Land Use and Management Effects on Sustainable Sugarcane-Derived Bioenergy
Abstract
1. Introduction
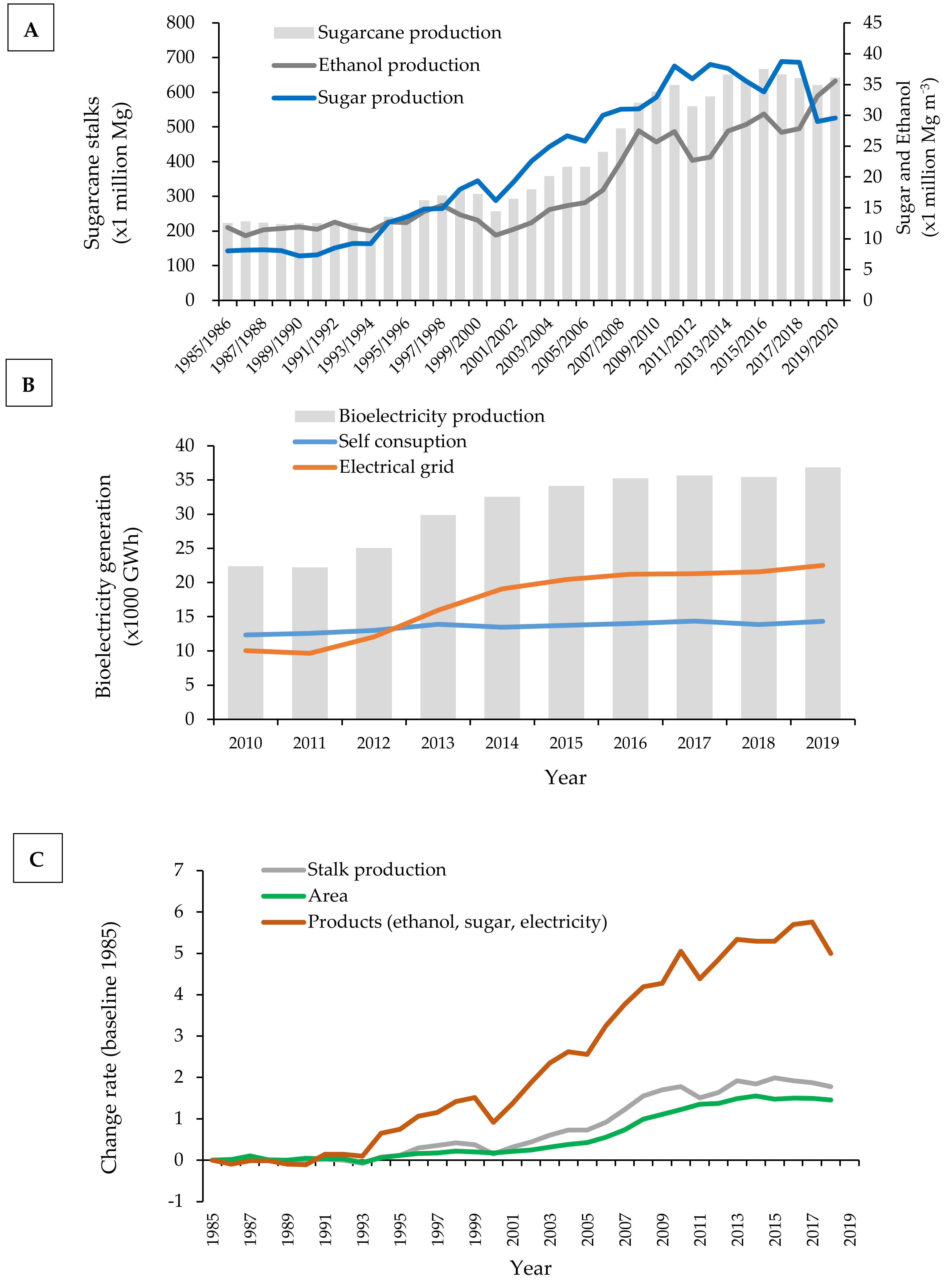
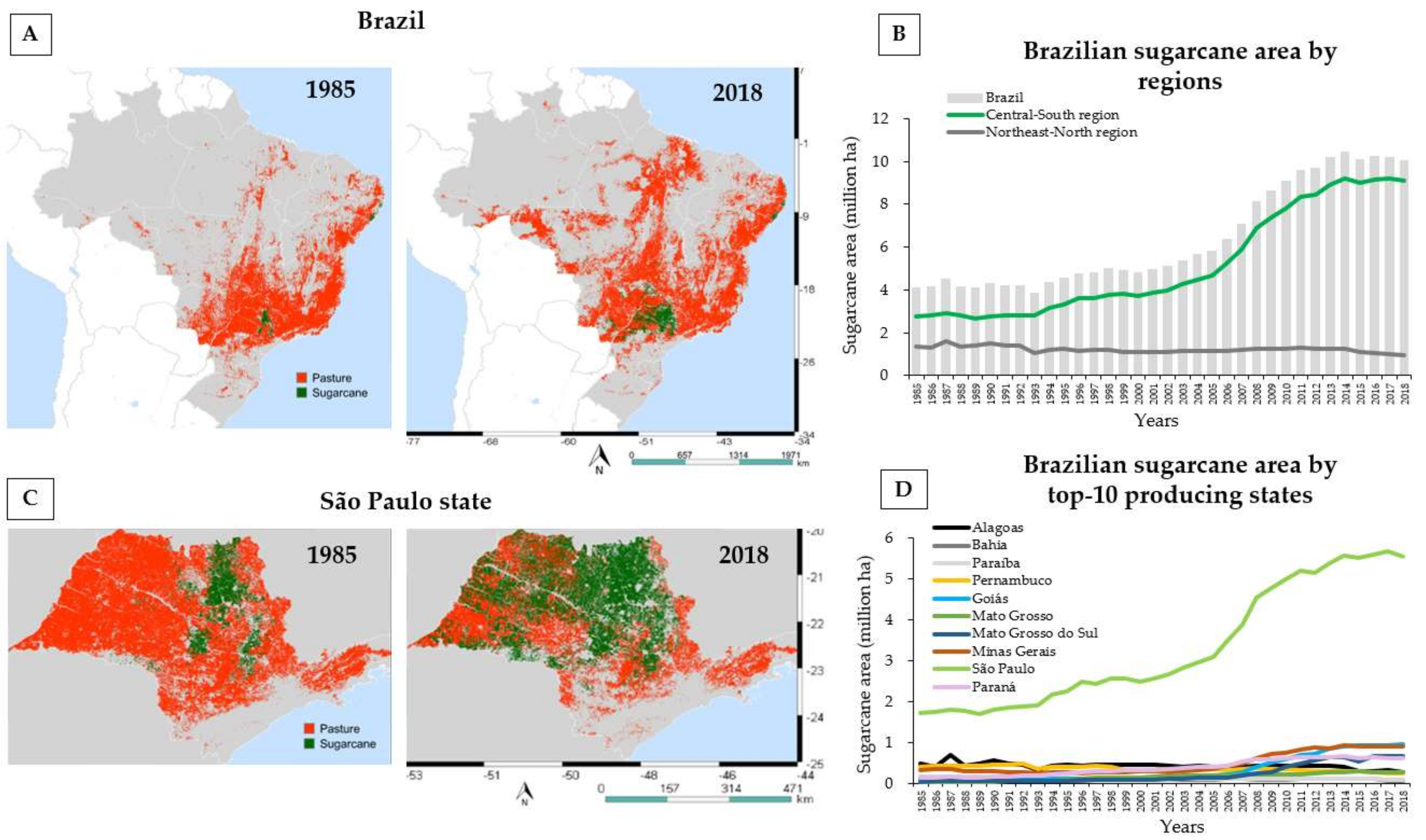
2. Land Use Change for Sugarcane Production in Brazil
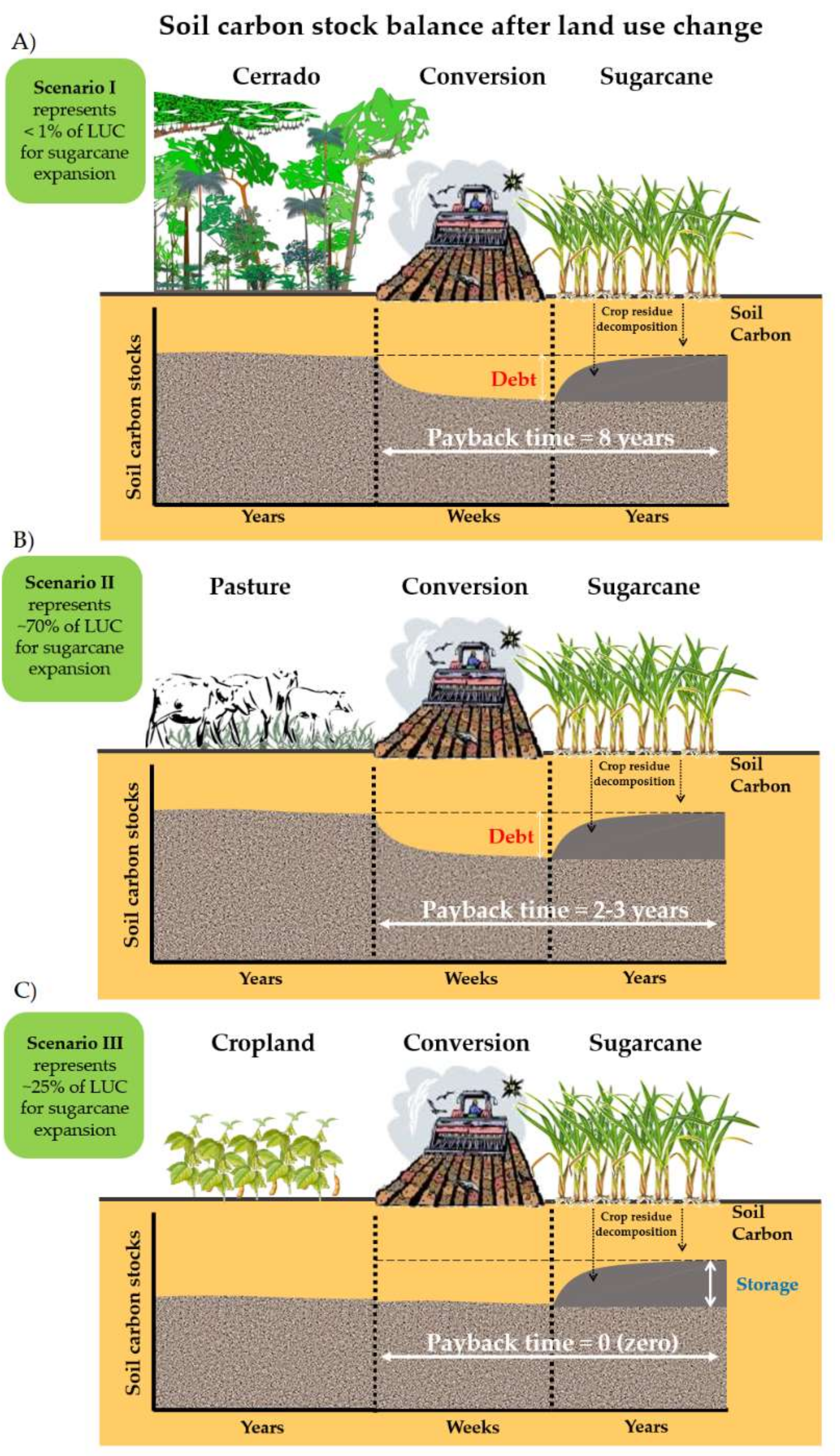
2.1. Soil C Sequestration in Land-Use Change Scenarios for Sugarcane Expansion
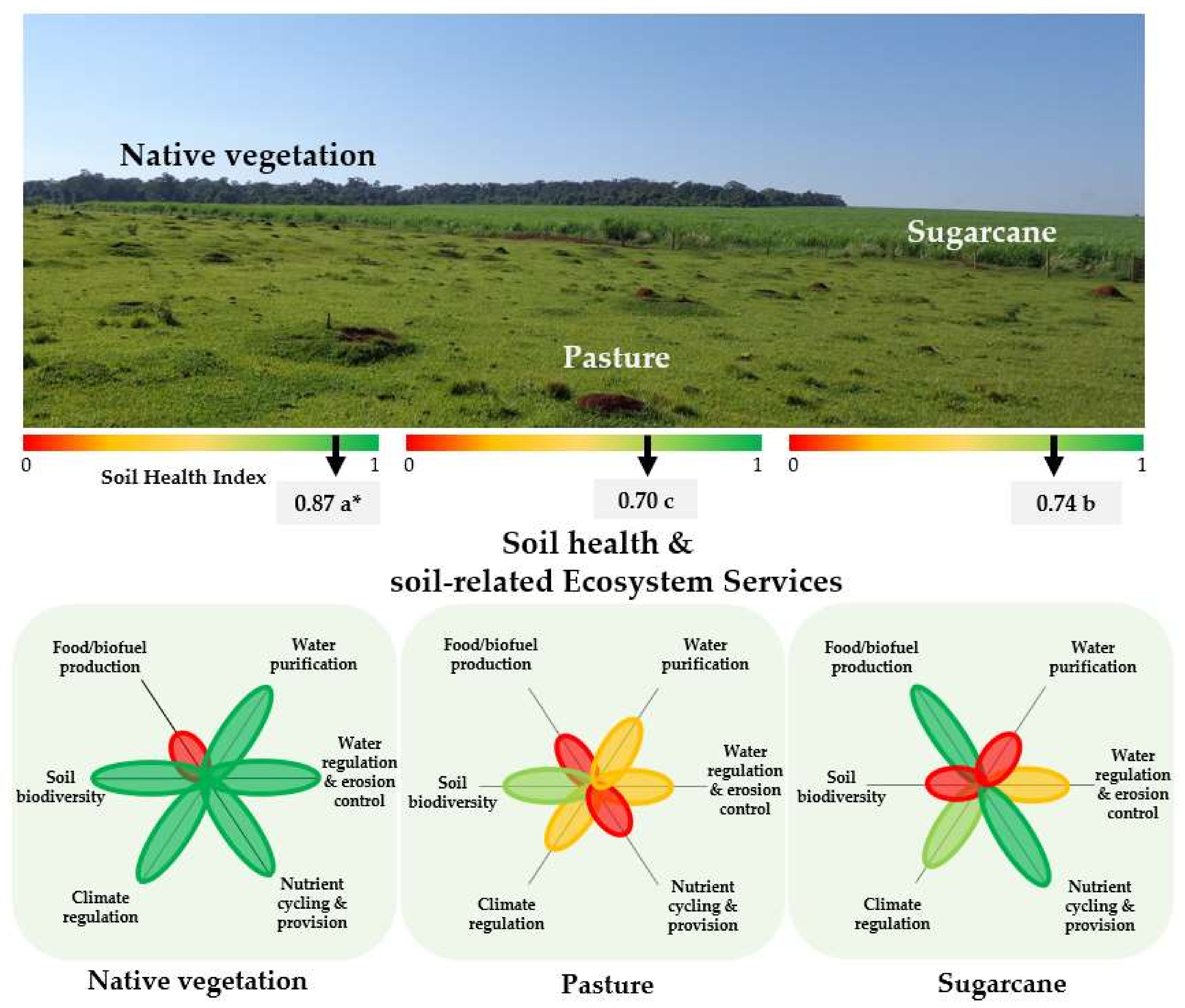
2.2. Effects of LUC for Sugarcane Expansion on Soil Health and Soil-Related Ecosystem Services
3. Best Management Practices towards Sustainable Sugarcane-Derived Bioenergy Production
3.1. Conservation Tillage and Crop Rotation
3.2. Rational Crop Residue Management
3.3. Fertilization Management and GHG Emissions in Sugarcane Fields
3.4. Recycling Sugarcane by-Products: Nutrient Savings and Promotion of the Circular Economy
4. Public Policies for Promoting Sustainable Bioenergy Production and Land Preservation
5. Sugarcane Potential for Bioenergy Production in Latin America and Africa
6. Final Remarks and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Country | Land under Permanent Meadows and Pastures (2018) (FAO, [6]) | Sugarcane Production (2018) [A] (FAO, [6]) | Sugarcane Potential Production on 1% of Pasture Land [B] | Potential Annual Bioenergy Production | |||
|---|---|---|---|---|---|---|---|
| Ethanol | Electricity | ||||||
| 1000 ha | % Country Area | 1000 Mg | 1000 Mg | % Increase | 1000 m3 | MWh | |
| (A/B) | |||||||
| Angola | 51,737 | 41.5% | 573 | 43,977 | 7676% | 3738 | 4837 |
| Cameroon | 2000 | 4.2% | 1287 | 1700 | 132% | 145 | 187 |
| Congo | 10,000 | 29.2% | 718 | 8500 | 1183% | 723 | 935 |
| Côte d’Ivoire | 13,200 | 40.9% | 1948 | 11,220 | 576% | 954 | 1234 |
| Eswatini | 1032 | 59.4% | 5596 | 877 | 16% | 75 | 96 |
| Ethiopia | 20,000 | 17.6% | 1475 | 17,000 | 1153% | 1445 | 1870 |
| Ghana | 7383 | 30.9% | 152 | 6275 | 4115% | 533 | 690 |
| Guinea | 10,700 | 43.5% | 312 | 9095 | 2919% | 773 | 1000 |
| Kenya | 21,300 | 36.7% | 5262 | 18,105 | 344% | 1539 | 1992 |
| Liberia | 1254 | 11.3% | 275 | 1066 | 387% | 91 | 117 |
| Madagascar | 37,295 | 63.5% | 3143 | 31,701 | 1009% | 2695 | 3487 |
| Malawi | 1850 | 15.6% | 3025 | 1573 | 52% | 134 | 173 |
| Mozambique | 35,464 | 44.4% | 3155 | 30,144 | 956% | 2562 | 3316 |
| Nigeria | 28,623 | 31.0% | 1423 | 24,330 | 1710% | 2068 | 2676 |
| Sierra Leone | 2200 | 30.4% | 78 | 1870 | 2384% | 159 | 206 |
| South Sudan | 25,773 | 40.7% | nad * | 21,907 | 1862 | 2410 | |
| Uganda | 5315 | 22.0% | 3977 | 4518 | 114% | 384 | 497 |
| Tanzania | 24,000 | 25.3% | 3052 | 20,400 | 668% | 1734 | 2244 |
| Zambia | 20,000 | 26.6% | 4461 | 17,000 | 381% | 1445 | 1870 |
| Zimbabwe | 12,100 | 31.0% | 3305 | 10,285 | 311% | 874 | 1131 |
| SSA | 331,227 | 33.0% | 43,217 | 281,543 | 651% | 23,931 | 30,970 |
| Argentina | 108,500 | 39.0% | 19,040 | 92,225 | 484% | 7839 | 10,145 |
| Bolivia | 33,000 | 30.0% | 9616 | 28,050 | 292% | 2384 | 3086 |
| Brazil | 173,361 | 20.4% | 746,828 | 147,357 | 20% | 12,525 | 16,209 |
| Colombia | 39,600 | 34.7% | 36,277 | 33,660 | 93% | 2861 | 3703 |
| Costa Rica | 1200 | 23.5% | 4421 | 1020 | 23% | 87 | 112 |
