CCR6 as a Potential Target for Therapeutic Antibodies for the Treatment of Inflammatory Diseases
Abstract
1. Introduction
2. Description of CCR6
2.1. Biochemical Characteristics and Structure
2.2. Expression of CCR6
2.3. Signaling Pathways of CCR6
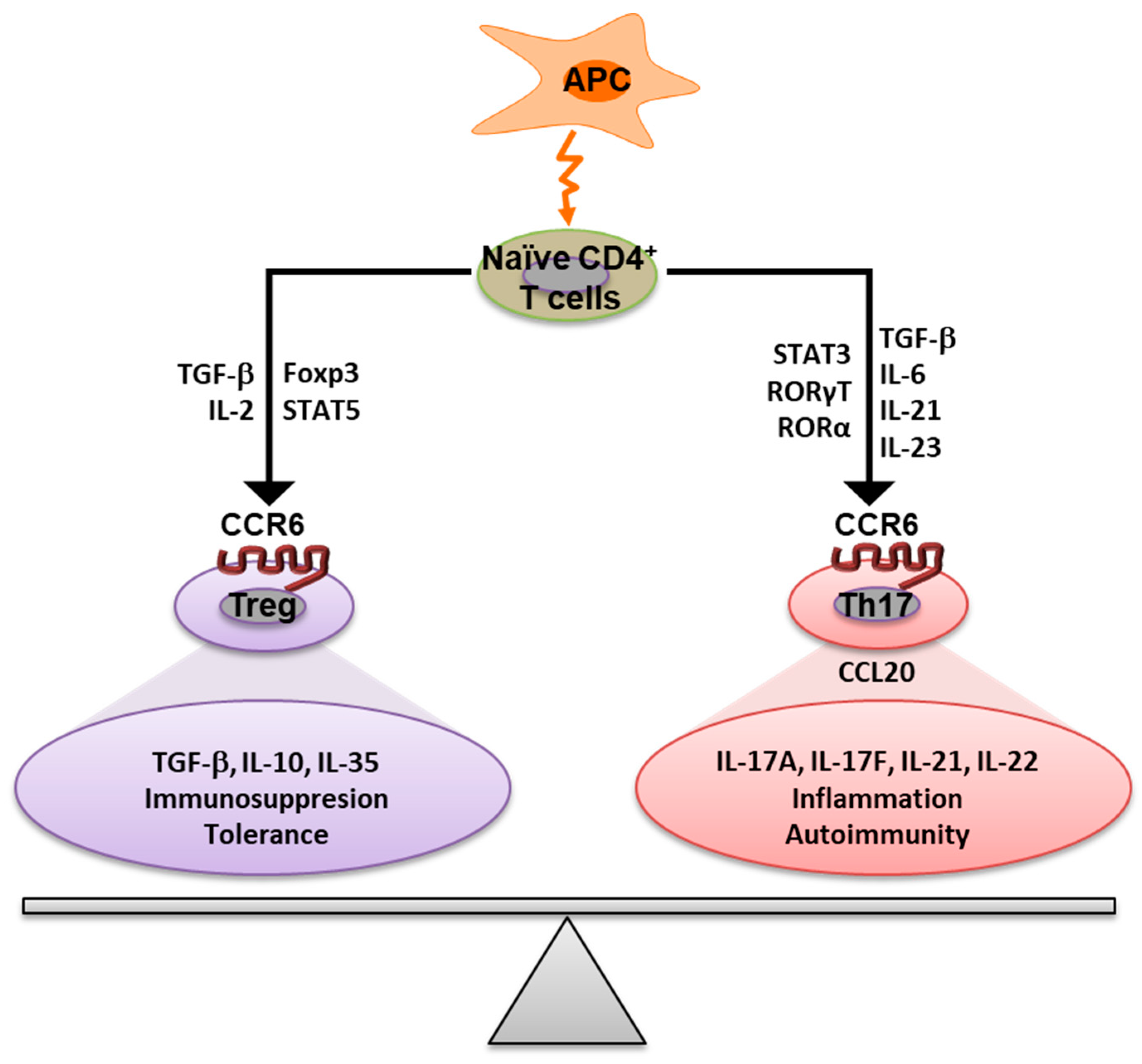
3. The Role of CCR6 in the Immune Response
4. Diseases Associated with CCR6
4.1. Lung Diseases
4.2. Cancer
4.3. Liver Diseases
4.4. Pancreas Diseases
4.5. Dry Eye Disease (DED)
4.6. Endometriosis (EM)
4.7. Renal Inflammation
4.8. Viral Infection
4.9. Inflammatory Bowel Disease (IBD)
4.10. Autoimmune Diseases
5. CCR6 Blocking Antibodies and Therapeutic Approaches
6. Future Perspectives
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Millar, R.P.; Newton, C.L. The year in G protein-coupled receptor research. Mol. Endocrinol. 2010, 24, 261–274. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.J.Y.; Gabriel, F.; Tandale, A.; Nietlispach, D. Structure and dynamics of GPCRs in lipid membranes: Physical principles and experimental approaches. Molecules 2020, 25, 4729. [Google Scholar] [CrossRef] [PubMed]
- Julian, B.; Gao, K.; Harwood, B.N.; Beinborn, M.; Kopin, A.S. Mutation-induced functional alterations of CCR6. J. Pharmacol. Exp. Ther. 2017, 360, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Zhou, Q.; Labroska, V.; Qin, S.; Darbalaei, S.; Wu, Y.; Yuliantie, E.; Xie, L.; Tao, H.; Cheng, J.; et al. G protein-coupled receptors: Structure- and function-based drug discovery. Signal Transduct. Target. Ther. 2021, 6, 7. [Google Scholar] [CrossRef] [PubMed]
- Jo, M.; Jung, S.T. Engineering therapeutic antibodies targeting G-protein-coupled receptors. Exp. Mol. Med. 2016, 48, e207. [Google Scholar] [CrossRef]
- White, G.E.; Iqbal, A.J.; Greaves, D.R. CC chemokine receptors and chronic inflammation-therapeutic opportunities and pharmacological challenges. Pharmacol. Rev. 2013, 65, 47–89. [Google Scholar] [CrossRef]
- Legler, D.F.; Thelen, M. New insights in chemokine signaling. F1000Research 2018, 7, 95. [Google Scholar] [CrossRef]
- Wasilko, D.J.; Johnson, Z.L.; Ammirati, M.; Che, Y.; Griffor, M.C.; Han, S.; Wu, H. Structural basis for chemokine receptor CCR6 activation by the endogenous protein ligand CCL20. Nat. Commun. 2020, 11, 3031. [Google Scholar] [CrossRef]
- Zhao, S.; Wu, B.; Stevens, R.C. Advancing Chemokine GPCR Structure Based Drug Discovery. Structure 2019, 27, 405–408. [Google Scholar] [CrossRef]
- Bobkov, V.; Arimont, M.; Zarca, A.; de Groof, T.W.M.; van der Woning, B.; de Haard, H.; Smit, M.J. Antibodies Targeting Chemokine Receptors CXCR4 and ACKR3. Mol. Pharmacol. 2019, 96, 753–764. [Google Scholar] [CrossRef]
- Ranasinghe, R.; Eri, R. Modulation of the CCR6-CCl20 axis: A potential therapeutic target in inflammation and cancer. Med. 2018, 54, 88. [Google Scholar] [CrossRef] [PubMed]
- Meitei, H.; Jadhav, N.; Lal, G. CCR6-CCL20 axis as a therapeutic target for autoimmune diseases. Autoimmun. Rev. 2021, 20, 102846. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.Y.S.; Körner, H. The CCR6-CCL20 axis in humoral immunity and T-B cell immunobiology. Immunobiology 2019, 224, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.Y.S.; Phan, T.K.; Hulett, M.D.; Körner, H. The relationship between CCR6 and its binding partners: Does the CCR6-CCL20 axis have to be extended? Cytokine 2015, 72, 97–101. [Google Scholar] [CrossRef]
