Abstract
While the desire to uncover the neural correlates of consciousness has taken numerous directions, self-face recognition has been a constant in attempts to isolate aspects of self-awareness. The neuroimaging revolution of the 1990s brought about systematic attempts to isolate the underlying neural basis of self-face recognition. These studies, including some of the first fMRI (functional magnetic resonance imaging) examinations, revealed a right-hemisphere bias for self-face recognition in a diverse set of regions including the insula, the dorsal frontal lobe, the temporal parietal junction, and the medial temporal cortex. In this systematic review, we provide confirmation of these data (which are correlational) which were provided by TMS (transcranial magnetic stimulation) and patients in which direct inhibition or ablation of right-hemisphere regions leads to a disruption or absence of self-face recognition. These data are consistent with a number of theories including a right-hemisphere dominance for self-awareness and/or a right-hemisphere specialization for identifying significant social relationships, including to oneself.
1. Introduction
The evolution of animal nervous system symmetry is complex, with many resulting variants [1,2,3,4,5]. Allowing for numerous phenotypic advantages, including those at both an individual and social/interactive level [6,7,8,9], the nervous systems of bilateral organisms have exploited the benefits of a lateralized nervous system for hundreds of millions of years [2,10] (but see [11]).
The human brain is no exception [2,12]. The first impression of the human brain was noted as far back as the Ancient Greeks as two distinct hemispheres. Except in rare cases of severe abnormal development, a human at any stage post-second trimester will anatomically have two distinctly visible hemispheres. These anatomical differences have given rise to functional differences, scientifically noted by Broca, Wernicke, and others in the late 1800s and early 1900s [13]. While language remains the most well-known of human brain lateralization, many other functions appear distinctly prominent in one hemisphere [14,15,16,17,18].
That being said, research concerning left and right hemisphere differences (LH/RH) in the brain appears to trend from ‘too simplistic’ to ‘explains everything’. While Roger Sperry’s Nobel Prize in 1981 seemed to cement the legitimacy of exploring hemispheric differences [19], the popular press has run with mythical notions such as people being ‘right- or left-brained’. The more measured approach is understanding both the ultimate and proximate reasons for asymmetries.
For example, the motor asymmetries observed in humans and other primates [12,20,21,22] have led to numerous theories the most useful of which include both the mechanisms and the underlying cost/benefit analyses. An example of this is that human cradling (mother/infant) is performed employing the left arm the majority of the time [8,23,24,25]. Such a bias is explained by socio-affective communication being RH dominant, which clearly taps into evolutionary explanations (i.e., facilitating social bonding) while also explaining functional brain hemispheric differences. That is, these data help to resolve why certain aspects of emotional communication may be RH dominant and how right-handedness itself may have evolved.
Here we describe the evidence that self-face recognition (SFR) is RH dominant and speculate that it is related to the underlying construct of self-awareness (SA; [26]), and we provide a review of the literature. It is recognized that while the evidence for RH dominance is robust [27], thus far the data are suggestive in terms of a link between SFR and SA [28,29,30]. In terms of evolution, we know even less. For example, while initial evidence indicates hemispheric differences in terms of SFR in chimpanzees [31,32], other animals (particularly the magpie, who may or may not have SFR: [33,34] may have an entirely different underlying neural structure that supports SFR and, potentially, SA. We therefore are unable to know at this point whether SFR has evolved independently or in a more homologous manner [35].
The cone of uncertainty widens in terms of SFR as we move phenotypically and phylogenetically further from Homo sapiens. In humans, we propose SFR as the benchmark against which all measures of self-awareness should be tested. We suggest, in fact, that despite numerous challenges, SFR in the great apes indicates SA, and we predict that in the near future, homologous neural underpinnings in the RH will be discovered that sustain SFR. We equally suggest that as we move to cetaceans, corvids, elephants, etc., SFR becomes more unstable, and both the underlying structures and the underlying indices of SFR become much less clear. Our purpose here is to highlight the main research that underlies these claims. While not a thorough review, this summary is intended to provide a clear and concise overview of SFR.
2. The History of Self-Face Recognition: Measuring Self-Awareness
Questions concerning self-awareness have been posed by almost all humans, including scientists, for millennia. From the Ancient Greek scholars, all the way through Gallup’s mirror self-recognition tests in the 1970s, to today’s modern brain imaging techniques, self-awareness has always been an intriguing topic to study and investigate.
Greek philosophers, including Socrates (b. 470 B.C.E.), believed that introspection was necessary for humans to be truly cognizant and pure. Plato (b. ca. 428 B.C.E.) took this concept of self even further and stated that introspection was a human obligation and that knowledge of “good” and “self” were needed in order to be honorable and principled. Importantly, Aristotle (b. 384 B.C.E.) took a comparative approach to look at differences between self-awareness in humans and nonhuman animals through studying cognitive intelligence. He concluded that both humans and animals had basic functions, such as sight, smell, taste, etc., but pure intellect was only found in humans, which made a large distinction between humans and animals. Additionally, Aristotle was one of the first to attempt to create a relationship between the self, soul, and body [36].
Most famously, the French mathematician René Descartes (b. 1596) took the study of consciousness further, as he is often considered among the first neuroscientists that attempted to localize the self. Many of his ideas are still commonly spoken of today, such as “Cogito, ergo sum” (“I think; therefore, I am”), which speculated that the self can exist because it can think of its own existence. Outside of just defining the self, Descartes attempted to actually locate the self in the brain. Although his determination of self in the brain as being located in the pineal gland (due to its centralized position in the brain) was ultimately wrong, his comparative look at humans and animals had a lasting influence [37,38,39,40]. Unlike Aristotle, Descartes believed that animals are intelligent but do not have a soul or self. He found that animals do not use language, behave on impulses, and are not adaptable, so they cannot have a self.
The Ancient Greek philosophers and Descartes laid the groundwork for formalizing scientific investigations of the self under the umbrella of psychology. Most famously, Sigmund Freud explained the deep, buried unconscious mind by using the self while simultaneously explaining the self [36]. Carl Jung believed that there were many common selves that people shared to some degree. Jean Piaget believed that children refine their self through assimilation and childhood experiences, which, in turn, play a key role in growing into adulthood. Many philosophers (including Locke, Sartre, Hegel, and Hume) and psychologists (e.g., Seligman, Beck, and Kohler) examined relationships between self-awareness and cognition.
It is noted that many individuals were addressing self/other distinctions as other sciences came into their own. In the 18th century, Carolus Linnaeus, a Swedish botanist, created a binomial classification system for living organisms. While it is known that many of his taxonomic names are still used today, such as kingdom, order, species, etc., we note here that one important classification was grouping humans along with other animals such as monkeys, apes, and bats as primates. Linnaeus remembered a Latin inscription, “Nosce te ipsum” (“Know thyself”), translated from the Greek above the Temple of Apollo at Delphi, from which he assumed that the distinction between humans and other primates is the capability for self-recognition and self-knowledge. Therefore, he categorized humans into Homo sapiens, wise men, since he believed that self-awareness was the highest form of uniquely human intelligence [41,42].
Up until this time, the self was quite an abstract concept with little concrete evidence, but that slowly changed when the mirror was seen as a tool to be used to measure cognitive abilities and self-awareness. Grant, in 1828, was the first person to our knowledge to use a mirror for a self-recognition study. The study found that monkeys, in general, had a surprised reaction when looking at mirror glass, but orangutans, in particular, had no emotional response to looking at the glass. The exact reaction of the orangutans was not recorded, just the lack of reaction, which was unfortunate, but enough of a reaction to record considering the monkeys’ strong reaction to the mirror. Soon after, Charles Darwin was one of the first to suggest using mirror recognition as a measure of higher cognitive abilities. His first recorded mirror test in 1840 examined the behavior of orangutans presented with a mirror, in which he recorded, and later published in 1876, that the orangutans would look at the mirror as if they were seeing another animal [43]. This led to Darwin’s conclusion that self-recognition was not an ability of nonhuman animals. Additionally, Darwin studied his 10 children as they grew up, starting in 1839, and did mirror tests with them. His conclusions were that self-awareness and self-knowing were tied to the ability to self-recognize [44].