| Cuba | 2738 | 24.9% | 19,648 | 2328 | 12% | 198 | 256 |
| Dominican Republic | 1197 | 24.6% | 5278 | 1017 | 19% | 86 | 112 |
| Ecuador | 3094 | 12.1% | 7502 | 2630 | 35% | 224 | 289 |
| El Salvador | 625 | 29.7% | 7046 | 531 | 8% | 45 | 58 |
| Guatemala | 1811 | 16.6% | 35,568 | 1539 | 4% | 131 | 169 |
| Guyana | 781 | 3.6% | 1214 | 664 | 55% | 56 | 73 |
| Honduras | 1760 | 15.6% | 5526 | 1496 | 27% | 127 | 165 |
| Mexico | 80,279 | 40.9% | 56,842 | 68,237 | 120% | 5800 | 7506 |
| Nicaragua | 3275 | 25.1% | 7224 | 2784 | 39% | 237 | 306 |
| Panama | 1509 | 20.0% | 2931 | 1283 | 44% | 109 | 141 |
| Paraguay | 17,000 | 41.8% | 6160 | 14,450 | 235% | 1228 | 1590 |
| Uruguay | 12,000 | 68.1% | 351 | 10,200 | 2910% | 867 | 1122 |
| Venezuela | 18,200 | 20.0% | 4167 | 15,470 | 371% | 1315 | 1702 |
| LAC | 499,931 | 28% | 975,640 | 424,941 | 44% | 36,120 | 46,744 |
| LAC w/o Brazil | 326,570 | 34% | 228812 | 277,584 | 121% | 23,595 | 30,534 |
References
- Hanssen, S.V.; Daioglou, V.; Steinmann, Z.J.N.; Doelman, J.C.; van Vuuren, D.P.; Huijbregts, M.A.J. The climate change mitigation potential of bioenergy with carbon capture and storage. Nat. Clim. Chang. 2020. [Google Scholar] [CrossRef]
- Arneth, A.; Barbosa, H.; Benton, T.; Calvin, K.; Calvo, E.; Connors, S. Summary For Policymakers. In Climate Change And Land: An IPCC Special Report on Climate Change, Desertification Land Degradation, Sustainable Land Management, Food Security, and Greenhouse Gas Fluxes in Terrestrial Ecosystems; IPCC: Geneva, Swizerland, 2019. [Google Scholar]
- Goldemberg, J. Ethanol for a sustainable energy future. Science 2007, 315, 808–810. [Google Scholar] [CrossRef] [PubMed]
- Bordonal, R.O.; Carvalho, J.L.N.; Lal, R.; de Figueiredo, E.B.; de Oliveira, B.G.; La Scala, N. Sustainability of sugarcane production in Brazil. A. review. Agron. Sustain. Dev. 2018, 38. [Google Scholar] [CrossRef]
- OECD/FAO. OECD‑FAO Agricultural Outlook 2019–2028; OECD: Rome, Italy, 2019. [Google Scholar]
- FAO. Food and Agriculture Organization of the United Nations—FAOStat, Land Use Data 2020. Available online: http://www.fao.org/faostat/en/#data/RL (accessed on 19 November 2020).
- Companhia Nacional de Abastecimento—CONAB. Acompanhamento de Safra Brasileira de Cana-de-açúcar. v. 7—Safra 2020/2021, no. 1—Primeiro Levantamento—Maio 2020. 2020. Available online: https://www.conab.gov.br/info-agro/safras/cana/boletim-da-safra-de-cana-de-acucar (accessed on 20 June 2020).
- SINDIPEÇAS/ABIPEÇAS. Relatório da Frota Circulante 2018. Sindicato Nacional Indústria Componentes Veículos Automotores SINDIPEÇAS Associação Brasileira Indústria Autopeças ABIPEÇAS. 2019. Available online: https://www.sindipecas.org.br/sindinews/Economia/2019/RelatorioFrotaCirculante_Maio_2019.pdf (accessed on 19 November 2020).
- EPE—Empresa de Pesquisa Energética. Brazilian Energy Balance 2020. Brasilia—DF. 2020. Available online: https://www.epe.gov.br/sites-en/publicacoes-dados-abertos/publicacoes/Paginas/Brazilian-Energy-Balance-2020.aspx (accessed on 19 November 2020).