- Baba, M.; Imai, T.; Nishimura, M.; Kakizaki, M.; Takagi, S.; Hieshima, K.; Nomiyama, H.; Yoshie, O. Identification of CCR6, the specific receptor for a novel lymphocyte- directed CC chemokine LARC. J. Biol. Chem. 1997, 272, 14893–14898. [Google Scholar] [CrossRef]
- Hutchings, C.J. A review of antibody-based therapeutics targeting G protein-coupled receptors: An update. Expert Opin. Biol. Ther. 2020, 20, 925–935. [Google Scholar] [CrossRef]
- Schutyser, E.; Struyf, S.; Van Damme, J. The CC chemokine CCL20 and its receptor CCR6. Cytokine Growth Factor Rev. 2003, 14, 409–426. [Google Scholar] [CrossRef]
- Katritch, V.; Cherezov, V.; Stevens, R.C. Diversity and modularity of G protein-coupled receptor structures. Trends Pharmacol. Sci. 2012, 33, 17–27. [Google Scholar] [CrossRef]
- Ranasinghe, R.; Eri, R. Pleiotropic Immune Functions of Chemokine Receptor 6 in Health and Disease. Medicines 2018, 5, 69. [Google Scholar] [CrossRef]
- Islam, S.; Shimizu, N.; Ohtsuki, T.; Jinno-Oue, A.; Tanaka, A.; Hoshino, H. CKR-L3, a deletion version CCR6-isoform shows coreceptor-activity for limited human and simian immunodeficiency viruses. BMC Infect. Dis. 2014, 14, P69. [Google Scholar] [CrossRef]
- Munk, C.; Isberg, V.; Mordalski, S.; Harpsøe, K.; Rataj, K.; Hauser, A.S.; Kolb, P.; Bojarski, A.J.; Vriend, G.; Gloriam, D.E. GPCRdb: The G protein-coupled receptor database—An introduction. Br. J. Pharmacol. 2016, 173, 2195–2207. [Google Scholar] [CrossRef]
- Ranasinghe, R.; Eri, R. CCR6–CCL20-Mediated Immunologic Pathways in Inflammatory Bowel Disease. Gastrointest. Disord. 2018, 1, 15–29. [Google Scholar] [CrossRef]
- Kadomoto, S.; Izumi, K.; Mizokami, A. The CCL20-CCR6 axis in cancer progression. Int. J. Mol. Sci. 2020, 21, 5186. [Google Scholar] [CrossRef] [PubMed]
- Hedrick, M.N.; Lonsdorf, A.S.; Hwang, S.T.; Farber, J.M. CCR6 as a possible therapeutic target in psoriasis. Expert Opin. Ther. Targets 2010, 14, 911–922. [Google Scholar] [CrossRef]
- Dorgham, K.; Dejou, C.; Piesse, C.; Gorochov, G.; Pène, J.; Yssel, H. Identification of the Single Immunodominant Region of the Native Human CC Chemokine Receptor 6 Recognized by Mouse Monoclonal Antibodies. PLoS ONE 2016, 11, e0157740. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Carson, W.F.; Cavassani, K.A.; Connett, J.M.; Kunkel, S.L. CCR6 as a mediator of immunity in the lung and gut. Exp. Cell Res. 2011, 317, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Charbonnier, A.S.; Kohrgruber, N.; Kriehuber, E.; Stingl, G.; Rot, A.; Maurer, D. Macrophage inflammatory protein 3α is involved in the constitutive trafficking of epidermal langerhans cells. J. Exp. Med. 1999, 190, 1755–1767. [Google Scholar] [CrossRef] [PubMed]
- Vilgelm, A.E.; Richmond, A. Chemokines Modulate Immune Surveillance in Tumorigenesis, Metastasis, and Response to Immunotherapy. Front. Immunol. 2019, 10, 333. [Google Scholar] [CrossRef]
- Furue, K.; Ito, T.; Tsuji, G.; Nakahara, T.; Furue, M. The CCL20 and CCR6 axis in psoriasis. Scand. J. Immunol. 2020, 91, e12846. [Google Scholar] [CrossRef]
- Jiang, Q.; Yang, G.; Xiao, F.; Xie, J.; Wang, S.; Lu, L.; Cui, D. Role of Th22 Cells in the Pathogenesis of Autoimmune Diseases. Front. Immunol. 2021, 12, 688066. [Google Scholar] [CrossRef]
- Single Cell Type—CCR6—The Human Protein Atlas. Available online: https://www.proteinatlas.org/ENSG00000112486-CCR6/single+cell+type (accessed on 29 March 2023).
