Venlafaxine Chiral Separation by Capillary Electrophoresis Using Cyclodextrin Derivatives as Chiral Selector and Experimental Design Method Optimization
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Instrumentation
2.3. Electrophoretic Procedure
2.4. Pharmaceutical Sample Preparation
3. Results
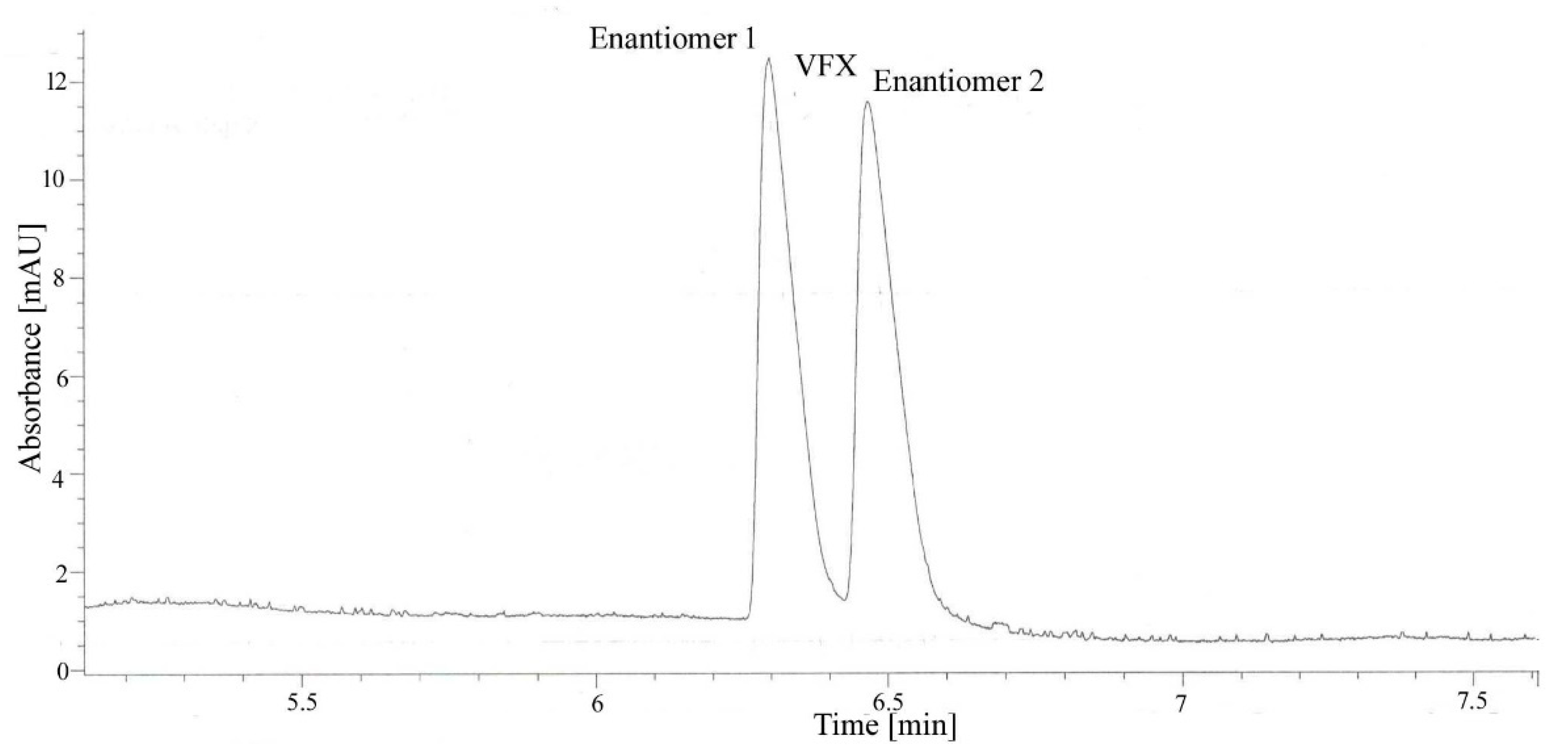
3.1. Preliminary Analysis
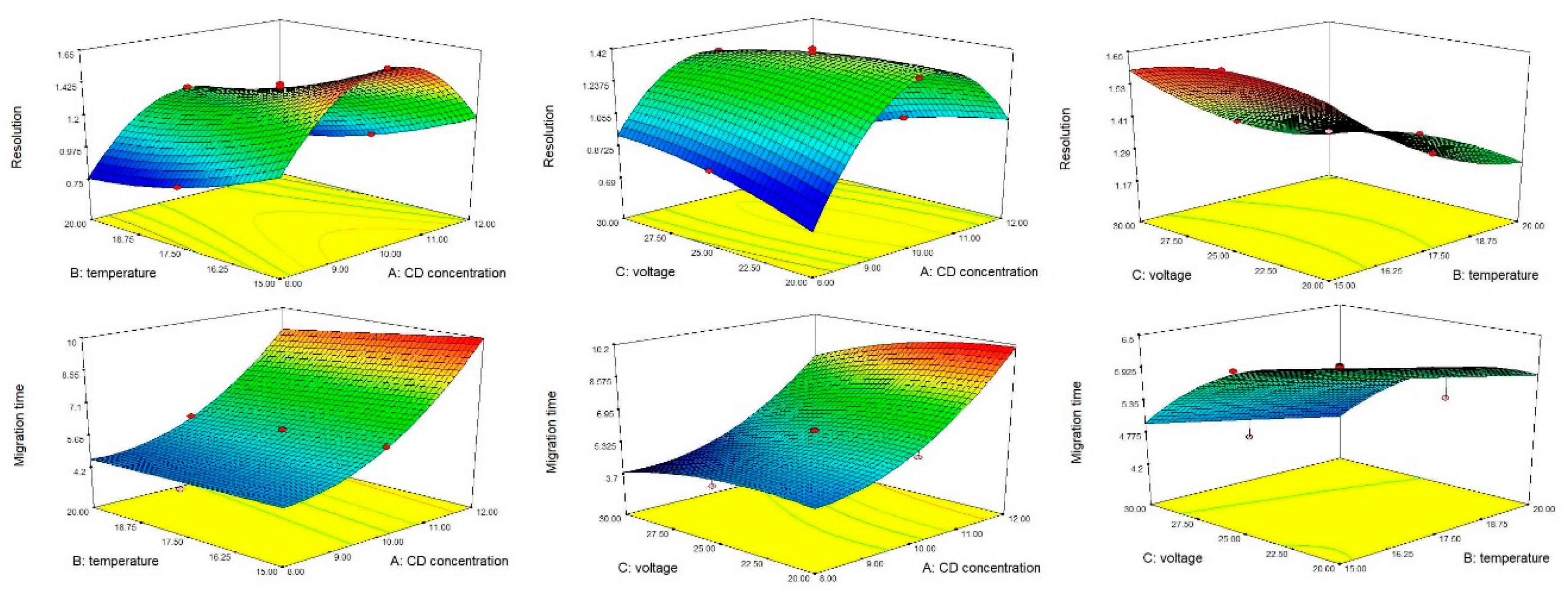
3.2. Method Optimization
3.3. Analytical Performance
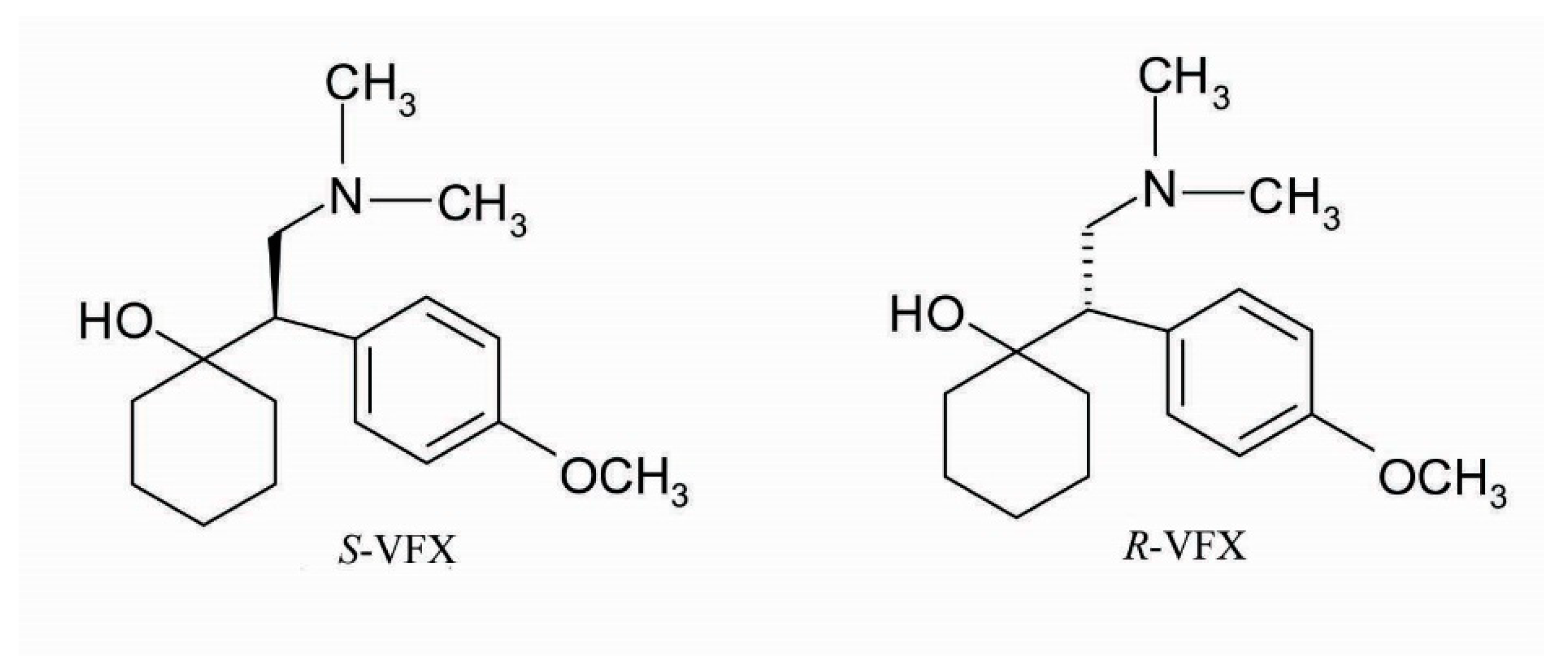
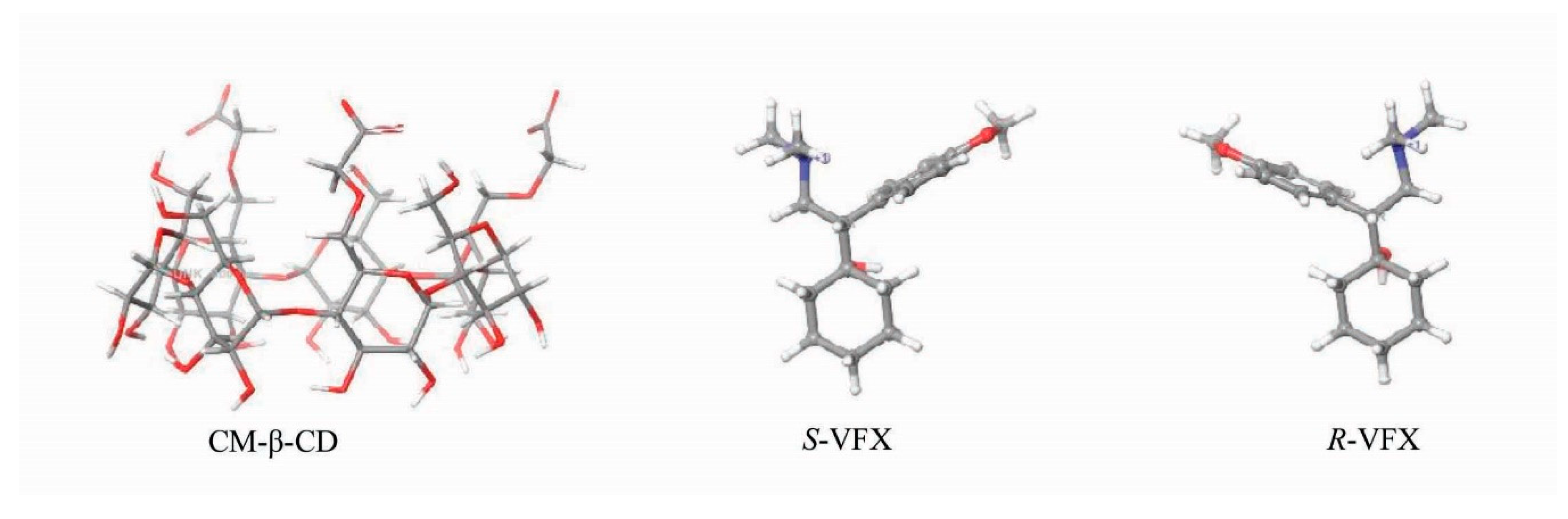
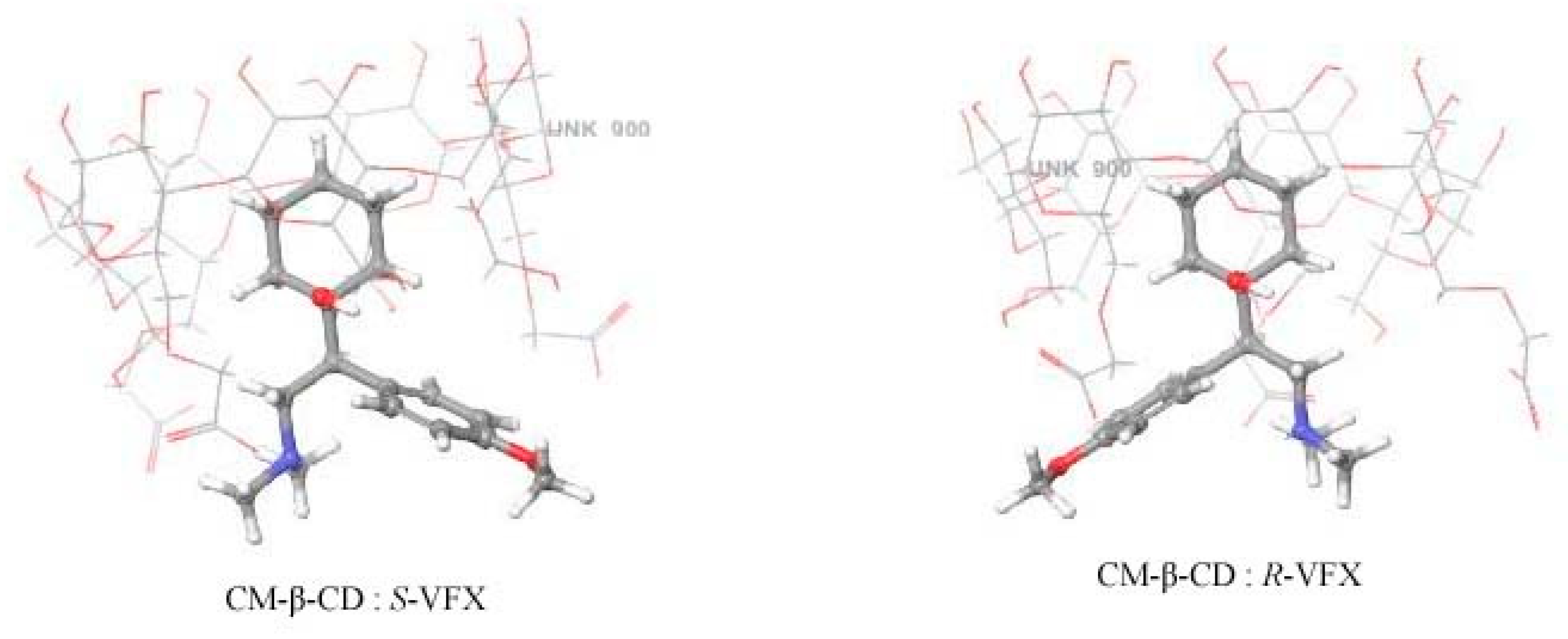
3.4. Molecular Modelling of VFX-CD Complexes
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Sekhon, B.S. Exploiting the power of stereochemistry in drugs: An overview of racemic and enantiopure drugs. J. Mod. Med. Chem. 2013, 1, 10–36. [Google Scholar] [CrossRef]
- Mehvar, R.; Brocks, D.R. Stereospecific pharmacokinetics and pharmacodynamics of beta-adrenergic blockers in humans. J. Pharm. Pharmaceut. Sci. 2001, 4, 185–201. [Google Scholar]
- Pai, V.; Pai, N. Recent advances in chirally pure proton pump inhibitors. J. Indian Med. Assoc. 2007, 105, 469–470. [Google Scholar]
- Hancu, G.; Câmpian, C.; Rusu, A.; Mircia, E.; Kelemen, H. Simultaneous determination of loratadine, desloratadine and cetirizine by capillary zone electrophoresis. Adv. Pharm. Bull. 2014, 4, 161–165. [Google Scholar]