- Brazil, “Lei No. 13576, de 26 de Dezembro de 2017—Dispõe Sobre a Política Nacional de Biocombustíveis (RenovaBio) e dá Outras Providências. 2017. Available online: https://www.planalto.gov.br/ccivil_03/_ato2015-2018/2017/lei/l13576.htm (accessed on 19 November 2020).
- Brazil, “Intended Nationally Determined Contributions (iNDC)—Brazil. 2015. Available online: http://www.mma.gov.br/images/arquivos/clima/convencao/indc/BRAZIL_iNDC_english.pdf (accessed on 19 November 2020).
- Carvalho, J.L.N.; Cerri, C.E.P.; Karlen, D.L. Sustainable Sugarcane Straw Special Issue: Considerations for Brazilian Bioenergy Production. Bioenergy Res. 2019, 12, 746–748. [Google Scholar] [CrossRef]
- Foley, J.A.; DeFries, R.; Asner, G.P.; Barford, C.; Bonan, G.; Carpenter, S.R.; Chapin, F.S.; Coe, M.T.; Daily, G.C.; Gibbs, H.K.; et al. Global consequences of land use. Science 2005, 309, 570–574. [Google Scholar] [CrossRef]
- Oliveira, D.M.S.; Cherubin, M.R.; Franco, A.L.C.; Santos, A.S.; Gelain, J.G.; Dias, N.M.S.; Diniz, T.R.; Almeida, A.N.; Feigl, B.J.; Davies, C.A.; et al. Is the expansion of sugarcane over pasturelands a sustainable strategy for Brazil’s bioenergy industry? Renew. Sustain. Energy Rev. 2019, 102, 346–355. [Google Scholar] [CrossRef]
- Nogueira, L.A.H.; Capaz, R.S. Biofuels in Brazil: Evolution, achievements and perspectives on food security. Glob. Food Sec. 2013, 2, 117–125. [Google Scholar] [CrossRef]
- Cherubin, M.R.; Lisboa, I.P.; Silva, A.G.B.; Varanda, L.L.; Bordonal, R.O.; Carvalho, J.L.N.; Otto, R.; Pavinato, P.S.; Soltangheisi, A.; Cerri, C.E.P. Sugarcane Straw Removal: Implications to Soil Fertility and Fertilizer Demand in Brazil. Bioenergy Res. 2019, 12, 888–900. [Google Scholar] [CrossRef]
- Cherubin, M.R.; Franco, A.L.C.; Cerri, C.E.P.; Oliveira, D.M.d.; Davies, C.A.; Cerri, C.C. Sugarcane expansion in Brazilian tropical soils-Effects of land use change on soil chemical attributes. Agric. Ecosyst. Environ. 2015, 211, 173–184. [Google Scholar] [CrossRef]
- Werling, B.P.; Dickson, T.L.; Isaacs, R.; Gaines, H.; Gratton, C.; Gross, K.L.; Liere, H.; Malmstrom, C.M.; Meehan, T.D.; Ruan, L.; et al. Perennial grasslands enhance biodiversity and multiple ecosystem services in bioenergy landscapes. Proc. Natl. Acad. Sci. USA 2014, 111, 1652–1657. [Google Scholar] [CrossRef]
- Verdade, L.M.; Piña, C.I.; Rosalino, L.M. Biofuels and biodiversity: Challenges and opportunities. Environ. Dev. 2015, 15, 64–78. [Google Scholar] [CrossRef]
- Romeu-Dalmau, C.; Gasparatos, A.; von Maltitz, G.; Graham, A.; Almagro-Garcia, J.; Wilebore, B.; Willis, K.J. Impacts of land use change due to biofuel crops on climate regulation services: Five case studies in Malawi, Mozambique and Swaziland. Biomass Bioenergy 2018, 114, 30–40. [Google Scholar] [CrossRef]
- Oliveira, D.M.S.; Paustian, K.; Davis, C.A.; Cherubin, M.R.; Franco, A.L.C.; Cerri, C.C.; Cerri, C.E.P. Soil carbon changes in areas undergoing expansion of sugarcane into pastures in south-central Brazil. Agric. Ecosyst. Environ. 2016, 228, 38–48. [Google Scholar] [CrossRef]
- UNICA—The Brazilian Sugarcane Industry Association. Observatório da Cana, 2020. Available online: https://observatoriodacana.com.br/ (accessed on 19 November 2020).