In 1878, Maximillian Schmidt reported similar findings to Grant and Darwin (that orangutans did not have a self-reaction to a mirror but seemed to understand the reflective properties of the mirror). Schmidt noted that the orangutan was able to identify a human reflection in the mirror of someone standing nearby [45]. These findings were the norm in many studies performed around this time with nonhuman mirror tests. Another study performed by J. von Fischer in 1876 observed monkeys and baboons in front of a mirror and, once again, a negative mirror self-recognition result was reported. It is noted that in these studies, rigorous methods to determine SFR were not employed. The general sense is that an organism was placed in front of a mirror and behavior was observed, such as attacking the mirror or apathy. The main conclusion was that the various monkeys tested by von Fischer reacted to the mirror as if it was another, novel monkey.
Not too long after, in 1889, Wilhelm Preyer, a German researcher, was able to define a definite sign of self through the use of mirrors. He studied how using only language would be an inadequate use to describe the self in children due to the lack of vocabulary, not the lack of understanding of the “I”, or ego. He created developmental timelines using mirror recognition, language, and other time measures, such as own-shadow recognition, to pinpoint a child’s timeline of self-recognition. Through this, Preyer was able to confirm the use of mirrors for self-recognition tests due to his orderly, thorough reports. Although Preyer did not specifically work with apes, he did record nonhuman animal reactions compared to the human reactions [46]. Unfortunately, most mirror self-recognition researchers did not communicate together at the time, so many went unnoticed, causing a dip in the study of mirror tests for self-recognition.
Outside the occasional mirror test on monkeys, orangutans, and chimpanzees, most mirror self-recognition slowed down, until 1929. Robert and Ada Yerkes found results suggesting no self-interest in mirrors by the nonhuman primates, noting that the animals were seeing ‘another animal’ in the mirror [47]. Following this, mirror tests were performed rarely and without any true lineage of experiments to follow. In 1940, C. W. Huntley also performed a little-known experiment that recorded human participants having a large emotional reaction to the realization that the recorded voice played back, hand pictures, and handwriting were indeed their own [48]. Arnold Gesell, a Yale child developmentalist, studied similar theories in the early to mid-20th century as Piaget and Preyer and studied many self-indicators of how a child’s timeline develops these indicators [49]. Unfortunately, lack of expansion on these previous theories with mirror tests led Gesell’s reports to fall through the cracks. In the 1940s, Jacques Lacan suggested that the formation of the self and mirror recognition were correlated, but due to a lack of mirror experimentation, his research was overlooked. In 1954, in a paper for the journal Human Biology, a photograph of a chimpanzee named Vicki using a mirror to guide pliers over her teeth was taken, but the authors, Keith and Catherine Hayes, did not discuss its relevance (that there was evidence that a nonhuman had self-recognition) and it went unrecognized [50].
3. Gallup’s Mirror Self-Recognition Test
The lull of interest in mirror tests finally changed abruptly in 1970 when Gallup published research of a nonhuman-animal positive mirror self-recognition test. Gallup’s mirror and mark test combined many previous ideas about self-recognition, and in the article, he commented that there is likely a connection between self-recognition and self-knowing. By creating a test that measured a real physical trait along with a well-thought-out process that eliminated random chance, Gallup made a solid argument for self-recognition in chimpanzees. However, he and others immediately picked up the notion that self-recognition in a mirror may, in fact, be evidence that humans are not the only self-aware organism on this planet.
The first test consisted of a 10-day period of a mirror placed in the testing site with the chimpanzees to allow them to acclimate with the mirror. This created a baseline behavior of the chimpanzees, which was typical mirror behavior, as seen with previous tests where there was no significant reaction to the mirror. Then, the chimpanzees were placed under anesthesia and a small mark that was dry and odorless was placed above the eyebrow where the chimpanzees could not see directly. After waking up from anesthesia, the chimpanzees’ behavior was observed without a mirror. It was found that the chimpanzees did not react to their new mark (in the absence of a mirror), no smelling or touching occurred, and therefore, it was concluded that they were unaware of the mark. After this short baseline period, mirrors were placed in the chimpanzee test site again and their reactions were observed. Upon seeing their reflections in the mirror, the chimpanzees would touch and smell the mark and investigate their hands after touching the mark. This indicated that the animals recognized that the mark had not been there previously and that they had to use a mirror to find the mark. This test itself was a breakthrough, but Gallup conducted two more tests to confirm this conclusion.
The second test was almost identical to the first, with the exception of the initial 10-day pre-mark mirror exposure. Gallup hypothesized that without the previous mirror exposure, the chimpanzees would not react to the mark as being odd on their face, which proved to be true. The chimpanzees were given the mark and indeed did not react to it in any significant manner, leading Gallup to conclude that the first group achieved mirror self-recognition (MSR). The third test Gallup conducted was identical to the first, but instead of chimpanzees, he used monkeys. He observed their reactions pre-mark and post-mark with a mirror, and the monkeys gave no sign of MSR [51].
The three tests put together create a clear, concise result that chimpanzees are able to recognize themselves in the mirror. Gallup published his findings in 1970 not only discussing the test and the breakthrough results he found, but also contemplating the higher consciousness of nonhuman animals. Before then, it was assumed that humans have the highest form of intelligence and cognition and that animals have some intelligence but not necessarily a “soul” or self. With these findings, Gallup opened up the discussion of nonhuman animals possibly having a higher-order cognitive process and internal world. With this mirror and mark test, Gallup also sparked a new method of research with the ability to test consciousness hypotheses.
Since Gallup’s initial findings, the literature has swayed from conservative (only humans, chimpanzees, and orangutans have self-face recognition) to today with more liberal interpretations that include the addition of animals such as elephants, dolphins, and magpies [52].
4. SFR in Animals
The advantages of Gallup’s test are many. It does not require language, which means it can be used in pre-/non-linguistic humans and nonhumans. It requires no special equipment; the equipment needed is portable and can be used in any environment. It requires little training to conduct, it is inexpensive, and it does not take months to perform. The test is noninvasive and generally culture-fair, though there are cultures where mirror exposure may be limited [42].
Gallup’s test involves a number of aspects that present difficulties in testing both human and nonhuman populations. The test relies on vision and memory, as well as intact motor systems, and alterations to these can influence results. The test requires the individual to inspect the mark to ‘pass’, which in the original form meant having limbs/arms capable of touching the mark. The test requires some aspect of ‘caring’ that one’s image is altered to the point of touching the mark. This last point is a major issue when testing special groups of humans, such as those with autism, who may know that there is a mark but not feel compelled to inspect the mark [53,54]. While there are other issues (e.g., attentional processing, what qualifies as a successful touch, and the role of training vs. spontaneous SFR), it should be apparent that direct comparison across populations is difficult if not impossible.
For example, dolphins and other marine mammals are notorious for being a ‘challenging’ population to test as mark-directed responses are near impossible to conduct [55,56,57]. Self-recognition has been claimed not via mark-directed touching but via inspection of the mark in a mirror and self-directed behavior conducted by use of the mirror. Horses have been tested via ‘scraping’ behavior of a mark on the cheek [58]. Elephants are considered to pass if they make a trunk-directed response [59,60,61].
It is possible that tests for SFR are most opaque in terms of the avians [62,63,64,65,66]. While an early report indicated magpies passing a modified version of Gallup’s test [64], this finding has not been consistent [33]. B.F. Skinner was one of the first to perform SFR avian studies when he and his colleagues conditioned pigeons to peck at a spot [67]; see [65,66], which caused others to emphasize that SFR must be spontaneous and not ‘taught’ [68]. Other attempts to modify the test have included making the test more phenotypically relevant by using olfaction rather than vision [69]. While this is but a brief summary, we remain skeptical about SFR in non-great apes (with the exception of dolphins) and conclude that more evidence is needed before we consider organisms outside of apes and dolphins to have SFR abilities [52].
5. Nonhumans and Brain Symmetry
Before presenting evidence of a lateralized system for SFR, it is worth examining the sparse evidence of the neural correlates of SFR in nonhumans. Bill Hopkins and his team examined mirror self-recognizers (MSR+) vs. non-recognizers (MSR−). Employing diffusion tensor imaging, a lateralized MSR+ vs. MSR− difference was found in the superior longitudinal fasciculus (including frontal lobe areas). The authors indicated that SFR was associated with a greater RH asymmetry [31].
