Abstract
As photoelectrochemical catalyst material, Z-scheme heterojunction 3D WO3@Co2SnO4 composites were designed through a hydrothermal-calcination method. The morphology and structure were characterized by SEM, EDS, XRD, XPS, DRS, and Mott–Schottky analysis, and the photoelectrochemical properties were explored with the transient photocurrent and electrochemical impedance. The construction of Z-scheme heterojunction markedly heightened the separation efficiency of photogenerated electron-hole pairs of WO3 and enhanced the light absorption intensity, retaining the strong redox ability of the photocatalyst. The 3D WO3@Co2SnO4 was used as a photocathode for production of H2O2. Under the optimal reaction conditions, the yield of H2O2 can reach 1335 μmol·L−1·h−1. The results of free radial capture and rotating disc test revealed the existence of direct one-step two-electron and indirect two-step one-electron oxygen reduction to produce H2O2. Based on the excellent H2O2 production performance of the Z-scheme heterojunction photoelectrocatalytic material, 3D WO3@Co2SnO4 and stainless-steel mesh were used to construct a dual-cathode photoelectric-Fenton system for in-situ degradation of a variety of pollutants in water, such as dye (Methyl orange, Rhodamine B), Tetracycline, sulfamethazine, and ciprofloxacin. The fluorescence spectrophotometry was used to detect hydroxyl radicals with terephthalic acid as a probe. Also, the photocatalytic degradation mechanism was revealed, indicating the dual-cathode photoelectron-Fenton system displayed satisfactory potential on degradation of different types of environmental pollutants. This work provided insights for designing high-activity photoelectrocatalytic materials to produce H2O2 and provided possibility for construction of a photoelectric-Fenton system without extra additions.
1. Introduction
Photoelectrocatalytic (PEC), as a new technology using solar energy, was developed not only for the catalytic production of energy, but also for the degradation of water pollutants, solving energy and environmental problems [1,2,3,4,5]. PEC can directly utilize the photogenerated electron-hole pairs in semiconductor catalysts to produce fuel and degrade pollutants, which was usually used for production of H2 and reduction of CO2 [6,7,8]. Furthermore, PEC production of hydrogen peroxide (H2O2) was also a new ideal solar energy conversion fuel technology [9,10,11]. As a kind of green and environmentally friendly chemical substance, H2O2 was widely used in the fields of pulp, chemical raw material synthesis and water purification because of its advantages, such as non-toxicity and high energy density, which was a promising clean fuel [12,13]. Nowadays, PEC production of H2O2 has been a widespread concern, though there are more problems, such as low stability of semiconductor catalysts, poor resistance to photo-corrosion, low photoelectric conversion efficiency, high recombination rate of photogenerated carriers, and so on [14]. Therefore, developing high efficiency, low-cost material for the production of H2O2 was an ideal way to promote clean energy utilization and reduce environmental pollution problems.
Tungsten trioxide (WO3), an n-type metal oxide semiconductor, was considered a benign visible-light response material, with a band gap of approximately 2.8 eV [15], because of its simple preparation method, eco-friendly attributes and stability in acidic and oxidative conditions. WO3 was considered one of the candidate semiconductor materials for PEC oxidation and degradation of organic pollutants due to its excellent absorption, extensive range of visible-light capability and efficient photogenerated electron-holes oxidative performance. Nevertheless, pure WO3 was plagued by some defects, that is, fast recombination of photogenerated hole-electron pairs and poor migration rates of interfacial photogenerated species, which caused unsatisfied reaction kinetics. It was manifested from multiple sources that construction of heterojunction can help WO3 to improve its visible-light adsorption property and accelerate the separation of photogenerated electron-holes, which was the effective action for enhancing the PEC performance [16,17,18,19,20].
Tin was a precious metal, and tin-based oxide was considered one of the most promising alternatives to precious metals, because of its high electrical conductance and optical properties [21,22,23]. The abundant active sites can be provided by the oxygen vacancies in Cobalt tin oxide (Co2SnO4), which was beneficial to increasing the conductance [24]. Meanwhile, Co2SnO4 can give good electrical conductivity and larger surface area when combined with other two-dimensional materials [25]. Herein, Co2SnO4 nanoparticles deposited and grew on the surface of WO3 nanobricks to construct Z-scheme 3D WO3@Co2SnO4 heterogeneous composites via a hydrothermal-calcination method. The PEC performances of WO3@Co2SnO4 heterogeneous for multifunction application were investigated, including production of H2O2 and photocatalytic degradation of organic pollutants in water. The PEC system for producing H2O2 was focused using 3D WO3@Co2SnO4 heterogeneous as a photocathode. Simultaneously, a dual-cathode photoelectron-Fenton system was investigated for PEC degradation of various environmental organic pollutants. The PEC reaction mechanisms were discussed in detail.
2. Materials and Methods
2.1. Synthesis of WO3 Nanobricks
The reagents used in this experiment were of analytical purity and did not undergo any further purification. The hydrothermal-calcination technique was employed for the synthesis of WO3 nanobricks. Initially, 0.1 g of NaWO4·2H2O (Sinopharm Chemical Reagent Co., Ltd., China) was added in deionized water (DI) with the addition of 4 mL HCl (Sinopharm Chemical Reagent Co., Ltd., China). Subsequently, a yellow flocculent precipitate was solubilized upon the introduction of 0.54 g of oxalic acid under continuous stirring. The homogeneous mixture underwent hydrothermal reaction at 180 °C for 8 h. Afterwards, the as-prepared products were centrifuged, washed three times with DI, and dried overnight at 60 °C. Finally, the precursors were calcined in a muffle furnace at 2 °C·min−1 to reach 500 °C for 2 h. Consequently, WO3 nanobricks in the form of light-yellow powder were obtained.
