Mathematical Modeling for Neuropathic Pain: Bayesian Linear Regression and Self-Organizing Maps Applied to Carpal Tunnel Syndrome
Abstract
1. Introduction
2. Methods
2.1. Participants
2.2. Pain and Related Disability Outcomes
2.3. Pressure and Thermal Pain Threshold Assessment
2.4. Pinch Tip Grip Force Assessment
2.5. Psychological Assessment
2.6. Data Overview and Preprocessing
2.7. Bayesian Linear Regression (BLR)
2.7.1. Method Overview
2.7.2. Bayesian vs. Frequentist Statistics
2.7.3. Bayesian Linear Models LR vs. Nonlinear Models
2.8. Self-Organizing Maps (SOM)
3. Results
3.1. Participants
3.2. Bayesian Linear Regression
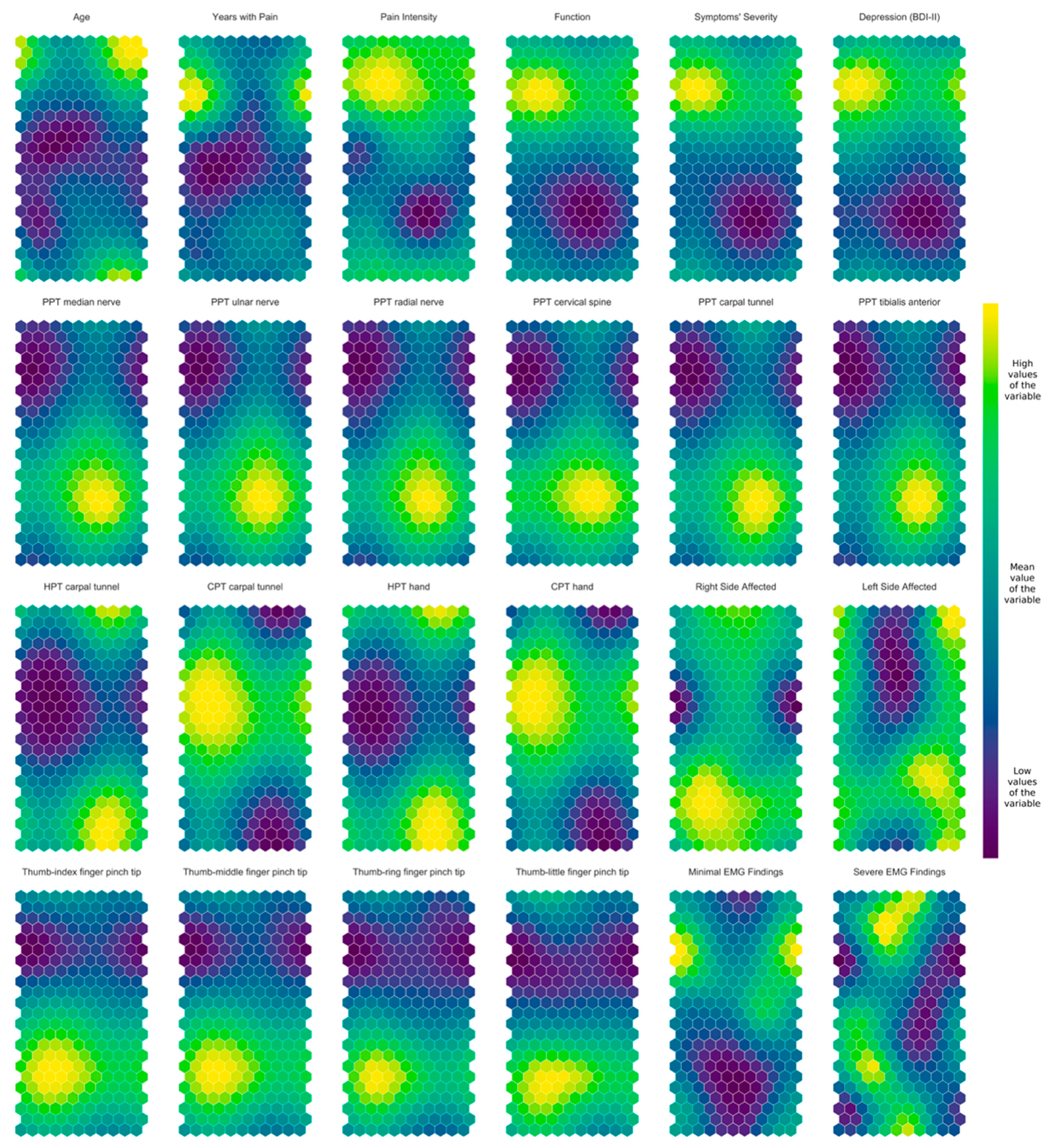
3.3. Self-Organizing Maps
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pourmemari, M.H.; Heliövaara, M.; Viikari-Juntura, E.; Shiri, R. Carpal tunnel release: Lifetime prevalence, annual incidence, and risk factors. Muscle Nerve 2018, 58, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Thiese, M.S.; Gerr, F.; Hegmann, K.T.; Harris-Adamson, C.; Dale, A.M.; Evanoff, B.; Eisen, E.A.; Kapellusch, J.; Garg, A.; Burt, S.; et al. Effects of varying case definition on carpal tunnel syndrome prevalence estimates in a pooled cohort. Arch. Phys. Med. Rehabil. 2014, 95, 2320–2326. [Google Scholar] [CrossRef] [PubMed]
- Epstein, S.; Sparer, E.H.; Tran, B.N.; Ruan, Q.Z.; Dennerlein, J.T.; Singhal, D.; Lee, B.T. Prevalence of Work-Related Musculoskeletal Disorders Among Surgeons and Interventionalists: A Systematic Review and Meta-analysis. JAMA Surg. 2018, 153, e174947. [Google Scholar] [CrossRef] [PubMed]
