Estimating the Impact of Inflow on the Chemistry of Two Different Caldera Type Lakes Located on the Bali Island (Indonesia)
Abstract
:1. Introduction
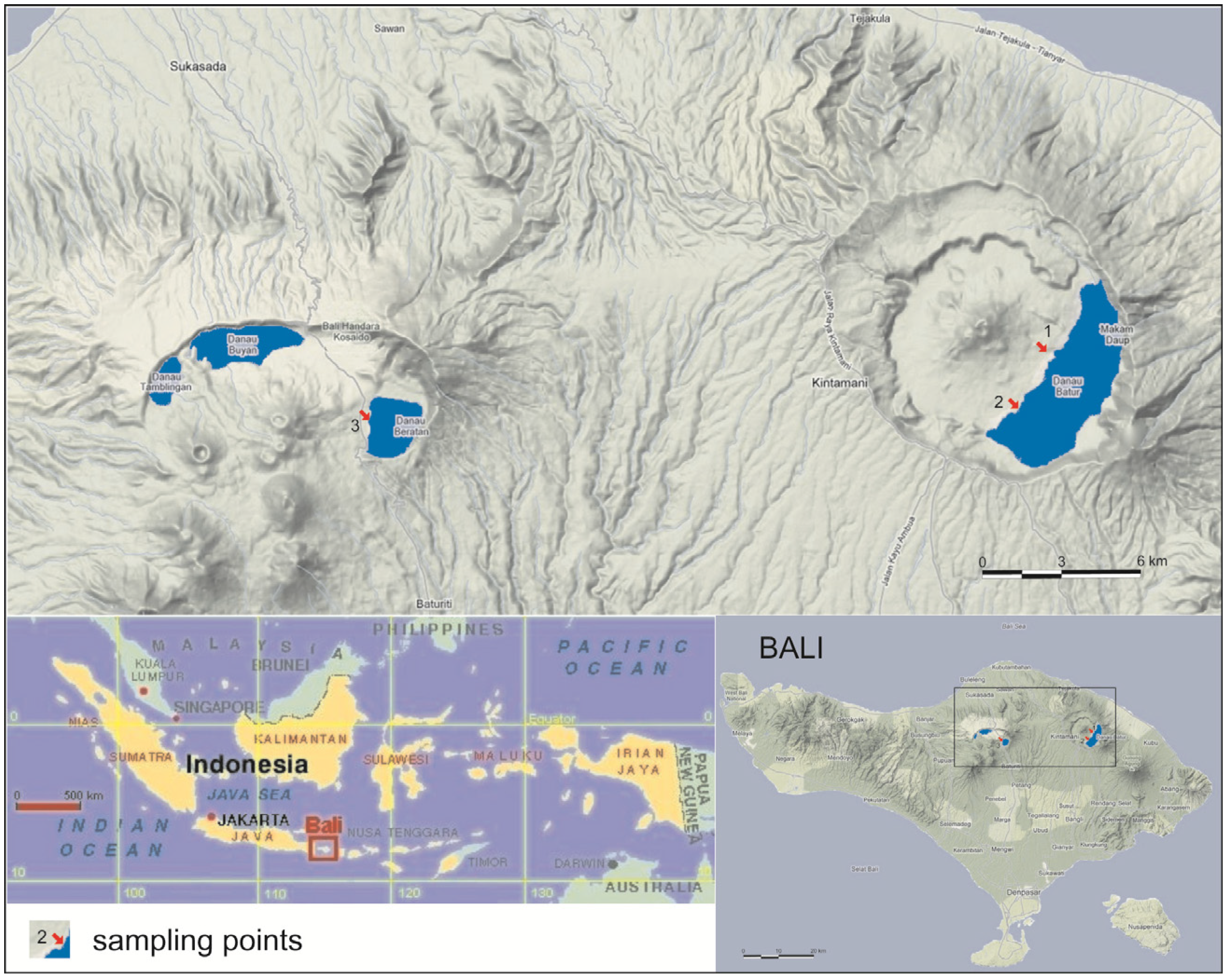
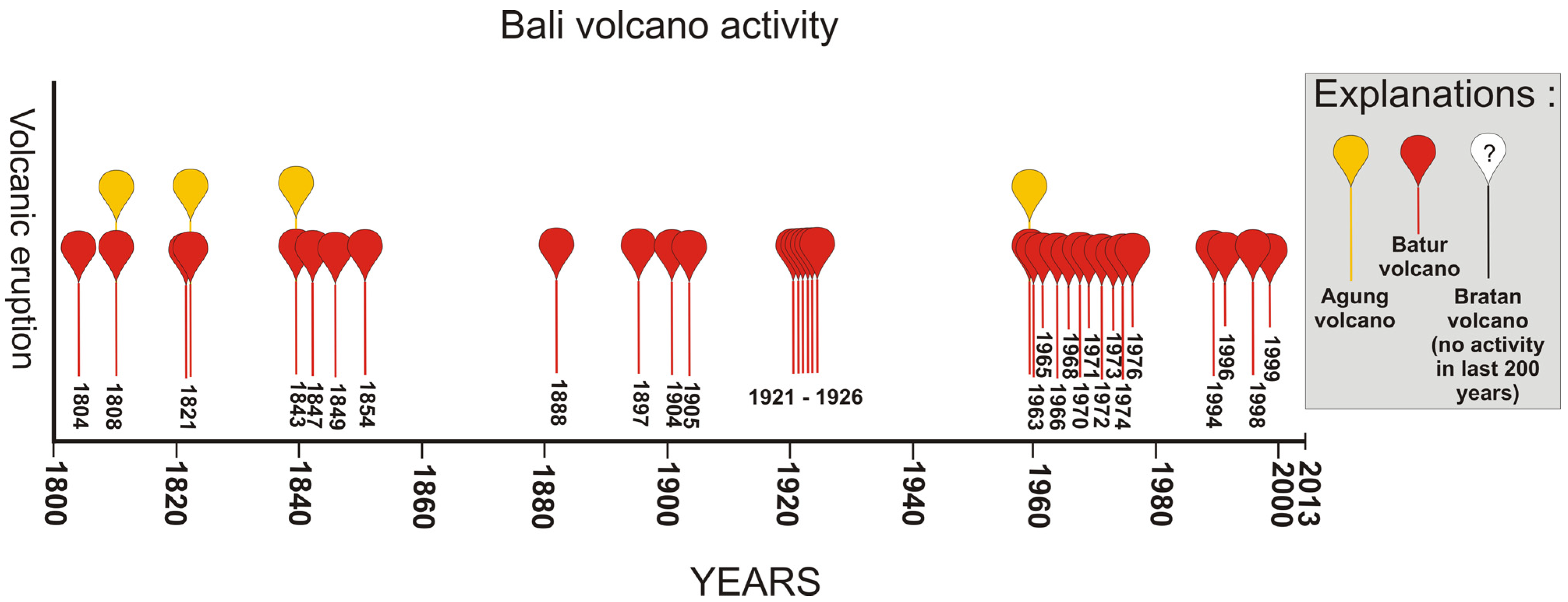
2. Experimental
2.1. Field Site
| Bratan Caldera | Batur Caldera |
|---|---|
| Morphological characteristics | |
| 1231 m above sea level, area 3.8 km2, maximum depth 22 m | 1031 m above sea level, area 15.9 km2, maximum depth 88 m |
| Volcanic activity | |
| Earlier than around 500 ka. BP | Around 500 ka. BP |
| No information about volcanic eruptions | 28 volcanic eruptions (from 1804 to 2000) |
| Geochemistry and petrology of rocks | |
|
|
| Lake characteristics | |
|
|
2.2. Sampling
2.3. Analytical Methods
| Parameter | Measurement Range | LOD | LOQ | CV (%) | Measurement Instrumentation | |
|---|---|---|---|---|---|---|
| Cations a,* | 0.03–500 | 0.01 | 0.030 | 0.9 | DIONEX 3000 chromatograph (DIONEX, Sunnyvale, CA, USA) | column: Ion Pac® CS14 (3 × 250 mm); suppressor: CSRS-300, 2 mm, mobile phase: 38 mM metasulfonic acid, flow rate: 0.36 mL·min−1, detection: conductivity |
| Anions a,* | 0.06–500 | 0.055–0.09 | 0.027–0.17 | 0.6 | column: Ion Pac®AS22 (2 × 250 mm); suppressor: ASRS-300, 2 mm, mobile phase: 4.5 mM CO32−, 1.4 mM HCO3−, flow rate: 0.38 mL·min−1, detection: conductivity | |
| TOC a,* | 0.5–500 | 0.17 | 0.50 | 5.0 | TOC analyzer (TOC-VCSH/CSN, SHIMADZU, Japan) | |
| Metals ** | 0.002–1000 | 0.0007 | 0.002 | 0.5–1.5 | Elan DRC, PerkinElmer, USA gas fed to the atomizer→Ar: 0.98 L·min−1, plasma gas→Ar: 15 L·min−1 | |
