Aboveground Biomass Distribution in a Multi-Use Savannah Landscape in Southeastern Kenya: Impact of Land Use and Fences
Abstract
1. Introduction
2. Material and Methods
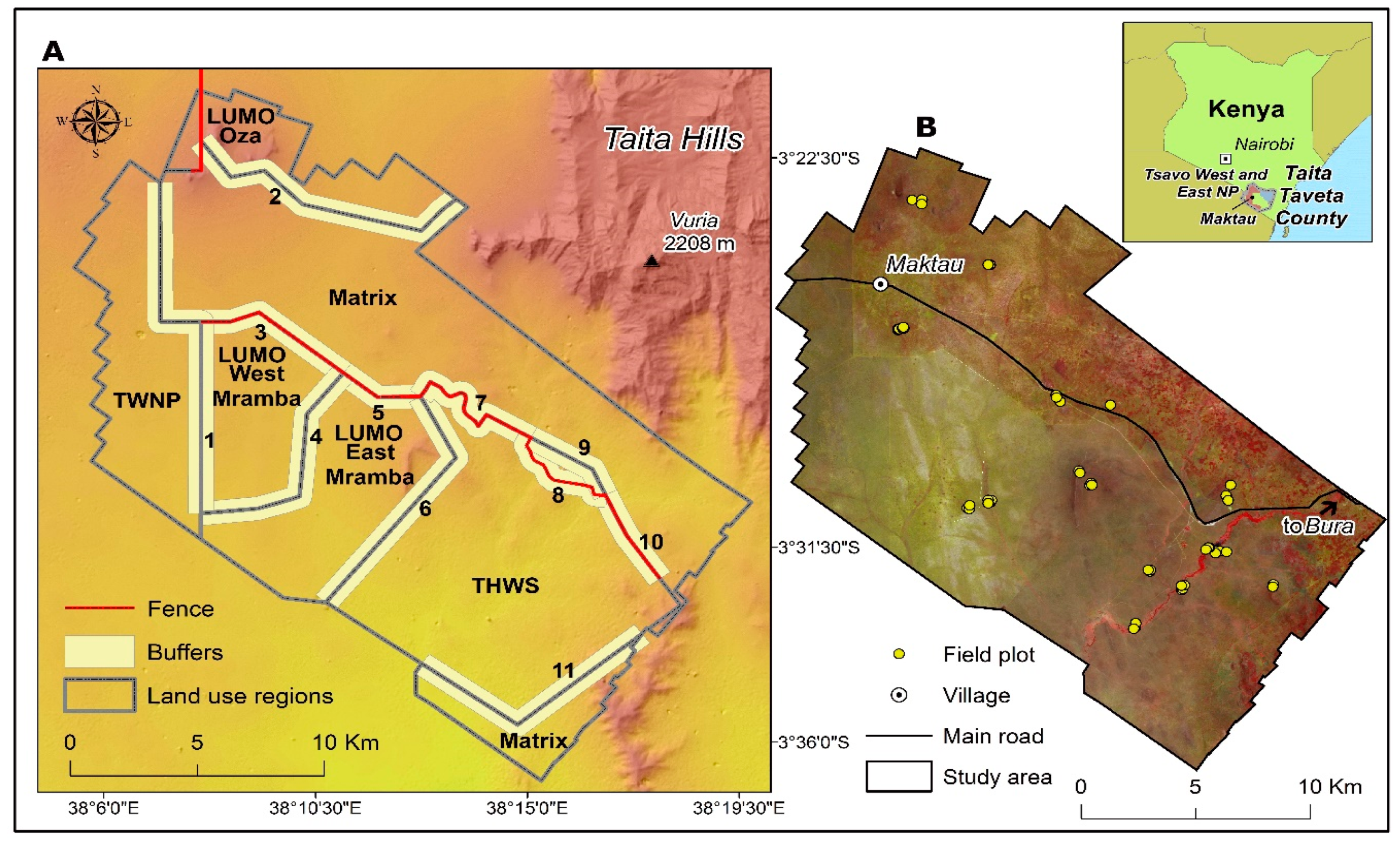
2.1. Study Area
2.2. Field Data
2.3. Airborne Laser Scanning Data (ALS) and Biomass Mapping
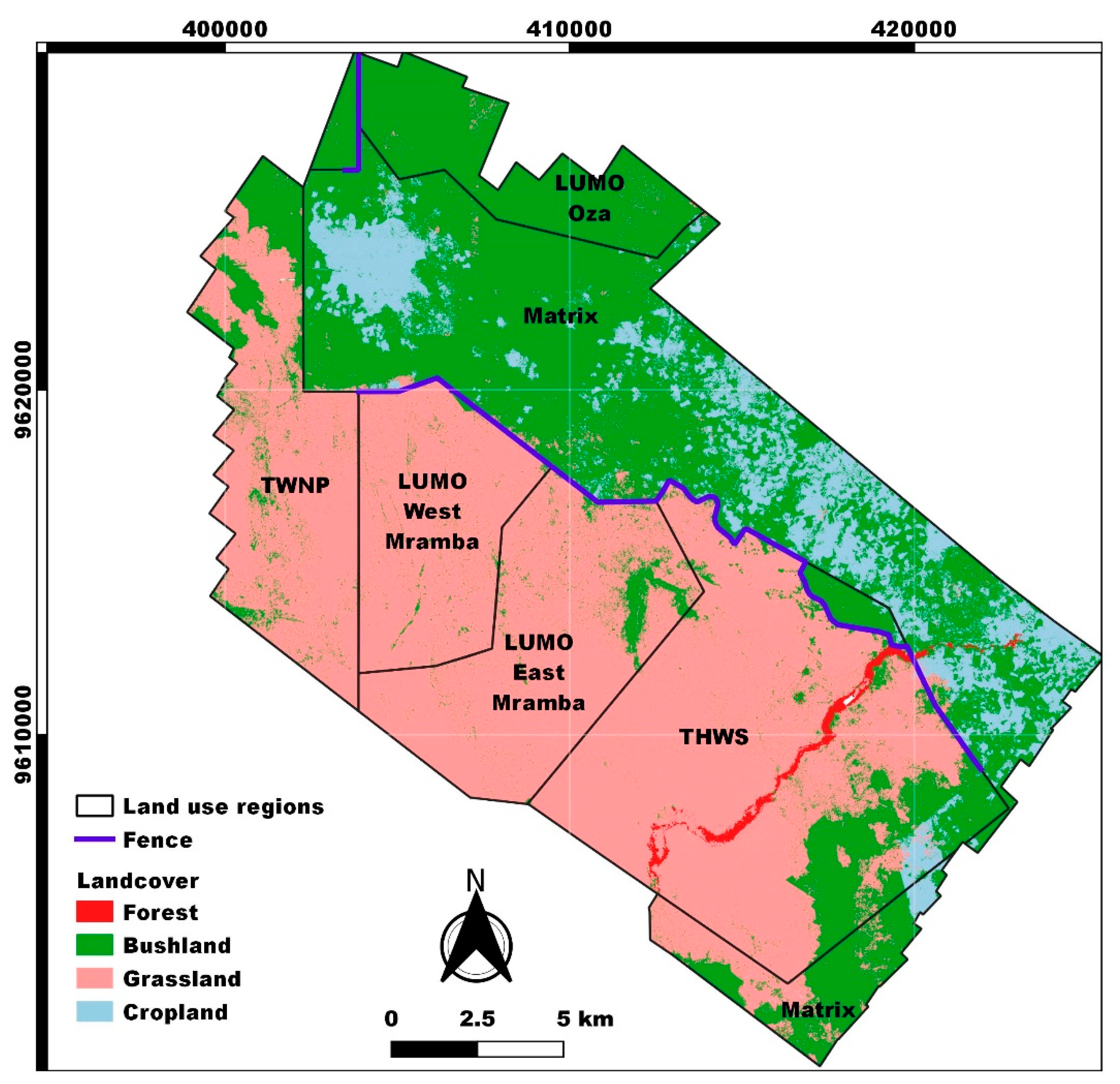
2.4. Satellite Imagery and Land Cover Mapping
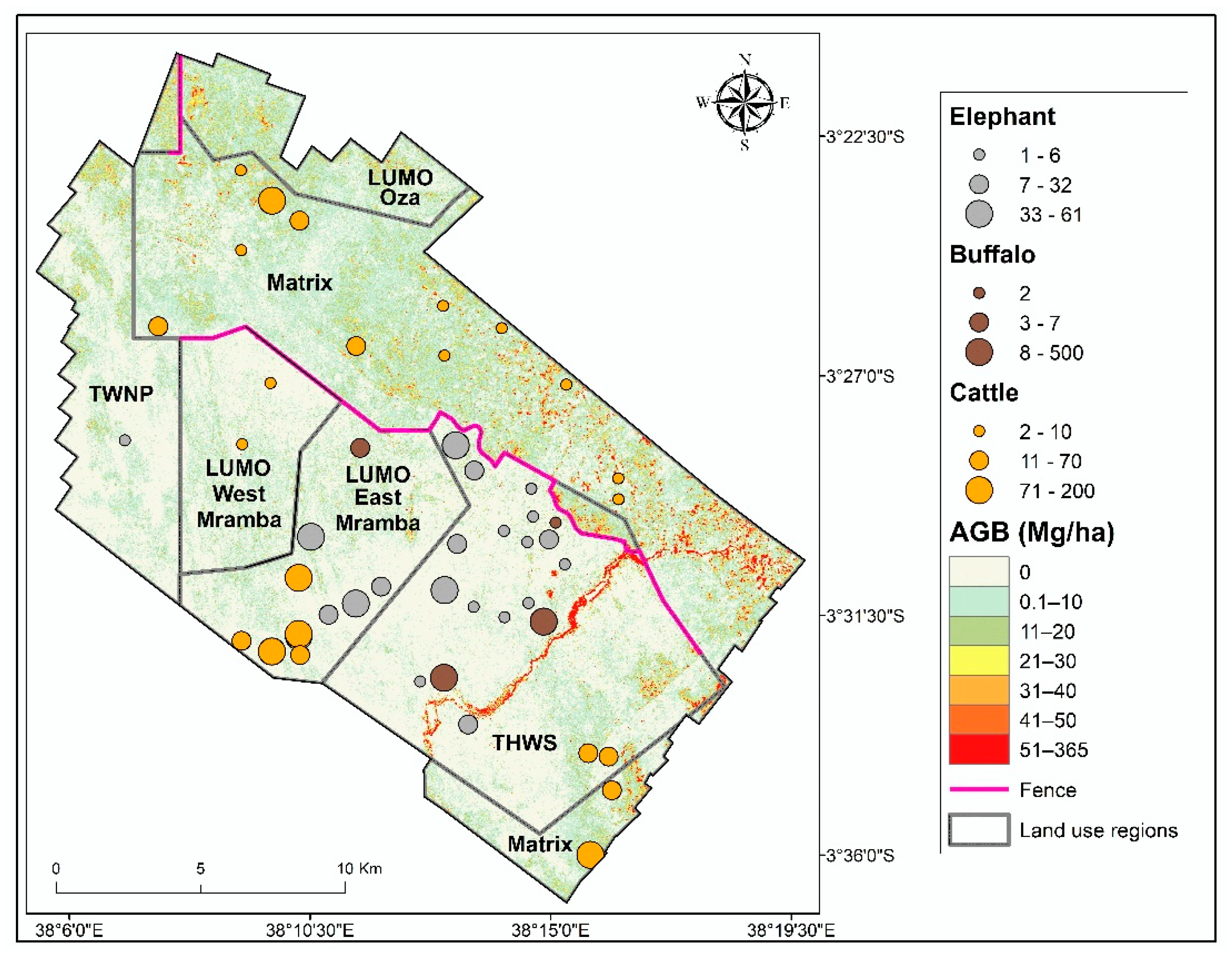
2.5. Wildlife and Livestock Data
2.6. Statistical Analyses of AGB Data
3. Results
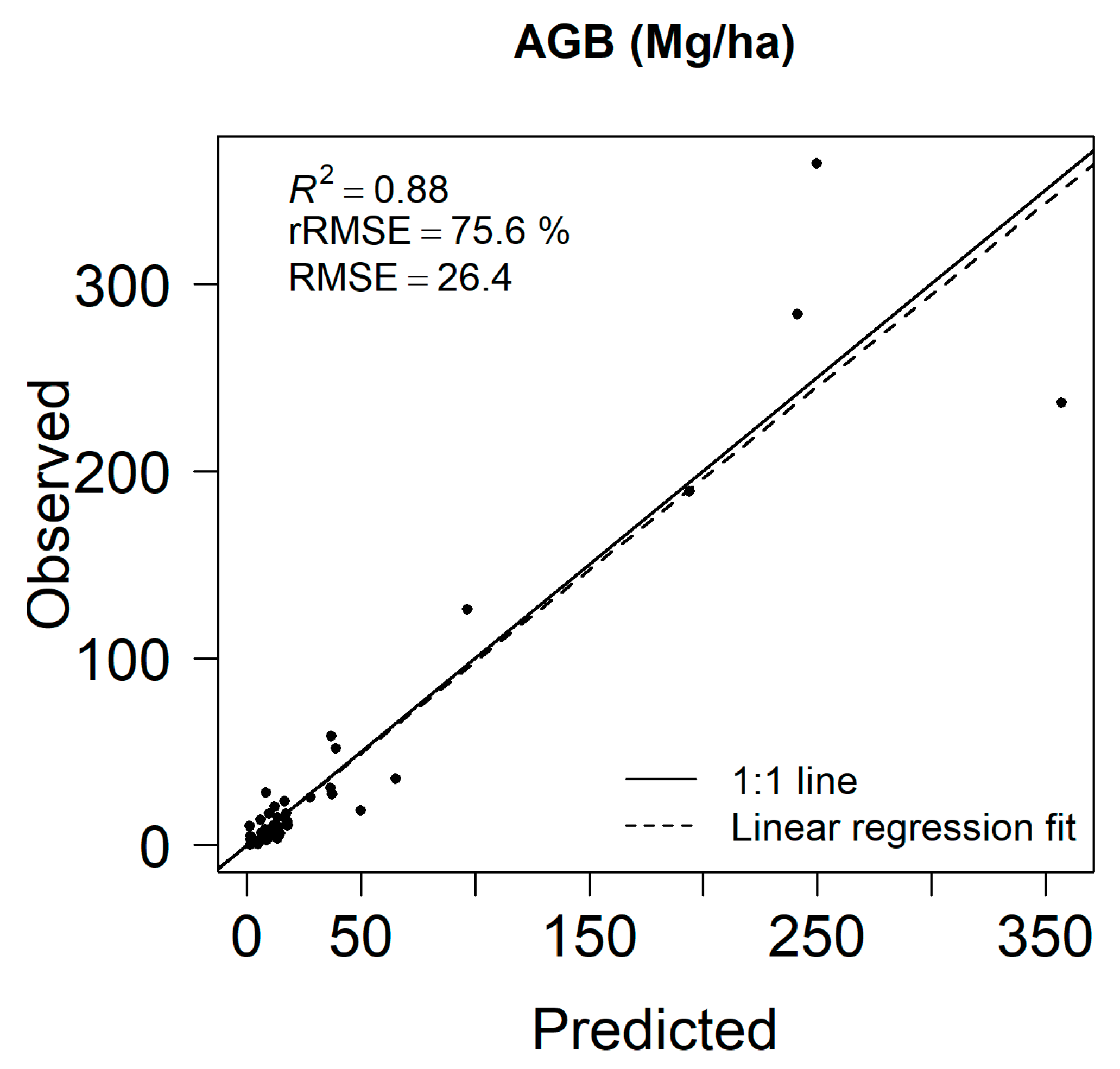
3.1. Aboveground Biomass Estimates and Map
3.2. Land Cover Classification
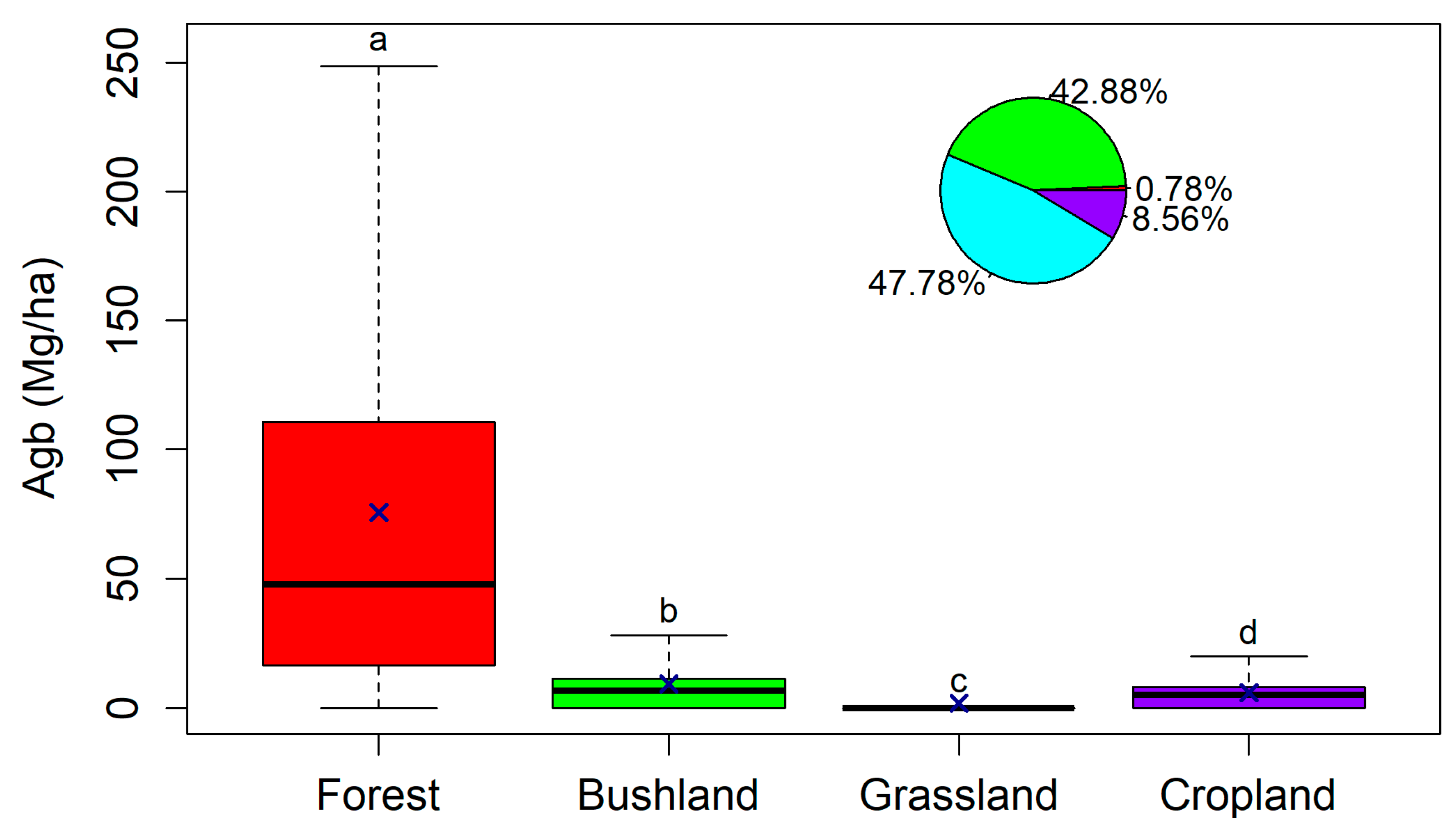
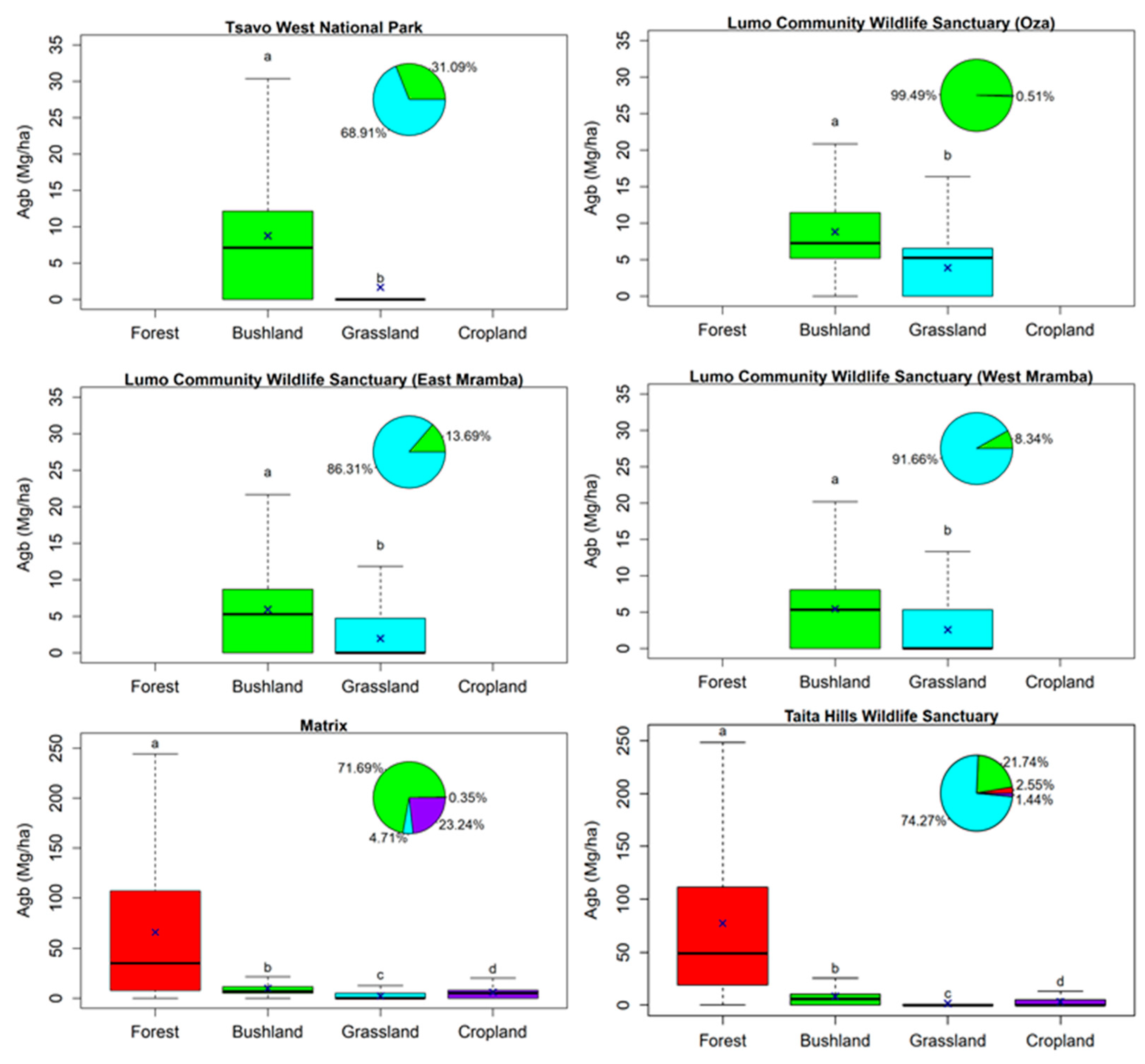
3.3. Effect of Land Cover and Land Use on Aboveground Biomass
3.4. Effect of Fences on Aboveground Biomass
4. Discussion
4.1. Remote Sensing—Based Biomass and Land Cover Maps
4.2. Effect of Land Cover and Land Use on Aboveground Biomass
4.3. Effect of Wildlife Fences on Biomass Distribution and Density
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Predictor | Description |
|---|---|
| H.p01, H.p05, H.p10, H.p20, H.p25, H.p30, H.p40, H.p50, H.p60, H.p70, H.p75, H.p80, H.p90, H.p95, H.p99 | 1st, 5th, 10th … and 99th percentile of return height > 3.5 m |
| H.L1, H.L2, H.L3, H.L4 | L-moments 1–4 of return height > 3.5 m |
| H.L.cv | L-moments coefficient of variation of return height > 3.5 m |
| H.L.skewness | L-moments skewness of return height > 3.5 m |
| H.L.kurtosis | L-moments kurtosis of return height > 3.5 m |
| H.max | Maximum of return height > 3.5 m |
| H.mean | Mean of return height > 3.5 m |
| H.min | Minimum of return height > 3.5 m |
| H.mode | Mode of return height > 3.5 m |
| H.cv | Coefficient of variation of return height > 3.5 m |
| H.v | Variance of return height > 3.5 m |
| H.stdev | Standard deviation of return height > 3,5 m |
| H.skewness | Skewness of return height > 3.5 m |
| H.kurtosis | Kurtosis of return height > 3.5 m |
| H.IQ | 75th percentile minus 25th percentile for cell |
| CC.first | First returns > 3.5 m/Total first returns * 100 |
| CC.all | All returns > 3.5 m/Total all returns * 100 |
| CC.all.first | All returns > 3.5 m/Total first returns * 100 |
| CC.first.mean | First returns above mean/Total first returns * 100 |
| CC.all.mean | All returns above mean/Total all returns * 100 |
| CC.all.mean.first | All returns above mean/Total first returns * 100 |
| CC.first.mode | First returns above mode/Total first returns * 100 |
| CC.all.mode | All returns above mode/Total all returns * 100 |
| CC.all.mode.first | All returns above mode/Total first returns * 100 |