A follow-up study found that MSR+ chimpanzees had increased cortical thickness bilaterally in the caudal anterior cingulate gyrus (mostly in the right hemisphere) and thinner cortex in the central portion of the pre- and postcentral gyri, primarily in the left hemisphere [32].
These results are too limited to draw distinct evolutionary patterns, but they are consistent with the RH asymmetry observed in humans in terms of SFR. That is, it is unclear whether the neural architecture that provides MSR in nonhumans is homologous to what occurs in humans and less clear whether there is a direct evolutionary path. However, taken together with what we find in humans, the data suggest that at least in chimpanzees, there are homologies rather than analogies. That is, the early data indicate that the same rightward bias for MSR that exists in chimpanzees also exists in humans.
6. Functional Imaging Indicates Right-Hemisphere Dominance in Self-Face Recognition
In the early 1990s through to today, research has examined how the brain actually allows MSR, treating it as an exceptional ability. Before neuroimaging, however, there were indicators of a possible right-hemisphere (RH) bias, as disorders of RH neural structures sometimes lead to a lack of own-body recognition [41,70,71,72,73] and disorders with self-awareness deficits appeared to be similar to RH disorder. Early attempts to determine the correlates of self-face recognition were made by pioneers such as Preilowski [74], who was the first to suggest a RH bias even though his methods involved indirect indicators.
In an early attempt to test Preilowski’s hypotheses, we employed the WADA method in which the anterior portion of one hemisphere is anesthetized. Using self-face morphs (e.g., self-morphed to Marylin Monroe), it was found that following RH anesthesia, patients had significant difficulty recognizing their own face (Figure 1 [75]). There were early attempts to use lateralized hand response differences as a further test of RH SFR [65,66]. However, as is the case in much of cognitive neuroscience, fMRI (functional magnetic resonance imaging) dominates the literature.
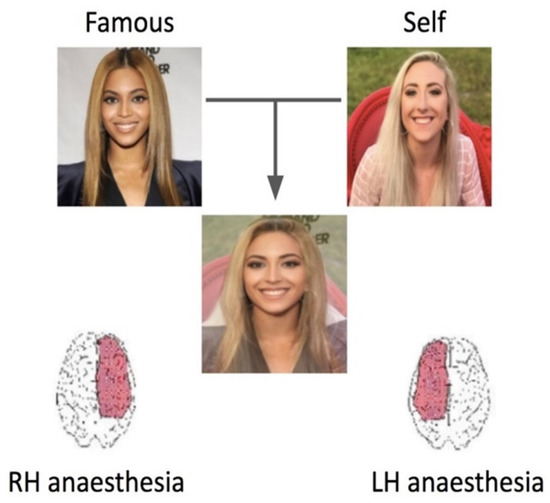
Figure 1.
Numerous methods have been employed to reach the conclusion that there is a RH lateralization in terms of MSR/SFR. Anesthesia applied to the brain in either the right or left hemisphere leads to differences in SFR. Namely, patients without a fully functioning RH see morphed images as not themselves. In this case, they report the image as Beyoncé under RH anesthesia conditions and their own face under LH anesthesia conditions [75].
Functional MRI (fMRI) of the self-face commenced in the mid-1990s with simple designs involving either unaltered self-faces contrasting with other faces or basic morphs [68]. Over time, the designs became more sophisticated, including examining affect, psychiatric disorders [76,77,78,79,80], and family [81]. While there is an overwhelming bias for RH activity in these studies, fMRI also revealed both a wide distribution of regions and the notion that many variables influence how the brain processes one’s own face [82,83]. Much of the variability across the studies is for reasons unknown as the studies have not been replicated. Therefore, the fact that in one study a face is presented for a certain duration (for example) may be the factor, or it may be the task itself. We do find convincing data that suggest the more the SFR task engages self-reflection, the greater the bilateral medial frontal activity is [27].
Sugiura’s group was one of the pioneers in determining the cortical correlates of SFR using fMRI [84,85,86,87,88,89,90,91]. Importantly, it was found that the brain has a number of distinct regions/networks associated with self–other distinctions. While there is overlap, the critical finding is that RH activation works in concert with medial frontal areas when SFR is performed in a social context. In other words, the social component of SFR appears to draw on the RH, as well as medial frontal networks.
Morita and colleagues also conducted numerous SFR fMRI studies and found a consistent RH bias [78,92,93,94,95,96]. In terms of brain symmetry, she solved a problem that has baffled researchers—the role that handedness plays in the lateralization of SFR [92]. They discovered that most right-handed participants exhibited a RH SFR bias. Likewise, most left-handers had RH bias, but there were significant numbers that had LH-localized SFR comparted to the right-handers. Therefore, it appears that, like handedness itself, which sometimes involves a shift in verbal language dominance, SFR may also shift to the LH in concert with some cases of left-handedness.
It may be surprising that three meta-analyses have been performed on SFR and the brain [27,97,98]. All three indicate a RH bias in SFR, though there are bilateral activations at a much lesser level. A key question that fMRI has helped to answer is the relationship between SFR and metacognition, either of oneself (SA) or of others (Theory of Mind: TOM). That is, if there is a relationship between SFR and TOM at a neurological level, such a relationship may be not just spurious but related in a meaningful way behaviorally and at an evolutionary level. It was found in two of the meta-analyses that SFR activates RH networks and overlaps with cortical midline structures in terms of metacognition, most likely critically involving medial regions of the frontal lobe [27,98]. We therefore conclude, based on fMRI, that the RH is likely necessary but not sufficient for SA.
Functional MRI provides only a correlational relationship between brain activity and behavior. By the 2000s, research was regularly appearing demonstrating a causal relationship. That is, by disrupting regions of the RH using noninvasive techniques, researchers were discovering that SFR was not just correlated with the RH but was actually involved in a causal relationship. The main methods employed thus far involve a version of a ‘virtual lesion’ in which a brain area is either temporarily taken offline or temporarily severely inhibited [99,100]. Basically, different regions of the brain were disrupted, and the subsequent changes in self-face recognition were measured, which established causality.
While early attempts using TMS (transcranial magnetic stimulation) did distinguish the right hemisphere as being necessary for self-face recognition [75], the most elegant of these studies was provided by Lucina Uddin and her colleagues [101]. She found that right parietal TMS disrupted self-face recognition, whereas left parietal TMS did not. Working in a somewhat ‘backwards’ manner, Uddin took these causal data and supported them (i.e., RH dominance) with correlational imaging data [102,103].
More recent studies have found that disruption of the RH is more dramatic for those individuals with subclinical grandiose narcissism [104]. That is, as narcissistic traits increase, TMS delivered to the RH causes a greater disruption of recognition of one’s own face. This study, unlike studies of autism (see below), is one in which we see an excess of SA (rather than a deficit) correlating with some measure of SFR. We speculate that hyperactivity of self-associated neural networks associated with narcissistic traits leads to a steeper decline in function when disrupted (compared to normal activity), though this hypothesis needs further testing.
An even more interesting study revealed that the RH bias for self-faces may in fact be subconscious, below one even needing to identify whether the face is their own. Using a mental rotation task involving either one’s own face or the face of another, Zeugin and his colleagues found that RH parietal TMS disrupted mental rotation of self-face compared to familiar faces in general [105]. This might indicate that the ‘specialness’ of the self-face is much ‘deeper’ in one’s cognitive schema and does not rely on conscious representation. It would be interesting to test narcissistic traits (as in [104]) to see whether there are similar contributions of narcissism at the implicit level, as we previously observed at the explicit level.
Confirming these TMS studies in terms of brain/behavior disruption, a similar technique known as tDCS (transcranial direct cortical stimulation) was employed to alter the brain with self-face identification being measured. It was found that disruption of the temporo-parietal lobe in the RH disrupted self-face perception [106].
As is often the case in neuroscience, we have to ask whether we can extrapolate the data from the lab to the clinic to the ‘real world’. While patient data from post-callosotomy (split-brain) surgery individuals [103] to those with autism [53,78,103] suggested a possible RH bias, a more specific method for detailing potential asymmetries is need. Specifically, we need to turn to those that lose self-face recognition in the absence of prosopagnosia (i.e., general face-recognition loss).