| Inlet | Carrier Gas: Helium Inlet Mode: Splitless | |
|---|---|---|
| Chromatography columns | GC1: 30 m ZB-5ms, film thickness 0.25 µm, inner diameter 0.25 mm. GC2: 1.5 m BPX50, film thickness 0.10 µm, inner diameter 0.1 mm. Postcolumn: 0.21 m BPX50, film thickness 0.10, inner diameter 0.1 mm | |
| Oven temperature | GC1: Target Temp (°C): 40 (Initial) Duration (min): 1.0 Rate (°C/min): 7.0 Target Temp (°C): 230 Duration (min): 10 | GC2: Target Temp (°C): 70 (Initial) Duration (min): 1.0 Rate (°C/min): 7.0 Target Temp (°C): 260 Duration (min): 10 |
| Modulation parameters | Modulation Period (s): 5.0 Hot Pulse Time: 1.0 Cool Time Between Stages: 1.5 | |
| Mass spectrometer settings | Start Mass (u): 33 End Mass (u): 400 Acquisition Rate (spectra/second): 200 Ion Source Temperature (°C): 250 | |
| Library | NIST 2011, contains 243,893 spectra of 212,961 compounds. | |
3. Results and Discussion
| Determined Parameter (mg·L−1) | Batur Lake | Batur Spring | Bratan Lake | Major Reservoirs in Indonesia | |||
|---|---|---|---|---|---|---|---|
| Year | 2013 | 1992 | 1993 | 2013 | 2013 | 1993 | 1989–1995 |
| pH | 8.54 | 8.5 | 8.8 | 7.37 | 7.00 | - | 6.8–8.8 |
| EC 1 (µS/cm) | 1,730 | - | 1.750–1.800 | 1,609 | 27.9 | 22–27 | 22–1,811 |
| Na+ | 287 | 355 | 350 | 248 | 3.40 | 1.3 | 1.3–350 |
| K+ | 55.7 | 31 | 22 | 55.3 | 0.388 | 0.45 | 0.45–22 |
| Mg2+ | 26.9 | 68 | 0.62 | 23.8 | 1.87 | 0.85 | 0.87–1,280 |
| Ca2+ | 18.2 | 35 | 35 | 29.6 | 0.082 | 1.9 | 1.9–32 |
| SO42− | 394 | 491 | 650–670 | 287 | 1.85 | 0.35–0.50 | 0.35–49,350 |