| Forest | Bushland | Grassland | Cropland | Row Total | Producer’s Accuracy | |
|---|---|---|---|---|---|---|
| Forest | 47 | 5 | 0 | 0 | 52 | 95.91 |
| Bushland | 1 | 59 | 4 | 2 | 66 | 76.62 |
| Grassland | 0 | 6 | 100 | 3 | 109 | 92.59 |
| Cropland | 1 | 7 | 4 | 55 | 67 | 91.66 |
| Column total | 49 | 77 | 108 | 60 | 294 | |
| User’s accuracy | 90.38 | 89.39 | 91.74 | 82.08 |
References
- Lehmann, C.E.R.; Archibald, S.A.; Hoffmann, W.A.; Bond, W.J. Deciphering the distribution of the savanna biome. New Phytol. 2011, 191, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Kaya, F.; Bibi, F.; Žliobaite, I.; Eronen, J.T.; Hui, T.; Fortelius, M. The rise and fall of the Old World savannah fauna and the origins of the African savannah biome. Nat. Ecol. Evol. 2018, 2, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Cawthra, H. Emergence of the African savannah. Nat. Geosci. 2019, 12, 588–589. [Google Scholar] [CrossRef]
- Barlow, J.; França, F.; Gardner, T.A.; Hicks, C.C.; Lennox, G.D.; Berenguer, E.; Castello, L.; Economo, E.P.; Ferreira, J.; Guénard, B.; et al. The future of hyperdiverse tropical ecosystems. Nature 2018, 559, 517–526. [Google Scholar] [CrossRef]
- Gaston, G.; Brown, S.; Lorenzini, M.; Singh, K.D. State and change in carbon pools in the forests of tropical Africa. Glob. Chang. Biol. 1998, 4, 97–114. [Google Scholar] [CrossRef]
- Glenday, J. Carbon storage and emissions offset potential in an African dry forest, the Arabuko-Sokoke Forest, Kenya. Environ. Monit. Assess. 2008, 142, 85–95. [Google Scholar] [CrossRef]
- Valentini, R.; Arneth, A.; Bombelli, A.; Castaldi, S.; Cazzolla Gatti, R.; Chevallier, F.; Ciais, P.; Grieco, E.; Hartmann, J.; Henry, M.; et al. A full greenhouse gases budget of Africa: Synthesis, uncertainties, and vulnerabilities. Biogeosciences 2014, 11, 381–407. [Google Scholar] [CrossRef]
- Pellikka, P.; Hakala, E. Climate change. In Megatreds in Africa; Vastapuu, L., Mattlin, M., Hakala, E., Pellikka, P., Eds.; Ministry for Foreign Affairs of Finland: Helsinki, Finland, 2019; pp. 7–13. [Google Scholar]
- Brink, A.B.; Eva, H.D. Monitoring 25 years of land cover change dynamics in Africa: A sample based remote sensing approach. Appl. Geogr. 2009, 29, 501–512. [Google Scholar] [CrossRef]
- Devine, A.P.; McDonald, R.A.; Quaife, T.; Maclean, I.M.D. Determinants of woody encroachment and cover in African savannas. Oecologia 2017, 183, 939–951. [Google Scholar] [CrossRef]
- Stevens, N.; Lehmann, C.E.R.; Murphy, B.P.; Durigan, G. Savanna woody encroachment is widespread across three continents. Glob. Chang. Biol. 2017, 23, 235–244. [Google Scholar] [CrossRef]
