Brain Symmetry Analysis during the Use of a BCI Based on Motor Imagery for the Control of a Lower-Limb Exoskeleton
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Equipment
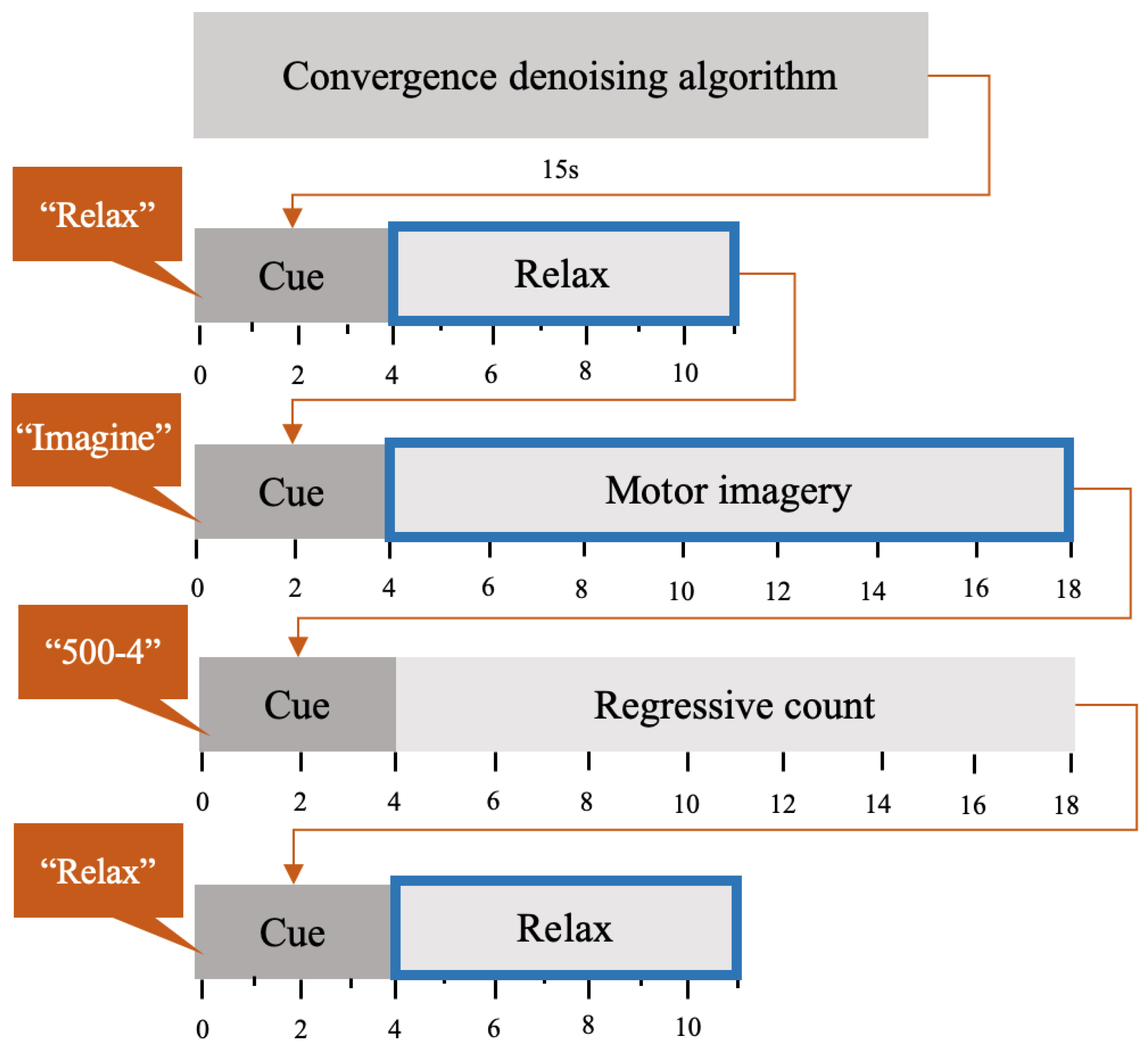
2.3. Experimental Design
2.4. BCI
2.4.1. Acquisition
2.4.2. Pre-Processing
2.4.3. Processing
FBCSP
ST and Welch’s PSD
2.4.4. Classification
2.4.5. Evaluation
3. Results
3.1. Methodology Influence
- Full-static: The accuracies of two trials were detected as outliers (one for Welch’s PSD and one for FBCSP). They were removed as well as the same value for all methodologies to be compared. Afterwards, Shapiro–Wilk test was employed to verify if the data from each group followed a normal distribution and check if the normality null hypothesis could not be rejected (p-value > 0.01). In addition, the sphericity assumption was assessed with Mauchly’s test. Results from the ANOVA test showed that the performance of FBCSP, ST and Welch’s method were statistically different (p-value < 0.01). Afterwards, pair-wise t-test comparisons were conducted, detecting that all methodologies differed significantly from each other (p-value < 0.01).
