Analysis of Human Health Risk Related to the Exposure of Arsenic Concentrations and Temporal Variation in Groundwater of a Semi-Arid Region in Mexico
Abstract
1. Introduction
2. Materials and Methods
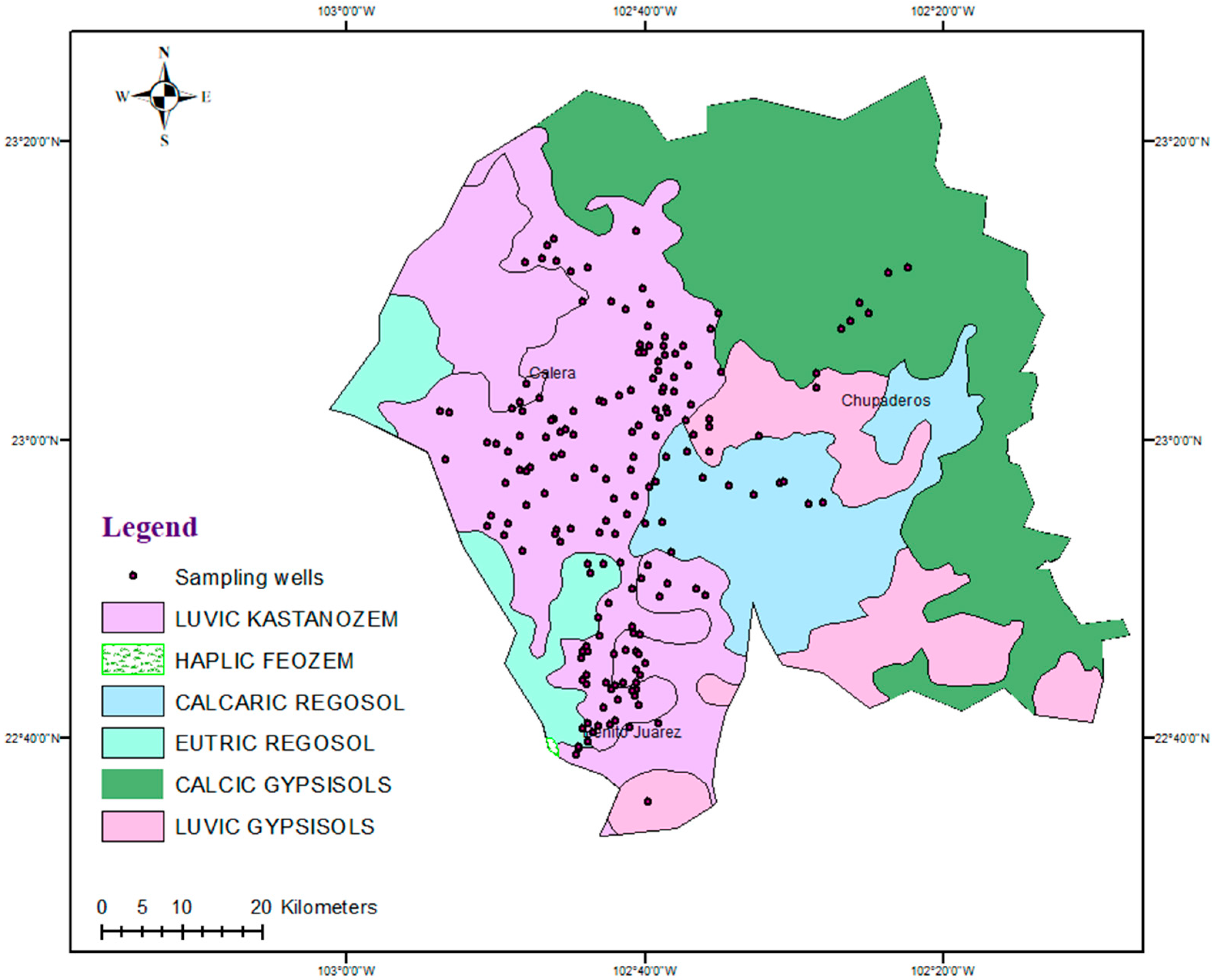
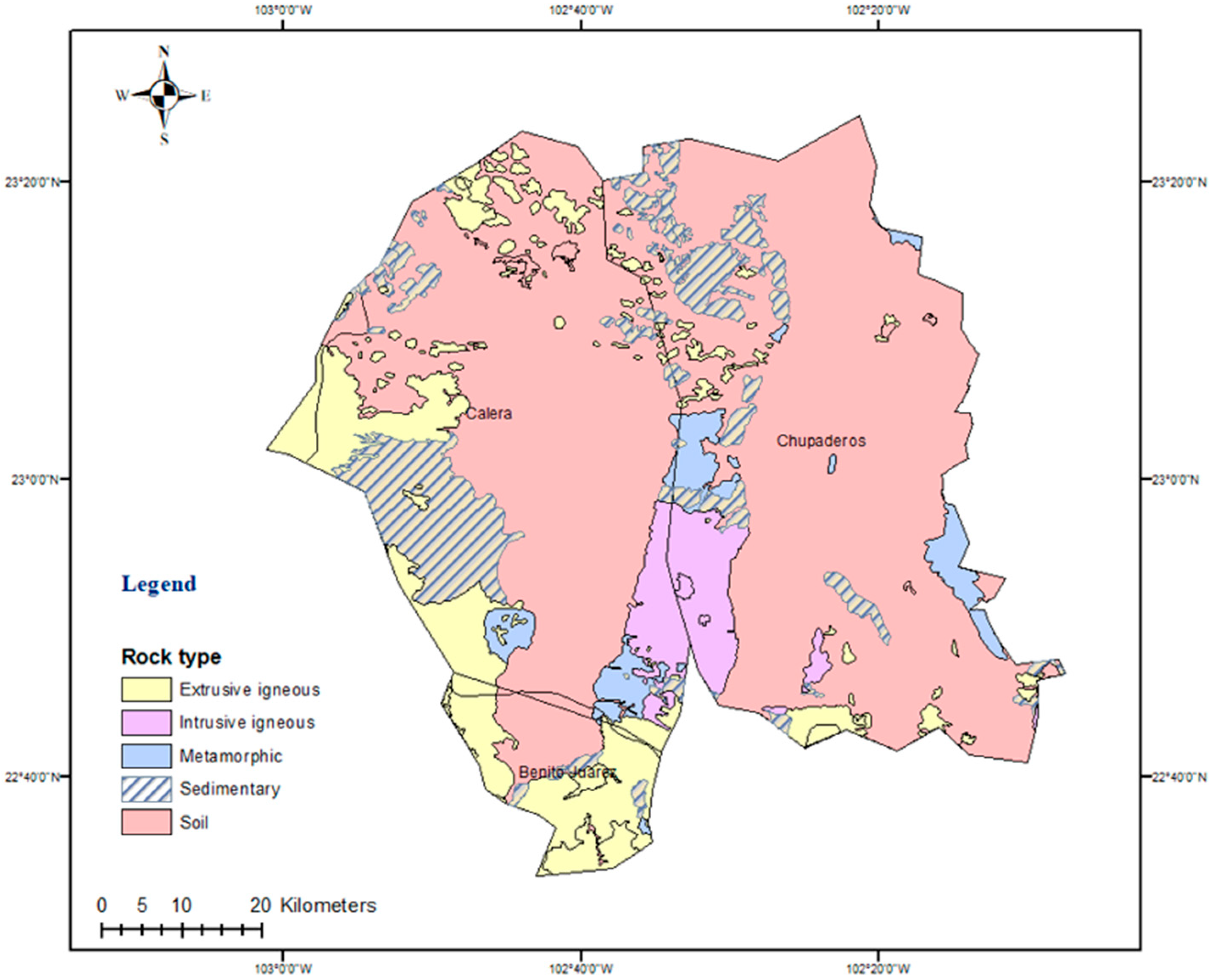
2.1. Study Area
2.2. Sample Collection and Concentration Determination
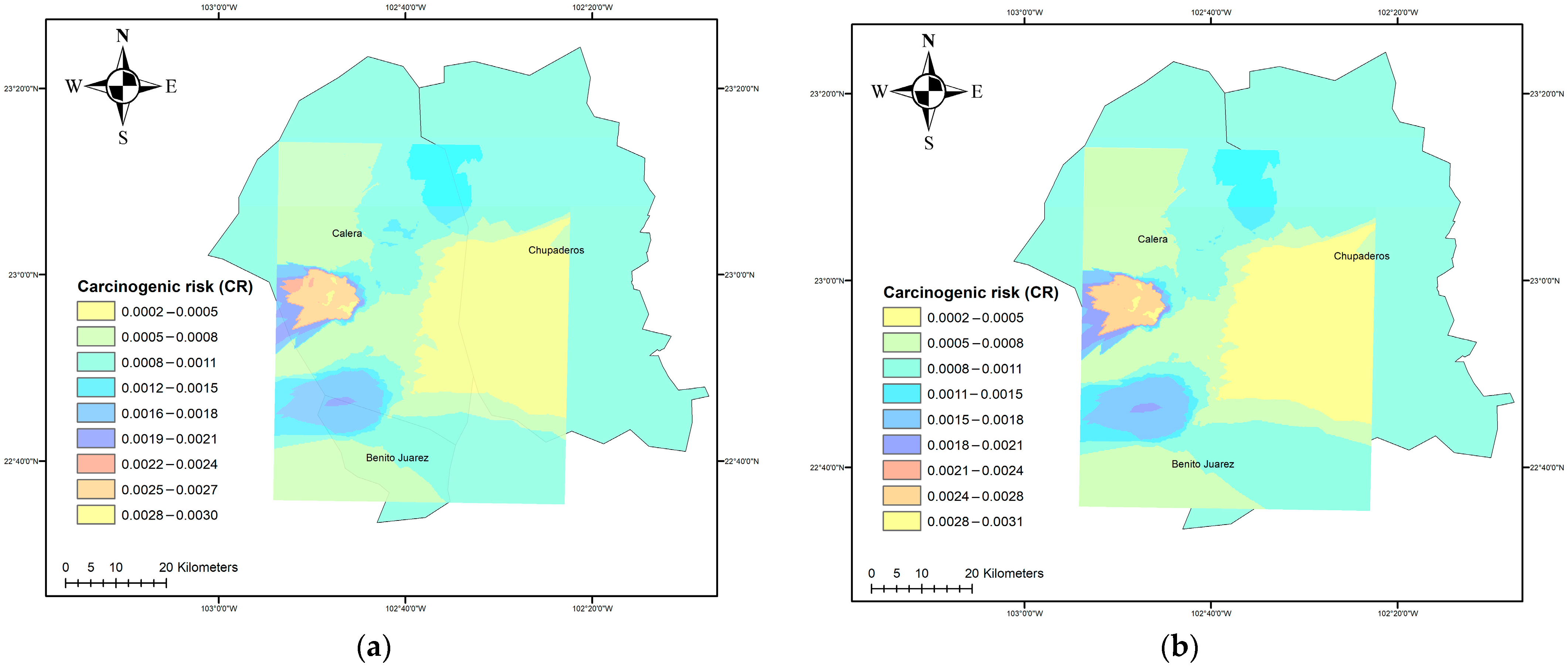
2.3. Mapping of Parameters
2.4. Human Health Risk Assessment
2.4.1. Non-Carcinogenic Risk
2.4.2. Carcinogenic Risk (CR)
2.5. Statistical Data
3. Results
3.1. Arsenic Concentrations
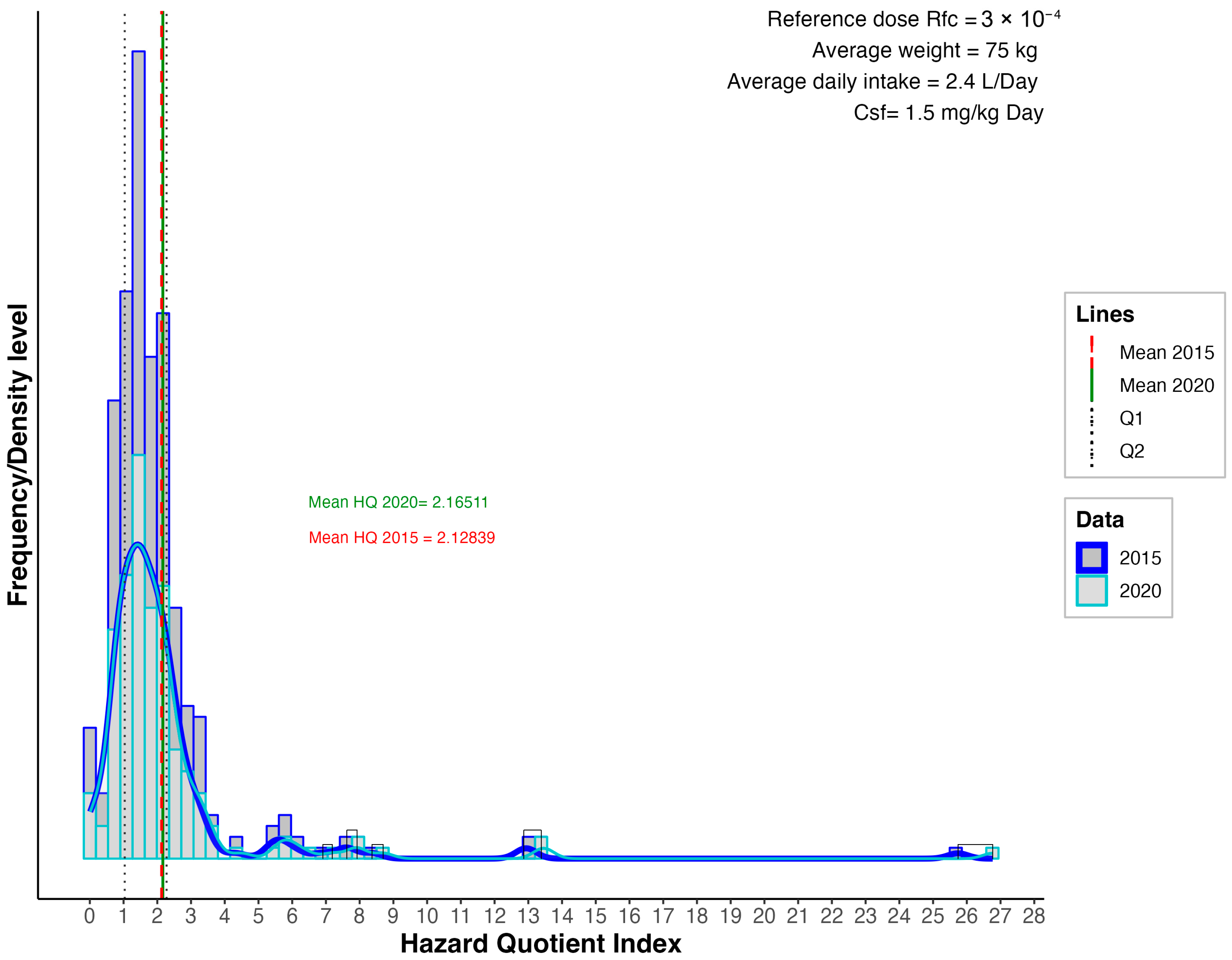
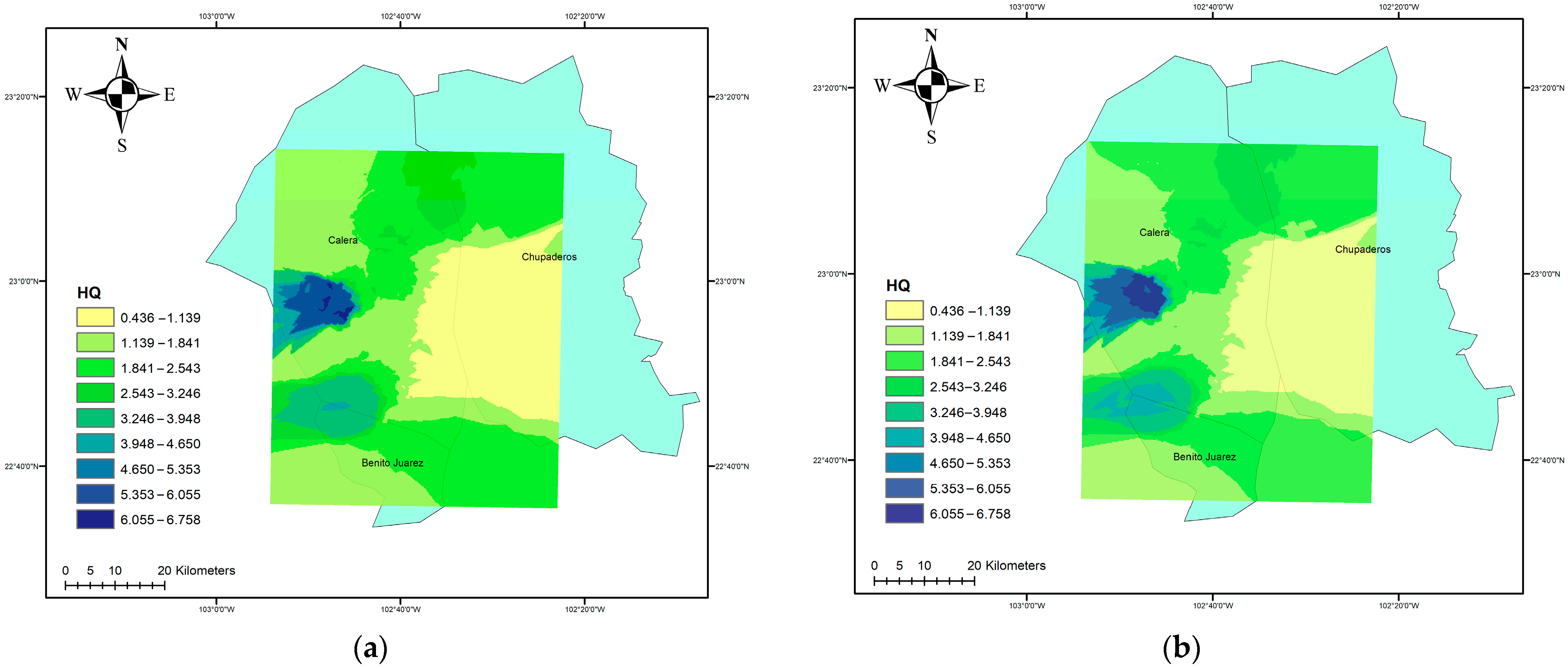
3.2. Non-Carcinogenic Risk
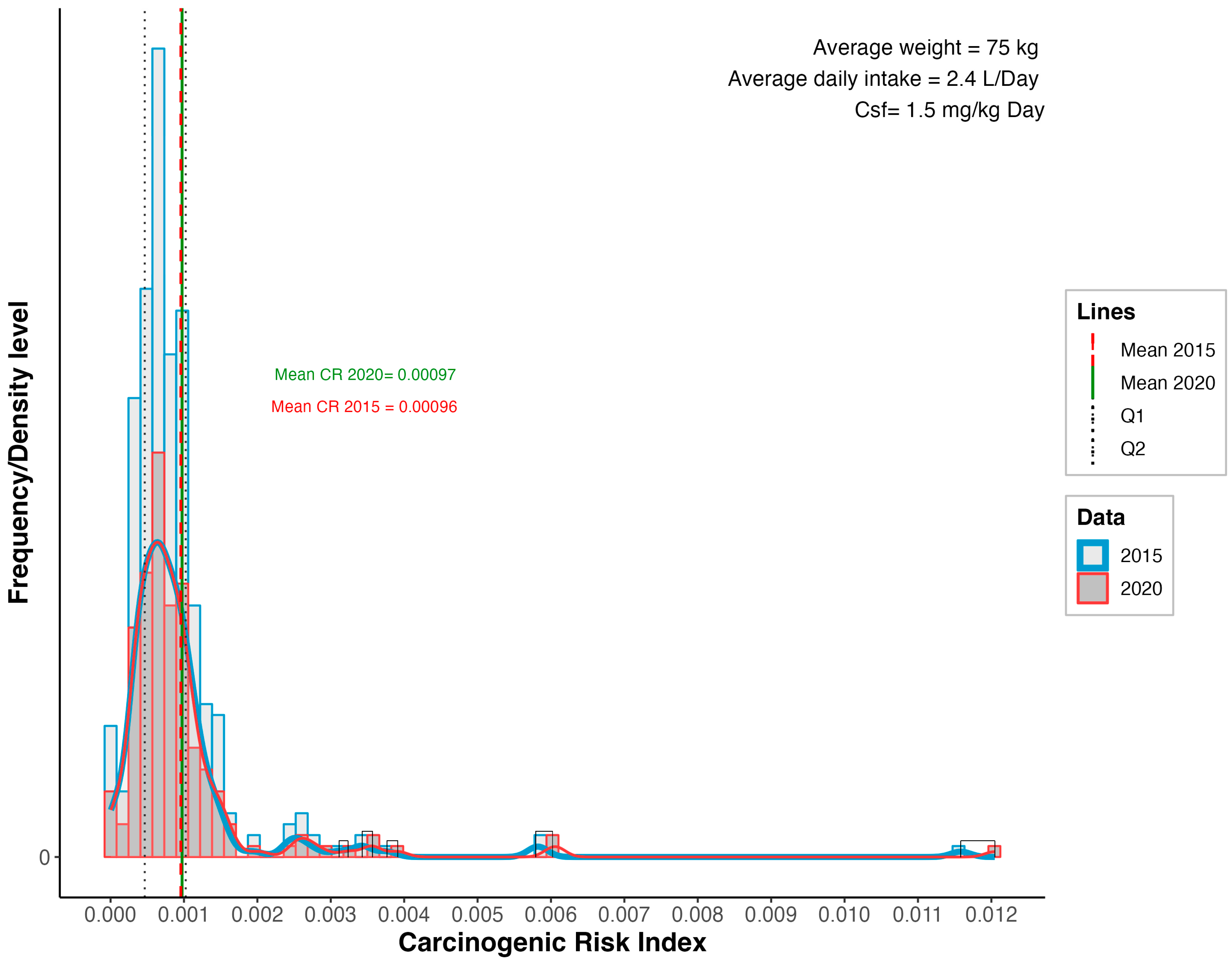
3.3. Carcinogenic Risk (CR)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| As | Arsenic |
| AAS | Administrative Aquifers |
| HQ | Hazard Quotient |
| ADD | Daily Dose |
| RFD | Reference Dose |
| CR | Carcinogenic Risk |
| CFS | Cancer Slope Factor |
References
- Boonkaewwan, S.; Sonthiphand, P.; Chotpantarat, S. Mechanisms of arsenic contamination associated with hydrochemical characteristics in coastal alluvial aquifers using multivariate statistical technique and hydrogeochemical modeling: A case study in Rayong province, eastern Thailand. Environ. Geochem. Health 2021, 43, 537–566. [Google Scholar] [CrossRef] [PubMed]
- Foust, R.D.; Mohapatra, P.; Compton-O’Brien, A.M.; Reifel, J. Groundwater arsenic in the Verde Valley in central Arizona, USA. Appl. Geochem. 2004, 19, 251–255. [Google Scholar] [CrossRef]