- Manzatto, C.V.; Assad, E.D.; Bacca, J.F.M.; Zaroni, M.J.; Pereira, S.E.M. Zoneamento Agroecológico da Cana-de Açúcar Expandir a produção, preservar a vida, garantir o future. Embrapa Solos-Documentos INFOTECA-E 2009, 110, 55. [Google Scholar]
- Strassburg, B.B.N.; Latawiec, A.; Barioni, L.G.; Nobre, C.A.; Silva, V.P.; Valentim, J.F.; Vianna, M.; Assad, E.D. When enough should be enough: Improving the use of current agricultural lands could meet production demands and spare natural habitats in Brazil. Glob. Environ. Chang. 2014, 28, 84–97. [Google Scholar] [CrossRef]
- Adami, M.; Rudorff, B.F.T.; Freitas, R.M.; Aguiar, D.A.; Sugawara, L.M.; Mello, M.P. Remote Sensing Time Series to Evaluate Direct Land Use Change of Recent Expanded Sugarcane Crop in Brazil. Sustainability 2012, 4, 574–585. [Google Scholar] [CrossRef]
- Dias, L.C.P.; Pimenta, F.M.; Santos, A.B.; Costa, M.H.; Ladle, R.J. Patterns of land use, extensification, and intensification of Brazilian agriculture. Glob. Chang. Biol. 2016, 22, 2887–2903. [Google Scholar] [CrossRef]
- IBGE—Instituto Brasileiro de Geografia e Estatística, “Levantamento Sistemático da Produção Agrícola. 2020. Available online: https://sidra.ibge.gov.br/tabela/1618 (accessed on 19 November 2020).
- MapBiomas. MapBiomas plataform—Uso e cobertura do solo. 2020. Available online: https://plataforma.mapbiomas.org/ (accessed on 19 November 2020).
- Hernandes, T.A.D.; Duft, D.G.; Luciano, A.C.S.; Leal, M.R.L.V.; Cavalett, O. Identifying suitable areas for expanding sugarcane ethanol production in Brazil under conservation of environmentally relevant Habitats. J. Clean. Prod. 2020, 125318. [Google Scholar] [CrossRef]
- Bungenstab, D.J.; de Almeida, R.G.; Laura, V.A.; Balbino, L.C.; Ferreira, A.D. ILPF Lavoura, Pecuária e Floresta; Embrapa: Brasília, Brazil, 2019; 835p. [Google Scholar]
- Salton, J.C.; Mercante, F.M.; Tomazi, M.; Zanatta, J.A.; Concenço, G.; Silva, W.M.; Retore, M. Integrated crop-livestock system in tropical Brazil: Toward a sustainable production system. Agric. Ecosyst. Environ. 2014, 190, 70–79. [Google Scholar] [CrossRef]
- Zilberman, D. Indirect land use change: Much ado about (almost) nothing. GCB Bioenergy 2017, 9, 485–488. [Google Scholar] [CrossRef]
- Wicke, B.; Verweij, P.; van Meijl, H.; van Vuuren, D.P.; Faaij, A.P.C. Indirect land use change: Review of existing models and strategies for mitigation. Biofuels 2012, 3, 87–100. [Google Scholar] [CrossRef]
- Fargione, J.; Hill, J.; Tilman, D.; Polasky, S.; Hawthorne, P. Land clearing and the biofuel carbon debt. Science 2008, 319, 1235–1238. [Google Scholar] [CrossRef] [PubMed]
- Lapola, D.M.; Schaldach, R.; Alcamo, J.; Dondeau, A.; Kock, J.; Koelking, C.; Priess, J.A. Indirect land-use changes can overcome carbon savings from biofuels in Brazil. Proc. Natl. Acad. Sci. USA 2010, 107, 3388–3393. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, H.K.; Johnston, M.; Foley, J.A.; Holloway, T.; Monfreda, C.; Ramankutty, N.; Zaks, D. Carbon payback times for crop-based biofuel expansion in the tropics: The effects of changing yield and technology. Environ. Res. Lett. 2008, 3, 034001. [Google Scholar] [CrossRef]
- Mello, F.F.C.; Cerri, C.E.P.; Davis, C.A.; Holbrook, N.M.; Paustian, K.; Maia, S.M.F.; Galdos, M.V.; Bernoux, M.; Cerri, C.C. Payback time for soil carbon and sugar-cane ethanol. Nat. Clim. Chang. 2014, 4, 605–609. [Google Scholar] [CrossRef]
- Alkimim, A.; Clarke, K.C. Land use change and the carbon debt for sugarcane ethanol production in Brazil. Land Use Policy 2016, 72, 65–73. [Google Scholar] [CrossRef]
- Franco, A.L.C.; Cherubin, M.R.; Pavinato, P.S.; Cerri, C.E.; Six, J.; Davies, C.A.; Cerri, C.C. Soil carbon, nitrogen and phosphorus changes under sugarcane expansion in Brazil. Sci. Total Environ. 2015, 515, 30–38. [Google Scholar] [CrossRef]
- Bordonal, R.O.; Lal, R.; Ronquim, C.C.; Figueiredo, E.B.; Carvalho, J.L.N.; Maldonado Jr., W.; Milori, D.M.B.P.; La Scala Jr., N. Changes in quantity and quality of soil carbon due to the land-use conversion to sugarcane (Saccharum officinarum) plantation in southern Brazil. Agric. Ecosyst. Environ. 2017, 240, 54–65. [Google Scholar] [CrossRef]
- Oliveira, D.M.S.; Williams, S.; Cerri, C.E.P.; Paustian, K. Predicting soil C changes over sugarcane expansion in Brazil using the DayCent model. GCB Bioenergy 2017, 9, 1436–1446. [Google Scholar] [CrossRef]
- Silva-Olaya, A.M.; Cerri, C.E.P.; Williams, S.; Cerri, C.C.; Davies, C.A.; Paustian, K. Modelling SOC response to land use change and management practices in sugarcane cultivation in South-Central Brazil. Plant. Soil 2017, 410, 483–498. [Google Scholar] [CrossRef]
- Oliveira, P.S.; Marquis, R.J. The Cerrados of Brazil: Ecology and Natural History of a Neotropical Savanna; Columbia University Press: New York, NY, USA, 2002. [Google Scholar]
- Carvalho, J.L.N.; Raucci, G.S.; Cerri, C.E.P.; Bernoux, M.; Feigl, B.J.; Wruck, F.J.; Cerri, C.C. Impact of pasture, agriculture and crop-livestock systems on soil C stocks in Brazil. Soil Tillage Res. 2010, 110, 175–186. [Google Scholar] [CrossRef]
- Lal, R. Soil carbon sequestration impacts on global climate change and food security. Science 2004, 304, 1623–1627. [Google Scholar] [CrossRef]
- Smith, P.; Davis, C.A.; Ogle, S.; Zanchi, G.; Bellarby, J.; Bird, N.; Boddey, R.M.; McNamara, N.P.; Powlson, D.; Cowie, A.; et al. Towards an integrated global framework to assess the impacts of land use and management change on soil carbon: Current capability and future vision. Glob. Chang. Biol. 2012, 18, 2089–2101. [Google Scholar] [CrossRef]
- Power, A.G. Ecosystem services and agriculture: Tradeoffs and synergies. Phil. Trans. R. Soc. B 2010, 365, 2959–2971. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.; Cotrufo, M.F.; Rumpel, C.; Paustian, K.; Kuikman, P.J.; Elliott, J.A.; McDowell, R.; Griffiths, R.I.; Asakawa, S.; Bustamante, M.; et al. Biogeochemical cycles and biodiversity as key drivers of ecosystem services provided by soils. Soil 2015, 2, 665–685. [Google Scholar] [CrossRef]
- Cherubin, M.R.; Karlen, D.L.; Cerri, C.E.P.; Franco, A.L.C.; Tormena, C.A.; Davies, C.A.; Cerri, C.C. Soil quality indexing strategies for evaluating sugarcane expansion in Brazil. PLoS ONE 2016, 11, e0150860. [Google Scholar] [CrossRef] [PubMed]
- Cherubin, M.R.; Karlen, D.L.; Franco, A.L.C.; Cerri, C.E.P.; Tormena, C.A.; Cerri, C.C. A Soil Management Assessment Framework (SMAF) Evaluation of Brazilian Sugarcane Expansion on Soil Quality. Soil Sci. Soc. Am. J. 2016, 80, 215–226. [Google Scholar] [CrossRef]
- Cherubin, M.R.; Karlen, D.L.; Franco, A.L.C.; Tormena, C.A.; Cerri, C.E.P.; Davies, C.A.; Cerri, C.C. Soil physical quality response to sugarcane expansion in Brazil. Geoderma 2016, 267, 156–168. [Google Scholar] [CrossRef]