- Full-motion: The accuracies of 10 trials were identified as outliers and removed (six for ST and four for FBCSP). Data from each methodology followed a normal distribution (p-value > 0.01), and the sphericity assumption was not violated. Results from ANOVA showed significant differences among methodologies (p-value < 0.01) showing the pair-wise t-test comparisons differences between CSP and ST and between ST and Welch PSD (p-value < 0.01).
3.2. Subject Influence
- Full-static: The accuracies of 10 trials were identified as outliers (four for S1, three for S2 and three for S3) and removed. Afterwards, Shapiro–Wilk test was applied, and results showed that the data from each subject followed a normal distribution (p-value > 0.01). Finally, the last assumption to check in order to apply the one-way ANOVA was the data homoscedasticity. This assumption was fulfilled, as assessed by Bartlett test (p-value > 0.01). The ANOVA test showed significant differences among subjects (p-value < 0.01), and the pair-wise t-test indicated that S1 had a different behavior than the rest (p-value < 0.01).
- Full-motion: The accuracies of eight trials were detected as outliers (one for S2, one for S3, one for S4 and five for S5) and removed from the analysis. Data from each subject were normally distributed as assessed by Shapiro–Wilk test (p-value > 0.01). However, the homoscedasticity assumption was violated, p-value < 0.01 from the Barlett test. Therefore, a non-parametric Kruskal–Wallis test was employed. Results from this test showed no significant differences among subjects (p-value > 0.01).
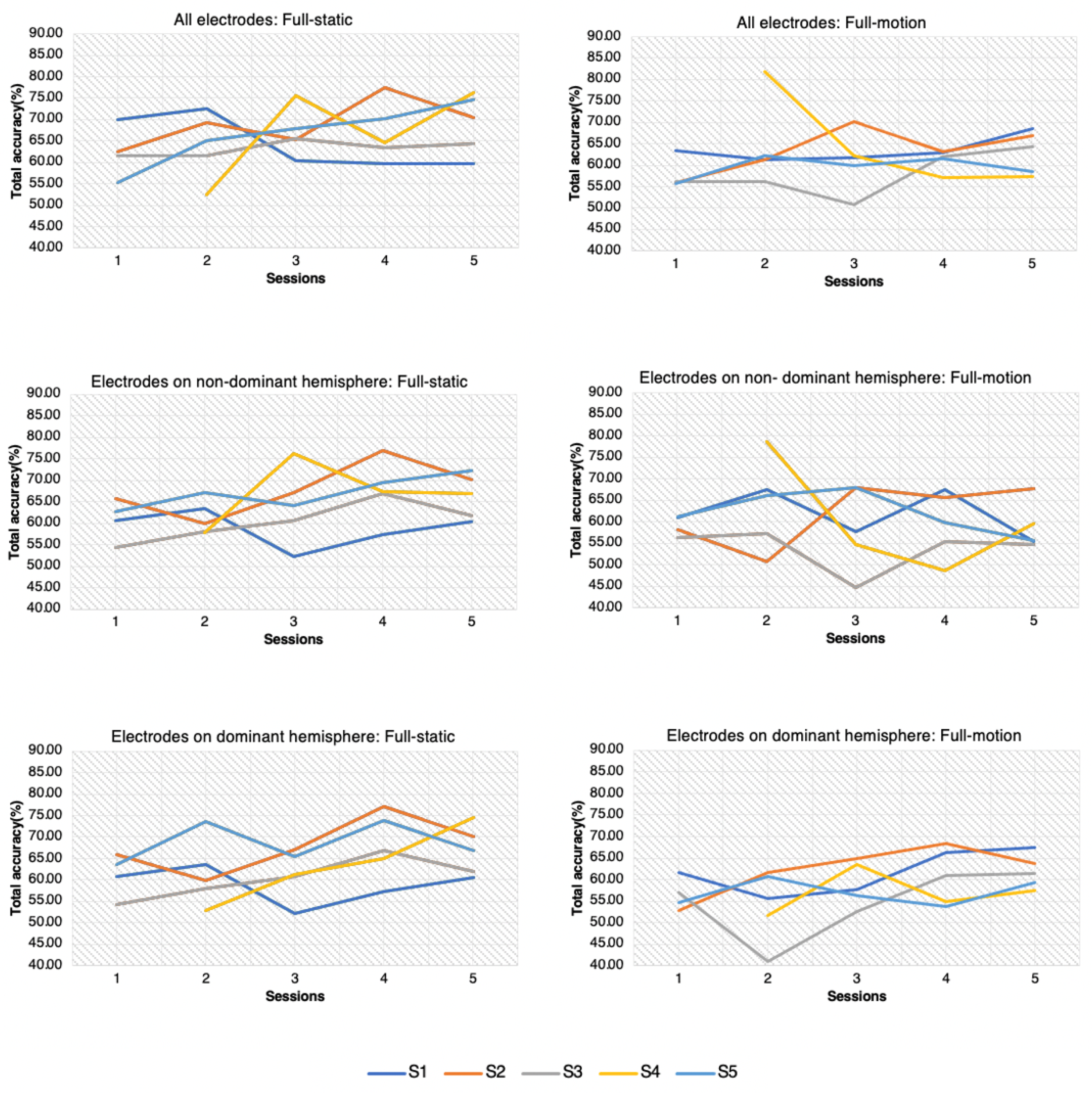
3.3. Training Evolution and Differences between Hemispheres
3.3.1. S1
- Full-static: Data of all sessions and electrode configurations did not follow a normal distribution. Therefore, instead of performing a two-way ANOVA test, three independent Kruskal–Wallis tests were employed: one assessing differences among sessions, another one with electrode configurations and the third one combining both variables as a new grouping variable. No differences were found for any group (p-value > 0.01).
- Full-motion: Initially, the ANOVA assumption for a normal distribution of the data was not fulfilled. The accuracies of 4 trials were detected as outliers and removed (one for EEG configuration with all electrodes, one for electrodes on the non-dominant hemisphere and two for the electrodes on the dominant hemisphere). With the remaining data, all the ANOVA test assumptions were matched, and results did not show statistically relevant differences (p-value > 0.01).
