Cadmium and Lead Sorption/Desorption on Non-Amended and By-Product-Amended Soil Samples and Pyritic Material
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Characterization of the Materials
2.2.2. Cd and Pb Sorption/Desorption Experiments
2.3. Data Analyses
3. Results
3.1. Cd Sorption on the Individual Materials
3.2. Pb Sorption on the Individual Materials
3.3. Cd and Pb Desorption from the Individual Materials
3.4. Cd and Pb Sorption on the Amended Materials
3.5. Cd and Pb Desorption from the Amended Materials
3.6. Implications of the Research
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Huang, B.; Tian, K.; Holm, P.E.; Zhang, Y. Heavy metals in intensive greenhouse vegetable production systems along Yellow Sea of China: Levels, transfer and health risk. Chemosphere 2017, 167, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Song, S.; Wang, R.; Liu, Z.; Meng, J.; Sweetman, A.J.; Jenkins, A.; Ferrier, R.C.; Li, H.; Luo, W.; et al. Impacts of soil and water pollution on food safety and health risks in China. Environ. Intern. 2015, 77, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Kang, H.; Zhang, X.; Shao, H.; Chu, L.; Ruan, C. A critical review on the bio-removal of hazardous heavy metals from contaminated soils: Issues, progress, eco-environmental concerns and opportunities. J. Hazard. Mater. 2010, 174, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.J.K.; Ahmaruzzaman, M. A review on potential usage of industrial waste materials for binding heavy metal ions from aqueous solutions. J. Water Process Eng. 2016, 10, 39–47. [Google Scholar] [CrossRef]
- Balmuri, S.R.; Selvaraj, U.; Kumar, V.V.; Anthony, S.P.; Tsatsakis, A.M.; Golokhvast, K.S.; Raman, T. Effect of surfactant in mitigating cadmium oxide nanoparticle toxicity: Implications for mitigating cadmium toxicity in environment. Environ. Res. 2017, 152, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Rehman, Z.U.; Khan, S.; Brusseau, M.L.; Shah, M.T. Lead and cadmium contamination and exposure risk assessment via consumption of vegetables grown in agricultural soils of five-selected regions of Pakistan. Chemosphere 2017, 168, 1589–1596. [Google Scholar] [CrossRef] [PubMed]
- Tong, S.; von Schirnding, Y.E.; Prapamontol, T. Environmental lead exposure: A public health problem of global dimensions. Bull. World Health Org. 2000, 78, 1068–1077. [Google Scholar] [PubMed]
- Zaborska, A. Anthropogenic lead concentrations and sources in Baltic Sea sediments based on lead isotopic composition. Mar. Pollut. Bull. 2014, 85, 99–113. [Google Scholar] [CrossRef] [PubMed]
- Gabaldón, C.; Marzal, P.; Seco, A.; González, J.A. Cadmium and copper removal by a granular activated carbon in laboratory column systems. Sep. Sci. Technol. 2000, 35, 1039–1053. [Google Scholar] [CrossRef]
- Gavrilescu, M. Removal of Heavy Metals from the Environment by Biosorption. Eng. Life Sci. 2004, 4, 219–232. [Google Scholar] [CrossRef]
- He, J.; Chen, J.P. A comprehensive review on biosorption of heavy metals by algal biomass: Materials, performances, chemistry, and modeling simulation tools. Bioresour. Technol. 2014, 160, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Jain, C.K.; Malik, D.S.; Yadav, A.K. Applicability of plant based biosorbents in the removal of heavy metals: A review. Environ. Process. 2016, 3, 495–5233. [Google Scholar] [CrossRef]
