4.1. Stocking Levels and the Potential of Fallows to Support Forest Recovery
Overall, the tree stocking parameters for the forests in the study site demonstrate the typical extent of stock depletion of Tanzanian Miombo woodlands [
34]. In general, tree stocking levels decrease from forest patches to fallows to farms, as was expected, but the level of stocking itself, especially for tree fallows, was significant. We found that stocking levels for the forests under study were lower than for the sub-montane forests of the Uluguru Mountains [
35]. If woody material in tree fallows is no longer harvested, forest patches may fully recover with time. Other studies have noted a positive relationship between basal area and fallow age in other parts of the Eastern Arc Mountains [
13]. We posit that the longevity of fallows determines their tree species composition, tree size, and the colonization of land towards forest transition.
The longevity of tree fallows, in turn, may depend upon factors such as distance from residential areas, type of terrain, and perceived soil fertility. Communities indicated that the cycle of tree fallows lasts between 10 and 15 years. Cycle duration depends on landforms, such as hilltops, hillsides, and valleys, and on proximity to residential areas. Tree fallows on hilltops persist longer than those on hillsides, in valleys, and close to homesteads, perhaps because hilltops are far from most homesteads and feature difficult terrain and apparently low soil fertility.
The presence of leftovers of large trees on fallows and the proximity of forest patches guarantee the availability of seed sources dispersed by wind and animals, but germination and recruitment remain challenging. Tree regeneration in the Kolero sub-catchment fallow occurs mainly through asexual propagation as cut stumps and roots sprout. Farmers do not uproot cut stumps, so most of them survive. Tree seedlings that make it to the sapling stage flourish, but most become suppressed and die during their infant stage, while others fail to germinate in the first place. We observed the presence of long grasses in fallows, which impact the recruitment of tree species. The dominance of long grasses that are persistent and aggressive on the tree fallow floor deprive tree seedlings of the chance to flourish from propagation; a similar phenomenon has been observed in the East Usambara Mountains [
27]. What is more, wildfires commonly occur at the study site in the dry season as a result of fire-based swidden methods. Wildfires suppress seed germination, root sprouting, and seedling recruitment, which has also been observed in the tropical woodlands of southeast Angola [
36].
Even after trees mature, there are other barriers to forest transition. For instance, extraction of woody material from tree fallows remains a common practice through selective harvesting of sizeable and desirable trees according to their usefulness; people believe forests and fallows in farmland are open access, especially those forests that lack traditional and formal institutional protections, but this practice slows progress in forest transition [
37]. Small- to medium-sized trees are extracted for energy, building materials, and various other uses, while large trees are left on-site. We observed more recent tree cuts in fallows than in forests and on farms. In addition, most large trees either are varieties that have no immediate economic value in the area, such as
Sterculia appendiculata and
Bombax rhodognaphalon, or have become overgrown and unsuitable for timber due to heart rot.
Yet good land management practices, informed by climate-smart agriculture, may boost the forest transition process. For example, attempts were made between 2011–2014 to improve tree cover on farm in Kinole sub-catchment. A climate-smart agriculture project (
www.fao.org/in-action/micca/knowledge/climate-smart-agriculture/en/, accessed 10 December 2020) led to planting of estimated 110,000 trees including species such as
Grevillea robusta, Khaya anthotheca, Tectona grandis,
Acacia crassicarpa, and
Terminalia cattapa. This project might help explain the unusually sharp surge in tree cover that occurred in the assessment period of 2014–2020.
4.2. Shrinking Farms, Expanding Fallows
Since the 1930s, population growth in the Uluguru Mountains has been linked to agricultural expansion [
38]. According to the population census, the number of people has doubled from 1,753,362 in 2002 to 2,218,492 in 2012 in Morogoro Region, and 73% of this population resides in rural areas [
26]. Our observation, to the contrary, indicates that overall population growth in the Kolero sub-catchment and the Uluguru Mountains at large does not correspond to expanding farms. From 1995–2000, indeed, the area occupied by farms consistently declined (
Table 5), and the stable, unchanged portion of farmland also continuously dwindled (
Table 6). The data imply an unexpected trend of halted agricultural expansion and increased dynamism in land use.