- Luz, F.B.; Carvalho, M.L.; de Borba, D.A.; Schiebelbein, B.E.; de Lima, R.P.; Cherubin, M.R. Linking Soil Water Changes to Soil Physical Quality in Sugarcane Expansion Areas in Brazil. Water 2020, 12, 3156. [Google Scholar]
- Franco, A.L.C.; Bartz, M.L.C.; Cherubin, M.R.; Baretta, D.; Cerri, C.E.P.; Feigl, B.J.; Wall, D.H.; Davis, C.A.; Cerri, C.C. Loss of soil (macro)fauna due to the expansion of Brazilian sugarcane acreage. Sci. Total Environ. 2016, 563, 160–168. [Google Scholar] [CrossRef]
- Franco, A.L.C.; Cherubin, M.R.; Cerri, C.E.P.; Six, J.; Wall, D.H.; Cerri, C.C. Linking soil engineers, structural stability, and organic matter allocation to unravel soil carbon responses to land-use change. Soil Biol. Biochem. 2020, 150. [Google Scholar] [CrossRef]
- Barbosa, L.C.; Magalhães, P.S.G.; Bordonal, R.O.; Cherubin, M.R.; Castioni, G.A.F.; Tenelli, S.; Franco, H.C.J.; Carvalho, J.L.N. Soil physical quality associated with tillage practices during sugarcane planting in south-central Brazil. Soil Tillage Res. 2019, 195, 104383. [Google Scholar] [CrossRef]
- Lehmann, J.; Bossio, D.A.; Knabner, I.K.; Rillig, M.C. The concept and future prospects of soil health. Nat. Rev. Earth Environ. 2020. [Google Scholar] [CrossRef] [PubMed]
- Vera, I.; Wicke, B.; van der Hilst, F. Spatial variation in environmental impacts of sugarcane expansion in Brazil. Land 2020, 9, 397. [Google Scholar] [CrossRef]
- Filoso, S.; Carmo, J.B.D.; Mardegan, S.F.; Lins, S.R.M.; Gomes, T.F.; Martinelli, L.A. Reassessing the environmental impacts of sugarcane ethanol production in Brazil to help meet sustainability goals. Renew. Sustain. Energy Rev. 2015, 52, 1847–1856. [Google Scholar] [CrossRef]
- Silva-Olaya, A.M.; Cerri, C.E.P.; La Scala, N.; Dias, C.T.S.; Cerri, C.C. Carbon dioxide emissions under different soil tillage systems in mechanically harvested sugarcane. Environ. Res. Lett. 2013, 8. [Google Scholar] [CrossRef]
- La Scala, N.; Bolonhezi, D.; Pereira, G.T. Short-term soil CO2 emission after conventional and reduced tillage of a no-till sugar cane area in southern Brazil. Soil Tillage Res. 2006, 91, 244–248. [Google Scholar] [CrossRef]
- Bento, C.B.; Filoso, S.; Pitombo, L.M.; Cantarella, H.; Rossetto, R.; Martinelli, L.A.; Carmo, J.B. Impacts of sugarcane agriculture expansion over low-intensity cattle ranch pasture in Brazil on greenhouse gases. J. Environ. Manag. 2018, 206, 980–988. [Google Scholar] [CrossRef]
- Gunkel, G.; Kosmol, J.; Sobral, M.; Rohn, H.; Montenegro, S.; Aureliano, J. Sugar cane industry as a source of water pollution—Case study on the situation in Ipojuca river, Pernambuco, Brazil. Water Air Soil Pollut. 2007, 180, 261–269. [Google Scholar] [CrossRef]
- Taniwaki, R.H.; Cassiano, C.C.; Filoso, S.; Ferraz, S.F.d.; de Camargo, P.B.; Martinelli, L.A. Impacts of converting low-intensity pastureland to high-intensity bioenergy cropland on the water quality of tropical streams in Brazil. Sci. Total Environ. 2017, 584, 339–347. [Google Scholar] [CrossRef]
- Wagg, C.; Bender, S.F.; Widmer, F.; van der Heijden, M.G.A. Soil biodiversity and soil community composition determine ecosystem multifunctionality. Proc. Natl. Acad. Sci. USA 2014, 111, 5266–5270. [Google Scholar] [CrossRef] [PubMed]
- de Weill, M.A.M.; Sparovek, G. Erosion study in the ceveiro watershed (Piracicaba, SP). II—Interpreting soil loss tolerance using the Soil Useful Life Index methodology. Rev. Bras. Cienc. do Solo 2008, 32, 815–824. [Google Scholar] [CrossRef][Green Version]
- Youlton, C.; Wendland, E.; Anache, J.A.A.; Poblete-Echeverría, C.; Dabney, S. Changes in erosion and runoff due to replacement of pasture land with sugarcane crops. Sustainability 2016, 8, 685. [Google Scholar] [CrossRef]
- Gomes, T.F.; Van de Broek, M.; Govers, G.; Silva, R.W.C.; Moraes, J.M.; Camargo, P.B.; Mazzi, E.A.; Martinelli, L.A. Runoff, soil loss, and sources of particulate organic carbon delivered to streams by sugarcane and riparian areas: An isotopic approach. Catena 2018, 181, 104083. [Google Scholar] [CrossRef]
- Carvalho, J.L.N.; Nogueirol, R.C.; Menandro, L.M.S.; de Oliveira Bordonal, R.; Borges, C.D.; Cantarella, H.; Franco, H.C.J. Agronomic and environmental implications of sugarcane straw removal:A major review. GCB Bioenergy 2017, 9, 1181–1195. [Google Scholar] [CrossRef]
- Blanco-Canqui, H.; Ruis, S.J. No-tillage and soil physical environment. Geoderma 2018, 326, 164–200. [Google Scholar] [CrossRef]
- Luo, Z.; Wang, E.; Sun, O.J. Can no-tillage stimulate carbon sequestration in agricultural soils? A meta-analysis of paired experiments. Agric. Ecosyst. Environ. 2010, 139, 224–231. [Google Scholar] [CrossRef]
- Segnini, A.; Carvalho, J.L.N.; Bolonhezi, D.; Milori, D.M.B.P.; da Silva, W.T.L.; Simões, M.L.; Cantarella, H.; de Maria, I.C.; Martin-Neto, L. Carbon stock and humification index of organic matter affected by sugarcane straw and soil management. Sci. Agric. 2013, 70, 321–326. [Google Scholar] [CrossRef]
- Tenelli, S.; Bordonal, R.O.; Barbosa, L.C.; Carvalho, J.L.N. Can reduced tillage sustain sugarcane yield and soil carbon if straw is removed? Bioenergy Res. 2019, 12, 764–777. [Google Scholar] [CrossRef]
- Bai, X.; Huang, Y.; ren, W.; Coyne, M.; Jacinthe, P.A.; Tao, B.; Hui, D.; Yang, J.; Matocha, C. Responses of soil carbon sequestration to climate-smart agriculture practices: A meta-analysis. Glob. Chang. Biol. 2019, 25, 2591–2606. [Google Scholar] [CrossRef]
- Sun, W.; Canadell, J.G.; Yu, L.; Yu, L.; Zhang, W.; Smith, P.; Fischer, T.; Huang, Y. Climate drives global soil carbon sequestration and crop yield changes under conservation agriculture. Glob. Chang. Biol. 2020, 26, 3325–3335. [Google Scholar] [CrossRef] [PubMed]
- Busari, M.A.; Kukal, S.S.; Kaur, A.; Bhatt, R.; Dulazi, A.A. Conservation tillage impacts on soil, crop and the environment. Int. Soil Water Conserv. Res. 2015, 3, 119–129. [Google Scholar] [CrossRef]
- Zotarelli, L.; Zatorre, N.P.; Boddey, R.M.; Urquiaga, S.; Jantalia, C.P.; Franchini, J.C.; Alves, B.J.R. Influence of no-tillage and frequency of a green manure legume in crop rotations for balancing N outputs and preserving soil organic C stocks. F. Crop. Res. 2012, 132, 185–195. [Google Scholar] [CrossRef]
- Cerri, C.C.; Galdos, M.V.; Maia, S.M.F.; Bernoux, M.; Feigl, B.J.; Powlson, D.; Cerri, C.E.P. Effect of sugarcane harvesting systems on soil carbon stocks in Brazil: An examination of existing data. Eur. J. Soil Sci. 2011, 62, 23–28. [Google Scholar] [CrossRef]
- Chagas, M.F.; Bordonal, R.O.; Cavalett, O.; Carvalho, J.L.N.; Bonomi, A.; La Scala, N. Environmental and economic impacts of different sugarcane production systems in the ethanol biorefinery. Biofuels Bioprod. Biorefining 2016, 10, 89–106. [Google Scholar] [CrossRef]