- Palma, G.; Freer, J.; Baeza, J. Removal of metal ions by modified Pinus radiata bark and tannins from water solutions. Water Res. 2003, 37, 4974–4980. [Google Scholar] [CrossRef] [PubMed]
- Vilar, V.J.P.; Botelho, C.M.S.; Loureiro, J.M.; Boaventura, R.A.R. Biosorption of copper by marine algae Gelidium and algal composite material in a packed bed column. Bioresour. Technol. 2008, 99, 5830–5838. [Google Scholar] [CrossRef] [PubMed]
- Coelho, G.F.; GonÇalves, A.C.; Nóvoa-Muñoz, J.C.; Fernández-Calviño, D.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. Competitive and non-competitive cadmium, copper and lead sorption/desorption on wheat straw affecting sustainability in vineyards. J. Clean. Prod. 2016, 139, 1496–1503. [Google Scholar] [CrossRef]
- Cutillas-Barreiro, L.; Ansias-Manso, L.; Fernandez Calviño, D.; Arias-Estévez, M.; Nóvoa-Muñoz, J.C.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. Pine bark as bio-adsorbent for Cd, Cu, Ni, Pb and Zn: Batch-type and stirred flow chamber experiments. J. Environ. Manag. 2014, 144, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Paradelo, R.; Cutillas-Barreiro, L.; Soto-Gomez, D.; Novoa-Muñoz, J.C.; Arias-Estevez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. Study of metal transport through pine bark for reutilization as a biosorbent. Chemosphere 2016, 149, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Peña, S.; Fernández, D.; Nóvoa, J.C.; Arias, M.; Núñez, A.; Fernández, M.J.; Álvarez, E. Kinetics of Hg(II) adsorption and desorption in calcined mussel shells. J. Hazard. Mater. 2010, 180, 622–627. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Pérez, A.M.; Paradelo, M.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; FernándezSanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. Heavy metal retention in copper mine soil treated with mussel shells: Batch and column experiments. J. Hazard. Mater. 2013, 248, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Calviño, D.; Garrido-Rodríguez, B.; Cutillas-Barreiro, L.; Araújo-Nespereira, P.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. Influence of mussel shell on As and Cr competitive and non-competitive sorption—Desorption kinetics in a mine soil: Stirred flow chamber experiments. Geoderma 2014, 232–234, 300–308. [Google Scholar] [CrossRef]
- Fernández-Pazos, M.T.; Garrido-Rodriguez, B.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Núñez-Delgado, A.; Álvarez, E. Cr(VI) adsorption and desorption on soils and bio-sorbents. Water Air Soil Pollut. 2013, 224, 1–12. [Google Scholar] [CrossRef]
- Osorio-López, C.; Seco-Reigosa, N.; Garrido-Rodríguez, B.; Cutillas-Barreiro, L.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. As(V) adsorption on forest and vineyard soils and pyritic material with or without mussel shell: Kinetics and fractionation. J. Taiwan Inst. Chem. Eng. 2014, 45, 1007–1014. [Google Scholar] [CrossRef]
- Otero, M.; Cutillas-Barreiro, L.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. Cr(VI) sorption/desorption on untreated and mussel-shell-treated soil materials: Fractionation and effects of pH and chromium concentration. Solid Earth 2015, 6, 373–382. [Google Scholar] [CrossRef]
- Seco, N.; Cutillas-Barreiro, L.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. Mixtures including wastes from the mussel shell processing industry: Retention of arsenic, chromium and mercury. J. Clean. Prod. 2014, 84, 680–690. [Google Scholar] [CrossRef]
