Chemical Models for Understanding the Emergence of Homo-Chirality of Phospholipids for Origin of Life Studies
Abstract
1. Introduction
1.1. Geochemical Scenarios
1.2. Homochirality Emergence and Origin of Life Studies
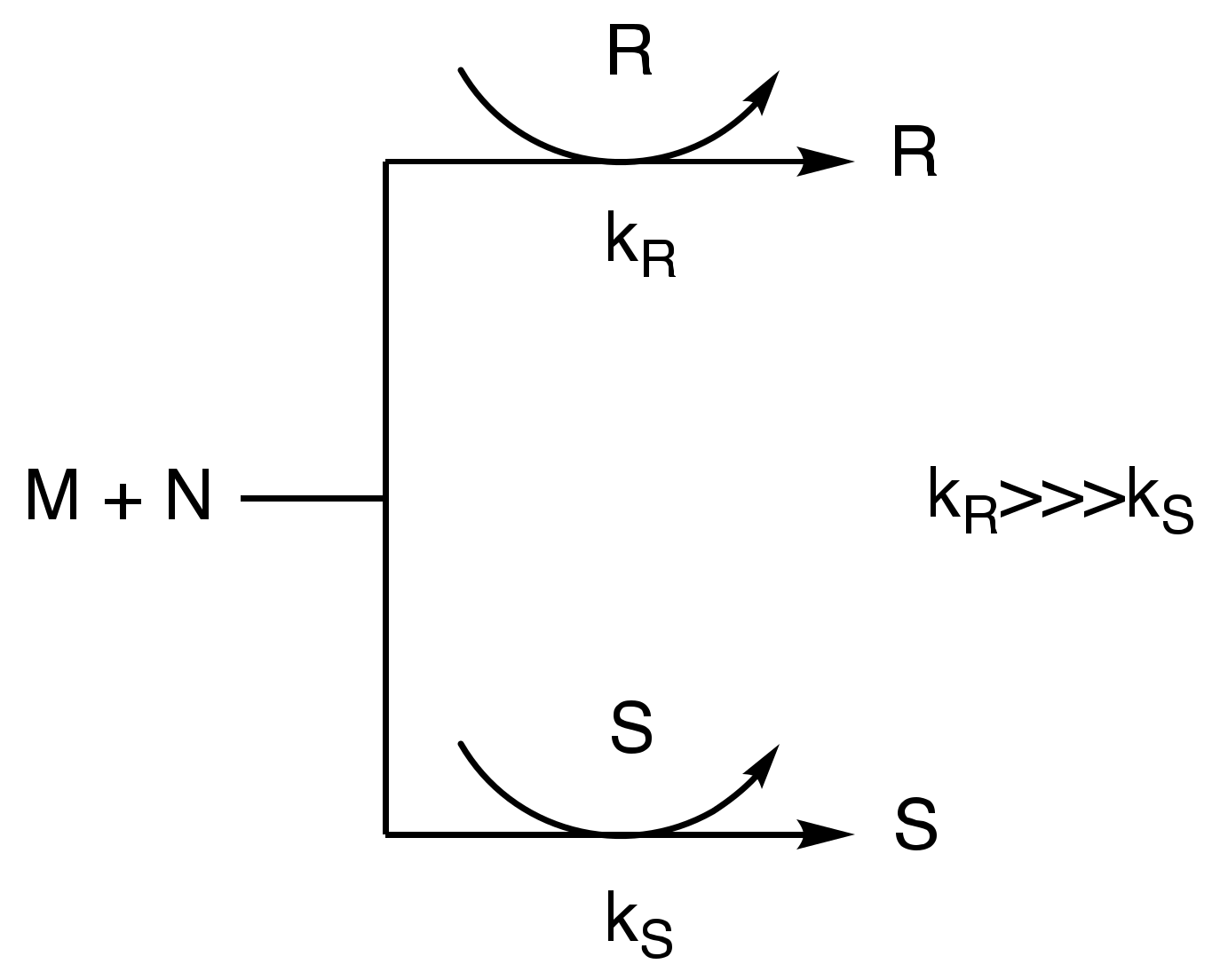
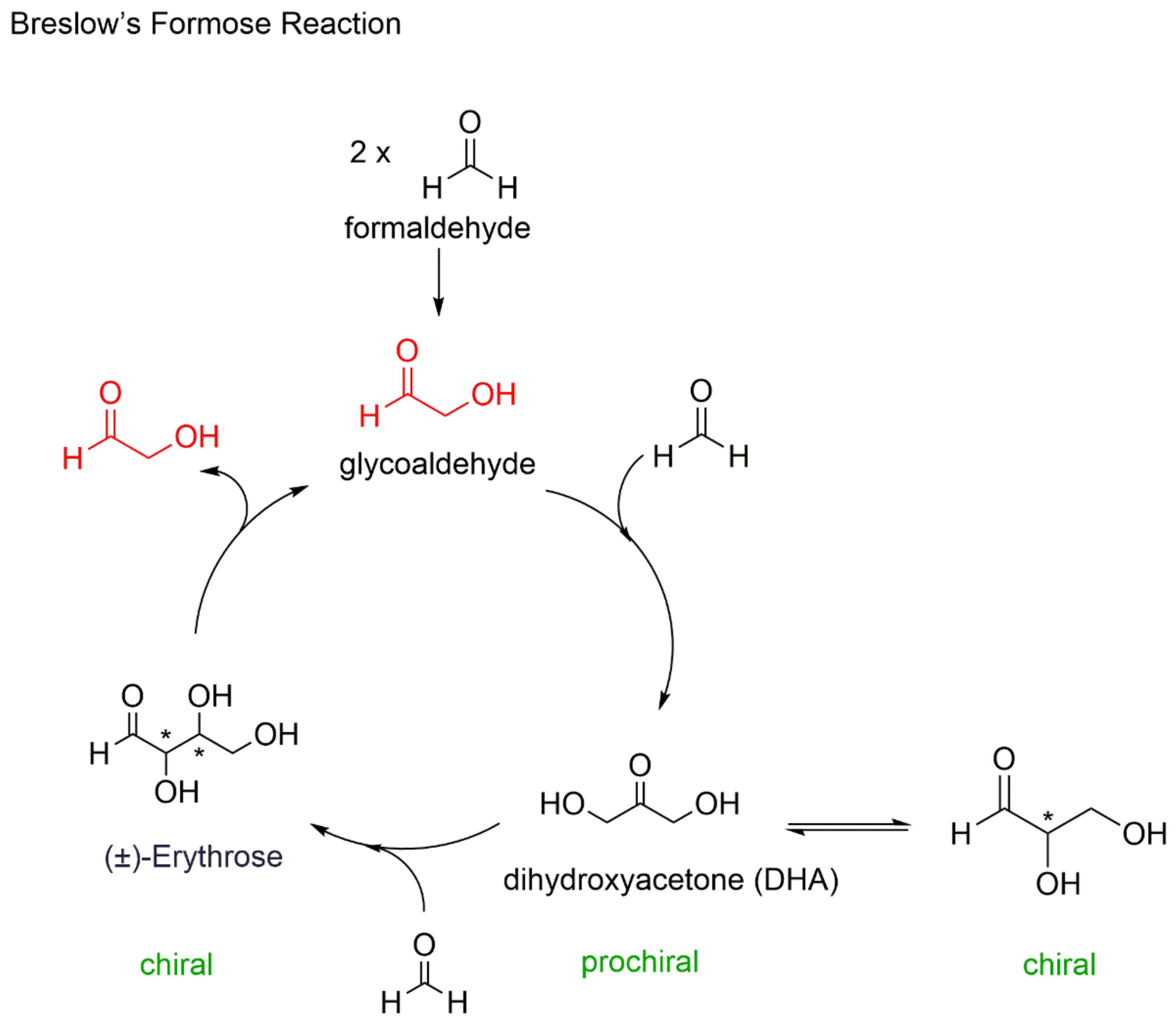
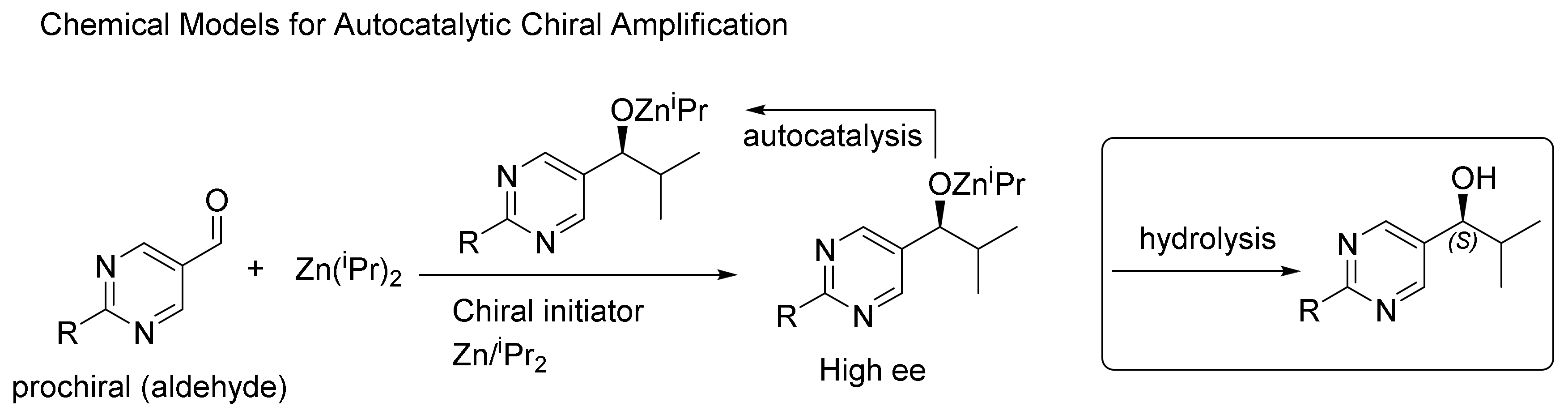
2. Autocatalysis and Symmetry Breaking
3. Crystallization Led to Homochiral Symmetry Breaking
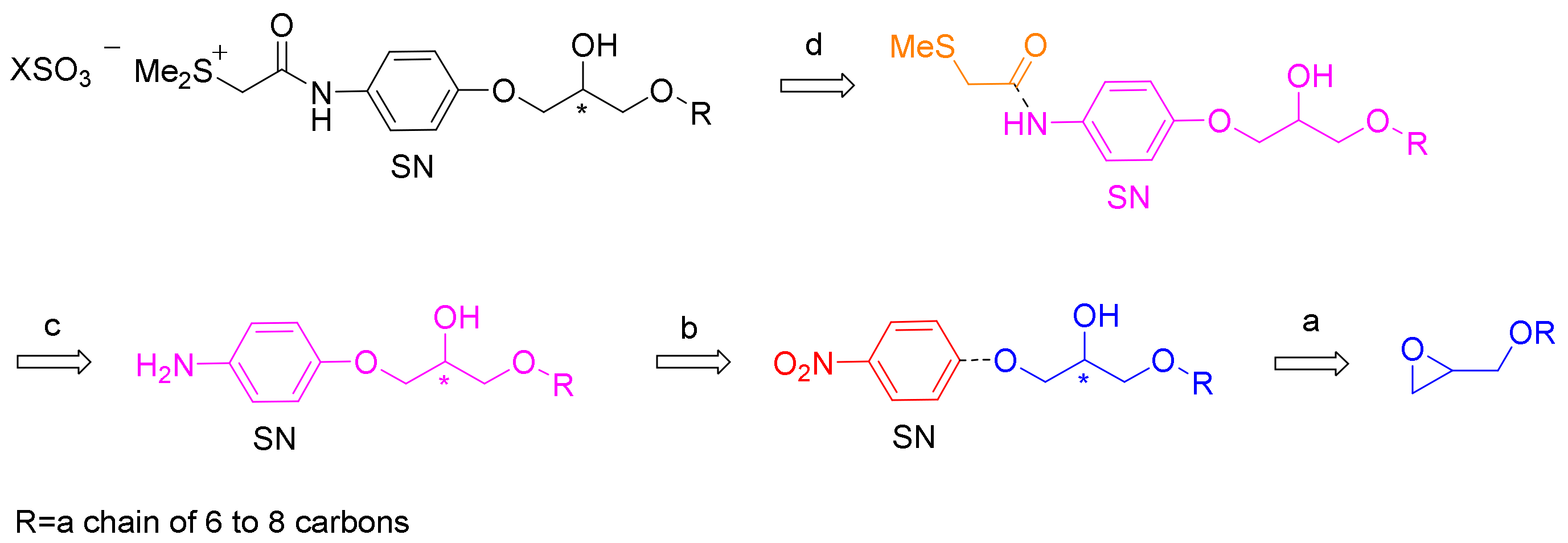
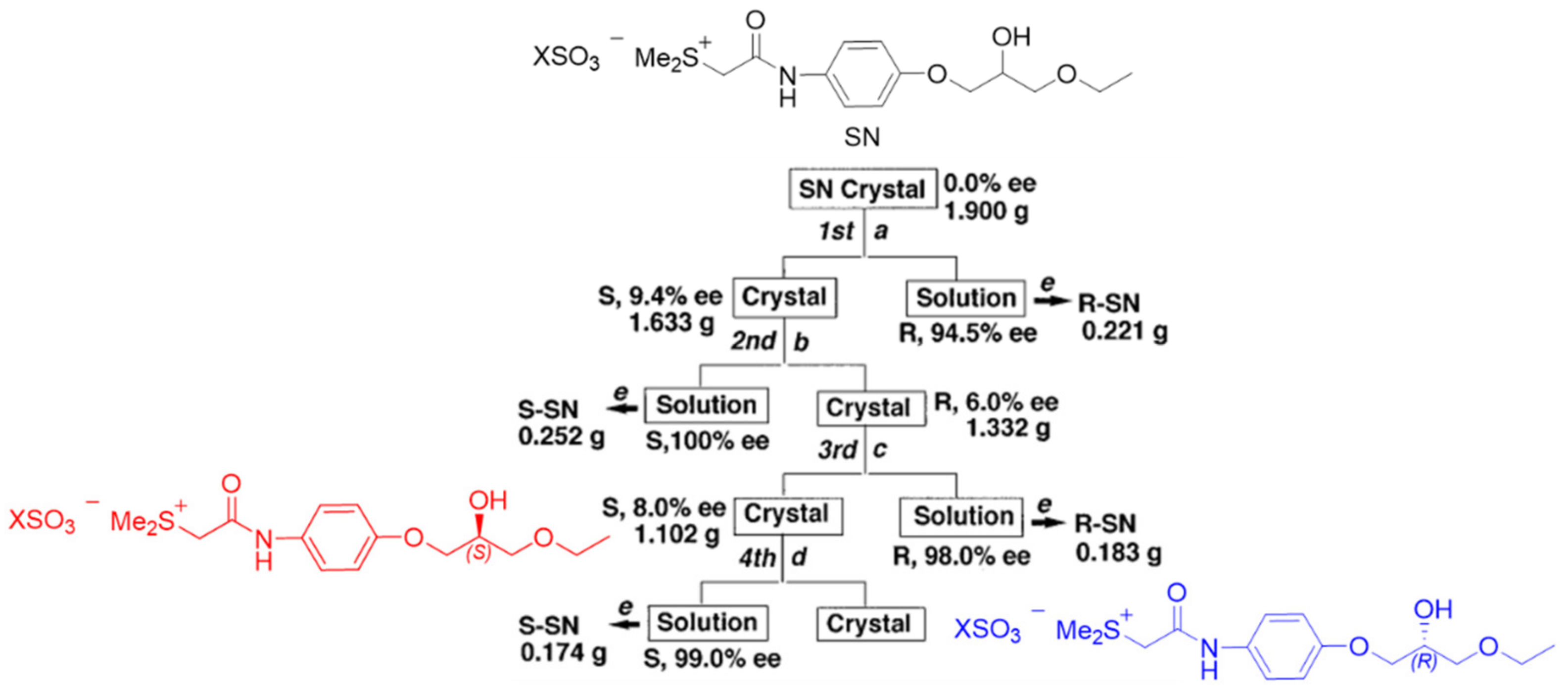
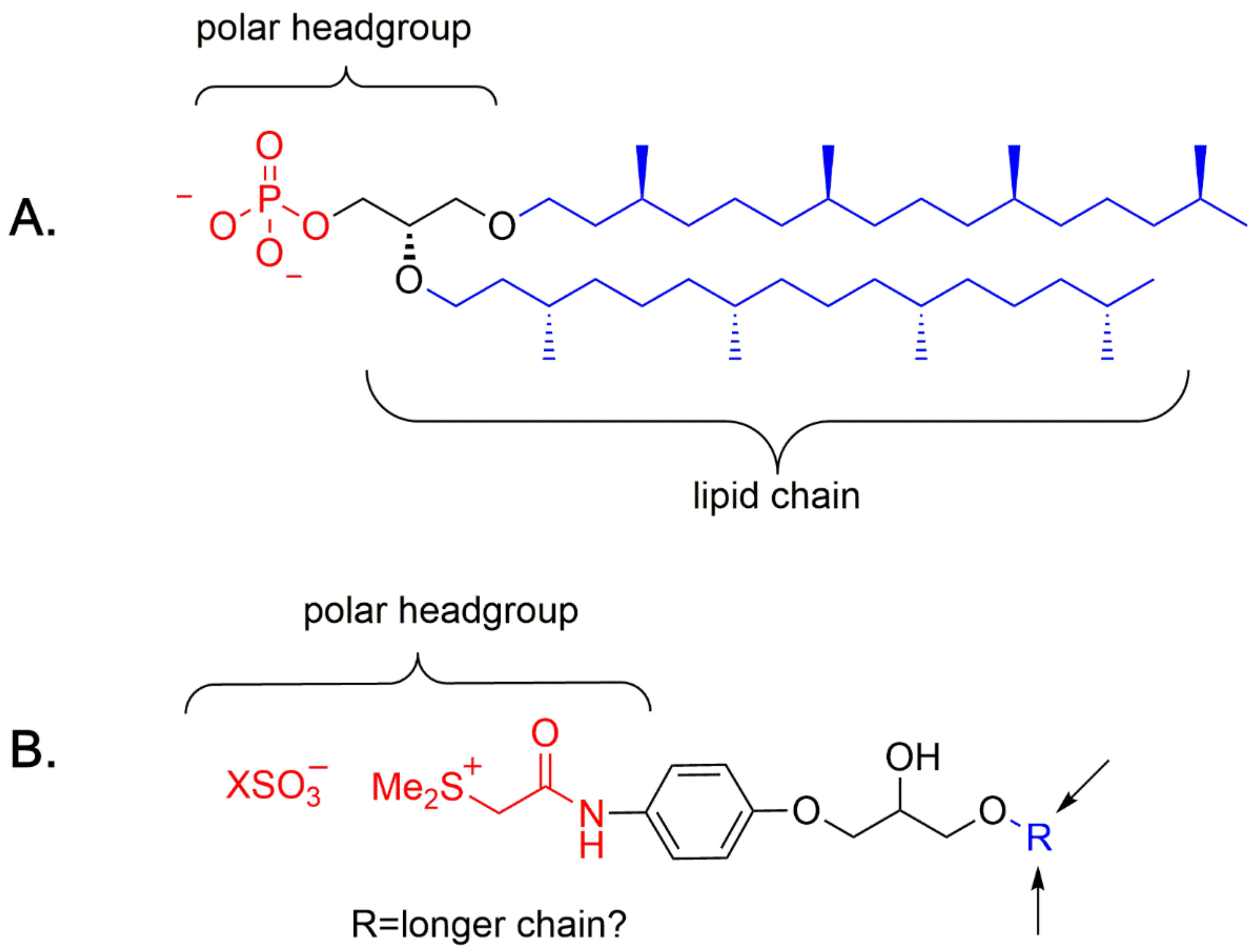
4. A Chemical Model for the Study of Symmetry Breaking of Phospholipids
Untangle a Racemic Conglomerate of Phospholipids
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Suh, I.-H.; Park, K.H.; Jensen, W.P.; Lewis, D.E. Molecules, Crystals, and Chirality. J. Chem. Educ. 1997, 74, 800. [Google Scholar] [CrossRef]
- Müller, F.; Escobar, L.; Xu, F.; Węgrzyn, E.; Nainytė, M.; Amatov, T.; Chan, C.; Pichler, A.; Carell, T. A prebiotically plausible scenario of an RNA–peptide world. Nature 2022, 605, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Airapetian, V.S.; Glocer, A.; Gronoff, G.; Hébrard, E.; Danchi, W. Prebiotic chemistry and atmospheric warming of early Earth by an active young Sun. Nat. Geosci. 2016, 9, 452–455. [Google Scholar] [CrossRef]
- Schmitt-Kopplin, P.; Gabelica, Z.; Gougeon, R.D.; Fekete, A.; Kanawati, B.; Harir, M.; Gebefuegi, I.; Eckel, G.; Hertkorn, N. High molecular diversity of extraterrestrial organic matter in Murchison meteorite revealed 40 years after its fall. Proc. Natl. Acad. Sci. USA 2010, 107, 2763–2768. [Google Scholar] [CrossRef] [PubMed]
- Kitadai, N.; Maruyama, S. Origins of building blocks of life: A review. Geosci. Front. 2018, 9, 1117–1153. [Google Scholar] [CrossRef]