- Dale, A.M.; Harris-Adamson, C.; Rempel, D.; Gerr, F.; Hegmann, K.; Silverstein, B.; Burt, S.; Garg, A.; Kapellusch, J.; Merlino, L.; et al. Prevalence and incidence of carpal tunnel syndrome in US working populations: Pooled analysis of six prospective studies. Scand. J. Work Environ. Health 2013, 39, 495–505. [Google Scholar] [CrossRef]
- Foley, M.; Silverstein, B. The long-term burden of work-related carpal tunnel syndrome relative to upper-extremity fractures and dermatitis in Washington State. Am. J. Ind. Med. 2015, 58, 1255–1269. [Google Scholar] [CrossRef]
- Genova, A.; Dix, O.; Saefan, A.; Thakur, M.; Hassan, A. Carpal tunnel syndrome: A review of literature. Cureus 2020, 12, e7333. [Google Scholar] [CrossRef]
- Fernández-de-las-Peñas, C.; Plaza-Manzano, G. Carpal tunnel syndrome: Just a peripheral neuropathy? Pain Manag. 2018, 8, 209–216. [Google Scholar] [CrossRef]
- Nunez, F.; Vranceanu, A.M.; Ring, D. Determinants of pain in patients with carpal tunnel syndrome. Clin. Orthop. Relat. Res. 2010, 468, 3328–3332. [Google Scholar] [CrossRef]
- Fernández-Muñoz, J.J.; Palacios-Ceña, M.; Cigarán-Méndez, M.; Ortega-Santiago, R.; de-la-Llave-Rincón, A.I.; Salom-Moreno, J.; Fernández-de-las-Peñas, C. Pain is associated to clinical, psychological, physical, and neurophysiological variables in women with carpal tunnel syndrome. Clin. J. Pain 2016, 32, 122–129. [Google Scholar] [CrossRef]