- Seco-Reigosa, N.; Bermúdez-Couso, A.; Garrido-Rodríguez, B.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. As(V) retention on soils and forest by-products and other waste materials. Environ. Sci. Pollut. Res. 2013, 20, 6574–6583. [Google Scholar] [CrossRef] [PubMed]
- Seco-Reigosa, N.; Peña-Rodriguez, S.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. Arsenic, chromium and mercury removal using mussel shell ash or a sludge/ashes waste mixture. Environ. Sci. Pollut. Res. 2013, 20, 2670–2678. [Google Scholar] [CrossRef] [PubMed]
- Seco-Reigosa, N.; Cutillas-Barreiro, L.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Álvarez-Rodríguez, E.; Fernández-Sanjurjo, M.J.; Núñez-Delgado, A. Adsorption, desorption and fractionation of As(V) on untreated and mussel shell-treated granitic material. Solid Earth 2015, 6, 337–346. [Google Scholar] [CrossRef]
- Alejano, L.R.; Perucho, A.; Olalla, C.; Jiménez, R. Rock Engineering and Rock Mechanics: Structures in and on Rock Masses; CRC Press: London, UK, 2014; p. 372. [Google Scholar]
- Álvarez, E.; Fernández-Sanjurjo, M.J.; Núñez, A.; Seco, N.; Corti, G. Aluminium fractionation and speciation in bulk and rhizosphere of a grass soil amended with mussel shells or lime. Geoderma 2012, 173, 322–329. [Google Scholar] [CrossRef]
- Brás, I.; Teixeira-Lemos, L.; Alves, A.; Pereira, M.F.R. Application of pine bark as a sorbent for organic pollutants in effluents. Manag. Environ. Qual. 2004, 15, 491–501. [Google Scholar] [CrossRef]
- Chatterjee, A.; Lal, R.; Wielopolski, L.; Martin, M.Z.; Ebinger, M.H. Evaluation of Different Soil Carbon Determination Methods. Crit. Rev. Plant. Sci. 2009, 28, 164–178. [Google Scholar] [CrossRef]
- Coelho, G.F.; ConÇalves, A.C., Jr.; Tarley, C.R.T.; Casarin, J.; Nacke, N.; Francziskowski, M.A. Removal of metal ions Cd (II), Pb (II) and Cr (III) from water by the cashew nut shell Anarcadium Occidentale L. Ecol. Eng. 2014, 73, 514–525. [Google Scholar] [CrossRef]
- Dlapa, P.; Bodí, M.B.; Mataix-Solera, J.; Cerdà, A.; Doerr, S.H. FT-IR spectroscopy reveals that ash water repellency is highly dependent on ash chemical composition. Catena 2013, 108, 35–43. [Google Scholar] [CrossRef]
- Fackler, K.; Stevanic, J.S.; Ters, T.; Hinterstoisser, B.; Schwanninger, M.; Salmén, L. Localisation and characterisation of incipient brown-rot decay within spruce wood cell walls using FT-IR imaging microscopy. Enzym. Microb. Technol. 2010, 47, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Haberhauer, G.; Gerzabek, M.H. Drift and transmission FT-IR spectroscopy of forest soils: An approach to determine decomposition processes of forest litter. Vib. Spectrosc. 1999, 19, 413–417. [Google Scholar] [CrossRef]
- Kamprath, E.J. Exchangeable aluminium as a criterion for liming leached mineral soils. Soil Sci. Soc. Am. Proc. 1970, 34, 252–254. [Google Scholar] [CrossRef]
- Margenot, A.J.; Calderón, F.J.; Goyne, K.W.; Mukome, F.N.D.; Parikh, S.J. IR Spectroscopy, Soil Analysis Applications. In Encyclopedia of Spectroscopy and Spectrometry, 3rd ed.; Lindon, J., Tranter, G., Koppenaal, D., Eds.; Oxford Academic Press: London, UK, 2017; pp. 448–454. [Google Scholar]