| F− | 2.71 | 0.86 | - | 2.73 | 0.504 | - | - |
| Cl− | 165 | 237 | 225 | 144 | 1.15 | 1.5 | 1.0–26,700 |
| Br− | 0.723 | - | - | 0.881 | 0.118 | - | - |
| Total N | 0.22 | - | 0.256–0.970 | 3.39 | 0.030 | 0.310–1.310 | 0.116–1.310 |
| Total P | 2.03 | - | 0.028 | 2.08 | 0.360 | 0.002 | 0–0.080 |
| Determined Parameter (µg·L−1) | Batur Lake 2013 | Batur Spring 2013 | Batur 1992 | Batur 1993 | Batur Rocks (ppm) | Bratan Lake 2013 | Major Reservoirs in Indonesia |
|---|---|---|---|---|---|---|---|
| Li | 423 | 361 | 400 | <LOD | |||
| B | 1440 | 2035 | 1490 | 53 | |||
| Al | 2.4 | 1.1 | 8.0 | ||||
| V | 34.8 | 80.0 | 5.7–309 | 0.4 | |||
| Cr | 3.8 | 6.2 | n.d.1 | 1.5–29 | 0.5 | n.d.1 | |
| Mn | 0.4 | <LOD | 0.4 | 0–0.14 | |||
| Co | <LOD | <LOD | 2–45 | <LOD | |||
| Ni | <LOD | <LOD | n.d.1 | 2.0–43 | <LOD | n.d.1 | |
| Cu | 2.2 | 2.6 | n.d.1 | 7.4–139 | 0.4 | n.d.1 | |
| Zn | 1.7 | 2.3 | 0–0.34 | 1.0 | 0–0.30 | ||
| As | 4.1 | 29.2 | 0.2 | ||||
| Se | 0.94 | 3.80 | <LOD | ||||
| Rb | 62.7 | 67.9 | 18–45 | 3.33 | |||
| Sr | 96 | 140 | 1500 | 168–484 | 11 | ||
| Mo | 3.15 | 5.76 | 0.6–2.2 | <LOD | |||
| Cd | <LOD | <LOD | n.d.1 | <LOD | n.d.1 | ||
| Sn | <LOD | 0.50 | <LOD | ||||
| Cs | 3.13 | 4.29 | 0.3–2.5 | 0.04 | |||
| Ba | 11.2 | 4.70 | 168–504 | 2.2 | |||
| Pb | <LOD | <LOD | n.d.1 | 3.3–12.9 | <LOD | n.d.1 | |
| U | 0.113 | 0.474 | 0.4–1.7 | <LOD | |||
| Be, Ag, Sb, Tl, Bi | <LOD | <LOD | <LOD |
3.1. Main Elements

| Compared Data | Regresion Curve | R2 |
|---|---|---|
| Batur Lake/Batur Spring | y = 0.926x − 9.02 | 0.997 |