- Budău, M.; Hancu, G.; Rusu, A.; Cârcu-dobrin, M.; Muntean, D.L. Chirality of modern antidepressants: An overview. Adv. Pharm. Bull. 2017, 7, 495–500. [Google Scholar] [CrossRef]
- Holliday, S.M.; Benfield, P. Venlafaxine. Drugs 1995, 49, 280–294. [Google Scholar] [CrossRef]
- Eap, C.B.; Lessard, E.; Baumann, P.; Brawand-AmeY, M.; Yessine, M.A.; O’Hara, G.; Turgeon, J. Role of CYP2D6 in the stereoselective disposition of venlafaxine in humans. Pharm. Genom. 2003, 13, 39–47. [Google Scholar]
- Magalhães, P.; Alves, G.; Llerena, A.; Falcão, A. Venlafaxine pharmacokinetics focused on drug metabolism and potential biomarkers. Drug Metab. Drug Interact. 2014, 29, 129–141. [Google Scholar] [CrossRef]
- Bernardo-Bermejo, S.; Sánchez-López, E.; Castro-Puyana, M.; Marina, M.L. Chiral Capillary Electrophoresis. TrAC Trends Anal. Chem. 2020, 124, 115807. [Google Scholar] [CrossRef]
- Řezanka, P.; Navrátilová, K.; Řezanka, M.; Král, V.; Sýkora, D. Application of cyclodextrins in chiral capillary electrophoresis. Electrophoresis 2014, 35, 2701–2721. [Google Scholar] [CrossRef]
- Rudaz, S.; Veuthy, J.L.; Desiderio, C.; Fanali, S. Enantioseparation of venlafaxine and O-desmethylvenlafaxine by capillary electrophoresis with mixed cyclodextrins. Chromatographia 1999, 50, 369–372. [Google Scholar] [CrossRef]