This shrinkage of farms might be due to the increasing area under fallow. Projections from 1967 estimated that fallows in the Uluguru Mountains constitute 30% of the farmland area [
39]. From 1995–2020, however, fallow area covered more than 40% of farmland, indicating that traditional tree fallows occupy a notable share of land in Kolero sub-catchment. The importance of fallows is further exemplified by the land use change matrix, which shows increments of unchanged fallow land in 2007–2020, a clear indication that the expansion of farms is contained.
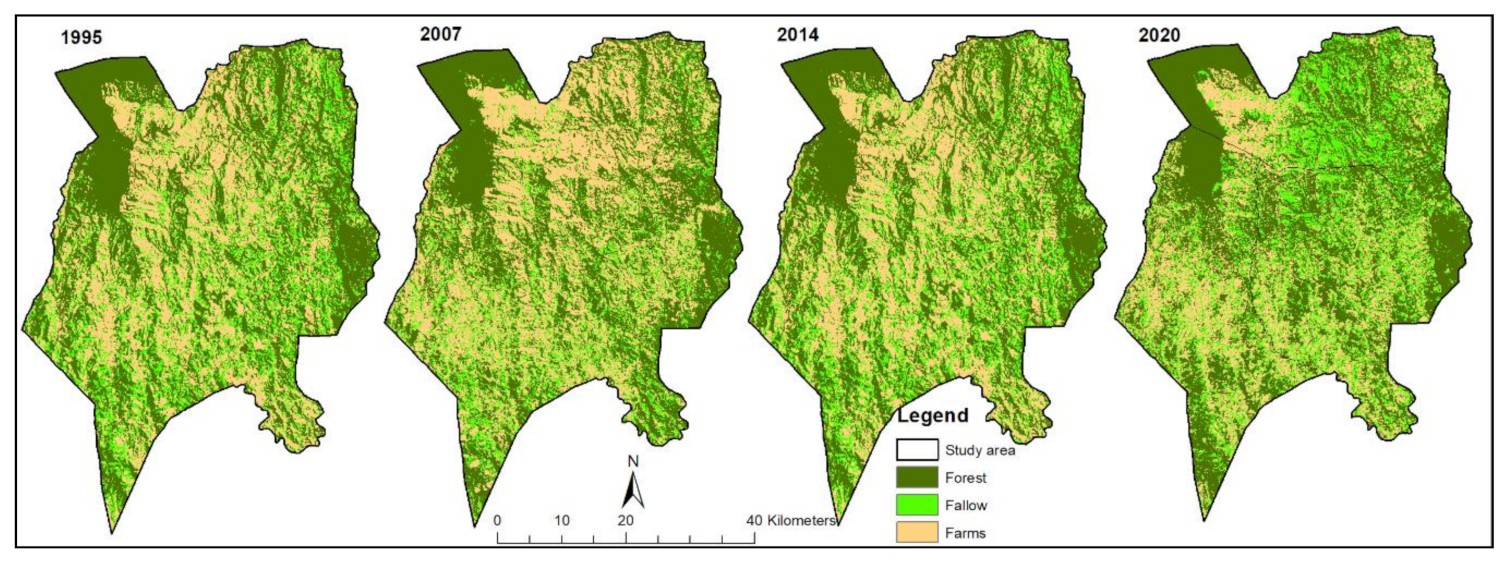
In recent years, meanwhile, forest cover loss has also slowed in the study area. From 1995–2007, forest cover loss was higher than in 2007–2014. Most recently, the period of 2014–2020 experienced forest cover gain (
Table 5), indicating a reduction in deforestation across the Kolero sub-catchment. Previous studies in the Uluguru Mountains highlighted similar trends of heightened deforestation prior to 2000 [
40], and reduced deforestation and/or forest recovery in the early 2000s [
41]. We speculate that the declining rate of forest loss in recent years may have arisen from a combination of factors including fewer forest disturbances, the diminishment of swidden, and the growing role of fallow in supplying forest produce.