- Venter, Z.S.; Cramer, M.D.; Hawkins, H.J. Drivers of woody plant encroachment over Africa. Nat. Commun. 2018, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mitchard, E.T.A.; Flintrop, C.M. Woody encroachment and forest degradation in sub-Saharan Africa’s woodlands and savannas 1982–2006. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 1–7. [Google Scholar] [CrossRef] [PubMed]
- DeVries, J. Shrublands in California: Literature Review and Research Needed for Management; California Water Resources Center, University of California: Davis, CA, USA, 1984. [Google Scholar]
- Barnes, M.D.; Craigie, I.D.; Harrison, L.B.; Geldmann, J.; Collen, B.; Whitmee, S.; Balmford, A.; Burgess, N.D.; Brooks, T.; Hockings, M.; et al. Wildlife population trends in protected areas predicted by national socio-economic metrics and body size. Nat. Commun. 2016, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Myers, N. National parks in Savannah Africa. Science 1972, 178, 1255–1263. [Google Scholar] [CrossRef]
- Pitman, R.T.; Fattebert, J.; Williams, S.T.; Williams, K.S.; Hill, R.A.; Hunter, L.T.B.; Slotow, R.; Balme, G.A. The Conservation Costs of Game Ranching. Conserv. Lett. 2017, 10, 402–412. [Google Scholar] [CrossRef]
- Craigie, I.D.; Baillie, J.E.M.; Balmford, A.; Carbone, C.; Collen, B.; Green, R.E.; Hutton, J.M. Large mammal population declines in Africa’s protected areas. Biol. Conserv. 2010, 143, 2221–2228. [Google Scholar] [CrossRef]
- Wilmshurst, J.F.; Fryxell, J.M.; Colucci, P.E. What constrains daily intake in Thomson’s gazelles? Ecology 1999, 80, 2338–2347. [Google Scholar] [CrossRef]
- Fynn, R.W.S.; Augustine, D.J.; Peel, M.J.S.; de Garine-Wichatitsky, M. Strategic management of livestock to improve biodiversity conservation in African savannahs: A conceptual basis for wildlife-livestock coexistence. J. Appl. Ecol. 2016, 53, 388–397. [Google Scholar] [CrossRef]
- Peters, M.K.; Hemp, A.; Appelhans, T.; Becker, J.N.; Behler, C.; Classen, A.; Detsch, F.; Ensslin, A.; Ferger, S.W.; Frederiksen, S.B.; et al. Climate–land-use interactions shape tropical mountain biodiversity and ecosystem functions. Nature 2019, 568, 88–92. [Google Scholar] [CrossRef]
- Niskanen, L. Situation Analysis—Conservation Areas & Species Diversity; Conservation Areas and Species Diversity Programme, IUCN Eastern and Southern Africa Programme Office: Gland, Switzerland, 2011. [Google Scholar]
- Vento, E. Divergent Objectives of Protected Area Management: Impact on Territorial Tourism Development in Taita Taveta County, Kenya. Master’s Thesis, University of Helsinki, Helsinki, Finland, October 2017. [Google Scholar]