- Powers, M.; Yracheta, J.; Harvey, D.; O’Leary, M.; Best, L.G.; Black Bear, A.; MacDonald, L.; Susan, J.; Hasan, K.; Thomas, E.; et al. Arsenic in groundwater in private wells in rural North Dakota and South Dakota: Water quality assessment for an intervention trial. Environ. Res. 2019, 168, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Alipour, E.; Aghapour, A.A.; Bahrami Asl, F. Concentration, spatial distribution, and non-carcinogenic risk assessment of arsenic, cadmium, chromium, and lead in drinking water in rural areas of eight cities of West Azarbaijan province, Iran. Environ. Sci. Pollut. Res. 2024, 31, 20222–20233. [Google Scholar] [CrossRef] [PubMed]
- Camacho, L.M.; Gutiérrez, M.; Alarcón-Herrera, M.T.; Villalba, M.d.L.; Deng, S. Occurrence and treatment of arsenic in groundwater and soil in northern Mexico and southwestern USA. Chemosphere 2011, 83, 211–225. [Google Scholar] [CrossRef] [PubMed]
- Alarcón-Herrera, M.T.; Bundschuh, J.; Nath, B.; Nicolli, H.B.; Gutierrez, M.; Reyes-Gomez, V.M.; Nuñez, D.; Martín-Dominguez, I.R.; Sracek, O. Co-occurrence of arsenic and fluoride in groundwater of semi-arid regions in Latin America: Genesis, mobility and remediation. J. Hazard. Mater. 2013, 262, 960–969. [Google Scholar] [CrossRef] [PubMed]
- Kumarathilaka, P.; Seneweera, S.; Meharg, A.; Bundschuh, J. Arsenic speciation dynamics in paddy rice soil-water environment: Sources, physico-chemical, and biological factors—A review. Water Res. 2018, 140, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Tume, P.; Melipichún, T.; Ferraro, F.; Sepúlveda, B.; Roca, N.; Bech, J. Contamination of As, Cd, Cr, Hg and Pb in soils in Arica commune (Chile). Environ. Geochem. Health 2023, 45, 9199–9213. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.S.; Pandey, P.K.; Martín-Ramos, P.; Corns, W.T.; Varol, S.; Bhattacharya, P.; Zhu, Y. A review on arsenic in the environment: Contamination, mobility, sources, and exposure. RSC Adv. 2023, 13, 8803–8821. [Google Scholar] [CrossRef] [PubMed]