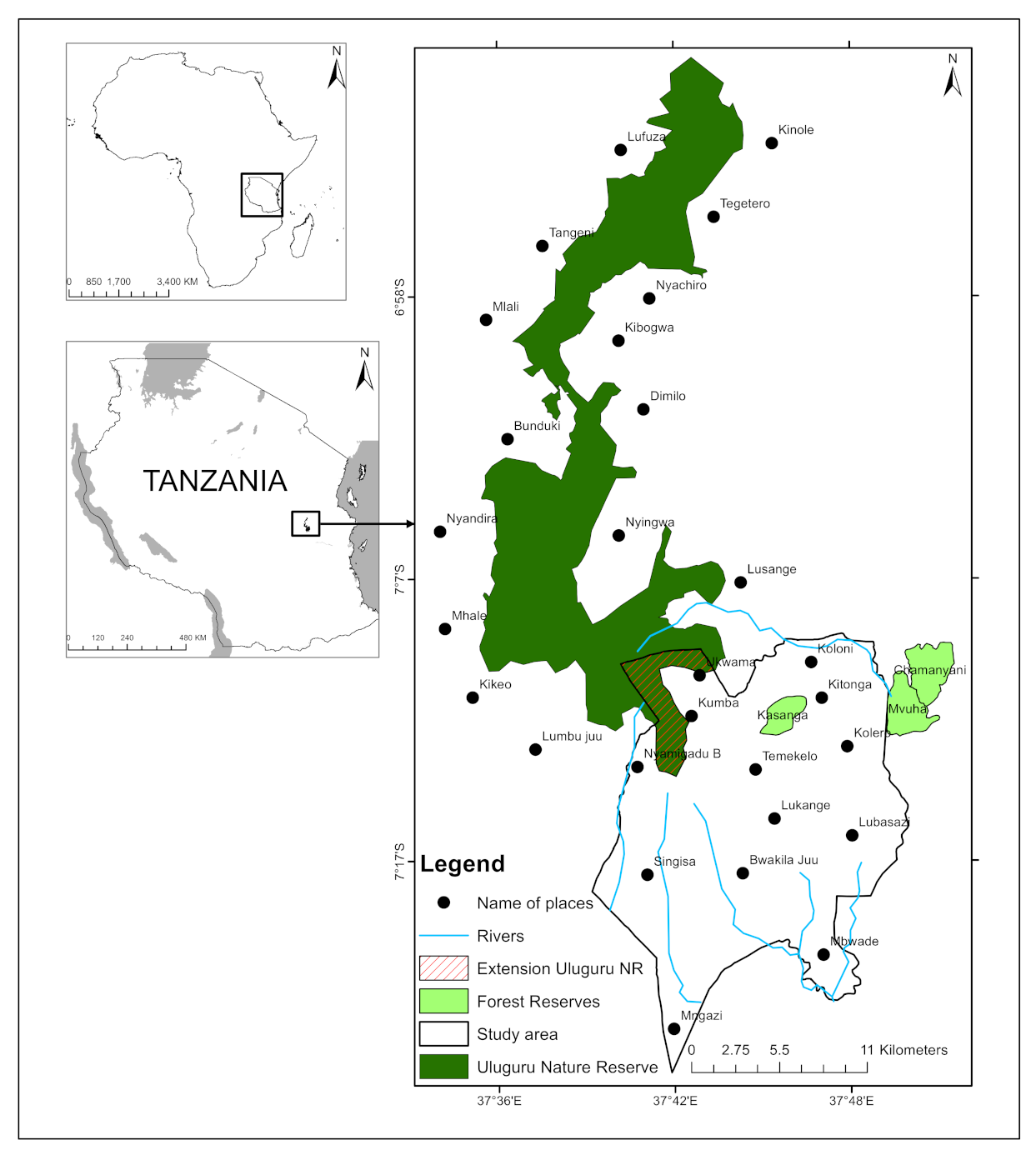
Accessibility from and proximity to villages may explain differences in forest cover changes because, as other studies have noted, there is often a relationship between accessibility and disturbances to forest vegetation [
42]. The two state-owned forests in the study area furnish a good example of this effect. From 1995–2020, an extension of the Uluguru Nature Reserve remained stable, whereas Kasanga Forest Reserve succumbed to degradation from 1995–2007 and later recovered from 2014–2020 (
Figure 2). The extension of Uluguru Nature Reserve is inaccessible, encompassing ridges with no roads, rough terrain, and deep valleys. It stands close to 1650 m above sea level, while the nearest villages, such as Mgata, Ukwama, and Longwe on the side of the Kolero sub-catchment, are located at 1250 m above sea level. Such physical barriers reduce and hinder human-induced disturbances. At 850 m above sea level, on the other hand, the Kasanga Forest Reserve is accessible from multiple directions and borders intensively cultivated farms, so it is prone to human-induced disturbances including fires.