7. Patient Data: Delusional Misidentification Syndrome
While TMS and other neuroimaging points towards a RH bias in SFR, the data become infinitely stronger if matched within a patient population. Losing the ability to recognize the self is a hallmark of late-stage dementia. However, this deficit tells us little about the locality of SFR. Although rare, there are in fact cases where the loss of SFR occurs against the background of relatively stable cognitive processing [107].
Delusional misidentification syndrome (DMS) refers to circumstances in which patients form a fixed, distorted belief regarding the identity of a person, place, or object [107,108]. These disorders include Capgras syndrome, in which a person that was once very familiar to the patient (e.g., their husband) is now perceived as a stranger. Amazingly, there are cases where Capgras exists exclusively for the self, in which a patient misidentifies him/herself as being either a stranger they have not met or a different, familiar person that is not the patient. This disorder is rare, and there are only a few cases in the existing literature. It is not agreed upon yet what the naming of this disorder is, but it tends to be referred to as mirror sign or Capgras for the self [109,110,111,112,113,114].
In the most substantive review to this point, David Roane et al. [115] examined 24 case reports of the mirror sign. Of note is that within most of these cases, the patients were successful in correctly identifying the mirror images of others, signifying that they do not have a general impairment in recognition of familiar faces and they understand what a mirror does [116]. That is, the loss of face recognition was exclusive to the self and not due to prosopagnosia or a lack of understanding of what a mirror does/how it functions. In terms of localization, there was a diverse range of methods employed to obtain anatomical and functional data. Of the 24 patients, 9 of them had clear evidence for RH damage including the “parietal, temporoparietal, occipito-temporal, dorsolateral frontal cortex, basal ganglia, and thalamus” [115]. Out of 24 cases detailing mirror sign, imaging data were reported in 20 of them. Of the 18 MRI findings reported, 13 showed patients with mild generalized atrophy, as well as atrophy in specific regions within the right hemisphere of the brain. Further, PET and EEG findings supported RH dysfunction, displaying hypermetabolism in the right prefrontal, parietal, and occipital–temporal cortex and right temporal slowing. Thus, DMS can be considered a RH disorder [70,71,72,73].
In particular, a case report of the mirror image followed an elderly 77-year-old right-handed woman by the name of SP. SP was hearing impaired from a young age and was known to have communicated through sign language and also by lip-reading those around her. The patient’s misidentification was regarded as highly selective as she was capable of readily identifying others in the mirror, though she regarded her own reflection as “the other SP”, a companion of sorts. As expected, SP’s lack of self-face recognition was supported upon her neurological examination; her MRI scan demonstrated clear RH damage [41].
The authors concluded that the association of RH involvement is consistent with previous work linking self-recognition to the right prefrontal and right frontoparietal cortex [42]. Overall, the most common findings were localized to the RH, which is not an unexpected finding [115].
8. Why Does This Make Sense?
The question remains as to why SFR is lateralized to the RH, and the clearest answer is that we do not know for certain. Most suggest that the RH has a specialization for social processing [117,118,119,120,121,122], though this ability is not exclusive to the RH. Rather, the RH (specifically the right TPJ) appears to be critical in correctly making self–other distinctions—an ability needed for empathy and TOM [123,124]. In fact, it is now well established that the right TPJ is critical for ‘feeling another’s pain’ and the ability to apply one’s own feelings to another [123].
Overall, social patterns and self–other distinctions are biased to the RH, and it is plausible that SFR is tapping into this to the point that in the absence of SFR, there may be social deficits. For example, there are SFR deficits in pervasive developmental disorders [53,54,80,125,126,127,128,129,130], though the range of deficit is from severe to nondetected. Quevedo’s group furthered this discussion by examining SFR in clinical populations including those with depression. Persons rated high in suicidal affect and cognitions, for example, have a unique neural response to the self-face compared to those without; self-faces involve differential neural circuits including the amygdala depending on clinical diagnosis and symptomatology [131,132,133,134,135]. These data indicate that SFR in its absence may indicate a lack of SA (i.e., autism) and that SFR in its presence may indicate different degrees of SA in different populations. In anorexia nervosa, a condition with a lack of accurate SA, there is difficulty in SFR [136]. Insight into one’s own schizophrenia (i.e., schizophrenics that have awareness of their condition) correlates positively with SFR [137]. Further, disorders of consciousness also correlate (as indicated by physiological measures) with SFR [137].
The false belief tasks that are prevalent in TOM testing tap into the notion that the human brain is capable of modeling two brains/belief systems simultaneously [138,139,140]. This differentiation is critical for our social interactions as humans. For example, I need not be in pain to know that you are in pain. Parenting, reproducing, successful predation and predator avoidance, etc., are all enhanced by the ability to separate one’s own thoughts from another’s thoughts. Thus, and somewhat ironically, empathy and TOM are increased by the ability to separate one’s SA from another’s SA.
We propose that RH development and laterality has evolved for numerous reasons, though like others, TOM is likely the main impetus. What we propose, however, is that the role of teaching is overlooked in this domain. Humans are specialists at active teaching, and given the plasticity of the human brain, active teaching is a critical component of human survival. We examined the role of TOM in teaching and learning, as well as teaching communication, in a task that involved building simple Lego models and found that the best teachers have high TOM [141]. This task was demanding in terms of active teaching and social interaction, and of interest is that the learner’s TOM was not critical (i.e., a learner can have low TOM and still learn), which models human–infant interactions.
Taken together, SFR appears to be an indicator of SA, and as SA fluctuates, so does SFR. RH patients are prone to deficits of SA [71,72,73,115,142]; thus, it is not surprising to see that SFR, SA, and the RH are related. It is worth noting that we do not know whether other non-ape organisms achieve SFR, or even if SFR is possible with very different brain mechanisms or if one needs a highly lateralized brain to have SFR and/or SA [42]. Finally, we are still unclear as to whether SA is truly indicated by SFR. As it is, 30 years of neuroimaging and patient data have left us with more questions than answers.
Author Contributions
Conceptualization, A.J., B.B., M.P., K.C., J.B., N.A., S.Z. and J.P.K.; methodology; software, N.A.; validation N.A.; formal analysis, N.A.; investigation, A.J., B.B., M.P., V.M. (Victoria Mistretta), K.C., J.B., T.S. and J.P.K.; resources, N.A.; data curation, N.A.; writing—original draft preparation, A.J., B.B., M.P., V.M. (Vanessa Martinez), K.C., J.B., T.S. and J.P.K.; writing—review and editing, A.J., B.B., M.P., V.M. (Victoria Mistretta), K.C., J.B., T.S., V.M. (Vanessa Martinez), K.P., N.A., S.Z., A.S., S.S. and J.P.K.; visualization, A.J. and J.P.K.; supervision, J.P.K.; project administration, J.P.K.; funding acquisition, J.P.K., K.C. and J.B. All authors have read and agreed to the published version of the manuscript.
