C2-Symmetric N-Heterocyclic Carbenes in Asymmetric Transition-Metal Catalysis
Abstract
:1. Introduction
2. Discussion
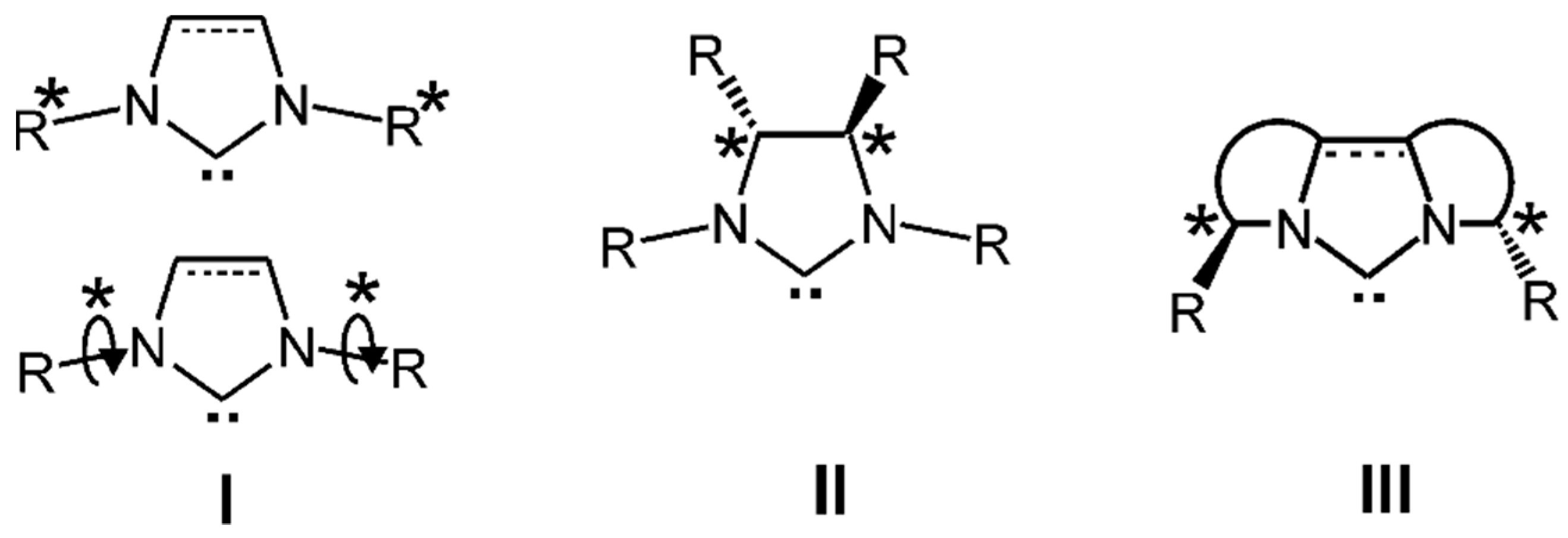
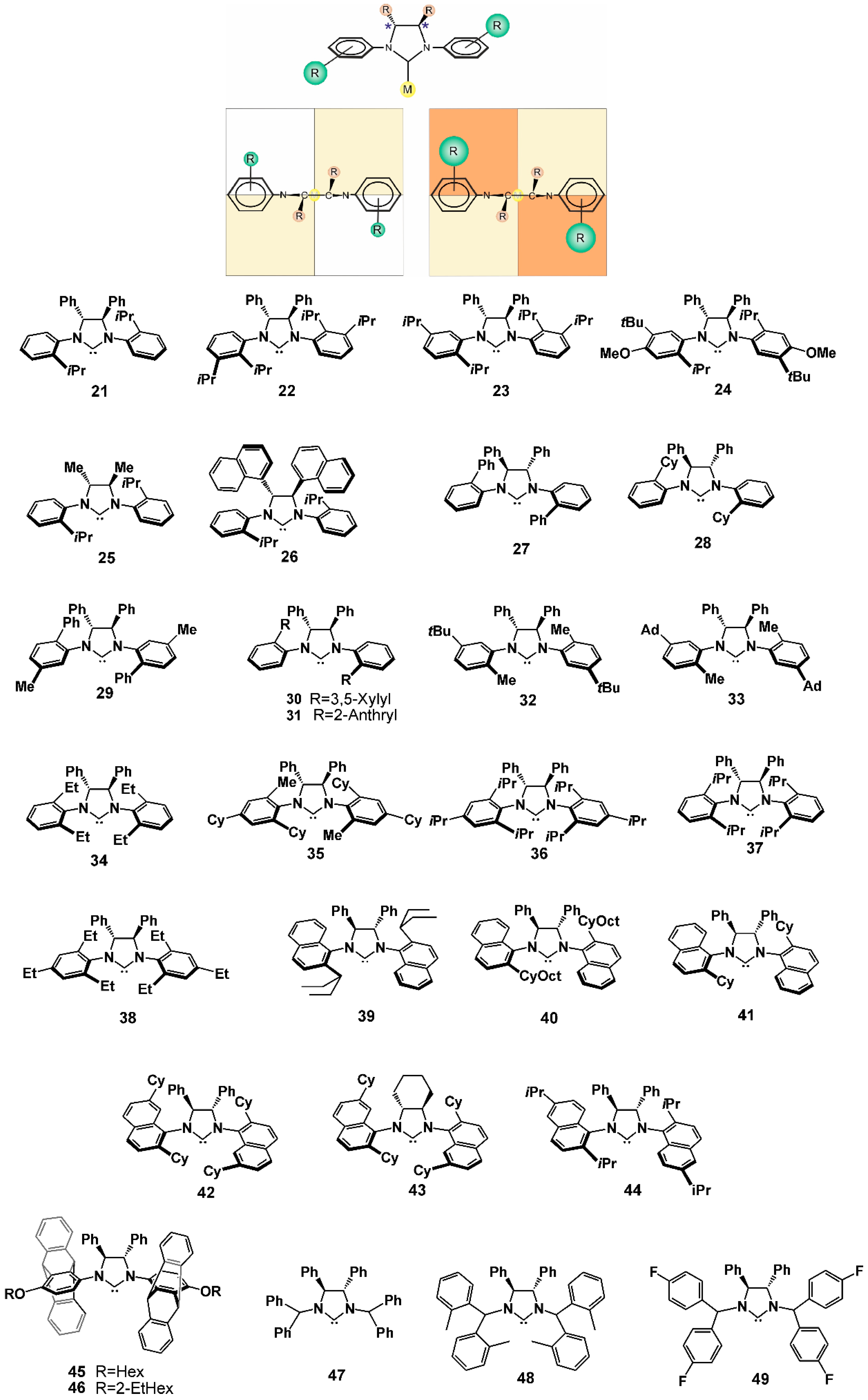
2.1. NHCs with Chiral N-Substituents
2.2. NHCs with Chiral Backbones
| Metal | NHC | Reactions | References |
|---|---|---|---|
| Cu | 28–31,34 44 45,46 47–49 | Conjugate sylil addition Cyanoborylation of allenes Borylation of α, β-unsaturated esters Allylic arylation | [61,62,63] [77] [78] [81,82,83] |
| Fe | 32,33 | C-H alkylation | [66] |
| Ir | 41–43 42 | Intramolecular hydroamination Ring-opening amination | [73,74,75] [76] |
| Ni | 21 26 27 28,38 34,35 36 | Synthesis of benzoxasiloles Three-component coupling Intermolecular [2 + 2 + 2] cycloaddition of enones with alkynes Cross-hydroalkenylation Desymmetrization and following [4 + 2] cycloaddition Reductive coupling of aldehydes and alkynes | [64] [60] [65] [70] [67] [68] |
| Pd | 37 39,40 | [3 + 2] cycloaddition of vinyl epoxides with allenic amides α-arylation of amides | [69] [71,72] |
| Ru | 21–25 | Ring-closing metathesis | [56,58,59] |
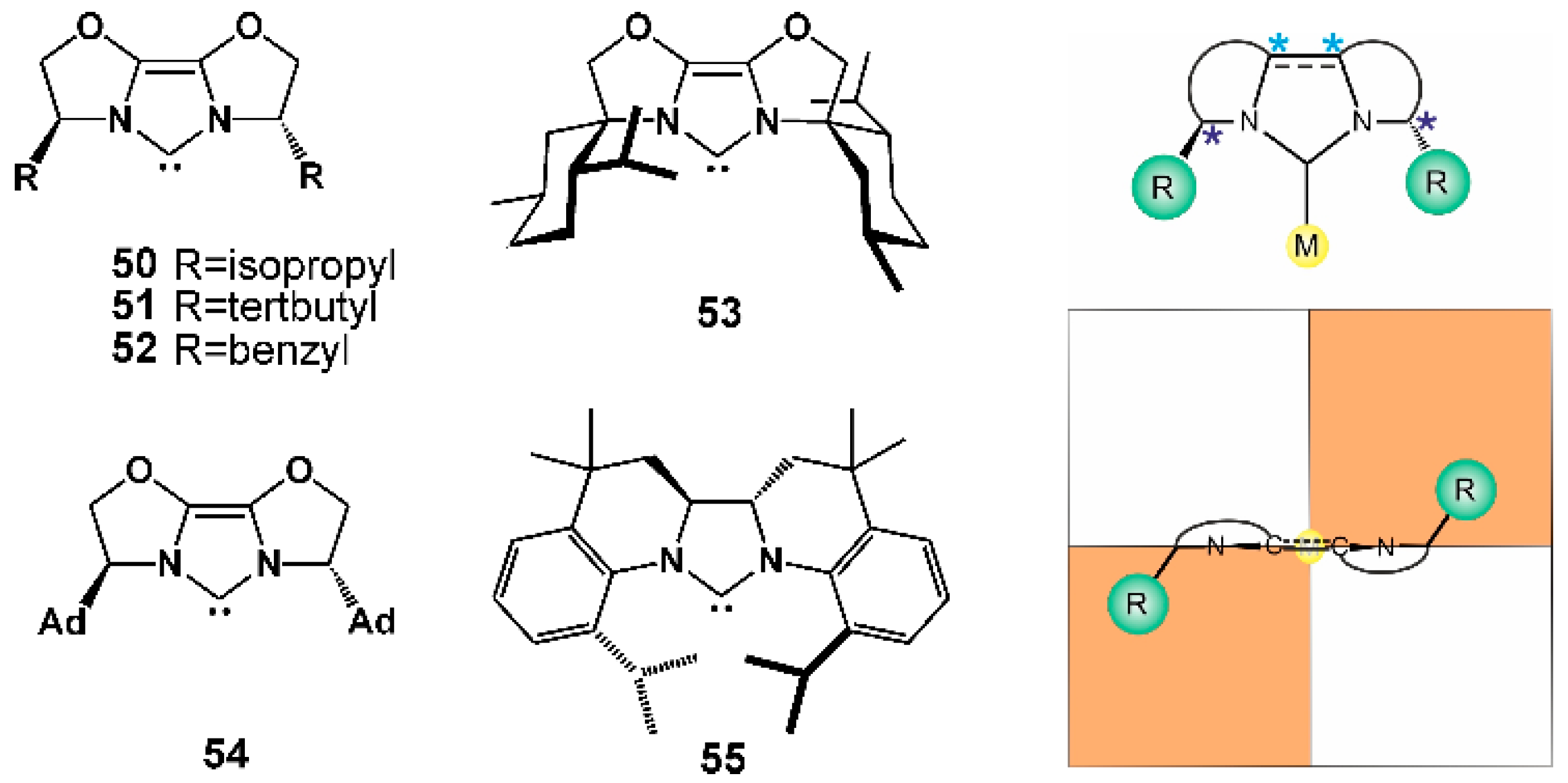
2.3. NHCs with Fused Cycles
3. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nolan, S.P. N-Heterocyclic Carbenes in Synthesis; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- Hopkinson, M.N.; Richter, C.; Schedler, M.; Glorius, F. An Overview of N-Heterocyclic Carbenes. Nature 2014, 510, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.C.Y.; Huang, R.T.W.; Lee, C.S.; Bhattacharyya, A.; Hwang, W.S.; Lin, I.J.B. Coinage Metal-N-Heterocyclic Carbene Complexes. Chem. Rev. 2009, 109, 3561–3598. [Google Scholar] [CrossRef] [PubMed]
- Nelson, D.J.; Nolan, S.P. Quantifying and understanding the electronic properties of N-heterocyclic carbenes. Chem. Soc. Rev. 2013, 42, 6723–6753. [Google Scholar] [CrossRef]
- Gómez-Suárez, A.; Nelson, D.J.; Nolan, S.P. Quantifying and Understanding the Steric Properties of N-Heterocyclic Carbenes. Chem. Commun. 2017, 53, 2650–2660. [Google Scholar] [CrossRef]
- Nolan, S.P. N-Heterocyclic Carbenes: Effective Tools for Organometallic Synthesis, 1st ed.; Wiley-VCH: Mannheim, Germany, 2014. [Google Scholar]
- Glorius, F. N-Heterocyclic Carbenes in Transition Metal Catalysis, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Huynh, H.V. The Organometallic Chemistry of N-Heterocyclic Carbenes, 1st ed.; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Peris, E. Smart N-Heterocyclic Carbene Ligands in Catalysis. Chem. Rev. 2018, 118, 9988–10031. [Google Scholar] [CrossRef]
- Visbal, R.; Gimeno, M.C. N-Heterocyclic Carbene Metal Complexes: Photoluminescence and Applications. Chem. Soc. Rev. 2014, 43, 3551–3574. [Google Scholar] [CrossRef]