| Batur Lake/Bratan Lake | y = 0.016x − 0.591 | 0.965 |
| Batur Spring/Bratan Lake | y = 0.017x − 0.464 | 0.980 |
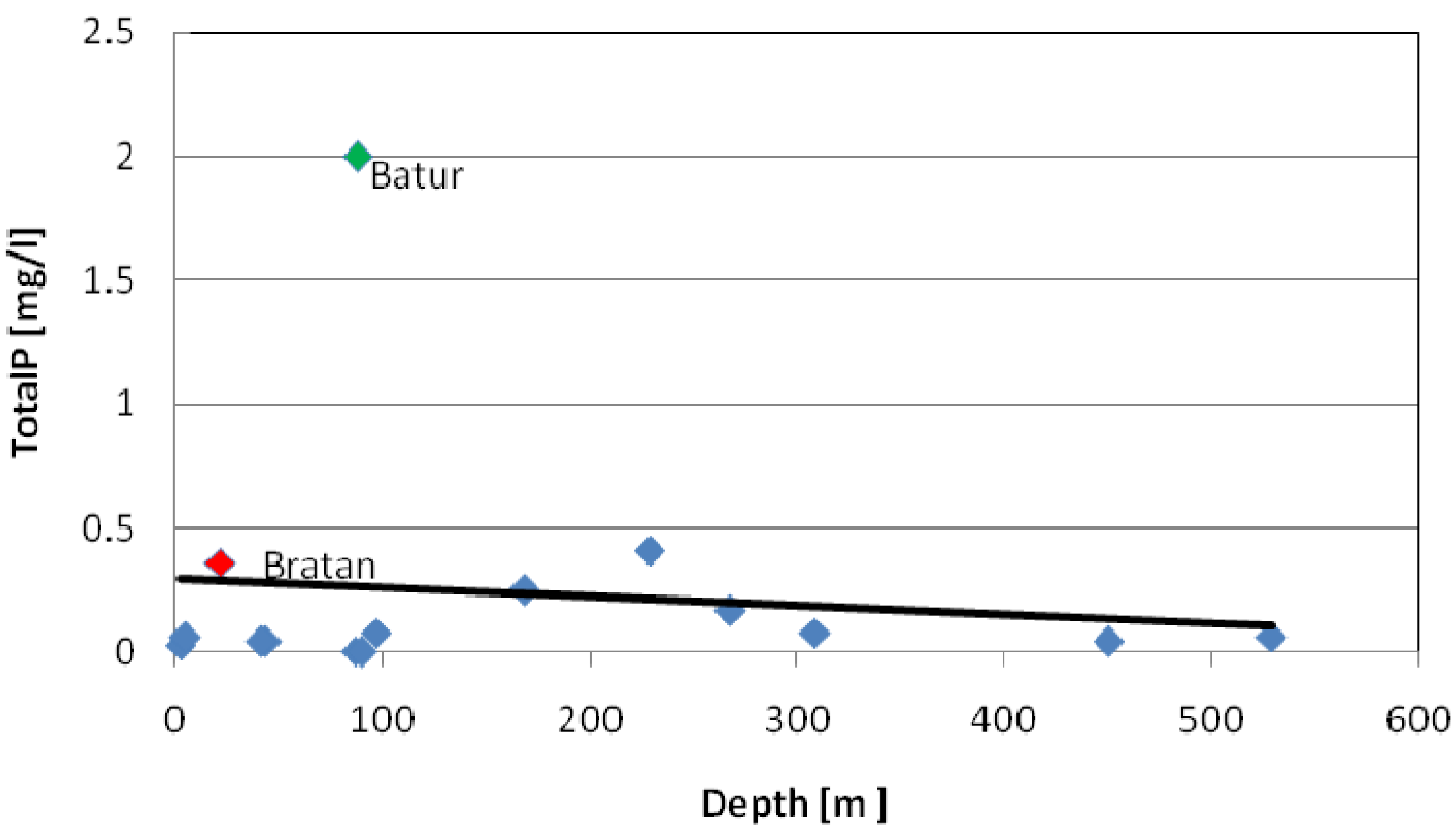
3.2. Trace Elements
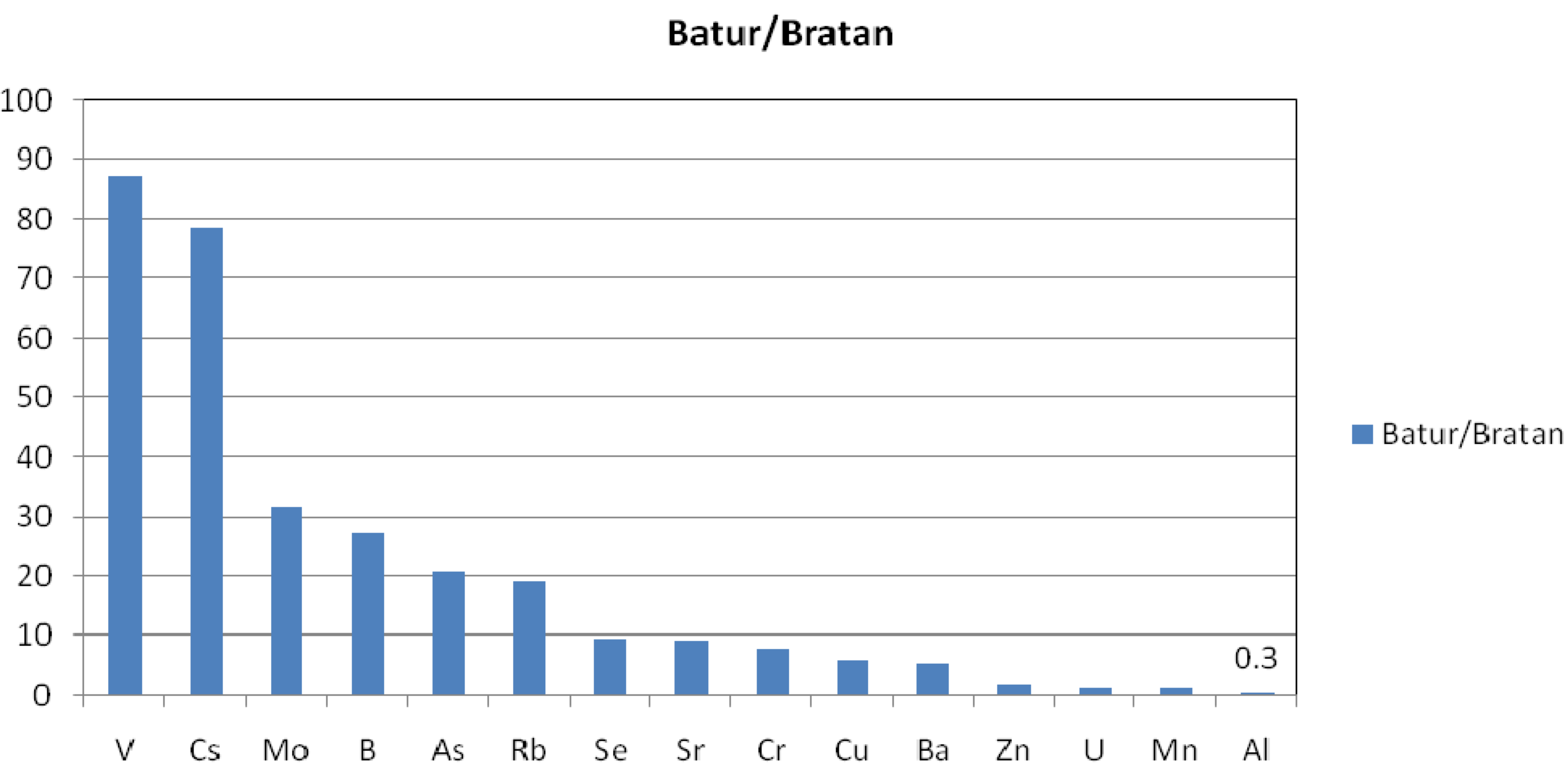
| Compared Data | Regresion Curve | R2 |
|---|---|---|
| Batur Lake/Batur Spring | y = 0.926x − 9.02 | 0.997 |
| Batur Lake/Bratan Lake | y = 0.016x − 0.591 | 0.965 |
| Batur Spring/Bratan Lake | y = 0.017x − 0.464 | 0.980 |
3.3. TOC and Organic Compounds
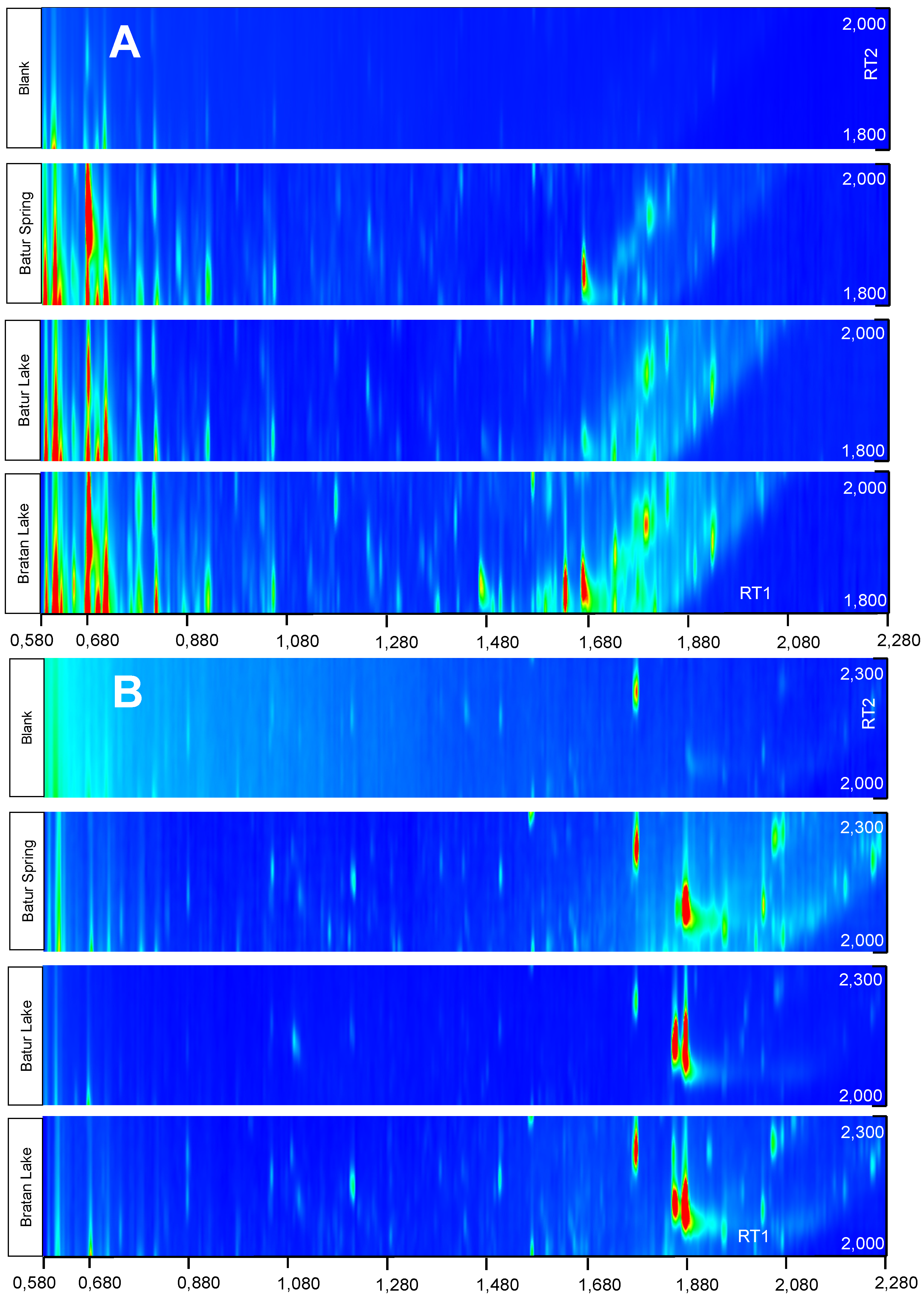

| Compounds Identified | Retention Time RT1; RT2 | Blank | Batur Lake | Batur Spring | Bratan Lake |
|---|---|---|---|---|---|
| RT: 1650–1800 | |||||
| Halocarbons | + | +++ | +++ | +++ | |
| RT: 1800–2000 | |||||
| 2-(dodecyloxy)-ethanol | 1630; 1830 | - | + | + | +++ |
| Tetradecanoic acid (Myristic acid) | 1665; 1845 | - | - | - | +++ |
| trans-2-Undecen-1-ol | 1725; 1845 | - | - | - | - |
| RT: 2000–2300 | |||||
| 5-butyldihydro-2(3H)-furanone | 1115; 2140 | - | + | - | - |
| cis-9-Hexadecenoic acid | 1855; 2150 | - | +++ | + | +++ |
| Hexadecanoic acid | 1875; 2155 | - | +++ | +++ | +++ |
| Diethylene glycol monododecyl ether | 1920; 2225 | - | - | - | + |
| 3-Heptadecanone | 2070; 2260 | - | - | + | + |
| RT2: 2300–3000 | |||||
| Dibutyl phthalate | 1880; 2505 | - | +++ | +++ | +++ |
| 2-methyl-Eicosane | 1995; 2675 | - | + | + | + |
| Oleic Acid | 2130; 2550 | - | +++ | + | + |
| 9,12,15-Octadecatrienoic acid | 2135; 2710 | - | +++ | + | + |
| Dodecanamide | 2210; 2990 | - | - | + | - |
