Botanical Composition and Species Diversity of Arid and Desert Rangelands in Tataouine, Tunisia
Abstract
1. Introduction
2. Materials and Methods
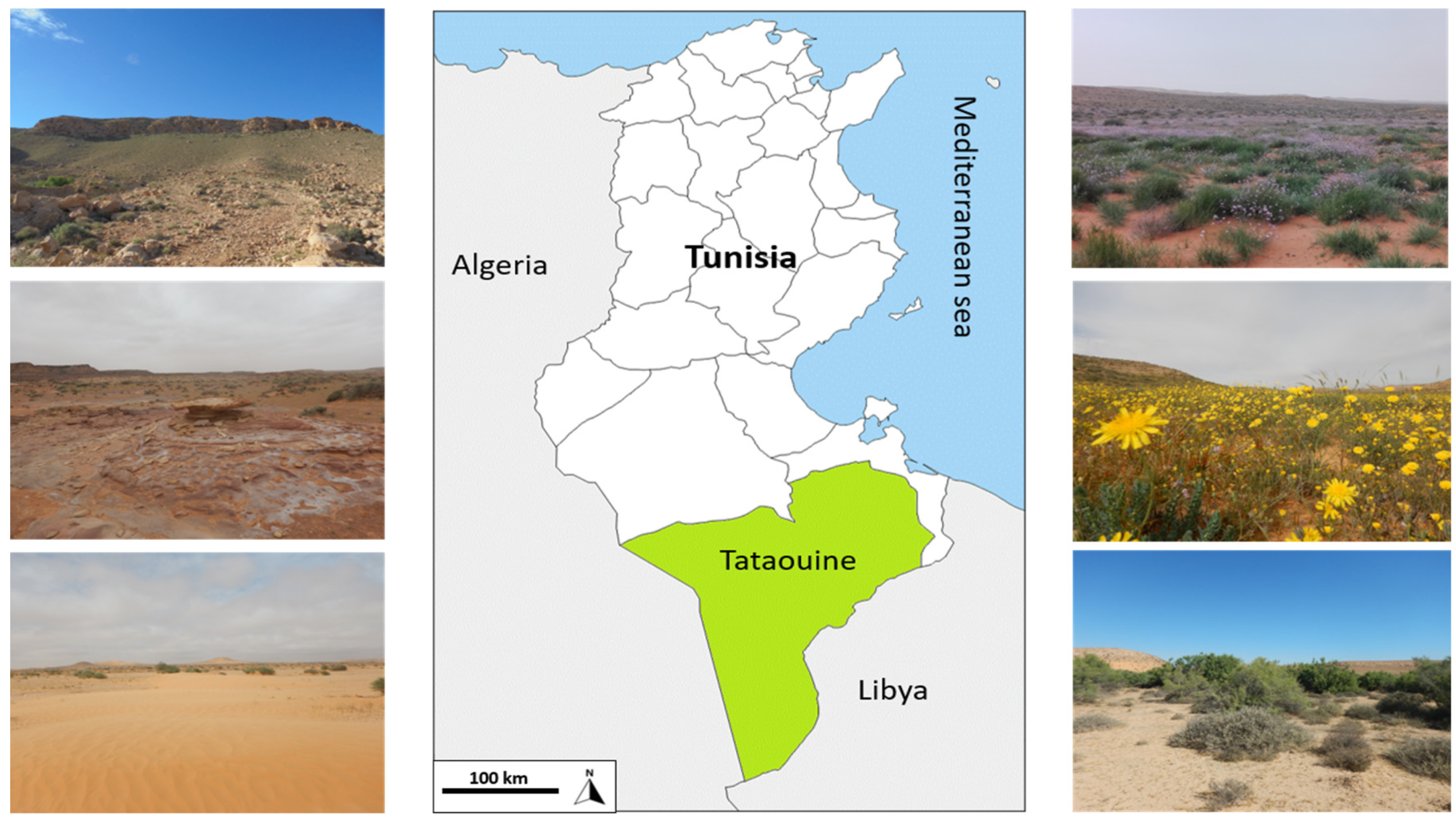
2.1. Study Area
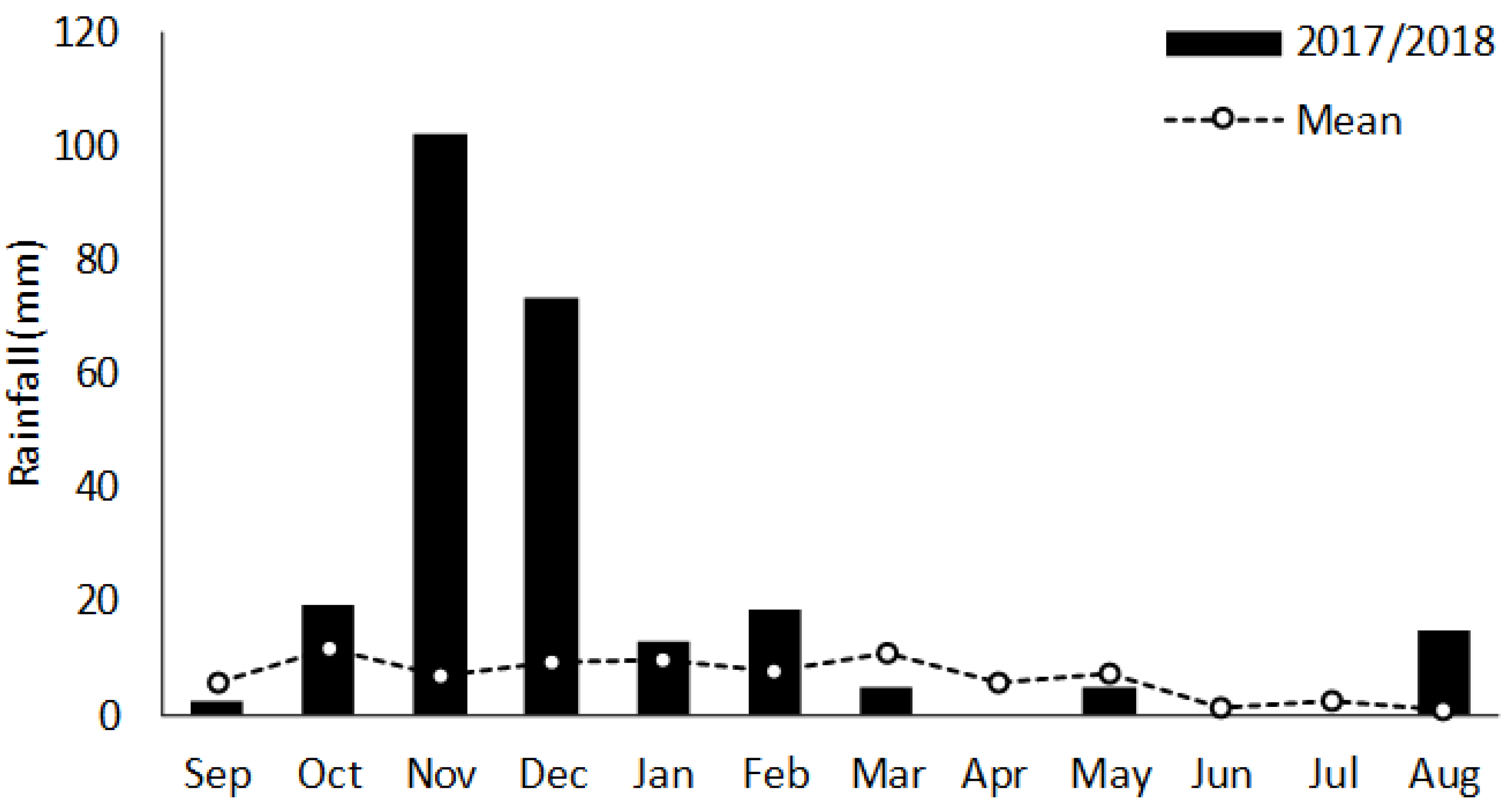
2.2. Data Collection
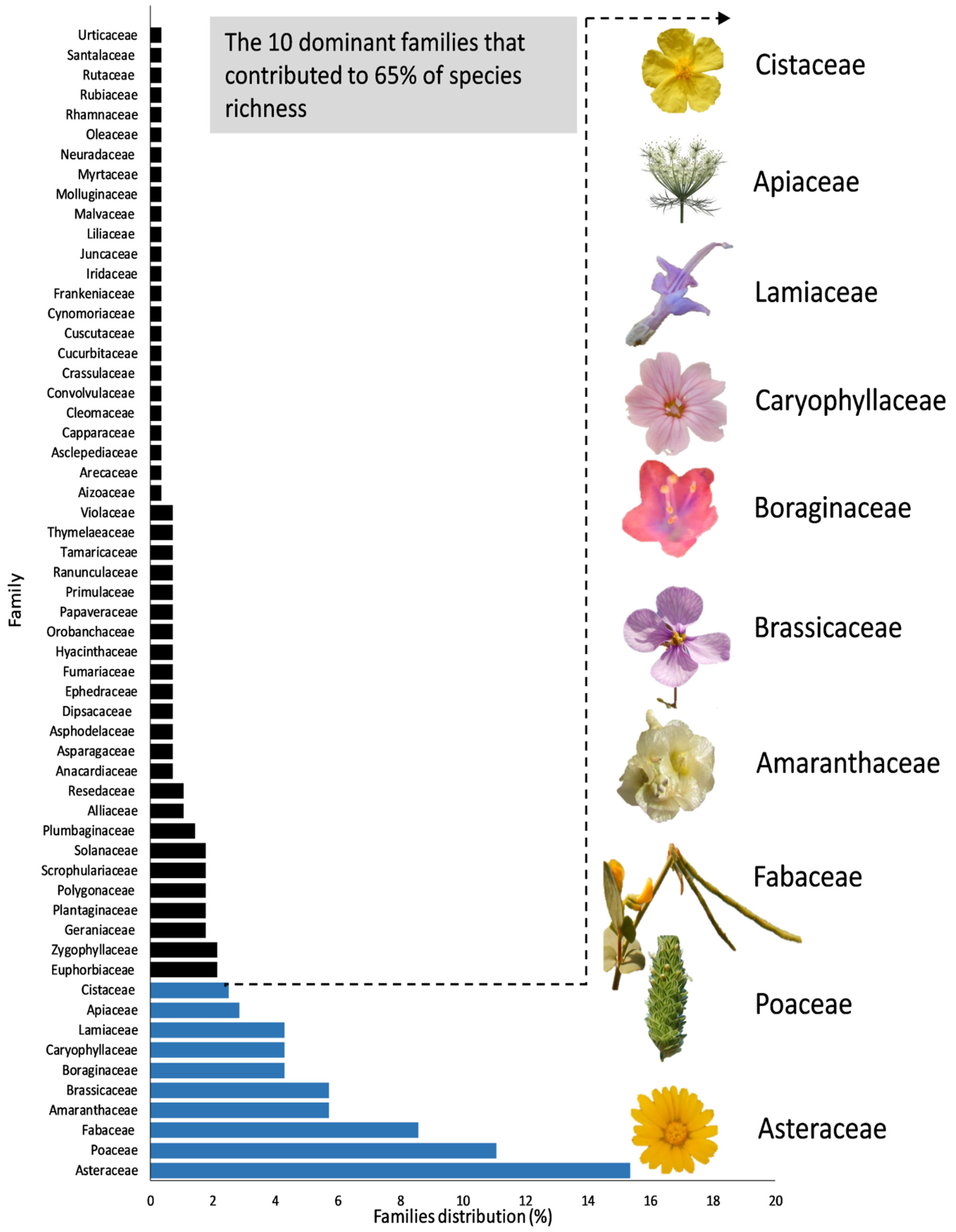
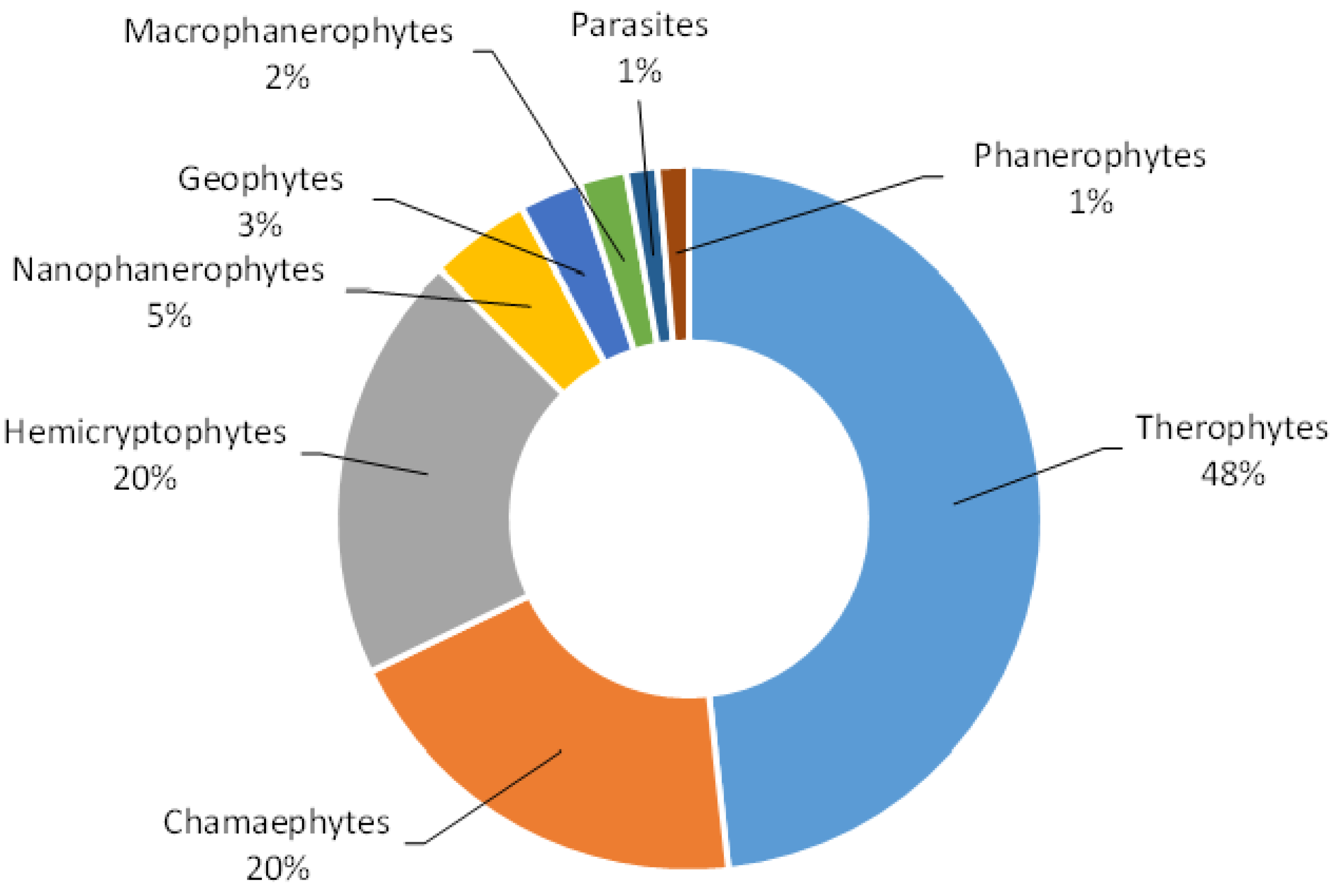
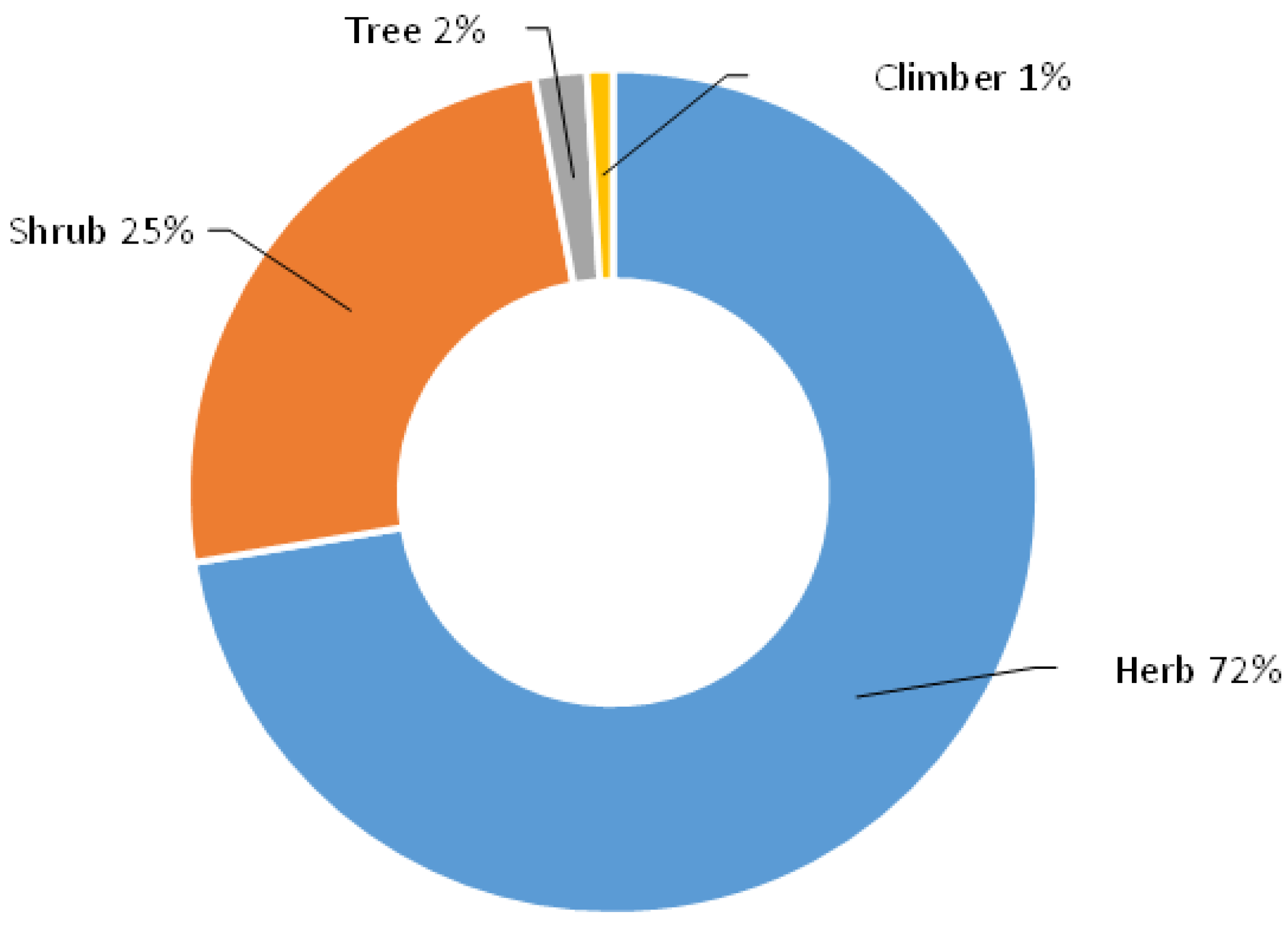
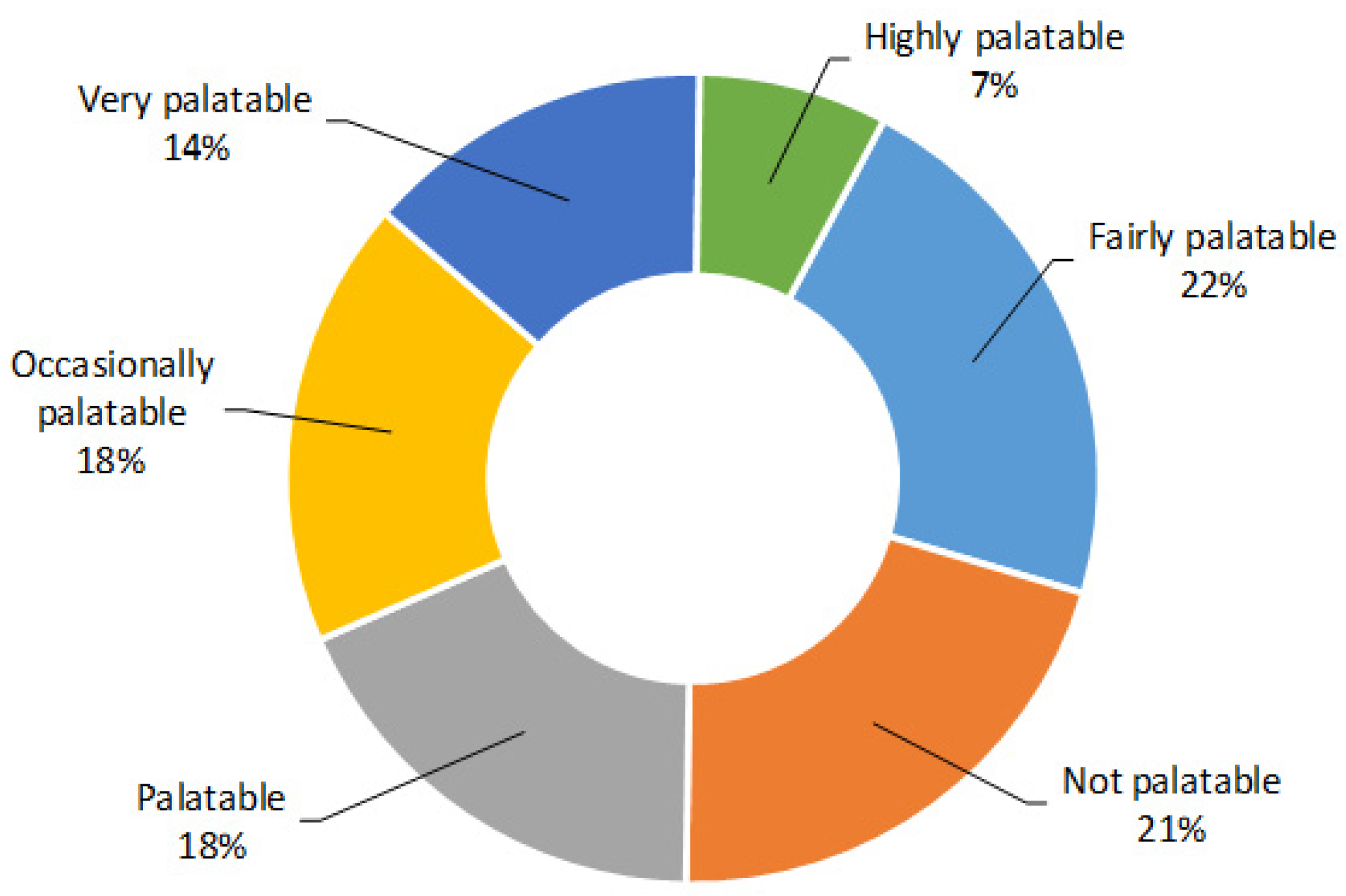
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- He, J.; Yan, C.; Holyoak, M.; Wan, X.; Ren, G.; Hou, Y.; Xie, Y.; Zhang, Z. Quantifying the effects of climate and anthropogenic change on regional species loss in China. PLoS ONE 2018, 13, e0199735. [Google Scholar] [CrossRef]
- Sala, O.E. Global biodiversity scenarios for the year 2100. Science 2000, 287, 1770–1774. [Google Scholar] [CrossRef]