- Velazquez, H.D.; Verpoort, F. N-Heterocyclic Carbene Transition Metal Complexes for Catalysis in Aqueous Media. Chem. Soc. Rev. 2012, 41, 7032–7060. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Meng, G.; Nolan, S.P.; Szostak, M. N-Heterocyclic Carbene Complexes in C-H Activation Reactions. Chem. Rev. 2020, 120, 1981–2048. [Google Scholar] [CrossRef] [PubMed]
- César, V.; Bellemin-Laponnaz, S.; Gade, L.H. Chiral N-heterocyclic carbenes as stereodirecting ligands in asymmetric catalysis. Chem. Soc. Rev. 2004, 33, 619–636. [Google Scholar] [CrossRef]
- Janssen-Müller, D.; Schlepphorst, C.; Glorius, F. Privileged chiral N-heterocyclic carbene ligands for asymmetric transition-metal catalysis. Chem. Soc. Rev. 2017, 46, 4845–4854. [Google Scholar] [CrossRef]
- Budagumpi, S.; Keri, R.S.; Achar, G.; Brinda, K.N. Coinage Metal Complexes of Chiral N-Heterocyclic Carbene Ligands: Syntheses and Applications in Asymmetric Catalysis. Adv. Synth. Catal. 2020, 362, 970–997. [Google Scholar] [CrossRef]
- Foster, D.; Borhanuddin, S.M.; Dorta, R. Designing successful monodentate N-heterocyclic carbene ligands for asymmetric metal catalysis. Dalton Trans. 2021, 50, 17467–17477. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, W.A.; Goossen, L.J.; Kocher, C.; Artus, G.R.J. Chiral Heterocylic Carbenes in Asymmetric Homogeneous Catalysis. Angew. Chem., Int. Ed. Engl. 1996, 35, 2805–2807. [Google Scholar] [CrossRef]
- Enders, D.; Gielen, H.; Raabe, G.; Runsink, J.; Teles, J.H. Synthesis and Stereochemistry of the First Chiral (Imidazolinylidene)- and (Triazolinylidene)palladium(ii) Complexes. Chem. Ber. 1996, 129, 1483–1488. [Google Scholar] [CrossRef]
- Alexakis, A.; Winn, C.L.; Guillen, F.; Pytkowicz, J.; Roland, S.; Mangeney, P. Asymmetric Synthesis with N-Heterocyclic Carbenes. Application to the Copper-Catalyzed Conjugate Addition. Adv. Synth. Catal. 2003, 345, 345–348. [Google Scholar] [CrossRef]
- Urban, S.; Ortega, N.; Glorius, F. Ligand-Controlled Highly Regioselective and Asymmetric Hydrogenation of Quinoxalines Catalyzed by Ruthenium N-Heterocyclic Carbene Complexes. Angew. Chem. Int. Ed. 2011, 50, 3803–3806. [Google Scholar] [CrossRef]
- Ortega, N.; Urban, S.; Beiring, B.; Glorius, F. Ruthenium NHC Catalyzed Highly Asymmetric Hydrogenation of Benzofurans. Angew. Chem. Int. Ed. 2012, 51, 1710–1713. [Google Scholar] [CrossRef] [PubMed]
- Ortega, N.; Beiring, B.; Urban, S.; Glorius, F. Highly Asymmetric Synthesis of (+)-corsifuran A. Elucidation of the Electronic Requirements in the Ruthenium NHC Catalyzed Hydrogenation of Benzofurans. Tetrahedron 2012, 68, 5185–5192. [Google Scholar] [CrossRef]
- Urban, S.; Beiring, B.; Ortega, N.; Paul, D.; Glorius, F. Asymmetric Hydrogenation of Thiophenes and Benzothiophenes. J. Am. Chem. Soc. 2012, 134, 15241–15244. [Google Scholar] [CrossRef]
- Zhao, D.; Beiring, B.; Glorius, F. Ruthenium–NHC-Catalyzed Asymmetric Hydrogenation of Flavones and Chromones: General Access to Enantiomerically Enriched Flavanones, Flavanols, Chromanones, and Chromanols. Angew. Chem. Int. Ed. 2013, 52, 8454–8458. [Google Scholar] [CrossRef]