3.3.2. S2
- Full-static: The ANOVA analysis of the results verified that there were not differences among the sessions and the electrode configurations. In addition, the interaction between the sessions and the configurations was not significant.
- Full-motion: ANOVA test revealed significant differences among sessions (p-value < 0.01). Sessions 1 and 2 showed worse accuracy than 3, 4, and 5, as was assessed with a pair-wise t-test.
3.3.3. S3
- Full-static: ANOVA test did not reveal statistically significant differences among sessions and electrode configurations. Furthermore, the interaction betweeen sessions and electrode configurations was not significant (p-value > 0.01).
- Full-motion: ANOVA test only indicated differences among sessions, and pair-wise t-test indicated that the behavior of session 3 was significantly worse in comparison with 1, 4 and 5.
3.3.4. S4
- Full-static: Data of all sessions and electrode configurations did not follow a normal distribution. Four outliers were detected and discarded (three for the EEG configuration with all electrodes and one for the configuration of electrodes on dominant hemisphere). However, data of all groups still did not follow a normal distribution. Three independent Kruskal–Wallis tests were performed as in S1. Results showed that there were statistically significant differences among sessions (p-value < 0.01). Performance of session 2 differed from the one of sessions 3 and 5, as verified with Dunn’s Kruskal–Wallis Multiple Comparisons. In addition, the interaction between electrode configurations and sessions was significant (p-value < 0.01). This means that the performance of the BCI with each electrode configuration varied from session to session. The performance of session 2 was better than sessions 3 and 5 with two electrode configurations, all electrodes and electrodes on the non-dominant hemisphere. In the case of the setup on the dominant-hemisphere, the performance of session 2 was worse.
- Full-motion: The performance was statistically different among different sessions, as assessed by ANOVA test. Furthermore, the interaction between session and configuration was statistically significant (p-value < 0.01).
3.3.5. S5
- Full-static: ANOVA test revealed significant differences among sessions (p-value < 0.01), and session 1 was significantly worse than session 5, as assessed with pair-wise t-test.