- Ross, D.S.; Deamer, D. Dry/wet cycling and the thermodynamics and kinetics of prebiotic polymer synthesis. Life 2016, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Damer, B.; Deamer, D. The hot spring hypothesis for an origin of life. Astrobiology 2020, 20, 429–452. [Google Scholar] [CrossRef] [PubMed]
- Reeves, E.P.; Seewald, J.S.; Saccocia, P.; Bach, W.; Craddock, P.R.; Shanks, W.C.; Sylva, S.P.; Walsh, E.; Pichler, T.; Rosner, M. Geochemistry of hydrothermal fluids from the PACMANUS, Northeast Pual and Vienna Woods hydrothermal fields, Manus Basin, Papua New Guinea. Geochim. Cosmochim. Acta 2011, 75, 1088–1123. [Google Scholar] [CrossRef]
- Miller, S.L.; Bada, J.L. Submarine hot springs and the origin of life. Nature 1988, 334, 609–611. [Google Scholar] [CrossRef] [PubMed]
- Damer, B.; Deamer, D. Coupled Phases and Combinatorial Selection in Fluctuating Hydrothermal Pools: A Scenario to Guide Experimental Approaches to the Origin of Cellular Life. Life 2015, 5, 872–887. [Google Scholar] [CrossRef]
- Deamer, D. The Role of Lipid Membranes in Life’s Origin. Life 2017, 7, 5. [Google Scholar] [CrossRef] [PubMed]
- Deamer, D. Origins of Life Research: The Conundrum between Laboratory and Field Simulations of Messy Environments. Life 2022, 12, 1429. [Google Scholar] [CrossRef] [PubMed]
- Schwarzenbach, E.M.; Früh-Green, G.L.; Bernasconi, S.M.; Alt, J.C.; Shanks, W.C.; Gaggero, L.; Crispini, L. Sulfur geochemistry of peridotite-hosted hydrothermal systems: Comparing the Ligurian ophiolites with oceanic serpentinites. Geochim. Cosmochim. Acta 2012, 91, 283–305. [Google Scholar] [CrossRef]