| Heptadecyl acetate | 2265; 2460 | - | + | + | + |
| RT2: 3000–4000 | |||||
| Hexanedioic acid, mono-(2-ethylhexyl)-ester | 1945; 3345 | - | + | + | + |
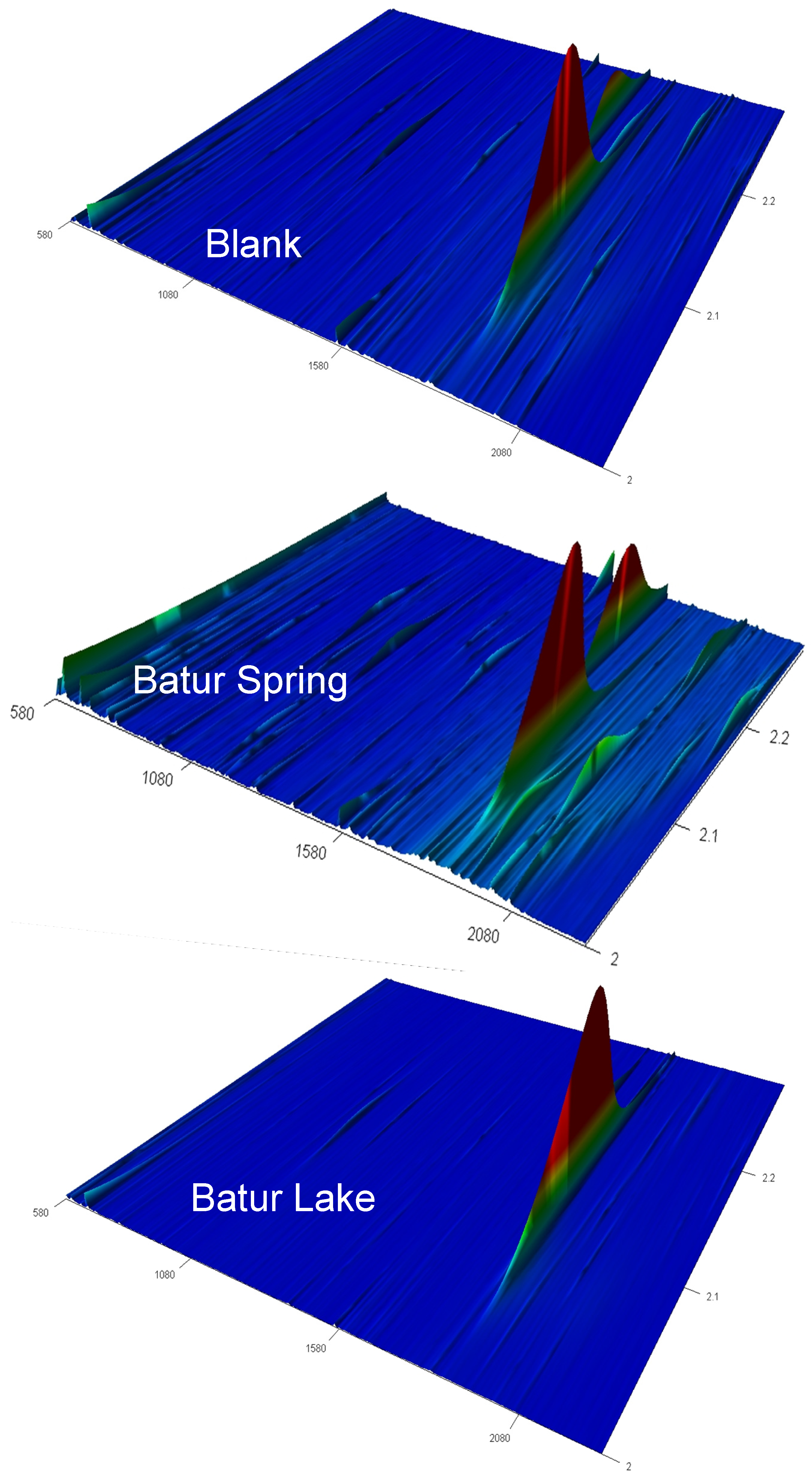
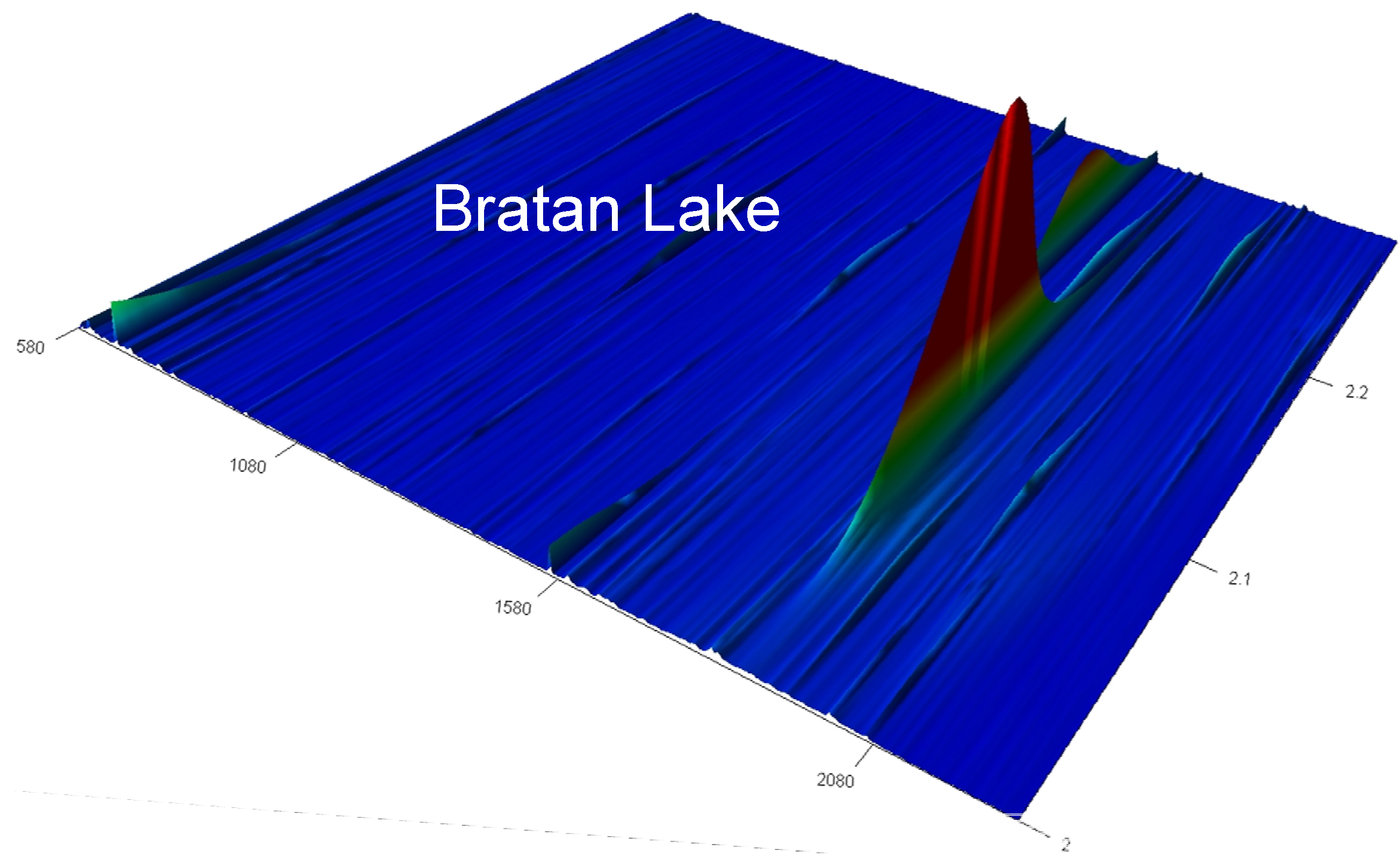
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Varekamp, J.C.; Kreulen, R. The stable isotope geochemistry of volcanic lakes, with examples from Indonesia. J. Volcanol. Geotherm. Res. 2000, 97, 309–327. [Google Scholar] [CrossRef]
- Cole, J.W.; Milne, D.M. Calderas and caldera structures: A review. Earth-Sci. Rev. 2005, 69, 1–26. [Google Scholar] [CrossRef]
- Varekamp, J.C.; Pasternack, G.B.; Rowe, G.L., Jr. Volcanic lake systematics II: Chemical constraints. J. Volcanol. Geotherm. Res. 2000, 97, 161–179. [Google Scholar] [CrossRef]
- Reubi, O.; Nicholls, I.A. Structure and Dynamics of a Silicic Magmatic System Associated with Caldera-Forming Eruptions at Batur Volcanic Field, Bali, Indonesia. J. Petrol. 2005, 46, 1367–1391. [Google Scholar] [CrossRef]
- Sutawidjaja, I.S. Ingnimbrite analyses of Batur Caldera Bali, based on 14C dating. J. Geol. Indones. 2008, 4, 189–202. [Google Scholar]
- Watanabe, K.; Yamanaka, T.; Harijoko, A.; Saitra, C.; Warmada, I.W. Caldera activities in North Bali Indonesia. J. Southeast Asian Appl. Geol. 2010, 2, 283–290. [Google Scholar]
- Lehmusluoto, P.; Machbub, B.; Terangna, N.; Rusmiputro, S.; Achmad, F.; Boer, L.; Brahmana, S.S.; Priadi, B.; Setiadji, B.; Sayuman, O.; et al. National Inventory of the Major Lakes and Reservoirs in Indonesia—General Limnology; Expedition Indodanau Technical Report; University of Helsinki: Helsinki, Finland, 1997. [Google Scholar]