- Full-motion: Results from ANOVA verified that there were no differences among sessions (p-value > 0.01).
4. Discussion
4.1. Study Limitations
4.2. Algorithm Influence
4.3. BCI Performance Comparison
4.4. Subject Influence
4.5. Hemisphere Dominance
4.6. Evolution of Performance with Practice
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mulder, T. Motor imagery and action observation: Cognitive tools for rehabilitation. J. Neural Transm. (Vienna Austria 1996) 2007, 114, 1265–1278. [Google Scholar] [CrossRef]
- Lee, K.; Liu, D.; Perroud, L.; Chavarriaga, R.; Millán, J.d.R. A brain-controlled exoskeleton with cascaded event-related desynchronization classifiers. Robot. Auton. Syst. 2017, 90, 15–23. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; Neuper, C.; Andrew, C.; Edlinger, G. Foot and hand area mu rhythms. Int. J. Psychophysiol. 1997, 26, 121–135. [Google Scholar] [CrossRef]
- Donati, A.R.; Shokur, S.; Morya, E.; Campos, D.S.; Moioli, R.C.; Gitti, C.M.; Augusto, P.B.; Tripodi, S.; Pires, C.G.; Pereira, G.A.; et al. Long-Term Training with a Brain-Machine Interface-Based Gait Protocol Induces Partial Neurological Recovery in Paraplegic Patients. Sci. Rep. 2016, 6, 30383. [Google Scholar] [CrossRef]
- He, Y.; Eguren, D.; Azorín, J.M.; Grossman, R.G.; Luu, T.P.; Contreras-Vidal, J.L. Brain-machine interfaces for controlling lower-limb powered robotic systems. J. Neural Eng. 2018, 15. [Google Scholar] [CrossRef]
- Soleimani Amiri, M.; Ramli, R.; Ibrahim, M.F.; Abd Wahab, D.; Aliman, N. Adaptive Particle Swarm Optimization of PID Gain Tuning for Lower-Limb Human Exoskeleton in Virtual Environment. Mathematics 2020, 8, 2040. [Google Scholar] [CrossRef]
- Kwak, N.S.; Müller, K.R.; Lee, S.W. A convolutional neural network for steady state visual evoked potential classification under ambulatory environment. PLoS ONE 2017, 12, e0172578. [Google Scholar] [CrossRef]
- Yu, Z.; Li, L.; Song, J.; Lv, H. The Study of Visual-Auditory Interactions on Lower Limb Motor Imagery. Front. Neurosci. 2018, 12, 509. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Prasad, S.; Kilicarslan, A.; Contreras-Vidal, J.L. Multiple kernel based region importance learning for neural classification of gait states from EEG signals. Front. Neurosci. 2017, 11, 170. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Chen, W.; Pei, Z.; Wang, J. A brain-controlled lower-limb exoskeleton for human gait training. Rev. Sci. Instr. 2017, 88, 104302. [Google Scholar] [CrossRef] [PubMed]
- Kilicarslan, A.; Grossman, R.G.; Contreras-Vidal, J.L. A robust adaptive denoising framework for real-time artifact removal in scalp EEG measurements. J. Neural Eng. 2016, 13, 026013. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; Neuper, C.; Flotzinger, D.; Pregenzer, M. EEG-based discrimination between imagination of right and left hand movement. Electroencephalogr. Clin. Neurophysiol. 1997, 103, 642–651. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; Lopes Da Silva, F.H. Event-related EEG/MEG synchronization and desynchronization: Basic principles. Clin. Neurophysiol. 1999, 110, 1842–1857. [Google Scholar] [CrossRef]
- Seeland, A.; Manca, L.; Kirchner, F.; Kirchner, E.A. Spatio-temporal comparison between ERD/ERS and MRCP-based movement prediction. In Proceedings of the BIOSIGNALS 20158th International Conference on Bio-Inspired Systems and Signal Processing, Proceedings, Part of 8th International Joint Conference on Biomedical Engineering Systems and Technologies, BIOSTEC 2015, Lisbon, Portugal, 12–15 January 2015; pp. 219–226. [Google Scholar] [CrossRef]
