Theoretical Analysis of a COVID-19 CF-Fractional Model to Optimally Control the Spread of Pandemic
Abstract
1. Introduction
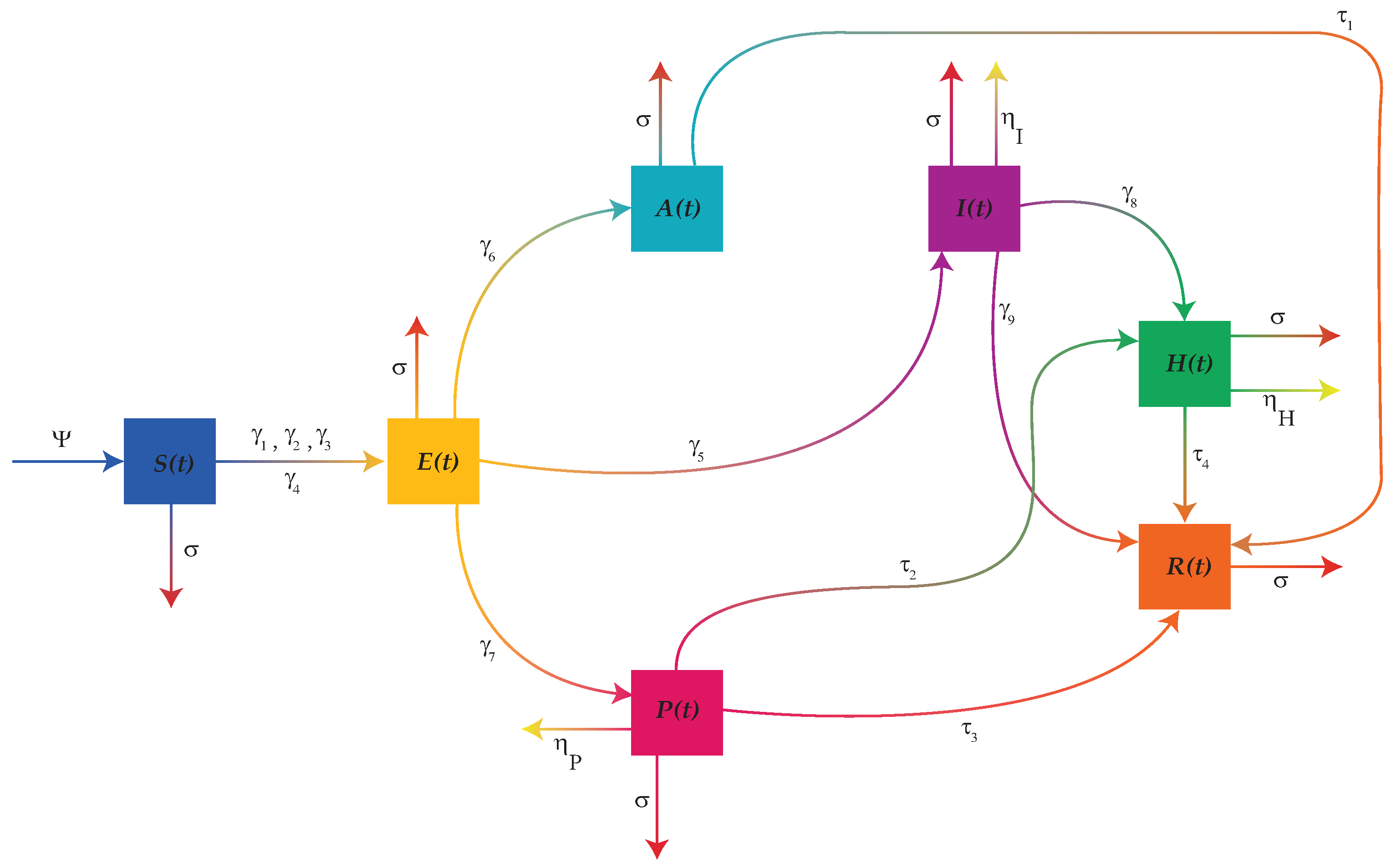
2. Mathematical Model
3. Fundamental and Biological Properties
3.1. Existence and Uniqueness