- Rudaz, S.; Stella, C.; Balant-Gorgia, A.E.; Fanali, S.; Veuthy, J.L. Simultaneous stereoselective analysis of venlafaxine and O-desmethylvenlafaxine enantiomers in clinical samples by capillary electrophoresis using charged cyclodextrins. J. Pharm. Biomed. Anal. 2000, 23, 107–115. [Google Scholar] [CrossRef]
- Fanali, S.; Rudaz, S.; Veuthey, J.L.; Desiderio, C. Use of vancomycin silica stationary phase in packed capillary electrochromatography II. Enantiomer separation of venlafaxine and O-desmethylvenlafaxine in human plasma. J. Chromatogr. A 2001, 919, 195–203. [Google Scholar] [CrossRef]
- Liu, Y.; Jann, M.; Vandenberg, C.; Eap, C.B.; Shamsi, S.A. Development of an enantioselective assay for simultaneous separation of venlafaxine and O-desmethylvenlafaxine by micellar electrokinetic chromatography-tandem mass spectrometry: Application to the analysis of drug–drug interaction. J. Chromatogr. A 2015, 1420, 119–128. [Google Scholar] [CrossRef]
- Elbashir, A.A.; Aboul-Enein, H.Y. Capillary electrophoresis and molecular modeling as a complementary technique for chiral recognition mechanism. Crit. Rev. Anal. Chem. 2013, 43, 131–137. [Google Scholar] [CrossRef]