- Blanco-Canqui, H.; Shaver, T.M.; Lindquist, J.L.; Shapiro, C.A.; Elmore, R.W.; Francis, C.A.; Hergert, G.W. Cover crops and ecosystem services: Insights from studies in temperate soils. Agron. J. 2015, 107, 2449–2474. [Google Scholar] [CrossRef]
- Kaye, J.P.; Finney, D.; White, C.; Bradley, B.; Schipanski, M.; Alonso-Ayuso, M.; Hunter, M.; Burgess, M.; Mejia, C. Managing nitrogen through cover crop species selection in the U.S. Mid-Atlantic. PLoS ONE 2019, 14, 1–23. [Google Scholar] [CrossRef]
- White, C.M.; DuPont, S.T.; Hautau, M.; Hartman, D.; Finney, D.; Bradley, B.; LaChance, J.C.; Kaye, J.P. Managing the trade off between nitrogen supply and retention with cover crop mixtures. Agric. Ecosyst. Environ. 2017, 237, 121–133. [Google Scholar] [CrossRef]
- Farhate, C.V.V.; Souza, Z.M.; Cherubin, M.R.; Lovera, L.H.; Oliveira, I.N.; Carneiro, M.P.; >La Scala, N., Jr. Abiotic Soil Health Indicators that Respond to Sustainable Management Practices in Sugarcane Cultivation. Sustainability 2020, 12, 9407. [Google Scholar] [CrossRef]
- Ambrosano, E.J.; Cantarella, H.; Ambrosano, G.M.B.; Schammas, E.A.; Dias, F.L.F.; Rossi, F.; Trivelin, P.C.O.; Muraoka, T.; Azcón, R. Produtividade da cana-de-açúcar após o cultivo de leguminosas. Bragantia 2011, 70, 810–818. [Google Scholar] [CrossRef]
- Park, S.E.; Webster, T.J.; Horan, H.L.; James, A.T.; Thorburn, P.J. A legume rotation crop lessens the need for nitrogen fertiliser throughout the sugarcane cropping cycle. F. Crop. Res. 2010, 119, 331–341. [Google Scholar] [CrossRef]
- Garside, A.L.; Bell, M. Fallow legume in the Australian sugar industry review of recent research findings and implications for the sugarcane cropping system. Proc. Aust. Soc. Sugar Cane Technol. 2001, 23, 230–235. [Google Scholar]
- Menandro, L.M.S.; Cantarella, H.; Franco, H.C.J.; Kölln, O.T.; Pimenta, M.T.B.; Sanches, G.M.; Rabelo, S.C.; Carvalho, J.L.N. Comprehensive assessment of sugarcane straw: Implications for biomass and bioenergy production. Biofuels Bioprod. Biorefining 2017, 11, 488–504. [Google Scholar] [CrossRef]
- Lisboa, I.P.; Cherubin, M.R.; Lima, R.P.; Cerri, C.C.; Satiro, L.S.; Wienhold, B.J.; Schmer, M.R.; Jin, V.L.; Cerri, C.E.P. Sugarcane straw removal effects on plant growth and stalk yield. Ind. Crops Prod. 2017, 111, 794–806. [Google Scholar] [CrossRef]
- de Aquino, G.S.; Medina, C.C.; Shahab, M.; Santiago, A.D.; Cunha, A.C.B.; Kussaba, D.A.O.; Carvalho, J.B.; Moreira, A. Does straw mulch partial-removal from soil interfere in yield and industrial quality sugarcane? A long term study. Ind. Crops Prod. 2018, 111, 573–578. [Google Scholar] [CrossRef]
- Carvalho, J.L.N.; Menandro, L.M.S.; de Castro, S.G.Q.; Cherubin, M.R.; de Oliveira Bordonal, R.; Barbosa, L.C.; Gonzaga, L.C.; Tenelli, S.; Franco, H.C.J.; Kolln, O.T.; et al. Multilocation Straw Removal Effects on Sugarcane Yield in South-Central Brazil. Bioenergy Res. 2019, 12, 813–829. [Google Scholar] [CrossRef]
- Bordonal, R.O.; Menandro, L.M.S.; Barbosa, L.C.; Lal, R.; Milori, D.M.B.P.; Kolln, O.T.; Franco, H.C.J.; Carvalho, J.L.N. Sugarcane yield and soil carbon response to straw removal in south-central Brazil. Geoderma 2018, 328, 79–90. [Google Scholar] [CrossRef]
- Morais, M.C.; Siqueira-Neto, M.; Guerra, H.P.; Satiro, L.S.; Soltangheisi, A.; Cerri, C.E.P.; Feigl, B.J.; Cherubin, M.R. Trade-offs between sugarcane straw removal and soil organic matter in Brazil. Sustainability 2020, 12, 9363. [Google Scholar] [CrossRef]
- Ruiz-Corrêa, S.T.; Barbosa, L.C.; Menandro, L.M.S.; Scarpare, F.V.; Reichardt, K.; Moraes, L.O.; Hernandes, T.A.D.; Franco, H.C.J.; Carvalho, J.L.N. Straw Removal Effects on Soil Water Dynamics, Soil Temperature, and Sugarcane Yield in South-Central Brazil. Bioenergy Res. 2019, 12, 749–763. [Google Scholar] [CrossRef]
- Gmach, M.R.; Scarpare, F.V.; Cherubin, M.R.; Lisboa, I.P.; Santos, A.K.B.; Cerri, C.E.P.; Cerri, C.C. Sugarcane straw removal effects on soil water storage and drainage in southeastern Brazil. J. Soil Water Conserv. 2019, 74, 466–476. [Google Scholar] [CrossRef]
- Castioni, G.A.; Cherubin, M.R.; Menandro, L.M.S.; Sanches, G.M.; de Oliveira Bordonal, R.; Barbosa, L.C.; Franco, H.C.J.; Carvalho, J.L.N. Soil physical quality response to sugarcane straw removal in Brazil: A multi-approach assessment. Soil Tillage Res. 2018, 184, 301–309. [Google Scholar] [CrossRef]
- Filho, M.V.M.; Liccioti, T.T.; Pereira, G.T.; Marques, J.M.; Sanchez, R.B. Perdas de solo e nutrientes por erosão num Argissolo com resíduos vegetais de cana-de-açúcar. Eng. Agric. 2009, 29, 8–18. [Google Scholar] [CrossRef]
- Morais, M.C.; Ferrari, B.M.; Borges, C.D.; Cherubin, M.R.; Tsai, S.M.; Cerri, C.C.; Cerri, C.E.P.; Feigl, B.J. Does Sugarcane Straw Removal Change the Abundance of Soil Microbes? Bioenergy Res. 2019, 12, 901–908. [Google Scholar] [CrossRef]
- Menandro, L.M.S.; de Moraes, L.O.; Borges, C.D.; Cherubin, M.R.; Castioni, G.A.; Carvalho, J.L.N. Soil Macrofauna Responses to Sugarcane Straw Removal for Bioenergy Production. Bioenergy Res. 2019, 12, 944–957. [Google Scholar] [CrossRef]
- Hassuani, S.; Leal, M.R.L.V.; Macedo, I. Biomass Power Generation: Sugar Cane, Bagasse and Trash; CTC: Piracicaba, Brazil, 2005. [Google Scholar]
- De Castro, S.G.Q.; Dinardo-Miranda, L.L.; Fracasso, J.V.; Bordonal, R.O.; Menandro, L.M.S.; Franco, H.C.J.; Carvalho, J.L.N. Changes in Soil Pest Populations Caused by Sugarcane Straw Removal in Brazil. Bioenergy Res. 2019, 12, 878–887. [Google Scholar] [CrossRef]
- Gonzaga, L.C.; Zotelli, L.C.; Castro, S.G.Q.; Oliveira, B.G.; Bordonal, R.O.; Cantarella, H.; Carvalho, J.L.N. Implications of Sugarcane Straw Removal for Soil Greenhouse Gas Emissions in São Paulo State, Brazil. Bioenergy Res. 2019, 12, 843–857. [Google Scholar] [CrossRef]
- Vasconcelos, A.L.S.; Cherubin, M.R.; Feigl, B.J.; Cerri, C.E.P.; Gmach, M.R.; Siqueira-Neto, M. Greenhouse gas emission responses to sugarcane straw removal. Biomass Bioenergy 2018, 113, 15–21. [Google Scholar] [CrossRef]
- Cherubin, M.R.; Oliveira, D.M.D.S.; Feigl, B.J.; Pimentel, L.G.; Lisboa, I.P.; Gmach, M.R.; Varanda, L.L.; Morais, M.C.; Satiro, L.S.; Popin, G.V.; et al. Crop residue harvest for bioenergy production and its implications on soil functioning and plant growth: A review. Sci. Agric. 2018, 75, 255–272. [Google Scholar] [CrossRef]
- Leal, M.R.L.V.; Galdos, M.V.; Scarpare, F.V.; Seabra, J.E.A.; Walter, A.; Oliveira, C.O.F. Sugarcane straw availability, quality, recovery and energy use: A literature review. Biomass Bioenergy 2013, 53, 11–19. [Google Scholar] [CrossRef]
- Silva, A.G.B.; Lisboa, I.P.; Cherubin, M.R.; Cerri, C.E.P. How Much Sugarcane Straw is Needed for Covering the Soil? Bioenergy Res. 2019, 12, 858–864. [Google Scholar] [CrossRef]
- Carvalho, J.L.N.; Hudiburg, T.W.; Franco, H.C.J.; DeLucia, E.H. Contribution of above- and belowground bioenergy crop residues to soil carbon. GCB Bioenergy 2017, 9, 1333–1343. [Google Scholar] [CrossRef]