3.2. Boundedness and Positivity of the Solutions
3.3. Equilibrium Points
3.4. Basic Reproduction Number
4. Stability Analysis
4.1. Local Behavior of the Model
4.2. Global Behavior of the Model
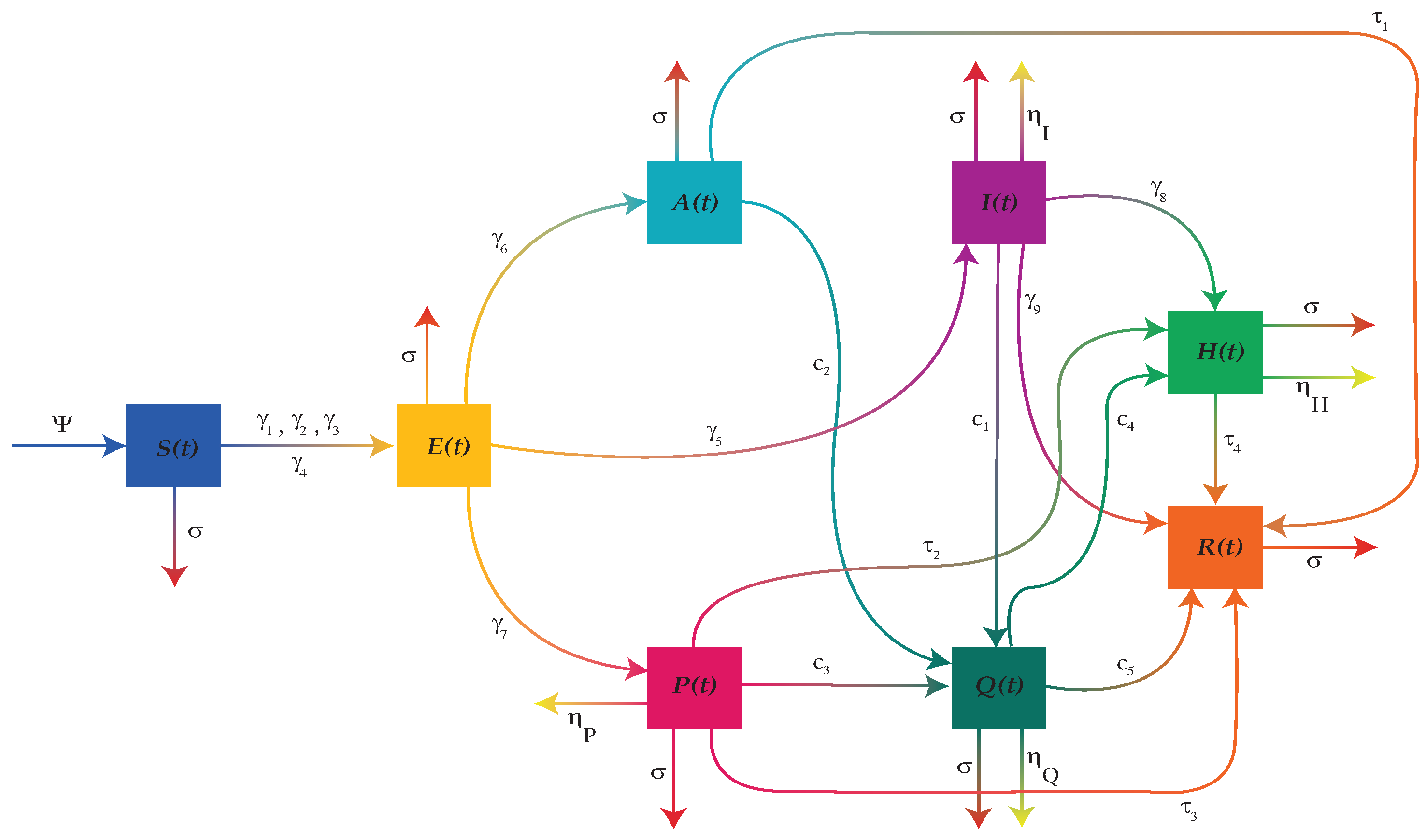
5. Updated Model with the Quarantine Compartment