- CCL20 Transcriptomics Data—The Human Protein Atlas. Available online: https://www.proteinatlas.org/ENSG00000115009-CCL20/summary/rna (accessed on 29 March 2023).
- Gómez-Melero, S.; García-Maceira, F.; García-Maceira, T.; Luna-Guerrero, V.; Montero-Peñalvo, G.; Túnez-Fiñana, I.; Paz-Rojas, E. Amino terminal recognition by a CCR6 chemokine receptor antibody blocks CCL20 signaling and IL-17 expression via β-arrestin. BMC Biotechnol. 2021, 21, 41. [Google Scholar] [CrossRef] [PubMed]
- Gurevich, V.V.; Gurevich, E.V. Arrestin-mediated signaling: Is there a controversy? World J. Biol. Chem. 2018, 9, 25–35. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Melero, S.; García-Maceira, F.I.; García-Maceira, T.; Luna-Guerrero, V.; Montero-Peñalvo, G.; Caballero-Villarraso, J.; Túnez, I.; Paz-Rojas, E. Development of a High-Throughput Calcium Mobilization Assay for CCR6 Receptor Coupled to Hydrolase Activity Readout. Biomedicines 2022, 10, 422. [Google Scholar] [CrossRef]
- Kumari, P.; Ghosh, E.; Shukla, A.K. Emerging Approaches to GPCR Ligand Screening for Drug Discovery. Trends Mol. Med. 2015, 21, 687–701. [Google Scholar] [CrossRef] [PubMed]
- Gurevich, V.V.; Gurevich, E.V. GPCR signaling regulation: The role of GRKs and arrestins. Front. Pharmacol. 2019, 10, 125. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.S.; Rajagopal, S. The β-Arrestins: Multifunctional regulators of G protein-coupled receptors. J. Biol. Chem. 2016, 291, 8969–8977. [Google Scholar] [CrossRef] [PubMed]
- Hoover, D.M.; Boulègue, C.; Yang, D.; Oppenheim, J.J.; Tucker, K.; Lu, W.; Lubkowski, J. The Structure of Human Macrophage Inflammatory Protein-3α/CCL20: Linking antimicrobial and cc chemokine receptor-6-binding activities with human β-defensins. J. Biol. Chem. 2002, 277, 37647–37654. [Google Scholar] [CrossRef]
- Riutta, S.J.; Larsen, O.; Getschman, A.E.; Rosenkilde, M.M.; Hwang, S.T.; Volkman, B.F. Mutational analysis of CCL20 reveals flexibility of N-terminal amino acid composition and length. J. Leukoc. Biol. 2018, 104, 423–434. [Google Scholar] [CrossRef]
- Korona, B.; Korona, D.; Zhao, W.; Wotherspoon, A.C.; Du, M.Q. CCR6 activation links innate immune responses to mucosa-associated lymphoid tissue lymphoma development. Haematologica 2022, 107, 1384–1396. [Google Scholar] [CrossRef]
- Ghannam, S.; Dejou, C.; Pedretti, N.; Giot, J.-P.; Dorgham, K.; Boukhaddaoui, H.; Deleuze, V.; Bernard, F.-X.; Jorgensen, C.; Yssel, H.; et al. CCL20 and β-Defensin-2 Induce Arrest of Human Th17 Cells on Inflamed Endothelium In Vitro under Flow Conditions. J. Immunol. 2011, 186, 1411–1420. [Google Scholar] [CrossRef]
- Lee, G.R. The Balance of Th17 versus Treg Cells in Autoimmunity. Int. J. Mol. Sci. 2018, 19, 730. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Paul, W.E. Peripheral CD4 T cell differentiation regulated by networks of cytokines and transcription factors. Immunol. Rev. 2010, 238, 247. [Google Scholar] [CrossRef] [PubMed]
- Renaude, E.; Kroemer, M.; Loyon, R.; Binda, D.; Borg, C.; Guittaut, M.; Hervouet, E.; Peixoto, P. The Fate of Th17 Cells is Shaped by Epigenetic Modifications and Remodeled by the Tumor Microenvironment. Int. J. Mol. Sci. 2020, 21, 1673. [Google Scholar] [CrossRef]
- Wang, C.; Kang, S.G.; Lee, J.; Sun, Z.; Kim, C.H. The roles of CCR6 in migration of Th17 cells and regulation of effector T-cell balance in the gut. Mucosal Immunol. 2009, 2, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Jatczak-Pawlik, I.; Wolinski, P.; Książek-Winiarek, D.; Pietruczuk, M.; Glabinski, A. CCR6 blockade on regulatory T cells ameliorates experimental model of multiple sclerosis. Cent. Eur. J. Immunol. 2020, 45, 256–266. [Google Scholar] [CrossRef]
