Evolution of Uranium Isotopic Compositions of the Groundwater and Rock in a Sandy-Clayey Aquifer
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Porcelli, D. Investigating groundwater processes using U- and Th-series nuclides. Radioact. Environ. 2008, 13, 105–153. [Google Scholar]
- Baskaran, M. Handbook of Environmental Isotope Geochemistry; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Dhaoui, Z.; Chkir, N.; Zouari, K.; Hadj Ammar, F.; Agoune, A. Investigation of uranium geochemistry along groundwater flow path in the Continental Intercalaire aquifer (Southern Tunisia). J. Environ. Radioact. 2016, 157, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Tan, L.; Liu, Q.; Li, R.; Li, Z.; Zhang, H.; Hu, S.; Liu, L.; Wang, J. Biosorption characteristics of Uranium (VI) from aqueous solution by pollen pini. J. Environ. Radioact. 2015, 150, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Yi, Z.; Yao, J.; Chen, H.-L.; Wang, F.; Yuan, Z.; Liu, X. Uranium biosorption from aqueous solution onto Eichhornia crassipes. J. Environ. Radioact. 2016, 154, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Manickum, T.; John, W.; Terry, S.; Hodgson, K. Preliminary study on the radiological and physicochemical quality of the Umgeni Water catchments and drinking water sources in KwaZulu-Natal, South Africa. J. Environ. Radioact. 2014, 137, 227–240. [Google Scholar] [CrossRef] [PubMed]
- Hall, S.M.; Mihalasky, M.J.; Tureck, K.R.; Hammarstrom, J.M.; Hannon, M.T. Genetic and grade and tonnage models for sandstone-hosted roll-type uranium deposits, Texas Coastal Plain. Ore Geol. Rev. 2017, 80, 716–753. [Google Scholar] [CrossRef]
- Cuvier, A.; Panza, F.; Pourcelot, L.; Foissard, B.; Cagnat, X.; Prunier, J.; van Beek, P.; Souhaut, M.; Le Roux, G. Uranium decay daughters from isolated mines: Accumulation and sources. J. Environ. Radioact. 2015, 149, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Keatley, A.C.; Scott, T.B.; Davis, S.; Jones, C.P.; Turner, P. An investigation into heterogeneity in a single vein-type uranium ore deposit: Implications for nuclear forensics. J. Environ. Radioact. 2015, 150, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Dosseto, A.; Schaller, M. The erosion response to Quaternary climate change quantified using uranium isotopes and in situ-produced cosmogenic nuclides. Earth-Sci. Rev. 2016, 155, 60–81. [Google Scholar] [CrossRef]