- Hudson, L.N.; Newbold, T.; Contu, S.; Hill, S.L.L.; Lysenko, I.; De Palma, A.; Phillips, H.R.P.; Senior, R.A.; Bennett, D.J.; Booth, H.; et al. The PREDICTS database: A global database of how local terrestrial biodiversity responds to human impacts. Ecol. Evol. 2014, 4, 4701–4735. [Google Scholar] [CrossRef] [PubMed]
- Elzinga, J.A.; Nouhuys, S.V.; Leeuwen, D.J.V.; Biere, A. Distribution and colonisation ability of three parasitoids and their herbivorous host in a fragmented landscape. Basic Appl. Ecol. 2007, 8, 75–88. [Google Scholar] [CrossRef]
- Luciano, C.; Graciela, V.; Adriana, S.; Marcelo, C.; Marcelo, Z. Habitat fragmentation and species loss across three interacting trophic levels: Effects of life-history and food-web traits. Conserv. Biol. 2009, 23, 1167–1175. [Google Scholar] [CrossRef]
- Stork, N.E. Re-assessing current extinction rates. Biodivers. Conserv. 2010, 19, 357–371. [Google Scholar] [CrossRef]
- Ouled Belgacem, A.; Louhaichi, M. The vulnerability of native rangeland plant species to global climate change in the West Asia and North African regions. Climat. Chang. 2013, 119, 451–463. [Google Scholar] [CrossRef]
- Louhaichi, M.; Ouled, B.A.; Hassan, S.; Petersen, L.S. Effects of climate change and grazing practices on shrub communities of West Asian rangelands. Int. J. Clim. Change Str. Manag. 2019, 11, 660–671. [Google Scholar] [CrossRef]
- Gamoun, M. Grazing intensity effects on the vegetation in desert rangelands of Southern Tunisia. J. Arid Land 2004, 6, 324–333. [Google Scholar] [CrossRef]
- Osem, Y.; Perevolotsky, A.; Kigel, J. Grazing effect on diversity of annual plant communities in a semi-arid rangeland: Interactions with small-scale spatial and temporal variation in primary productivity. J. Ecol. 2002, 90, 936–946. [Google Scholar] [CrossRef]
- Fensham, R.J.; Silcock, J.L.; Dwyer, J.M. Plant species richness responses to grazing protection and degradation history in a low productivity landscape. J. Veg. Sci. 2011, 22, 997–1008. [Google Scholar] [CrossRef]