- Alexander, J.M.; Clark, J.L.; Brett, T.J.; Stezowski, J.J. Chiral discrimination in cyclodextrin complexes of amino acid derivatives: β-cyclodextrin/N-acetyl-L-phenylalanine and N-acetyl-D-phenylalanine complexes. Proc. Natl. Acad. Sci. USA 2002, 99, 5115–5120. [Google Scholar] [CrossRef]
- Van Eupen, J.T.H.; Elffrink, W.W.J.; Keltjens, R.; Bennema, P.; De Gelder, R.; Smits, J.M.M.; van Eck, E.R.H.; Kentgens, A.P.M.; Deij, M.A.; Vlieg, E. Polymorphism and migratory chiral resolution of the free base of venlafaxine. A remarkable topotactical solid state transition from a racemate to a racemic conglomerate. Cryst. Growth Des. 2008, 8, 71–79. [Google Scholar] [CrossRef]
- Groom, C.R.; Bruno, I.J.; Lightfoot, M.P.; Ward, S.C. The Cambridge structural database. Acta Crystallogr. Sect. B Struct. Sci. Cryst. Eng. Mater. 2016, 72, 171–179. [Google Scholar] [CrossRef]
- Fejős, I.; Varga, E.; Benkovics, G.; Malanga, M.; Sohajda, T.; Szemán, J.; Béni, S. Characterization of a single-isomer carboxymethyl-beta-cyclodextrin in chiral capillary electrophoresis. Electrophoresis 2017, 38, 1869–1877. [Google Scholar] [CrossRef]
- Altria, K.D.; Clark, B.J.; Filbey, S.D.; Kelly, M.A.; Rudd, D.R. Application of chemometric experimental designs in capillary electrophoresis: A review. Electrophoresis 1995, 16, 2143–2148. [Google Scholar] [CrossRef]
- Fukuda, I.M.; Pinto, C.F.F.; Moreira, C.D.S.; Saviano, A.M.; Lourenço, F.R. Design of Experiments (DoE) applied to pharmaceutical and analytical Quality by Design (QbD). Braz. J. Pharm. Sci. 2018, 54, e01006. [Google Scholar] [CrossRef]