- Mo, C.; Zeng, Z.; Deng, Q.; Ding, Y.; Xiao, R. Imbalance between T helper 17 and regulatory T cell subsets plays a significant role in the pathogenesis of systemic sclerosis. Biomed. Pharmacother. 2018, 108, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, T.; Yang, X.O.; Chung, Y.; Fukunaga, A.; Nurieva, R.; Pappu, B.; Martin-Orozco, N.; Kang, H.S.; Ma, L.; Panopoulos, A.D.; et al. CCR6 Regulates the Migration of Inflammatory and Regulatory T Cells. J. Immunol. 2008, 181, 8391–8401. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Zhang, H.; Xie, H.; Chen, L.; Li, S.; Zheng, J.; Chai, R.; Wang, Z.; Zang, Y.; He, S. Reduced CCR6+IL-17A+Treg Cells in Blood and CCR6-Dependent Accumulation of IL-17A+Treg Cells in Lungs of Patients with Allergic Asthma. Front. Immunol. 2021, 12, 3382. [Google Scholar] [CrossRef]
- Thomas, S.Y.; Banerji, A.; Medoff, B.D.; Lilly, C.M.; Luster, A.D. Multiple chemokine receptors, including CCR6 and CXCR3, regulate antigen-induced T cell homing to the human asthmatic airway. J. Immunol. 2007, 179, 1901–1912. [Google Scholar] [CrossRef]
- Nakayama, T.; Fujisawa, R.; Yamada, H.; Horikawa, T.; Kawasaki, H.; Hieshima, K.; Izawa, D.; Fujiie, S.; Tezuka, T.; Yoshie, O. Inducible expression of a CC chemokine liver- and activation-regulated chemokine (LARC)/macrophage inflammatory protein (MIP)-3 alpha/CCL20 by epidermal keratinocytes and its role in atopic dermatitis. Int. Immunol. 2001, 13, 95–103. [Google Scholar] [CrossRef]
- Rafaels, N.M.; Beck, L.A.; Gao, P.S.; Hand, T.; Boguniewicz, M.; Hata, T.R.; Schneider, L.; Hanifin, J.M.; Gallo, R.L.; Gao, L. Variations in the CCL20 and CCR6 Genes are Associated with Atopic Dermatitis and Eczema Herpeticum in Populations of European and African descent. J. Allergy Clin. Immunol. 2009, 123, S70. [Google Scholar] [CrossRef]
- Kadomoto, S.; Izumi, K.; Hiratsuka, K.; Nakano, T.; Naito, R.; Makino, T.; Iwamoto, H.; Yaegashi, H.; Shigehara, K.; Kadono, Y.; et al. Tumor-associated macrophages induce migration of renal cell carcinoma cells via activation of the CCL20-CCR6 axis. Cancers 2020, 12, 89. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wang, W.; Zhang, N.; Di, W. The role of CCL20-CCR6 axis in ovarian cancer metastasis. Onco. Targets. Ther. 2020, 13, 12739–12750. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.P.; Hu, Z.J.; Meng, A.H.; Duan, G.C.; Zhao, Q.T.; Yang, J. Role of CCL20/CCR6 and the ERK signaling pathway in lung adenocarcinoma. Oncol. Lett. 2017, 14, 8183–8189. [Google Scholar] [CrossRef] [PubMed]
- Rubie, C.; Frick, V.O.; Ghadjar, P.; Wagner, M.; Grimm, H.; Vicinus, B.; Justinger, C.; Graeber, S.; Schilling, M.K. CCL20/CCR6 expression profile in pancreatic cancer. J. Transl. Med. 2010, 8, 45. [Google Scholar] [CrossRef]
- Yu, Q.; Lou, X.M.; He, Y. Preferential recruitment of Th17 cells to cervical cancer via CCR6-CCL20 pathway. PLoS ONE 2015, 10, 0120855. [Google Scholar] [CrossRef]
- Korbecki, J.; Grochans, S.; Gutowska, I.; Barczak, K.; Baranowska-Bosiacka, I. CC chemokines in a tumor: A review of pro-cancer and anti-cancer properties of receptors CCR5, CCR6, CCR7, CCR8, CCR9, and CCR10 ligands. Int. J. Mol. Sci. 2020, 21, 7619. [Google Scholar] [CrossRef]
- Chen, W.; Qin, Y.; Liu, S. CCL20 Signaling in the Tumor Microenvironment. Adv. Exp. Med. Biol. 2020, 1231, 53–65. [Google Scholar] [CrossRef]
- Du, D.; Liu, Y.; Qian, H.; Zhang, B.; Tang, X.; Zhang, T.; Liu, W. The effects of the CCR6/CCL20 biological axis on the invasion and metastasis of hepatocellular carcinoma. Int. J. Mol. Sci. 2014, 15, 6441–6452. [Google Scholar] [CrossRef]
- Liu, F.; Lv, H.; Jia, X.; Liu, G.; Li, T.; Xu, Z.; Li, J. CC chemokine receptor 6 expression predicts poor prognosis in hepatocellular carcinoma. J. Surg. Oncol. 2014, 110, 151–155. [Google Scholar] [CrossRef]
- Kapur, N.; Mir, H.; Clark, C.E.; Krishnamurti, U.; Beech, D.J.; Lillard, J.W.; Singh, S. CCR6 expression in colon cancer is associated with advanced disease and supports epithelial-to-mesenchymal transition. Br. J. Cancer 2016, 114, 1343–1351. [Google Scholar] [CrossRef] [PubMed]
- Rubie, C.; Oliveira, V.; Kempf, K.; Wagner, M.; Tilton, B.; Rau, B.; Kruse, B.; König, J.; Schilling, M. Involvement of chemokine receptor CCR6 in colorectal cancer metastasis. Tumor Biol. 2006, 27, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Boyle, S.T.; Faulkner, J.W.; McColl, S.R.; Kochetkova, M. The chemokine receptor CCR6 facilitates the onset of mammary neoplasia in the MMTV-PyMT mouse model via recruitment of tumor-promoting macrophages. Mol. Cancer 2015, 14, 115. [Google Scholar] [CrossRef] [PubMed]