- Mönkkönen, M. Managing Nordic boreal forest landscapes for biodiversity: Ecological and economic perspectives. Biodivers. Conserv. 1999, 8, 85–99. [Google Scholar] [CrossRef]
- Shachak, M.; Gosz, J.R.; Pickett, S.T.A.; Perevolotsky, A. Biodiversity in Drylands: Toward a Unified Framework; Oxford University Press: Oxford, UK, 2005. [Google Scholar]
- Ramawat, K.G. Desert Plants: Biology and Biotechnology; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Tarhouni, M.; Ben Hmida, W.; Ouled Belgacem, A.; Louhaichi, M.; Neffati, M. Is long-term protection useful for the regeneration of disturbed plant communities in dry areas? Afr. J. Ecol. 2017, 55, 509–517. [Google Scholar] [CrossRef]
- Gamoun, M.; Ouled Belgacem, A.; Louhaichi, M. Diversity of desert rangelands of Tunisia. Plant Divers. 2018, 40, 217–225. [Google Scholar] [CrossRef]
- Ouled Belgacem, A.; Ben Salem, F.; Gamoun, M.; Chibani, R.; Louhaichi, M. Revival of traditional best practices for rangeland restoration under climate change in the dry areas: A case study from Southern Tunisia. Int. J. Clim. Chang. Strateg. Manag. 2019, 11, 643–659. [Google Scholar] [CrossRef]
- Primack, R.B. Essentials of Conservation Biology, 3rd ed.; Sinauer Ass: Sunderland, MA, USA, 2002. [Google Scholar]
- Meir, E.; Andelman, S.; Possingham, H.P. Does conservation planning matter in a dynamic and uncertain world? Ecol. Lett. 2004, 7, 615–622. [Google Scholar] [CrossRef]
- Matthies, P.L.; Bose, E. Species richness, structural diversity and species composition in meadows created by visitors of a botanical garden in Switzerland. Landsc. Urban Plan. 2007, 79, 298–307. [Google Scholar] [CrossRef]
- ODS. Tataouine Governorate in Numbers; Republic of Tunisia Ministry of Development, Investment and International Cooperation, Southern Development Office: Tunis, Tunisia, 2018.
- Pottier-Aiapetite, G. Flore de la Tunisie: Angiospermes, Dicotyledones Apetales-Dialypetales; Ministère de l’Enseignement Supérieur et de la Recherche Scientifique et le Ministère de l’Agriculture: Tunis, Tunisia, 1979; pp. 1–654.
- Pottier Alapetite, G. Flore de la Tunisie: Angiospermes-Dicotylédones, Gamopétales. Programme Flore et Végétation Tunisiennes; Ministère de l’Enseignement Supérieur et de la Recherche Scientifique et le Ministère de l’Agriculture: Tunis, Tunisia, 1981.
- Dobignard, A.; Chatelain, C. Synonymic and Bibliographical Index of the Flora of North Africa; Conservatoire et Jardin Botaniques de la Ville de Genève: Geneva, Switzerland, 2010–2013.
- POWO. Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. Available online: http://www.plantsoftheworldonline.org/2020 (accessed on 11 February 2021).
