otolith Morphometrics. Can. J. Fish. Aquat. Sci. 2022, 79, 7. [Google Scholar] [CrossRef]
- Schulz-Mirbach, T.; Olbinado, M.; Rack, A.; Mittone, A.; Bravin, A.; Melzer, R.R.; Ladich, F.; Heß, M. In-Situ Visualization of Sound-Induced Otolith Motion Using Hard X-Ray Phase Contrast Imaging. Sci. Rep. 2018, 8, 3121. [Google Scholar] [CrossRef]
- Schulz-Mirbach, T.; Ladich, F.; Plath, M.; Heß, M. Enigmatic Ear Stones: What We Know about the Functional Role and Evolution of Fish Otoliths. Biol. Rev. 2019, 94, 457–482. [Google Scholar] [CrossRef]
- Schulz-Mirbach, T.; Ladich, F.; Mittone, A.; Olbinado, M.; Bravin, A.; Maiditsch, I.P.; Melzer, R.R.; Krysl, P.; Heß, M. Auditory Chain Reaction: Effects of Sound Pressure and Particle Motion on Auditory Structures in Fishes. PLoS ONE 2020, 15, e0230578. [Google Scholar] [CrossRef]
- Wei, C.; McCauley, R.D. Numerical Modeling of the Impacts of Acoustic Stimulus on Fish Otoliths from Two Directions. J. Acoust. Soc. Am. 2022, 152, 3226–3234. [Google Scholar] [CrossRef]
- Mille, T.; Mahe, K.; Villanueva, M.C.; De Pontual, H.; Ernande, B. Sagittal Otolith Morphogenesis Asymmetry in Marine Fishes. J. Fish Biol. 2015, 87, 646–663. [Google Scholar] [CrossRef]
- Mille, T. Sources de Variation Intra-Populationnelle de La Morphologie Des Otolithes: Asymétrie Directionnelle et Régime Alimentaire. Ph.D. Thesis, Université de Lille 1—Sciences et Technologies, Lille, France, 2015; p. 222. [Google Scholar]
- Palmer, A.R. What Determines Direction of Asymmetry: Genes, Environment or Chance? Philos. Trans. R. Soc. B Biol. Sci. 2016, 371, 20150417. [Google Scholar] [CrossRef]
- Mahé, K.; Ider, D.; Massaro, A.; Hamed, O.; Jurado-Ruzafa, A.; Gonçalves, P.; Anastasopoulou, A.; Jadaud, A.; Mytilineou, C.; Elleboode, R.; et al. Directional Bilateral Asymmetry in Otolith Morphology May Affect Fish Stock Discrimination Based on Otolith Shape Analysis. ICES J. Mar. Sci. 2018, 76, 232–243. [Google Scholar] [CrossRef]
- Conides, A.; Papaconstantinou, C. Commercial Fisheries in the Mediterranean, Focusing on the Environmental Status and the Corresponding Management Measures. Aquac. Fish. Stud. 2020, 2. [Google Scholar] [CrossRef]
- Spedicato, M.T.; Zupa, W.; Carbonara, P.; Fiorentino, F.; Follesa, M.; Galgani, F.; Garcia, C.; Jadaud, A.; Ioakeimidis, C.; Lazarakis, G.; et al. Spatial Distribution of Marine Macro-Litter on the Seafloor in the Northern Mediterranean Sea: The MEDITS Initiative. Sci. Mar. 2019, 83, 257–270. [Google Scholar] [CrossRef]
- Andrialovanirina, N.; Hache, A.; Mahé, K.; Couette, S.; Caillault, E.P. Automatic Method to Transform Routine Otolith Images for a Standardized Otolith Database Using R. Cybium 2023, 47, 31–42. [Google Scholar] [CrossRef]
- Laffont, R.; Navarro, N. Digit3DLand: Digitalization of 3D Landmarks on Mesh. R package version 0.2.3. 2019. Available online: https://github.com/morphOptics/digit3DLand (accessed on 13 June 2022).