Funding
The work was funded by LSAMP (Louis Stokes Alliance for Minority Participation), The Crawford Foundation, and the Wehner Fund. Josh and Judy Weston provided funding as well as the Kennedy Foundation.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Aboitiz, F.; Montiel, J.; Morales, D.; Concha, M. Evolutionary divergence of the reptilian and the mammalian brains: Considerations on connectivity and development. Brain Res. Rev. 2002, 39, 141–153. [Google Scholar] [CrossRef]
- Corballis, M.C. Of mice and men—And lopsided birds. Cortex 2008, 44, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Corballis, M.C. Evolution of cerebral asymmetry. Chang. Brains Appl. Brain Plast. Adv. Recover Hum. Abil. 2019, 250, 153–178. [Google Scholar] [CrossRef]
- Genikhovich, G.; Technau, U. On the evolution of bilaterality. Development 2017, 144, 3392–3404. [Google Scholar] [CrossRef] [PubMed]
- Manuel, M. Early evolution of symmetry and polarity in metazoan body plans. C. R. Biol. 2009, 332, 184–209. [Google Scholar] [CrossRef]
- Rogers, L.J.; Vallortigara, G. Complementary Specializations of the Left and Right Sides of the Honeybee Brain. Front. Psychol. 2019, 10, 280. [Google Scholar] [CrossRef]
- Rogers, L.J.; Zucca, P.; Vallortigara, G. Advantages of having a lateralized brain. Proc. R. Soc. B Boil. Sci. 2004, 271, S420–S422. [Google Scholar] [CrossRef]
- Tommasi, L. Mechanisms and functions of brain and behavioural asymmetries. Philos. Trans. R. Soc. B Biol. Sci. 2008, 364, 855–859. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vallortigara, G.; Rogers, L.J. Survival with an asymmetrical brain: Advantages and disadvantages of cerebral lateralization. Behav. Brain Sci. 2005, 28, 575–589. [Google Scholar] [CrossRef]
- Huber, B.A.; Sinclair, B.J.; Schmitt, M. The evolution of asymmetric genitalia in spiders and insects. Biol. Rev. 2007, 82, 647–698. [Google Scholar] [CrossRef]
- Cheng, K. Learning in Cnidaria: A systematic review. Learn. Behav. 2021, 1–15. [Google Scholar] [CrossRef]
- Hopkins, W.D.; Misiura, M.; Pope, S.M.; Latash, E.M. Behavioral and brain asymmetries in primates: A preliminary evaluation of two evolutionary hypotheses. Ann. N. Y. Acad. Sci. 2015, 1359, 65–83. [Google Scholar] [CrossRef]
- Henderson, V.W. Paul Broca’s Less Heralded Contributions to Aphasia Research. Arch. Neurol. 1986, 43, 609–612. [Google Scholar] [CrossRef]
- de Haan, E.H.; Fabri, M.; Dijkerman, H.C.; Foschi, N.; Lattanzi, S.; Pinto, Y. Unified tactile detection and localisation in split-brain patients. Cortex 2020, 124, 217–223. [Google Scholar] [CrossRef]
- Esteves, M.; Lopes, S.S.; Almeida, A.; Sousa, N.; Leite-Almeida, H. Unmasking the relevance of hemispheric asymmetries—Break on through (to the other side). Prog. Neurobiol. 2020, 192, 101823. [Google Scholar] [CrossRef]
- Francks, C. Exploring human brain lateralization with molecular genetics and genomics. Ann. N. Y. Acad. Sci. 2015, 1359, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Lemée, J.-M.; Bernard, F.; Ter Minassian, A.; Menei, P. Right Hemisphere Cognitive Functions: From Clinical and Anatomical Bases to Brain Mapping During Awake Craniotomy. Part II: Neuropsychological Tasks and Brain Mapping. World Neurosurg. 2018, 118, 360–367. [Google Scholar] [CrossRef]
- Pinto, Y.; De Haan, E.H.; Lamme, V.A. The Split-Brain Phenomenon Revisited: A Single Conscious Agent with Split Perception. Trends Cogn. Sci. 2017, 21, 835–851. [Google Scholar] [CrossRef] [PubMed]
- Pearce, J. The “split brain” and Roger Wolcott Sperry (1913–1994). Rev. Neurol. 2019, 175, 217–220. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, W.D. A review of performance asymmetries in hand skill in nonhuman primates with a special emphasis on chimpanzees. Chang. Brains Appl. Brain Plast. Adv. Recover Hum. Abil. 2018, 238, 57–89. [Google Scholar] [CrossRef]
- Meguerditchian, A.; Vauclair, J.; Hopkins, W.D. On the origins of human handedness and language: A comparative review of hand preferences for bimanual coordinated actions and gestural communication in nonhuman primates. Dev. Psychobiol. 2013, 55, 637–650. [Google Scholar] [CrossRef]
- Ocklenburg, S.; Beste, C.; Arning, L.; Peterburs, J.; Güntürkün, O. The ontogenesis of language lateralization and its relation to handedness. Neurosci. Biobehav. Rev. 2014, 43, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Malatesta, G.; Marzoli, D.; Apicella, F.; Abiuso, C.; Muratori, F.; Forrester, G.S.; Vallortigara, G.; Scattoni, M.L.; Tommasi, L. Received Cradling Bias during the First Year of Life: A Retrospective Study on Children With Typical and Atypical Development. Front. Psychiatry 2020, 11, 91. [Google Scholar] [CrossRef] [PubMed]
- Malatesta, G.; Marzoli, D.; Rapino, M.; Tommasi, L. The left-cradling bias and its relationship with empathy and depression. Sci. Rep. 2019, 9, 6141. [Google Scholar] [CrossRef] [PubMed]
- Malatesta, G.; Marzoli, D.; Tommasi, L. The association between received maternal cradling and neurodevelopment: Is left better? Med. Hypotheses 2020, 134, 109442. [Google Scholar] [CrossRef]
- Gallup, G.G.; Platek, S.M.; Spaulding, K.N. The nature of visual self-recognition revisited. Trends Cogn. Sci. 2014, 18, 57–58. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Di, X.; Eickhoff, S.B.; Zhang, M.; Peng, K.; Guo, H.; Sui, J. Distinct and common aspects of physical and psychological self-representation in the brain: A meta-analysis of self-bias in facial and self-referential judgements. Neurosci. Biobehav. Rev. 2016, 61, 197–207. [Google Scholar] [CrossRef]
- Carmody, D.P.; Lewis, M. Self Representation in Children with and without Autism Spectrum Disorders. Child Psychiatry Hum. Dev. 2011, 43, 227–237. [Google Scholar] [CrossRef]
- Lewis, M.; Carmody, D.P. Self-representation and brain development. Dev. Psychol. 2008, 44, 1329–1334. [Google Scholar] [CrossRef]
- Lewis, M.; Sullivan, M.W.; Stanger, C.; Weiss, M.; Sullivan, M.W. Self Development and Self-Conscious Emotions. Child Dev. 1989, 60, 146–156. [Google Scholar] [CrossRef]
- Hecht, E.; Mahovetz, L.M.; Preuss, T.M.; Hopkins, W.D. A neuroanatomical predictor of mirror self-recognition in chimpanzees. Soc. Cogn. Affect. Neurosci. 2016, 12, 37–48. [Google Scholar] [CrossRef]
- Hopkins, W.D.; Latzman, R.D.; Mahovetz, L.M.; Li, X.; Roberts, N. Investigating individual differences in chimpanzee mirror self-recognition and cortical thickness: A vertex-based and region-of-interest analysis. Cortex 2019, 118, 306–314. [Google Scholar] [CrossRef]
- Soler, M.; Colmenero, J.M.; Pérez-Contreras, T.; Peralta-Sánchez, J.M. Replication of the mirror mark test experiment in the magpie (Pica pica) does not provide evidence of self-recognition. J. Comp. Psychol. 2020, 134, 363–371. [Google Scholar] [CrossRef]
- Soler, M.; Pérez-Contreras, T.; Peralta-Sánchez, J.M. Mirror-Mark Tests Performed on Jackdaws Reveal Potential Methodological Problems in the Use of Stickers in Avian Mark-Test Studies. PLoS ONE 2014, 9, e86193. [Google Scholar] [CrossRef] [PubMed]
- Povinelli, D.J. Arboreal Clambering and the Evolution of Self-Conception. Q. Rev. Biol. 1995, 70, 393–421. [Google Scholar] [CrossRef]