- CRDA. Tataouine; Commissariat Régional au Développement Agricole de Tataouine: Tunis, Tunisia, 2019. [Google Scholar]
- Noy-Meir, I. Desert ecosystems: Environment and producers. Annu. Rev. Ecol. Syst. 1973, 4, 25–51. [Google Scholar] [CrossRef]
- Yan, H.; Liang, C.; Li, Z.; Liu, Z.; Miao, B.; He, C.; Sheng, L. Impact of precipitation patterns on biomass and species richness of annuals in a dry steppe. PLoS ONE 2015, 10, e0125300. [Google Scholar] [CrossRef]
- García-Vega, D.; Newbold, T. Assessing the effects of land use on biodiversity in the world’s drylands and Mediterranean environments. Biodivers. Conserv. 2019, 29, 393–408. [Google Scholar] [CrossRef]
- Bertiller, M.B.; Beeskow, A.M.; Coronato, F. Seasonal environmental variation and plant phenology in arid Patagonia (Argentina). J. Arid Environ. 1991, 21, 1–11. [Google Scholar] [CrossRef]
- Shinoda, M.; Ito, S.; Nachinshonhor, G.U.; Erdenetsetseg, D. Phenology of Mongolian grasslands and moisture conditions. J. Meteorol. Soc. Jpn 2007, 85, 359–367. [Google Scholar] [CrossRef]
- John, R.; Jiquan, C.; Nan, L.; Ke, G.; Cunzhu, L.; Yafen, W.; Asko, N.; Keping, M.; Xingguo, H. Predicting plant diversity based on remote sensing products in the semi-arid region of Inner Mongolia. Remote Sens. Environ. 2008, 112, 2018–2032. [Google Scholar] [CrossRef]
- Hannah, L.; Midgley, G.F.; Lovejoy, T.; Bond, W.J.; Bush, M.L.; Scott, D.; Woodward, F.I. Conservation of biodiversity in a changing climate. Conserv. Biol. 2002, 16, 11–15. [Google Scholar] [CrossRef]
- Mathias, A.; Chesson, P. Coexistence and evolutionary dynamics mediated by seasonal environmental variation in annual plant communities. Theor. Popul. Biol. 2013, 84, 56–71. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.D.; Keenan, T.F.; Migliavacca, M.; Ryu, Y.; Sonnentag, O.; Toomey, M. Climate change, phenology, and phenological control of vegetation feedbacks to the climate system. Agric. For. Meteorol. 2013, 169, 156–173. [Google Scholar] [CrossRef]
- Tilman, D.; El Haddi, A. Drought and biodiversity in grasslands. Oecologia 1992, 89, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Le Houèrou, H.N. Climate change, drought and desertification. J. Arid. Environ. 1996, 34, 133–185. [Google Scholar] [CrossRef]
- Seghieri, J.; Galle, S. Run-on contribution to a Sahelian two-phase mosaic system: Soil water regime and vegetation life cycles. Acta Oecologica 1999, 20, 209–217. [Google Scholar] [CrossRef]
- Loeser, M.R.R.; Sisk, T.D.; Crews, T.E. Impact of grazing intensity during drought in an Arizona grassland. Conserv. Biol. 2007, 21, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Lin, S.; Taube, F.; Pan, Q.; Dittert, K. Above and belowground net primary productivity of grassland influenced by supplemental water and nitrogen in Inner Mongolia. Plant Soil 2011, 340, 53–64. [Google Scholar] [CrossRef]
- Ni, J. Plant functional types and climate along a precipitation gradient in temperate grasslands, north-east China and south-east Mongolia. J. Arid Environ. 2003, 53, 501–516. [Google Scholar] [CrossRef][Green Version]
- Bai, Y.F.; Han, X.G.; Wu, J.G.; Chen, Z.; Li, L. Ecosystem stability and compensatory effects in the Inner Mongolia grassland. Nature 2004, 431, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Tsubo, M.; Ito, T.Y.; Nishihara, E.; Shinoda, M. Impact of rainfall variability and grazing pressure on plant diversity in Mongolian grasslands. J. Arid Environ. 2011, 75, 471–476. [Google Scholar] [CrossRef]
- Kinugasa, T.; Tsunekawa, A.; Shinoda, M. Increasing nitrogen deposition enhances post-drought recovery of grassland productivity in the Mongolian steppe. Oecologia 2012, 170, 857–865. [Google Scholar] [CrossRef] [PubMed]
- Holzaphel, C.; Schmidt, W.; Shmida, A. Effects of human-caused distribution on the flora along a Mediterranean-desert gradient. Flora 1992, 186, 261–270. [Google Scholar] [CrossRef]
- Hobbs, R.J. Synergisms among habitat fragmentation, livestock grazing, and biotic invasions in southwestern Australia. Conserv. Biol. 2001, 15, 1522–1528. [Google Scholar] [CrossRef]
- Gamoun, M.; Tarhouni, M.; Ouled Belgacem, A.; Hanchi, B.; Neffati, M. Response of different arid rangelands to protection and drought. Arid Land Res. Manag. 2011, 25, 372–378. [Google Scholar] [CrossRef]
- Gamoun, M. The Impact of Rest from Grazing on Vegetation Dynamics: Application to Sustainable Management of Saharan Rangelands in Southern Tunisia. Ph.D. Thesis, University of Tunis El Manar, Tunis, Tunisia, 2012. [Google Scholar]
- Raunkiaer, C. The Life Form of Plants and Statistical Plant Geography; Collected Papers; Clarendon Press: Oxford, UK, 1934. [Google Scholar]