- McLean, E.O. Soil pH and Lime Requirement. In Methods of Soil Analysis, Part 2, Chemical and Microbiological Properties; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA: Madison, WI, USA, 1982; pp. 199–223. [Google Scholar]
- Mimura, A.M.S.; Vieira, T.V.A.; Martinelli, P.B.; Gorgulho, H.F. Utilization of rice husk to remove Cu2+, Al3+, Ni2+ and Zn2+ from wastewater. Quím. Nova 2010, 33, 1279–1284. [Google Scholar] [CrossRef]
- Movasaghi, Z.; Rehman, S.; Rehman, I. Fourier Transform Infrared (FTIR) Spectroscopy of Biological Tissues. Appl. Spectrosc. Rev. 2008, 43, 134–179. [Google Scholar] [CrossRef]
- Nóbrega, J.A.; Pirola, C.; Fialho, L.L.; Rota, G.; de Campos, C.E.; Pollo, F. Microwave-assisted digestion of organic samples: How simple can it become? Talanta 2012, 98, 272–276. [Google Scholar] [CrossRef] [PubMed]
- Pavia, D.L.; Lampman, G.M.; Kriz, G.S.; Vyvyan, J.R. Introdução à Espectroscopia, 4th ed.; Cengage Learning: São Paulo, Brazil, 2010; p. 700. [Google Scholar]
- Rubio, F.; GonÇalves, A.C., Jr.; Meneghel, A.P.; Tarley, C.R.T.; Schwantes, D.; Coelho, G.F. Removal of cadmium from water using by-product Crambe abyssinica Hochst seeds as biosorbent material. Water Sci. Technol. 2013, 68, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Saikia, B.J.; Parthasarathy, G. Fourier Transform Infrared Spectroscopic Characterization of Kaolinite from Assam and Meghalaya, Northeastern India. J. Mod. Phys. 2010, 1, 206–210. [Google Scholar] [CrossRef]
- Sila, A.M.; Shepherd, K.D.; Pokhariyal, G.P. Evaluating the utility of mid-infrared spectral subspaces for predicting soil properties. Chemom. Intell. Lab. Syst. 2016, 153, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Smidt, W.; Meissl, K. The applicability of Fourier transform infrared (FT-IR) spectroscopy in waste management. Waste Manag. 2007, 27, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Sumner, M.E.; Miller, W.P. Cation exchange capacity and exchange coefficients. In Methods of Soil Analysis, Part 3, Chemical Methods; Page, D.L., Ed.; ASA: Madison, WI, USA, 1996; pp. 437–474. [Google Scholar]
- Tan, K.H. Soil Sampling, Preparation, and Analysis; Marcel Dekker: New York, NY, USA, 1996. [Google Scholar]
- Tarley, C.R.T.; Arruda, M.A.Z. Biosorption of heavy metals using rice milling by-products. Characterisation and application for removal of metals from aqueous effluents. Chemosphere 2004, 54, 987–995. [Google Scholar] [CrossRef] [PubMed]
- Tinti, A.; Tugnoli, V.; Bonora, S.; Francioso, O. Recent applications of vibrational mid-Infrared (IR) spectroscopy for studying soil components: A review. J. Cent. Eur. Agric. 2015, 16, 1–22. [Google Scholar] [CrossRef]
- Quintáns-Fondo, A.; Fernández-Calviño, D.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A. As(V) Sorption/Desorption on Different Waste Materials and Soil Samples. Int. J. Environ. Res. Public Health 2017, 14, 803. [Google Scholar] [CrossRef] [PubMed]
- Appel, C.; Ma, L.Q. Concentration, pH, and surface charge effects on cadmium and lead sorption in three tropical soils. J. Environ. Qual. 2002, 31, 581–589. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.R.; Owens, G.O.; Naidu, R. Heavy metal distribution, bioaccessibility, and phytoavailability in long-term contaminated soils from Lake Macquarie, Australia. Aust. J. Soil Res. 2009, 47, 166–176. [Google Scholar] [CrossRef]