- Wysocki, J.; Ortega, N.; Glorius, F. Asymmetric Hydrogenation of Disubstituted Furans. Angew. Chem. Int. Ed. 2014, 53, 8751–8755. [Google Scholar] [CrossRef] [PubMed]
- Schlepphorst, C.; Wiesenfeldt, M.P.; Glorius, F. Enantioselective Hydrogenation of Imidazo[1,2-a]pyridines. Chem.-Eur. J. 2018, 24, 356–359. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wiesenfeldt, M.P.; Glorius, F. Ruthenium–NHC–Diamine Catalyzed Enantioselective Hydrogenation of Isocoumarins. J. Am. Chem. Soc. 2017, 139, 2585–2588. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Wagener, T.; Hellmann, L.; Daniliuc, C.G.; Mück-Lichtenfeld, C.; Neugebauer, J.; Glorius, F. Design of Ru(II)-NHC-Diamine Precatalysts Directed by Ligand Cooperation: Applications and Mechanistic Investigations for Asymmetric Hydrogenation. J. Am. Chem. Soc. 2020, 142, 7100–7107. [Google Scholar] [CrossRef]
- Sato, Y.; Hinata, Y.; Seki, R.; Oonishi, Y.; Saito, N. Nickel-Catalyzed Enantio- and Diastereoselective Three-Component Coupling of 1,3-Dienes, Aldehydes, and Silanes Using Chiral N-Heterocyclic Carbenes as Ligands. Org. Lett. 2007, 9, 5597–5599. [Google Scholar] [CrossRef]
- Kündig, P.; Seidel, T.; Jia, Y.; Bernardinelli, G. Bulky Chiral Carbene Ligands and Their Application in the Palladium-Catalyzed Asymmetric Intramolecular a-Arylation of Amides. Angew. Chem. Int. Ed. 2007, 46, 8484–8487. [Google Scholar] [CrossRef]
- Jia, Y.-X.; Hillgren, J.M.; Watson, E.L.; Mardsen, S.P.; Kündig, E.P. Chiral N-heterocyclic carbene ligands for asymmetric catalytic oxindole synthesis. Chem. Commun. 2008, 34, 4040–4042. [Google Scholar] [CrossRef]
- Nakanishi, M.; Katayev, D.; Besnard, C.; Kündig, E.P. Fused Indolines by Palladium-Catalyzed Asymmetric C-C Coupling Involving an Unactivated Methylene Group. Angew. Chem. Int. Ed. 2011, 50, 7438–7441. [Google Scholar] [CrossRef]
- Katayev, D.; Nakanishi, M.; Bürgi, T.; Kündig, E.P. Asymmetric C(sp3)-H/C(Ar) coupling reactions. Highly enantio-enriched indolines via regiodivergent reaction of a racemic mixtur. Chem. Sci. 2012, 3, 1422–1425. [Google Scholar] [CrossRef]
- Katayev, D.; Larionov, E.; Nakanishi, M.; Besnard, C.; Kündig, E.P. Palladium-N-Heterocyclic Carbene (NHC)-Catalyzed Asymmetric Synthesis of Indolines through Regiodivergent C(sp3)—H Activation: Scope and DFT Study. Chem.-Eur. J. 2014, 20, 15021–15030. [Google Scholar] [CrossRef]
- Yeung, K.; Ruscoe, R.E.; Rae, J.; Pulis, A.P.; Procter, D.J. Enantioselective Generation of Adjacent Stereocenters in a Copper-Catalyzed Three-Component Coupling of Imines, Allenes, and Diboranes. Angew. Chem., Int. Ed. 2016, 55, 11912–11916. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; Ohmiya, H.; Sawamura, M. Enantioselective Conjugate Addition of Alkylboranes Catalyzed by a Copper–N-Heterocyclic Carbene Complex. J. Am. Chem. Soc. 2012, 134, 11896–11899. [Google Scholar] [CrossRef] [PubMed]