- Tresan, D.I. This new science of ours: A more or less systematic history of consciousness and transcendence Part I. J. Anal. Psychol. 2004, 49, 193–216. [Google Scholar] [CrossRef]
- Bassiri, N. Material translations in the Cartesian brain. Stud. Hist. Philos. Sci. Part C Stud. Hist. Philos. Biol. Biomed. Sci. 2012, 43, 244–255. [Google Scholar] [CrossRef] [PubMed]
- Berhouma, M. Beyond the pineal gland assumption: A neuroanatomical appraisal of dualism in Descartes’ philosophy. Clin. Neurol. Neurosurg. 2013, 115, 1661–1670. [Google Scholar] [CrossRef]
- López-Muñoz, F.; Marín, F.; Alamo, C. The historical background of the pineal gland: I. From a spiritual valve to the seat of the soul. Revista de Neurología 2010, 50, 50–57. [Google Scholar] [PubMed]
- López-Muñoz, F.; Rubio, G.; Molina, J.; Alamo, C. La glándula pineal como instrumento físico de las facultades del alma: Una conexión histórica persistente. Neurología 2012, 27, 161–168. [Google Scholar] [CrossRef]
- Feinberg, T.E.; Keenan, J.P. Where in the brain is the self? Conscious. Cogn. 2005, 14, 661–678. [Google Scholar] [CrossRef] [PubMed]
- Keenan, J.P.; Falk, D.; Gallup, G.G. Face in the Mirror; Harper Collins: New York, NY, USA, 2001. [Google Scholar]
- Darwin, C. Sexual Selection in Relation to Monkeys. Nat. Cell Biol. 1876, 15, 18–19. [Google Scholar] [CrossRef]
- Darwin, C. A Biographical Sketch of an Infant. Ann. Neurosci. 2010, 17, 187–190. [Google Scholar] [CrossRef][Green Version]
- Schmidt, M. Beobachtungen am Orang-Utan; Zoologische Garten: Berlin, Germany, 1878; Volume 19. [Google Scholar]
- Preyer, W. The Mind of the Child, Part II. In The Development of the Intellect; Appleton and Company: New York, NY, USA, 1889. [Google Scholar]
- Yerkes, R.M.; Yerkes, A. The Great Apes; Yale University Press: New Haven, CT, USA, 1929. [Google Scholar]
- Huntley, C.W. Judgments of self based upon records of expressive behavior. J. Abnorm. Soc. Psychol. 1940, 35, 398–427. [Google Scholar] [CrossRef]
- Gesell, A.; Illg, F.L. Infant and Child in the Culture of Today. Am. Sociol. Rev. 1943, 8, 489. [Google Scholar] [CrossRef]
- Hayes, K.J.; Hayes, C. The cultural capacity of chimpanzee. Hum. Biol. 1954, 26, 288–303. [Google Scholar] [PubMed]
- Gallup, G.G. Chimpanzees: Self-Recognition. Science 1970, 167, 86–87. [Google Scholar] [CrossRef] [PubMed]
- Gallup, G.G.; Anderson, J.R. The “olfactory mirror” and other recent attempts to demonstrate self-recognition in non-primate species. Behav. Process. 2018, 148, 16–19. [Google Scholar] [CrossRef]
- Chakraborty, A.; Chakrabarti, B. Is it me? Self-recognition bias across sensory modalities and its relationship to autistic traits. Mol. Autism 2015, 6, 20. [Google Scholar] [CrossRef]
- Chakraborty, A.; Chakrabarti, B. Looking at My Own Face: Visual Processing Strategies in Self–Other Face Recognition. Front. Psychol. 2018, 9, 121. [Google Scholar] [CrossRef]
- Delfour, F.; Marten, K. Mirror image processing in three marine mammal species: Killer whales (Orcinus orca), false killer whales (Pseudorca crassidens) and California sea lions (Zalophus californianus). Behav. Process. 2001, 53, 181–190. [Google Scholar] [CrossRef]
- Morrison, R.; Reiss, D. Precocious development of self-awareness in dolphins. PLoS ONE 2018, 13, e0189813. [Google Scholar] [CrossRef]
- Reiss, D.; Marino, L. Mirror self-recognition in the bottlenose dolphin: A case of cognitive convergence. Proc. Natl. Acad. Sci. USA 2001, 98, 5937–5942. [Google Scholar] [CrossRef]
- Baragli, P.; Demuru, E.; Scopa, C.; Palagi, E. Are horses capable of mirror self-recognition? A pilot study. PLoS ONE 2017, 12, e0176717. [Google Scholar] [CrossRef]
- Hamdan, A.; Ab Latip, M.Q.; Abu Hassim, H.; Noor, M.H.M.; Azizan, T.R.P.T.; Mustapha, N.M.; Ahmad, H. A preliminary study of mirror-induced self-directed behaviour on wildlife at the Royal Belum Rainforest Malaysia. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Plotnik, J.M.; de Waal, F.B.; Moore, D.; Reiss, D. Self-recognition in the Asian elephant and future directions for cognitive research with elephants in zoological settings. Zoo Biol. 2010, 29, 179–191. [Google Scholar] [CrossRef]
- Plotnik, J.M.; de Waal, F.B.M.; Reiss, D. Self-recognition in an Asian elephant. Proc. Natl. Acad. Sci. USA 2006, 103, 17053–17057. [Google Scholar] [CrossRef] [PubMed]
- Brecht, K.F.; Müller, J.; Nieder, A. Carrion crows (Corvus corone corone) fail the mirror mark test yet again. J. Comp. Psychol. 2020, 134, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Kraft, F.-L.; Forštová, T.; Urhan, A.U.; Exnerová, A.; Brodin, A. No evidence for self-recognition in a small passerine, the great tit (Parus major) judged from the mark/mirror test. Anim. Cogn. 2017, 20, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Prior, H.; Schwarz, A.; Güntürkün, O. Mirror-Induced Behavior in the Magpie (Pica pica): Evidence of Self-Recognition. PLoS Biol. 2008, 6, e202. [Google Scholar] [CrossRef]
- Uchino, E.; Watanabe, S. Self-recognition in pigeons revisited. J. Exp. Anal. Behav. 2014, 102, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Vanhooland, L.-C.; Bugnyar, T.; Massen, J.J.M. Crows (Corvus corone ssp.) check contingency in a mirror yet fail the mirror-mark test. J. Comp. Psychol. 2020, 134, 158–169. [Google Scholar] [CrossRef] [PubMed]
- Epstein, R.; Lanza, R.; Skinner, B.F. Self-Awareness in the Pigeon. Science 1981, 212, 695–696. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.R.; Gallup, G.G. Mirror self-recognition: A review and critique of attempts to promote and engineer self-recognition in primates. Primates 2015, 56, 317–326. [Google Scholar] [CrossRef]
- Horowitz, A. Smelling themselves: Dogs investigate their own odours longer when modified in an “olfactory mirror” test. Behav. Process. 2017, 143, 17–24. [Google Scholar] [CrossRef]
- Feinberg, T.E.; Roane, D. Self-representation in delusional misidentification and confabulated “others”. Cortex 2017, 87, 118–128. [Google Scholar] [CrossRef]
- Feinberg, T.E.; Roane, D.M. Delusional Misidentification. Psychiatr. Clin. N. Am. 2005, 28, 665–683. [Google Scholar] [CrossRef]
- Feinberg, T.E.; Shapiro, R.M. Misidentification-reduplication and the right hemisphere. Neuropsychiatry Neuropsychol. Behav. Neurol. 1989, 2, 39–48. [Google Scholar]
- Feinberg, T.E.; Venneri, A.; Simone, A.M.; Fan, Y.; Northoff, G. The neuroanatomy of asomatognosia and somatoparaphrenia. J. Neurol. Neurosurg. Psychiatry 2009, 81, 276–281. [Google Scholar] [CrossRef]
- Preilowski, B. Self-recognition as a test of consciousness in the left and right hemisphere of “split-brain” patients. Act. Nerv. Super. 1977, 19 (Suppl. 2), 343–344. [Google Scholar]
- Keenan, J.P.; Nelson, A.C.G.; O’Connor, M.; Pascual-Leone, A. Self-recognition and the right hemisphere. Nat. Cell Biol. 2001, 409, 305. [Google Scholar] [CrossRef]
- Kim, M.-K.; Yoon, H.-J.; Shin, Y.-B.; Lee, S.-K.; Kim, J.-J. Neural basis of distorted self-face recognition in social anxiety disorder. NeuroImage Clin. 2016, 12, 956–964. [Google Scholar] [CrossRef] [PubMed]
- Kurth, S.; Moyse, E.; Bahri, M.A.; Salmon, E.; Bastin, C. Recognition of personally familiar faces and functional connectivity in Alzheimer’s disease. Cortex 2015, 67, 59–73. [Google Scholar] [CrossRef]