- Jamieson, R.A.; Baldini, J.U.L.; Brett, M.J.; Taylor, J.; Ridley, H.E.; Ottley, C.J.; Prufer, K.M.; Wassenburg, J.A.; Scholz, D.; Breitenbach, S.F.M. Intra- and inter-annual uranium concentration variability in a Belizean stalagmite controlled by prior aragonite precipitation: A new tool for reconstructing hydro-climate using aragonitic speleothems. Geochim. Cosmochim. Acta 2016, 190, 332–346. [Google Scholar] [CrossRef]
- Wang, Q.; Song, J.; Li, X.; Yuan, H.; Li, N.; Cao, L. Environmental evolution records reflected by radionuclides in the sediment of coastal wetlands: A case study in the Yellow River Estuary wetland. J. Environ. Radioact. 2016, 162–163, 87–96. [Google Scholar] [CrossRef] [PubMed]
- International Atomic Energy Agency. World Distribution of Uranium Deposits (UDEPO), with Uranium Deposit Classification; IAEA-TECDOC-1629; IAEA: Vienna, Austria, 2009. [Google Scholar]
- Malov, A.I. Estimation of uranium migration parameters in sandstone aquifers. J. Environ. Radioact. 2016, 153, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Malov, A.I.; Bolotov, I.N.; Pokrovsky, O.S.; Zykov, S.B.; Tokarev, I.V.; Arslanov, K.A.; Druzhinin, S.V.; Lyubas, A.A.; Gofarov, M.Y.; Kostikova, I.A.; et al. Modeling past and present activity of a subarctic hydrothermal system using O, H, C, U and Th isotopes. Appl. Geochem. 2015, 63, 93–104. [Google Scholar] [CrossRef]
- Han, L.F.; Plummer, N. A review of single-sample-based models and other approaches for radiocarbon dating of dissolved inorganic carbon in groundwater. Earth-Sci. Rev. 2016, 152, 119–142. [Google Scholar] [CrossRef]
- Malov, A.I. The use of the geological benchmarks to assess the residence time of groundwater in the aquifer using uranium isotopes on the example of the Northern Dvina basin. Lithol. Miner. Resour. 2013, 48, 254–265. [Google Scholar] [CrossRef]