- Rickard, D. Pyrite and the Origins of Life. In Pyrite; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Avalos, M.; Babiano, R.; Cintas, P.; Jimenez, J.L.; Palacios, J.C. Symmetry Breaking by Spontaneous Crystallization—Is it the Most Plausible Source of Terrestrial Handedness we have Long Been Looking for?—A Reappraisal. Orig. Life Evol. Biosph. 2004, 34, 391–405. [Google Scholar] [CrossRef]
- Chen, Y.; Ma, W. The origin of biological homochirality along with the origin of life. PLoS Comput. Biol. 2020, 16, e1007592. [Google Scholar] [CrossRef] [PubMed]
- Gal, J. When did Louis Pasteur present his memoir on the discovery of molecular chirality to the Académie des sciences? Analysis of a discrepancy. Chirality 2008, 20, 1072–1084. [Google Scholar] [CrossRef]
- Blackmond, D.G. The origin of biological homochirality. Cold Spring Harb. Perspect. Biol. 2019, 2, a002147. [Google Scholar] [CrossRef]
- Cronin, J.R.; Gandy, W.E.; Pizzarello, S. Amino acids of the murchison meteorite: I. Six Carbon Acyclic Primary α-Amino Alkanoic Acids. J. Mol. Evol. 1981, 17, 265–272. [Google Scholar] [CrossRef]
- Engel, M.H.; Nagy, B.; Bada, J.L.; Cronin, J.R.; Ho, M.-S.S.; Kvenvolden, K.A.; Lawless, J.G.; Miller, S.L.; Oro, J.; Steinberg, S. On the reported optical activity of amino acids in the Murchison meteorite (reply). Nature 1983, 301, 496–497. [Google Scholar] [CrossRef]
- Cronin, J.R.; Pizzarello, S. Enantiomeric excesses in meteoritic amino acids. Science 1997, 275, 951–955. [Google Scholar] [CrossRef]
- Cronin, J.R.; Pizzarello, S. Amino acids of the Murchison meteorite. III. Seven carbon acyclic primary α-amino alkanoic acids1. Geochim. Cosmochim. Acta 1986, 50, 2419–2427. [Google Scholar] [CrossRef]
- Garcia, A.; Meinert, C.; Sugahara, H.; Jones, N.; Hoffmann, S.; Meierhenrich, U. The Astrophysical Formation of Asymmetric Molecules and the Emergence of a Chiral Bias. Life 2019, 9, 29. [Google Scholar] [CrossRef]