5.1. Solution Approximating Technique
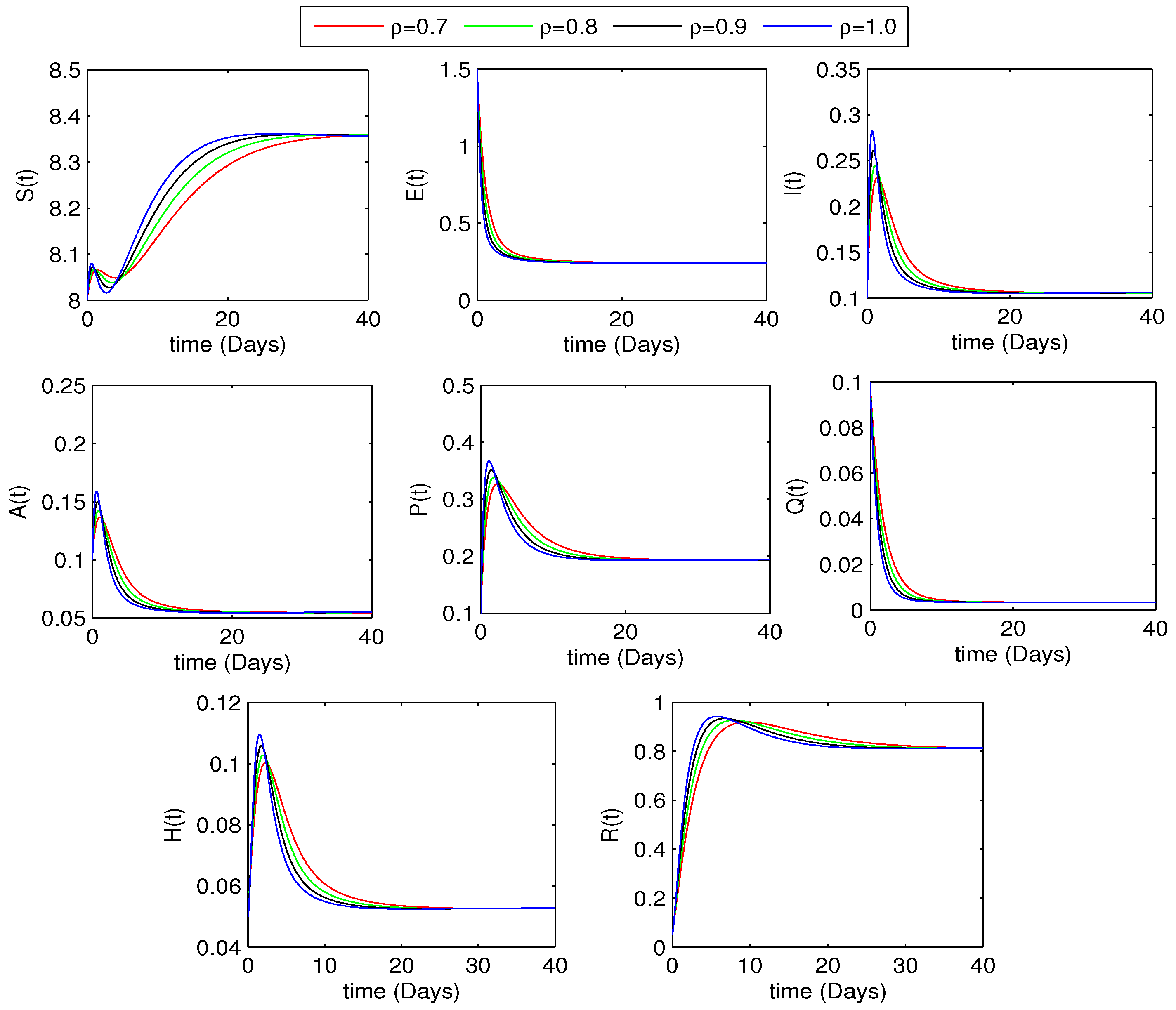
5.2. Effect of the Fractional Order on Disease Dynamics
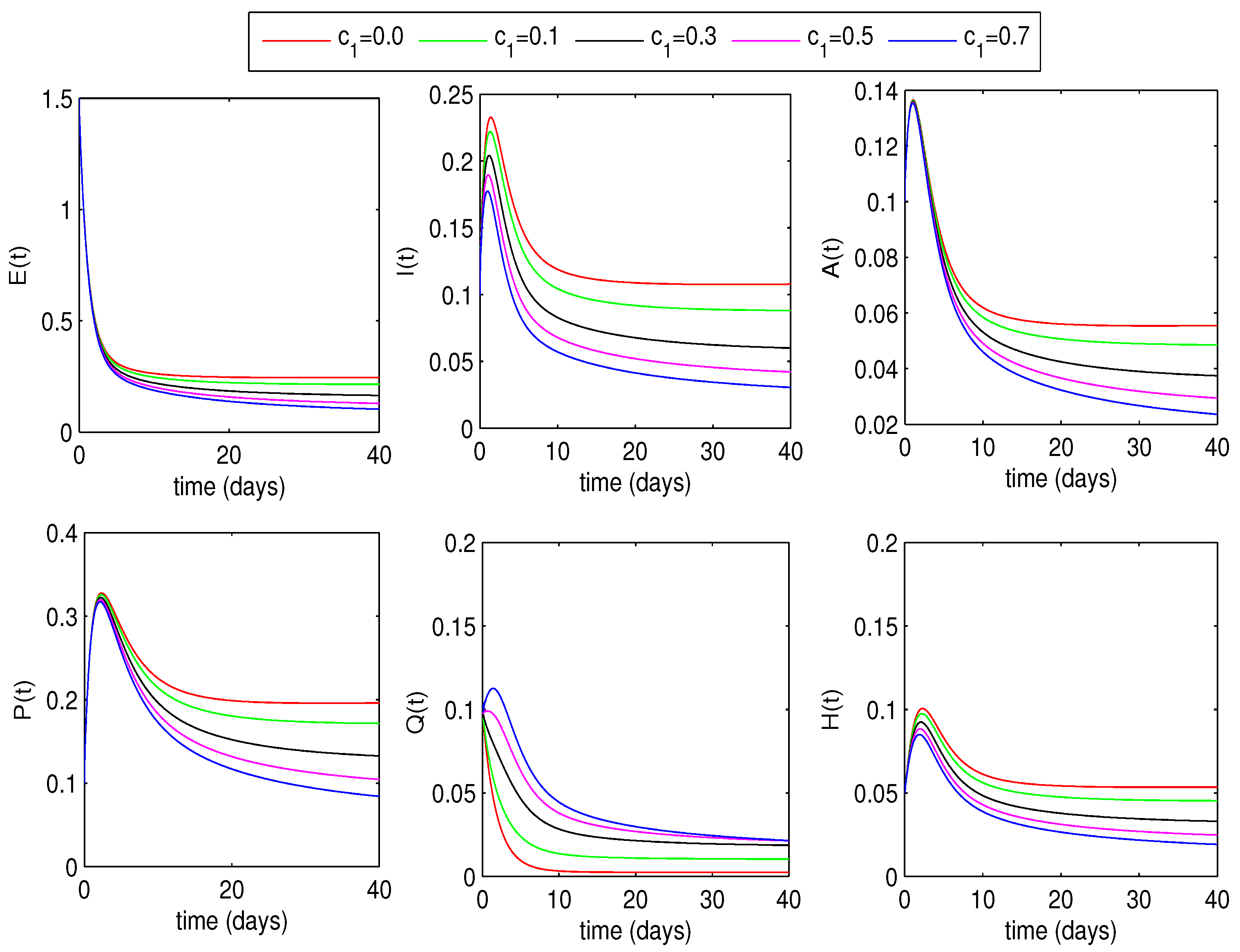
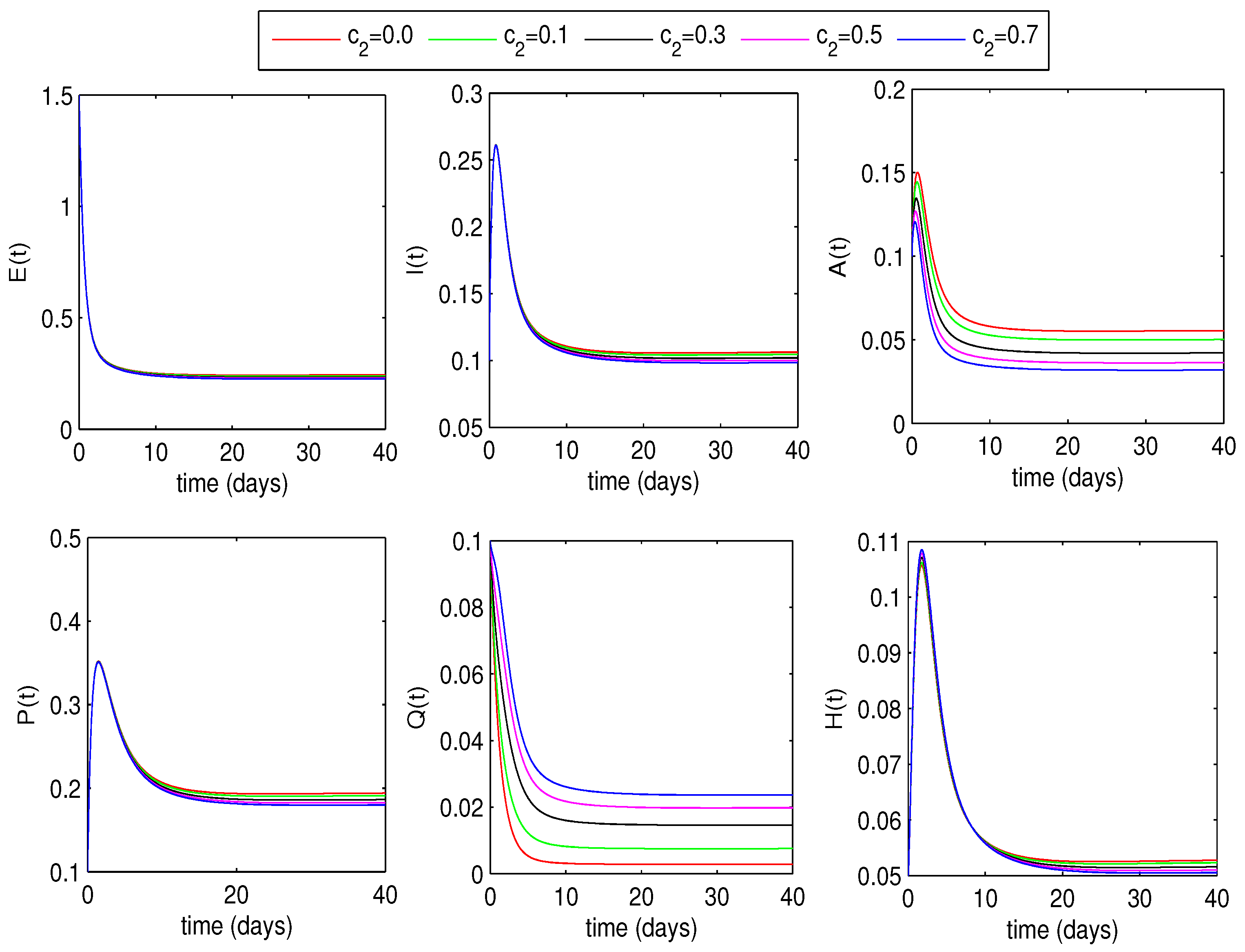
5.3. Effect of Quarantine Rates on Disease Dynamics