- Fernández-de-las-Peñas, C.; Cleland, J.A.; Plaza-Manzano, G.; Ortega-Santiago, R.; de-la-Llave-Rincón, A.I.; Martínez-Perez, A.; Arroyo-Morales, M. Clinical, physical, and neurophysiological impairments associated with decreased function in women with carpal tunnel syndrome. J. Orthop. Sports Phys. Ther. 2013, 43, 641–649. [Google Scholar] [CrossRef]
- Permai, S.D.; Tanty, H. Linear regression model using bayesian approach for energy performance of residential building. Procedia Comput. Sci. 2018, 135, 671–677. [Google Scholar] [CrossRef]
- Dunson, D.B.; Neelon, B. Bayesian inference on order-constrained parameters in generalized linear models. Biometrics 2003, 59, 286–295. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Taylor, J.M.G.; Vokonas, P.S.; Park, S.K.; Mukherjee, B. Improving estimation and prediction in linear regression incorporating external information from an established reduced model. Stat. Med. 2018, 37, 1515–1530. [Google Scholar] [CrossRef] [PubMed]
- Kohonen, T. Self-Organizing Maps, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2001. [Google Scholar]
- Riesenhuber, M.; Bauer, H.U.; Brockmann, D.; Geisel, T. Breaking rotational symmetry in a self-organizing map model for orientation map development. Neural Comput. 1998, 10, 717–730. [Google Scholar] [CrossRef]
- Aoki, T.; Ota, K.; Kurata, K.; Aoyagi, T. Ordering process of self-organizing maps improved by asymmetric neighborhood function. Cogn. Neurodyn. 2009, 3, 9–15. [Google Scholar] [CrossRef]
- Caldas, R.; Rativa, D.; Lima Neto, F. Clustering of Self-Organizing Maps as a means to support gait kinematics analysis and symmetry evaluation. Med. Eng. Phys. 2018, 62, 46–52. [Google Scholar] [CrossRef]