- Junior, J.G.d.A.S.; Cherubin, M.R.; Oliveira, B.G.; Cerri, C.E.P.; Cerri, C.C.; Feigl, B.J. Three-Year Soil Carbon and Nitrogen Responses to Sugarcane Straw Management. Bioenergy Res. 2018, 11, 249–261. [Google Scholar] [CrossRef]
- Weiler, D.A.; Moro, W.J.; Awe, G.O.; Oliveira, D.M.S.; Cerri, C.E.P.; Reichert, J.M.; Giacomini, S.J. Carbon Balance in Sugarcane Areas Under Different Tillage Systems. Bioenergy Res. 2019, 12, 778–788. [Google Scholar] [CrossRef]
- Carmo, J.B.d.; Filoso, S.; Zotelli, L.C.; Sousa Neto, E.R.; Pitombo, L.M.; Duarte-Neto, P.J.; Vargas, V.P.; Andrade, C.A.; Gava, G.J.; Rosseto, R.; et al. Infield greenhouse gas emissions from sugarcane soils in brazil: Effects from synthetic and organic fertilizer application and crop trash accumulation. GCB Bioenergy 2013, 5, 267–280. [Google Scholar] [CrossRef]
- Pitombo, L.M.; Cantarella, H.; Packer, A.P.C.; Ramos, N.P.; do Carmo, J.B. Straw preservation reduced total N2O emissions from a sugarcane field. Soil Use Manag. 2017, 33, 583–594. [Google Scholar] [CrossRef]
- IPPC. Climate Change 2014—Synthesis Report, Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Core Writing Team, Pachauri, R.K., Meyer, L.A., Eds.; IPCC: Geneva, Switzerland, 2014. [Google Scholar]
- Snyder, C.S.; Bruulsema, T.W.; Jensen, T.L.; Fixen, P.E. Review of greenhouse gas emissions from crop production systems and fertilizer management effects. Agric. Ecosyst. Environ. 2009, 133, 247–266. [Google Scholar] [CrossRef]
- Lisboa, C.C.; Butterbach-Bahl, K.; Mauder, M.; Kiese, R. Bioethanol production from sugarcane and emissions of greenhouse gases—known and unknowns. GCB Bioenergy 2011, 3, 277–292. [Google Scholar] [CrossRef]
- Jantalia, C.P.; Santos, H.P.D.; Urquiaga, S.; Boddey, R.M.; Alves, B.J.R. Fluxes of nitrous oxide from soil under different crop rotations and tillage systems in the South of Brazil. Nutr. Cycl. Agroecosyst. 2008, 82, 161–173. [Google Scholar] [CrossRef]
- Soares, J.R.; Cassman, N.A.; Kielak, A.M.; Pijl, A.; Carmo, J.B. Nitrous oxide emission related to ammonia-oxidizing bacteria and mitigation options from N fertilization in a tropical soil. Sci. Rep. 2016, 6, 30349. [Google Scholar] [CrossRef]
- Mutton, M.; Rossetto, R.; Mutton, M.J.R. Agricultural use of stillage. In Sugarcane Bioethano; Cortez, L.A.B., Ed.; Blucher: São Paulo, Brazil, 2010; pp. 381–403. [Google Scholar]
- Lourenço, K.S.; Rosseto, R.; Vitti, A.C.; Montezano, Z.F.; Soares, J.R.; Sousa, R.M.; Carmo, J.B.; Kuramae, E.E.; Cantarella, H. Strategies to mitigate the nitrous oxide emissions from nitrogen fertilizer applied with organic fertilizers in sugarcane. Sci. Total Environ. 2019, 650, 1476–1486. [Google Scholar] [CrossRef]
- Neto, M.S.; Galdos, M.V.; Feigl, B.J.; Cerri, C.E.P.; Cerri, C.C. Direct N2O emission factors for synthetic N-fertilizer and organic residues applied on sugarcane for bioethanol production in Central-Southern Brazil. GCB Bioenergy 2016, 8, 269–280. [Google Scholar] [CrossRef]
- Cantarella, H.; de Oliveira, B.G.; Lourenço, K.S.; Soares, J.R.; Gonzaga, L.C.; Carvalho, J.L.N. Nitrification inhibitors reduce the carbon footprint of sugarcane production. Pak. Sugar J. 2020, 35, 26. [Google Scholar]
- Gonzaga, L.C.; Carvalho, J.L.N.; de Oliveira, B.G.; Soares, J.R.; Cantarella, H. Crop residue removal and nitrification inhibitor application as strategies to mitigate N2O emissions in sugarcane fields. Biomass Bioenergy 2018, 119, 206–216. [Google Scholar] [CrossRef]
- Cantarella, H.; Rossetto, R. Fertilizer concerns. In Sustainability of Sugarcane Bioenergy; Poppe, M.K., Cortez, L.A.B., Eds.; CGEE: Brasilia—DF, Brasil, 2012; pp. 71–94. [Google Scholar]
- Rossetto, R.; Crusciol, C.A.C.; Cantarella, H.; Carmo, J.B.d.; Nascimento, C.A.C. Residues uses and environment sustainability. In Sustainable Sugarcane Production; Singh, P., Tiwari, A.K., Eds.; Apple Academic Press: Waretown, NJ, USA, 2018; pp. 162–187. [Google Scholar]
- Câmara, E.; Pinto, R.C.A.; Rocha, J.C. Setting process on mortars containing sugarcane bagasse ash. Rev. IBRACON Estruturas Mater. 2016, 9, 617–629. [Google Scholar] [CrossRef][Green Version]
- CETESB Norma P4.231: Stillage—Criteria and procedures for agricultural soil application. Cia. Ambient. Estado São Paulo 2015, 3, 1–15.
- Brandani, C.B.; Abbruzzini, T.F.; Williams, S.; Easter, M.; Cerri, C.E.P.; Paustian, K. Simulation of management and soil interactions impacting SOC dynamics in sugarcane using the CENTURY Model. GCB Bioenergy 2015, 7, 646–657. [Google Scholar] [CrossRef]
- Zani, C.F.; Barneze, A.S.; Robertson, A.D.; Keith, A.M.; Cerri, C.E.P.; McNamara, N.P.; Cerri, C.C. Vinasse application and cessation of burning in sugarcane management can have positive impact on soil carbon stocks. PeerJ 2018, 8, 1–30. [Google Scholar] [CrossRef]
- Van Raij, B.; Cantarella, H.; Quaggio, J.; Furlani, A.M. Recomendações de Adubação e Calagem para o Estado de São Paulo; Instituto Agronômico de Campinas: Campinas, São Paulo, Brazil, 1997. [Google Scholar]
- Borges, B.M.M.N.; Strauss, M.; Camelo, P.A.; Sohi, S.P.; Franco, H.C.J. Re-use of sugarcane residue as a novel biochar fertilizer—Increased phosphorus use efficiency and plant yield. J. Clean. Prod. 2020, 262, 121406. [Google Scholar] [CrossRef]
- Spokas, K.A.; Reicosky, D.C. Impacts of sixteen different biochars on soil greenhouse gas production. Ann. Environ. Sci. 2009, 3, 179–193. Available online: https://pubag.nal.usda.gov/download/47667/PDF (accessed on 10 January 2021).
- Scheer, C.; Grace, P.R.; Rowlings, D.W.; Kimber, S.; van Zwieten, L. Effect of biochar amendment on the soil-atmosphere exchange of greenhouse gases from an intensive subtropical pasture in northern New South Wales, Australia. Plant. Soil 2011, 345, 47–58. [Google Scholar] [CrossRef]
- Cayuela, M.L.; Sánchez-Monedero, M.A.; Roig, A.; Hanley, K.; Enders, A.; Lehmann, J. Biochar and denitrification in soils: When, how much and why does biochar reduce N2O emissions? Sci. Rep. 2013, 3, 1732. [Google Scholar] [CrossRef]
- Abbruzzini, T.F.; Zenero, M.D.O.; de Andrade, P.A.M.; Andreote, F.D.; Campo, J.; Cerri, C.E.P. Effects of Biochar on the Emissions of Greenhouse Gases from Sugarcane Residues Applied to Soils. Agric. Sci. 2017, 08, 869–886. [Google Scholar] [CrossRef]
- Lefebvre, D.; Williams, A.; Meersmans, J.; Kirk, G.J.D.; Sohi, S.; Goglio, P.; Smith, P. Modelling the potential for soil carbon sequestration using biochar from sugarcane residues in Brazil. Sci. Rep. 2020, 10, 19479. [Google Scholar] [CrossRef] [PubMed]
- Brazil Lei 12651 de 25 de Maio de 2012—Código Florestal Brasileiro. 2012. Available online: http://www.planalto.gov.br/ccivil_03/_ato2011-2014/2012/lei/l12651.htm (accessed on 19 November 2020).