2.2. Synthesis of WO3@Co2SnO4 Composites
The synthesis process of WO3@Co2SnO4 was displayed in Scheme 1. The molar ratio of CoCl2·6H2O (Sinopharm Chemical Reagent Co., Ltd., China) to SnCl4·5H2O (Sinopharm Chemical Reagent Co., Ltd., China) in a 2:1 proportion was dissolved in 60 mL DI under continuous stirring. Subsequently, a blue precipitate was obtained upon the addition of 20 mL NaOH (Sinopharm Chemical Reagent Co., Ltd., China) solution (1 mol·L−1). The previously prepared WO3 nanobricks were introduced into the mixture with stirring for 30 min. The obtained dispersion was poured into a Teflon-lined steel autoclave to react at 180 °C for 10 h. Following centrifugation and triple DI washing, the resulting product, namely WO3@Co2SnO4 composite, was dried overnight at 60 °C and then annealed at heating rate of 2 °C·min−1 to reach 500 °C for a duration of 2 h. The WO3@Co2SnO4 composites with varying content ratios (10%, 20%, 30%, and 50%, which is mass ratio of Co2SnO4 and WO3) were prepared by adjusting the quantities of CoCl2·6H2O and SnCl4·5H2O, denoted as WCSO-1, WCSO-2, WCSO-3, and WCSO-5, respectively. Also, Co2SnO4 loaded WO3 without heterojunctions (WO3+Co2SnO4), that is Co2SnO4 mixed WO3 simply, was used as another control group.
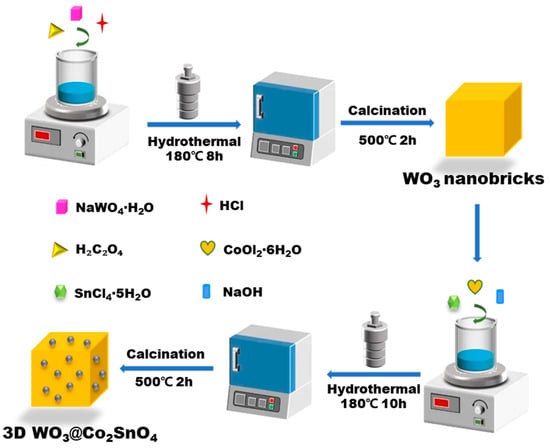
Scheme 1.
Illustration of synthesis strategy for 3D WO3@Co2SnO4 heterojunction.
2.3. Preparation of Photocathode
The carbon paper (2 × 3 cm2) was activated following the methodology described in the literature [26]. Then, 3 mg of WO3@Co2SnO4 composites were ultrasonically dissolved in 0.5 mL DI. The resulting WO3@Co2SnO4 suspension was dropped onto the lower part of carbon paper, covering approximately two-thirds of the carbon paper’s area. After allowing the suspension to dry, the catalyst materials formed a uniform film on the drop-coated region of the carbon paper. The procedure was followed for the preparation of all the photocathode films.
2.4. Characterization
All the catalyst samples were characterized through Scanning electron microscopy (SEM) equipped with X-ray energy-dispersive spectroscopy (EDS) (HITACHI SU-8000, Japan) and X-ray diffraction (XRD) (Bruck D8 Siemens D5000, Germany); PHI 5700 (USA) was used to detect X-ray photoelectron spectroscopy (XPS) of the catalysts, the UV–Vis spectra (Hitachi U3010), and photoluminescence (PL).
2.5. PEC H2O2 Production
PEC H2O2 production was performed in a quartz reactor under 300 W Xenon light illumination (λ ≥ 420 nm). The performance of WO3@Co2SnO4 photocathode was detected in a three-electrode configuration with a Pt counter electrode and Ag/AgCl reference electrode under −0.6 V (vs. Ag/AgCl) bias conditions. A 0.1 mol·L−1 Na2SO4 solution (20 mL, pH = 3 with 0.5 mol·L−1 HClO4). Prior to PEC measurement, the electrolytes were purged with O2 gas under continuous stirring for 30 min, which ensured the adsorption–desorption equilibrium. In the whole reaction process, the concentration of H2O2 was determined using a spectrophotometric method with potassium titanium oxalate.
2.6. PEC Degradation of Contaminations
The performance of the WO3@Co2SnO4 photocathode in terms of PEC was assessed using a double cathode. A stainless-steel mesh (SSM) measuring 2 × 3 m2 was utilized as the second cathode, while a platinum plate served as the anode, and an Ag/AgCl electrode functioned as reference electrode. The PEC degradation of organic pollutants was conducted under the following conditions: 0.1 M Na2SO4 as the supporting electrolyte containing an initial concentration of 10 mg·L−1 organic pollutants, Methylene blue (MB), Rhodamine B (RhB), Tetracycline (TC), Sulfamerazine (SMR), and Ciprofloxacin (CIP); pH value at 3 adjusted by 0.5 mol·L−1 HClO4, the bias voltage as −0.6 V (vs. Ag/AgCl). Prior to PEC measurement, oxygen purging with continuous magnetic stirring for a duration of 30 min ensured adsorption–desorption balance. Subsequently, a 300 W Xenon lamp from Beijing Perfect Light Co., Ltd. (Beijing, China) served as the light source for illumination purposes. At specific time intervals, UV–Vis spectrophotometry monitored changes in concentrations of different organic pollutants.
3. Results
3.1. Characteristic of WO3@Co2SnO4 Composite
The morphology of various materials is characterized in Figure 1a–c. Pure WO3, obtained through the hydrothermal-calcination method, exhibited a smooth-surface stack of nanobricks measuring approximately 300–500 nm in length and width, and a height of about 50–300 nm (Figure 1a). Each nanobrick can serve as a support, providing an optimal contact interface for loading with other semiconductors to construct heterojunctions. Figure 1b displays the morphology of Co2SnO4, which was a was spherical nanoparticle in the diameter range of 20–30 nm. After the combination of WO3 and Co2SnO4, numerous secondary Co2SnO4 nanoparticles can be observed to grow epitaxially on WO3 nanobricks, making the surface of WO3 nanobricks rough (Figure 1c). That indicated that the formation of a large number of heterojunction interfaces was conducive to shortening the ion diffusion and reaction distance. Subsequently, as illustrated in Figure 1d, the existence of W, Co, Sn, and O elements in the materials was ascertained via EDS analysis on the WO3@Co2SnO4 composite, which suggested that WO3 nanobricks were successfully combined with Co2SnO4 nanoparticles.
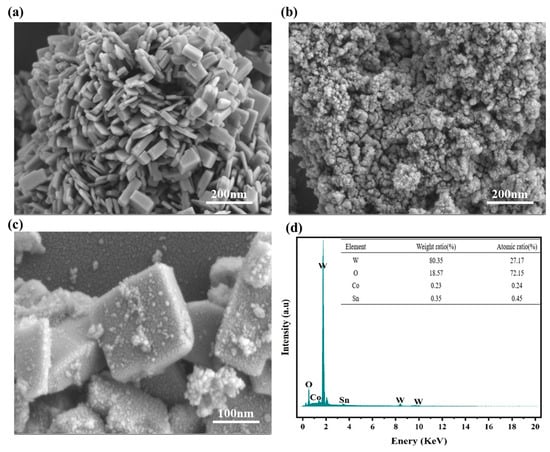
Figure 1.