- Orshan, G.; Montenegro, G.; Avila, G.; Aljaro, M.E.; Walckowiaak, A.; Mujica, A.M. Plant growth forms of Chilean matoral. A monocharacter growth form analysis along an altitudinal transect from sea level to 2000 MASL. Bull. Soc. Bot. Fr. 1984, 131, 411–425. [Google Scholar]
- Floret, C.; Galan, M.J.; LeFloc’h, E.; Orshan, G.; Romane, F. Growth forms and phenomorphology traits along an environmental gradient: Tools for studying vegetation. J. Veg. Sci. 1990, 1, 71–80. [Google Scholar] [CrossRef]
- Jauffret, S.; Visser, M. Assigning life history traits to plant species to better qualify arid land degradation in Presaharian Tunisia. J. Arid. Environ. 2003, 55, 1–28. [Google Scholar] [CrossRef]
- Kahmen, S.; Poschlod, P. Effects of grassland management on plant functional trait composition. Agric. Ecosyst. Environ. 2008, 128, 137–145. [Google Scholar] [CrossRef]
- Gamoun, M.; Ouled Belgacem, A.; Hanchi, B.; Neffati, M.; Gillet, F. Impact of grazing on the floristic diversity of arid rangelands in South Tunisia. Rev. Ecol. 2012, 67, 271–282. [Google Scholar]
- Evenari, M. The desert environment. In Ecosystems of the World. Hot Deserts and Arid Shrublands 12A; Evenari, M., Noy-Meir, I., Goodall, D.W., Eds.; Elsevier: Amsterdam, The Netherlands, 1985; pp. 1–22. [Google Scholar]
- Le Houérou, H.N. Les Pâturages Naturels de la Tunisie Aride et Désertique; Institut des Sciences Economiques Appliquées: Tunis, Tunisia, 1962. [Google Scholar]
- Le Houérou, H.N. Improvements of Natural Pastures and Fodder Resources; Report to the Government of Libya. EPTA Rep. no. 1979; FAO: Rome, Italy, 1965. [Google Scholar]
- Le Houérou, H.N.; Ionesco, T. Appétabilité des Espèces Végétales de la Tunisie Steppique; AG-TUN 71/ 525; FAO: Rome, Italy, 1973. [Google Scholar]
- Sarson, M.; Salmon, P. Appétabilité de Certaines Espèces Vivaces Spontanées ou Introduites au Maroc et en Afrique du Nord; FAO: Rabat, Maroc; Rome, Italy, 1976. [Google Scholar]
- Bhatnagar, A.; Bhatnagar, M. Microbial diversity in desert ecosystems. Curr. Sci. 2005, 89, 91–100. [Google Scholar]
- Le Floc’h, E. Contribution à une Étude Ethnobotanique de la Flore Tunisienne; Imprimerie Officielle de la République Tunisienne: Tunis, Tunisia, 1983.
- Neffati, M.; Najjaa, H.; Máthé, A. Medicinal and Aromatic Plants of the World—Africa; Springer Nature: Berin/Heidelberg, Germany, 2017; Volume 3. [Google Scholar]
- Chaieb, M.; Boukhris, M. Flore Succinte et Illustrée des Zones Arides et Désertiques de Tunisie; Association pour la Protection de la Nature et de l’Environnement: Sfax, Tunisia, 1998. [Google Scholar]
- Jendoubi, R.; Neffati, M.; Henchi, B.; Yobi, A. Système de reproduction et variabilité morphologique chez Allium roseum L. Plant Genet. Res. Newsl. 2001, 127, 29–34. [Google Scholar]
- Neffati, M.; Ghrabi-Gammar, Z.; Akrimi, N.; Henchi, B. Les plantes endémiques de la Tunisie. Flora Mediterr. 1999, 9, 163–174. [Google Scholar]
- Laity, J. Deserts and Desert Environments; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008. [Google Scholar]
- Isbell, F.; Adler, P.R.; Eisenhauer, N.; Fornara, D.; Kimmel, K.; Kremen, C.; Letourneau, D.K.; Liebman, M.; Polley, H.W.; Quijas, S.; et al. Benefits of increasing plant diversity in sustainable agroecosystems. J. Ecol. 2017, 105, 871–879. [Google Scholar] [CrossRef]
- Pohl, M.; Graf, F.; Buttler, A.; Rixen, C. The relationship between plant species richness and soil aggregate stability can depend on disturbance. Plant Soil. 2012, 355, 87–102. [Google Scholar] [CrossRef]
- Gould, I.J.; Quinton, J.N.; Weigelt, A.; De Deyn, G.B.; Bardgett, R.D. Plant diversity and root traits benefit physical properties key to soil function in grasslands. Ecol. Lett. 2016, 19, 1140–1149. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gamoun, M.; Louhaichi, M. Botanical Composition and Species Diversity of Arid and Desert Rangelands in Tataouine, Tunisia. Land 2021, 10, 313. https://doi.org/10.3390/land10030313
Gamoun M, Louhaichi M. Botanical Composition and Species Diversity of Arid and Desert Rangelands in Tataouine, Tunisia. Land. 2021; 10(3):313. https://doi.org/10.3390/land10030313
Chicago/Turabian StyleGamoun, Mouldi, and Mounir Louhaichi. 2021. "Botanical Composition and Species Diversity of Arid and Desert Rangelands in Tataouine, Tunisia" Land 10, no. 3: 313. https://doi.org/10.3390/land10030313
APA StyleGamoun, M., & Louhaichi, M. (2021). Botanical Composition and Species Diversity of Arid and Desert Rangelands in Tataouine, Tunisia. Land, 10(3), 313. https://doi.org/10.3390/land10030313