- Raju, N.J. Arsenic in the geo-environment: A review of sources, geochemical processes, toxicity and removal technologies. Environ. Res. 2022, 203, 111782. [Google Scholar] [CrossRef] [PubMed]
- Fazi, S.; Amalfitano, S.; Casentini, B.; Davolos, D.; Pietrangeli, B.; Crognale, S.; Lotti, F.; Rossetti, S. Arsenic removal from naturally contaminated waters: A review of methods combining chemical and biological treatments. Rend. Lincei 2016, 27, 51–58. [Google Scholar] [CrossRef]
- Chen, Y.f.; He, M.x.; Feng, H.m.; Liu, Q.; Chen, J.x.; Li, T. Health risk study of cadmium, chromium, lead and arsenic in reservoir water of Changzhou, China. J. Environ. Sci. Health Part A 2023, 58, 680–693. [Google Scholar] [CrossRef] [PubMed]
- Kanel, S.R.; Das, T.K.; Varma, R.S.; Kurwadkar, S.; Chakraborty, S.; Joshi, T.P.; Bezbaruah, A.N.; Nadagouda, M.N. Arsenic Contamination in Groundwater: Geochemical Basis of Treatment Technologies. ACS Environ. Au 2023, 3, 135–152. [Google Scholar] [CrossRef] [PubMed]
- Planer-Friedrich, B.; Armienta, M.A.; Merkel, B.J. Origin of arsenic in the groundwater of the Rioverde basin, Mexico. Environ. Geol. 2001, 40, 1290–1298. [Google Scholar] [CrossRef]
- Costa, M. Review of arsenic toxicity, speciation and polyadenylation of canonical histones. Toxicol. Appl. Pharmacol. 2019, 375, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, H.; Kay, P.; Slack, R.; Gong, Y.Y.; Carter, A. Human exposure assessment of different arsenic species in household water sources in a high risk arsenic area. Sci. Total Environ. 2017, 584–585, 631–641. [Google Scholar] [CrossRef] [PubMed]
- U.S. EPA. 2010 U.S. Environmental Protection Agency (EPA) Decontamination Research and Development Conference; U.S. Environmental Protection Agency: Washington, DC, USA, 2011.