- Ivanovich, M.; Fröhlich, K.; Hendry, M.J. Uranium-series radionuclides in fluids and solids, Milk River aquifer, AB, Canada. Appl. Geochem. 1991, 6, 405–418. [Google Scholar] [CrossRef]
- Grazhdankin, D.V.; Podkovyrov, V.N.; Maslov, A.V. Paleoclimatic Environments of the Formation of Upper Vendian Rocks on the Belomorian-Kuloi Plateau, Southeastern White Sea Region. Lithol. Miner. Resour. 2005, 40, 232–244. [Google Scholar] [CrossRef]
- Rozanov, A.Y.; Bessonova, V.Y.; Brangulis, A.P.; Giants, V.A. Paleogeography and Lithology of the Vendian and Cambrian of the Western Part of the Eastern European Platform; Nauka: Moscow, Russia, 1980. [Google Scholar]
- Barnes, C.E.; Cochran, J.K. Uranium geochemistry in estuarine sediments: Controls on removal and release processes. Geochim. Cosmochim. Acta 1993, 57, 555–589. [Google Scholar] [CrossRef]
- Mikhailov, B.M. The Forecast of the Supergene Zones at Solid Minerals; VSEGEI: St. Petersburg, Russia, 1998. [Google Scholar]
- Shore, G.M.; Starchenko, V.V.; Myronyuk, E.P.; Kudryavtsev, V.E.; Radyukevich, N.M.; Alekseenko, V.D.; Shipov, R.V. Requirements to Maps of the Ore-Bearing of the Supergene Zones; VSEGEI: St. Petersburg, Russia, 2005. [Google Scholar]
- Goretsky, G.I. Alluvium of the Great Anthropogenic Paleorivers of the Russian Plain; Nauka: Moscow, Russia, 1964. [Google Scholar]
- Kashtanov, S.G. The geological data of Pliocene age of the Kazanka and Sviyaga rivers valleys. Uchenye Zap. Kazan. Univ. 1954, 114, 155–163. [Google Scholar]
- Kashtanov, S.G. New data to the history of the Paleo-Kama. Dokl. Earth Sci. 1956, 106, 708–711. [Google Scholar]
- Malov, A.I. Water-Rock Interaction in Vendian Sandy-Clayey Rocks of the Mezen Syneclise. Lithol. Miner. Resour. 2004, 39, 345–356. [Google Scholar] [CrossRef]
- Zverev, V.P.; Malov, A.I.; Kostikova, I.A. Geochemical state groundwater of the active water exchange zone at the Lomonosov diamond deposit. Geoecology 2005, 4, 298–303. [Google Scholar]
- Cheng, H.; Edwards, R.L.; Hoff, J.; Gallupb, C.D.; Richardsc, D.A.; Asmeromd, A. The half-lives of uranium-234 and thorium-230. Chem. Geol. 2000, 169, 17–33. [Google Scholar] [CrossRef]