| BGE Concentration [mM] | CD Concentration [mM] | Voltage (kV) | Temperature (°C) | Injection Pressure (mbar/s) | R | t1 (min) | t2 (min) |
|---|---|---|---|---|---|---|---|
| 25 | 5 | 25 | 20 | 50 | 0.59 | 3.22 | 3.27 |
| 50 | 5 | 25 | 20 | 50 | 0.52 | 4.45 | 4.51 |
| 100 | 5 | 25 | 20 | 50 | 0.50 | 6.42 | 6.49 |
| 25 | 10 | 25 | 20 | 50 | 1.03 | 4.24 | 4.34 |
| 25 | 15 | 25 | 20 | 50 | 0.55 | 7.13 | 7.19 |
| 25 | 10 | 20 | 20 | 50 | 0.98 | 6.28 | 6.42 |
| 25 | 10 | 30 | 20 | 50 | 0.87 | 2.78 | 2.84 |
| 25 | 10 | 15 | 15 | 50 | 1.12 | 4.59 | 4.71 |
| 25 | 10 | 25 | 25 | 50 | 0.92 | 3.91 | 3.99 |
| 25 | 10 | 25 | 15 | 40 | 1.20 | 4.22 | 4.31 |
| 25 | 10 | 25 | 15 | 30 | 1.31 | 4.21 | 4.29 |
| Run | Factor A | Factor B | Factor C | Response 1 | Response 2 |
|---|---|---|---|---|---|
| 1 | 10.00 | 17.50 | 30.00 | 1.32 | 4.29 |
| 2 | 8.00 | 17.50 | 25.00 | 0.87 | 4.35 |
| 3 | 10.00 | 17.50 | 25.00 | 1.36 | 5.97 |
| 4 | 10.00 | 17.50 | 25.00 | 1.36 | 5.94 |
| 5 | 12.00 | 20.00 | 20.00 | 0.90 | 9.75 |
| 6 | 8.00 | 20.00 | 30.00 | 0.82 | 3.84 |
| 7 | 10.00 | 20.00 | 25.00 | 1.27 | 5.58 |
| 8 | 10.00 | 15.00 | 25.00 | 1.65 | 6.20 |
| 9 | 12.00 | 15.00 | 30.00 | 0.98 | 8.65 |
| 10 | 10.00 | 17.50 | 20.00 | 1.36 | 5.77 |