- Morita, T.; Kosaka, H.; Saito, D.N.; Ishitobi, M.; Munesue, T.; Itakura, S.; Omori, M.; Okazawa, H.; Wada, Y.; Sadato, N. Emotional responses associated with self-face processing in individuals with autism spectrum disorders: An fMRI study. Soc. Neurosci. 2012, 7, 223–239. [Google Scholar] [CrossRef] [PubMed]
- Pujol, J.; Giménez, M.; Ortiz, H.; Soriano-Mas, C.; Lopez-Sola, M.; Farre, M.; Deus, J.; Merlo-Pich, E.; Harrison, B.J.; Cardoner, N.; et al. Neural response to the observable self in social anxiety disorder. Psychol. Med. 2012, 43, 721–731. [Google Scholar] [CrossRef] [PubMed]
- Uddin, L.Q. The self in autism: An emerging view from neuroimaging. Neurocase 2011, 17, 201–208. [Google Scholar] [CrossRef]
- Platek, S.M.; Kemp, S.M. Is family special to the brain? An event-related fMRI study of familiar, familial, and self-face recognition. Neuropsychologia 2009, 47, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Kircher, T.T.; Senior, C.; Phillips, M.L.; Benson, P.J.; Bullmore, E.; Brammer, M.; Simmons, A.; Williams, S.C.; Bartels, M.; David, A.S. Towards a functional neuroanatomy of self processing: Effects of faces and words. Cogn. Brain Res. 2000, 10, 133–144. [Google Scholar] [CrossRef]
- Kircher, T.T.; Senior, C.; Phillips, M.L.; Rabe-Hesketh, S.; Benson, P.J.; Bullmore, E.T.; Brammer, M.; Simmons, A.; Bartels, M.; David, A.S. Recognizing one’s own face. Cognition 2001, 78, B1–B15. [Google Scholar] [CrossRef]
- Oikawa, H.; Sugiura, M.; Sekiguchi, A.; Tsukiura, T.; Miyauchi, C.M.; Hashimoto, T.; Takano-Yamamoto, T.; Kawashima, R. Self-face evaluation and self-esteem in young females: An fMRI study using contrast effect. NeuroImage 2012, 59, 3668–3676. [Google Scholar] [CrossRef]
- Sugiura, M. Neural basis of self-face recognition: Social aspects. Brain Nerve 2012, 64, 753–760. [Google Scholar]
- Sugiura, M. Three faces of self-face recognition: Potential for a multi-dimensional diagnostic tool. Neurosci. Res. 2015, 90, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, M.; Miyauchi, C.M.; Kotozaki, Y.; Akimoto, Y.; Nozawa, T.; Yomogida, Y.; Hanawa, S.; Yamamoto, Y.; Sakuma, A.; Nakagawa, S.; et al. Neural Mechanism for Mirrored Self-face Recognition. Cereb. Cortex 2014, 25, 2806–2814. [Google Scholar] [CrossRef]
- Sugiura, M.; Sassa, Y.; Jeong, H.; Horie, K.; Sato, S.; Kawashima, R. Face-specific and domain-general characteristics of cortical responses during self-recognition. NeuroImage 2008, 42, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, M.; Sassa, Y.; Jeong, H.; Miura, N.; Akitsuki, Y.; Horie, K.; Sato, S.; Kawashima, R. Multiple brain networks for visual self-recognition with different sensitivity for motion and body part. NeuroImage 2006, 32, 1905–1917. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, M.; Sassa, Y.; Jeong, H.; Wakusawa, K.; Horie, K.; Sato, S.; Kawashima, R. Self-face recognition in social context. Hum. Brain Mapp. 2011, 33, 1364–1374. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, M.; Watanabe, J.; Maeda, Y.; Matsue, Y.; Fukuda, H.; Kawashima, R. Cortical mechanisms of visual self-recognition. NeuroImage 2005, 24, 143–149. [Google Scholar] [CrossRef]
- Morita, T.; Asada, M.; Naito, E. Right-hemispheric Dominance in Self-body Recognition is Altered in Left-handed Individuals. Neuroscience 2020, 425, 68–89. [Google Scholar] [CrossRef]
- Morita, T.; Itakura, S.; Saito, D.N.; Nakashita, S.; Harada, T.; Kochiyama, T.; Sadato, N. The Role of the Right Prefrontal Cortex in Self-evaluation of the Face: A Functional Magnetic Resonance Imaging Study. J. Cogn. Neurosci. 2008, 20, 342–355. [Google Scholar] [CrossRef]
- Morita, T.; Saito, D.N.; Ban, M.; Shimada, K.; Okamoto, Y.; Kosaka, H.; Okazawa, H.; Asada, M.; Naito, E. Self-face recognition shares brain regions active during proprioceptive illusion in the right inferior fronto-parietal superior longitudinal fasciculus III network. Neuroscience 2017, 348, 288–301. [Google Scholar] [CrossRef] [PubMed]
- Morita, T.; Saito, D.N.; Ban, M.; Shimada, K.; Okamoto, Y.; Kosaka, H.; Okazawa, H.; Asada, M.; Naito, E. Self-Face Recognition Begins to Share Active Region in Right Inferior Parietal Lobule with Proprioceptive Illusion During Adolescence. Cereb. Cortex 2018, 28, 1532–1548. [Google Scholar] [CrossRef]
- Morita, T.; Tanabe, H.C.; Sasaki, A.T.; Shimada, K.; Kakigi, R.; Sadato, N. The anterior insular and anterior cingulate cortices in emotional processing for self-face recognition. Soc. Cogn. Affect. Neurosci. 2013, 9, 570–579. [Google Scholar] [CrossRef]
- Platek, S.M.; Wathne, K.; Tierney, N.G.; Thomson, J.W. Neural correlates of self-face recognition: An effect-location meta-analysis. Brain Res. 2008, 1232, 173–184. [Google Scholar] [CrossRef]
- Van Veluw, S.J.; Chance, S.A. Differentiating between self and others: An ALE meta-analysis of fMRI studies of self-recognition and theory of mind. Brain Imaging Behav. 2013, 8, 24–38. [Google Scholar] [CrossRef]
- Weissman-Fogel, I.; Granovsky, Y. The “virtual lesion” approach to transcranial magnetic stimulation: Studying the brain–behavioral relationships in experimental pain. Pain Rep. 2019, 4, e760. [Google Scholar] [CrossRef]
- Ziemann, U. TMS in cognitive neuroscience: Virtual lesion and beyond. Cortex 2010, 46, 124–127. [Google Scholar] [CrossRef]
- Uddin, L.Q.; Molnar-Szakacs, I.; Zaidel, E.; Iacoboni, M. rTMS to the right inferior parietal lobule disrupts self–other discrimination. Soc. Cogn. Affect. Neurosci. 2006, 1, 65–71. [Google Scholar] [CrossRef]
- Kaplan, J.T.; Aziz-Zadeh, L.; Uddin, L.Q.; Iacoboni, M. The self across the senses: An fMRI study of self-face and self-voice recognition. Soc. Cogn. Affect. Neurosci. 2008, 3, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Uddin, L.Q.; Mooshagian, E.; Zaidel, E.; Scheres, A.; Margulies, D.; Kelly, C.; Shehzad, Z.; Adelstein, J.S.; Castellanos, F.; Biswal, B.B.; et al. Residual functional connectivity in the split-brain revealed with resting-state functional MRI. NeuroReport 2008, 19, 703–709. [Google Scholar] [CrossRef]
- Kramer, R.; Duran, K.; Soder, H.; Applegate, L.; Youssef, A.; Criscione, M.; Keenan, J.P. The Special Brain: Subclinical Grandiose Narcissism and Self-Face Recognition in the Right Prefrontal Cortex. Am. J. Psychol. 2020, 133, 487–500. [Google Scholar] [CrossRef]
- Zeugin, D.; Notter, M.P.; Knebel, J.-F.; Ionta, S. Temporo-parietal contribution to the mental representations of self/other face. Brain Cogn. 2020, 143, 105600. [Google Scholar] [CrossRef]
- Payne, S.; Tsakiris, M. Anodal transcranial direct current stimulation of right temporoparietal area inhibits self-recognition. Cogn. Affect. Behav. Neurosci. 2017, 17, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Barrelle, A.; Luauté, J.-P. Capgras Syndrome and Other Delusional Misidentification Syndromes. Front. Neurol. Neurosci. 2017, 42, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Kakegawa, Y.; Isono, O.; Hanada, K.; Nishikawa, T. Incidence and lesions causative of delusional misidentification syndrome after stroke. Brain Behav. 2020, 10, 01829. [Google Scholar] [CrossRef]