- Vicory, J.; Pascal, L.; Hernandez, P.; Fishbaugh, J.; Prieto, J.; Mostapha, M.; Huang, C.; Shah, H.; Hong, J.; Liu, Z.; et al. SlicerSALT: Shape AnaLysis Toolbox. In Shape in Medical Imaging—ShapeMI 2018; Reuter, M., Wachinger, C., Lombaert, H., Paniagua, B., Lüthi, M., Egger, B., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 65–72. [Google Scholar]
- Kuhl, F.P.; Giardina, C.R. Elliptic Fourier Features of a Closed Contour. Comput. Graph. Image Process. 1982, 18, 236–258. [Google Scholar] [CrossRef]
- Lestrel, P.E. Fourier Descriptors and Their Applications in Biology. Cambridge University Press: Cambridge, UK, 2008; p. 482. ISBN 978-0-521-45201-4. [Google Scholar]
- Bonhomme, V.; Picq, S.; Gaucherel, C.; Claude, J. Momocs: Outline Analysis Using R. J. Stat. Softw. 2014, 56, 1–24. [Google Scholar] [CrossRef]
- Shen, L.; Farid, H.; McPeek, M.A. Modeling Three-Dimensional Morphological Structures Using Spherical Harmonics. Evolution 2009, 63, 1003–1016. [Google Scholar] [CrossRef]
- Shen, L.; Makedon, F. Spherical Mapping for Processing of 3D Closed Surfaces. Image Vis. Comput. 2006, 24, 743–761. [Google Scholar] [CrossRef]
- Rohlf, F.J.; Archie, J.W. A Comparison of Fourier Methods for the Description of Wing Shape in Mosquitoes (Diptera: Culicidae). Syst. Biol. 1984, 33, 302–317. [Google Scholar] [CrossRef]
- Legendre, P.; Legendre, L. Numerical Ecology; Elsevier: Amsterdam, The Netherlands, 2012; ISBN 978-0-444-53869-7. [Google Scholar]
- Plate, T.; Heiberger, R. abind: Combine Multidimensional Arrays. R package version 1.4-5. 2016. Available online: https://CRAN.R-project.org/package=abind (accessed on 18 July 2022).
- Fox, J.; Weisberg, S. An R Campanion to Applied Regression; Sage: Thousand Oaks, CA, USA, 2019; ISBN 978-1-412-97514-8. [Google Scholar]
- Fox, J.; Hong, J. Effect Displays in R for Multinomial and Proportional-Odds Logit Models: Extensions to the Effects Package. J. Stat. Softw. 2010, 32, 1–24. [Google Scholar] [CrossRef]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using Lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Bates, D.; Maechler, M.; Jagan, M. Matrix: Sparse and Dense Matrix Classes and Methods. R package version 1.5-1. 2022. Available online: https://CRAN.R-project.org/package=Matrix (accessed on 18 July 2022).
- Pinheiro, J.C.; Bates, D.M. Mixed-Effects Models in S and S-PLUS; Springer: New York, NY, USA, 2000. [Google Scholar] [CrossRef]
- Pinheiro, J.C.; Bates, D.M.; R Core Team. nlme: Linear and Nonlinear Mixed Effects Models. R package version 3.1-160. 2022. Available online: https://CRAN.R-project.org/package=nlme (accessed on 18 July 2022).
- Borchers, H. pracma: Practical Numerical Math Functions, R package version 2.4.2. Available online: https://CRAN.R-project.org/package=pracma (accessed on 18 July 2022).
- Bivand, R.; Rundel, C. rgeos: Interface to Geometry Engine—Open Source (’GEOS’). R package version 0.5-9. 2021. Available online: https://CRAN.R-project.org/package=rgeos (accessed on 18 July 2022).
- Murdoch, D.; Adler, D. rgl: 3D Visualization Using OpenGL. R package version 1.0.1. 2023. Available online: https://CRAN.R-project.org/package=rgl (accessed on 18 July 2022).