- Albright, A.; Eddings, D.; Black, R.; Welch, C.J.; Gerasimchuk, N.N.; Gawley, R.E. Design and Synthesis of C2-Symmetric N-Heterocyclic Carbene Precursors and Metal Carbenoids. J. Org. Chem. 2011, 76, 7341–7351. [Google Scholar] [CrossRef]
- Albright, A.; Gawley, R.E. Application of a C2-Symmetric Copper Carbenoid in the Enantioselective Hydrosilylation of Dialkyl and Aryl–Alkyl Ketones. J. Am. Chem. Soc. 2011, 133, 19680–19683. [Google Scholar] [CrossRef] [PubMed]
- Diesel, J.; Finogenova, A.M.; Cramer, N. Nickel-Catalyzed Enantioselective Pyridone C-H Functionalizations Enabled by a Bulky N-Heterocyclic Carbene Ligand. J. Am. Chem. Soc. 2018, 140, 4489–4493. [Google Scholar] [CrossRef]
- Diesel, J.; Grosheva, D.; Kodama, S.; Cramer, N. A Bulky Chiral N-Heterocyclic Carbene Nickel Catalyst Enables Enantioselective C-H Functionalizations of Indoles and Pyrroles. Angew. Chem. Int. Ed. 2019, 58, 11044–11048. [Google Scholar] [CrossRef]
- Cai, Y.; Yang, X.-T.; Zhang, S.-Q.; Li, F.; Li, Y.-Q.; Ruan, L.-X.; Hong, X.; Shi, S.-L. Copper-Catalyzed Enantioselective Markovnikov Protoboration of α-Olefins Enabled by a Buttressed N-Heterocyclic Carbene Ligand. Angew. Chem. Int. Ed. 2018, 57, 1376–1380. [Google Scholar] [CrossRef]
- Cai, Y.; Zhang, J.-W.; Li, F.; Liu, J.-M.; Shi, S.-L. Nickel/N-Heterocyclic Carbene Complex-Catalyzed Enantioselective Redox-Neutral Coupling of Benzyl Alcohols and Alkynes to Allylic Alcohols. ACS Catal. 2019, 9, 1–6. [Google Scholar] [CrossRef]
- Zhang, W.-B.; Yang, X.-T.; Ma, J.-B.; Su, Z.-M.; Shi, S.-L. Regio- and Enantioselective C–H Cyclization of Pyridines with Alkenes Enabled by a Nickel/N-Heterocyclic Carbene Catalysis. J. Am. Chem. Soc. 2019, 141, 5628–5634. [Google Scholar] [CrossRef]
- Shen, D.; Zhang, W.-B.; Li, Z.; Shi, S.-L.; Xu, Y. Nickel/NHC-Catalyzed Enantioselective Cyclization of Pyridones and Pyrimidones with Tethered Alkenes. Adv. Synth. Catal. 2020, 362, 1125–1130. [Google Scholar] [CrossRef]
- Cai, Y.; Ye, X.; Liu, S.; Shi, S.-L. Nickel/NHC-Catalyzed Asymmetric C-H Alkylation of Fluoroarenes with Alkenes: Synthesis of Enantioenriched Fluorotetralins. Angew. Chem. Int. Ed. 2019, 58, 13433–13437. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.-W.; Li, R.; Li, J.-F.; Sun, J.; Ye, M. NHC ligand-enabled Ni-catalyzed reductive coupling of alkynes and imines using isopropanol as a reductant. Green Chem. 2019, 21, 2240–2244. [Google Scholar] [CrossRef]
- Cai, Y.; Ruan, L.-X.; Rahman, A.; Shi, S.-L. Fast Enantio- and Chemoselective Arylation of Ketones with Organoboronic Esters Enabled by Nickel/N-Heterocyclic Carbene Catalysis. Angew. Chem. Int. Ed. 2021, 60, 5262–5267. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Shi, S.-L. Enantioconvergent Arylation of Racemic Secondary Alcohols to Chiral Tertiary Alcohols Enabled by Nickel/N-Heterocyclic Carbene Catalysis. J. Am. Chem. Soc. 2021, 143, 11963–11968. [Google Scholar] [CrossRef]