- Basu, A.; Saha, D.; Saha, R.; Ghosh, T.; Saha, B. A review on sources, toxicity and remediation technologies for removing arsenic from drinking water. Res. Chem. Intermed. 2014, 40, 447–485. [Google Scholar] [CrossRef]
- Lapworth, D.; Boving, T.; Brauns, B.; Dottridge, J.; Hynds, P.; Kebede, S.; Kreamer, D.; Misstear, B.; Mukherjee, A.; Re, V.; et al. Groundwater quality: Global challenges, emerging threats and novel approaches. Hydrogeol. J. 2023, 31, 15–18. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2011; Available online: https://www.who.int/publications/i/item/9789241548151 (accessed on 26 June 2025).
- Nickson, R.; McArthur, J.; Burgess, W.; Matin Ahmed, K.; Ravenscroft, P.; Rahman, M. Arsenic poisoning of Bangladesh groundwater. Nature 1998, 395, 338. [Google Scholar] [CrossRef] [PubMed]
- Ullah, Z.; Rashid, A.; Ghani, J.; Talib, M.A.; Shahab, A.; Lun, L. Arsenic Contamination, Water Toxicity, Source Apportionment, and Potential Health Risk in Groundwater of Jhelum Basin, Punjab, Pakistan. Biol. Trace Elem. Res. 2022, 201, 514–524. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Kim, K.H.; Kumar, V.; Kim, S. A review of functional sorbents for adsorptive removal of arsenic ions in aqueous systems. J. Hazard. Mater. 2020, 388, 121815. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.Y.; Jeon, H.; Shin, H.S. Risk Assessment and Determination of Arsenic and Heavy Metals in Fishery Products in Korea. Foods 2023, 12, 3750. [Google Scholar] [CrossRef] [PubMed]
- Jaafarzadeh, N.; Poormohammadi, A.; Almasi, H.; Ghaedrahmat, Z.; Rahim, F.; Zahedi, A. Arsenic in drinking water and kidney cancer: A systematic review. Rev. Environ. Health 2023, 38, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Samiee, F.; Leili, M.; Faradmal, J.; Torkshavand, Z.; Asadi, G. Exposure to arsenic through breast milk from mothers exposed to high levels of arsenic in drinking water: Infant risk assessment. Food Control 2019, 106, 106–669. [Google Scholar] [CrossRef]
- Zhao, C.; Yang, J.; Zheng, Y.; Yang, J.; Guo, G.; Wang, J.; Chen, T. Effects of environmental governance in mining areas: The trend of arsenic concentration in the environmental media of a typical mining area in 25 years. Chemosphere 2019, 235, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Guo, H.; Wang, Y.; Wang, G.; Wang, H.; Zang, X.; Zhu, J. Groundwater hydrogeochemical characteristics and quality suitability assessment for irrigation and drinking purposes in an agricultural region of the North China plain. Environ. Earth Sci. 2021, 80, 162. [Google Scholar] [CrossRef]