6. Formulation of an Optimal Control Problem
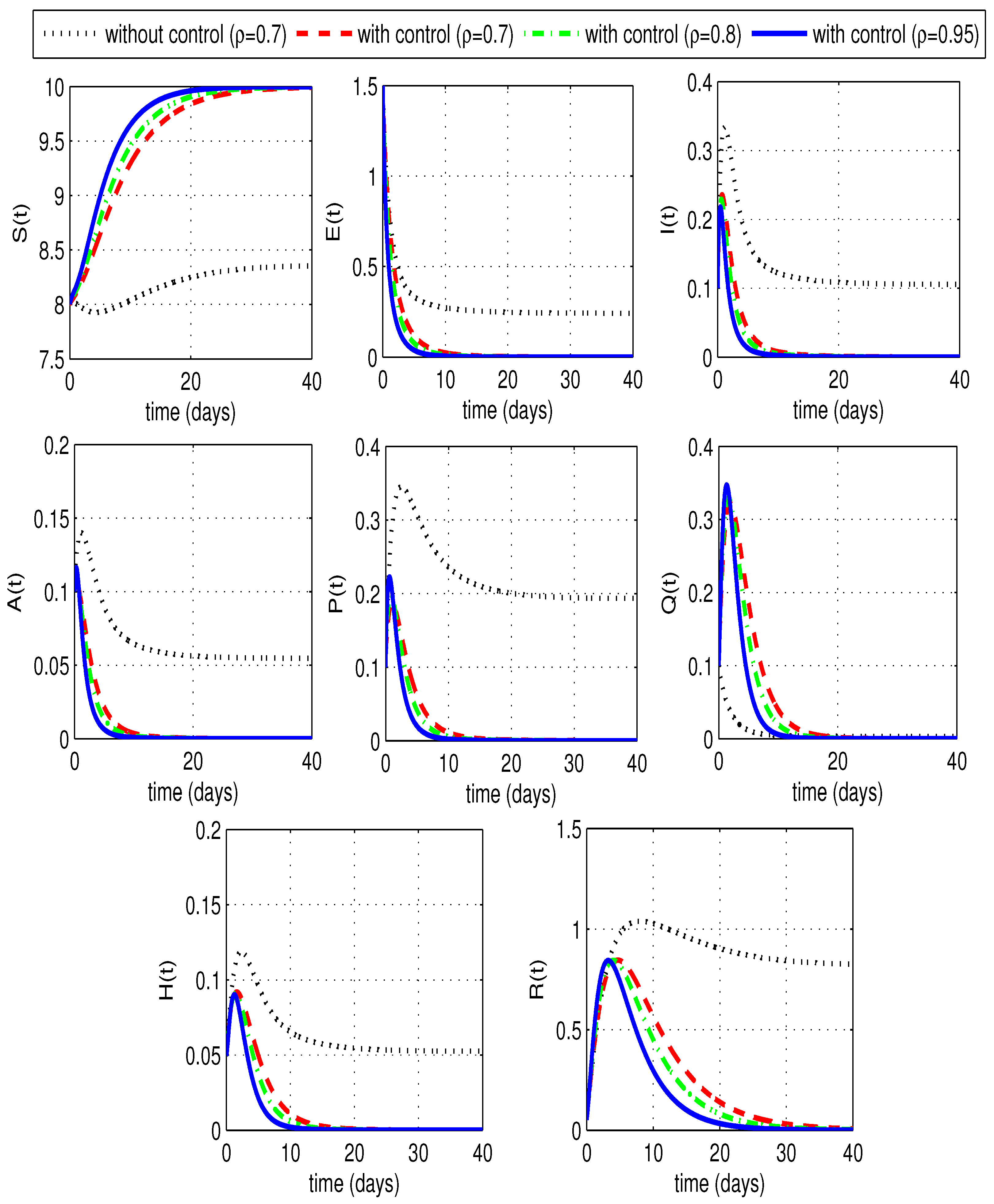
6.1. Numerical Study of an Optimal Control Problem
6.1.1. Solution Algorithm
| Algorithm 1: |
|
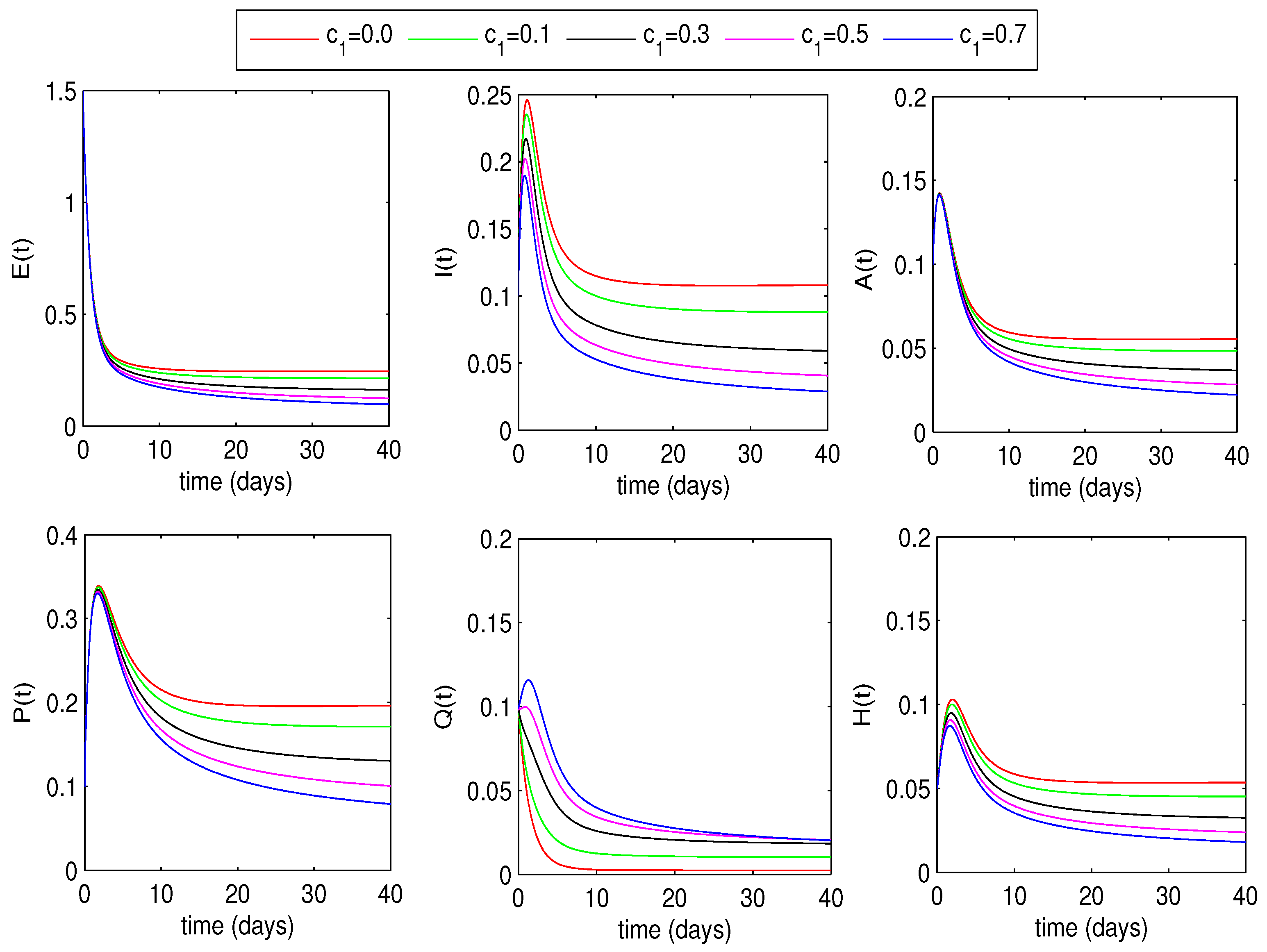
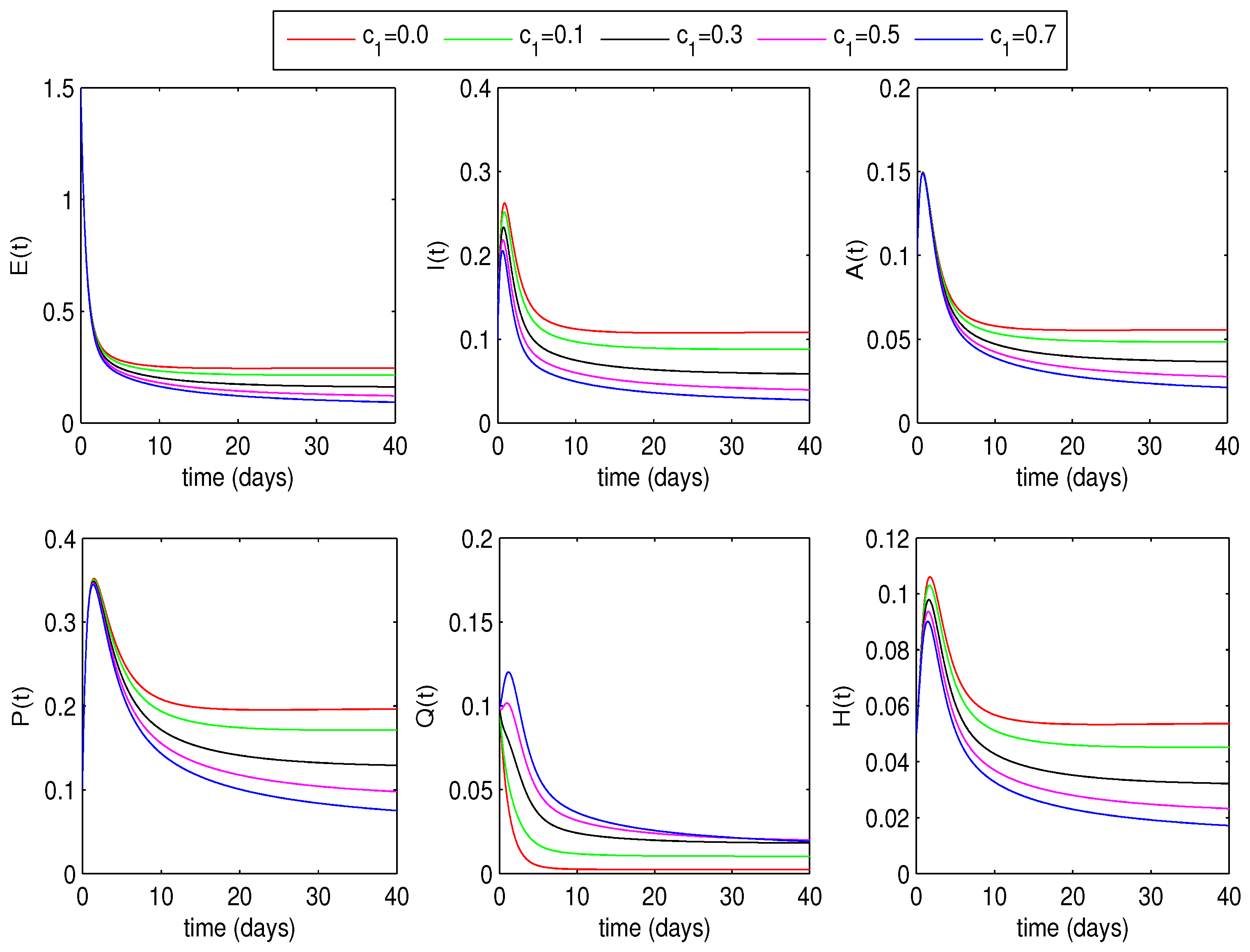