- Ghadjar, P.; Loddenkemper, C.; Coupland, S.E.; Stroux, A.; Noutsias, M.; Thiel, E.; Christoph, F.; Miller, K.; Scheibenbogen, C.; Keilholz, U. Chemokine receptor CCR6 expression level and aggressiveness of prostate cancer. J. Cancer Res. Clin. Oncol. 2008, 134, 1181–1189. [Google Scholar] [CrossRef] [PubMed]
- Oo, Y.H.; Banz, V.; Kavanagh, D.; Liaskou, E.; Withers, D.R.; Humphreys, E.; Reynolds, G.M.; Lee-Turner, L.; Kalia, N.; Hubscher, S.G.; et al. CXCR3-dependent recruitment and CCR6-mediated positioning of Th-17 cells in the inflamed liver. J. Hepatol. 2012, 57, 1044–1051. [Google Scholar] [CrossRef]
- Hammerich, L.; Bangen, J.M.; Govaere, O.; Zimmermann, H.W.; Gassler, N.; Huss, S.; Liedtke, C.; Prinz, I.; Lira, S.A.; Luedde, T.; et al. Chemokine receptor CCR6-dependent accumulation of γδ T cells in injured liver restricts hepatic inflammation and fibrosis. Hepatology 2014, 59, 630–642. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Namkoong, H.; Yang, Y.; Huang, H.; Heller, D.; Szot, G.L.; Davis, M.M.; Husain, S.Z.; Pandol, S.J.; Bellin, M.D.; et al. Single-cell sequencing unveils distinct immune microenvironments with CCR6-CCL20 crosstalk in human chronic pancreatitis. Gut 2022, 71, 1831–1842. [Google Scholar] [CrossRef]
- Demedts, I.K.; Bracke, K.R.; Van Pottelberge, G.; Testelmans, D.; Verleden, G.M.; Vermassen, F.E.; Joos, G.F.; Brusselle, G.G. Accumulation of Dendritic Cells and Increased CCL20 Levels in the Airways of Patients with Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2012, 175, 998–1005. [Google Scholar] [CrossRef]
- Bracke, K.R.; Demedts, I.K.; Joos, G.F.; Brusselle, G.G. CC-chemokine receptors in chronic obstructive pulmonary disease. Inflamm. Allergy Drug Targets 2007, 6, 75–79. [Google Scholar] [CrossRef]
- Saris, A.; Reijnders, T.D.Y.; Reijm, M.; Hollander, J.C.; de Buck, K.; Schuurman, A.R.; Duitman, J.W.; Heunks, L.; Aman, J.; Bogaard, H.J.; et al. Enrichment of CCR6+ CD8+ T cells and CCL20 in the lungs of mechanically ventilated patients with COVID-19. Eur. J. Immunol. 2021, 51, 1535–1538. [Google Scholar] [CrossRef]
- Lee, S.M.; Yang, H.; Tartar, D.M.; Gao, B.; Luo, X.; Ye, S.Q.; Zaghouani, H.; Fang, D. Prevention and treatment of diabetes with resveratrol in a non-obese mouse model of type 1 diabetes. Diabetologia 2011, 54, 1136. [Google Scholar] [CrossRef] [PubMed]
- Dohlman, T.H.; Chauhan, S.K.; Kodati, S.; Hua, J.; Chen, Y.; Omoto, M.; Sadrai, Z.; Dana, R. The CCR6/CCL20 axis mediates Th17 cell migration to the ocular surface in dry eye disease. Investig. Ophthalmol. Vis. Sci. 2013, 54, 4081–4091. [Google Scholar] [CrossRef] [PubMed]
- Coursey, T.G.; Gandhi, N.B.; Volpe, E.A.; Pflugfelder, S.C.; De Paiva, C.S. Chemokine receptors CCR6 and CXCR3 are necessary for CD4(+) T cell mediated ocular surface disease in experimental dry eye disease. PLoS ONE 2013, 8, 0078508. [Google Scholar] [CrossRef] [PubMed]
- Hirata, T.; Osuga, Y.; Takamura, M.; Kodama, A.; Hirota, Y.; Koga, K.; Yoshino, O.; Harada, M.; Takemura, Y.; Yano, T.; et al. Recruitment of CCR6-expressing Th17 cells by CCL 20 secreted from IL-1β-, TNF-α-, and IL-17A-stimulated endometriotic stromal cells. Endocrinology 2010, 151, 5468–5476. [Google Scholar] [CrossRef]
- Tan, J.; Xu, T.; Gou, Y.; Wang, H.; Liang, Z.; Cao, Y.; Wang, H.; Yu, Y.; Jiao, N.; Zhang, Z. CCL20/CCR6 axis mediates macrophages to promote proliferation and migration of ESCs by blocking autophagic flux in endometriosis. Stem Cell Res. Ther. 2022, 13, 294. [Google Scholar] [CrossRef]
- Welsh-Bacic, D.; Lindenmeyer, M.; Cohen, C.D.; Draganovici, D.; Mandelbaum, J.; Edenhofer, I.; Ziegler, U.; Regele, H.; Wüthrich, R.P.; Segerer, S. Expression of the chemokine receptor CCR6 in human renal inflammation. Nephrol. Dial. Transplant. 2011, 26, 1211–1220. [Google Scholar] [CrossRef]
- Turner, J.E.; Paust, H.J.; Steinmetz, O.M.; Peters, A.; Riedel, J.H.; Erhardt, A.; Wegscheid, C.; Velden, J.; Fehr, S.; Mittrücker, H.W.; et al. CCR6 recruits regulatory T cells and Th17 cells to the kidney in glomerulonephritis. J. Am. Soc. Nephrol. 2010, 21, 974–985. [Google Scholar] [CrossRef]