- Head, M.J.; Gibbard, P.L. Early-Middle Pleistocene transitions: Linking terrestrial and marine realms. Quat. Int. 2015, 389, 7–46. [Google Scholar] [CrossRef]
- Azzaroli, A.; De Giuli, C.; Ficcarelli, G.; Torre, D. Late pliocene to early mid-pleistocene mammals in Eurasia: Faunal succession and dispersal events. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1988, 66, 1–143. [Google Scholar] [CrossRef]
- Markova, A.K.; Vislobokova, I.A. Mammal faunas in Europe at the end of the Early—Beginning of the Middle Pleistocene. Quat. Int. 2016, 420, 363–377. [Google Scholar] [CrossRef]
- Muttonia, G.; Giancarlo Scardiab, G.; Kentc, D.V. Human migration into Europe during the late Early Pleistocene climate transition. Palaeogeogr. Palaeoclimatol. Palaeoecol. 2010, 296, 79–93. [Google Scholar] [CrossRef]
- Ehlers, J.; Astakhov, V.; Gibbard, P.L.; Mangerud, J.; Svendsen, J.I. GLACIATIONS|Middle Pleistocene in Eurasia. In Reference Module in Earth Systems and Environmental Sciences, Encyclopedia of Quaternary Science, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2013; pp. 172–179. [Google Scholar]
- Lisitsyn, A.P. (Ed.) The White Sea System; Nauchn. Mir: Moscow, Russia, 2010. (In Russian) [Google Scholar]
- Molodkov, A.N.; Bolikhovskaya, N.S. Long-term palaeoenvironmental changes recorded in palynologically studied loess-palaeosol and ESR-dated marine deposits of Northern Eurasia: Implications for sea–land correlation. Quat. Int. 2006, 152, 37–47. [Google Scholar] [CrossRef]
- Glaznev, V.N.; Kukkonen, I.T.; Rajewsky, A.B.; Ekinen, J. New data on the heat flow in the central part of the Kola Bay. Dokl. Earth Sci. 2004, 396, 102–104. [Google Scholar]
- DePaolo, D.J.; Maher, K.; Christensen, J.N.; McManus, J. Sediment transport time measured with U-series isotopes: Results from ODP North Atlantic drift site 984. Earth Planet. Sci. Lett. 2006, 248, 394–410. [Google Scholar] [CrossRef]
- Malov, A.I.; Kiselev, G.P.; Rudik, G.P. Uranium in the groundwater of the Mezen Syneclise. Dokl. Earth Sci. 2008, 421, 965–968. [Google Scholar] [CrossRef]
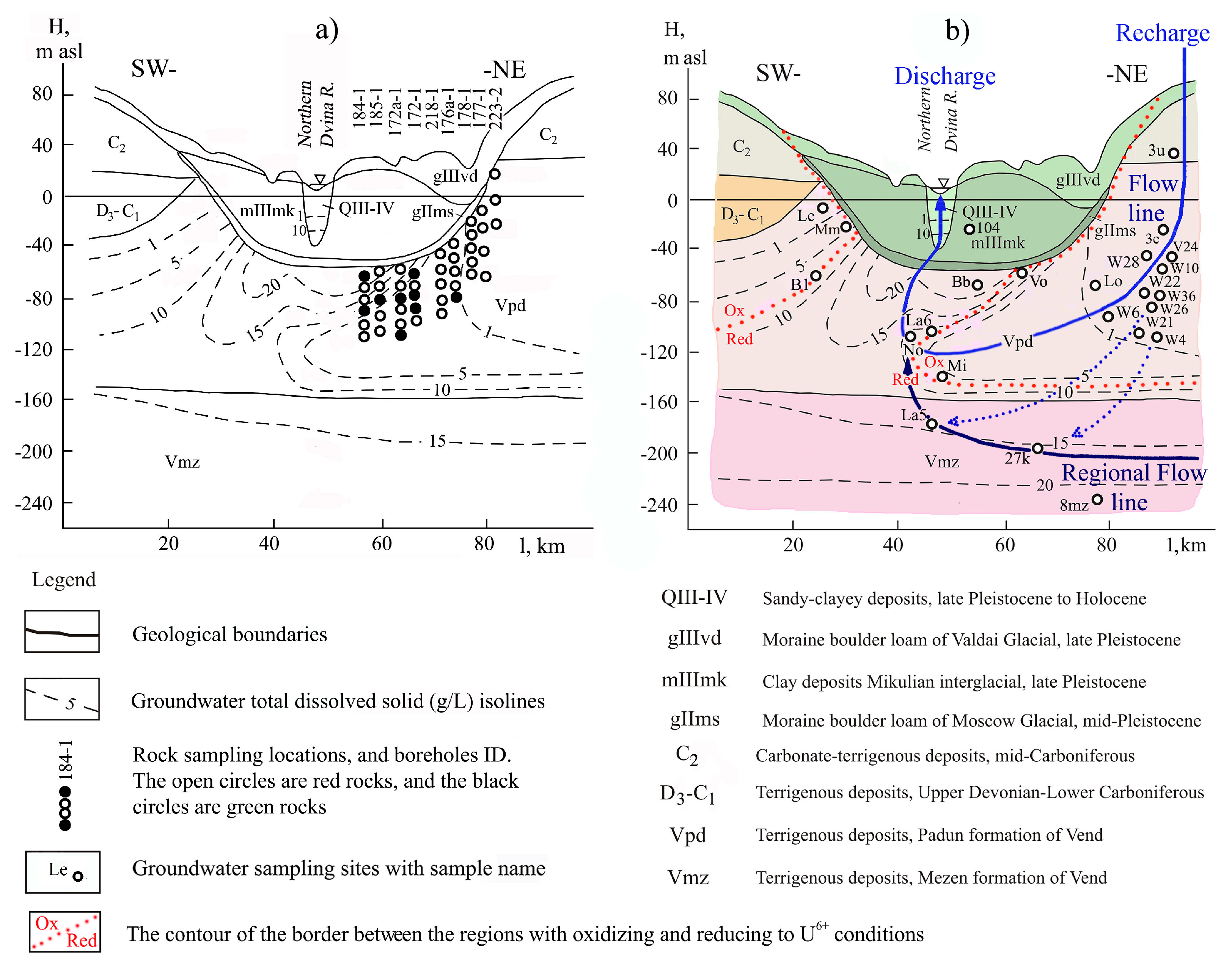
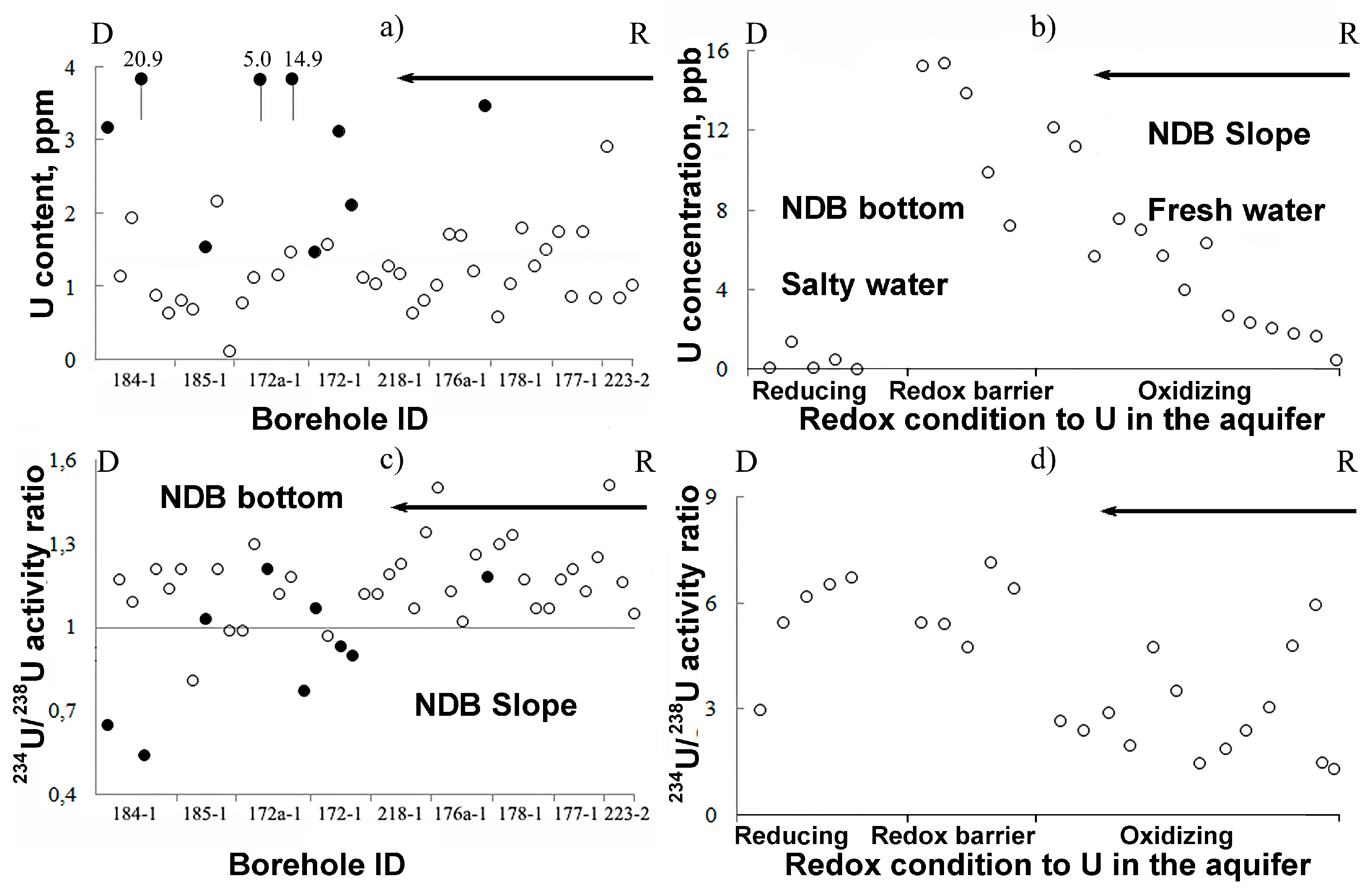