- Tsuzuki, M.; Ohnuma, E.; Sato, N.; Takaku, T.; Kawaguchi, A. Effects of CO2 Concentration during Growth on Fatty Acid Composition in Microalgae. Plant Physiol. 1990, 93, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Delmelle, P.; Bernard, A. Downstream composition changes of acidic volcanic waters discharged into the Banyupahit stream, Ijen caldera, Indonesia. J. Volcanol. Geotherm. Res. 2000, 97, 55–75. [Google Scholar] [CrossRef]
- Reubi, O.; Nicholls, I.A. Magmatic evolution at Batur volcanic field, Bali, Indonesia: Petrological evidence for polybaric fractional crystallization and implications for caldera-forming eruptions. J. Volcanol. Geotherm. Res. 2004, 138, 345–369. [Google Scholar] [CrossRef]
- Moune, S.; Gauthier, P.J.; Delmelle, P. Trace elements in the particulate phase of the plume of Masaya Volcano, Nicaragua. J. Volcanol. Geotherm. Res. 2010, 193, 232–244. [Google Scholar] [CrossRef]
- Delmelle, P. Environmental impacts of tropospheric volcanic gas plumes. Volcan. Degassing 2003, 213, 381–399. [Google Scholar]
- Kim, J. Fate of THMs and HAAs in low TOC surface water. Environ. Res. 2009, 109, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Parszuto, K.; Teodorowicz, M.; Grochowska, J. Relationship between organic carbon and other measures of organic matter in the waters of Lake Isąg. Limnol. Rev. 2006, 6, 233–238. [Google Scholar]