- Breen, N.; Caine, D.; Coltheart, M. Mirrored-self Misidentification: Two Cases of Focal Onset Dementia. Neurocase 2001, 7, 239–254. [Google Scholar] [CrossRef]
- Gil-Ruiz, N.; Osorio, R.S.; Cruz, I.; Agüera-Ortiz, L.; Olazarán, J.; Sacks, H.; Álvarez-Linera, J.; Martínez-Martín, P.; Alzheimer Center of the Queen Sofia Foundation; Multidisciplinary Therapy Group. An effective environmental intervention for management of the ‘mirror sign’ in a case of probable Lewy body dementia. Neurocase 2013, 19, 1–13. [Google Scholar] [CrossRef]
- Mulcare, J.L.; Nicolson, S.E.; Bisen, V.S.; Sostre, S.O. The Mirror Sign: A Reflection of Cognitive Decline? J. Psychosom. Res. 2012, 53, 188–192. [Google Scholar] [CrossRef]
- Phillips, M.L. Mirror, Mirror on the Wall, Who? Towards a Model of Visual Self-recognition. Cogn. Neuropsychiatry 1996, 1, 153–164. [Google Scholar] [CrossRef] [PubMed]
- Villarejo, A.; Puertas-Martin, V.; Moreno-Ramos, T.; Camacho, A.; Porta-Etessam, J.; Bermejo-Pareja, F. Mirrored-self misidentification in a patient without dementia: Evidence for right hemispheric and bifrontal damage. Neurocase 2010, 17, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Yuki, N.; Nakagawa, M. Complex visual hallucination and mirror sign in posterior cortical atrophy. Acta Psychiatr. Scand. 2006, 114, 62–65. [Google Scholar] [CrossRef] [PubMed]
- Roane, D.M.; Feinberg, T.E.; Liberta, T.A. Delusional Misidentification of the Mirror Image. Curr. Neurol. Neurosci. Rep. 2019, 19, 55. [Google Scholar] [CrossRef]
- Bauer, R.M. The Cognitive Psychophysiology of Prosopagnosia. In Aspects of Face Processing; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 1986; pp. 253–267. [Google Scholar]
- Chen, Y.; Slinger, M.; Edgar, J.C.; Bloy, L.; Kuschner, E.S.; Kim, M.; Green, H.L.; Chiang, T.; Yount, T.; Liu, S.; et al. Maturation of hemispheric specialization for face encoding during infancy and toddlerhood. Dev. Cogn. Neurosci. 2021, 48, 100918. [Google Scholar] [CrossRef]
- Gainotti, G. The influence of handedness on hemispheric representation of tools: A survey. Brain Cogn. 2015, 94, 10–16. [Google Scholar] [CrossRef]
- Hewetson, R.; Cornwell, P.; Shum, D.H.K. Relationship and Social Network Change in People with Impaired Social Cognition Post Right Hemisphere Stroke. Am. J. Speech-Lang. Pathol. 2021, 30, 962–973. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, K.; Marsh, A.A.; Edwards, D.F.; Dromerick, A.W.; Seydell-Greenwald, A. Emotion recognition impairments and social well-being following right-hemisphere stroke. Neuropsychol. Rehabil. 2021, 384, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Okubo, M.; Ishikawa, K.; Kobayashi, A. No trust on the left side: Hemifacial asymmetries for trustworthiness and emotional expressions. Brain Cogn. 2013, 82, 181–186. [Google Scholar] [CrossRef]
- Sinha, R.; Dijkshoorn, A.B.C.; Li, C.; Manly, T.; Price, S.J. Glioblastoma surgery related emotion recognition deficits are associated with right cerebral hemisphere tract changes. Brain Commun. 2020, 2, fcaa169. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Chen, C.; Lin, C.-P.; Chou, K.-H.; Decety, J. Love hurts: An fMRI study. NeuroImage 2010, 51, 923–929. [Google Scholar] [CrossRef] [PubMed]
- Fourie, M.M.; Stein, D.J.; Solms, M.; Gobodo-Madikizela, P.; Decety, J. Empathy and moral emotions in post-apartheid South Africa: An fMRI investigation. Soc. Cogn. Affect. Neurosci. 2017, 12, 881–892. [Google Scholar] [CrossRef]
- Dawson, G.; McKissick, F.C. Self-recognition in autistic children. J. Autism Dev. Disord. 1984, 14, 383–394. [Google Scholar] [CrossRef]
- Dunphy-Lelii, S.; Wellman, H.M. Delayed self-recognition in autism: A unique difficulty? Res. Autism Spectr. Disord. 2012, 6, 212–223. [Google Scholar] [CrossRef][Green Version]
- Ferrari, M.; Matthews, W.S. Self-recognition deficits in autism: Syndrome-specific or general developmental delay? J. Autism Dev. Disord. 1983, 13, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, R.W. A comparison of the self-awareness and kinesthetic-visual matching theories of self-recognition: Autistic children and others. Ann. N. Y. Acad. Sci. 1997, 818, 39–62. [Google Scholar] [CrossRef] [PubMed]
- Neuman, C.J.; Hill, S.D. Self-recognition and stimulus preference in autistic children. Dev. Psychobiol. 1978, 11, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Spiker, D.; Ricks, M. Visual self-recognition in autistic children: Developmental relationships. Child Dev. 1984, 55, 214–225. [Google Scholar] [CrossRef]
- Liu, G.; Zhang, N.; Teoh, J.Y.; Egan, C.; Zeffiro, T.A.; Davidson, R.J.; Quevedo, K. Self-compassion and dorsolateral prefrontal cortex activity during sad self-face recognition in depressed adolescents. Psychol. Med. 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Quevedo, K.; Harms, M.; Sauder, M.; Scott, H.; Mohamed, S.; Thomas, K.M.; Schallmo, M.-P.; Smyda, G. The neurobiology of self face recognition among depressed adolescents. J. Affect. Disord. 2018, 229, 22–31. [Google Scholar] [CrossRef]
- Quevedo, K.; Liu, G.; Teoh, J.Y.; Ghosh, S.; Zeffiro, T.; Ahrweiler, N.; Zhang, N.; Wedan, R.; Oh, S.; Guercio, G.; et al. Neurofeedback and neuroplasticity of visual self-processing in depressed and healthy adolescents: A preliminary study. Dev. Cogn. Neurosci. 2019, 40, 100707. [Google Scholar] [CrossRef]
- Quevedo, K.; Ng, R.; Scott, H.; Martin, J.; Smyda, G.; Keener, M.; Oppenheimer, C.W. The neurobiology of self-face recognition in depressed adolescents with low or high suicidality. J. Abnorm. Psychol. 2016, 125, 1185–1200. [Google Scholar] [CrossRef]
- Quevedo, K.; Teoh, J.Y.; Engstrom, M.; Wedan, R.; Santana-Gonzalez, C.; Zewde, B.; Porter, D.; Kadosh, K.C. Amygdala Circuitry During Neurofeedback Training and Symptoms’ Change in Adolescents With Varying Depression. Front. Behav. Neurosci. 2020, 14, 110. [Google Scholar] [CrossRef]
- Hirot, F.; Lesage, M.; Pedron, L.; Meyer, I.; Thomas, P.; Cottencin, O.; Guardia, D. Impaired processing of self-face recognition in anorexia nervosa. Eat. Weight Disord. Stud. Anorex. Bulim. Obes. 2016, 21, 31–40. [Google Scholar] [CrossRef]
- Heinisch, C.; Wiens, S.; Grundl, M.; Juckel, G.; Brüne, M. Self-face recognition in schizophrenia is related to insight. Eur. Arch. Psychiatry Clin. Neurosci. 2013, 263, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Barone, P.; Corradi, G.; Gomila, A. Infants’ performance in spontaneous-response false belief tasks: A review and meta-analysis. Infant Behav. Dev. 2019, 57, 101350. [Google Scholar] [CrossRef] [PubMed]
- Frank, C.K. Reviving pragmatic theory of theory of mind. AIMS Neurosci. 2018, 5, 116–131. [Google Scholar] [CrossRef]
- Tomasello, M. How children come to understand false beliefs: A shared intentionality account. Proc. Natl. Acad. Sci. USA 2018, 115, 8491–8498. [Google Scholar] [CrossRef]
- Krych-Appelbaum, M.; Law, J.B.; Jones, D.; Barnacz, A.; Johnson, A.; Keenan, J.P. “I think I know what you mean”: The role of theory of mind in collaborative communication. Interact. Stud. 2007, 8, 267–280. [Google Scholar] [CrossRef]
- Feinberg, T. Altered Egos: How the Brain Creates the Self; Oxford University Press: Oxford, UK, 2002. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).