- Lee, A.Y.S.; Körner, H. CCR6/CCL20 chemokine axis in human immunodeficiency virus immunity and pathogenesis. J. Gen. Virol. 2017, 98, 338–344. [Google Scholar] [CrossRef]
- Gosselin, A.; Salinas, T.R.W.; Planas, D.; Wacleche, V.S.; Zhang, Y.; Fromentin, R.; Chomont, N.; Cohen, E.A.; Shacklett, B.; Mehraj, V.; et al. HIV persists in CCR6+CD4+ T cells from colon and blood during antiretroviral therapy. AIDS 2017, 31, 35. [Google Scholar] [CrossRef]
- Varona, R.; Cadenas, V.; Flores, J.; Martínez, A.C.; Márquez, G. CCR6 has a non-redundant role in the development of inflammatory bowel disease. Eur. J. Immunol. 2003, 33, 2937–2946. [Google Scholar] [CrossRef]
- Ranasinghe, R.; Fernando, R.; Perera, A.P.; Shastri, M.; Basheer, W.; Scowen, P.; Pinfold, T.; Eri, R. Ccr6 Deficiency Attenuates Spontaneous Chronic Colitis in Winnie. Gastrointest. Disord. 2020, 2, 27–47. [Google Scholar] [CrossRef]
- Skovdahl, H.K.; Van Beelen Granlund, A.; Østvik, A.E.; Bruland, T.; Bakke, I.; Torp, S.H.; Damas, J.K.; Sandvik, A.K. Expression of CCL20 and Its Corresponding Receptor CCR6 Is Enhanced in Active Inflammatory Bowel Disease, and TLR3 Mediates CCL20 Expression in Colonic Epithelial Cells. PLoS ONE 2015, 10, 0141710. [Google Scholar] [CrossRef] [PubMed]
- Lees, C.W.; Barrett, J.C.; Parkes, M.; Satsangi, J. New IBD genetics: Common pathways with other diseases. Gut 2011, 60, 1739–1753. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.J.; Mu, R.; Li, C.; Nath, S.K.; Zhang, Y.M.; Qi, Y.Y.; Li, Z.G.; Zhao, M.H.; Zhang, H. Variants in CCR6 are associated with susceptibility to lupus nephritis in Chinese. Arthritis Rheumatol. 2015, 67, 3091. [Google Scholar] [CrossRef]
- Lee, A.Y.S.; Körner, H. CC chemokine receptor 6 (CCR6) in the pathogenesis of systemic lupus erythematosus. Immunol. Cell Biol. 2020, 98, 845–853. [Google Scholar] [CrossRef]
- Comerford, I.; Bunting, M.; Fenix, K.; Haylock-Jacobs, S.; Litchfield, W.; Harata-Lee, Y.; Turvey, M.; Brazzatti, J.; Gregor, C.; Nguyen, P.; et al. An immune paradox: How can the same chemokine axis regulate both immune tolerance and activation? CCR6/CCL20: A chemokine axis balancing immunological tolerance and inflammation in autoimmune disease. BioEssays 2010, 32, 1067–1076. [Google Scholar] [CrossRef]
- Reboldi, A.; Coisne, C.; Baumjohann, D.; Benvenuto, F.; Bottinelli, D.; Lira, S.; Uccelli, A.; Lanzavecchia, A.; Engelhardt, B.; Sallusto, F. C-C chemokine receptor 6–regulated entry of TH-17 cells into the CNS through the choroid plexus is required for the initiation of EAE. Nat. Immunol. 2009, 10, 514–523. [Google Scholar] [CrossRef]
- Latek, D.; Modzelewska, A.; Trzaskowski, B.; Palczewski, K.; Filipek, S. G protein-coupled receptors-recent advances. Acta Biochim. Pol. 2012, 59, 515–529. [Google Scholar] [CrossRef]
- Shi, Z.; Garcia-Melchor, E.; Wu, X.; Getschman, A.E.; Nguyen, M.; Rowland, D.J.; Wilson, M.; Sunzini, F.; Akbar, M.; Huynh, M.; et al. Targeting the CCR6/CCL20 Axis in Entheseal and Cutaneous Inflammation. Arthritis Rheumatol. 2021, 73, 2271–2281. [Google Scholar] [CrossRef]
- Getschman, A.E.; Imai, Y.; Larsen, O.; Peterson, F.C.; Wu, X.; Rosenkilde, M.M.; Hwang, S.T.; Volkman, B.F. Protein engineering of the chemokine CCL20 prevents psoriasiform dermatitis in an IL-23–dependent murine model. Proc. Natl. Acad. Sci. USA 2017, 114, 12460–12465. [Google Scholar] [CrossRef]
- Hedrick, M.N.; Lonsdorf, A.S.; Shirakawa, A.K.; Lee, C.C.R.; Liao, F.; Singh, S.P.; Zhang, H.H.; Grinberg, A.; Love, P.E.; Hwang, S.T.; et al. CCR6 is required for IL-23–induced psoriasis-like inflammation in mice. J. Clin. Invest. 2009, 119, 2317. [Google Scholar] [CrossRef] [PubMed]
- Mabuchi, T.; Singh, T.P.; Takekoshi, T.; Jia, G.F.; Wu, X.; Kao, M.C.; Weiss, I.; Farber, J.M.; Hwang, S.T. CCR6 is required for epidermal trafficking of γδ-T cells in an IL-23-induced model of psoriasiform dermatitis. J. Invest. Dermatol. 2013, 133, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Corvaisier, M.; Delneste, Y.; Jeanvoine, H.; Preisser, L.; Blanchard, S.; Garo, E.; Hoppe, E.; Barré, B.; Audran, M.; Bouvard, B.; et al. IL-26 Is Overexpressed in Rheumatoid Arthritis and Induces Proinflammatory Cytokine Production and Th17 Cell Generation. PLoS Biol. 2012, 10, 1001395. [Google Scholar] [CrossRef]