- Sauvé, S.; Hendershot, W.; Allen, H.E. Solid-solution partitioning of metals in contaminated soils: Dependence on pH, total metal burden, and organic matter. Environ. Sci. Technol. 2000, 34, 1125–1131. [Google Scholar] [CrossRef]
- Shaheen, M.; Tsadilas, C.D.; Rinklebe, J. A review of the distribution coefficients of trace elements in soils: Influence of sorption system, element characteristics, and soil colloidal properties. Adv. Colloid Interface Sci. 2013, 201–202, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.S.; Angle, J.S.; Chaney, R.L.; Delorme, T.A.; Reeves, R.D. Soil pH effects on uptake of Cd and Zn by Thlaspi caerulescens. Plant Soil 2006, 281, 325–337. [Google Scholar] [CrossRef]
- Memon, S.Q.; Memon, N.; Shah, S.W.; Khuhawar, M.Y.; Bhanger, M.I. Sawdust—A green and economical sorbent for the removal of cadmium(II) ions. J. Hazard. Mater. 2007, 139, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Kabata-Pendias, A.; Pendias, H. Trace Elements in Soils; Boca Ratón: New York, NY, USA, 2001. [Google Scholar]
- Alloway, B.J. Heavy Metals in Soils, 2nd ed.; Blackie Academic and Professional: London, UK, 1995. [Google Scholar]
- Taty-Costodes, V.C.; Fauduet, H.; Porte, C.; Delacroix, A. Removal of Cd(II) and Pb(II) ions, from aqueous solutions, by adsorption onto sawdust of Pinus sylvestris. J. Hazard. Mater. 2003, 105, 121–142. [Google Scholar] [CrossRef] [PubMed]
- Pagnanelli, F.; Esposito, A.; Toroa, L.; Veglió, F. Metal speciation and pH effect on Pb, Cu, Zn and Cd biosorption onto Sphaerotilus natans: Langmuir-type empirical model. Water Res. 2003, 37, 627–633. [Google Scholar] [CrossRef]
- Van Herwijnen, R.; Hutchings, T.R.; Al-Tabba, A.; Moffat, A.J.; Johns, M.L.; Ouki, S.K. Remediation of metal contaminated soil with mineral-amended composts. Environ. Pollut. 2007, 150, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Lamb, D.; Paneerselvam, P.; Choppala, G.; Bolan, N.; Chung, J.W. Role of organic amendments on enhanced bioremediation of heavy metal(loid) contaminated soils. J. Hazard. Mater. 2011, 185, 549–574. [Google Scholar] [CrossRef] [PubMed]
- Antoniadis, V.; Tsadilas, C.D.; Ashworth, D.J. Monometal and competitive adsorption of heacy metals by sewage sludge-amended soil. Chemosphere 2007, 68, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, S.M. Sorption and lability of cadmium and lead in different soils from Egypt and Greece. Geoderma 2009, 153, 61–68. [Google Scholar] [CrossRef]
- Stevenson, F.J. Humus Chemistry Genesis Composition Reactions; John Wiley: New York, NY, USA, 1982. [Google Scholar]
- Chen, C.; Liu, H.; Chen, T.; Chena, D.; Frost, R.L. An insight into the removal of Pb(II), Cu(II), Co(II), Cd(II), Zn(II), Ag(I), Hg(I), Cr(VI) by Na(I)-montmorillonite and Ca(II)montmorillonite. Appl. Clay Sci. 2015, 118, 239–247. [Google Scholar] [CrossRef]
- Serrano, S.; Garrido, F.; Campbell, C.G.; Garcia-Gonzalez, M.T. Competitive sorption of cadmium and lead in acid soils of Central Spain. Geoderma 2005, 124, 91–104. [Google Scholar] [CrossRef]
- McBride, M.B. Reactions controlling heavy metal solubility in soils. Adv. Soil Sci. 1989, 10, 1–56. [Google Scholar]
- Stipp, S.L.; Hochella, M.F.; Parks, G.A.; Leckie, J.O. Cd2+ uptake by calcite, solid-state diffusion, and the formation of solid-solution: Interface processes observed with near-surface sensitive techniques (XPS, LEED, and AES). Geochim. Cosmochim. Acta 1992, 56, 1941–1954. [Google Scholar] [CrossRef]