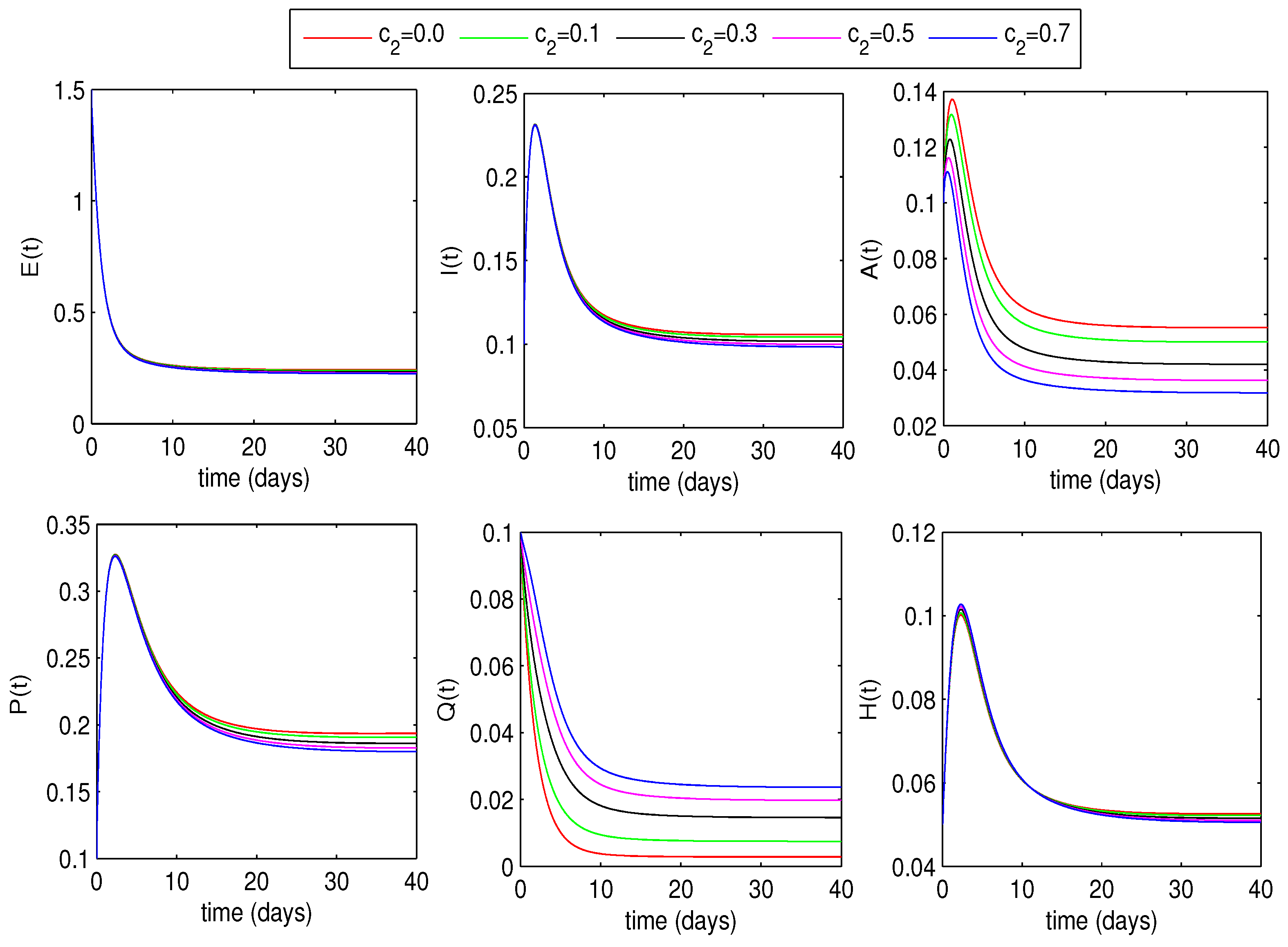
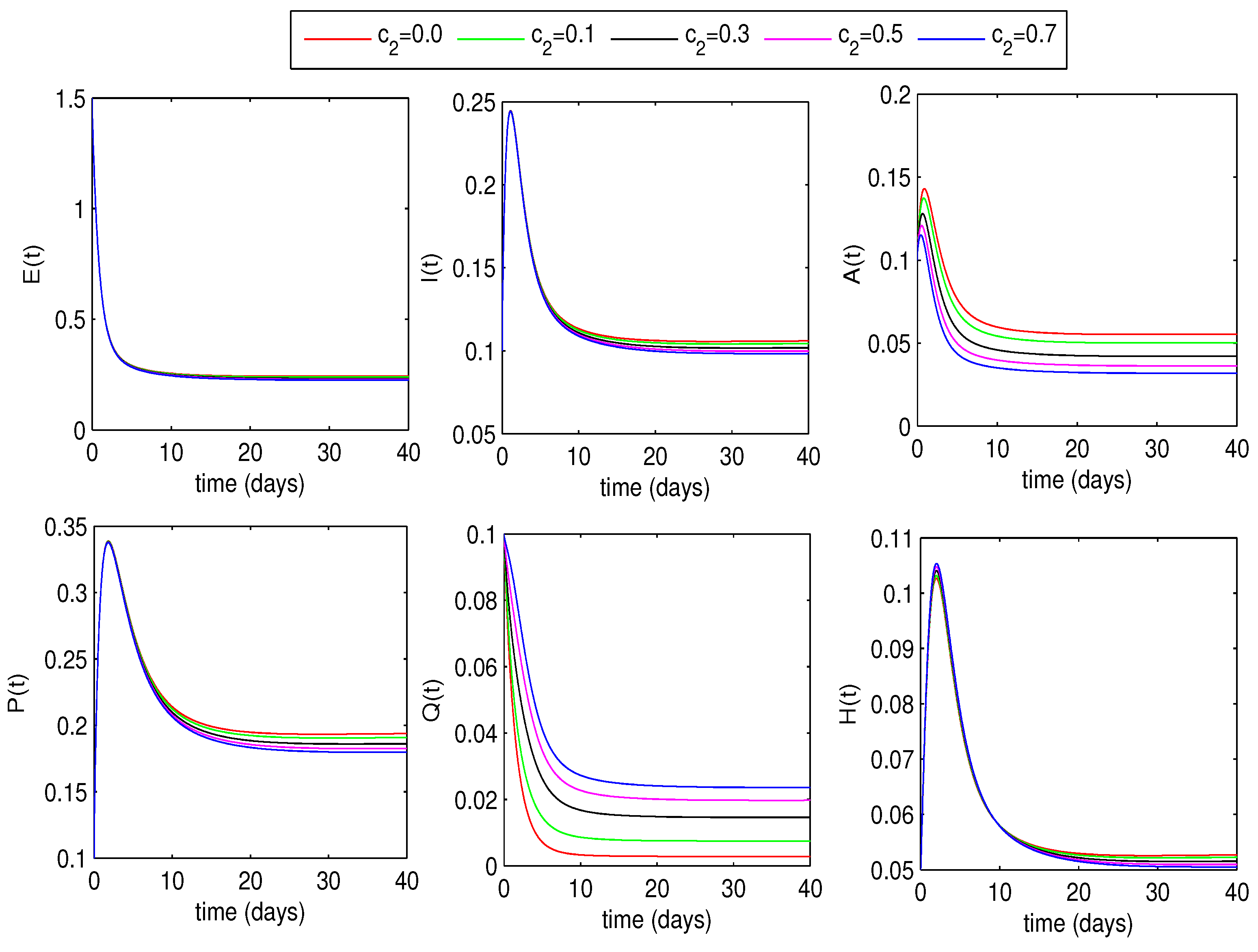
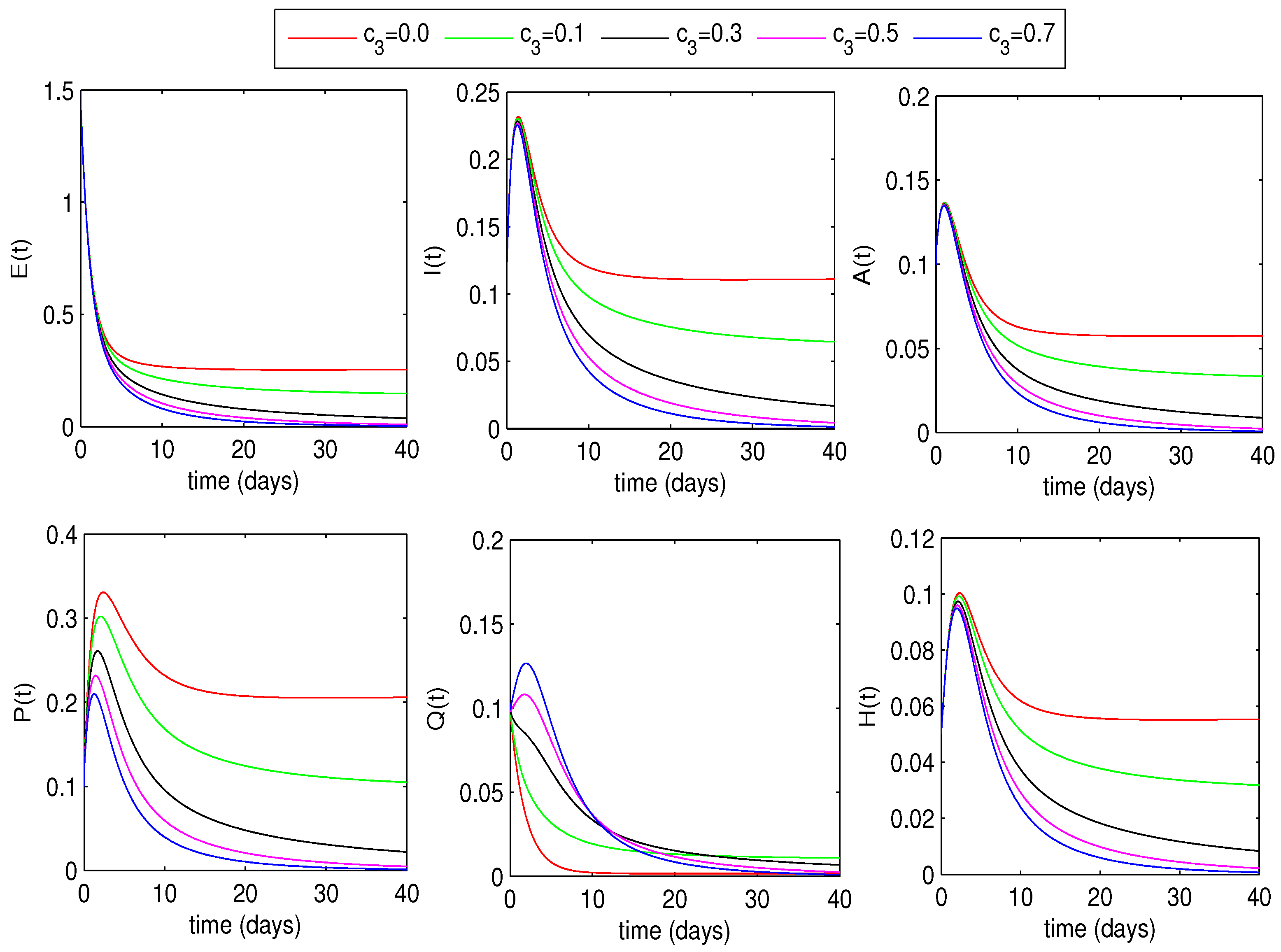
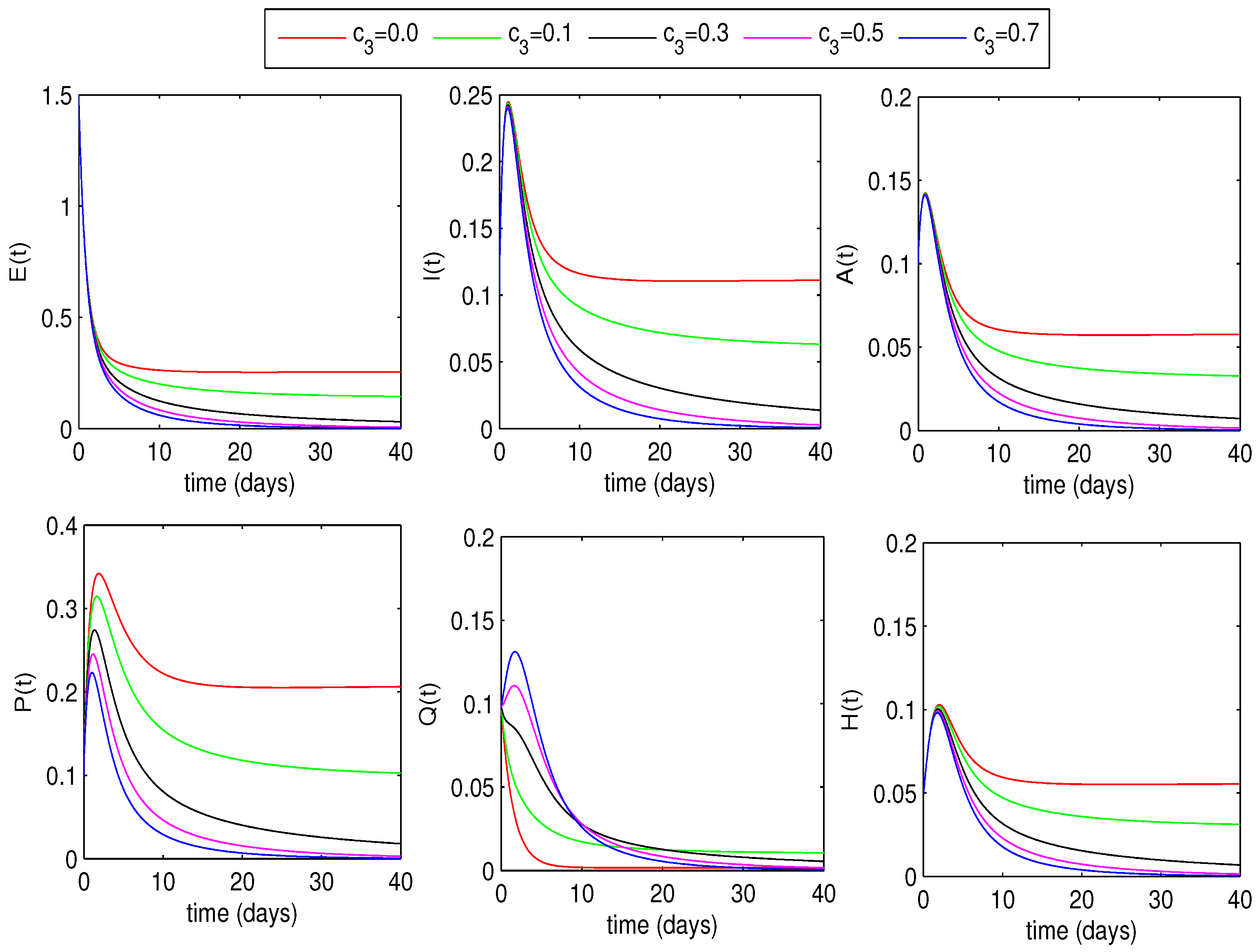
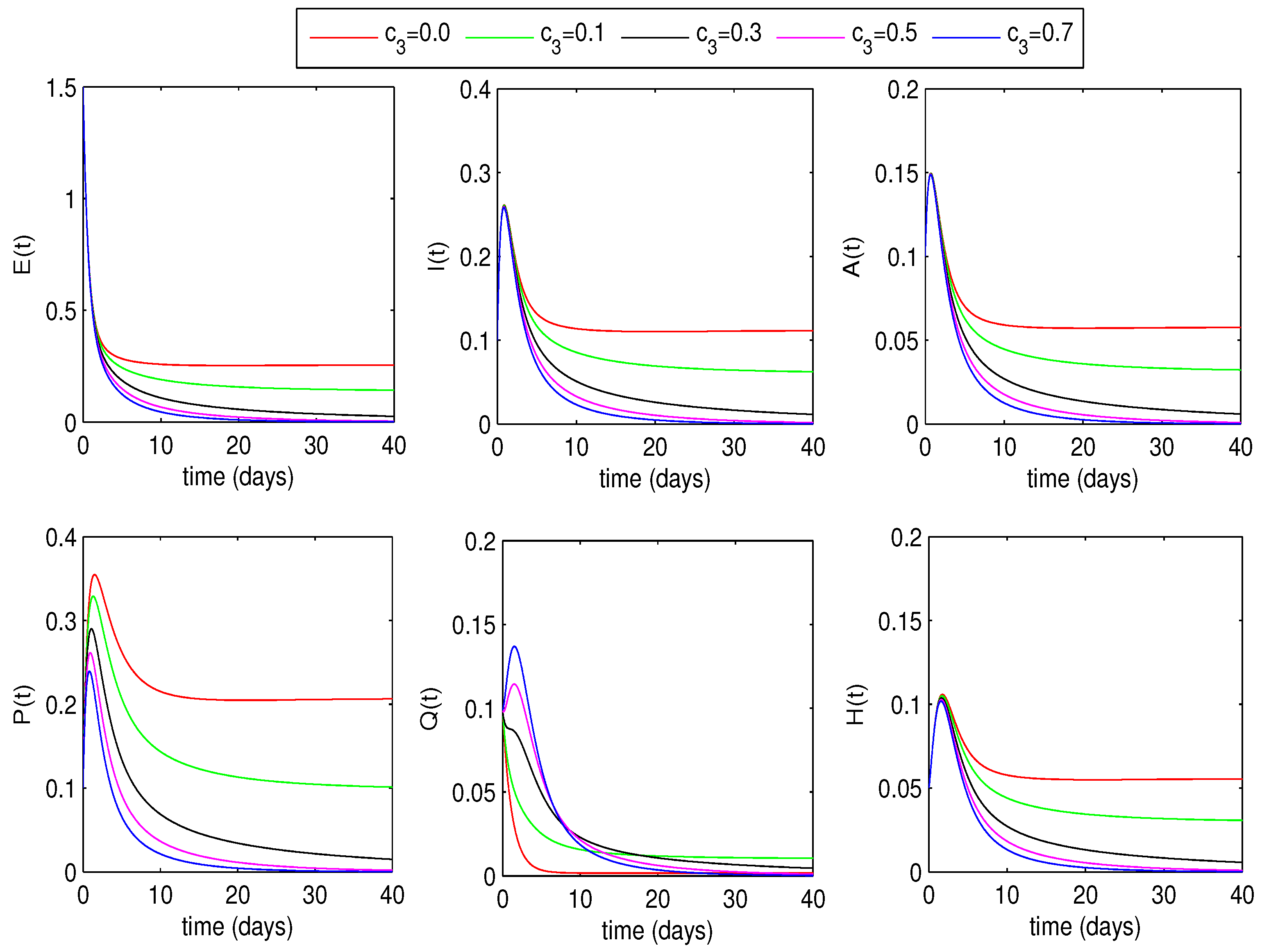
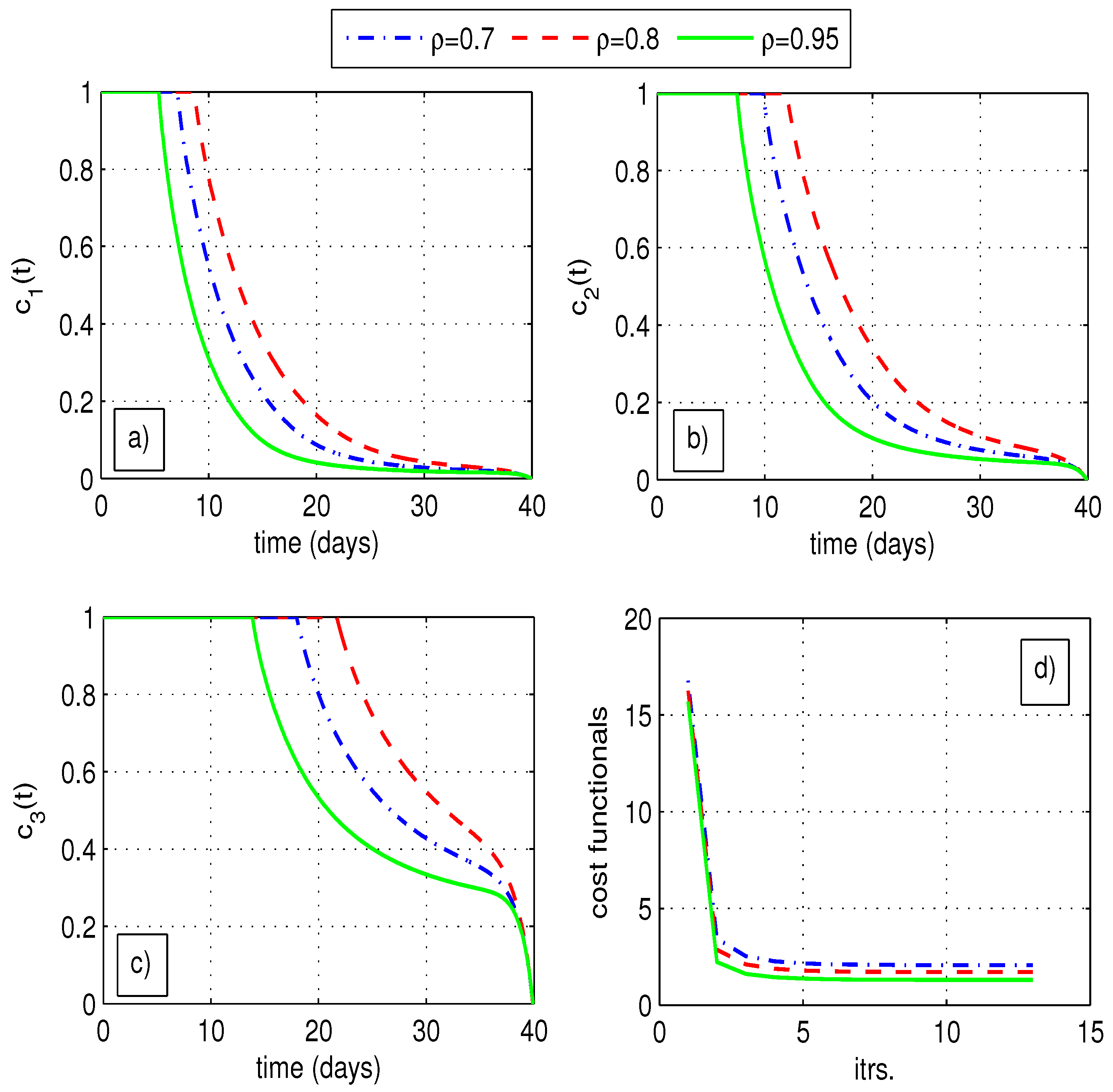
6.1.2. Optimal Quarantine Rates
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World of Health Organization. Novel Coronavirus (2019-nCoV)-SITUATION REPORT. Available online: https://apps.who.int/iris/bitstream/handle/10665/330760/nCoVsitrep21Jan2020-eng.pdf?sequence=3&isAllowed=y (accessed on 1 January 2023).