- Molina-Frechero, N.; Nevarez-Rascón, M.; Tremillo-Maldonado, O.; Vergara-Onofre, M.; Gutiérrez-Tolentino, R.; Gaona, E.; Castañeda, E.; Jarquin-Yañez, L.; Bologna-Molina, R. Environmental exposure of arsenic in groundwater associated to carcinogenic risk in underweight children exposed to fluorides. Int. J. Environ. Res. Public Health 2020, 17, 724. [Google Scholar] [CrossRef] [PubMed]
- Alejandro, I.; Guerrero, P. Evaluación de las Pérdidas de Transmisión y su Efecto en la Recarga de Agua Subterránea. Ph.D. Thesis, Universidad Autónoma de Zacatecas, Zacatecas, Mexico, 2020. [Google Scholar]
- Anuard, P.-G.; Julián, G.-T.; Hugo, J.-F.; Carlos, B.-C.; Arturo, H.-A.; Edith, O.-T.; Claudia, Á.-S. Integration of Isotopic (2H and 18O) and Geophysical Applications to Define a Groundwater Conceptual Model in Semiarid Regions. Water 2019, 11, 488. [Google Scholar] [CrossRef]
- Comisión Nacional del Agua. Actualización de la Disponibilidad Media Anual de Agua en el Acuífero Calera (3225), Estado de Zacatecas; Diario Oficial de la Federación: Mexico City, Mexico, 2018.
- Varsányi, I. Arsenic in Deep Groundwater. In Water-Rock Interaction (WRI-6), Proceedings of the 6th International Symposium, Malvern, UK, 3–6 August 1989; Miles, D.L., Ed.; CRC Press: Boca Raton, FL, USA, 1989; pp. 715–718. ISBN 9789061919704. [Google Scholar]
- Tyagi, S.; Sharma, B.; Singh, P.; Dobhal, R. Water Quality Assessment in Terms of Water Quality Index. Am. J. Water Resour. 2020, 1, 34–38. [Google Scholar] [CrossRef]
- Litter, M.I.; Ingallinella, A.M.; Olmos, V.; Savio, M.; Difeo, G.; Botto, L.; Torres, E.M.F.; Taylor, S.; Frangie, S.; Herkovits, J.; et al. Arsenic in Argentina: Technologies for arsenic removal from groundwater sources, investment costs and waste management practices. Sci. Total Environ. 2019, 690, 778–789. [Google Scholar] [CrossRef] [PubMed]
- Sohrabi, N.; Kalantari, N.; Amiri, V.; Saha, N.; Berndtsson, R.; Bhattacharya, P.; Ahmad, A. A probabilistic-deterministic analysis of human health risk related to the exposure to potentially toxic elements in groundwater of Urmia coastal aquifer (NW of Iran) with a special focus on arsenic speciation and temporal variation. Stoch. Environ. Res. Risk Assess. 2021, 35, 1509–1528. [Google Scholar] [CrossRef]
- Fadel, M.; Courcot, D.; Afif, C.; Ledoux, F. Methods for the assessment of health risk induced by contaminants in atmospheric particulate matter: A review. Environ. Chem. Lett. 2022, 20, 3289–3311. [Google Scholar] [CrossRef]