- Schlager, S. Chapter 9—Morpho and Rvcg—Shape Analysis in R: R-Packages for Geometric Morphometrics, Shape Analysis and Surface Manipulations. In Statistical Shape and Deformation Analysis; Zheng, G., Li, S., Székely, G., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 217–256. ISBN 978-0-12-810493-4. [Google Scholar]
- Bivand, R.S.; Pebesma, E.; Gomez-Rubio, V. Applied Spatial Data Analysis with R., 2nd ed.; Springer: New York, NY, USA, 2013. [Google Scholar]
- Oksanen, J.; Simpson, G.; Blanchet, F.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, R.; Solymos, P.; Stevens, M.; Szoecs, E.; et al. vegan: Community ecology package. R package version 2.2-0. 2013. Available online: https://CRAN.R-project.org/package=vegan (accessed on 18 July 2022).
- Pawson, M.G.; Jennings, S. A Critique of Methods for Stock Identification in Marine Capture Fisheries. Fish. Res. 1996, 25, 203–217. [Google Scholar] [CrossRef]
- Ponton, D. Is Geometric Morphometrics Efficient for Comparing Otolith Shape of Different Fish Species? J. Morphol. 2006, 267, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Garcia, A.; Mattiucci, S.; Damiano, S.; Santos, M.N.; Nascetti, G. Metazoan Parasites of Swordfish, Xiphias gladius (Pisces: Xiphiidae) from the Atlantic Ocean: Implications for Host Stock Identification. ICES J. Mar. Sci. 2011, 68, 175–182. [Google Scholar] [CrossRef]
- ICES. Report of the Stock Identification Methods Working Group (SIMWG); “CES CM 2016/SSGEPI:16”; ICES: Toronto, ON, Canada, 2016; p. 47. [Google Scholar] [CrossRef]
- Pita, A.; Casey, J.; Hawkins, S.J.; Villarreal, M.R.; Gutiérrez, M.-J.; Cabral, H.; Carocci, F.; Abaunza, P.; Pascual, S.; Presa, P. Conceptual and Practical Advances in Fish Stock Delineation. Fish. Res. 2016, 173, 185–193. [Google Scholar] [CrossRef]
- Nasreddine, K.; Benzinou, A.; Fablet, R. Shape Geodesics for the Classification of Calcified Structures: Beyond Fourier Shape Descriptors. Fish. Res. 2009, 98, 8–15. [Google Scholar] [CrossRef]
- Benzinou, A.; Carbini, S.; Nasreddine, K.; Elleboode, R.; Mahé, K. Discriminating Stocks of Striped Red Mullet (Mullus surmuletus) in the Northwest European Seas Using Three Automatic Shape Classification Methods. Fish. Res. 2013, 143, 153–160. [Google Scholar] [CrossRef]
- Duarte-Neto, P.; Lessa, R.; Stosic, B.; Morize, E. The Use of Sagittal Otoliths in Discriminating Stocks of Common Dolphinfish (Coryphaena hippurus) off Northeastern Brazil Using Multishape Descriptors. ICES J. Mar. Sci. 2008, 65, 1144–1152. [Google Scholar] [CrossRef]
- Mapp, J.; Hunter, E.; van der Kooij, J.; Songer, S.; Fisher, M. Otolith Shape and Size: The Importance of Age When Determining Indices for Fish-Stock Separation. Fish. Res. 2017, 190, 43–52. [Google Scholar] [CrossRef]
- Cadrin, S.X.; Friedland, K.D. The Utility of Image Processing Techniques for Morphometric Analysis and Stock Identification. Fish. Res. 1999, 43, 129–139. [Google Scholar] [CrossRef]