(a) SEM images of WO3 nanobricks, (b) SEM images of Co2SnO4 nanoparticles, (c) SEM images of WO3@Co2SnO4, (d) EDS spectrum of WO3@Co2SnO4.
To examine the crystallographic composition of the samples, XRD analysis was conducted and presented in Figure 2a. The obtained results indicated a good match between pure WO3 and monoclinic system WO3 corresponding to JCPDS No. 43-1035 [27]. Additionally, besides the diffraction peak of WO3, the XRD pattern of WO3@Co2SnO4 composite also revealed distinct peaks associated with Co2SnO4 at (220), (222), (420), and (533) located at 29.56°, 38.25°, 52.21°, and 59.52°, respectively, according to JCPDS No.29-0514 [28]. These findings provided confirmation for the successful preparation of heterojunction composites consisting of WO3@Co2SnO4.
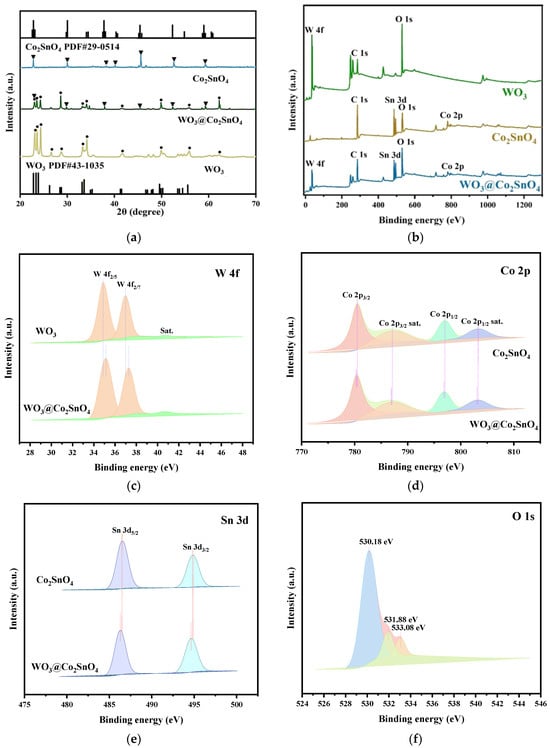
Figure 2.
(a) XRD patterns, (b) comparative XPS survey scan of WO3, Co2SnO4, and WO3@Co2SnO4, (c–f) relative high-resolution XPS spectra of W 4f, Co 2p, Sn 3d, and O1s regions.
XPS analysis was carried out to investigate the chemical constitution and surface states of WO3, Co2SnO4, and WO3@Co2SnO4. Figure 2b illustrates the survey XPS spectra obtained for WO3, Co2SnO4, and WO3@Co2SnO4. In the composite WO3@Co2SnO4, distinct peaks corresponding to W, Sn, O, and Co peaks were observed consistently with the EDS results. Additionally, the reference peak at 284.8 eV in all survey spectra corresponded to C 1s. Figure 2c–f illustrates detailed scans of W 4f, Co 2p, Sn 3d, and O 1s peaks in pure materials and WO3@Co2SnO4 composite in sequence. Two peaks at 34.88 eV and 36.98 eV in Figure 2c correspond well to the W6+ states of W 4f5/2 and W 4f7/2, respectively. The binding energy peaks at 780.28 eV and 796.98 eV in Figure 2d can be ascribed to Co 2p3/2 and Co 2p1/2 states, which was consistent with the results for Co2+ [29]. Furthermore, the satellite peaks located at 787.08 eV and 803.18 eV were associated with Co 2p3/2 and Co 2p1/2 states [26,30]. The peaks at 486.15 eV and 494.48 eV belonged to Sn 3d5/2 and Sn 3d3/2 states, respectively. The O 1s binding energy peaks were detected at three different values: 530.18 eV, 531.88 eV, and 533.08 eV, corresponding to Metal-O (M-O), O-H of H2O, and absorbed O, respectively. Based on the XPS analysis results, it can be observed that Co2SnO4 was incorporated into the heterogeneous structure of WO3. During the heterojunction formation process, there was an increase in electron cloud density around the semiconductor, resulting in a negative shift in XPS spectral peaks while the semiconductor exhibited positive shift in its XPS spectra peaks. In the case of WO3@Co2SnO4 composite, there was a positive shift in binding energy for W 4f, and both the binding energy of Co 2p peaks and that of Sn 3d peaks were shifted negatively. All these XPS results collectively indicated successful construction of heterojunction.
The optical properties of semiconductor materials were crucial factors for the performance of PEC systems. Figure 3a illustrated the UV–Vis absorption spectra of WO3, Co2SnO4, and WO3@Co2SnO4, demonstrating enhanced absorption in the UV–Vis range after combing WO3 with Co2SnO4. Additionally, there was a red shift in the absorption range along with a broad peak spanning from 550 to 650 nm. The incorporation of Co2SnO4 into WO3 was inferred to significantly broaden the absorption region, indicating enhanced light-absorption capabilities. As displayed in Figure 3b, the band gap energy (Eg) for WO3, Co2SnO4, and WO3@Co2SnO4 calculated using the Kubelka–Munk function equation [31] were 2.75 eV, 1.62 eV, and 2.47 eV, respectively. This suggested introduction of Co2SnO4 reduced the band gap of WO3 and noticeably improved its light-absorption ability.
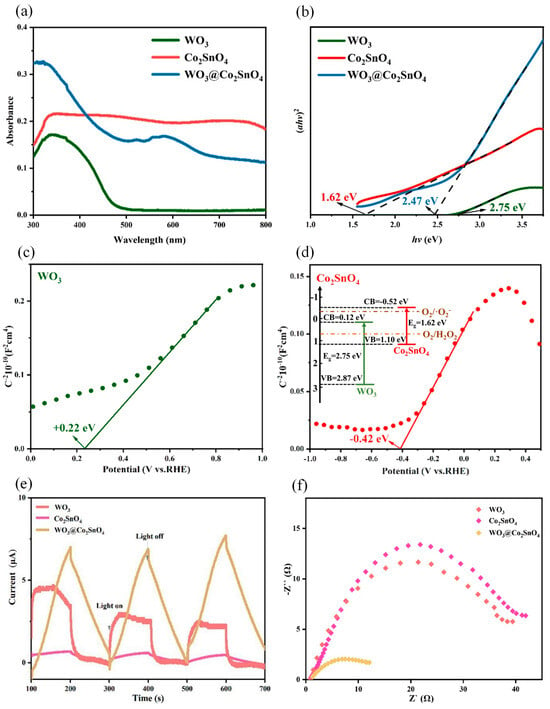
Figure 3.