| 11 | 8.00 | 15.00 | 20.00 | 0.93 | 4.91 |
| 12 | 12.00 | 17.50 | 25.00 | 0.92 | 9.15 |
| 13 | 10.00 | 17.50 | 25.00 | 1.40 | 5.95 |
| 14 | 10.00 | 17.50 | 25.00 | 1.40 | 5.95 |
| 15 | 10.00 | 17.50 | 25.00 | 1.42 | 5.97 |
| Enantiomer | Time (min) | Area | Height | Symmetry | Nr Theoretical Plates | Rs | α |
|---|---|---|---|---|---|---|---|
| 1 | 5.77 | 48.77 | 9.60 | 0.28 | 36790 | - | - |
| 2 | 5.95 | 52.61 | 9.24 | 0.27 | 31879 | 1.64 | 1.02 |
| VFX | Enantiomer 1 | Enantiomer 2 | |
|---|---|---|---|
| Precision | |||
| Intra-day precision (sample concentration = 0.5 mg/mL, n = 6) | RSD%, migration time | 0.16 | 0.17 |
| RSD%, peak area | 1.33 | 1.30 | |
| Inter-day precision (sample concentration = 0.5 mg/mL, n = 18) | RSD%, migration time | 0.29 | 0.30 |
| RSD%, peak area | 1.55 | 1.71 | |
| Accuracy (recovery, %) | |||
| 0.5 mg/mL (n = 6) | 101.06 | 99.42 | |
| 0.25 mg/mL (n = 6) | 101.35 | 101.96 | |
| Linearity | |||
| Regression equation (0.1–2 mg/mL) | y = 68.837x+ 2.564 | y = 70.916x+4.498 | |
| Coefficient of correlation | 0.998 | 0.997 | |
| LOD (mg/mL) | 0.07 | 0.06 | |
| LOQ (mg/mL) | 0.21 | 0.18 | |
| Pharmaceutical Product | Declared Enantiomers Quantity (mg) | Found Enantiomer Quantity (mg) ± SD (n = 3) | ||
|---|---|---|---|---|
| Enantiomer 1 | Enantiomer 2 | Enantiomer 1 | Enantiomer 2 | |