- Morat, F.; Letourneur, Y.; Nérini, D.; Banaru, D.; Batjakas, I.E. Discrimination of Red Mullet Populations (Teleostean, Mullidae) along Multi-Spatial and Ontogenetic Scales within the Mediterranean Basin on the Basis of Otolith Shape Analysis. Aquat. Living Resour. 2012, 25, 27–39. [Google Scholar] [CrossRef]
- Castonguay, M.; Simard, P.; Gagnon, P. Usefulness of Fourier Analysis of Otolith Shape for Atlantic Mackerel (Scomber scombrus) Stock Discrimination. Can. J. Fish. Aquat. Sci. 1991, 48, 296–302. [Google Scholar] [CrossRef]
- Petursdottir, G.; Begg, G.A.; Marteinsdottir, G. Discrimination between Icelandic Cod (Gadus morhua L.) Populations from Adjacent Spawning Areas Based on Otolith Growth and Shape. Fish. Res. 2006, 80, 182–189. [Google Scholar] [CrossRef]
- Mahé, K.; Evano, H.; Mille, T.; Muths, D.; Bourjea, J. Otolith Shape as a Valuable Tool to Evaluate the Stock Structure of Swordfish Xiphias Gladius in the Indian Ocean. Afr. J. Mar. Sci. 2016, 38, 457–464. [Google Scholar] [CrossRef]
- Palmer, A.R. Animal Asymmetry. Curr. Biol. 2009, 19, R473–R477. [Google Scholar] [CrossRef]
- Lemberget, T.; McCormick, M.I. Replenishment Success Linked to Fluctuating Asymmetry in Larval Fish. Oecologia 2009, 159, 83–93. [Google Scholar] [CrossRef]
- Green, A.; Mosaliganti, K.; Swinburne, I.; Obholzer, N.; Megason, S. Recovery of Shape and Size in a Developing Organ Pair. Dev. Dyn. 2017, 246, 451–465. [Google Scholar] [CrossRef]
- Mahé, K.; MacKenzie, K.; Ider, D.; Massaro, A.; Hamed, O.; Jurado-Ruzafa, A.; Gonçalves, P.; Anastasopoulou, A.; Jadaud, A.; Mytilineou, C.; et al. Directional Bilateral Asymmetry in Fish Otolith: A Potential Tool to Evaluate Stock Boundaries? Symmetry 2021, 13, 987. [Google Scholar] [CrossRef]
- Hilbig, R.; Knie, M.; Shcherbakov, D.; Anken, R.H. Analysis of Behaviour and Habituation of Fish Exposed to Diminished Gravity in Correlation to Inner Ear Stone Formation-A Sounding Rocket Experiment (TEXUS 45). In Proceedings of the 20th ESA Symposium on Europe Rocket and Balloon Programmes and Related Research, Hyere, France, 22–26 May 2011. [Google Scholar]
- Lychakov, D.V.; Rebane, Y.T.; Lombarte, A.; Demestre, M.; Fuiman, L.A. Saccular Otolith Mass Asymmetry in Adult Flatfishes. J. Fish Biol. 2008, 72, 2579–2594. [Google Scholar] [CrossRef]
- Beier, M.; Anken, R.H.; Rahmann, H. Susceptibility to Abnormal (Kinetotic) Swimming Fish Correlates with Inner Ear Carbonic Anhydrase-Reactivity. Neurosci. Lett. 2002, 335, 17–20. [Google Scholar] [CrossRef]
- Hilbig, R.; Anken, R.H.; Rahmann, H. On the Origin of Susceptibility to Kinetotic Swimming Behaviour in Fish: A Parabolic Aircraft Flight Study. J. Vestib. Res. 2003, 12, 185–189. [Google Scholar] [CrossRef]
- Dunkelberger, D.G.; Dean, J.M.; Watabe, N. The Ultrastructure of the Otolithic Membrane and Otolith in the Juvenile Mummichog, Fundulus heteroclitus. J. Morphol. 1980, 163, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, P.; Mahe, K.; Elleboode, R.; Chantre, C.; Murta, A.; Avila De Melo, A.; Cabral, H. Blue Whiting Otoliths Pair’s Symmetry Side Effect. Int. J. Fish. Aquat. Stud. 2017, 5, 06–09. [Google Scholar]