- Wang, Z.C.; Xie, P.P.; Xu, Y.; Hong, X.; Shi, S.-L. Low-Temperature Nickel-Catalyzed C−N Cross-Coupling via Kinetic Resolution Enabled by a Bulky and Flexible Chiral N -Heterocyclic Carbene Ligand. Angew. Chem. Int. Ed. 2021, 60, 16077–16084. [Google Scholar] [CrossRef]
- Wang, Z.C.; Gao, J.; Cai, Y.; Ye, X.; Shi, S.-L. Chemo- and Enantioselective Arylation and Alkenylation of Aldehydes Enabled by Nickel/N-Heterocyclic Carbene Catalysis. CCS Chem. 2022, 4, 1169–1179. [Google Scholar] [CrossRef]
- Shen, D.; Xu, Y.; Shi, S.-L. A Bulky Chiral N-Heterocyclic Carbene Palladium Catalyst Enables Highly Enantioselective Suzuki−Miyaura Cross-Coupling Reactions for the Synthesis of Biaryl Atropisomers. J. Am. Chem. Soc. 2019, 141, 14938–14945. [Google Scholar] [CrossRef]
- Ma, Y.; Song, C.; Ma, C.; Sun, Z.; Chai, Q.; Andrus, M. Asymmetric Addition of Aryl Boron Reagents to Enones with Rhodium Dicyclophane Imidazolium Carbene Catalysis. Angew. Chem. Int. Ed. 2003, 42, 5871–5874. [Google Scholar] [CrossRef]
- Song, C.; Ma, C.; Ma, Y.; Feng, W.; Ma, S.; Chai, Q.; Andrus, M. Bis-paracyclophane N-heterocyclic carbene-ruthenium catalyzed asymmetric ketone hydrosilylation. Tetrahedron Lett. 2005, 46, 3241–3244. [Google Scholar] [CrossRef]
- Chen, J.; Duan, W.; Chen, Z.; Ma, M.; Song, C.; Ma, Y. [2.2]Paracyclophane-based carbene–copper catalyst tuned by transannular electronic effects for asymmetric boration. RSC Adv. 2016, 6, 75144–75151. [Google Scholar] [CrossRef]
- Kong, L.; Morvan, J.; Pichon, D.; Joan, M.; Albalat, M.; Vives, T.; Colombel-Rouen, S.; Giorgi, M.; Dorcet, V.; Roisnel, T.; et al. From Prochiral N-Heterocyclic Carbenes to Optically Pure Metal Complexes: New Opportunities in Asymmetric Catalysis. J. Am. Chem. Soc. 2020, 142, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Seiders, T.J.; Ward, D.W.; Grubbs, R.H. Enantioselective Ruthenium-Catalyzed Ring-Closing Metathesis. Org. Lett. 2001, 3, 3225–3228. [Google Scholar] [CrossRef] [PubMed]
- Costabile, C.; Cavallo, L. Origin of Enantioselectivity in the Asymmetric Ru-Catalyzed Metathesis of Olefins. J. Am. Chem. Soc. 2004, 126, 9592–9600. [Google Scholar] [CrossRef]
- Funk, T.W.; Berlin, J.M.; Grubbs, R.H. Highly Active Chiral Ruthenium Catalysts for Asymmetric Ring-Closing Olefin Metathesis. J. Am. Chem. Soc. 2006, 128, 1840–1846. [Google Scholar] [CrossRef] [PubMed]
- Costabile, C.; Mariconda, A.; Cavallo, L.; Longo, P.; Bertolasi, V.; Ragone, F.; Grisi, F. The Pivotal Role of Symmetry in the Ruthenium-Catalyzed Ring-Closing Metathesis of Olefins. Chem. Eur. J. 2011, 17, 8618–8629. [Google Scholar] [CrossRef] [PubMed]
- Ahlin, J.S.E.; Cramer, N. Chiral N Heterocyclic Carbene Ligand Enabled Nickel(0)- Catalyzed Enantioselective Three-Component Couplings as Direct Access to Silylated Indanols. Org. Lett. 2016, 18, 3242–3245. [Google Scholar] [CrossRef]