- Bikdeli, B.; Madhavan, M.V.; Jimenez, D.; Chuich, T.; Dreyfus, I.; Driggin, E.; Nigoghossian, C.D.; Ageno, W.; Madjid, M.; Guo, Y.; et al. COVID-19 and thrombotic or thromboembolic disease: Implications for prevention, antithrombotic therapy, and follow-up. J. Am. Cardiol. 2020, 75, 2950–2973. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.-Y.; He, S.; Rong, L.-B.; Tang, S.-Y. The effect of control measures on COVID-19 transmission in Italy: Comparison with Guangdong province in China. Infect. Dis. Poverty 2020, 130, 9. [Google Scholar] [CrossRef] [PubMed]
- Mekonen, K.G.; Balcha, S.F.; Obsu, L.L.; Hassen, A. Mathematical Modeling and Analysis of TB and COVID- 19 Coinfection. J. Appl. Math. 2022, 2022, 2449710. [Google Scholar] [CrossRef]
- Liang, T. Handbook of COVID-19 prevention and treatment, The First Affiliated Hospital, Zhejiang University School of Medicine. Compil. Accord. Clin. Exp. 2020, 68. [Google Scholar]
- Ahmad, W.; Abbas, M.; Rafiq, M.; Baleanu, D. Mathematical analysis for the effect of voluntary vaccination on the propagation of Corona virus pandemic. Results Phys. 2021, 31, 104917. [Google Scholar] [CrossRef]
- Hui, D.S.; Azhar, E.I.; Madani, T.A.; Ntoumi, F.; Kock, R.; Dar, O.; Petersen, E. The continuing 2019-nCoV epidemic threat of novel corona viruses to global health: The latest 2019 novel coronavirus outbreak in Wuhan, China. Int. J. Infect. Dis. 2020, 91, 264–266. [Google Scholar] [CrossRef]
- Volpert, V.; Banerjee, M.; Petrovskii, S. On a quarantine model of Coronavirus infection and data analysis. Math. Model. Nat. Phenom. 2020, 15, 24. [Google Scholar] [CrossRef]
- Mohsen, A.A.; L-Husseiny, H.F.A.; Zhou, X.; Hattaf, K. Global stability of COVID-19 model involving the quarantine strategy and media coverage effects. AIMS Public Health 2020, 7, 587–605. [Google Scholar] [CrossRef]
- De la Sen, M.; Ibeas, A.; Agarwal, R.P. On confinement and quarantine concerns on an SEIAR epidemic model with simulated parameterizations for the COVID-19 pandemic. Symmetry 2020, 12, 1646. [Google Scholar] [CrossRef]
- Chowdhury, A.; Kabir, K.M.A.; Tanimoto, J. How quarantine and social distancing policy can suppress the outbreak of novel coronavirus in developing or under poverty level countries: A mathematical and statistical analysis. Res. Sq. 2020. [Google Scholar] [CrossRef]
- Hui, D.S.; Azhar, E.I.; Kim, Y.-J.; Memish, Z.A.; Oh, M.-D.; Zulma, A. Middle East respiratory syndrome coronavirus: Risk factors and determinants of primary, household, and nosocomial transmission. Lancet Infect. Dis. 2018, 18, 217–227. [Google Scholar] [CrossRef]
- Wilder-Smith, A.; Freedman, D.O. Isolation, quarantine, social distancing and community containment: Pivotal role for old-style public health measures in the novel coronavirus (2019-ncov) outbreak. J. Travel Med. 2020, 27, 1–4. [Google Scholar] [CrossRef]
- Rafiq, M.; Macias-Diaz, J.E.; Raza, A.; Ahmed, N. Design of a nonlinear model for the propagation of COVID-19 and its efficient nonstandard computational implementation. Appl. Math. Model. 2021, 89, 1835–1846. [Google Scholar] [CrossRef]
- Gao, Q.; Zhuang, J.; Wu, T.; Shen, H. Transmission dynamics and quarantine control of COVID-19 in cluster community: A new transmission-quarantine model with case study for diamond princess. Math. Model. Methods Appl. Sci. 2021, 31, 619–648. [Google Scholar] [CrossRef]
- Marshall, P. The impact of quarantine on COVID-19 infections. Epidemiol. Methods 2021, 10, 20200038. [Google Scholar] [CrossRef]
- Feng, L.-X.; Jing, S.L.; Hu, S.-K.; Wang, D.-F.; Huo, H.-F. Modelling the effects of media coverage and quarantine on the COVID-19 infections in the UK. Math. Biosci. Eng. 2020, 17, 3618–3636. [Google Scholar] [CrossRef]
- Aronna, M.S.; Guglielmi, R.; Moschen, L.M. A model for COVID-19 with isolation, quarantine and testing as control measures. Epidemics 2021, 34, 100437. [Google Scholar] [CrossRef]
- Acuna-Zegarra, M.A.; Diaz-Infanteb, S.; Baca-Carrasco, D.; Liceaga, D.O. COVID-19 optimal vaccination policies: A modeling study on efficacy, natural and vaccine-induced immunity responses. medRxiv 2020. [Google Scholar] [CrossRef]
- Belete, T.M. A review on Promising vaccine development progress for COVID-19 disease. Vacunas 2020, 21, 121–128. [Google Scholar] [CrossRef]
- Kaur, S.P.; Gupta, V. COVID-19 Vaccine: A comprehensive status report. Virus Res. 2020, 288, 198114. [Google Scholar] [CrossRef]
- Shah, A.; Marks, P.W. Unwavering Regulatory Safeguards for COVID-19 Vaccines. J. Am. Med. Assoc. 2020, 324, 931–932. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, P. Russia Says Its Sputnik v Covid-19 Vaccine is 92% Effective. Press Release 11 November 2020. Available online: https://www.reuters.com/article/us-health-coronavirus-russia-vaccine-idCAKBN27R0Z6 (accessed on 1 January 2023).
- Cohen, E. Moderna’s Coronavirus Vaccine is 94.5% Effective, According to Company Data. Press Release Updated 16 November 2020. Available online: https://edition.cnn.com/2020/11/16/health/moderna-vaccine-results-coronavirus/index.html (accessed on 1 January 2023).