- Torres, G.J.; Lombarte, A.; Morales-Nin, B. Variability of the Sulcus Acusticus in the Sagittal Otolith of the Genus Merluccius (Merlucciidae). Fish. Res. 2000, 46, 5–13. [Google Scholar] [CrossRef]
- Chollet-Villalpando, J.G.; García-Rodríguez, F.J.; De Luna, E.; De La Cruz-Agüero, J. Geometric Morphometrics for the Analysis of Character Variation in Size and Shape of the Sulcus acusticus of Sagittae Otolith in Species of Gerreidae (Teleostei: Perciformes). Mar. Biodiv. 2019, 49, 2323–2332. [Google Scholar] [CrossRef]
- D’Iglio, C.; Famulari, S.; Albano, M.; Carnevale, A.; Fresco, D.D.; Costanzo, M.; Lanteri, G.; Spanò, N.; Savoca, S.; Capillo, G. Intraspecific Variability of the Saccular and Utricular Otoliths of the Hatchetfish Argyropelecus hemigymnus (Cocco, 1829) from the Strait of Messina (Central Mediterranean Sea). PLoS ONE 2023, 18, e0281621. [Google Scholar] [CrossRef] [PubMed]

| Type of Data | Response Variable | Explanatory Variable | χ2 | Df | p-Values |
|---|---|---|---|---|---|
| 2D | PCs | PCs | 436,088.849 | 24 | <0.001 *** |
| PCs:SI | 27.216 | 24 | 0.290 | ||
| PCs:GSA | 921.882 | 48 | <0.001 *** | ||
| PCs:SI:GSA | 66.359 | 48 | 0.087 | ||
| 3D | PCs | PCs | 0.289 | 15 | 0.134 |
| PCs:SI | 42.967 | 15 | <0.001 *** | ||
| PCs:GSA | 83.682 | 30 | <0.001 *** | ||
| PCs:SI:GSA | 67.176 | 30 | <0.001 *** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andrialovanirina, N.; Caillault, É.P.; Couette, S.; Laffont, R.; Poloni, L.; Lutet-Toti, C.; Mahé, K. Asymmetry of Sagittal Otolith Shape Based on Inner Ear Side Tested on Mediterranean Red Mullet (Mullus barbatus Linnaeus, 1758): Comparative Analysis of 2D and 3D Otolith Shape Data. Symmetry 2023, 15, 1067. https://doi.org/10.3390/sym15051067
Andrialovanirina N, Caillault ÉP, Couette S, Laffont R, Poloni L, Lutet-Toti C, Mahé K. Asymmetry of Sagittal Otolith Shape Based on Inner Ear Side Tested on Mediterranean Red Mullet (Mullus barbatus Linnaeus, 1758): Comparative Analysis of 2D and 3D Otolith Shape Data. Symmetry. 2023; 15(5):1067. https://doi.org/10.3390/sym15051067
Chicago/Turabian StyleAndrialovanirina, Nicolas, Émilie Poisson Caillault, Sébastien Couette, Rémi Laffont, Lauriane Poloni, Camille Lutet-Toti, and Kélig Mahé. 2023. "Asymmetry of Sagittal Otolith Shape Based on Inner Ear Side Tested on Mediterranean Red Mullet (Mullus barbatus Linnaeus, 1758): Comparative Analysis of 2D and 3D Otolith Shape Data" Symmetry 15, no. 5: 1067. https://doi.org/10.3390/sym15051067
APA StyleAndrialovanirina, N., Caillault, É. P., Couette, S., Laffont, R., Poloni, L., Lutet-Toti, C., & Mahé, K. (2023). Asymmetry of Sagittal Otolith Shape Based on Inner Ear Side Tested on Mediterranean Red Mullet (Mullus barbatus Linnaeus, 1758): Comparative Analysis of 2D and 3D Otolith Shape Data. Symmetry, 15(5), 1067. https://doi.org/10.3390/sym15051067