| Well ID | Rock a | Depth (m) | 238U (ppm) | 234U/238U (Bk/Bk) | Well ID | Rock | Depth (m) | 238U (ppm) | 234U/238U (Bk/Bk) |
|---|---|---|---|---|---|---|---|---|---|
| Samples taken from rocks under the central part of the NDB (NDB Bottom) | Samples taken from rocks on the slope of the NDB (NDB Slope) | ||||||||
| 184-1 | GSi | 106.0 | 3.15 ± 0.33 | 0.65 ± 0.10 | 218-1 | RSi | 72.8 | 1.02 ± 0.19 | 1.12 ± 0.19 |
| RSi | 116.4 | 1.13 ± 0.22 | 1.17 ± 0.17 | RM | 82.5 | 1.26 ± 0.21 | 1.19 ± 0.20 | ||
| RSi | 127.2 | 1.92 ± 0.36 | 1.09 ± 0.15 | RSa | 89.4 | 1.16 ± 0.18 | 1.23 ± 0.21 | ||
| GSi | 127.2 | 20.9 ± 3.52 | 0.54 ± 0.08 | RSi | 108.1 | 0.63 ± 0.11 | 1.07 ± 0.18 | ||
| RSi | 143.2 | 0.86 ± 0.15 | 1.21 ± 0.18 | RSi | 114 | 0.81 ± 0.14 | 1.34 ± 0.21 | ||
| RSi | 156.0 | 0.63 ± 0.11 | 1.14 ± 0.16 | 176а-1 | RSa | 68.5 | 1.01 ± 0.16 | 1.50 ± 0.25 | |
| 185-1 | RSi | 122.8 | 0.80 ± 0.14 | 1.21 ± 0.18 | RSi | 72.1 | 1.70 ± 0.26 | 1.13 ± 0.19 | |
| RSi | 133.0 | 0.68 ± 0.11 | 0.81 ± 0.12 | RSi | 85.6 | 1.69 ± 0.25 | 1.02 ± 0.18 | ||
| GM | 144.1 | 1.52 ± 0.28 | 1.03 ± 0.15 | RM | 97.0 | 1.20 ± 0.19 | 1.26 ± 0.20 | ||
| VSi | 153.4 | 2.16 ± 0.37 | 1.21 ± 0.18 | GM | 97.0 | 3.45 ± 0.48 | 1.18 ± 0.19 | ||
| RSi | 160.0 | 0.11 ± 0.03 | 0.99 ± 0.15 | 177-1 | RSS | 52.0 | 0.58 ± 0.12 | 1.30 ± 0.21 | |
| 172а-1 | RSS | 110.6 | 0.76 ± 0.13 | 0.99 ± 0.15 | RSi | 60.8 | 1.03 ± 0.16 | 1.33 ± 0.22 | |
| VSi | 120.0 | 1.11 ± 0.22 | 1.30 ± 0.19 | RSi | 78.1 | 1.79 ± 0.27 | 1.17 ± 0.19 | ||
| GSi | 133.6 | 4.96 ± 0.75 | 1.21 ± 0.18 | RM | 89.0 | 1.26 ± 0.21 | 1.07 ± 0.18 | ||
| RSi | 133.6 | 1.14 ± 0.23 | 1.12 ± 0.16 | 178-1 | RSi | 72.5 | 1.50 ± 0.23 | 1.07 ± 0.18 | |
| RSi | 150.9 | 1.45 ± 0.27 | 1.18 ± 0.17 | RSa | 79.4 | 1.74 ± 0.27 | 1.17 ± 0.19 | ||
| GSi | 150.9 | 14.9 ± 2.31 | 0.77 ± 0.11 | RSi | 83.0 | 0.85 ± 0.15 | 1.21 ± 0.21 | ||
| 172-1 | GSi | 114.0 | 1.46 ± 0.23 | 1.07 ± 0.15 | RSa | 92.0 | 1.73 ± 0.27 | 1.13 ± 0.18 | |
| RSi | 119.6 | 1.57 ± 0.25 | 0.97 ± 0.15 | RSa | 101.5 | 0.84 ± 0.15 | 1.25 ± 0.20 | ||
| GSi | 119.6 | 3.11 ± 0.38 | 0.93 ± 0.14 | 223-2 | RSi | 49.0 | 2.89 ± 0.78 | 1.51 ± 0.25 | |
| GSi | 131.6 | 2.10 ± 0.35 | 0.90 ± 0.14 | RSi | 70.0 | 0.83 ± 0.15 | 1.16 ± 0.19 | ||