- Hirota, K.; Yoshitomi, H.; Hashimoto, M.; Maeda, S.; Teradaira, S.; Sugimoto, N.; Yamaguchi, T.; Nomura, T.; Ito, H.; Nakamura, T.; et al. Preferential recruitment of CCR6-expressing Th17 cells to inflamed joints via CCL20 in rheumatoid arthritis and its animal model. J. Exp. Med. 2007, 204, 2803–2812. [Google Scholar] [CrossRef]
- Akhtar, M.; Khan, S.; Ali, Y.; Haider, S.; Ud Din, J.; Islam, Z.-U.; Jalil, F. Association study of CCR6 rs3093024 with Rheumatoid Arthritis in a Pakistani cohort. Saudi J. Biol. Sci. 2020, 27, 3354–3358. [Google Scholar] [CrossRef]
- Cheng, P.; Zhang, Y.; Huang, H.; Zhang, W.; Yang, Q.; Guo, F.; Chen, A. Association between CCR6 and rheumatoid arthritis: A meta-analysis. Int. J. Clin. Exp. Med. 2015, 8, 5388. [Google Scholar]
- Quan, C.; Ren, Y.Q.; Xiang, L.H.; Sun, L.D.; Xu, A.E.; Gao, X.H.; Chen, H.D.; Pu, X.M.; Wu, R.N.; Liang, C.Z.; et al. Genome-wide association study for vitiligo identifies susceptibility loci at 6q27 and the MHC. Nat. Genet. 2010, 42, 614–618. [Google Scholar] [CrossRef]
- Affo, S.; Rodrigo-Torres, D.; Blaya, D.; Morales-Ibanez, O.; Coll, M.; Millan, C.; Altamirano, J.; Arroyo, V.; Caballeria, J.; Bataller, R.; et al. Chemokine Receptor Ccr6 Deficiency Alters Hepatic Inflammatory Cell Recruitment and Promotes Liver Inflammation and Fibrosis. PLoS ONE 2015, 10, 0145147. [Google Scholar] [CrossRef]
- Shimizu, Y.; Murata, H.; Kashii, Y.; Hirano, K.; Kunitani, H.; Higuchi, K.; Watanabe, A. CC-chemokine receptor 6 and its ligand macrophage inflammatory protein 3α might be involved in the amplification of local necroinflammatory response in the liver. Hepatology 2001, 34, 311–319. [Google Scholar] [CrossRef]
- Sriram, K.; Insel, P.A. G Protein-Coupled Receptors as Targets for Approved Drugs: How Many Targets and How Many Drugs? Mol. Pharmacol. 2018, 93, 251–258. [Google Scholar] [CrossRef]
- Mackay, C.R. Moving targets: Cell migration inhibitors as new anti-inflammatory therapies. Nat. Immunol. 2008, 9, 988–998. [Google Scholar] [CrossRef] [PubMed]
- Bachelerie, F.; Ben-Baruch, A.; Burkhardt, A.M.; Combadiere, C.; Farber, J.M.; Graham, G.J.; Horuk, R.; Sparre-Ulrich, A.H.; Locati, M.; Luster, A.D.; et al. International Union of Pharmacology. LXXXIX. Update on the Extended Family of Chemokine Receptors and Introducing a New Nomenclature for Atypical Chemokine Receptors. Pharmacol. Rev. 2014, 66, 1. [Google Scholar] [CrossRef] [PubMed]
- Martina, M.G.; Giorgio, C.; Allodi, M.; Palese, S.; Barocelli, E.; Ballabeni, V.; Szpakowska, M.; Chevigné, A.; Piet van Hamburg, J.; Davelaar, N.; et al. Discovery of small-molecules targeting the CCL20/CCR6 axis as first-in-class inhibitors for inflammatory bowel diseases. Eur. J. Med. Chem. 2022, 243, 114703. [Google Scholar] [CrossRef] [PubMed]
- Tawaraishi, T.; Sakauchi, N.; Hidaka, K.; Yoshikawa, K.; Okui, T.; Kuno, H.; Chisaki, I.; Aso, K. Identification of a novel series of potent and selective CCR6 inhibitors as biological probes. Bioorg. Med. Chem. Lett. 2018, 28, 3067–3072. [Google Scholar] [CrossRef]
- Amgen to Acquire ChemoCentryx for $3.7B, Bolstering Autoimmune Portfolio. Available online: https://www.genengnews.com/topics/drug-discovery/therapeutics/autoimmune-disease-drugs/amgen-to-acquire-chemocentryx-for-3-7b-bolstering-autoimmune-portfolio/ (accessed on 21 February 2023).
- Dairaghi, D.; Zhang, P.; Leleti, M.; Berahovich, R.; Ebsworth, K.; Ertl, L.; Miao, S.; Miao, Z.; Seitz, L.; Tan, J.; et al. Inhibition of Chemokine Receptors CCR1 and CCR6 as Promising Therapies for Autoimmune Diseases Such as Rheumatoid Arthritis and Psoriasis—ACR Meeting Abstracts. Available online: https://acrabstracts.org/abstract/inhibition-of-chemokine-receptors-ccr1-and-ccr6-as-promising-therapies-for-autoimmune-diseases-such-as-rheumatoid-arthritis-and-psoriasis/ (accessed on 10 February 2023).