(a) DRS spectra of WO3, Co2SnO4, and WO3@Co2SnO4, (b) the Kubelka-Munk plots converted from the UV-Vis DRS spectra of WO3, Co2SnO4, and WO3@Co2SnO4, (c) Mott-Schottky plots of WO3, (d) Mott-Schottky plots of Co2SnO4, (inset: the relationship of band gap of WO3 and Co2SnO4), (e) photocurrent responses of WO3, Co2SnO4, and WO3@Co2SnO4, (f) electrochemical impedance spectra (Nyquist plots) of WO3, Co2SnO4, and WO3@Co2SnO4.
The Mott-Schottky plots were conducted to estimate the semiconductor type and the flat-band potential (Efb) values of pure WO3 and Co2SnO4, as depicted in Figure 3c,d. Both WO3 and Co2SnO4 exhibited n-type semiconducting behavior with a positive slope, confirming that WO3 had an Efb of 0.22 eV (vs. RHE) while Co2SnO4 had an Efb of −0.42 eV (vs. RHE). Generally, for n-type semiconductors, the Efb was typically 0.1–0.3 eV higher than the ECB. Thus, the ECB values for WO3 and Co2SnO4 were determined to be 0.12 eV and −0.42 eV, respectively. The inset of Figure 3d illustrated the band gap relationship between WO3 and Co2SnO4 (inset of Figure 3d), revealing a favorable alignment of band positions which facilitates efficient transfer of photogenerated electron-holes within the heterostructure.
In order to inquire about the separation and transfer of photogenerated carriers at the heterojunction interface, we measured the transient photocurrent response of samples under simulated sunlight (Xenon lamp). Generally, a higher photocurrent indicated better separation of photogenerated carriers and a faster migration rate. As depicted in Figure 3e, compared to WO3 and Co2SnO4, the WO3@Co2SnO4 composite displayed the highest photocurrent. This can be put down to the construction of a heterojunction and an internal electric field within the composite, which significantly improved the efficiency of separating photoexcited carrier. The Electrochemical Impedance Spectroscopy (EIS, Nyquist plots) analysis (Figure 3f) demonstrated that the WO3@Co2SnO4 electrode displayed a significantly reduced arc radius in comparison to the WO3 and Co2SnO4 electrodes, indicating its exceptional ability to efficiently separate and transport photogenerated carriers for enhanced photoelectrocatalytic reactions.
3.2. PEC Performances
3.2.1. In-Situ Generation of H2O2 on the WO3@Co2SnO4 Photocathode
The PEC activities of various catalysts for in-situ generation of H2O2 were conducted under oxygen saturation condition with a pH of 3 and visible-light irradiation for 60 min. As depicted in Figure 4a, WCSO-2 exhibited the highest H2O2 production (1335.3 μmol·L−1·h−1) compared to pure WO3 (33.13 μmol·L−1·h−1), pure Co2SnO4 (279.1 μmol·L−1·h−1), WCSO-1 (673.3 μmol·L−1·h−1), WCSO-3 (956.7 μmol·L−1·h−1), WCSO-3 (956.7 μmol·L−1·h−1), WCSO-5 (824.5 μmol·L−1·h−1), and WO3+Co2SnO4(125.6 μmol·L−1·h−1), respectively. The low yield of H2O2 observed for the pure catalysts can be due to the high photogenerated carrier recombination efficiency and inappropriate conduction potential. However, the PEC production capability of H2O2 gradually increased with different loading amounts of Co2SnO4 in the WO3@Co2SnO4 composites. This indicated that the introduction of Co2SnO4 can enhance the PEC properties, while insufficient Co2SnO4 was not conducive to constructing a heterojunction structure, and excessive Co2SnO4 may obstruct active sites on the catalyst. If there was no special illustration, WCSO-2 was selected as the catalyst material for the whole experiment. In Figure 4b, under the optimal PEC H2O2 production conditions on WO3@Co2SnO4 photocathode, that is O2-saturated solution with pH value at 3 with visible-light irradiation, the cumulative concentration of H2O2 was 3120 μmol·L−1 for 240 min, which indicated that the WO3@Co2SnO4 photocathode possessed excellent PEC H2O2 production performance.
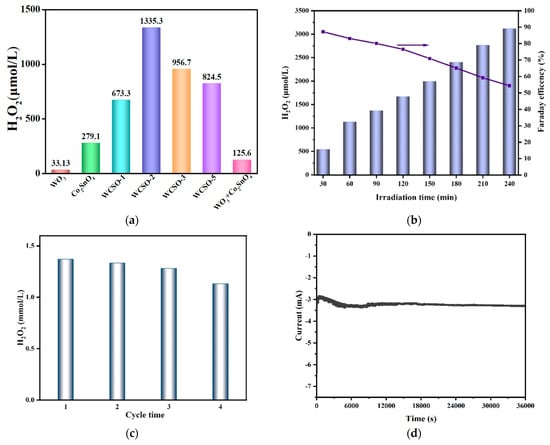
Figure 4.
(a) The H2O2 production with different proportion of the electrode materials, (b) the yield of H2O2 on WO3@Co2SnO4 photocathode and Faraday efficiency over time, (c) recycling stability of WO3@Co2SnO4 photocathode on production of H2O2, (d) long-term photostability under WO3@Co2SnO4 photocathode.
The recycling stability for an electrode was one of the important criteria to evaluate the quality. The recyclability of the as-prepared WO3@Co2SnO4 photocathode was investigated for four cycles in Figure 4c, which exhibited that the yield of H2O2 still can reach more than 1 mmol·L−1 in the four cycles. Figure 4d demonstrated that the photocurrent remained stable for 36,000 s. The results above showed that the WO3@Co2SnO4 photocathode had satisfied stability and can be reused many times.