- Lee, K.S.; Hoveyda, A.H. Enantioselective Conjugate Silyl Additions to Cyclic and Acyclic Unsaturated Carbonyls Catalyzed by Cu Complexes of Chiral N-Heterocyclic Carbenes. J. Am. Chem. Soc. 2010, 132, 2898–2900. [Google Scholar] [CrossRef]
- Lee, K.S.; Wu, H.; Haeffner, F.; Hoveyda, A.H. NHC–Cu-Catalyzed Silyl Conjugate Additions to Acyclic and Cyclic Dienones and Dienoates. Efficient Site-, Diastereo- and Enantioselective Synthesis of Carbonyl-Containing Allylsilanes. Organometallics 2012, 31, 7823–7826. [Google Scholar] [CrossRef]
- Shen, J.J.; Gao, Q.; Wang, G.; Tong, M.; Chen, L.; Xu, S. Cu-NHC-Catalyzed Enantioselective Conjugate Silyl addition to Indol-1-ylacrylate Derivatives. Chem. Sel. 2019, 4, 11358–11361. [Google Scholar] [CrossRef]
- Kumar, R.; Hoshimoto, Y.; Yabuki, H.; Ohashi, M.; Ogoshi, S. Nickel(0)-Catalyzed Enantio- and Diastereoselective Synthesis of Benzoxasiloles: Ligand-Controlled Switching from Inter- to Intramolecular Aryl-Transfer Process. J. Am. Chem. Soc. 2015, 137, 11838–11845. [Google Scholar] [CrossRef]
- Kumar, R.; Tokura, H.; Nishimura, A.; Mori, T.; Hoshimoto, Y.; Ohashi, M.; Ogoshi, S. Nickel(0)/N-Heterocyclic Carbene-Catalyzed Asymmetric [2 + 2 + 2] Cycloaddition of Two Enones and an Alkyne: Access to Cyclohexenes with Four Contiguous StereogenicCenters. Org. Lett. 2015, 17, 6018–6021. [Google Scholar] [CrossRef] [PubMed]
- Loup, J.; Zell, D.; Oliveira, J.C.A.; Keil, H.; Stalke, D.; Ackermann, L. Asymmetric Iron-Catalyzed C-H Alkylation Enabled by Remote Ligand meta-Substitution. Angew. Chem. Int. Ed. 2017, 56, 14197–14201. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Hoshimoto, Y.; Tamai, E.; Ohashi, M.; Ogoshi, S. Two-step synthesis of chiral fused tricyclic scaffolds from phenols via desymmetrization on nickel. Nat. Commun. 2017, 8, 32. [Google Scholar] [CrossRef]
- Weng, H.; Lu, G.; Sormunen, G.J.; Malik, H.A.; Liu, P.; Montgomery, J. NHC Ligands Tailored for Simultaneous Regio- and Enantiocontrol in Nickel-Catalyzed Reductive Couplings. J. Am. Chem. Soc. 2017, 139, 9317–9324. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.Y.; Wu, J.Y.; Liu, Q.R.; Liu, X.Y.; Ding, C.H.; Hou, X.L. Palladium/N-Heterocyclic Carbene (NHC)-Catalyzed Asymmetric [3 + 2] Cycloaddition Reaction of Vinyl Epoxides with Allenic Amides. Org. Lett. 2018, 20, 4773–4776. [Google Scholar] [CrossRef]
- Chen, Y.; Dang, L.; Ho, C.Y. NHC-Ni catalyzed enantioselective synthesis of 1,4-dienes by cross-hydroalkenylation of cyclic 1,3-dienes and heterosubstituted terminal olefins. Nat. Commun. 2020, 11, 1–6. [Google Scholar] [CrossRef]
- Luan, X.; Wu, L.; Drinkel, E.; Mariz, R.; Gatti, M.; Dorta, R. Highly Chemo- and Enantioselective Synthesis of 3-Allyl-3-aryl Oxindoles via the Direct Palladium-Catalyzedα-Arylation of Amides. Org. Lett. 2010, 12, 1912–1915. [Google Scholar] [CrossRef]
- Wu, L.; Falivene, L.; Drinkel, E.; Grant, S.; Linden, A.; Cavallo, L.; Dorta, R. Synthesis of 3-Fluoro-3-aryl Oxindoles: Direct Enantioselective α Arylation of Amides. Angew. Chem. Int. Ed. 2012, 51, 2870–2873. [Google Scholar] [CrossRef]