- Zhao, X.; Jiang, T.; Du, B. Effect of organic matter and calcium carbonate on behaviors of cadmium adsorption–desorption on/from purple paddy soils. Chemosphere 2014, 99, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Irani, M.; Amjadib, M.; Mousaviana, M.A. Comparative study of lead sorption onto natural perlite, dolomite and diatomite. Chem. Eng. J. 2011, 178, 317–323. [Google Scholar] [CrossRef]
- Petruzelli, G.; Guidi, G.; Lubrano, L.Z. The effect os sewage sludges and compost on the extractability of heavy metals from soil. Environ. Technol. Lett. 1981, 2, 449–456. [Google Scholar] [CrossRef]
- Reddy, K.J.; Wang, L.; Gloss, S.P. Solubility and mobility of copper, zinc and lead in acidic environments. Plant Soil 1995, 171, 53–58. [Google Scholar] [CrossRef]
- McKenzie, R.M. The adsorption of lead and other heavy metals on oxides of manganese and iron. Aust. J. Soil Res. 1980, 18, 61–73. [Google Scholar] [CrossRef]
- Tofan, L.; Paduraru, C.; Volf, I.; Balan, C. Removal of lead (II) from aqueous solution on natural hemp fibers. Sci. Pap. J. Agro Ser. 2010, 53, 15–153. [Google Scholar]
- Papandreou, A.D.; Stournaras, C.J.; Panias, D.; Paspaliaris, I. Adsorption of Pb(II), Zn(II) and Cr(III) on coal fly ash porous pellets. Min. Eng. 2011, 24, 1495–1501. [Google Scholar] [CrossRef]
- Reddy, H.K.; Seshaiaha, K.; Reddy, A.V.R.; Madhava Rao, M.; Wang, M.C. Biosorption of Pb2+ from aqueous solutions by Moringa oleifera bark: Equilibrium and kinetic studies. J. Hazard. Mater. 2010, 174, 831–838. [Google Scholar] [CrossRef] [PubMed]
- Tunali, S.; Çabuk, A.; Akar, T. Removal of lead and copper ions from aqueous solutions by bacterial strain isolated from soil. Chem. Eng. J. 2006, 115, 203–211. [Google Scholar] [CrossRef]
- Karami, N.; Clemente, R.; Moreno-Jiménez, E.; Lepp, N.W.; Beesley, L. Efficiency of green waste compost and biochar soil amendments for reducing lead and copper mobility and uptake to ryegrass. J. Hazard. Mater. 2011, 191, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, M.; Rout, K.; Mohapatra, B.K.; Anand, S. Sorption behavior of Pb(II) and Cd(II) on iron ore slime and characterization of metal ion loaded sorbent. J. Hazard. Mater. 2009, 166, 1506–1513. [Google Scholar] [CrossRef] [PubMed]
- Papandreou, A.; Stournaras, C.J.; Panias, D. Copper and cadmium adsorption on pellets made from fired coal fly ash. J. Hazard. Mater. 2007, 148, 538–547. [Google Scholar] [CrossRef] [PubMed]
- Vega, F.A.; Covelo, E.F.; Andrade, M.L. Effects of sewage sludge and barley straw treatment on the sorption and retention of Cu, Cd and Pb by coppermine Anthropic Regosols. J. Hazard. Mater. 2009, 169, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Prieto, M.; Cubillas, P.; Fernández-González, A. Uptake of dissolved Cd by biogenic and biogenic aragonite: A comparison with sorption onto calcite. Geochim. Cosmochim. Acta 2003, 67, 3859–3869. [Google Scholar] [CrossRef]
- Shaheen, S.M.; Rinklebe, J. Impact of emerging and low cost alternative amendments on the (im)mobilization and phytoavailability of Cd and Pb in a contaminated floodplain soil. Ecol. Eng. 2015, 74, 319–326. [Google Scholar] [CrossRef]
- Fernández-Calviño, D.; Pérez-Armada, L.; Cutillas-Barreiro, L.; Paradelo-Núñez, R.; Núñez-Delgado, A.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Arias-Estévez, M. Changes in Cd, Cu, Ni, Pb and Zn fractionation and liberation due to mussel shell amendment on a mine soil. Land Degrad. Dev. 2016, 27, 1276–1285. [Google Scholar] [CrossRef]