- Jablecki, C.K.; Andary, M.T.; Floeter, M.K.; Miller, R.G.; Quartly, C.A.; Vennix, M.J.; Wilson, J.R. Practice parameter: Electro-diagnostic studies in carpal tunnel syndrome. Report of the American Association of Electrodiagnostic Medicine, American Academy of Neurology, American Academy of Physical Medicine and Rehabilitation. Neurology 2002, 58, 1589–1592. [Google Scholar] [CrossRef]
- Jensen, M.P.; Turbner, J.A.; Romano, J.M.; Fisher, L. Comparative reliability and validity of chronic pain intensity measures. Pain 1999, 83, 157–162. [Google Scholar] [CrossRef]
- Levine, D.W.; Simmons, B.; Koris, M.; Daltroy, L.; Hohl, G.; Fossel, A.; Katz, J. A self-administered questionnaire for the assessment of severity of symptoms and functional status in carpal tunnel syndrome. J. Bone Jt. Surg. Am. 1993, 75, 1585–1592. [Google Scholar] [CrossRef]
- Carvalho-Leite, J.; Jerosch-Herold, C.; Song, F. A systematic review of the psychometric properties of the Boston Carpal Tunnel Questionnaire. BMC Musculoskelet. Disord. 2006, 7, 78. [Google Scholar]
- Rosales, R.S.; Benseny, E.; Díez de la Lastra-Bosch, I. Evaluation of the Spanish version of the DASH and carpal tunnel syndrome health-related quality of life instruments: Cross cultural adaptation process and reliability. J. Hand Surg. 2002, 27, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Fernández-de-las-Peñas, C.; de la Llave-Rincón, A.I.; Fernández-Carnero, J.; Cuadrado, M.L.; Arendt-Nielsen, L.; Pareja, J.A. Bilateral widespread mechanical pain sensitivity in carpal tunnel syndrome: Evidence of central processing in unilateral neuropathy. Brain 2009, 132, 1472–1479. [Google Scholar] [CrossRef] [PubMed]