- Sipos, G.; Ou, A.; Skelton, B.; Falivene, L.; Cavallo, L.; Dorta, R. Unusual NHC–Iridium(I) Complexes and Their Use in the Intramolecular Hydroamination of Unactivated Aminoalkenes. Chem.-Eur. J. 2016, 22, 6939–6946. [Google Scholar] [CrossRef]
- Gao, P.; Sipos, G.; Foster, D.; Dorta, R. Developing NHC-Iridium Catalysts for the Highly Efficient Enantioselective Intramolecular Hydroamination Reaction. ACS Catal. 2017, 7, 6060–6064. [Google Scholar] [CrossRef]
- Foster, D.; Gao, P.; Zhang, Z.; Sipos, G.; Sobolev, A.N.; Nealon, G.; Falivene, L.; Cavallo, L.; Dorta, R. Design, scope and mechanism of highly active and selective chiral NHC–iridium catalysts for the intramolecular hydroamination of a variety of unactivatedaminoalkenes. Chem. Sci. 2021, 12, 3751–3767. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Foster, D.; Sipos, G.; Skelton, B.W.; Sobolev, A.N.; Dorta, R. Chiral NHC-Iridium Complexes and Their Performance in Enantioselective Intramolecular Hydroamination and Ring-Opening Amination Reactions. Organometallics 2020, 39, 556–573. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, L.; Nishiura, M.; Luo, G.; Luo, Y.; Hou, Z. Enantioselective Cyanoborylation of Allenes by N-Heterocyclic Carbene-Copper Catalysts. ACS Catal. 2020, 10, 11685–11692. [Google Scholar] [CrossRef]
- Savka, R.; Bergmann, M.; Kanai, Y.; Foro, S.; Plenio, H. Triptycene-Based Chiral and meso-N-Heterocyclic Carbene Ligands and Metal Complexes. Chem. Eur. J. 2016, 22, 9667–9675. [Google Scholar] [CrossRef]
- Guillen, F.; Winn, C.L.; Alexakis, A. Enantioselective copper-catalyzed conjugate addition using chiral diaminocarbene ligands. Tetrahedron Asymmetry 2001, 12, 2083–2086. [Google Scholar] [CrossRef]
- Grisi, F.; Costabile, C.; Gallo, E.; Mariconda, A.; Tedesco, C.; Longo, P. Ruthenium-Based Complexes Bearing Saturated Chiral N-Heterocyclic Carbene Ligands: Dynamic Behavior and Catalysis. Organometallics 2008, 27, 4649–4656. [Google Scholar] [CrossRef]
- Selim, K.B.; Matsumoto, Y.; Yamada, K.; Tomioka, K. Efficient Chiral N-Heterocyclic Carbene/Copper(I)-Catalyzed Asymmetric Allylic Arylation with Aryl Grignard Reagents. Angew. Chem. Int. Ed. 2009, 48, 8733–8735. [Google Scholar] [CrossRef]
- Selim, K.B.; Nakanishi, H.; Matsumoto, Y.; Yamamoto, Y.; Yamada, K.; Tomioka, K. Chiral N-Heterocyclic Carbene−Copper(I)-Catalyzed Asymmetric Allylic Arylation of Aliphatic Allylic Bromides: Steric and Electronic Effects on γ-Selectivity. J. Org. Chem. 2011, 76, 1398–1408. [Google Scholar] [CrossRef]
- Guduguntla, S.; Hornillos, V.; Tessier, R.; Fananas-Mastraland, M.; Feringa, B.L. Chiral Diarylmethanes via Copper-Catalyzed Asymmetric Allylic Arylation with Organolithium Compounds. Org. Lett. 2016, 18, 252–255. [Google Scholar] [CrossRef]
- Glorius, F.; Altenhoff, G.; Goddard, R.; Lehmann, C. Oxazolines as chiral building blocks for imidazolium salts and N-heterocyclic carbene ligands. Chem. Commun. 2002, 22, 2704–2705. [Google Scholar] [CrossRef]