| RSi | 145.0 | 1.11 ± 0.22 | 1.12 ± 0.16 | RSi | 92.0 | 1.01 ± 0.17 | 1.05 ± 0.17 | ||
| Average | 3.07 ± 0.50 | 1.03 ± 0.16 | Average | 1.36 ± 0.23 | 1.20 ± 0.20 | ||||
| Total average 238U = 2.22 ± 0.36 ppm, 234U/238U = 1.12 ± 0.18 | |||||||||
| Green rock average 238U = 6.17 ± 0.99 ppm, 234U/238U = 0.92 ± 0.15 | |||||||||
| Red rock average 238U = 1.20 ± 0.20 ppm, 234U/238U = 1.16 ± 0.19 | |||||||||
| Sample Name | TDS (mg/L) | pH | DO (mg/L) | T (°C) | 14C (pmc) | δ13C (‰) | Age (ka) | Eh (mV) | Alkalinity (meq/L) | Fe (µg/L) | C8W (ppb) | ARt (Bk/Bk) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Samples taken from groundwater under the central part of the NDB (NDB Bottom) | ||||||||||||
| 8 mz2014 | 22,246 | 7.6 | 0 | 6.7 | 0 | 7.6 | 460 ± 70 b | NA | 0.16 | 8097 | 0.2 ± 0.004 | 2.97 ± 0.45 |
| 272012 | 15,724 | 7.9 | 1.1 | 5.9 | NA | NA | 160 ± 25 c | −23 | 0.92 | 826 | 1.39 ± 0.03 | 5.45 ± 0.81 |
| La52012 | 15,158 | 9 | 0 | 6.8 | NA | NA | 110 ± 17 c | −151 | 0.15 | 6031 | 0.12 ± 0.002 | 6.19 ± 0.93 |
| La62012 | 9065 | 8.4 | 0.3 | 6.3 | NA | NA | 90 ± 15 c | −42 | 0.7 | 708 | 0.47 ± 0.01 | 6.53 ± 0.98 |
| No2014 | 8954 | 8.3 | 1.2 | 6.8 | 0 | NA | 80 ± 14 c | −82 | 0.57 | 1352 | 0.28 ± 0.01 | 6.72 ± 1.05 |
| Bb2003 | 20,619 | NA | NA | NA | NA | NA | NA | NA | 3.97 | 400 | NA | NA |
| B12012 | 8399 | 7.7 | 1.2 | 5.7 | 5.78 ± 0.24 | −16.6 | 27.3 ± 0.6 a | −38 | 4.18 | 1872 | 15.22 ± 0.3 | 5.46 ± 0.82 |
| B12014 | 9193 | 7.6 | NA | 5.8 | 5.79 ± 0.19 | −15.6 | 26.1 ± 0.6 a | NA | 4.07 | 776 | 15.38 ± 0.3 | 5.41 ± 0.81 |
| Vo2012 | 13,370 | 7.7 | 0 | 5.3 | NA | NA | 22.2 ± 3.3 b | NA | 2.18 | NA | 13.84 ± 0.3 | 4.75 ± 0.71 |
| Mi2014 | 5317 | 7.4 | 0 | 5 | 1.70 ± 0.26 | −14.6 | 33.0 ± 2.3 a | −25 | 1.74 | 439 | 9.86 ± 0.2 | 7.16 ± 0.94 |
| MM2012 | 4362 | 7.8 | NA | 4.8 | NA | NA | 16.6 ± 2.4 b | 2 | 3.75 | 443 | 7.24 ± 0.14 | 6.4 ± 0.96 |
| Samples taken from groundwater on the slope of the NDB (NDB Slope) | ||||||||||||
| 3e2015 | 138 | 8.7 | 3.5 | 4.8 | 34.18 ± 0.59 | −12.3 | 2.1 ± 0.3 a | −68 | 1.62 | 203 | 12.15 ± 0.24 | 2.26 ± 0.34 |
| W102014 | 387 | 9.1 | 1.4 | 4.1 | NA | NA | 11.7 ± 1.8 b | 101 | 3.67 | 5.6 | 11.22 ± 0.22 | 2.39 ± 0.36 |