Other positive factors may have influenced the expansion of fallow and increased tree cover. For instance, it seems that more farms were left as tree fallow and that the trend towards tree-based farming systems such as agroforestry has increased from 2007–2020. Between 2011 and 2014, conservation agriculture was promoted in the study site, but farmers did not view the returns as profitable [
43], which may have dampened adoption rates [
44]. However, we suggest that this effort may have increased awareness of agricultural intensification and so contributed to the reduction of swidden practices.
Traditional institutions are one of the key success factors in promoting community-led forest management in Tanzania [
10]. Sacred forests under community-based forest management (
Table 2) were found to be more protected and stable than forest patches that had no traditional significance. Despite their small size, sacred forests that are protected under traditional norms and customs provide important refugia for plant species, because access is limited to a few non-destructive utilizations. On the other hand, forest patches in farmland without community-based forest management arrangements faced excessive exploitation including conversion to other uses. Similarly, in other parts of Tanzania, traditionally protected forests have withstood exploitation pressure and remained intact [
45].
4.3. The Threat of Deforestation
The study area faces a grave trend toward fragmentation that if not checked will result in complete deforestation. Across the whole of the Eastern Arc Mountains, deforestation has caused habitat reduction and forest fragmentation that has impacted more than 77% of the original forest cover in the past 2000 years [
46]. At local and regional scales, forest fragmentation has caused a loss of biodiversity, continues to undermine forest transition in the Uluguru Mountains, and accelerates the extinction of important species such as birds [
47,
48].
The trend toward increasing forest fragmentation in the Kolero sub-catchment landscape is notable, defined by the increasing isolation of continuous forests into smaller patches (
Figure 3,
Table 7). Forest fragmentation is exhibited both in spatial and temporal patterns. Temporal fragmentation can lead either to perforation (subdivision) or attrition (shrinkage) of the forest. Our observations on fragmentation (
Table 7) conform with the tree inventory results (
Table 3), indicating low stocking levels typical of perforated forests. In other parts of the Eastern Arc Mountains, forest fragmentation has involved encroachment affecting both protected and non-protected forests [
49].
Forest fragmentation in the Kolero sub-catchment results from deforestation and forest degradation mainly due to anthropogenic factors such as expanding agricultural frontiers, excessive extraction of woody products, and fires. The scale and trends of forest fragmentation (
Figure 3,
Table 7) suggests the continuous decline of perforated forests throughout the assessment period of 1995–2020, especially in the eastern, western, and southern parts of the study site. Edge and small-core forests scattered across the study landscape also experienced shrinkage. Our field observations indicate that forests are not opened up in large-scale clearing but rather in small, incremental areas adjacent to existing farms. Similar observations were made previously of significant forest fragmentation in less dense forest classes in the Uluguru Mountains [
50].