- Burton, A.; Berger, E. Insights into Abiotically-Generated Amino Acid Enantiomeric Excesses Found in Meteorites. Life 2018, 8, 14. [Google Scholar] [CrossRef] [PubMed]
- Parker, E.T.; Cleaves, H.J.; Dworkin, J.P.; Glavin, D.P.; Callahan, M.; Aubrey, A.; Lazcano, A.; Bada, J.L. Primordial synthesis of amines and amino acids in a 1958 Miller H 2S-rich spark discharge experiment. Proc. Natl. Acad. Sci. USA 2011, 108, 5526–5531. [Google Scholar] [CrossRef]
- Engel, M.H.; Macko, S.A.; Silfer, J.A. Carbon isotope composition of individual amino acids in the Murchison meteorite. Nature 1990, 348, 47–49. [Google Scholar] [CrossRef] [PubMed]
- Engel, M.H.; Nagy, B. Distribution and enantiomeric composition of amino acids in the Murchison meteorite. Nature 1982, 296, 837–840. [Google Scholar] [CrossRef]
- Cronin, J.R.; Pizzarello, S.; Yuen, G.U. Amino acids of the Murchison meteorite: II. Five carbon acyclic primary β-, γ-, and δ-amino alkanoic acids. Geochim. Cosmochim. Acta 1985, 49, 2259–2265. [Google Scholar] [CrossRef]
- Kvenvolden, K.A.; Lawless, J.G.; Ponnamperuma, C. Nonprotein Amino Acids in the Murchison Meteorite. Proc. Natl. Acad. Sci. USA 1971, 68, 486–490. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.L. A Production of Amino Acids Under Possible Primitive Earth Conditions. Science 1953, 117, 528–529. [Google Scholar] [CrossRef]
- Skolnick, J.; Zhou, H.; Gao, M. On the possible origin of protein homochirality, structure, and biochemical function. Proc. Natl. Acad. Sci. USA 2019, 116, 26571–26579. [Google Scholar] [CrossRef] [PubMed]