- Letechipia, J.O.; González-Trinidad, J.; Júnez-Ferreira, H.E.; Bautista-Capetillo, C.; Robles-Rovelo, C.O.; Rodríguez, A.R.C.; Dávila-Hernández, S. Aqueous Arsenic Speciation with Hydrogeochemical Modeling and Correlation with Fluorine in Groundwater in a Semiarid Region of Mexico. Water 2022, 14, 519. [Google Scholar] [CrossRef]
- Ortiz-Letechipia, J.; González-Trinidad, J.; Júnez-Ferreira, H.E.; Bautista-Capetillo, C.; Dávila-Hernández, S. Evaluation of groundwater quality for human consumption and irrigation in relation to arsenic concentration in flow systems in a semi-arid mexican region. Int. J. Environ. Res. Public Health 2021, 18, 8045. [Google Scholar] [CrossRef] [PubMed]
- Bundschuh, J.; Litter, M.I.; Parvez, F.; Román-Ross, G.; Nicolli, H.B.; Jean, J.S.; Liu, C.W.; López, D.; Armienta, M.A.; Guilherme, L.R.G.; et al. One century of arsenic exposure in Latin America: A review of history and occurrence from 14 countries. Sci. Total Environ. 2012, 429, 2–35. [Google Scholar] [CrossRef] [PubMed]
- Litter, M.I.; Armienta, M.A.; Villanueva Estrada, R.E.; Villaamil Lepori, E.C.; Olmos, V. Arsenic in Latin America: Part II BT—Arsenic in Drinking Water and Food; Srivastava, S., Ed.; Springer: Singapore, 2020; pp. 113–182. ISBN 978-981-13-8587-2. [Google Scholar]
- Navarro, O.; González, J.; Júnez-Ferreira, H.E.; Bautista, C.-F.; Cardona, A. Correlation of Arsenic and Fluoride in the Groundwater for Human Consumption in a Semiarid Region of Mexico. Procedia Eng. 2017, 186, 333–340. [Google Scholar] [CrossRef]
- García-Rico, L.; Valenzuela-Rodríguez, M.P.; Meza-Montenegro, M.M.; Lopez-Duarte, A.L. Arsenic in rice and rice products in Northwestern Mexico and health risk assessment. Food Addit. Contam. Part B Surveill. 2020, 13, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Ayala, M.B.; Megdal, S.B. An Overview of Managed Aquifer Recharge in Mexico and Its Legal Framework. Water 2020, 12, 474. [Google Scholar] [CrossRef]
- Adimalla, N.; Qian, H. Groundwater quality evaluation using water quality index (WQI) for drinking purposes and human health risk (HHR) assessment in an agricultural region of Nanganur, south India. Ecotoxicol. Environ. Saf. 2019, 176, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Zhang, Y.; Luo, J.; Wang, T.; Lian, H.; Ding, Z. Bioaccessibility and health risk of arsenic, mercury and other metals in urban street dusts from a mega-city, Nanjing, China. Environ. Pollut. 