- Yang, W. Modeling COVID-19 pandemic with hierarchical quarantine and time delay. Dyn. Games Appl. 2021, 11, 892–914. [Google Scholar] [CrossRef] [PubMed]
- Ajbar, A.; Alqahtani, R.T.; Boumaza, M. Dynamics of a COVID-19 model with a nonlinear incidence rate, quarantine, media effects, and number of hospital beds. Symmetry 2021, 13, 947. [Google Scholar] [CrossRef]
- Podlubny, I. Fractional differential equations: An introduction to fractional derivatives. In Fractional Differential Equations, to Methods of Their Solution and Some of Their Applications; Elsevier: Amsterdam, The Netherlands, 1999. [Google Scholar]
- Samko, S.G.; Kilbas, A.A.; Marichev, O.I. Fractional Integrals and Derivatives: Theory and Applications; Gordon and Breach Science Publishers: Philadelphia, PA, USA, 1993. [Google Scholar]
- Khan, M.F.; Alrabaiah, H.; Ullah, S.; Khan, M.A.; Farooq, M.; Mamat, M.B.; Asjad, M.I. A new fractional model for vector-host disease with saturated treatment function via singular and non-singular operators. Alex. Eng. J. 2021, 60, 629–645. [Google Scholar] [CrossRef]
- Peter, O.J.; Oguntolu, F.A.; Ojo, M.; Oyeniyi, A.O.; Jan, R.; Khan, I. Fractional order mathematical model of monkeypox transmission dynamics. Phys. Scr. 2022, 97, 084005. [Google Scholar] [CrossRef]
- Hadi, M.S.; Bilgehan, B. Fractional COVID-19 Modeling and Analysis on Successive Optimal Control Policies. Fractal Fract. 2022, 6, 533. [Google Scholar] [CrossRef]
- Shaikh, A.S.; Shaikh, I.N.; Nisar, K.S. A mathematical model of COVID-19 using fractional derivative: Outbreak in India with dynamics of transmission and control. Adv. Differ. Equations 2020, 2020, 373. [Google Scholar] [CrossRef]
- Ali, Z.; Rabiei, F.; Rashidi, M.M.; Khodadadi, T. A fractional-order mathematical model for COVID-19 outbreak with the effect of symptomatic and asymptomatic transmissions. Eur. Phys. J. Plus 2022, 137, 395. [Google Scholar] [CrossRef]
- Tunç, O.; Tunç, C. Solution estimates to Caputo proportional fractional derivative delay integro-differential equations. Rev. R. Acad. Cienc. Exactas Fís. Nat. Ser. A Mat. RACSAM 2023, 117, 12. [Google Scholar] [CrossRef]
- Tunç, C.; Tunç, O.; Yao, J.-C. On the new qualitative results in integro-differential equations with Caputo fractional derivative and multiple kernels and delays. J. Nonlinear Convex Anal. 2022, 23, 2577–2591. [Google Scholar]
- Butt, A.I.K.; Chamaleen, M.I.D.B.D.; B, S. Optimal control strategies for the reliable and competitive mathematical analysis of COVID-19 pandemic model. Math. Meth. Appl. Sci. 2023, 46, 1528–1555. [Google Scholar] [CrossRef]
- Losada, J.; Nieto, J.J. Properties of a new fractional derivative without singular kernel. Prog. Fract. Differ. Appl. 2015, 1, 87–92. [Google Scholar]
- Caputo, M.; Fabrizio, M. A new definition of fractional derivative without singular kernel. Prog. Fract. Differ. Appl. 2015, 1, 73–85. [Google Scholar]
- Hanif, A.; Butt, A.I.K.; Ahmad, S.; Din, R.U.; Mustafa Inc. A new fuzzy fractional-order model of transmission of COVID-19 with quarantine class. Eur. Phys. J. Plus 2021, 136, 1179. [Google Scholar] [CrossRef]
- Ahmad, W.; Abbas, M. Effect of quarantine on transmission dynamics of Ebola virus epidemic: A mathematical analysis. Eur. J. Plus 2021, 136, 355. [Google Scholar] [CrossRef]
- Ahmad, W.; Rafiq, M.; Abbas, M. Mathematical analysis to control the spread of Ebola virus epidemic through voluntary vaccination. Eur. Phys. J. Plus 2020, 135, 775. [Google Scholar] [CrossRef]
- Rafiq, M.; Ahmad, W.; Abbas, M.; Baleanu, D. A reliable and competitive mathematical analysis of Ebola epidemic model. Adv. Differ. Equations 2020, 2020, 540. [Google Scholar] [CrossRef]
- Butt, A.I.K.; Abbas, M.; Ahmad, W. A mathematical analysis of an isothermal tube drawing process. Alexanderia Eng. J. 2020, 59, 3419–3429. [Google Scholar] [CrossRef]
- Castillo-Chavez, C.; Feng, Z.; Huanz, W.; Driessche, P.V.D.; Kirschner, D.E. On the computation of RO and its role in global stability. In Mathematical Approaches for Emerging and Reemerging Infectious Diseases: An Introduction; Springer: Berlin/Heidelberg, Germany, 2002. [Google Scholar]
- Thabet, S.T.M.; Abdo, M.S.; Shah, K. Theoretical and numerical analysis for transmission dynamics of COVID-19 mathematical model involving Caputo-Fabrizio derivative. Adv. Differ. Equ. 2021, 2021, 184. [Google Scholar] [CrossRef]
- Vargas-De-Len, C. Volterra-type Lyapunov functions for fractional-order epidemic systems. Commun. Nonlinear Sci. Numer. Simul. 2015, 24, 75–85. [Google Scholar] [CrossRef]
- Moore, E.J.; Sirisubtawee, S.; Koonprasert, S. A Caputo-Fabrizio fractional differential equation model for HIV/AIDS with treatment compartment. Adv. Differ. Equations 2019, 2019, 200. [Google Scholar] [CrossRef]
- Lenhart, S.; Workman, J.T. Optimal Control Applied to Biological Models; Chapman & Hall/CRC: Boca Raton, FL, USA, 2007. [Google Scholar]
- Wang, H.; Jahanshahi, H.; Wang, M.K.; Bekiros, S.; Liu, J.; Aly, A.A. A Caputo-Fabrizio fractional-order model of HIV/AIDS with a treatment compartment: Sensitivity analysis and optimal control strategies. Entropy 2021, 23, 610. [Google Scholar] [CrossRef] [PubMed]
| Parameter | Description | Values | Reference |
|---|---|---|---|
| Transmission rate of S to E after contacting I | 0.866 | [36] | |
| Transmission rate of S to E after contacting A | 0.16 | [36,39] | |
| Transmission rate of S to E after contacting P | 0.8 | [36] | |
| Transmission rate of S to E after contacting H | 0.0131 | [36,39] | |
| Translation of E to I | 0.235 | [36,39] | |
| Translation of E to A | 0.26 | [36,39] | |
| Translation of E to P | 0.56 | [36] | |
| Translation of I to H | 0.45 | [36,39] | |
| Translation of I to R | 0.6381 | [36,39] | |
| Translation of A to R | 0.8 | [36,39] | |
| Translation of P to H | 0.1 | [36] | |
| Translation of P to R | 0.3 | [36] | |
| Translation of H to R | 0.54 | [36,39] | |
| Death induced by disease in I | 0.08 | [36] | |
| Death induced by disease in P | 0.092 | [36,39] | |
| Death induced by disease in H | 0.485 | [36] | |
| Susceptible recruit rate | 2.5 | [36] | |
| Natural death rate | 0.241 | [36] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Butt, A.I.K.; Imran, M.; Batool, S.; Nuwairan, M.A. Theoretical Analysis of a COVID-19 CF-Fractional Model to Optimally Control the Spread of Pandemic. Symmetry 2023, 15, 380. https://doi.org/10.3390/sym15020380
Butt AIK, Imran M, Batool S, Nuwairan MA. Theoretical Analysis of a COVID-19 CF-Fractional Model to Optimally Control the Spread of Pandemic. Symmetry. 2023; 15(2):380. https://doi.org/10.3390/sym15020380
Chicago/Turabian StyleButt, Azhar Iqbal Kashif, Muhammad Imran, Saira Batool, and Muneerah AL Nuwairan. 2023. "Theoretical Analysis of a COVID-19 CF-Fractional Model to Optimally Control the Spread of Pandemic" Symmetry 15, no. 2: 380. https://doi.org/10.3390/sym15020380
APA StyleButt, A. I. K., Imran, M., Batool, S., & Nuwairan, M. A. (2023). Theoretical Analysis of a COVID-19 CF-Fractional Model to Optimally Control the Spread of Pandemic. Symmetry, 15(2), 380. https://doi.org/10.3390/sym15020380