- Grishin, D.V.; Zhdanov, D.D.; Pokrovskaya, M.V.; Sokolov, N.N. D-amino acids in nature, agriculture and biomedicine. All Life 2020, 13, 11–22. [Google Scholar] [CrossRef]
- Lopez, A.; Fiore, M. Investigating Prebiotic Protocells for a Comprehensive Understanding of the Origins of Life: A Prebiotic Systems Chemistry Perspective. Life 2019, 9, 49. [Google Scholar] [CrossRef]
- Fiore, M.; Buchet, R. Symmetry breaking of phospholipids. Symmetry 2020, 12, 1488. [Google Scholar] [CrossRef]
- Frank, F.C. On spontaneous asymmetric synthesis. Biochim. Biophys. Acta 1953, 11, 459–463. [Google Scholar] [CrossRef]
- Micheau, J.-C.; Coudret, C.; Cruz, J.-M.; Buhse, T. Amplification of enantiomeric excess, mirror-image symmetry breaking and kinetic proofreading in Soai reaction models with different oligomeric orders. Phys. Chem. Chem. Phys. 2012, 14, 13239. [Google Scholar] [CrossRef]
- Breslow, R. On the mechanisms of formose reaction. Tetrahedron Lett. 1959, 1, 22–26. [Google Scholar] [CrossRef]
- Breslow, R.; Cheng, Z.L. L-amino acids catalyze the formation of an excess of D-glyceraldehyde, and thus of other D sugars, under credible prebiotic conditions. Proc. Natl. Acad. Sci. USA 2010, 107, 5723–5725. [Google Scholar] [CrossRef]
- Soai, K.; Kawasaki, T.; Matsumoto, A. Role of Asymmetric Autocatalysis in the Elucidation of Origins of Homochirality of Organic Compounds. Symmetry 2019, 11, 694. [Google Scholar] [CrossRef]
- Hawbaker, N.A.; Blackmond, D.G. Energy threshold for chiral symmetry breaking in molecular self-replication. Nat. Chem. 2019, 11, 957–962. [Google Scholar] [CrossRef]
- Geiger, Y. One Soai reaction, two mechanisms? Chem. Soc. Rev. 2022, 51, 1206–1211. [Google Scholar] [CrossRef]