- Greenwood, P.F.; Brocks, J.J.; Grice, K.; Schwark, L.; Jaraula, C.M.B.; Dick, J.M.; Evans, K.A. Organic geochemistry and mineralogy. I: Characterization of organic matter associated with metal deposits. Ore Geol. Rev. 2013, 50, 1–27. [Google Scholar] [CrossRef]
- Baecelona, M.J. TOC determinations in ground water. Ground Water 1984, 22, 18–24. [Google Scholar] [CrossRef]
- Henriksen, A.; Brakke, D.F.; Nortonc, S.A. Total organic carbon concentrations in acidic lakes in Southern Norway. Environ. Sci. Technol. 1988, 22, 1103–1105. [Google Scholar] [CrossRef] [PubMed]
- Řezanka, T.; Dor, I.; Prell, A.; Dembitsky, V.M. Fatty acid composition of six freshwater wild cyanobacterial species. Folia Microbiol. 2003, 48, 71–75. [Google Scholar] [CrossRef]
- Kaneshiro, T.; Nakamura, L.K.; Bagby, M.O. Oleic acid transformations by selected strains of Sphingobacterium thalpophilum and Bacillus cereus from composted manure. Curr. Microbiol. 1995, 31, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Sushanta, K.S.; Lakshmanan, U.; Gopalakrishnan, S. Nitrogen stress induced changes in the marine cyanobacterium Oscillatoria willei BDU 130511, FEMS. Microbiol. Ecol. 2003, 45, 263–272. [Google Scholar] [CrossRef]
- Liu, J.; Shi, L. Analysis of chemical composition of precipitation in a typical background site. J. Environ. Sci. 1998, 10, 325–333. [Google Scholar]
- Bruijnzeel, L.A. Nutrient content of bulk precipitation in south-central Java, Indonesia. J. Trop. Ecol. 1989, 5, 187–202. [Google Scholar] [CrossRef]
- Sanford, W.E.; Konikow, L.F.; Rowe, G.; Brantley, S.L. Ground-water transport of crater-lake brine at Poas Volcano, Costa Rica. J. Volcanol. Geotherm. Res. 1995, 64, 271–297. [Google Scholar]