3.2.2. PEC Degradation of Organic Pollutants on Double Photocathode
A double-cathode that is the WO3@Co2SnO4 photocathode coupled with the SSM photoelectric-Fenton system was carried out to investigate PEC degradation of organic pollutants in water. Based on the excellent H2O2 production performance of the WO3@Co2SnO4 photocathode, the generated H2O2 from WO3@Co2SnO4 photocathode and the electrons (e−) by the SSM were in-situ converted into hydroxyl radial (·OH), that is H2O2 + e− →·OH + OH−, to achieve degradation of common pollutants in water.
Firstly, the effect of different cathode systems, namely the single-cathode system and double-cathode system, was investigated for the degradation of tetracycline (TC). The PEC degradation of TC was conducted in a 20 mL solution containing 10 mg·L−1 TC with saturated oxygen (0.1 mol·L−1 Na2SO4, pH = 3). As shown in Figure 5a, the degradation efficiency of TC in a double-cathode system reached 96% for PEC 60 min and achieved complete degradation after PEC 75 min. In contrast, the single-cathode system only exhibited a degradation efficiency of 47% for PEC 90 min. These confirmed that the double-cathode configuration outperformed the single-cathode configuration in terms of PEC degradation of pollutants. Meanwhile, the constant of apparent reaction rates of the two different systems were calculated and illustrated in Figure 5b, which indicated that the PEC degradation reaction rate was obeyed by the linearly correlated slopes. The double-cathode system displayed the faster reaction rate, and the apparent kinetic rate constant was 0.07565 min−1 which was obviously higher than the single-cathode system (0.01153 min−1). It was illustrated that the introduction of SSM accelerated the PEC degradation reaction, and further verified that the double-cathode system can prompt the ·OH generation to improve the degradation effect of TC.
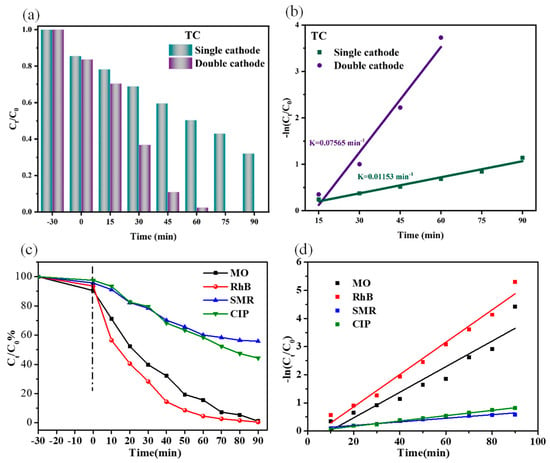
Figure 5.
(a) PEC degradation of TC with single/double cathode, (b) the kinetic curves of PEC degradation of TC with single/double cathode, (c) PEC degradation of different organic pollutants with double cathode, (d) the kinetic curves of PEC degradation.
To further investigate the PEC capability of the double-cathode system, the degradation effect of organic pollutants (MO, RhB, SMR, and CIP) was examined in Figure 5c. After reaching adsorption-desorption equilibrium for 30 min, satisfactory degradation efficiencies were achieved by the double-cathode system. Figure 5d shows the degradation kinetics curves of four organic pollutants, which can be fitted into first-order kinetic model. Figure S1 displays UV-Vis spectra of organic dyes (MO and RhB), SMR, and CIP. It can be seen that the double-cathode system was fit for the degradation of different types of pollutants.
3.3. Deduced PEC Reaction Mechanisms
3.3.1. PEC H2O2 Production Mechanism
To investigate the mechanism of PEC H2O2 production, various sacrificial agents, Potassium Persulfate (K2S2O8, scavenger of e−) [32], Ethyl Alcohol (CH3CH2OH, h+ scavenger) [33], 2,2,6,6-Tetramethylpiperidoxyl (TEMPO, scavenger of ∙O2−) [34], and Isopropanol (IPA, ∙OH scavenger) [35] were added into the reaction system. It can be found in Figure 6a that yield of H2O2 decreased obviously upon the introduction of K2S2O8 and TEMPO, indicating that e− and ∙O2− played important roles during the process of PEC H2O2 production. When IPA was added, the yield of H2O2 only reduced 5.4%, suggesting ∙OH gave little inhibited effect on H2O2 production. The addition of CH3CH2OH slightly improved the yield of H2O2 because the recombination of photogenerated carriers was lowed after trapping h+. It can be found that the yield of H2O2 still remained at 294.5 mmol·L−1 after trapping ∙O2− species, which showed that indirect ORR and direct ORR existed simultaneously, and indirect ORR was dominant in the PEC H2O2.
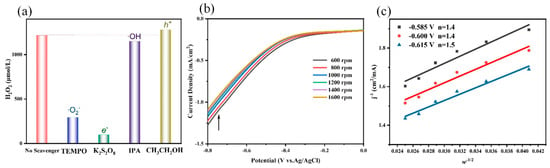
Figure 6.
(a) The effect of various scavengers on PEC H2O2 production, (b) LSVs at different scan rates on RRED, (c) the Koutecky-Levich plots by RRED measurements at different potentials in a buffered pH 3 of 0.1 M Na2SO4.
Furthermore, a rotating ring disk electrode (RRDE) was employed to record and calculate the number of e− transfers (n) in the PEC H2O2 production process. The linear sweep voltammetry (LSVs) at different scan rates on RRED was measured in an O2—saturated buffered of 0.1 mol·L−1 Na2SO4 (pH of 3) with the scan rates range of 600–1600 rpm (Figure 6b). As displayed in Figure 6c, based on the Koutecky-Levich (KL) equation (1/j = 1/jk + kw − 1/2) [36,37], the number of e− transfers (n) at the voltages of −0.585 V, −0.600 V, and −0.615 V was 1.4, 1.4, and 1.5, respectively, indicating both one-electron and two-electron transfer existed during the process of PEC H2O2 production. Namely, the direct one-step two-electron ORR coexisted with the indirect two-step one-electron ORR process. These results were consistent with those of free radical trapping experiments.
3.3.2. PEC Degradation of Organic Pollutants Mechanism
The fluorescent spectrometry with terephthalic acid (TA, 5 × 10−4 mol·L−1) as a probe was used to detect the existence of ∙OH with λex = 315 nm and λem = 340–600 nm. Figure 7a shows that the fluorescent intensity of TA increased continuously at 425 nm along with the time, indicating that the content of ∙OH was increasing. This confirmed the ∙OH in double-cathode system came from the transformation of H2O2 in the PEC process and as the main active substance. In order to further explore the primary active species in the double-cathode system, the trapping examinations were performed in Figure 7b. After capture of ∙OH by IPA, the degradation efficiency of TC decreased obviously, indicating that ∙OH was the dominating role during the PEC degradation of pollutants process. Meanwhile, the degradation efficiency of TC declined 22.6% after trapping h+ by CH3CH2OH, which demonstrated that h+ plays a subsidiary role.