- Athavale, S.V.; Simon, A.; Houk, K.N.; Denmark, S.E. Demystifying the asymmetry-amplifying, autocatalytic behaviour of the Soai reaction through structural, mechanistic and computational studies. Nat. Chem. 2020, 12, 412–423. [Google Scholar] [CrossRef]
- Viedma, C. Experimental evidence of chiral symmetry breaking in crystallization from primary nucleation. J. Cryst. Growth 2004, 261, 118–121. [Google Scholar] [CrossRef]
- Kondepudi, D.K.; Kaufman, R.J.; Singh, N. Chiral Symmetry Breaking in Sodium Chlorate Crystallization. Science 1990, 250, 975–976. [Google Scholar] [CrossRef]
- Tamura, R.; Iwama, S.; Takahashi, H. Chiral Symmetry Breaking Phenomenon Caused by a Phase Transition. Symmetry 2010, 2, 112–135. [Google Scholar] [CrossRef]
- Tamura, R.; Takahashi, H.; Hirotsu, K.; Nakajima, Y.; Ushio, T.; Toda, F. Unusual disordered crystal structure of a racemate exhibiting a novel enantiomeric resolution: Preferential enrichment. Angew. Chemie-Int. Ed. 1998, 37, 2876–2878. [Google Scholar] [CrossRef]
- Ruiz-Mirazo, K.; Briones, C.; De La Escosura, A. Prebiotic systems chemistry: New perspectives for the origins of life. Chem. Rev. 2014, 114, 285–366. [Google Scholar] [CrossRef]
- Fiore, M.; Chieffo, C.; Lopez, A.; Fayolle, D.; Ruiz, J.; Soulère, L.; Oger, P.; Altamura, E.; Popowycz, F.; Buchet, R. Synthesis of Phospholipids Under Plausible Prebiotic Conditions and Analogies with Phospholipid Biochemistry for Origin of Life Studies. Astrobiology 2022, 22, 598–627. [Google Scholar] [CrossRef]
- Patel, B.H.; Percivalle, C.; Ritson, D.J.; Duffy, C.D.; Sutherland, J.D. Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism. Nat. Chem. 2015, 7, 301–307. [Google Scholar] [CrossRef]
- Bonfio, C.; Valer, L.; Scintilla, S.; Shah, S.; Evans, D.J.; Jin, L.; Szostak, J.W.; Sasselov, D.D.; Sutherland, J.D.; Mansy, S.S. UV-light-driven prebiotic synthesis of iron-sulfur clusters. Nat. Chem. 2017, 9, 1229–1234. [Google Scholar] [CrossRef]