- Bond, W.J.; Loffell, D. Introduction of giraffe changes acacia distribution in a South African savanna. Afr. J. Ecol. 2001, 39, 286–294. [Google Scholar] [CrossRef]
- Bergstorm, R. Browse characteristics and impact of browsing on trees and shrubs in African savannas. J. Veg. Sci. 1992, 3, 315–324. [Google Scholar] [CrossRef]
- Schmitz, O.J. Herbivory from Individuals Herbivory from Individuals to Ecosystems. Annu. Rev. Ecol. Evol. Syst. 2016, 39, 133–152. [Google Scholar] [CrossRef]
- Augustine, D.J.; McNaughton, S.J. Interactive effects of ungulate herbivores, soil fertility, and variable rainfall on ecosystem processes in a semi-arid savanna. Ecosystems 2006, 9, 1242–1256. [Google Scholar] [CrossRef]
- Vesala, R.; Harjuntausta, A.; Hakkarainen, A.; Rönnholm, P.; Pellikka, P.; Rikkinen, J. Termite mound architecture regulates nest temperature and correlates with species identities of symbiotic fungi. PeerJ 2019, 2019, 1–20. [Google Scholar] [CrossRef]
- Vesala, R.; Niskanen, T.; Liimatainen, K.; Boga, H.; Pellikka, P.; Rikkinen, J. Diversity of fungus-growing termites (Macrotermes) and their fungal symbionts (Termitomyces) in the semiarid Tsavo Ecosystem, Kenya. Biotropica 2017, 49, 402–412. [Google Scholar] [CrossRef]
- Massey, A.L.; King, A.A.; Foufopoulos, J. Fencing protected areas: A long-term assessment of the effects of reserve establishment and fencing on African mammalian diversity. Biol. Conserv. 2014, 176, 162–171. [Google Scholar] [CrossRef]
- Xu, L.; Nie, Y.; Chen, B.; Xin, X.; Yang, G.; Xu, D.; Ye, L. Effects of fence enclosure on vegetation community characteristics and productivity of a degraded temperate meadow steppe in Northern China. Appl. Sci. 2020, 10, 2952. [Google Scholar] [CrossRef]
- Nagaike, T.; Ohkubo, E.; Hirose, K. Vegetation Recovery in Response to the Exclusion of Grazing by Sika Deer (Cervus nippon) in Seminatural Grassland on Mt. Kushigata, Japan. ISRN Biodivers. 2014, 2014, 1–6. [Google Scholar] [CrossRef]
- Packer, C.; Loveridge, A.; Canney, S.; Caro, T.; Garnett, S.T.; Pfeifer, M.; Zander, K.K.; Swanson, A.; MacNulty, D.; Balme, G.; et al. Conserving large carnivores: Dollars and fence. Ecol. Lett. 2013, 16, 635–641. [Google Scholar] [CrossRef]
- Li, Q.; Zhou, D.W.; Jin, Y.H.; Wang, M.L.; Song, Y.T.; Li, G. Di Effects of fencing on vegetation and soil restoration in a degraded alkaline grassland in northeast China. J. Arid Land 2014, 6, 478–487. [Google Scholar] [CrossRef]
- Pekor, A.; Miller, J.R.B.; Flyman, M.V.; Kasiki, S.; Kesch, M.K.; Miller, S.M.; Uiseb, K.; van der Merve, V.; Lindsey, P.A. Fencing Africa’s protected areas: Costs, benefits, and management issues. Biol. Conserv. 2019, 229, 67–75. [Google Scholar] [CrossRef]