- De-la-Llave-Rincón, A.I.; Fernández-de-las-Peñas, C.; Fernández-Carnero, J.; Padua, L.; Arendt-Nielsen, L.; Pareja, J.A. Bilateral hand/wrist heat and cold hyperalgesia, but not hypoesthesia, in unilateral carpal tunnel syndrome. Exp. Brain Res. 2009, 198, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.H.; Kilgour, R.D.; Comtois, A.S. Test-retest reliability of pressure pain threshold measurements of the upper limb and torso in young healthy women. J. Pain 2007, 8, 650–656. [Google Scholar] [CrossRef]
- Moloney, N.A.; Hall, T.M.; O’Sullivan, T.C.; Doody, C.M. Reliability of thermal quantitative sensory testing of the hand in a cohort of young, healthy adults. Muscle Nerve 2011, 44, 547–552. [Google Scholar] [CrossRef]
- Fernández-de-las-Peñas, C.; Pérez-de-Heredia-Torres, M.; Martínez-Piédrola, R.; De-la-Llave-Rincón, A.I.; Cleland, J.A. Bilateral deficits in fine motor control and pinch grip force in patients with unilateral carpal tunnel syndrome. Exp. Brain Res. 2009, 194, 29–37. [Google Scholar] [CrossRef]
- Schreuders, T.; Roebroeck, M.; Goumans, J.; Van Nieuwenhuijzen, J.; Stijnen, T. Measurement error in grip and pinch force measurements in patients with hand injuries. Phys. Ther. 2003, 83, 806–815. [Google Scholar] [CrossRef]
- Beck, A.T.; Steer, R.A.; Brown, G.K. Beck Depression Inventory, 2nd ed.; The Psychological Corporation: San Antonio, TX, USA, 1996. [Google Scholar]