- Wheller, G.E.; Varne, R. Genesis of dacitic magmatism at Batur volcano, Bali, Indonesia: Implications for the origins of stratovolcano calderas. J. Volcanol. Geotherm Res. 1986, 28, 363–378. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Polkowska, Ż.; Wolska, L.; Łęczyński, L.; Ruman, M.; Lehmann, S.; Kozak, K.; Matysik, M.; Absalon, D. Estimating the Impact of Inflow on the Chemistry of Two Different Caldera Type Lakes Located on the Bali Island (Indonesia). Water 2015, 7, 1712-1730. https://doi.org/10.3390/w7041712
Polkowska Ż, Wolska L, Łęczyński L, Ruman M, Lehmann S, Kozak K, Matysik M, Absalon D. Estimating the Impact of Inflow on the Chemistry of Two Different Caldera Type Lakes Located on the Bali Island (Indonesia). Water. 2015; 7(4):1712-1730. https://doi.org/10.3390/w7041712
Chicago/Turabian StylePolkowska, Żaneta, Lidia Wolska, Leszek Łęczyński, Marek Ruman, Sara Lehmann, Katarzyna Kozak, Magdalena Matysik, and Damian Absalon. 2015. "Estimating the Impact of Inflow on the Chemistry of Two Different Caldera Type Lakes Located on the Bali Island (Indonesia)" Water 7, no. 4: 1712-1730. https://doi.org/10.3390/w7041712
APA StylePolkowska, Ż., Wolska, L., Łęczyński, L., Ruman, M., Lehmann, S., Kozak, K., Matysik, M., & Absalon, D. (2015). Estimating the Impact of Inflow on the Chemistry of Two Different Caldera Type Lakes Located on the Bali Island (Indonesia). Water, 7(4), 1712-1730. https://doi.org/10.3390/w7041712