- Fernández-Calviño, D.; Cutillas-Barreiro, L.; Paradelo-Núñez, R.; Nóvoa-Muñoz, J.C.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Núñez-Delgado, A.; Arias-Estévez, M. Heavy metals fractionation and desorption in pine bark amended mine soils. J. Environ. Manag. 2017, 192, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Madejón, P.; Murillo, J.M.; Marañón, T.; Cabrera, F.; López, R. Bioaccumulation of As, Cd, Cu, Fe and Pb in wild grasses affected by the Aznalcóllar mine spill (SW Spain). Sci. Total Environ. 2002, 290, 105–120. [Google Scholar] [CrossRef]
- Cui, Y.-J.; Zhu, Y.-G.; Zhai, R.-H.; Chen, D.-Y.; Huang, Y.-Z.; Qiu, Y.; Liang, J.-Z. Transfer of metals from soil to vegetables in an area near a smelter in Nanning, China. Environ. Int. 2004, 30, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Probst, A.; Liao, B. Metal contamination of soils and crops affected by the Chenzhou lead/zinc mine spill (Hunan, China). Sci. Total Environ. 2005, 339, 153–166. [Google Scholar] [CrossRef] [PubMed]
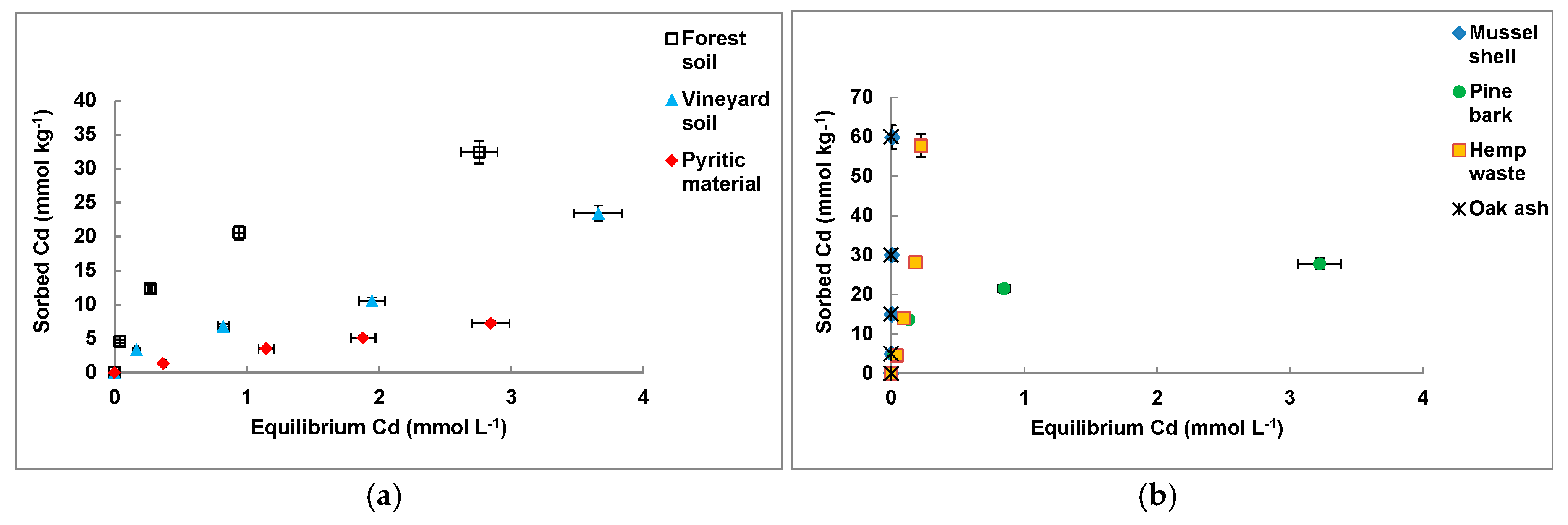
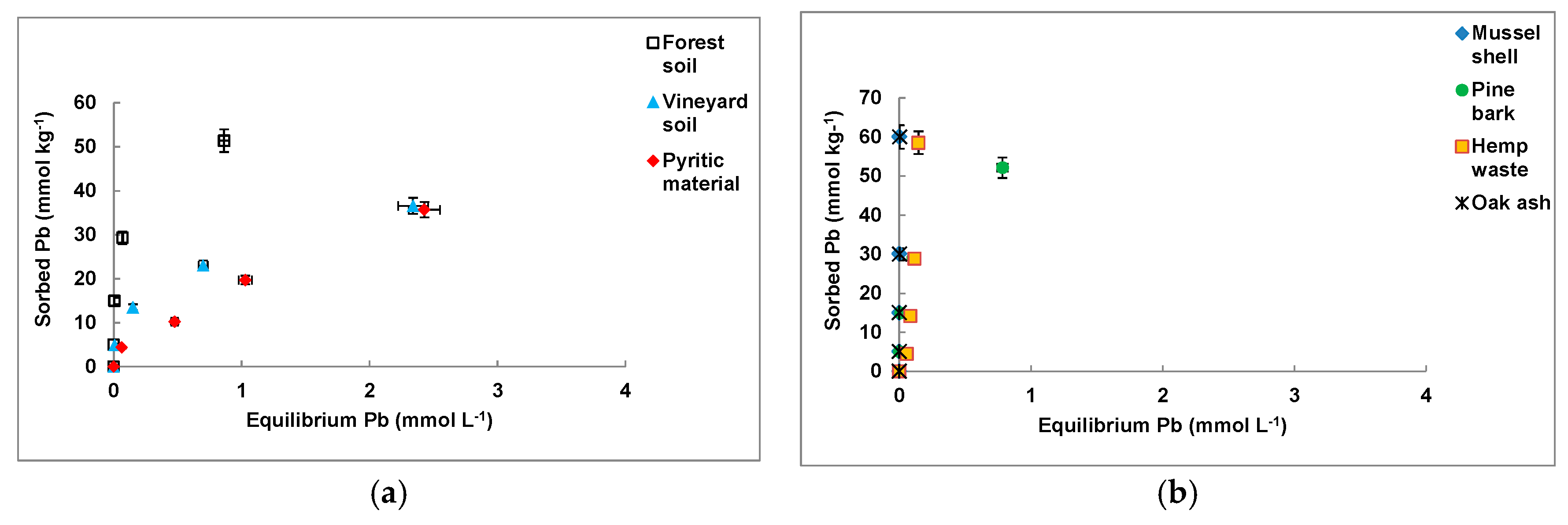

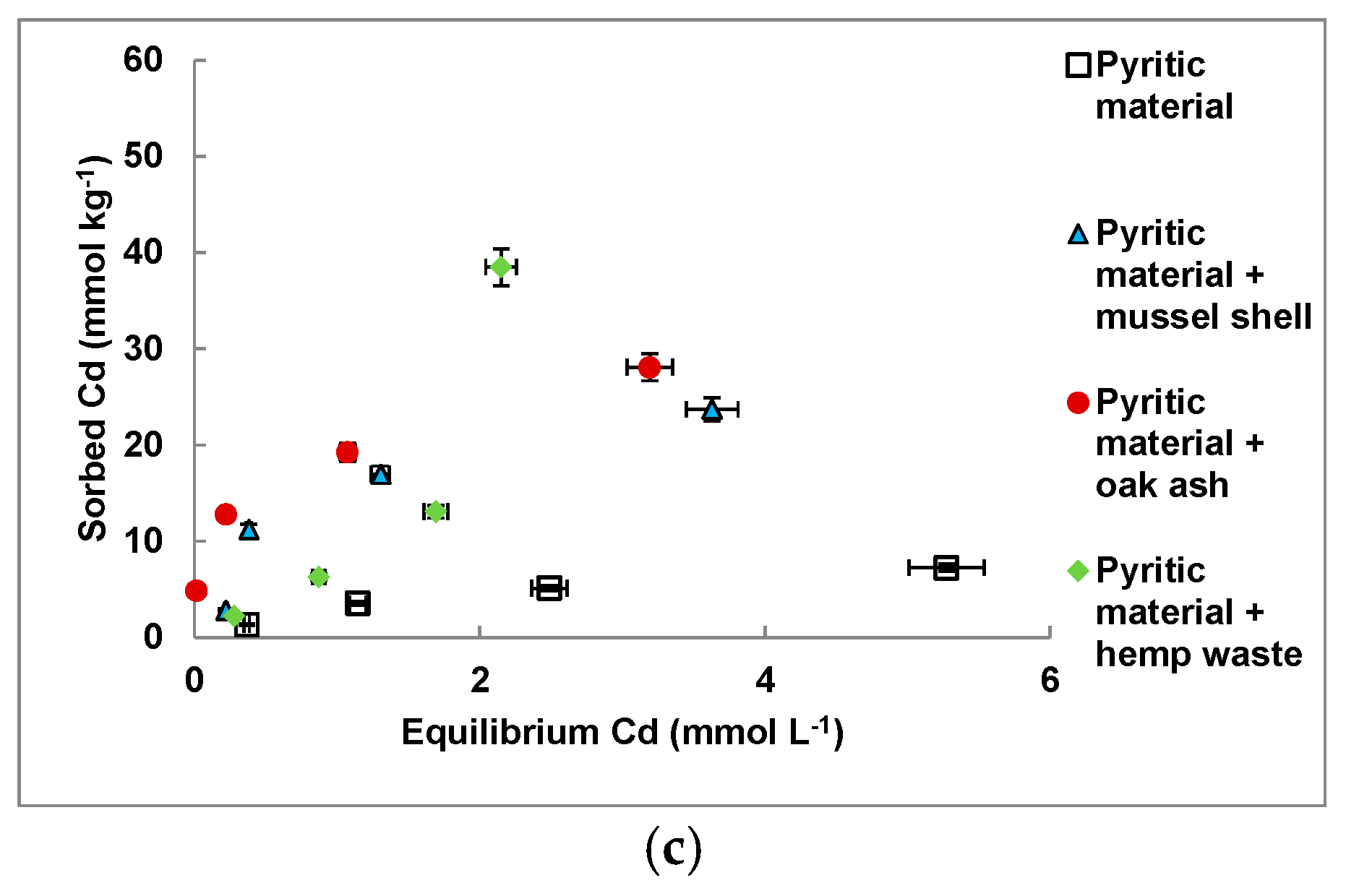
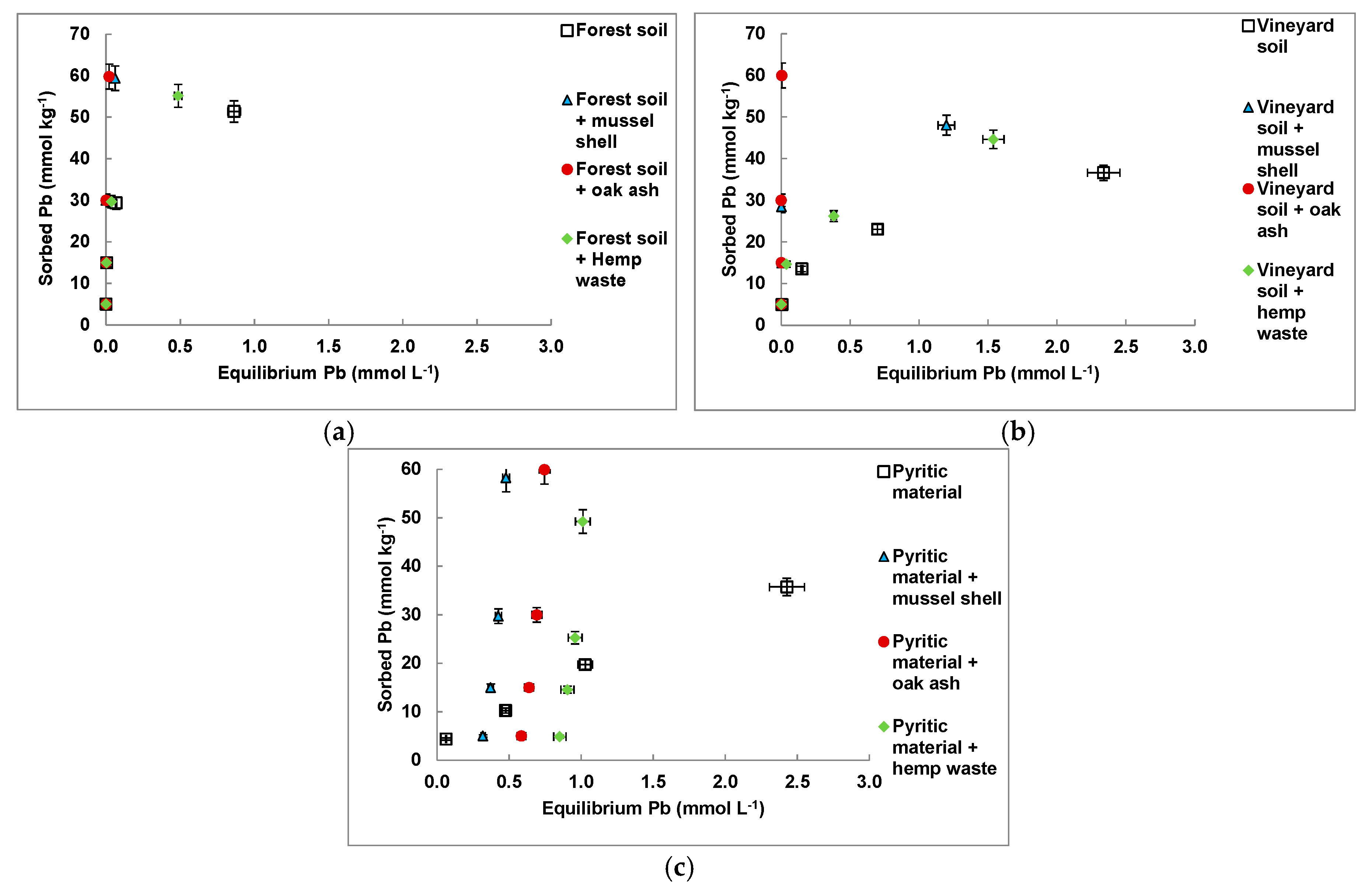
| Sorbent Material | Freundlich | Langmuir | ||||
|---|---|---|---|---|---|---|
| KF (Ln kg−1 mmol(1−n)) | n | R2 | KL (L mmol−1) | Qm (mmol kg−1) | R2 | |
| Forest soil | 21.0 ± 0.4 | 0.43 ± 0.02 | 0.998 | 1.5 ± 0.6 | 39.2 ± 5.3 | 0.971 |
| Vineyard soil | 6.9 ± 1.7 | 0.92 ± 0.21 | 0.955 | - | - | - |
| Pyritic material | 3.0 ± 0.3 | 0.54 ± 0.06 | 0.983 | 0.4 ± 0.1 | 10.5 ± 0.7 | 0.996 |
| Mussel shell | - | - | - | - | - | - |
| Pine bark | 21.0 ± 1.4 | 0.27 ± 0.05 | 0.960 | 7.3 ± 2.4 | 27.3 ± 1.9 | 0.973 |
| Hemp waste | - | - | - | - | - | - |
| Oak ash | - | - | - | - | - | - |
| Sorbent Material | Freundlich | Langmuir | ||||
|---|---|---|---|---|---|---|
| KF (Ln kg−1 mmol(1−n)) | n | R2 | KL (L mmol−1) | Qm (mmol kg−1) | R2 | |
| Forest soil | 54.0 ± 2.3 | 0.25 ± 0.02 | 0.99 | 29.6 ± 19.3 | 51.1 ± 7.1 | 0.93 |
| Vineyard soil | 26.8 ± 0.3 | 0.36 ± 0.01 | 1.00 | 2.5 ± 1.2 | 41.2 ± 6.0 | 0.96 |
| Pyritic material | 19.1 ± 0.7 | 0.70 ± 0.05 | 1.00 | 0.3 ± 0.1 | 84.7 ± 21.5 | 0.99 |
| Mussel shell | - | - | - | - | - | - |
| Pine bark | 56.8 ± 2.1 | 0.31 ± 0.02 | 0.99 | 15.9 ± 8.4 | 54.3 ± 7.1 | 0.95 |
| Hemp waste | - | - | - | - | - | - |
| Oak ash | - | - | - | - | - | - |
| Sorbent Material | Added Cd or Pb (mmol L−1) | Cd Desorption (mmol kg−1) | Cd Desorption (%) | Pb Desorption (mmol kg−1) | Pb Desorption (%) |
|---|---|---|---|---|---|
| Forest soil | 0.5 | 0.10 | 2 | 0.01 | 0.2 |
| 1.5 | 0.57 | 5 | 0.02 | 0.1 | |
| 3.0 | 1.68 | 8 | 0.01 | 0.3 | |
| 6.0 | 4.11 | 13 | 0.93 | 1.8 | |
| Vineyard soil | 0.5 | 0.52 | 15 | 0.02 | 0.3 |
| 1.5 | 1.50 | 22 | 0.17 | 1.2 | |
| 3.0 | 2.86 | 27 | 0.79 | 3.4 | |
| 6.0 | 4.56 | 19 | 2.03 | 5.5 | |
| Pyritic material | 0.5 | 0.72 | 54 | 0.34 | 7.9 |
| 1.5 | 1.91 | 54 | 0.99 | 9.7 | |
| 3.0 | 3.79 | 74 | 1.80 | 9.1 | |
| 6.0 | 5.47 | 76 | 3.00 | 8.4 | |
| Mussel shell | 0.5 | 0.03 | 0.62 | 0.01 | 0.25 |
| 1.5 | 0.04 | 0.27 | 0.02 | 0.13 | |
| 3.0 | 0.04 | 0.13 | 0.02 | 0.05 | |
| 6.0 | 0.05 | 0.08 | 0.03 | 0.04 | |
| Pine bark | 0.5 | 0.07 | 1.4 | 0.01 | 0.26 |