- Bonfio, C.; Caumes, C.; Duffy, C.D.; Patel, B.H.; Percivalle, C.; Tsanakopoulou, M.; Sutherland, J.D. Length-Selective Synthesis of Acylglycerol-Phosphates through Energy-Dissipative Cycling. J. Am. Chem. Soc. 2019, 141, 3934–3939. [Google Scholar] [CrossRef]
- Liu, Z.; Wu, L.; Xu, J.; Bonfio, C.; Russell, D.A.; Pascal, R.; Sutherland, J.D. Harnessing Chemical Energy for the Activation and Joining of Prebiotic Building Blocks. Nat. Chem. 2019, 12, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Lopez, A.; Chieffo, C.; Fiore, M. Abiotic Synthesis and Role of Amphiphiles in the Encapsulation Process in Life’s Origin. In Prebiotic Chemistry and The Origin of Life; Neubeck Anna, M.S., Ed.; Springer: Berlin/Heidelberg, Germany, 2021; pp. 139–164. ISBN 978-3-030-81038-2. [Google Scholar]
- Altamura, E.; Comte, A.; D’Onofrio, A.; Roussillon, C.; Fayolle, D.; Buchet, R.; Mavelli, F.; Stano, P.; Fiore, M.; Strazewski, P. Racemic Phospholipids for Origin of Life Studies. Symmetry 2020, 12, 1108. [Google Scholar] [CrossRef]
- Fayolle, D.; Altamura, E.; D’Onofrio, A.; Madanamothoo, W.; Fenet, B.; Mavelli, F.; Buchet, R.; Stano, P.; Fiore, M.; Strazewski, P. Crude phosphorylation mixtures containing racemic lipid amphiphiles self-assemble to give stable primitive compartments. Sci. Rep. 2017, 7, 18106. [Google Scholar] [CrossRef]
- Wick, R.; Walde, P.; Luisi, P.L. Giant Vesicles. In Self-Production of Supramolecular Structures; Springer: Dordrecht, The Netherlands, 1994; pp. 295–299. [Google Scholar]
- Jordan, S.F.; Rammu, H.; Zheludev, I.N.; Hartley, A.M.; Maréchal, A.; Lane, N. Promotion of protocell self-assembly from mixed amphiphiles at the origin of life. Nat. Ecol. Evol. 2019, 3, 1705–1714. [Google Scholar] [CrossRef]