- Erb, K.H.; Kastner, T.; Plutzar, C.; Bais, A.L.S.; Carvalhais, N.; Fetzel, T.; Gingrich, S.; Haberl, H.; Lauk, C.; Niedertscheider, M.; et al. Unexpectedly large impact of forest management and grazing on global vegetation biomass. Nature 2018, 553, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Lynch, J.; Maslin, M.; Balzter, H.; Sweeting, M. Choose satellites to monitor deforestation. Nature 2013, 496, 293–294. [Google Scholar] [CrossRef] [PubMed]
- McNicol, I.M.; Ryan, C.M.; Mitchard, E.T.A. Carbon losses from deforestation and widespread degradation offset by extensive growth in African woodlands. Nat. Commun. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Saatchi, S.S.; Shapiro, A.; Meyer, V.; Ferraz, A.; Yang, Y.; Bastin, J.F.; Banks, N.; Boeckx, P.; Verbeeck, H.; et al. Spatial Distribution of Carbon Stored in Forests of the Democratic Republic of Congo. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Kumar, L.; Mutanga, O. Remote sensing of above-ground biomass. Remote Sens. 2017, 9, 935. [Google Scholar] [CrossRef]
- Karlson, M.; Ostwald, M.; Reese, H.; Sanou, J.; Tankoano, B.; Mattsson, E. Mapping tree canopy cover and aboveground biomass in Sudano-Sahelian woodlands using Landsat 8 and random forest. Remote Sens. 2015, 7, 10017–10041. [Google Scholar] [CrossRef]
- Gorelick, N.; Hancher, M.; Dixon, M.; Ilyushchenko, S.; Thau, D.; Moore, R. Google Earth Engine: Planetary-scale geospatial analysis for everyone. Remote Sens. Environ. 2017, 202, 18–27. [Google Scholar] [CrossRef]
- Zolkos, S.G.; Goetz, S.J.; Dubayah, R. A meta-analysis of terrestrial aboveground biomass estimation using lidar remote sensing. Remote Sens. Environ. 2013, 128, 289–298. [Google Scholar] [CrossRef]
- Egberth, M.; Nyberg, G.; Næsset, E.; Gobakken, T.; Mauya, E.; Malimbwi, R.; Katani, J.; Chamuya, N.; Bulenga, G.; Olsson, H. Combining airborne laser scanning and Landsat data for statistical modeling of soil carbon and tree biomass in Tanzanian Miombo woodlands. Carbon Balance Manag. 2017, 12. [Google Scholar] [CrossRef]
- Heiskanen, J.; Adhikari, H.; Piiroinen, R.; Packalen, P.; Pellikka, P.K.E. Do airborne laser scanning biomass prediction models benefit from Landsat time series, hyperspectral data or forest classification in tropical mosaic landscapes? Int. J. Appl. Earth Obs. Geoinf. 2019, 81, 176–185. [Google Scholar] [CrossRef]
- Kenya National Bureau of Statistic (KNBS). Kenya Population and Housing Census; KNBS: Nairobi, Kenya, 2019; Volume I, ISBN 9789966102096.
- County Integrated Development Plan 2018–2022; County Government of Taita Taveta: Taita Taveta County, Kenya, 2018.