| 1.5 | 0.40 | 2.9 | 0.04 | 0.26 | |
| 3.0 | 2.05 | 9.5 | 0.25 | 0.87 | |
| 6.0 | 3.95 | 14.2 | 2.32 | 4.45 | |
| Hemp waste | 0.5 | 0.08 | 1.72 | 0.16 | 3.7 |
| 1.5 | 0.24 | 1.69 | 0.41 | 2.9 | |
| 3.0 | 0.47 | 1.68 | 0.50 | 1.7 | |
| 6.0 | 0.84 | 1.46 | 0.89 | 1.5 | |
| Oak ash | 0.5 | 0.001 | 0.013 | 0.008 | 0.16 |
| 1.5 | 0.001 | 0.004 | 0.009 | 0.06 | |
| 3.0 | 0.001 | 0.002 | 0.010 | 0.03 | |
| 6.0 | 0.001 | 0.001 | 0.011 | 0.02 |
| Amended Material | Added Cd or Pb (mmol L−1) | Cd Desorption (mmol kg−1) | Cd Desorption (%) | Pb Desorption (mmol kg−1) | Pb Desorption (%) |
|---|---|---|---|---|---|
| FS + MS | 0.5 | 0.01 | 0.3 | 0.01 | 0.16 |
| 1.5 | 0.09 | 0.6 | 0.01 | 0.09 | |
| 3.0 | 0.27 | 1.0 | 0.02 | 0.06 | |
| 6.0 | 1.19 | 2.6 | 0.04 | 0.06 | |
| FS + OA | 0.5 | 0.01 | 0.2 | 0.01 | 0.24 |
| 1.5 | 0.08 | 0.5 | 0.03 | 0.17 | |
| 3.0 | 0.27 | 0.9 | 0.02 | 0.08 | |
| 6.0 | 1.19 | 2.4 | 0.03 | 0.04 | |
| FS + HW | 0.5 | 0.08 | 1.7 | 0.01 | 0.24 |
| 1.5 | 0.33 | 2.6 | 0.02 | 0.12 | |
| 3.0 | 1.21 | 5.4 | 0.05 | 0.17 | |
| 6.0 | 2.46 | 7.1 | 0.57 | 1.04 | |
| VS + MS | 0.5 | 0.08 | 1.8 | 0.01 | 0.17 |
| 1.5 | 0.26 | 2.1 | 0.01 | 0.08 | |
| 3.0 | 0.73 | 3.4 | 0.05 | 0.17 | |
| 6.0 | 1.69 | 5.2 | 0.24 | 0.50 | |
| VS + OA | 0.5 | 0.03 | 0.5 | 0.02 | 0.31 |
| 1.5 | 0.09 | 0.6 | 0.04 | 0.23 | |
| 3.0 | 0.23 | 0.8 | 0.04 | 0.14 | |
| 6.0 | 0.77 | 1.6 | 0.03 | 0.06 | |
| VS + HW | 0.5 | 0.21 | 5.1 | 0.02 | 0.33 |
| 1.5 | 0.86 | 7.4 | 0.04 | 0.29 | |
| 3.0 | 1.93 | 11.1 | 0.22 | 0.84 | |
| 6.0 | 3.67 | 14.6 | 1.54 | 3.45 | |
| PM + MS | 0.5 | 0.22 | 8.0 | 0.00 | 0.09 |
| 1.5 | 0.85 | 7.6 | 0.01 | 0.06 | |
| 3.0 | 1.23 | 7.2 | 0.07 | 0.23 | |
| 6.0 | 3.83 | 16.1 | 0.12 | 0.21 | |
| PM + OA | 0.5 | 0.04 | 0.7 | 0.01 | 0.10 |
| 1.5 | 0.18 | 1.4 | 0.01 | 0.05 | |
| 3.0 | 4.02 | 20.9 | 0.02 | 0.07 | |
| 6.0 | 1.86 | 6.6 | 0.13 | 0.22 | |
| PM + HW | 0.5 | 0.56 | 25.2 | 0.07 | 1.44 |
| 1.5 | 1.57 | 25.0 | 0.47 | 3.26 | |
| 3.0 | 4.03 | 30.8 | 1.04 | 4.11 | |
| 6.0 | 3.01 | 7.8 | 2.75 | 5.58 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Núñez-Delgado, A.; Romar-Gasalla, A.; Santás-Miguel, V.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M. Cadmium and Lead Sorption/Desorption on Non-Amended and By-Product-Amended Soil Samples and Pyritic Material. Water 2017, 9, 886. https://doi.org/10.3390/w9110886
Núñez-Delgado A, Romar-Gasalla A, Santás-Miguel V, Fernández-Sanjurjo MJ, Álvarez-Rodríguez E, Nóvoa-Muñoz JC, Arias-Estévez M. Cadmium and Lead Sorption/Desorption on Non-Amended and By-Product-Amended Soil Samples and Pyritic Material. Water. 2017; 9(11):886. https://doi.org/10.3390/w9110886
Chicago/Turabian StyleNúñez-Delgado, Avelino, Aurora Romar-Gasalla, Vanesa Santás-Miguel, María J. Fernández-Sanjurjo, Esperanza Álvarez-Rodríguez, Juan Carlos Nóvoa-Muñoz, and Manuel Arias-Estévez. 2017. "Cadmium and Lead Sorption/Desorption on Non-Amended and By-Product-Amended Soil Samples and Pyritic Material" Water 9, no. 11: 886. https://doi.org/10.3390/w9110886
APA StyleNúñez-Delgado, A., Romar-Gasalla, A., Santás-Miguel, V., Fernández-Sanjurjo, M. J., Álvarez-Rodríguez, E., Nóvoa-Muñoz, J. C., & Arias-Estévez, M. (2017). Cadmium and Lead Sorption/Desorption on Non-Amended and By-Product-Amended Soil Samples and Pyritic Material. Water, 9(11), 886. https://doi.org/10.3390/w9110886