| Year | Amino Acids | Comments/References |
|---|---|---|
| 2011 a | Gly, Ala, Ser, Thr, Asp, Glu, Met, Val, Leu, Phe, β-Ala, Iso-serine, Homocysteic acid, α-ABA β-ABA, α-AIB, β-AIB, γ-ABI, S-Methyl Cysteine, Iso-valine, Methionine sulfoxide, Methionine sulfone, Iso-leucine, Ethionine, MA, EA, Cysteamine, Ethanolamine | Analysis of the original samples from Miller—Bada experiments. Reactions were carried out in the presence of H2S in addition to CH4, NH3 and H2O [25] |
| From 1969 | Glu (0.322); Asp (0.202); Pro (0.342); Leu (0.166); Ala (0.682) | Murchinson meteorite extracts [26,27]: predominancy of l-configuration; more than 20 amino acids were found in the Murchinson meteorite [19,20,22,23,28,29] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skorda, F.; Chieffo, C.; Fiore, M. Chemical Models for Understanding the Emergence of Homo-Chirality of Phospholipids for Origin of Life Studies. Symmetry 2022, 14, 2109. https://doi.org/10.3390/sym14102109
Skorda F, Chieffo C, Fiore M. Chemical Models for Understanding the Emergence of Homo-Chirality of Phospholipids for Origin of Life Studies. Symmetry. 2022; 14(10):2109. https://doi.org/10.3390/sym14102109
Chicago/Turabian StyleSkorda, Fryni, Carolina Chieffo, and Michele Fiore. 2022. "Chemical Models for Understanding the Emergence of Homo-Chirality of Phospholipids for Origin of Life Studies" Symmetry 14, no. 10: 2109. https://doi.org/10.3390/sym14102109
APA StyleSkorda, F., Chieffo, C., & Fiore, M. (2022). Chemical Models for Understanding the Emergence of Homo-Chirality of Phospholipids for Origin of Life Studies. Symmetry, 14(10), 2109. https://doi.org/10.3390/sym14102109