| W62014 | 738 | 8.9 | 0.6 | 4.5 | 25.30 ± 0.64 | −9.6 | 4.0 ± 0.3 a | 99 | 3.75 | 9.5 | 5.21 ± 0.92 | 2.86 ± 0.42 |
| W212014 | 647 | 8.6 | 2.9 | 4.6 | NA | NA | 4.8 ± 0.8 b | 23 | 3.44 | 18.2 | 7.55 ± 0.15 | 1.99 ± 0.3 |
| W42012 | 638 | 9 | 0 | 4.7 | NA | NA | 16.4 ± 2.4 b | 106 | 3.97 | 3.9 | 7.0 ± 0.14 | 4.76 ± 0.72 |
| V242012 | 307 | 8.6 | 1.5 | 3.8 | NA | NA | 8.9 ± 1.4 b | −12 | 3.33 | 71.9 | 5.71 ± 0.11 | 3.51 ± 0.52 |
| 3u2014 | 285 | 7.7 | NA | 4.0 | 44.97 ± 1.08 | −8.7 | 1.3 ± 0.2 b | NA | 3.21 | 65.7 | 4.01 ± 0.84 | 1.46 ± 0.22 |
| 3u2015 | 93 | 7.8 | NA | 4.9 | NA | NA | NA | NA | 1.02 | 94.9 | 0.25 ± 0.01 | 1.28 ± 0.21 |
| W222014 | 383 | 8.4 | 1.2 | 4.1 | 24.86 ± 0.43 | −10.1 | 4.6 ± 0.4 a | −8 | 3.02 | 14.3 | 6.37 ± 0.13 | 1.63 ± 0.24 |
| W282014 | 260 | 8.2 | 3.3 | 3.9 | 58.40 ± 0.89 | −11.0 | 2.0 ± 0.3 b | 106 | 3.05 | 9.3 | 2.99 ± 0.06 | 2.39 ± 0.36 |
| W262014 | 365 | 8.1 | 0.8 | 4.4 | NA | NA | 2.8 ± 0.4 b | −34 | 3.61 | 10.8 | 2.38 ± 0.05 | 3.04 ± 0.45 |
| W362014 | 348 | 8.2 | 2.7 | 5.2 | 25.01 ± 0.47 | −11.7 | 5.8 ± 0.4 a | −62 | 3.56 | 59.1 | 2.0 ± 0.04 | 4.81 ± 0.62 |
| Lo2014 | 790 | 9.0 | 1.8 | 4.7 | 37.03 ± 0.67 | −11.2 | 5.4 ± 0.8 b | NA | 5.25 | 26.4 | 1.78 ± 0.04 | 5.94 ± 0.84 |
| Le2012 | 209 | 8.2 | 6 | 5 | 51.67 ± 0.63 | −11.5 | 0.3 ± 0.05 b | -24 | 2.61 | 165 | 1.70 ± 0.03 | 1.43 ± 0.21 |
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malov, A.I. Evolution of Uranium Isotopic Compositions of the Groundwater and Rock in a Sandy-Clayey Aquifer. Water 2017, 9, 910. https://doi.org/10.3390/w9120910
Malov AI. Evolution of Uranium Isotopic Compositions of the Groundwater and Rock in a Sandy-Clayey Aquifer. Water. 2017; 9(12):910. https://doi.org/10.3390/w9120910
Chicago/Turabian StyleMalov, Alexander I. 2017. "Evolution of Uranium Isotopic Compositions of the Groundwater and Rock in a Sandy-Clayey Aquifer" Water 9, no. 12: 910. https://doi.org/10.3390/w9120910
APA StyleMalov, A. I. (2017). Evolution of Uranium Isotopic Compositions of the Groundwater and Rock in a Sandy-Clayey Aquifer. Water, 9(12), 910. https://doi.org/10.3390/w9120910