2011, 159, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Alidadi, H.; Tavakoly Sany, S.B.; Zarif Garaati Oftadeh, B.; Mohamad, T.; Shamszade, H.; Fakhari, M. Health risk assessments of arsenic and toxic heavy metal exposure in drinking water in northeast Iran. Environ. Health Prev. Med. 2019, 24. [Google Scholar] [CrossRef] [PubMed]
- Rapant, S.; Krěmová, K. Health risk assessment maps for arsenic groundwater content: Application of national geochemical databases. Environ. Geochem. Health 2007, 29, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Hamidian, A.H.; Razeghi, N.; Zhang, Y.; Yang, M. Spatial distribution of arsenic in groundwater of Iran, a review. J. Geochemical Explor. 2019, 201, 88–98. [Google Scholar] [CrossRef]
- Shah, A.H.; Shahid, M.; Khalid, S.; Natasha; Shabbir, Z.; Bakhat, H.F.; Murtaza, B.; Farooq, A.; Akram, M.; Shah, G.M. Assessment of arsenic exposure by drinking well water and associated carcinogenic risk in peri-urban areas of Vehari, Pakistan. Environ. Geochem. Health 2020, 42, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Niknejad, H.; Ala, A.; Ahmadi, F.; Mahmoodi, H.; Saeedi, R.; Gholami-Borujeni, F.; Abtahi, M. Carcinogenic and non-carcinogenic risk assessment of exposure to trace elements in groundwater resources of Sari city, Iran. J. Water Health 2023, 21, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Asere, T.G.; Stevens, C.V.; Du Laing, G. Use of (modified) natural adsorbents for arsenic remediation: A review. Sci. Total Environ. 2019, 676, 706–720. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, S.I.; Naushad, M.; Chaudhry, S.A. Promising prospects of nanomaterials for arsenic water remediation: A comprehensive review. Process Saf. Environ. Prot. 2019, 126, 60–97. [Google Scholar] [CrossRef]
- Abejón, A.; Garea, A.; Irabien, A. Arsenic removal from drinking water by reverse osmosis: Minimization of costs and energy consumption. Sep. Purif. Technol. 2015, 144, 46–53. [Google Scholar] [CrossRef]
- Weerasundara, L.; Ok, Y.-S.; Bundschuh, J. Selective removal of arsenic in water: A critical review. Environ. Pollut. 2021, 268, 115668. [Google Scholar] [CrossRef] [PubMed]
- Biedunkova, O.; Kuznietsov, P. Integration of water management in the assessment of the impact of heavy metals discharge from the power plant with mitigation strategies. Ecol. Indic. 2025, 175, 113618. [Google Scholar] [CrossRef]
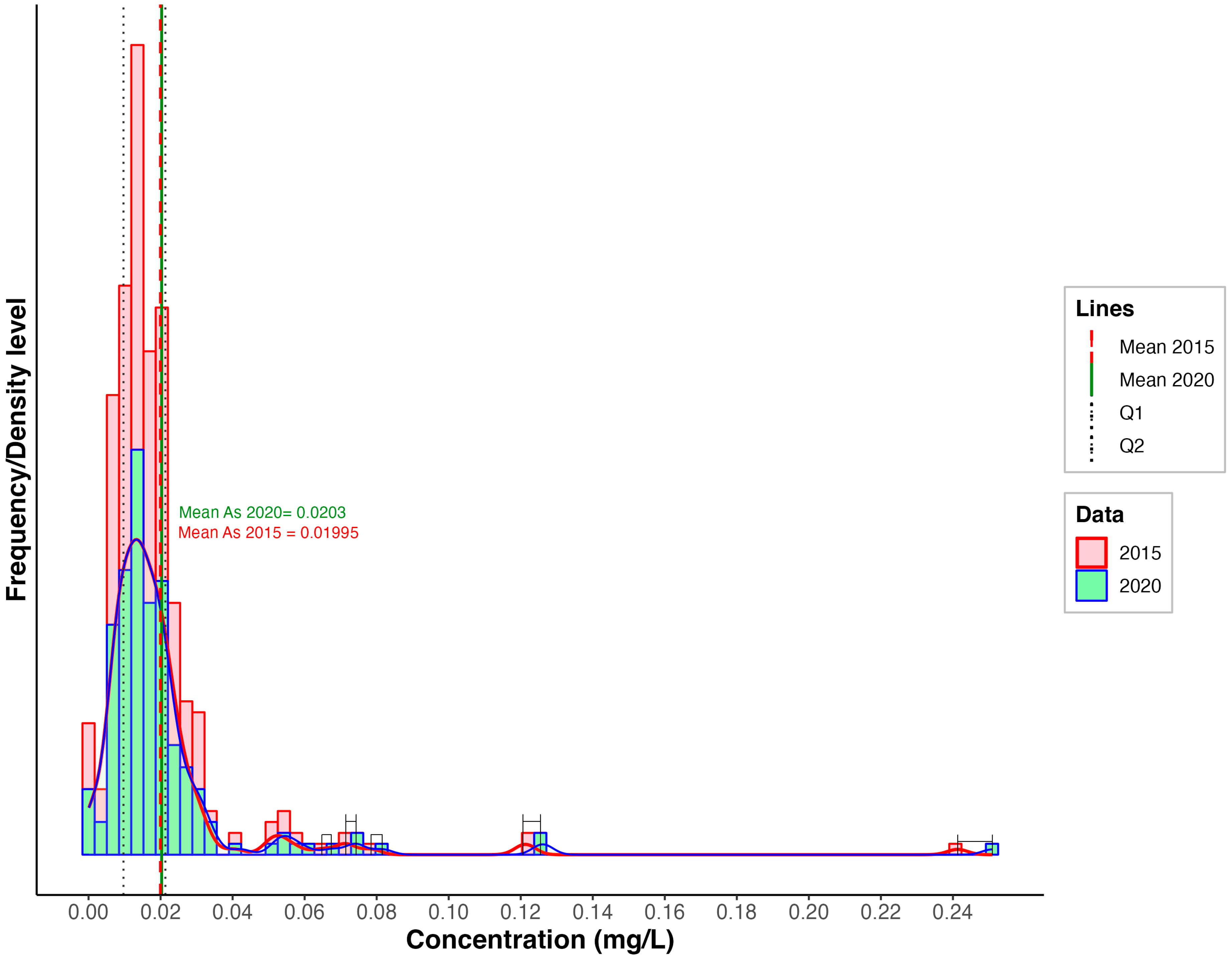