- Wang, Y.P.; Gorenstein, C. Assessment of depression in medical patients: A systematic review of the utility of the Beck Depression Inventory-II. Clinics 2013, 68, 1274–1287. [Google Scholar] [CrossRef]
- Salvatier, J.; Wiecki, T.V.; Fonnesbeck, C. Probabilistic programming in Python using PyMC3. Peer J. Comput. Sci. 2016, 2, e55. [Google Scholar] [CrossRef]
- Hoffman, M.D.; Gelman, A. The no-U-turn sampler: Adaptively setting path lengths in Hamiltonian Monte Carlo. J. Mach. Learn. Res. 2014, 15, 1593–1623. [Google Scholar]
- Chen, T.; Guestrin, C. Xgboost: A scalable tree boosting system. In Proceedings of the 22nd ACM Sigkdd International Conference on Knowledge Discovery and Data Mining, Association for Computing Machinery, San Francisco, CA, USA, 13–17 August 2016; pp. 785–794. [Google Scholar]
- Kohonen, T. The Self-Organizing Map. Proc. IEEE 1990, 78, 1464–1480. [Google Scholar] [CrossRef]
- Comitani, F. SimpSOM, a lightweight Python library for Kohonen Self Organising Maps. Available online: https://zenodo.org/record/2621560 (accessed on 29 August 2020).
- Seventer, R.V.; Serpell, M.; Bach, F.W.; Morlion, B.; Zlateva, G.; Bushmakin, A.G.; Cappelleri, J.C.; Nimour, M. Relationships between changes in pain severity and other patient-reported outcomes: An analysis in patients with posttraumatic peripheral neuropathic pain. Health Qual. Life Outcomes 2011, 9, 17. [Google Scholar] [CrossRef] [PubMed]
- McWilliams, L.A.; Cox, B.J.; Enns, M.W. Mood and anxiety disorders associated with chronic pain: An examination in a nationally representative sample. Pain 2003, 106, 127–133. [Google Scholar] [CrossRef]

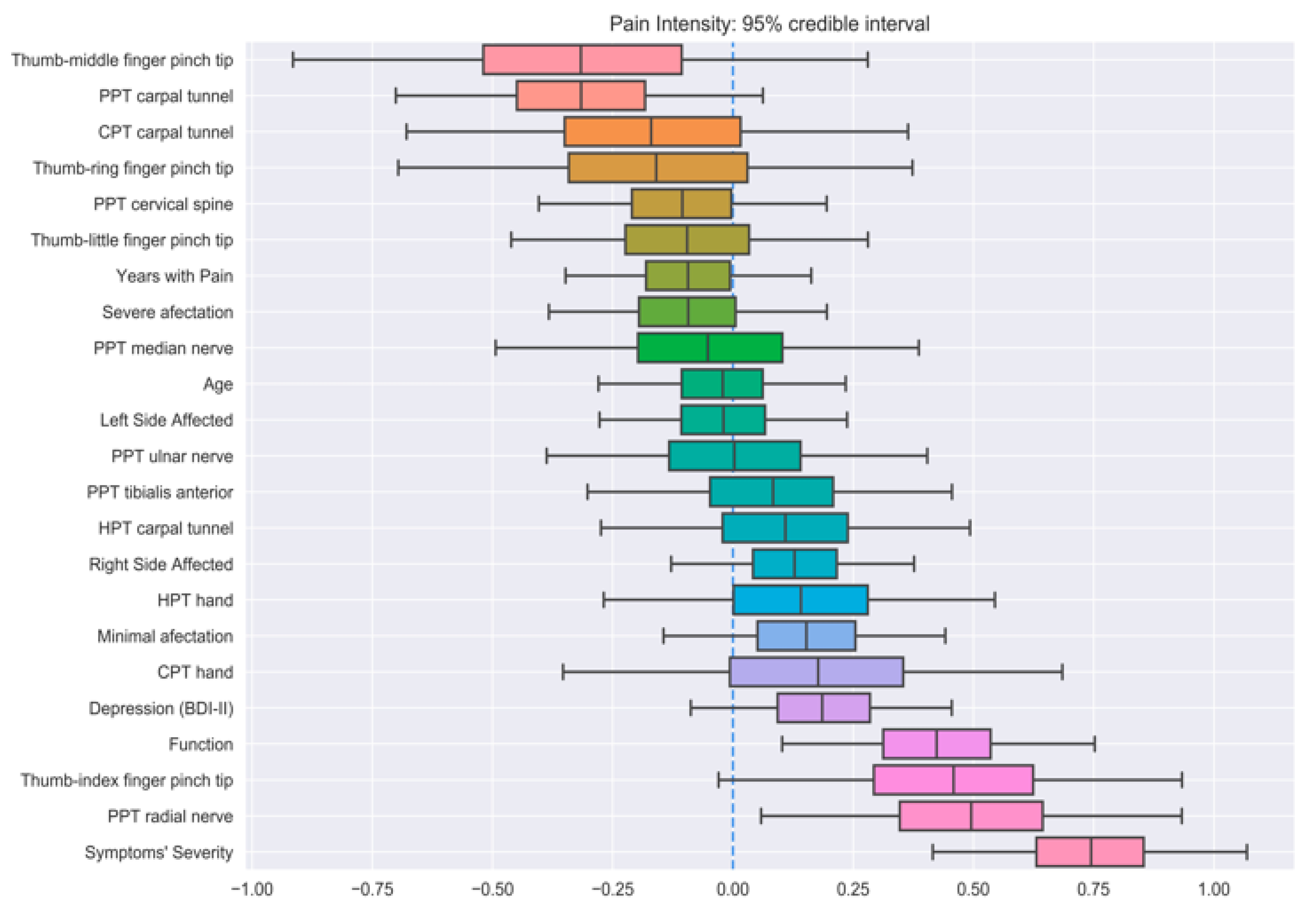