- Pellikka, P.K.E.; Clark, B.J.F.; Gosa, A.G.; Himberg, N.; Hurskainen, P.; Maeda, E.; Mwang’ombe, J.; Omoro, L.M.A.; Siljander, M. Chapter 13—Agricultural expansion and its consequences in the Taita Hills, Kenya. In Developments in Earth Surface Processes; Paolo Paron, D.O.O., Christian Thine, O., Eds.; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Tuure, J.; Korpela, A.; Hautala, M.; Rautkoski, H.; Hakojärvi, M.; Mikkola, H.; Duplissy, J.; Pellikka, P.; Petäjä, T.; Kulmala, M.; et al. Comparing plastic foils for dew collection: Preparatory laboratory-scale method and field experiment in Kenya. Biosyst. Eng. 2020, 196, 145–158. [Google Scholar] [CrossRef]
- Räsänen, M.; Aurela, M.; Vakkari, V.; Beukes, J.; Tuovinen, J.-P.; Josipovic, M.; Siebert, S.; Laurila, T.; Kulmala, M.; Laakso, L.; et al. The effect of rainfall amount and timing on annual transpiration in grazed savanna grassland. Hydrol. Earth Syst. Sci. Discuss. 2020, 1–31. [Google Scholar] [CrossRef]
- Smith, R.J.; Kasiki, S.M. A Spatial Analysis of Human-Elephant Conflict in the Tsavo Ecosystem. Kenya; African Elephant Specialist Group, Human-Elephant Conflict Task Force of IUCN: Gland, Switzerland, 2000. [Google Scholar]
- Ogutu, J.O.; Piepho, H.P.; Said, M.Y.; Ojwang, G.O.; Njino, L.W.; Kifugo, S.C.; Wargute, P.W. Extreme wildlife declines and concurrent increase in livestock numbers in Kenya: What are the causes? PLoS ONE 2016, 11, 1–46. [Google Scholar] [CrossRef] [PubMed]
- Ogutu, J.; Piepho, H.; Kuloba, B.; Kanga, E. Wildlife population dynamics in human-dominated landscapes under community-based conservation: The example of Nakuru Wildlife Conservancy, Kenya. PLoS ONE 2017, 12. [Google Scholar] [CrossRef]
- Muteti, D.; Maloba, M. Taita Hills Wildlife Sanctuary: Dry season total ground wildlife census. 2013; unpublished. [Google Scholar]
- Ngene, S.; Lala, F.; Nzisa, M.; Kimitei, K.; Mukeka, J.; Kiambi, J.; Davidson, Z.; Bakari, S.; Lyimo, E.; Khayale, C.; et al. Aerial Total Count of Elephants, Buffalo and Giraffe in the Tsavo-Mkomazi Ecosystem; Kenya Wildlife Service, Kenya and Tanzania Wildlife Research Institute: Arusha, Tanzania, 2017. [Google Scholar]
- Vågen, T.-G.; Winowiecki, L.; Walsh, M.G.; Tamene Desta, L.; Tondoh, J.E. The Land Degradation Surveillance Framework (LDSF): Field Guide; World Agroforestry Centre (ICRAF): Nairobi, Kenya, 2010. [Google Scholar]
- Chave, J.; Réjou-Méchain, M.; Búrquez, A.; Chidumayo, E.; Colgan, M.S.; Delitti, W.B.C.; Duque, A.; Eid, T.; Fearnside, P.M.; Goodman, R.C.; et al. Improved allometric models to estimate the aboveground biomass of tropical trees. Glob. Chang. Biol. 2014, 20. [Google Scholar] [CrossRef]
- Réjou-Méchain, M.; Tanguy, A.; Piponiot, C.; Chave, J.; Hérault, B. Biomass: An R Package for Estimating Above-Ground Biomass and Its Uncertainty in Tropical Forests. Methods Ecol. Evol. 2017, 8, 1163–1167. [Google Scholar] [CrossRef]
- Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria; Available online: https://www.R-project.org/ (accessed on 11 August 2018).
- Conti, G.; Gorné, L.D.; Zeballos, S.R.; Lipoma, M.L.; Gatica, G.; Kowaljow, E.; Whitworth-Hulse, J.I.; Cuchietti, A.; Poca, M.; Pestoni, S.; et al. Developing allometric models to predict the individual aboveground biomass of shrubs worldwide. Glob. Ecol. Biogeogr. 2019, 28, 961–975. [Google Scholar] [CrossRef]
- Paul, K.I.; Roxburgh, S.H.; Chave, J.; England, J.R.; Zerihun, A.; Specht, A.; Lewis, T.; Bennett, L.T.; Baker, T.G.; Adams, M.A.; et al. Testing the generality of above-ground biomass allometry across plant functional types at the continent scale. Glob. Chang. Biol. 2016, 22, 2106–2124. [Google Scholar] [CrossRef]
- McGaughey, R. FUSION/LDV: Software for LIDAR Data Analysis and Visualization; US Department of Agriculture, Forest Service, Pacific Northwest Research Station: Seattle, WA, USA, 2016.
- Adhikari, H.; Valbuena, R.; Pellikka, P.K.E.; Heiskanen, J. Mapping forest structural heterogeneity of tropical montane forest remnants from airborne laser scanning and Landsat time series. Ecol. Indic. 2020, 108, 105739. [Google Scholar] [CrossRef]
- Package ‘Leaps’. Available online: https://cran.r-project.org/web/packages/leaps/leaps.pdf (accessed on 16 January 2020).
- Adhikari, H.; Heiskanen, J.; Siljander, M.; Maeda, E.; Heikinheimo, V.; Pellikka, P.K.E. Determinants of aboveground biomass across an afromontane landscape Mosaic in Kenya. Remote Sens. 2017, 9, 827. [Google Scholar] [CrossRef]
- Rouse, J.W.; Hass, R.H.; Schell, J.A.; Deering, D.W. Monitoring vegetation systems in the Great Plains with ERTS. Third Earth Resour. Technol. Satell. Symp. 1973, 1, 309–317. [Google Scholar]
- Huete, A.R.; Liu, H.Q.; Batchily, K.; Van Leeuwen, W. A comparison of vegetation indices over a global set of TM images for EOS-MODIS. Remote Sens. Environ. 1997, 59, 440–451. [Google Scholar] [CrossRef]
- Jiang, Z.; Huete, A.R.; Didan, K.; Miura, T. Development of a two-band enhanced vegetation index without a blue band. Remote Sens. Environ. 2008, 112, 3833–3845. [Google Scholar] [CrossRef]
- Serrano, L.; Ustin, S.L.; Roberts, D.A.; Gamon, J.A.; Peñuelas, J. Deriving water content of chaparral vegetation from AVIRIS data. Remote Sens. Environ. 2000, 74, 570–581. [Google Scholar] [CrossRef]
- Clevers, J.G.P.W.; Gitelson, A.A. Remote estimation of crop and grass chlorophyll and nitrogen content using red-edge bands on sentinel-2 and-3. Int. J. Appl. Earth Obs. Geoinf. 2013, 23, 344–351. [Google Scholar] [CrossRef]
- Vågen, T.-G.; Winowiecki, L.A.; Tamene Desta, L.; Tondoh, J.E. Land Degradation Surveillance Framework (LDSF)—Field Guide; World Agroforestry Center (ICRAF): Nairobi, Kenya, 2013. [Google Scholar]
- Breiman, L.; Friedman, J.H.; Stone, C.J.S.; Olshen, R.A. Classification and Regression Trees, 1st ed.; Wadsworth & Brooks/Cole Advanced Books & Software: Monterey, CA, USA, 1984; 358p. [Google Scholar]
- Ngene, S.; Njumbi, S.; Nzisa, M.; Kimitei, K.; Mukeka, J.; Muya, S.; Ihwagi, F.; Omondi, P. Status and trends of the elephant population in the Tsavo–Mkomazi ecosystem. Pachyderm 2013, 53, 38–50. [Google Scholar]
- Nyangala, E.B. Livestock Officer, Mwatate Sub County, Taita Taveta County, Kenya, 2019; unpublished.