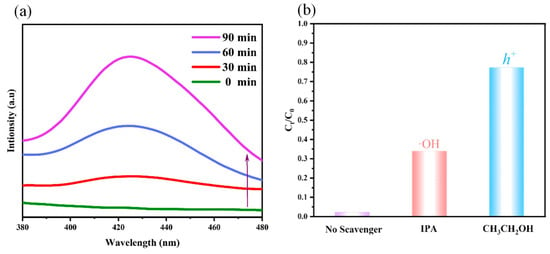
Figure 7.
(a) The fluorescence intensity of TA at different times, (b) trapping experiment of different actives species.
Combined with the band gap of WO3 and Co2SnO4 (inset of Figure 3b), it can be found that the ECB of WO3 and Co2SnO4 was +0.12 eV and −0.52 eV; the EVB of WO3 and Co2SnO4 was 2.87 eV and 1.10 eV. Under visible-light irradiation, both WO3 and Co2SnO4 can be irradiated to generate electron-hole (e−-h+) pairs. WO3+Co2SnO4 without heterojunctions cannot provide efficient way to transfer the photogenerated carriers and separate the photogenerated electron-hole pairs, which will hinder the PEC reaction. In addition, if WO3 and Co2SnO4 formed a conventional WO3-Co2SnO4 heterostructure, the generated e− from the CB of Co2SnO4 will transfer to the CB of WO3; meanwhile, the h+ on the VB of WO3 can migrate to the VB of Co2SnO4. However, the potential of O2/O2− was −0.33 eV, and the ECB of WO3 was insufficient to reduce O2 to form ∙O2−. Through the trapping experiment results, ∙O2− was one of the main active radicals. Based on the above results, the h+ on the VB of WO3 and the photogenerated e− on the CB of Co2SnO4 participated directly in the PEC degradation process, and also retained high redox properties. These testified WO3@Co2SnO4 composites executed a new type of Z-scheme mode for PEC degradation of organic pollutants. As demonstrated in Scheme 2, the photogenerated e− on the CB of WO3 were combined directly with the h+ on the VB of Co2SnO4. Then the h+ on the VB of WO3 and the generated e− on the CB of Co2SnO4 can participate in the PEC process directly, that is, WO3@Co2SnO4 composite followed the principal of Z-scheme carriers conduction mode. Based on the above analysis results, the deduced reaction equations are summarized below:
WO3 + hv → h+(VB) + e−(CB)
Co2SnO4 + hv → e−(CB) + h+(VB)
O2 + 2H+ + 2e− → H2O2
O2 + e− → ∙O2−;
O2− + 2H+ + e− → H2O2
The degradation reaction equation was as follows:
H2O2 + e− → ∙OH + OH
OH + Organic pollutants → CO2 + H2O
h+(VB of WO3) + Organic pollutants → CO2 + H2O
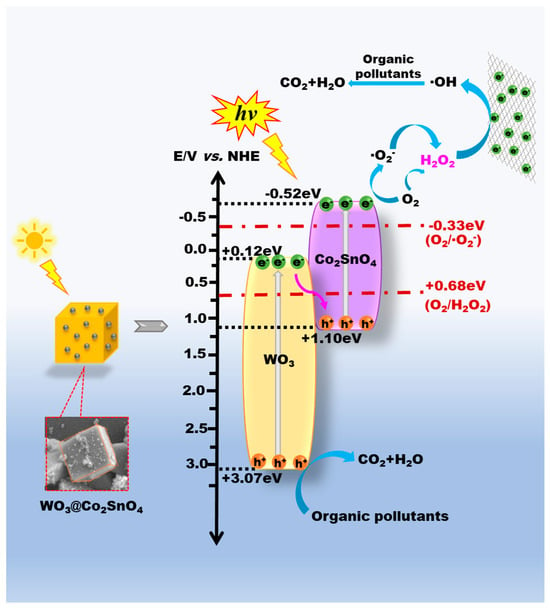
Scheme 2.
The deduced PEC mechanism for Z-scheme 3D WO3@Co2SnO4 heterojunction composite under two-cathode photoelectron-Fenton system.
4. Conclusions
- (1)
- Z-scheme 3D WO3@Co2SnO4 heterojunction composites were prepared through a hydrothermal and then calcining technique. By employing the characterization methods of DRS, Mott-Schottky plots, Transient photocurrent, and electrochemical impedance spectroscopy, the bandgap structure of WO3@Co2SnO4 and its separation capability of photogenerated carriers were detected.
- (2)
- As a photocathode, 3D WO3@Co2SnO4 was one of the satisfied materials for production of H2O2. Under the optimal reaction conditions, the yield of H2O2 reached 1335 μmol·L−1·h−1. The reaction path of H2O2 was investigated by capturing experiments and the rotating disk electrode test, which was mainly an indirect two-step one-electron ORR process.
- (3)
- As the other cathode was designed to conduct a two-cathode photoelectron-Fenton system, 3D WO3@Co2SnO4 as photocathode and stainless-steel mess was carried out in-situ PEC degradation of different organic environmental pollutants. The PEC degradation mechanism was explored, finding that it obeyed a new type of Z-scheme mode through fluorescence spectrophotometry and trapping agents experiments.
- (4)
- This work provided a new idea for preparation of multifunction materials, gave an economic and environmentally friendly way for photoelectrocatalysis production of H2O2, and established a photoelectron-Fenton system without additional additives.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/w16030406/s1, Figure S1: Change in UV–Vis spectra of different organic pollutants.
Author Contributions
L.Z. and D.Z. conceived and designed the study. M.W. and C.A. performed the experiments. D.Z. wrote the paper. L.Z. and D.Z. reviewed and edited the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by National Nature Science Foundation of China (NSFC52072164), Liaoning Provincial Natural Science Foundation of China (2022-MS-366), Pharmaceutical cleaner production and industrialization innovation team of Liaoning Institute of Science and Technology (XKT202304).
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The data are not publicly available due to a confidentiality agreement signed at the beginning of the study.
Acknowledgments
We thank everyone who helped us in the process of writing the manuscript, and we also thank the reviewers for their useful comments and suggestions. The authors also thank their colleagues who participated in this work.