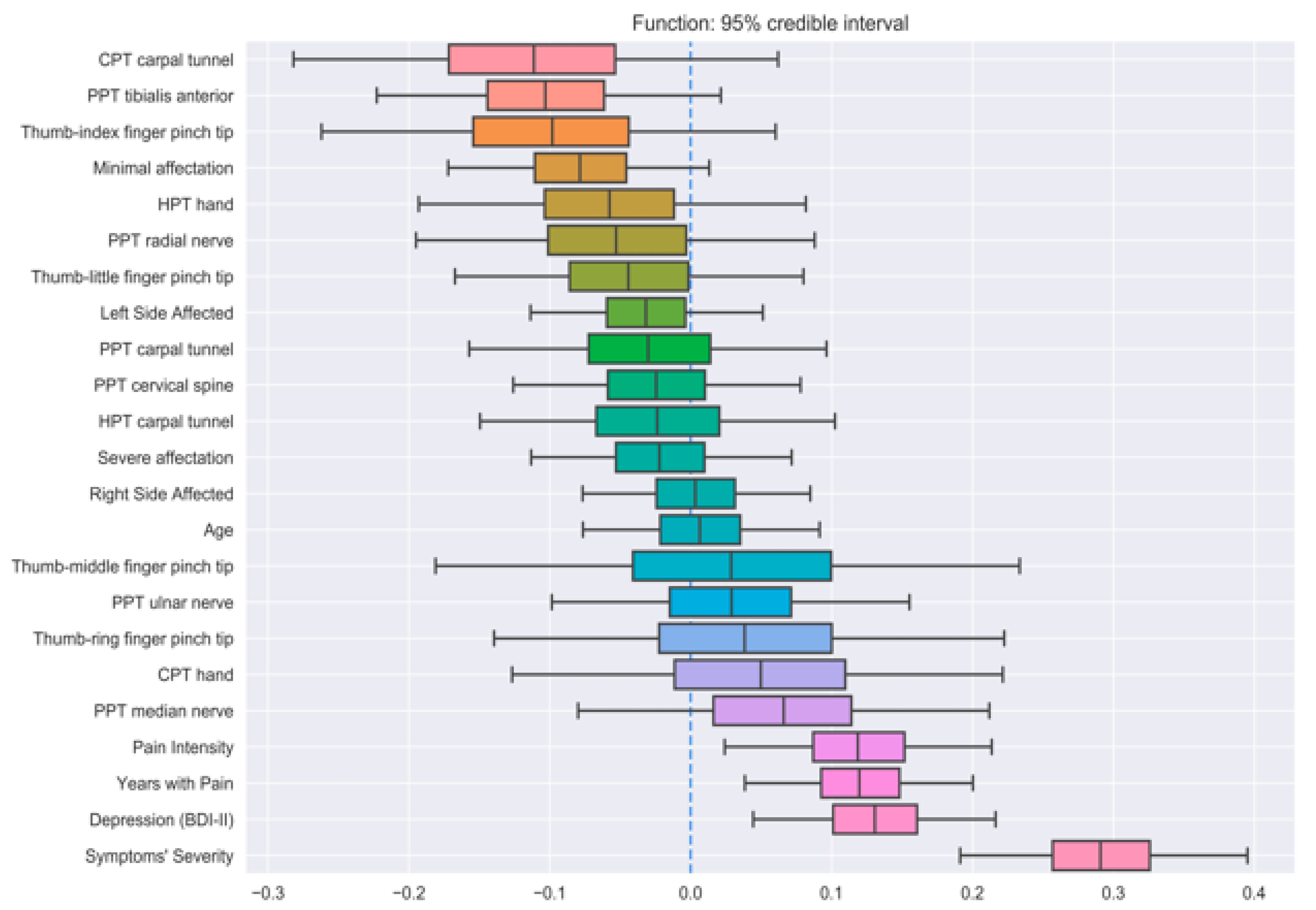
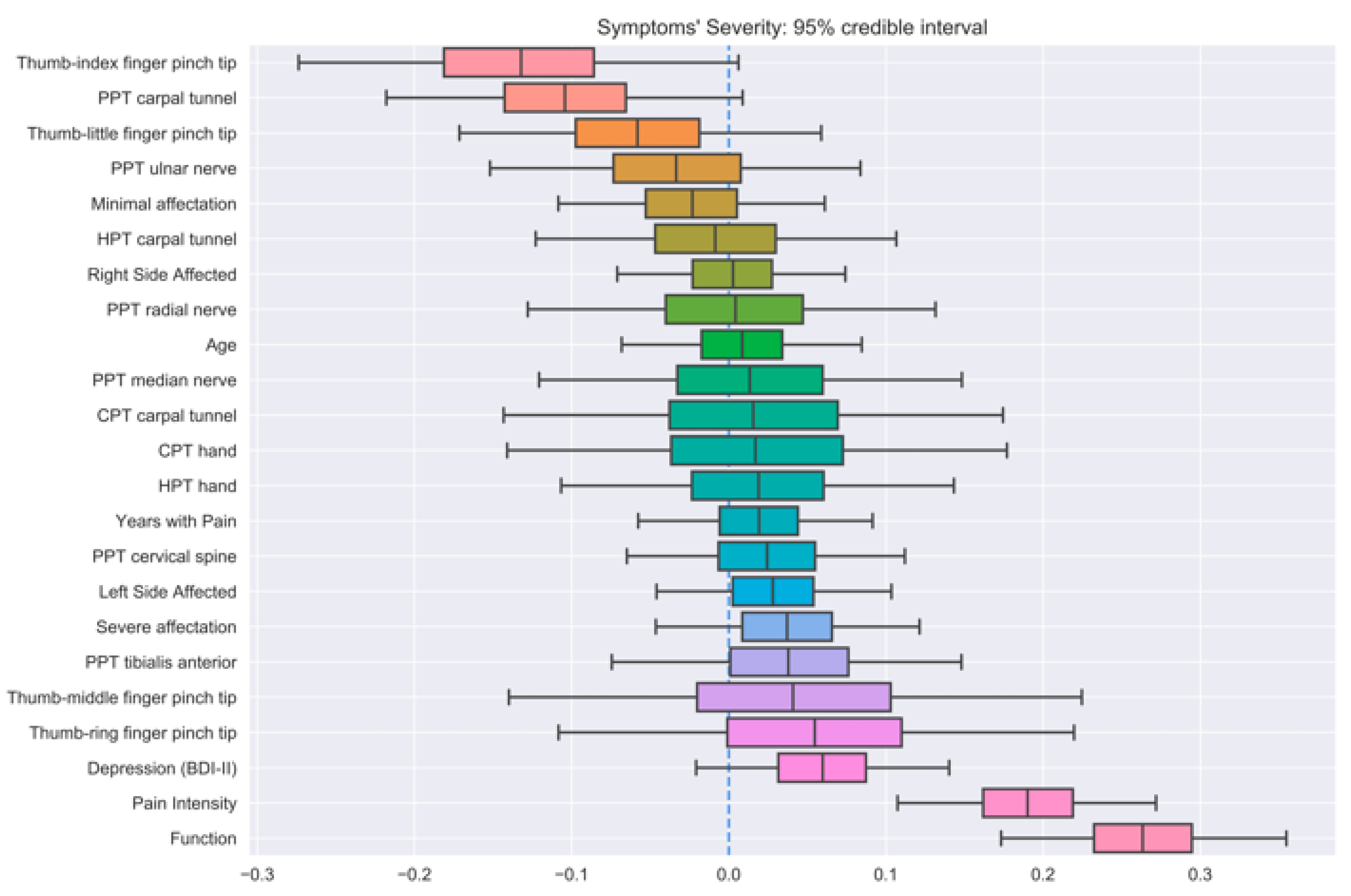
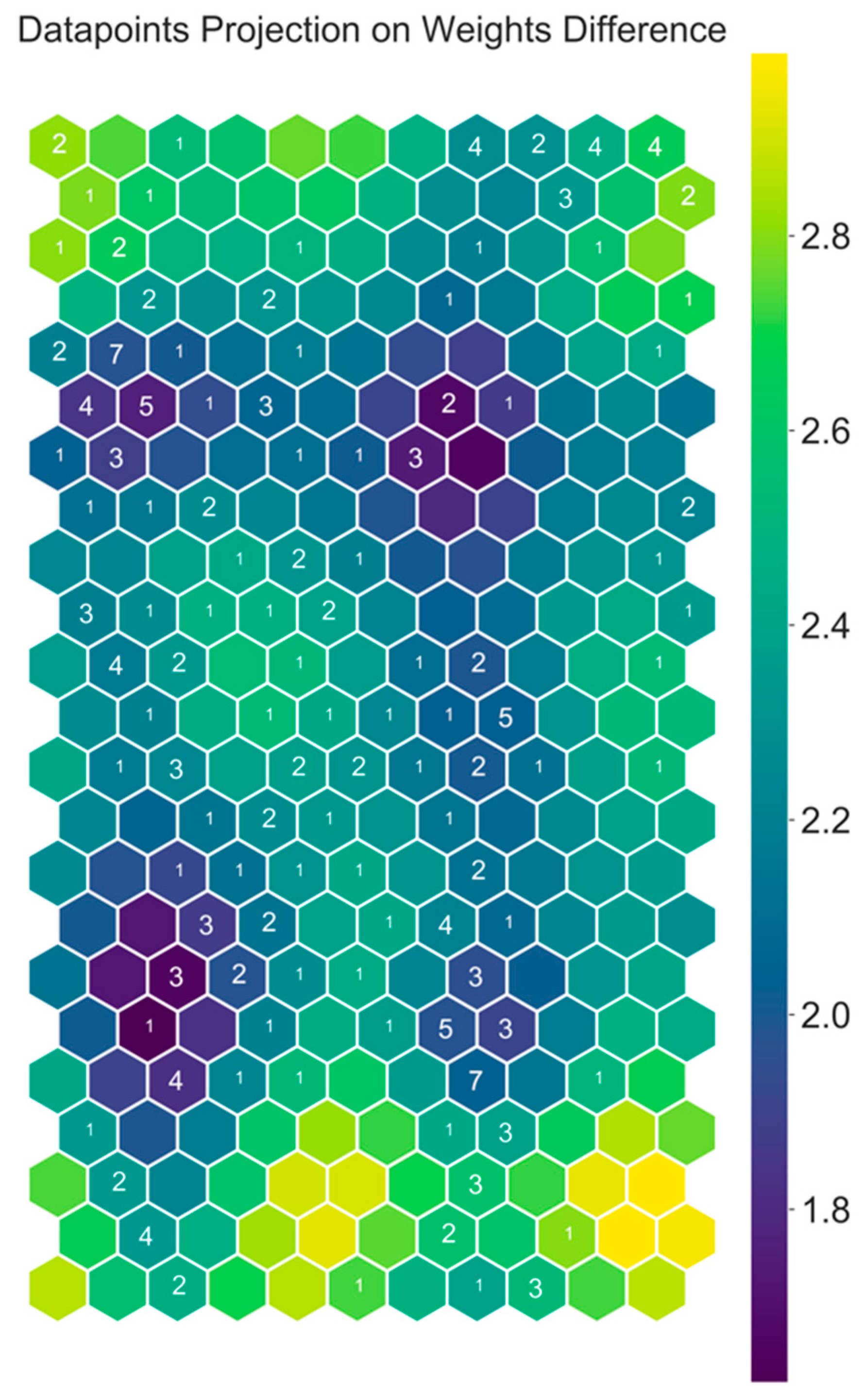
| Output Feature | Mean Train Correlation | Mean CV Correlation | ||||
|---|---|---|---|---|---|---|
| BLR | xgboost | NN | BLR | xgboost | NN | |
| Function | 0.724 | 0.981 | 0.697 | 0.596 | 0.604 | 0.607 |
| Symptoms’ Severity | 0.723 | 0.980 | 0.694 | 0.628 | 0.633 | 0.632 |
| Pain Intensity | 0.622 | 0.978 | 0.764 | 0.457 | 0.597 | 0.499 |
| Mean | SD | Min | Max | |
|---|---|---|---|---|
| Age | 45.5 | 9.1 | 21.0 | 64.00 |
| Years with pain | 3.5 | 3.0 | 0.5 | 17.00 |
| Right side affected * | 0.9 | 0.3 | 0.00 | 1.00 |
| Left side affected * | 0.75 | 0.45 | 0.00 | 1.00 |
| EMG minimal affectation # | 0.3 | 0.45 | 0.00 | 1.00 |
| EMG severe affectation # | 0.4 | 0.5 | 0.00 | 1.00 |
| Pain intensity | 5.8 | 2.1 | 0.00 | 10.00 |
| Symptom severity | 2.75 | 0.7 | 1.25 | 5.00 |
| Function | 2.4 | 0.75 | 1.0 | 4.62 |
| Depression (BDI-II) | 4.6 | 2.9 | 0.0 | 15.0 |
| CPT carpal tunnel | 19.4 | 6.7 | 5.00 | 30.2 |
| CPT hand | 19.2 | 6.45 | 5.00 | 29.75 |
| HPT carpal tunnel | 39.9 | 2.6 | 35.2 | 48.45 |
| HPT hand | 40.1 | 2.85 | 32.1 | 48.2 |
| PPT median nerve | 192.55 | 50.7 | 57.65 | 365.5 |
| PPT ulnar nerve | 293.7 | 73.6 | 115.5 | 465.5 |
| PPT radial nerve | 225.25 | 61.9 | 109.5 | 433.5 |
| PPT cervical spine | 171.1 | 53.75 | 57.0 | 499.5 |
| PPT carpal tunnel | 346.05 | 95.4 | 130.5 | 731.0 |
| PPT tibialis anterior | 322.85 | 85.5 | 110.5 | 652.5 |
| Thumb–index finger pinch tip | 4.15 | 1.7 | 0.5 | 8.5 |