- Choi, J.; Kim, K.T.; Jeong, J.H.; Kim, L.; Lee, S.J.; Kim, H. Developing a Motor Imagery-Based Real-Time Asynchronous Hybrid BCI Controller for a Lower-Limb Exoskeleton. Sensors 2020, 20, 7309. [Google Scholar] [CrossRef] [PubMed]
- Ferrero, L.; Quiles, V.; Ortiz, M.; Iáñez, E.; Azorín, J.M. A BMI Based on Motor Imagery and Attention for Commanding a Lower-Limb Robotic Exoskeleton: A Case Study. Appl. Sci. 2021, 11, 4106. [Google Scholar] [CrossRef]
- Gharabaghi, A. What Turns Assistive into Restorative Brain-Machine Interfaces? Front. Neurosci. 2016, 10, 456. [Google Scholar] [CrossRef] [PubMed]
- Tomasi, D.; Volkow, N.D. Laterality patterns of brain functional connectivity: Gender effects. Cereb. Cortex (New York NY 1991) 2012, 22, 1455–1462. [Google Scholar] [CrossRef]
- Ocklenburg, S.; Hirnstein, M.; Beste, C.; Güntürkün, O. Lateralization and cognitive systems. Front. Psychol. 2014, 5, 1143. [Google Scholar] [CrossRef]
- Sokołowska, B. A novel virtual reality approach for functional lateralization in healthy adults. Brain Res. 2021, 1766, 147537. [Google Scholar] [CrossRef]
- Parsons, L.M.; Gabrieli, J.D.; Phelps, E.A.; Gazzaniga, M.S. Cerebrally lateralized mental representations of hand shape and movement. J. Neurosci. 1998, 18, 6539–6548. [Google Scholar] [CrossRef]
- Zapała, D.; Zabielska-Mendyk, E.; Augustynowicz, P.; Cudo, A.; Jaśkiewicz, M.; Szewczyk, M.; Kopiś, N.; Francuz, P. The effects of handedness on sensorimotor rhythm desynchronization and motor-imagery BCI control. Sci. Rep. 2020, 10, 2087. [Google Scholar] [CrossRef]
- Shieh, C.P.; Yang, S.H.; Liu, Y.S.; Kuo, Y.T.; Lo, Y.C.; Kuo, C.H.; Chen, Y.Y. Simultaneously Spatiospectral Pattern Learning and Contaminated Trial Pruning for Electroencephalography-Based Brain Computer Interface. Symmetry 2020, 12, 1387. [Google Scholar] [CrossRef]
- Pfurtscheller, G.; Brunner, C.; Schlo, A.; Lopes, F.H. Mu rhythm (de) synchronization and EEG single-trial classification of different motor imagery tasks. NeuroImage 2006, 31, 153–159. [Google Scholar] [CrossRef]
- Tariq, M.; Trivailo, P.M.; Simic, M. Mu-Beta event-related (de)synchronization and EEG classification of left-right foot dorsiflexion kinaesthetic motor imagery for BCI. PLoS ONE 2020, 15, e0230184. [Google Scholar] [CrossRef]
- Tariq, M.; Uhlenberg, L.; Trivailo, P.; Munir, K.S.; Simic, M. Mu-beta rhythm ERD/ERS quantification for foot motor execution and imagery tasks in BCI applications. In Proceedings of the 2017 8th IEEE International Conference on Cognitive Infocommunications (CogInfoCom), Debrecen, Hungary, 11–14 September 2017; pp. 91–96. [Google Scholar] [CrossRef]
- Crémers, J.; Dessoullières, A.; Garraux, G. Hemispheric specialization during mental imagery of brisk walking. Hum. Brain Mapp. 2012, 33, 873–882. [Google Scholar] [CrossRef]
- Jahn, K.; Deutschländer, A.; Stephan, T.; Strupp, M.; Wiesmann, M.; Brandt, T. Brain activation patterns during imagined stance and locomotion in functional magnetic resonance imaging. NeuroImage 2004, 22, 1722–1731. [Google Scholar] [CrossRef]