Conflicts of Interest
The authors declare that they have no competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- Yao, T.; An, X.; Han, H.; Chen, J.Q.; Li, C. Photoelectrocatalytic Materials for Solar Water Splitting. Adv. Energy Mater. 2018, 8, 1800210. [Google Scholar] [CrossRef]
- Huang, H.; Steiniger, K.A.; Lambert, T.H. Electrophotocatalysis: Combining Light and Electricity to Catalyze Reactions. J. Am. Chem. Soc. 2022, 144, 12567–12583. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, S.; Wang, W.; Zhang, H. Photoelectrocatalytic Principles for Meaningfully Studying Photocatalyst Properties and Photocatalysis Processes: From Fundamental Theory to Environmental Applications. J. Energy Chem. 2023, 86, 84–117. [Google Scholar] [CrossRef]
- Daghrir, R.; Drogui, P.; Robert, D. Photoelectrocatalytic Technologies for Environmental Applications. J. Photochem. Photobiol. A 2012, 238, 41–52. [Google Scholar] [CrossRef]
- Yang, C.; Wu, H.; Cai, M.; Zhou, Y.; Guo, C.; Han, Y.; Zhang, L. Valorization of Biomass-Derived Polymers to Functional Biochar Materials for Supercapacitor Applications via Pyrolysis: Advances and Perspectives. Polymers 2023, 15, 2741. [Google Scholar] [CrossRef] [PubMed]
- Hansora, D.; Yoo, J.W.; Mehrotra, R.; Byun, W.J.; Lim, D.; Kim, Y.K.; Noh, E.; Lim, H.; Jang, J.-W.; Seok, S.I.; et al. All-perovskite-based unassisted photoelectrochemical water splitting system for efficient, stable and scalable solar hydrogen production. Nat. Energy 2024. [Google Scholar] [CrossRef]
- Kumaravel, V.; Bartlett, J.; Pillai, S.C. Photoelectrochemical Conversion of Carbon Dioxide (CO2) into Fuels and Value-Added Products. ACS Energy Lett. 2020, 5, 486–519. [Google Scholar] [CrossRef]
- Muzzillo, C.P.; Klein, W.E.; Li, Z.; DeAngelis, A.D.; Horsley, K.; Zhu, K.; Gaillard, N. Low-Cost, Efficient, and Durable H2 Production by Photoelectrochemical Water Splitting with CuGa3Se5 Photocathodes. ACS Appl. Mater. Inter. 2018, 10, 19573–19579. [Google Scholar] [CrossRef]
- Lu, H.; Li, X.; Monny, S.A.; Wang, Z.; Wang, L. Photoelectrocatalytic Hydrogen Peroxide Production Based on Transition-Metal-Oxide Semiconductors. Chin. J. Catal. 2022, 43, 1204–1215. [Google Scholar] [CrossRef]
- Baran, T.; Wojtyła, S.; Vertova, A.; Minguzzi, A.; Rondinini, S. Photoelectrochemical and Photocatalytic Systems Based on Titanates for Hydrogen Peroxide Formation. Electroanal. Chem. 2018, 808, 395–402. [Google Scholar] [CrossRef]
- Xue, Y.; Wang, Y.; Pan, Z.; Sayama, K. Electrochemical and Photoelectrochemical Water Oxidation for Hydrogen Peroxide Production. Angew. Chem. Int. Edit. 2021, 60, 10469–10480. [Google Scholar] [CrossRef]
- Chao, C.; Yasugi, M.; Yu, L.; Teng, Z.; Ohno, T. Visible Light-Driven H2O2 Synthesis by a Cu3BiS3 Photocathode Via a Photoelectrochemical Indirect Two-Electron Oxygen Reduction Reaction. Appl. Catal. B-Environ. 2022, 307, 121152. [Google Scholar]
- Zhiyuan, P.; Su, Y.; Siaj, M. Encapsulation of Tin Oxide Layers on Gold Nanoparticles Decorated One-Dimensional CdS Nanoarrays for Pure Z-Scheme Photoanodes Towards Solar Hydrogen Evolution. Appl. Catal. B-Environ. 2023, 330, 122614. [Google Scholar]
- Sotirios, M.; Göltz, M.; Perry, S.C.; Bogdan, F.; Leung, P.K.; Rosiwal, S.; Wang, L.; de León, C.P. Effective Hydrogen Peroxide Production from Electrochemical Water Oxidation. ACS Energy Lett. 2021, 6, 2369. [Google Scholar]
- Sivula, K.; van de Krol, R. Semiconducting Materials for Photoelectrochemical Energy Conversion. Nat. Rev. Mater. 2016, 1, 15010. [Google Scholar] [CrossRef]
- Shan, Y.; Xiujuan, T.; Dongsheng, S.; Zhiruo, Z.; Meizhen, W. A Review of Tungsten Trioxide (WO3)-Based Materials for Antibiotics Removal Via Photocatalysis. Ecotoxicol. Environ. Saf. 2023, 259, 114988. [Google Scholar]
- Qingyi, Z.; Li, J.; Li, L.; Bai, J.; Xia, L.; Zhou, B. Synthesis of WO3/BiVO4 Photoanode Using a Reaction of Bismuth Nitrate with Peroxovanadate on Wo3 Film for Efficient Photoelectrocatalytic Water Splitting and Organic Pollutant Degradation. Appl. Catal. B-Environ. 2017, 217, 21–29. [Google Scholar]
- Asma, M.A.; Taha, T.A.M.; Abdullah, M.; Abid, A.G.; Manzoor, S.; Khosa, R.Y.; Farid, H.M.T.; Trukhanov, S.; Sayyed, M.I.; Tishkevich, D.; et al. Highly Performed Tungsten Trioxide-Polyaniline Composite Thin Film and Their Accelerated Oxygen Evolution Electrocatalyst Activity. Electroanal. Chem. 2023, 941, 117550. [Google Scholar]
- Razali, M.; Aqilah, N.; Salleh, W.N.W.; Aziz, F.; Jye, L.W.; Yusof, N.; Ismail, A.F. Review on Tungsten Trioxide as a Photocatalysts for Degradation of Recalcitrant Pollutants. J. Clean. Prod. 2021, 309, 127438. [Google Scholar] [CrossRef]