- Asner, G.P.; Mascaro, J.; Muller-Landau, H.C.; Vieilledent, G.; Vaudry, R.; Rasamoelina, M.; Hall, J.S.; van Breugel, M. A universal airborne LiDAR approach for tropical forest carbon mapping. Oecologia 2012, 168, 1147–1160. [Google Scholar] [CrossRef]
- Mascaro, J.; Detto, M.; Asner, G.P.; Muller-Landau, H.C. Evaluating uncertainty in mapping forest carbon with airborne LiDAR. Remote Sens. Environ. 2011, 115, 3770–3774. [Google Scholar] [CrossRef]
- Asner, G.P.; Flint Hughes, R.; Varga, T.A.; Knapp, D.E.; Kennedy-Bowdoin, T. Environmental and biotic controls over aboveground biomass throughout a tropical rain forest. Ecosystems 2009, 12, 261–278. [Google Scholar] [CrossRef]
- Chen, Q.; Vaglio Laurin, G.; Valentini, R. Uncertainty of remotely sensed aboveground biomass over an African tropical forest: Propagating errors from trees to plots to pixels. Remote Sens. Environ. 2015, 160, 134–143. [Google Scholar] [CrossRef]
- Gonzalez, P.; Asner, G.P.; Battles, J.J.; Lefsky, M.A.; Waring, K.M.; Palace, M. Forest carbon densities and uncertainties from Lidar, QuickBird, and field measurements in California. Remote Sens. Environ. 2010, 114, 1561–1575. [Google Scholar] [CrossRef]
- Asner, G.P.; Clark, J.K.; Mascaro, J.; Vaudry, R.; Chadwick, K.D.; Vieilledent, G.; Rasamoelina, M.; Balaji, A.; Kennedy-Bowdoin, T.; Maatoug, L.; et al. Human and environmental controls over aboveground carbon storage in Madagascar. Carbon Balance Manag. 2012, 7, 1–13. [Google Scholar] [CrossRef]
- Mauya, E.W.; Ene, L.T.; Bollandsås, O.M.; Gobakken, T.; Næsset, E.; Malimbwi, R.E.; Zahabu, E. Modelling aboveground forest biomass using airborne laser scanner data in the miombo woodlands of Tanzania. Carbon Balance Manag. 2015, 10, 28. [Google Scholar] [CrossRef] [PubMed]
- Heiskanen, J.; Liu, J.; Valbuena, R.; Aynekulu, E.; Packalen, P.; Pellikka, P. Remote sensing approach for spatial planning of land management interventions in West African savannas. J. Arid Environ. 2017, 140, 29–41. [Google Scholar] [CrossRef]
- Forkuor, G.; Benewinde Zoungrana, J.-B.; Dimobe, K.; Ouattara, B.; Vadrevu, K.P.; Tondoh, J.E. Above-ground biomass mapping in West African dryland forest using Sentinel-1 and 2 datasets—A case study. Remote Sens. Environ. 2020, 236, 111496. [Google Scholar] [CrossRef]
- Venter, F.J.; Naiman, R.J.; Biggs, H.C.; Pienaar, D.J. The evolution of conservation management philosophy: Science, environmental change and social adjustments in Kruger National Park. Ecosystems 2008, 11, 173–192. [Google Scholar] [CrossRef]
- Pellikka, P.K.E.; Heikinheimo, V.; Hietanen, J.; Schäfer, E.; Siljander, M.; Heiskanen, J. Impact of land cover change on aboveground carbon stocks in Afromontane landscape in Kenya. Appl. Geogr. 2018, 94, 178–189. [Google Scholar] [CrossRef]
- Vuorinne, I. Geoinformatics Assessing Agave Sisalana Biomass from Leaf to Plantation Using Field Measurements and Multispectral Satellite Imagery. Master’s Thesis, University of Helsinki, Helsinki, Finland, 2020. [Google Scholar]
- Davidson, E.A.; De Araüjo, A.C.; Artaxo, P.; Balch, J.K.; Brown, I.F.; Mercedes, M.M.; Coe, M.T.; Defries, R.S.; Keller, M.; Longo, M.; et al. The Amazon basin in transition. Nature 2012, 481, 321–328. [Google Scholar] [CrossRef]
- Paruelo, J.M.; Burke, I.C.; Lauenroth, W.K. Land-use impact on ecosystem functioning in eastern Colorado, USA. Glob. Chang. Biol. 2001, 7, 631–639. [Google Scholar] [CrossRef]
- Morgan, J.A.; Lecain, D.R.; Pendall, E.; Blumenthal, D.M.; Kimball, B.A.; Carrillo, Y.; Williams, D.G.; Heisler-White, J.; Dijkstra, F.A.; West, M. C4 grasses prosper as carbon dioxide eliminates desiccation in warmed semi-arid grassland. Nature 2011, 476, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Scheiter, S.; Higgins, S.I.; Beringer, J.; Hutley, L.B. Climate change and long-term fire management impacts on Australian savannas. New Phytol. 2015, 205, 1211–1226. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.F.; Bartholomew, D.C.; Amakobe, B.; Githiru, M. Environmental factors affecting the distribution of African elephants in the Kasigau wildlife corridor, SE Kenya. Afr. J. Ecol. 2018, 56, 244–253. [Google Scholar] [CrossRef]
- Abera, T.A.; Heiskanen, J.; Pellikka, P.K.E.; Adhikari, H.; Maeda, E.E. Climatic impacts of bushland to cropland conversion in Eastern Africa. Sci. Total Environ. 2020, 717, 137255. [Google Scholar] [CrossRef]
- Aalto, I. Assessing the Cooling Impact of Tree Canopies in an Intensively Modified Tropical Landscape. Master’s Thesis, University of Helsinki, Helsinki, Finland, 2020. [Google Scholar]
- Liedloff, A.C.; Cook, G.D. Modelling the effects of rainfall variability and fire on tree populations in an Australian tropical savanna with the Flames simulation model. Ecol. Modell. 2007, 201, 269–282. [Google Scholar] [CrossRef]
- Kenya Wildlife Service. Tsavo Conservation Area Management Plan 2008–2018; Kenya Wildlife Service: Nairobi, Kenya, 2008. [Google Scholar]
- Stoldt, M.; Göttert, T.; Mann, C.; Zeller, U. Transfrontier Conservation Areas and Human-Wildlife Conflict: The Case of the Namibian Component of the Kavango-Zambezi (KAZA) TFCA. Sci. Rep. 2020, 10, 1–16. [Google Scholar] [CrossRef]
- Durant, S.M.; Becker, M.S.; Creel, S.; Bashir, S.; Dickman, A.J.; Beudels-Jamar, R.C.; Lichtenfeld, L.; Hilborn, R.; Wall, J.; Wittemyer, G.; et al. Developing fencing policies for dryland ecosystems. J. Appl. Ecol. 2015, 52, 544–551. [Google Scholar] [CrossRef]
- Dupuis-Desormeaux, M.; Davidson, Z.; Pratt, L.; Mwololo, M.; MacDonald, S.E. Testing the effects of perimeter fencing and elephant exclosures on lion predation patterns in a Kenyan wildlife conservancy. PeerJ 2016, 2016, 1–19. [Google Scholar] [CrossRef]