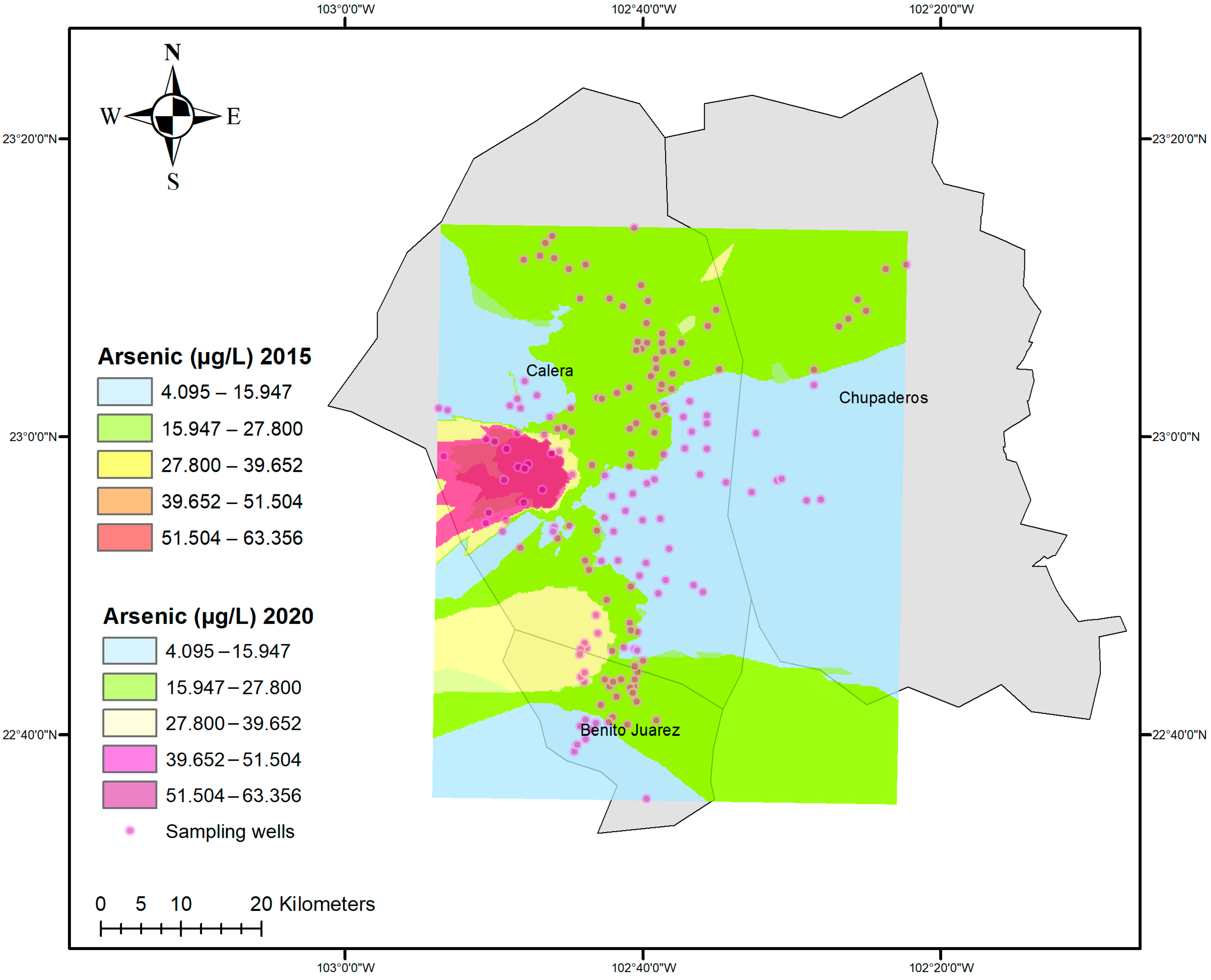
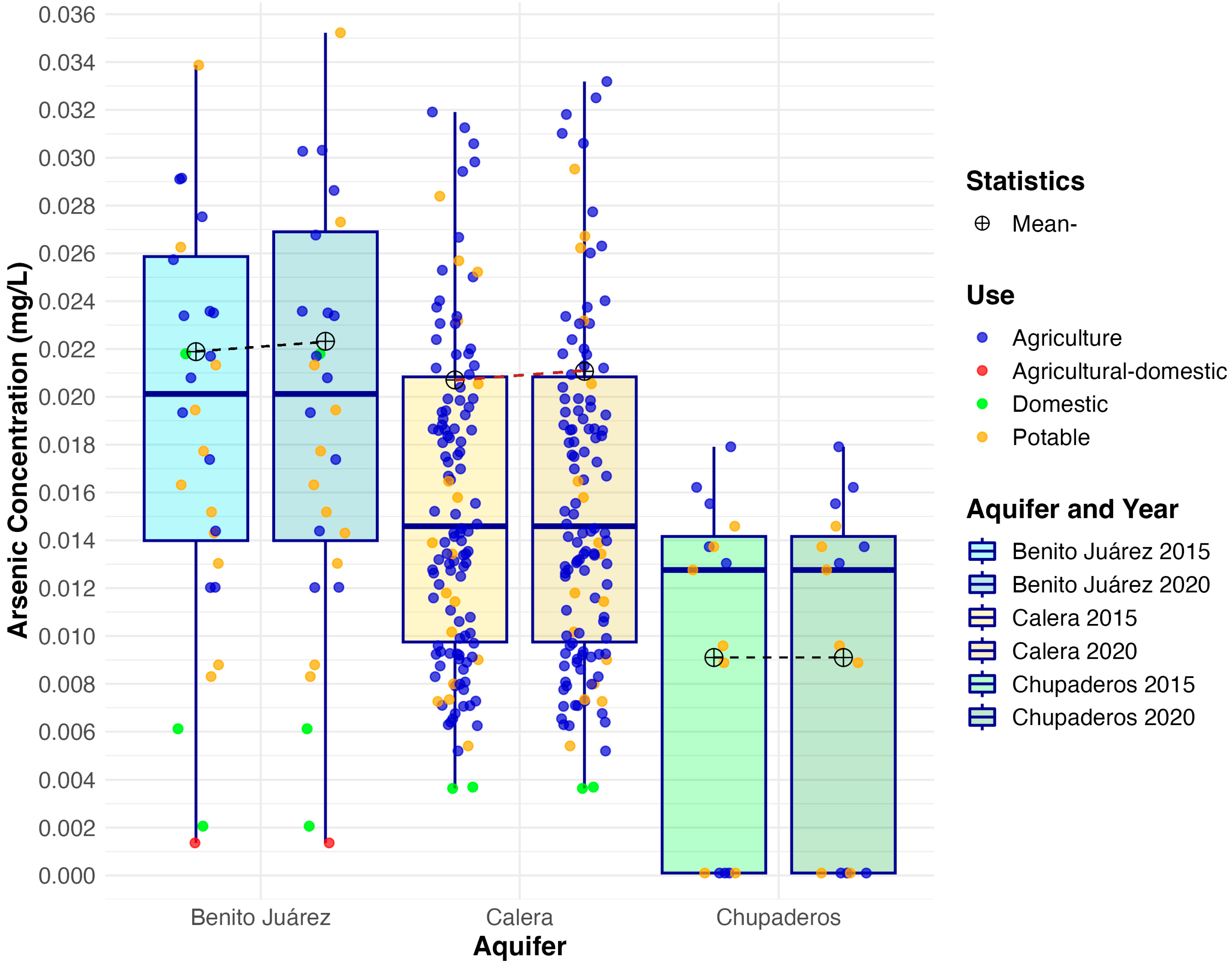
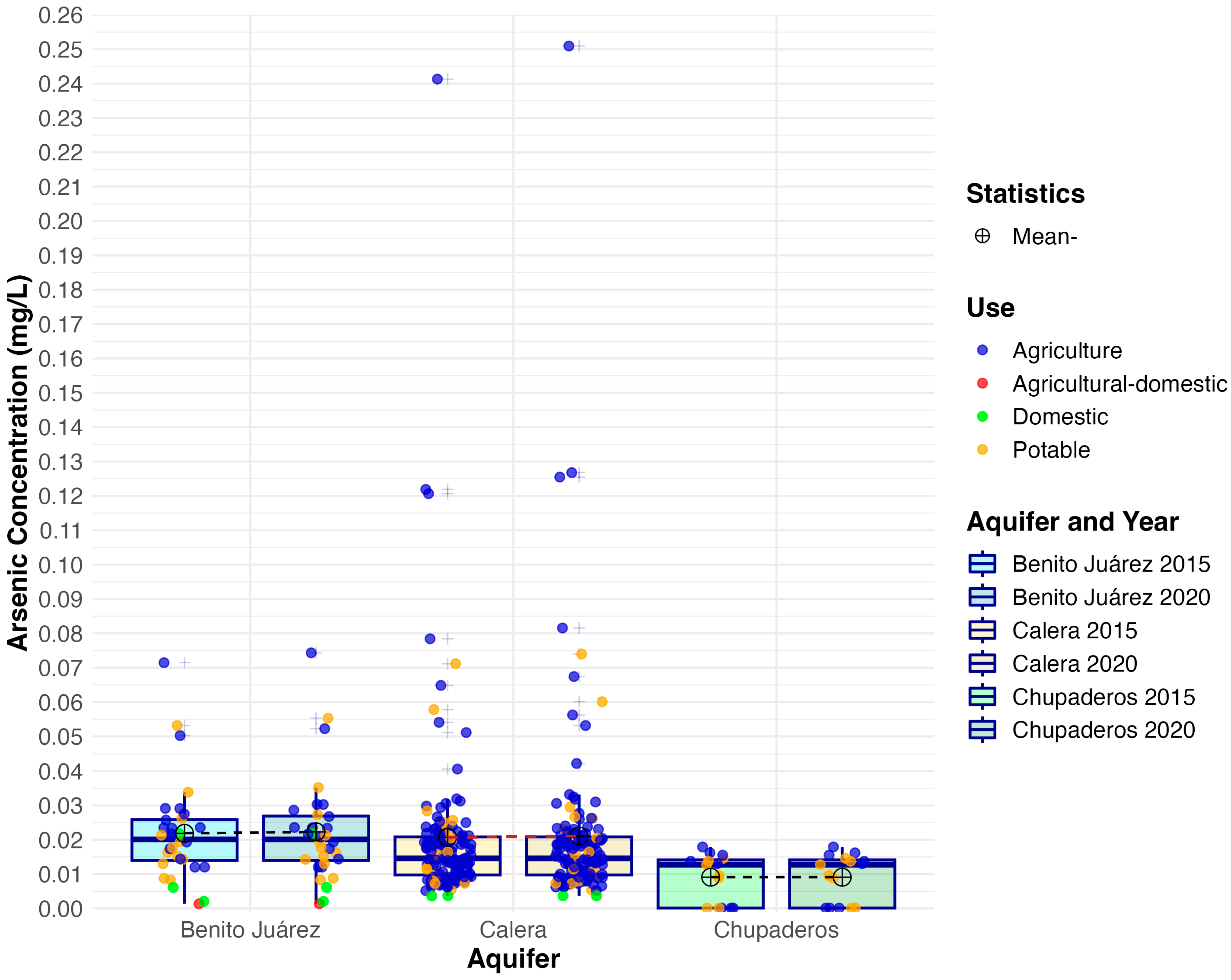
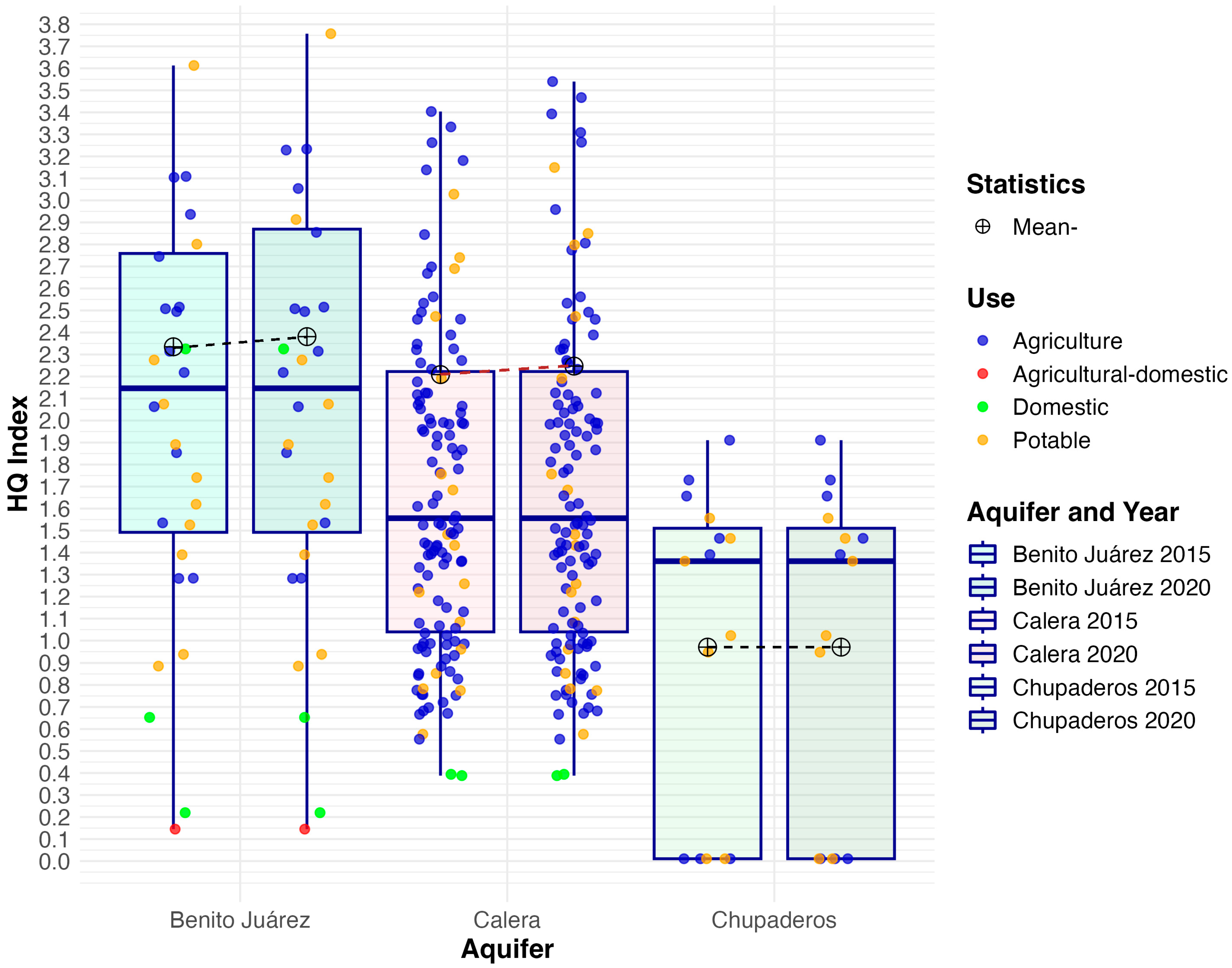
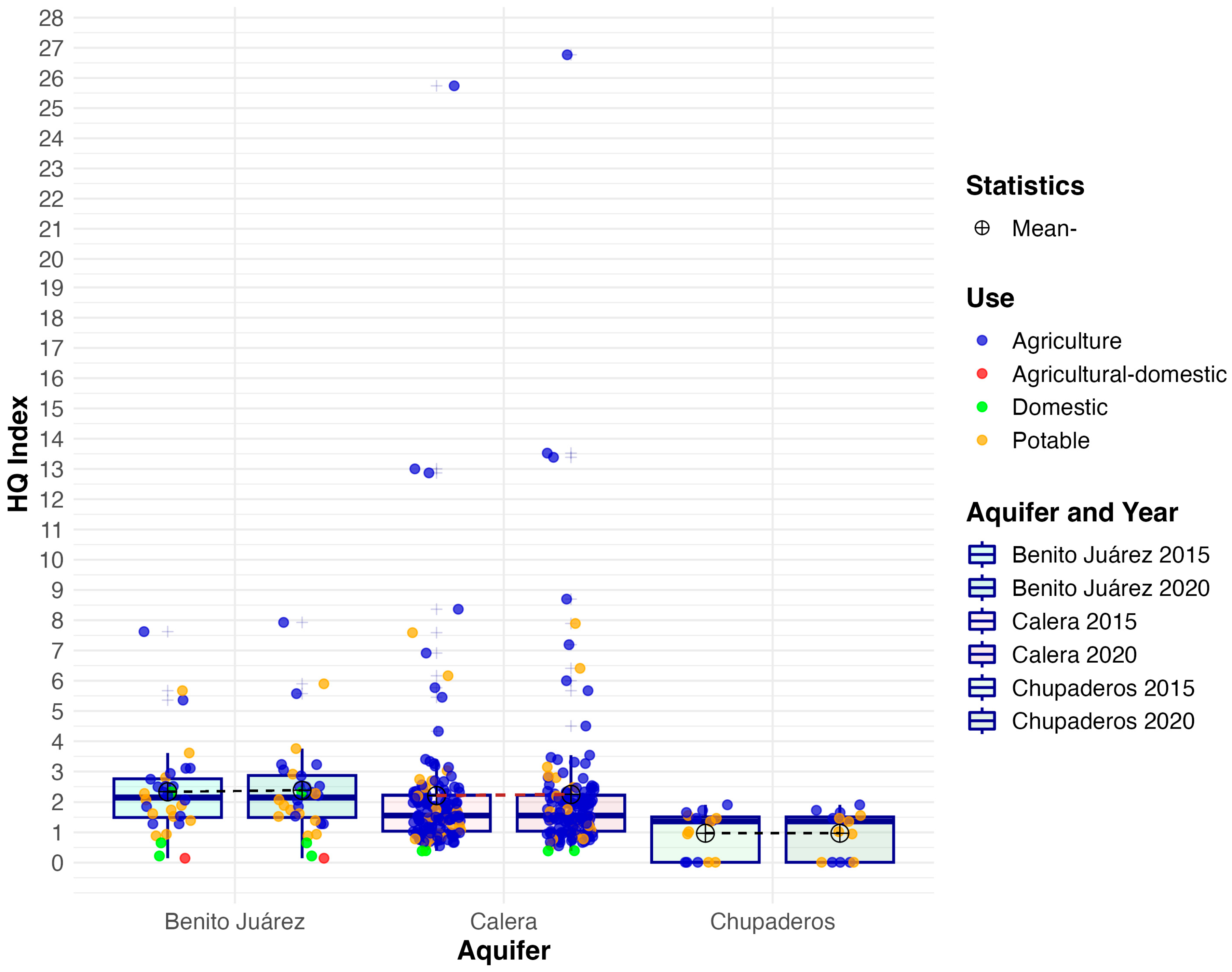
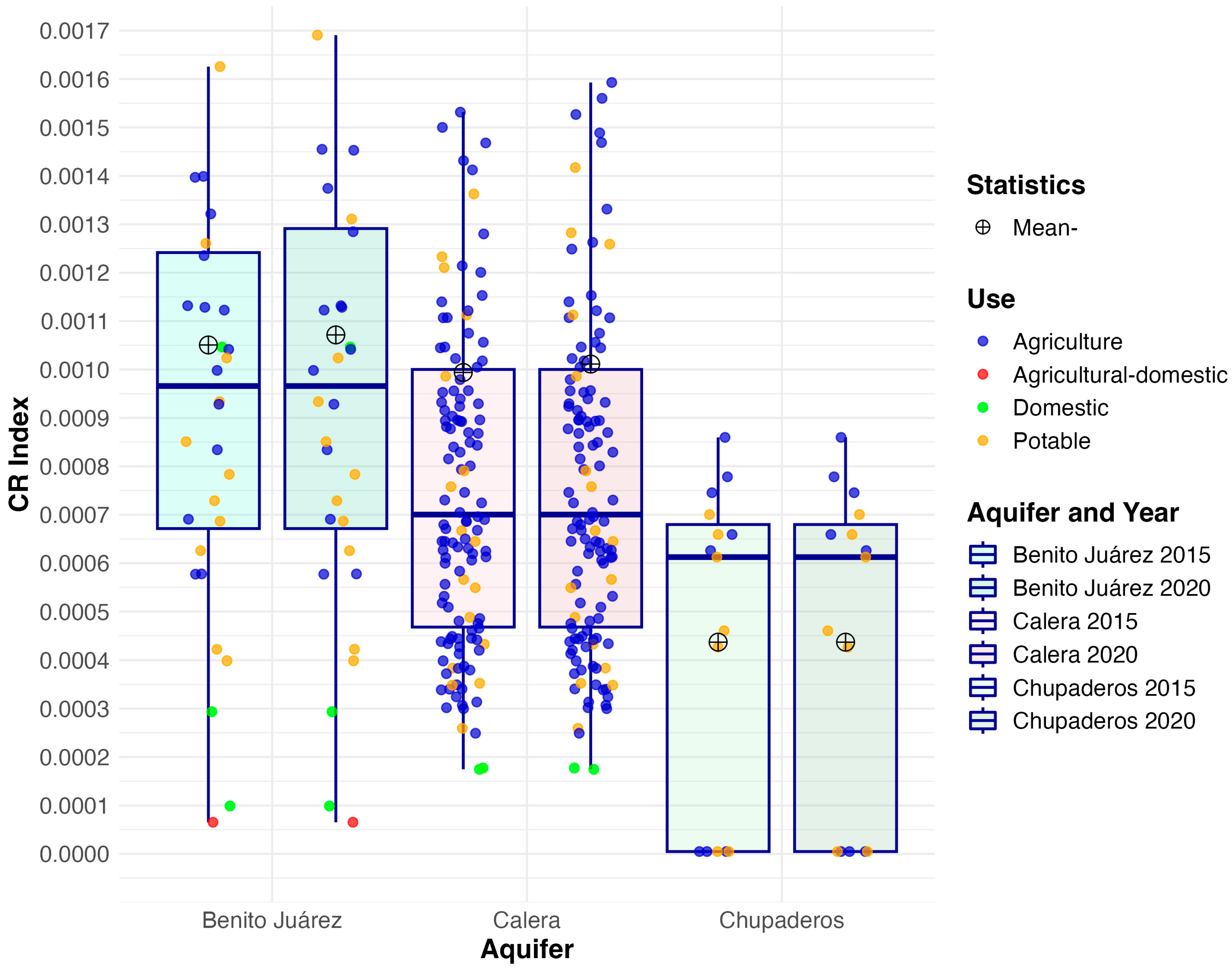
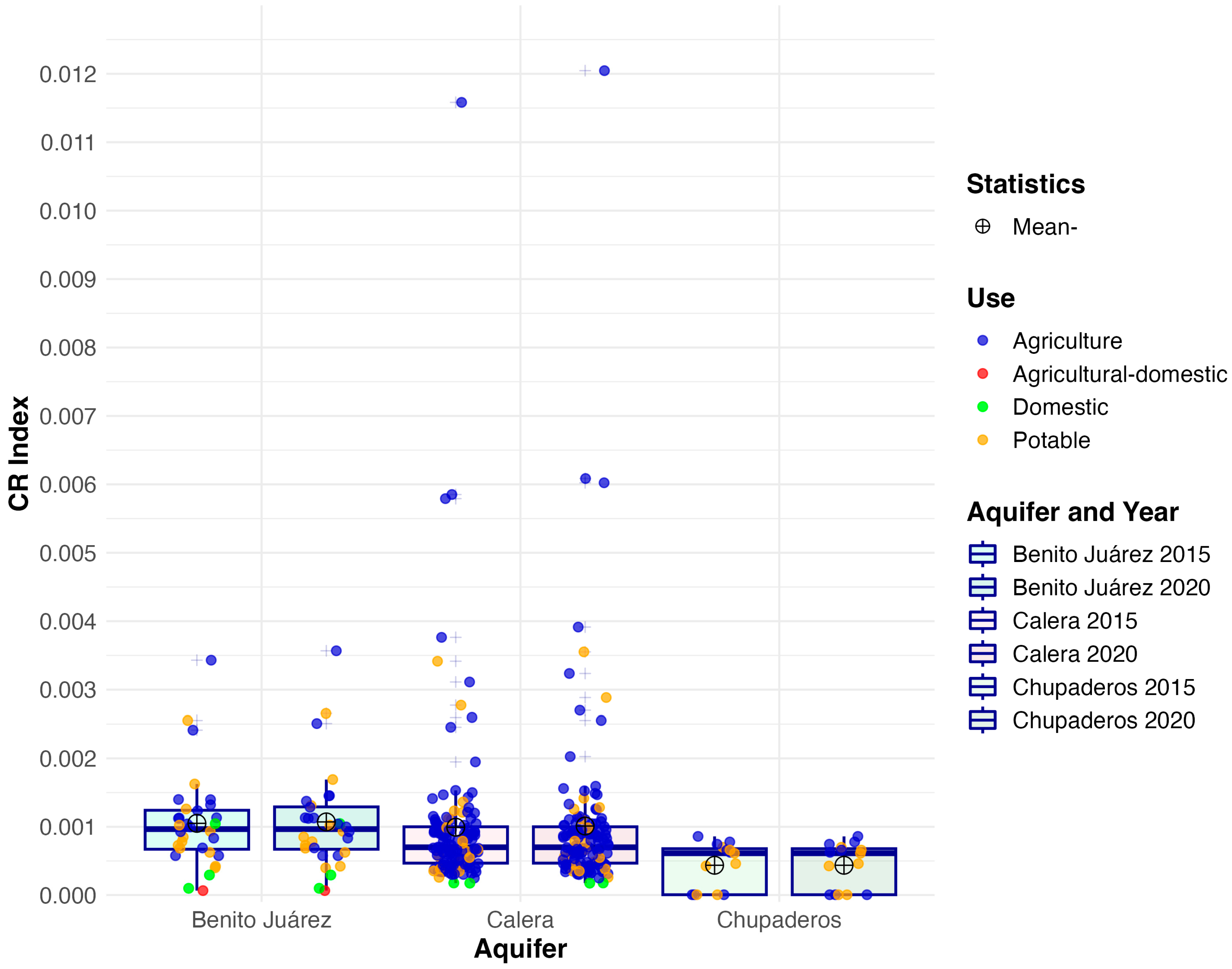
| Aquifer | Mean 2015 | Mean 2020 | Median 2015 | Median 2020 | IQR 2015 | IQR 2020 | SD 2015 | SD 2020 | Skew 2015 | Skew 2020 | Kurt. 2015 | Kurt. 2020 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| B. Juárez | 0.022 | 0.023 | 0.020 | 0.020 | 0.012 | 0.013 | 0.015 | 0.015 | 1.530 | 1.560 | 2.810 | 2.810 |
| Calera | 0.021 | 0.021 | 0.015 | 0.015 | 0.011 | 0.011 | 0.026 | 0.027 | 5.630 | 5.620 | 39.700 | 39.500 |
| Chupaderos | 0.009 | 0.009 | 0.013 | 0.013 | 0.014 | 0.014 | 0.007 | 0.007 | −0.368 | −0.368 | −1.710 | −1.710 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortiz Letechipia, J.; Pinedo Vega, M.E.; González Trinidad, J.; Júnez-Ferreira, H.E.; Veyna Gómez, A.I.; Contreras Rodríguez, A.R.; Robles Rovelo, C.O.; Dávila Hernández, S. Analysis of Human Health Risk Related to the Exposure of Arsenic Concentrations and Temporal Variation in Groundwater of a Semi-Arid Region in Mexico. Water 2025, 17, 2143. https://doi.org/10.3390/w17142143
Ortiz Letechipia J, Pinedo Vega ME, González Trinidad J, Júnez-Ferreira HE, Veyna Gómez AI, Contreras Rodríguez AR, Robles Rovelo CO, Dávila Hernández S. Analysis of Human Health Risk Related to the Exposure of Arsenic Concentrations and Temporal Variation in Groundwater of a Semi-Arid Region in Mexico. Water. 2025; 17(14):2143. https://doi.org/10.3390/w17142143
Chicago/Turabian StyleOrtiz Letechipia, Jennifer, Miguel Eduardo Pinedo Vega, Julián González Trinidad, Hugo Enrique Júnez-Ferreira, Ana Isabel Veyna Gómez, Ada Rebeca Contreras Rodríguez, Cruz Octavio Robles Rovelo, and Sandra Dávila Hernández. 2025. "Analysis of Human Health Risk Related to the Exposure of Arsenic Concentrations and Temporal Variation in Groundwater of a Semi-Arid Region in Mexico" Water 17, no. 14: 2143. https://doi.org/10.3390/w17142143
APA StyleOrtiz Letechipia, J., Pinedo Vega, M. E., González Trinidad, J., Júnez-Ferreira, H. E., Veyna Gómez, A. I., Contreras Rodríguez, A. R., Robles Rovelo, C. O., & Dávila Hernández, S. (2025). Analysis of Human Health Risk Related to the Exposure of Arsenic Concentrations and Temporal Variation in Groundwater of a Semi-Arid Region in Mexico. Water, 17(14), 2143. https://doi.org/10.3390/w17142143