- Ang, K.K.; Chin, Z.Y.; Zhang, H.; Guan, C. Filter Bank Common Spatial Pattern (FBCSP) in brain–computer interface. In Proceedings of the International Joint Conference on Neural Networks, Hong Kong, China, 1–8 June 2008; pp. 2390–2397. [Google Scholar] [CrossRef]
- Ortiz, M.; Rodríguez-Ugarte, M.; Iáñez, E.; Azorín, J.M. Application of the Stockwell Transform to Electroencephalographic Signal Analysis during Gait Cycle. Front. Neurosci. 2017, 11, 660. [Google Scholar] [CrossRef] [PubMed]
- Solomon, O.M.J. PSD Computations Using Welch’s Method. [Power Spectral Density (PSD)]; Technical Report; Sandia National Labs.: Albuquerque, NM, USA, 1991. [Google Scholar] [CrossRef]
- Ramoser, H.; Müller-Gerking, J.; Pfurtscheller, G. Optimal Spatial Filtering of Single Trial EEG During Imagined Hand Movement. IEEE Trans. Rehabil. Eng. A Publ. IEEE Eng. Med. Biol. Soc. 2001, 8, 441–446. [Google Scholar] [CrossRef] [PubMed]
- McFarland, D.J.; McCane, L.M.; David, S.V.; Wolpaw, J.R. Spatial filter selection for EEG-based communication. Electroencephalogr. Clin. Neurophysiol. 1997, 103, 386–394. [Google Scholar] [CrossRef]
- Izenman, A. Linear Discriminant Analysis. In Modern Multivariate Statistical Techniques; Springer Texts in Statistics; Springer: New York, NY, USA, 2013. [Google Scholar]
- Ferrero, L.; Quiles, V.; Ortiz, M.; Iáñez, E.; Azorín, J.M. BCI Based on Lower-Limb Motor Imagery and a State Machine for Walking on a Treadmill. In Proceedings of the International IEEE EMBS Conference on Neural Engineering, Virtual, Italy, 4–6 May 2021. [Google Scholar]
- Ferrero, L.; Ortiz, M.; Quiles, V.; Iáñez, E.; Azorín, J.M. Improving Motor Imagery of Gait on a Brain–Computer Interface by Means of Virtual Reality: A Case of Study. IEEE Access 2021, 9, 49121–49130. [Google Scholar] [CrossRef]
- Tariq, M.; Trivailo, P.M.; Simic, M. Classification of left and right foot kinaesthetic motor imagery using common spatial pattern. Biomed. Phys. Eng. Express 2019, 6, 15008. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Eskandarian, A. A Computationally Efficient Multiclass Time-Frequency Common Spatial Pattern Analysis on EEG Motor Imagery. In Proceedings of the 2020 42nd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Montreal, QC, Canada, 20–24 July 2020. [Google Scholar]
- Leeuwis, N.; Paas, A.; Alimardani, M. Vividness of Visual Imagery and Personality Impact Motor-Imagery Brain Computer Interfaces. Front. Hum. Neurosci. 2021, 15, 634748. [Google Scholar] [CrossRef]
- Lotte, F.; Bougrain, L.; Cichocki, A.; Clerc, M.; Congedo, M.; Rakotomamonjy, A.; Yger, F. A review of classification algorithms for EEG-based brain—Computer interfaces: A 10 year update. J. Neural Eng. 2018, 15, 031005. [Google Scholar] [CrossRef]
- Gordleeva, S.Y.; Lukoyanov, M.V.; Mineev, S.A.; Khoruzhko, M.A.; Mironov, V.I.; Kaplan, A.Y.; Kazantsev, V.B. Exoskeleton control system based on motor-imaginary brain–computer interface. Sovrem. Tehnol. V Med. 2017, 9, 31–36. [Google Scholar] [CrossRef]
- Ortiz, M.; Ferrero, L.; Iáñez, E.; Azorín, J.M.; Contreras-Vidal, J.L. Sensory Integration in Human Movement: A New Brain-Machine Interface Based on Gamma Band and Attention Level for Controlling a Lower-Limb Exoskeleton. Front. Bioeng. Biotechnol. 2020, 8, 735. [Google Scholar] [CrossRef]