- Hayward, M.W.; Kerley, G.I.H. Fencing for conservation: Restriction of evolutionary potential or a riposte to threatening processes? Biol. Conserv. 2009, 142, 1–13. [Google Scholar] [CrossRef]
- Ringma, J.L.; Wintle, B.; Fuller, R.A.; Fisher, D.; Bode, M. Minimizing species extinctions through strategic planning for conservation fencing. Conserv. Biol. 2017, 31, 1029–1038. [Google Scholar] [CrossRef]
- Soto-Shoender, J.R.; McCleery, R.A.; Monadjem, A.; Gwinn, D.C. The importance of grass cover for mammalian diversity and habitat associations in a bush encroached savanna. Biol. Conserv. 2018, 221, 127–136. [Google Scholar] [CrossRef]
- Staver, A.C.; Bond, W.J. Is there a “browse trap”? Dynamics of herbivore impacts on trees and grasses in an African savanna. J. Ecol. 2014, 102, 595–602. [Google Scholar] [CrossRef]
- Sankaran, M.; Augustine, D.J.; Ratnam, J. Native ungulates of diverse body sizes collectively regulate long-term woody plant demography and structure of a semi-arid savanna. J. Ecol. 2013, 101, 1389–1399. [Google Scholar] [CrossRef]
- Miller, G.R. The Effects of Mammalian Herbivores on Natural Regeneration of Upland, Native Woodland; Scottish Natural Heritage: Perth, Scotland, 2015. [Google Scholar]
- Leuthold, W. Changes in tree populations of Tsavo East National Park, Kenya. Afr. J. Ecol. 1977, 15, 61–69. [Google Scholar] [CrossRef]

| DBH Class | AGB (Mg/ha) | |||||
|---|---|---|---|---|---|---|
| Mean | Min | Max | SD | IQR | Median | |
| DBH > 5 cm | 42.15 | 0.28 | 364.04 | 85.41 | 20.94 | 7.91 |
| DBH 1–5 cm | 3.69 | 0.00 | 19.46 | 4.98 | 4.03 | 2.07 |
| DBH < 1 cm | 0.52 | 0.00 | 2.56 | 0.60 | 0.49 | 0.35 |
| Total | 38.02 | 0.00 | 364.54 | 78.27 | 21.56 | 10.04 |
| Animal | Land-Use Region (area) | ||||
|---|---|---|---|---|---|
| TWNP (48.92 km2) | LUMO East Mramba (47.24 km2) | LUMO West Mramba (33.92 km2) | THWS (101.50 km2) | Matrix (141.63 km2) | |
| Elephant | 5 (0.10) | 137 (2.90) | 0 (0) | 237 (2.33) | 0 (0) |
| Cattle | 0 (0) | 540 (11.43) | 20 (0.59) | 100 (0.99) | 623 (4.40) |
| Buffalo | 2 (0.04) | 7 (0.15) | 0 (0) | 802 (7.90) | 0 (0) |
| Land-Use Region | Land Cover | AGB (Mg/ha) | |||||
|---|---|---|---|---|---|---|---|
| Mean | Min | Max | SD | IQR | Median | ||
| LUMO Oza | Bushland | 8.8 | 0.0 | 82.3 | 7.9 | 6.3 | 7.3 |
| Grassland | 3.9 | 0.0 | 20.1 | 4.4 | 6.6 | 5.2 | |
| All | 8.8 | 0.0 | 82.3 | 7.9 | 6.2 | 7.2 | |
| LUMO East Mramba | Bushland | 5.9 | 0.0 | 106.2 | 7.6 | 8.7 | 5.3 |
| Grassland | 2.0 | 0.0 | 50.9 | 3.8 | 4.7 | 0.0 | |
| All | 2.4 | 0.0 | 106.2 | 4.6 | 5.1 | 0.0 | |
| LUMO West Mramba | Bushland | 5.4 | 0.0 | 104.4 | 6.3 | 8.1 | 5.3 |
| Grassland | 2.6 | 0.0 | 55.6 | 4.2 | 5.3 | 0.0 | |
| All | 2.6 | 0.0 | 104.4 | 4.4 | 5.4 | 0.0 | |
| THWS | Forest | 77.4 | 0.0 | 353.0 | 79.0 | 92.5 | 49.0 |
| Bushland | 8.1 | 0 | 159.3 | 11.9 | 10.1 | 5.5 | |
| Grassland | 1.4 | 0 | 237.3 | 4.2 | 0 | 0 | |
| Cropland | 3.0 | 0.0 | 100.3 | 7.0 | 5.1 | 0.0 | |
| All | 4.8 | 0.0 | 353.0 | 18.6 | 5.1 | 0.0 | |
| TWNP | Bushland | 8.8 | 0 | 71.8 | 8.3 | 12.2 | 7.1 |
| Grassland | 1.7 | 0 | 100.9 | 3.4 | 0 | 0 | |
| All | 3.8 | 0.0 | 100.9 | 6.3 | 6.2 | 0.0 | |
| Matrix | Forest | 66.0 | 0.0 | 346.7 | 73.8 | 99.3 | 35.0 |
| Bushland | 9.9 | 0.0 | 353 | 13.3 | 6.6 | 6.9 | |
| Grassland | 2.2 | 0.0 | 54.5 | 4.0 | 5.1 | 0.0 | |
| Cropland | 6.0 | 0.0 | 241.3 | 9.4 | 8.1 | 5.2 | |
| All | 8.9 | 0.0 | 353 | 13.5 | 10.5 | 6.3 | |
| All Land cover | Forest | 75.5 | 0.0 | 353.0 | 78.3 | 94.2 | 47.8 |
| Bushland | 9.2 | 0 | 353 | 11.9 | 11.2 | 6.7 | |
| Grassland | 1.8 | 0 | 237.3 | 4 | 0 | 0 | |
| Cropland | 5.8 | 0.0 | 241.3 | 9.3 | 7.9 | 5.1 | |
| All | 5.9 | 0.0 | 353.0 | 13.1 | 7.6 | 0.0 | |
| Side 1 | Side 2 | Fence | Percentage Zero AGB | Median for Non-Zero AGB | |||
|---|---|---|---|---|---|---|---|
| Side 1 | Side 2 | Side 1 | Side 2 | P Value | |||
| TWNP_1 | LUMO West Mramba_1 | No | 57.6 | 52.9 | 7.1 | 6.6 | <0.001 |
| LUMO Oza_2 | Matrix_2 | No | 20.7 | 17.0 | 8.2 | 8.2 | >0.05 |
| LUMO West Mramba_3 | Matrix_3 | Yes | 84.3 | 44.1 | 6.5 | 6.4 | >0.05 |
| LUMO East Mramba_4 | LUMO West Mramba_4 | No | 63.2 | 58.2 | 6.7 | 6.8 | >0.05 |
| Matrix_5 | LUMO East Mramba_5 | Yes | 32.6 | 62.6 | 7.2 | 7.2 | >0.05 |
| LUMO East Mramba_6 | THWS_6 | No | 85.9 | 84.9 | 6.5 | 6.9 | <0.001 |
| THWS_7 | Matrix_7 | Yes | 75.2 | 22.0 | 6.9 | 8.8 | <0.001 |
| THWS1_8 | THWS2_8 | Yes | 5.3 | 72.0 | 12.6 | 8.0 | <0.001 |
| Matrix_9 | THWS_9 | No | 31.0 | 10.6 | 9.1 | 10.0 | <0.001 |
| Matrix_10 | THWS_10 | Yes | 56.2 | 61.9 | 6.7 | 9.7 | <0.001 |
| THWS_11 | Matrix_11 | No | 69.9 | 56.4 | 6.6 | 6.8 | >0.05 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amara, E.; Adhikari, H.; Heiskanen, J.; Siljander, M.; Munyao, M.; Omondi, P.; Pellikka, P. Aboveground Biomass Distribution in a Multi-Use Savannah Landscape in Southeastern Kenya: Impact of Land Use and Fences. Land 2020, 9, 381. https://doi.org/10.3390/land9100381
Amara E, Adhikari H, Heiskanen J, Siljander M, Munyao M, Omondi P, Pellikka P. Aboveground Biomass Distribution in a Multi-Use Savannah Landscape in Southeastern Kenya: Impact of Land Use and Fences. Land. 2020; 9(10):381. https://doi.org/10.3390/land9100381
Chicago/Turabian StyleAmara, Edward, Hari Adhikari, Janne Heiskanen, Mika Siljander, Martha Munyao, Patrick Omondi, and Petri Pellikka. 2020. "Aboveground Biomass Distribution in a Multi-Use Savannah Landscape in Southeastern Kenya: Impact of Land Use and Fences" Land 9, no. 10: 381. https://doi.org/10.3390/land9100381
APA StyleAmara, E., Adhikari, H., Heiskanen, J., Siljander, M., Munyao, M., Omondi, P., & Pellikka, P. (2020). Aboveground Biomass Distribution in a Multi-Use Savannah Landscape in Southeastern Kenya: Impact of Land Use and Fences. Land, 9(10), 381. https://doi.org/10.3390/land9100381