| Alventa capsule (75 mg VFX) | 37.5 | 37.5 | 36.65 +/− 0.56 | 36.63 +/− 0.29 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milan, A.; Hancu, G.; Lupu, D.; Budău, M.; Garaj, V.; Kelemen, H. Venlafaxine Chiral Separation by Capillary Electrophoresis Using Cyclodextrin Derivatives as Chiral Selector and Experimental Design Method Optimization. Symmetry 2020, 12, 849. https://doi.org/10.3390/sym12050849
Milan A, Hancu G, Lupu D, Budău M, Garaj V, Kelemen H. Venlafaxine Chiral Separation by Capillary Electrophoresis Using Cyclodextrin Derivatives as Chiral Selector and Experimental Design Method Optimization. Symmetry. 2020; 12(5):849. https://doi.org/10.3390/sym12050849
Chicago/Turabian StyleMilan, Andreea, Gabriel Hancu, Daniela Lupu, Monica Budău, Vladimir Garaj, and Hajnal Kelemen. 2020. "Venlafaxine Chiral Separation by Capillary Electrophoresis Using Cyclodextrin Derivatives as Chiral Selector and Experimental Design Method Optimization" Symmetry 12, no. 5: 849. https://doi.org/10.3390/sym12050849
APA StyleMilan, A., Hancu, G., Lupu, D., Budău, M., Garaj, V., & Kelemen, H. (2020). Venlafaxine Chiral Separation by Capillary Electrophoresis Using Cyclodextrin Derivatives as Chiral Selector and Experimental Design Method Optimization. Symmetry, 12(5), 849. https://doi.org/10.3390/sym12050849