- Würtz, S.; Lohre, C.; Frohlich, R.; Bergander, K.; Glorius, F. IBiox[(-)-menthyl]: A sterically demanding chiral NHC ligand. J. Am. Chem. Soc. 2009, 131, 8344–8345. [Google Scholar] [CrossRef] [PubMed]
- Bexrud, J.; Lautens, M. A Rhodium IBiox[(−)-menthyl] Complex as a Highly Selective Catalyst for the Asymmetric Hydroarylation of Azabicyles: An Alternative Route to Epibatidine. Org. Lett. 2010, 12, 3160–3163. [Google Scholar] [CrossRef] [PubMed]
- Melot, R.; Zuccarello, M.; Cavalli, D.; Niggli, N.; Devereux, M.; Bürgi, T.; Baudoin, O. Palladium(0)-Catalyzed Enantioselective Intramolecular Arylation of Enantiotopic Secondary C-H Bonds. Angew. Chem. Int. Ed. 2021, 60, 7245–7250. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Ishida, N.; Ashida, S.; Murakami, M. Synthesis of Chiral N-Heterocyclic Carbene Ligands with Rigid Backbones and Application to the Palladium-Catalyzed Enantioselective Intramolecular α-Arylation of Amides. Org. Lett. 2011, 13, 1666–1669. [Google Scholar] [CrossRef]

| Metal | NHC | Reactions | References |
|---|---|---|---|
| Cu | 2,7 6 8 9 18 19 | Conjugate addition Three-component coupling Hydrosylilation Protoboration Boration of α, β-unsaturated esters Allylic alkylation | [19,37] [35] [37,38] [41] [44] [55] |
| Ni | 3 9 9 10 10 10 10 11 11 13 | Three-component coupling Arylation of secondary alcohols C-N cross-coupling C-H alkylation of polyfluoroarenes Reductive coupling of alkynes and imines Arylboration of ketones Arylboration and alkenylboration of aldehydes Reductive coupling of alcohols and alkynes C-H cyclization of pyridines, pyridones and pyrimidines C-H functionalization of indoles and pyrroles | [29] [48] [49] [45] [46] [47] [50] [42] [43,44] [40] |
| Pd | 4,5,20 4,6 13 | Intramolecular α-arylation of amides C(sp3)-H/C(Ar) coupling Suzuki-Miyaura cross-coupling | [30,31,55] [32,33,34] [51] |
| Rh | 15–18 | Conjugate addition | [52] |
| Ru | 2 14, 15, 17 | Hydrogenation Hydrosilylation of ketones | [19,20,21,22,23,24,25,26] [53] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costabile, C.; Pragliola, S.; Grisi, F. C2-Symmetric N-Heterocyclic Carbenes in Asymmetric Transition-Metal Catalysis. Symmetry 2022, 14, 1615. https://doi.org/10.3390/sym14081615
Costabile C, Pragliola S, Grisi F. C2-Symmetric N-Heterocyclic Carbenes in Asymmetric Transition-Metal Catalysis. Symmetry. 2022; 14(8):1615. https://doi.org/10.3390/sym14081615
Chicago/Turabian StyleCostabile, Chiara, Stefania Pragliola, and Fabia Grisi. 2022. "C2-Symmetric N-Heterocyclic Carbenes in Asymmetric Transition-Metal Catalysis" Symmetry 14, no. 8: 1615. https://doi.org/10.3390/sym14081615
APA StyleCostabile, C., Pragliola, S., & Grisi, F. (2022). C2-Symmetric N-Heterocyclic Carbenes in Asymmetric Transition-Metal Catalysis. Symmetry, 14(8), 1615. https://doi.org/10.3390/sym14081615