- Sheng, W.; Shi, X.; Shao, G.; Duan, X.; Yang, H.; Wang, T. Preparation, Characterization and Photocatalytic Activity of Multi-Walled Carbon Nanotube-Supported Tungsten Trioxide Composites. J. Phys. Chem. Solids 2008, 69, 2396–2400. [Google Scholar]
- Ştefan, N.; Neaţu, F.; Diculescu, V.C.; Trandafir, M.M.; Petrea, N.; Somacescu, S.; Krumeich, F.; Wennmacher, J.T.C.; Knorpp, A.J.; van Bokhoven, J.A.; et al. Undoped SnO2 as a Support for Ni Species to Boost Oxygen Generation through Alkaline Water Electrolysis. ACS Appl. Mater. Interfaces 2020, 12, 18407–18420. [Google Scholar]
- Shunyan, Z.; Li, C.; Liu, J.; Liu, N.; Qiao, S.; Han, Y.; Huang, H.; Liu, Y.; Kang, Z. Carbon Quantum Dots/SnO2–Co3O4 Composite for Highly Efficient Electrochemical Water Oxidation. Carbon 2015, 92, 64–73. [Google Scholar]
- Raj, G.S.; Bhuvaneshwari, S.; Wu, J.J.; Asiri, A.M.; Anandan, S. Sonochemical Synthesis of Co2SnO4 Nanocubes for Supercapacitor Applications. Ultrason. Sonochem. 2018, 41, 435. [Google Scholar]
- Hwa, K.Y.; Santhan, A.; Sharma, T.S.K. One-Dimensional Self-Assembled Co2SnO4 Nanosphere to Nanocubes Intertwined in Two-Dimensional Reduced Graphene Oxide: An Intriguing Electrocatalytic Sensor Toward mesalamine Detection. Mater. Today Chem. 2022, 23, 100739. [Google Scholar] [CrossRef]
- Chang, C.; Ru, Q.; Hu, S.; An, B.; Song, X.; Hou, X. Co2SnO4 Nanocrystals Anchored on Graphene Sheets as High-Performance Electrodes for Lithium-Ion Batteries. Electrochim. Acta 2015, 151, 203. [Google Scholar]
- Lei, Z.; Hu, Y.; Zheng, J. Fabrication of 3D Hierarchical CoSnO3@CoO Pine Needle-Like Array Photoelectrode for Enhanced Photoelectrochemical Properties. J. Mater. Chem. A 2017, 5, 18664–18673. [Google Scholar]
- Cheng, Z.; Zheng, X.; Ning, Y.; Li, Z.; Wu, Z.; Feng, X.; Li, G.; Huang, Z.; Hu, Z. Enhancing Long-Term Stability of Bio-Photoelectrochemical Cell by Defect Engineering of a WO3- Photoanode. J. Energy Chem. 2023, 80, 584. [Google Scholar]
- Karuppaiya, P.; Raman, N. Electrochemical Detection of 2-Nitroaniline at a Novel Sphere-Like Co2SnO4 Modified Glassy Carbon Electrode. New J. Chem. 2020, 44, 8454. [Google Scholar]
- Cao, G.; Li, X.; Yu, H.; Mao, L.; Wong, L.H.; Yan, Q.; Wang, J. A Novel Hollowed CoO-in-CoSnO3 Nanostructure with Enhanced Lithium Storage Capabilities. Nanoscale 2014, 6, 13824. [Google Scholar]
- Yile, W.; Yu, D.; Wang, W.; Gao, P.; Zhang, L.; Zhong, S.; Liu, B. The Controllable Synthesis of Novel Heterojunction CoO/BiVO4 Composite Catalysts for Enhancing Visible-Light Photocatalytic Property. Colloid. Surfaces 2019, 578, 123608. [Google Scholar]
- Danfeng, Z.; Zhang, G.; Wang, Q.; Zhang, L. Dual-Functional Catalytic Materials: Magnetically Hollow Prous Ni-Manganese Oxides Microspheres/Cotton Cellulose Fiber. J. Taiwan Inst. Chem. E 2017, 77, 311. [Google Scholar]
- Kadarkarai, G.; Chandran, H.T.; Mohan, R.; Maheswari, S.U.; Murali, R. Electron Scavenger-Assisted Photocatalytic Degradation of Amido Black 10B Dye with Mn3O4 Nanotubes: A Response Surface Methodology Study with Central Composite Design. J. Photochem. Photobiol. A Chem. 2017, 341, 146. [Google Scholar]
- Doan, N.T.T.; Nguyen, D.; Vo, P.P.; Doan, H.N.; Pham, H.T.N.; Hoang, V.H.; Le, K.T.; Kinashi, K.; Huynh, V.T.; Nguyen, P.T. The Roles of Ethanol and Isopropanol as Hole Scavengers in the Photoreduction Reaction of Graphene Oxide by TiO2: A Competition of Oxygenated Groups Removal and Carbon Defects Invasion. J. Mol. Liq. 2023, 381, 121831. [Google Scholar]
- Mateusz, M.; Szlachcikowska, D.; Stępień, K.; Kielar, P.; Galiniak, S. Two Faces of Tempo (2,2,6,6-Tetramethylpiperidinyl-1-Oxyl)—An Antioxidant or a Toxin? BBA—Mol. Cell Res. 2023, 1870, 119412. [Google Scholar]
- Mengyu, L.; Han, R.; Si, C.; Han, X.; Lu, Q.; Pang, Y. NiO/Bi2MoxW1−XO6 (0 ≤ X ≤ 1) Nanofibers as a Bifunctional Platform Boosting Photocatalytic Tetracycline Conversion and Electrocatalytic Oxygen Reduction. J. Alloys Compd. 2023, 947, 169607. [Google Scholar]
- Shicheng, X.; Kim, Y.; Higgins, D.; Yusuf, M.; Jaramillo, T.F.; Prinz, F.B. Building Upon the Koutecky-Levich Equation for Evaluation of Next-Generation Oxygen Reduction Reaction Catalysts. Electrochim. Acta 2017, 255, 99. [Google Scholar]
- Yu, D.; Yu, Z.-Y.; Yang, L.; Zheng, L.-R.; Zhang, C.-T.; Yang, X.-T.; Gao, F.-Y.; Zhang, X.-L.; Yu, X.; Liu, R.; et al. Bimetallic Nickel-Molybdenum/Tungsten Nanoalloys for High-Efficiency Hydrogen Oxidation Catalysis in Alkaline Electrolytes. Nat. Commun. 2020, 11, 4789. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).