| Thumb–little finger pinch tip | 1.1 | 0.8 | 0.0 | 5.5 |
| Thumb–middle finger pinch tip | 4.0 | 1.9 | 0.0 | 9.5 |
| Thumb–ring finger pinch tip | 2.45 | 1.4 | 0.0 | 6.35 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pellicer-Valero, O.J.; Martín-Guerrero, J.D.; Cigarán-Méndez, M.I.; Écija-Gallardo, C.; Fernández-de-las-Peñas, C.; Navarro-Pardo, E. Mathematical Modeling for Neuropathic Pain: Bayesian Linear Regression and Self-Organizing Maps Applied to Carpal Tunnel Syndrome. Symmetry 2020, 12, 1581. https://doi.org/10.3390/sym12101581
Pellicer-Valero OJ, Martín-Guerrero JD, Cigarán-Méndez MI, Écija-Gallardo C, Fernández-de-las-Peñas C, Navarro-Pardo E. Mathematical Modeling for Neuropathic Pain: Bayesian Linear Regression and Self-Organizing Maps Applied to Carpal Tunnel Syndrome. Symmetry. 2020; 12(10):1581. https://doi.org/10.3390/sym12101581
Chicago/Turabian StylePellicer-Valero, Oscar J., José D. Martín-Guerrero, Margarita I. Cigarán-Méndez, Carmen Écija-Gallardo, César Fernández-de-las-Peñas, and Esperanza Navarro-Pardo. 2020. "Mathematical Modeling for Neuropathic Pain: Bayesian Linear Regression and Self-Organizing Maps Applied to Carpal Tunnel Syndrome" Symmetry 12, no. 10: 1581. https://doi.org/10.3390/sym12101581
APA StylePellicer-Valero, O. J., Martín-Guerrero, J. D., Cigarán-Méndez, M. I., Écija-Gallardo, C., Fernández-de-las-Peñas, C., & Navarro-Pardo, E. (2020). Mathematical Modeling for Neuropathic Pain: Bayesian Linear Regression and Self-Organizing Maps Applied to Carpal Tunnel Syndrome. Symmetry, 12(10), 1581. https://doi.org/10.3390/sym12101581