- Strassburg, B.B.N.; Beyer, H.L.; Crouzeilles, R.; Iribarrem, A.; Barros, F.; Siqueira, M.F.; Sánchez-Tapia, A.; Balmford, A.; Sansevero, J.B.B.; Brancalion, P.H.S.; et al. Strategic approaches to restoring ecosystems can triple conservation gains and halve costs. Nat. Ecol. E 2019, 3, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Tavares, P.A.; Brites, A.D.; Sparovek, G.; Guidotti, V.; Cerignoni, F.; Aguiar, D.; Metzger, J.P.; Rodrigues, R.R.; Pinto, L.F.G.; Mello, K.; et al. Unfolding additional massive cutback effects of the Native Vegetation Protection Law on Legal Reserves, Brazil. Biota Neotrop. 2019, 19, e20180658. [Google Scholar] [CrossRef]
- Rodrigues, R.R.; Gandolfi, S.; Nave, A.G.; Aronson, J.; Barreto, T.E.; Vidal, C.Y.; Brancalion, P.H.S. Large-scale ecological restoration of high-diversity tropical forests in SE Brazil. For. Ecol. Manag. 2011, 261, 1605–1613. [Google Scholar] [CrossRef]
- De Freitas, F.L.M.; Sparovek, G.; Mörtberg, U.; Silveira, S.; Klug, I.; Berndes, G. Offsetting legal deficits of native vegetation among Brazilian landholders: Effects on nature protection and socioeconomic development. Land Use Policy 2017, 68, 189–199. [Google Scholar] [CrossRef]
- Grassi, M.C.B.; Pereira, G.A.G. Energy-cane and RenovaBio: Brazilian vectors to boost the development of Biofuels. Ind. Crops Prod. 2019, 129, 201–205. [Google Scholar] [CrossRef]
- MME—Brazil Resolução N°15, de 18 de Agosto de 2020 (Renovabio)—Decarbonization Targets. Ministério de Minas e Energia—CNPE: Conselho Nacional de Política Energética. 2020. Available online: https://www.gov.br/mme/pt-br/assuntos/conselhos-e-comites/cnpe/resolucoes-do-cnpe/arquivos/2020/resolucao_8_cnpe_metas_compulsorias.pdf (accessed on 25 November 2020).
- Valor Economico RenovaBio Credits Start to Take off. 2020. Available online: https://valor.globo.com/agronegocios/noticia/2020/10/19/creditos-do-renovabio-comecam-a-deslanchar.ghtml (accessed on 19 October 2020).
- Trindade, S.C.; Nogueira, L.A.H.; Souza, G.M. Relevance of LACAf biofuels for global sustainability. Biofuels 2019, 1–11. [Google Scholar] [CrossRef]
- Bioenergy for Sustainable Development in Africa; Janssen, R., Rutz, D., Eds.; Springer: Dordrecht, The Netherlands, 2012. [Google Scholar]
- Sugarcane Bioenergy in Southern Africa: Economic Potential for Sustainable Scale-up; IRENA: Abu Dhabi, UAE, 2019.
- Sugarcane Bioenergy for Sustainable Development—Expanding Production in Latin America and Africa, 1st ed.; Cortez, L.A.B., Leal, M.R.L.V., Nogueira, L.A.H., Eds.; Routledge: Abingdon, UK, 2019. [Google Scholar]
- Watson, H.K. Potential to expand sustainable bioenergy from sugarcane in southern Africa. Energy Policy 2011, 39, 5746–5750. [Google Scholar] [CrossRef]
- Souza, S.P.; Nogueira, L.A.H.; Watson, H.K.; Lynd, L.R.; Elmissiry, M.; Cortez, L.A.B. Potential of sugarcane in modern energy development in Southern Africa. Front. Energy Res. 2016, 4, 1–11. [Google Scholar] [CrossRef]
- Leal, M.R.L.V.; Leite, J.G.D.B.; Chagas, M.F.; da Maia, R.; Cortez, L.A.B. Feasibility assessment of converting sugar mills to bioenergy production in Africa. Agriculture 2016, 6, 45. [Google Scholar] [CrossRef]
- Souza, S.P.; Nogueira, L.A.H.; Martinez, J.; Cortez, L.A.B. Sugarcane can afford a cleaner energy profile in Latin America & Caribbean. Renew. Energy 2018, 121, 164–172. [Google Scholar] [CrossRef]
- Dallemand, J.F.; Hilbert, J.A.; Georgakaki, A.; Monforti, F. Bioenergy and Latin America: A Multi-Country Perspective; Publications Office of the European Union: Luxembourg, 2015. [Google Scholar]
- Dufey, A.; Stange, D. Estudio Regional Sobre la Economía de los Biocombustibles en 2010: Temas Clave Para los Países de América Latina y el Caribe. Santiago de Chile, 2010. Available online: http://repositorio.cepal.org/bitstream/handle/11362/3906/LCW412_es.pdf?sequence=1 (accessed on 10 January 2021).
- Compodónico, M.; Diaz, M.A.; Veras, A.I.; Cárceres, R. Experiencia y Perspectivas em América Latina sobre Alcohol Carburante; IICA: Mexico City, Mexico, 1984. [Google Scholar]
- Baulcombe, D.; Crute, I.; Davies, B.; Dunwell, J.; Gale, M.; Jones, J.; Pretty, J.; Sutherland, W.; Toulmin, C. Reaping the Benefits: Science and the Sustainable Intensification of Global Agriculture; The Royal Society: London, UK, 2009. [Google Scholar]
- BNDES/CGEE Sugarcane-based Bioethanol: Energy for Sustainable Development Rio de Janeiro. 2008. Available online: https://web.bndes.gov.br/bib/jspui/bitstream/1408/6305/1/2008_Sugarcane-based%20bioethanol_energy%20for%20sustainable_P.pdf (accessed on 10 January 2021).
- Masih, I.; Maskey, S.; Mussá, F.E.F.; Trambauer, P. A review of droughts on the African continent: A geospatial and long-term perspective. Hydrol. Earth Syst. Sci. 2014, 18, 3635–3649. [Google Scholar] [CrossRef]
- Berenguer, E.; Ferreira, J.; Gardner, T.A.; Aragão, L.E.O.C.; Camargo, P.B.; Cerri, C.E.P.; Durigan, M.; Oliveira Junior, R.C.; Vieira, I.C.G.; Barlow, J. A large-scale field assessment of carbon stocks in human-modified tropical forests. Glob. Chang. Biol. 2014, 20, 3713–3726. [Google Scholar] [CrossRef] [PubMed]
| By-Product (*) | Amount Recycled | Recycled Nutrients and Organic Matter (**) | |||
|---|---|---|---|---|---|
| N | P | K | Organic Matter | ||
| Mg or m3 | Mg year−1 | ||||
| Filter cake | 10,500 | 15 | 89 | 4 | 3150 |
| Ash | 12,000 | 0 | 36 | 132 | 0 |
| Vinasse | 1,274,000 | 510 | 111 | 2123 | 35,672 |
| Total | - | 524 | 235 | 2259 | 38,822 |
| Region | Land under Permanent Meadows and Pastures (2018) (FAO, [6]) | Sugarcane Production (2018) [A] (FAO, [6]) | Sugarcane Potential Production on 1% of Pasture Land [B] | Potential Annual Bioenergy Production | |||
|---|---|---|---|---|---|---|---|
| Ethanol | Electricity | ||||||
| 1000 ha | % Country Area | 1000 Mg | 1000 Mg | % Increase (A/B) | 1000 m3 | MWh | |
| SSA (20 countries) (*) | 331,227 | 33% | 43,217 | 281,543 | 651% | 23,931 | 30,970 |
| LAC (18 countries) (**) | 499,931 | 28% | 975,640 | 424,941 | 44% | 36,120 | 46,744 |
| LAC w/o Brazil (17 countries) | 326,570 | 34% | 228,812 | 277,584 | 121% | 23,595 | 30,534 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cherubin, M.R.; Carvalho, J.L.N.; Cerri, C.E.P.; Nogueira, L.A.H.; Souza, G.M.; Cantarella, H. Land Use and Management Effects on Sustainable Sugarcane-Derived Bioenergy. Land 2021, 10, 72. https://doi.org/10.3390/land10010072
Cherubin MR, Carvalho JLN, Cerri CEP, Nogueira LAH, Souza GM, Cantarella H. Land Use and Management Effects on Sustainable Sugarcane-Derived Bioenergy. Land. 2021; 10(1):72. https://doi.org/10.3390/land10010072
Chicago/Turabian StyleCherubin, Maurício Roberto, João Luís Nunes Carvalho, Carlos Eduardo Pellegrino Cerri, Luiz Augusto Horta Nogueira, Glaucia Mendes Souza, and Heitor Cantarella. 2021. "Land Use and Management Effects on Sustainable Sugarcane-Derived Bioenergy" Land 10, no. 1: 72. https://doi.org/10.3390/land10010072
APA StyleCherubin, M. R., Carvalho, J. L. N., Cerri, C. E. P., Nogueira, L. A. H., Souza, G. M., & Cantarella, H. (2021). Land Use and Management Effects on Sustainable Sugarcane-Derived Bioenergy. Land, 10(1), 72. https://doi.org/10.3390/land10010072