- Zhang, T.; Liu, T.; Li, F.; Li, M.; Liu, D.; Zhang, R.; He, H.; Li, P.; Gong, J.; Luo, C.; et al. Structural and functional correlates of motor imagery BCI performance: Insights from the patterns of fronto-parietal attention network. NeuroImage 2016, 134, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, J.A.; Zielinski, B.A.; Ferguson, M.A.; Lainhart, J.E.; Anderson, J.S. An Evaluation of the Left-Brain vs. Right-Brain Hypothesis with Resting State Functional Connectivity Magnetic Resonance Imaging. PLoS ONE 2013, 8, e71275. [Google Scholar]
- Sabaté, M.; Gonzalez, B.; Díaz, M. Brain lateralization of motor imagery: Motor planning asymmetry as a cause of movement lateralization. Neuropsychologia 2004, 42, 1041–1049. [Google Scholar] [CrossRef]
- Labriffe, M.; Annweiler, C.; Amirova, L.E.; Gauquelin-Koch, G.; Ter Minassian, A.; Leiber, L.M.; Beauchet, O.; Custaud, M.A.; Dinomais, M. Brain Activity during Mental Imagery of Gait Versus Gait-Like Plantar Stimulation: A Novel Combined Functional MRI Paradigm to Better Understand Cerebral Gait Control. Front. Hum. Neurosci. 2017, 11, 106. [Google Scholar] [CrossRef]
- Li, G.L.; Wu, J.T.; Xia, Y.H.; He, Q.G.; Jin, H.G. Review of semi-dry electrodes for EEG recording. J. Neural Eng. 2020, 17, 51004. [Google Scholar] [CrossRef]
- Li, G.; Wang, S.; Li, M.; Duan, Y.Y. Towards real-life EEG applications: Novel superporous hydrogel-based semi-dry EEG electrodes enabling automatically ‘charge–discharge’ electrolyte. J. Neural Eng. 2021, 18, 046016. [Google Scholar] [CrossRef] [PubMed]
- Zich, C.; De Vos, M.; Kranczioch, C.; Debener, S. Wireless EEG with individualized channel layout enables efficient motor imagery training. Clin. Neurophysiol. 2015, 126, 698–710. [Google Scholar] [CrossRef] [PubMed]

| FBCSP | ST | Welch’s PSD | ||
|---|---|---|---|---|
| Full-static | %MI | |||
| % Relax | ||||
| %Total | ||||
| Full-motion | %MI | |||
| % Relax | ||||
| %Total |
| S1 | S2 | S3 | S4 | S5 | ||
|---|---|---|---|---|---|---|
| Full-static | %MI | 59.5 ± 7.6 | 70.4 ± 6.4 | 63.4 ± 4.6 | 64.7 ± 6.6 | 65.4 ± 4.0 |
| % Relax | 69.4 ± 7.1 | 67.7 ± 7.5 | 63.1 ± 3.3 | 69.2 ± 16.0 | 67.8 ± 10.5 | |
| %Total | 64.4 ± 6.2 | 69.0 ± 5.7 | 63.3 ± 1.7 | 67.2 ± 11.2 | 67.2 ± 11.2 | |
| Full-motion | %MI | 62.8 ± 7.7 | 64.3 ± 9.0 | 54.9 ± 9.0 | 64.2 ± 11.0 | 59.3 ± 5.4 |
| % Relax | 64.5 ± 3.5 | 62.7 ± 6.0 | 60.7 ± 11.5 | 65.1 ± 13.0 | 59.8 ± 6.0 | |
| %Total | 63.6 ± 3.0 | 63.5 ± 5.5 | 57.8 ± 5.4 | 57.8 ± 5.4 | 57.8 ± 5.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrero, L.; Ortiz, M.; Quiles, V.; Iáñez, E.; Flores, J.A.; Azorín, J.M. Brain Symmetry Analysis during the Use of a BCI Based on Motor Imagery for the Control of a Lower-Limb Exoskeleton. Symmetry 2021, 13, 1746. https://doi.org/10.3390/sym13091746
Ferrero L, Ortiz M, Quiles V, Iáñez E, Flores JA, Azorín JM. Brain Symmetry Analysis during the Use of a BCI Based on Motor Imagery for the Control of a Lower-Limb Exoskeleton. Symmetry. 2021; 13(9):1746. https://doi.org/10.3390/sym13091746
Chicago/Turabian StyleFerrero, Laura, Mario Ortiz, Vicente Quiles, Eduardo Iáñez, José A. Flores, and José M. Azorín. 2021. "Brain Symmetry Analysis during the Use of a BCI Based on Motor Imagery for the Control of a Lower-Limb Exoskeleton" Symmetry 13, no. 9: 1746. https://doi.org/10.3390/sym13091746
APA StyleFerrero, L., Ortiz, M., Quiles, V., Iáñez, E., Flores, J. A., & Azorín, J. M. (2021). Brain Symmetry Analysis during the Use of a BCI Based on Motor Imagery for the Control of a Lower-Limb Exoskeleton. Symmetry, 13(9), 1746. https://doi.org/10.3390/sym13091746