- Campbell, J.J.; Ebsworth, K.; Ertl, L.S.; McMahon, J.P.; Newland, D.; Wang, Y.; Liu, S.; Miao, Z.; Dang, T.; Zhang, P.; et al. IL-17–Secreting γδ T Cells Are Completely Dependent upon CCR6 for Homing to Inflamed Skin. J. Immunol. 2017, 199, 3129–3136. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J.J.; Ebsworth, K.; Ertl, L.S.; McMahon, J.P.; Wang, Y.; Yau, S.; Mali, V.R.; Chhina, V.; Kumamoto, A.; Liu, S.; et al. Efficacy of Chemokine Receptor Inhibition in Treating IL-36α–Induced Psoriasiform Inflammation. J. Immunol. 2019, 202, 1687–1692. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Vasilyeva, V.; Abbasova, S.; Ivanova, E.; Ovchinnikova, E.; Ulitin, A.; Mikhaylov, R. Human Monoclonal Antibodies against Human Chemokine Receptor CCR6. WO2013184218A1. 2013. Available online: https://patents.google.com/patent/WO2013184218A1/pt-PT (accessed on 11 April 2023).
- Yuya, I.; Mami, K.; Yutaka, K.; Kazuya, Y. Anti-Human CCR6 Antibody. 2013. Available online: https://patents.google.com/patent/WO2013005649A1/en (accessed on 11 April 2023).
- Lissilaa, R.; Walmsley, A.; Blein, S.; Ollier, R.; Hou, S.; Loyau, J. Antibodies that Bind to CCR6 and their Uses. 2015. Available online: https://patents.google.com/patent/WO2016059253A1/en (accessed on 11 April 2023).
- Partnered Programs—Sosei Heptares. Available online: https://soseiheptares.com/our-pipeline/rd-pipeline/partnered-programs.html (accessed on 26 February 2023).
- Castelli, M.S.; McGonigle, P.; Hornby, P.J. The pharmacology and therapeutic applications of monoclonal antibodies. Pharmacol. Res. Perspect. 2019, 7, e00535. [Google Scholar] [CrossRef]
- Ju, M.S.; Jung, S.T. Antigen design for successful isolation of highly challenging therapeutic anti-gpcr antibodies. Int. J. Mol. Sci. 2020, 21, 8240. [Google Scholar] [CrossRef]
- Kim, S.K.; Fristrup, P.; Abrol, R.; Goddard, W.A. Structure-based prediction of subtype-selectivity of Histamine H3 receptor selective antagonists in clinical trials. J. Chem. Inf. Model. 2011, 51, 3262. [Google Scholar] [CrossRef]

| Cell Type | Reference |
|---|---|
| B cell | [19,22,23,24] |
| Immature DC | [19,22,24,25] |
| ICL-3 | [19,22,25] |
| Langerhans cell | [26,27] |
| NK cell | [17,28] |
| NKT cell | [19,22,26] |
| Neutrophils | [19,22] |
| Th9 cell | [28] |
| Th17 cell | [19,22,23,29] |
| Th22 cell | [28,30] |
| Treg cell | [19,22,23,24] |
| γδT cell | [28] |
| Disease | Reference |
|---|---|
| Asthma | [26,50,51] |
| Atopic dermatitis | [52,53] |
| Cancer | [23,54,55,56,57,58,59,60,61,62,63,64,65,66] |
| Cholestatic liver diseases | [67] |
| Chronic liver diseases | [68] |
| Chronic pancreatitis | [69] |
| COPD | [70,71] |
| COVID-19 | [72] |
| Diabetes | [73] |
| DED | [74,75] |
| Endometriosis | [76,77] |
| Glomerulonephritis | [78,79] |
| HIV | [80,81] |
| IBD | [22,26,82,83,84,85] |
| Systemic Lupus erythematosus | [86,87] |
| Multiple sclerosis | [25,47,88,89] |
| Psoriasis | [24,29,90,91,92,93,94] |
| Rheumatoid arthritis | [25,95,96,97,98] |
| Vitiligo | [99] |
| Name | Company | Type of Inhibitor | Reference |
|---|---|---|---|
| Compound 35 | Takeda Pharmaceutical | Small molecule | [106] |
| Compound 1b | MedChemExpress | Small molecule | [105] |
| CCX587 | Chemocentryx | Small molecule | [107] |
| CCX9664 | Chemocentryx | Small molecule | [108] |
| CCX2553 | Chemocentryx | Small molecule | [109] |
| CCX624 | Chemocentryx | Small molecule | [29,110] |
| WO2013184218A1 | Msm Protein Technologies | Antibody | [111] |
| WO2013005649A1 | Kyowa Hakko Kirin | Antibody | [112] |
| WO2016059253A1 | Glenmark Pharmaceuticals | Antibody | [113] |
| PF-07054894 | Pfizer | Small molecule | [114] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gómez-Melero, S.; Caballero-Villarraso, J. CCR6 as a Potential Target for Therapeutic Antibodies for the Treatment of Inflammatory Diseases. Antibodies 2023, 12, 30. https://doi.org/10.3390/antib12020030
Gómez-Melero S, Caballero-Villarraso J. CCR6 as a Potential Target for Therapeutic Antibodies for the Treatment of Inflammatory Diseases. Antibodies. 2023; 12(2):30. https://doi.org/10.3390/antib12020030
Chicago/Turabian StyleGómez-Melero, Sara, and Javier Caballero-Villarraso. 2023. "CCR6 as a Potential Target for Therapeutic Antibodies for the Treatment of Inflammatory Diseases" Antibodies 12, no. 2: 30. https://doi.org/10.3390/antib12020030
APA StyleGómez-Melero, S., & Caballero-Villarraso, J. (2023). CCR6 as a Potential Target for Therapeutic Antibodies for the Treatment of Inflammatory Diseases. Antibodies, 12(2), 30. https://doi.org/10.3390/antib12020030







