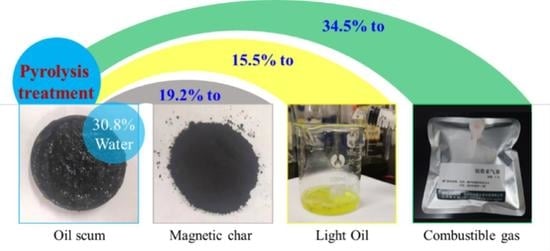
Catalytic Performance of Fe-Rich Sludge in Pyrolysis of Waste Oil Scum as Volatiles and Magnetic Char
Abstract
1. Introduction
2. Materials and Methods
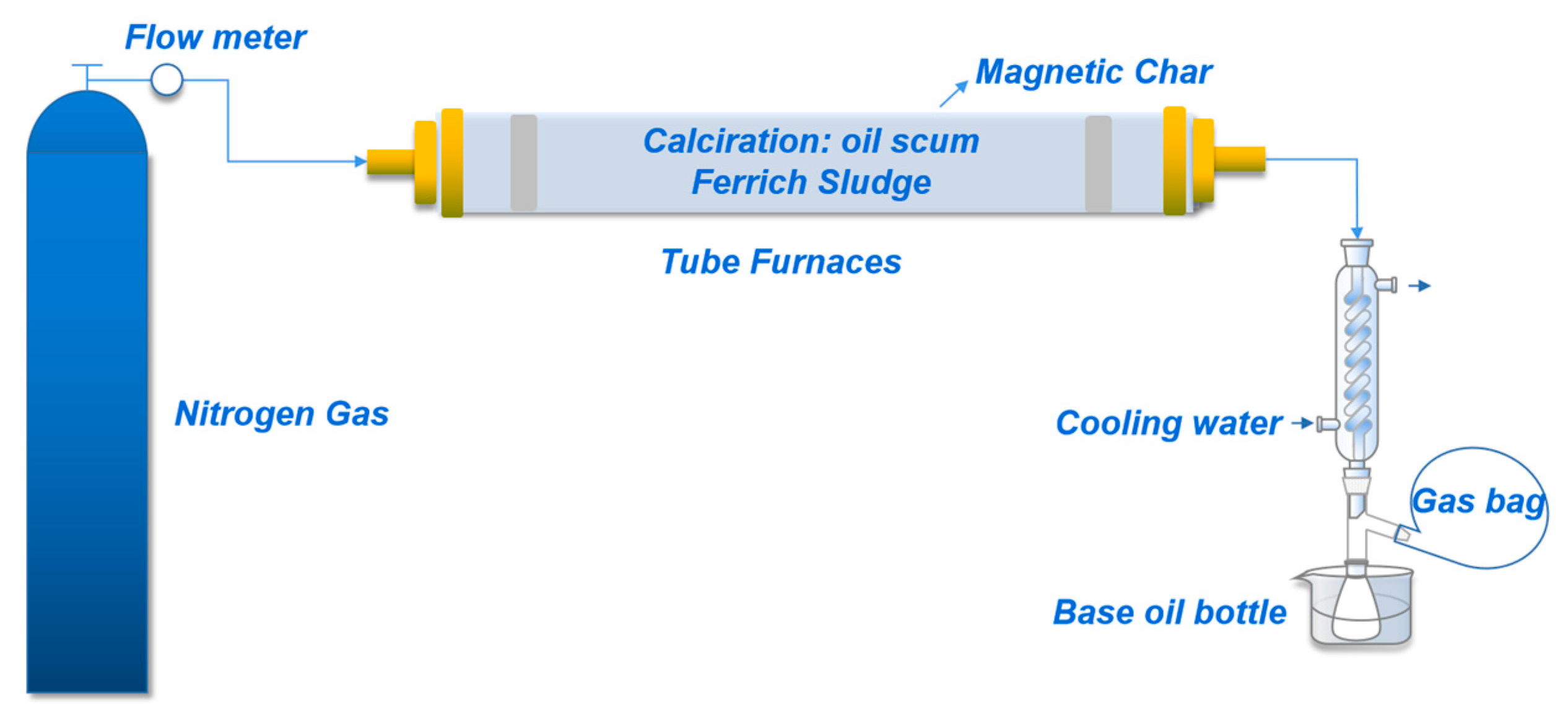
2.1. Oil Scum Pyrolysis
2.2. Chemical Reagent Experiment
2.3. Adsorption Experiment
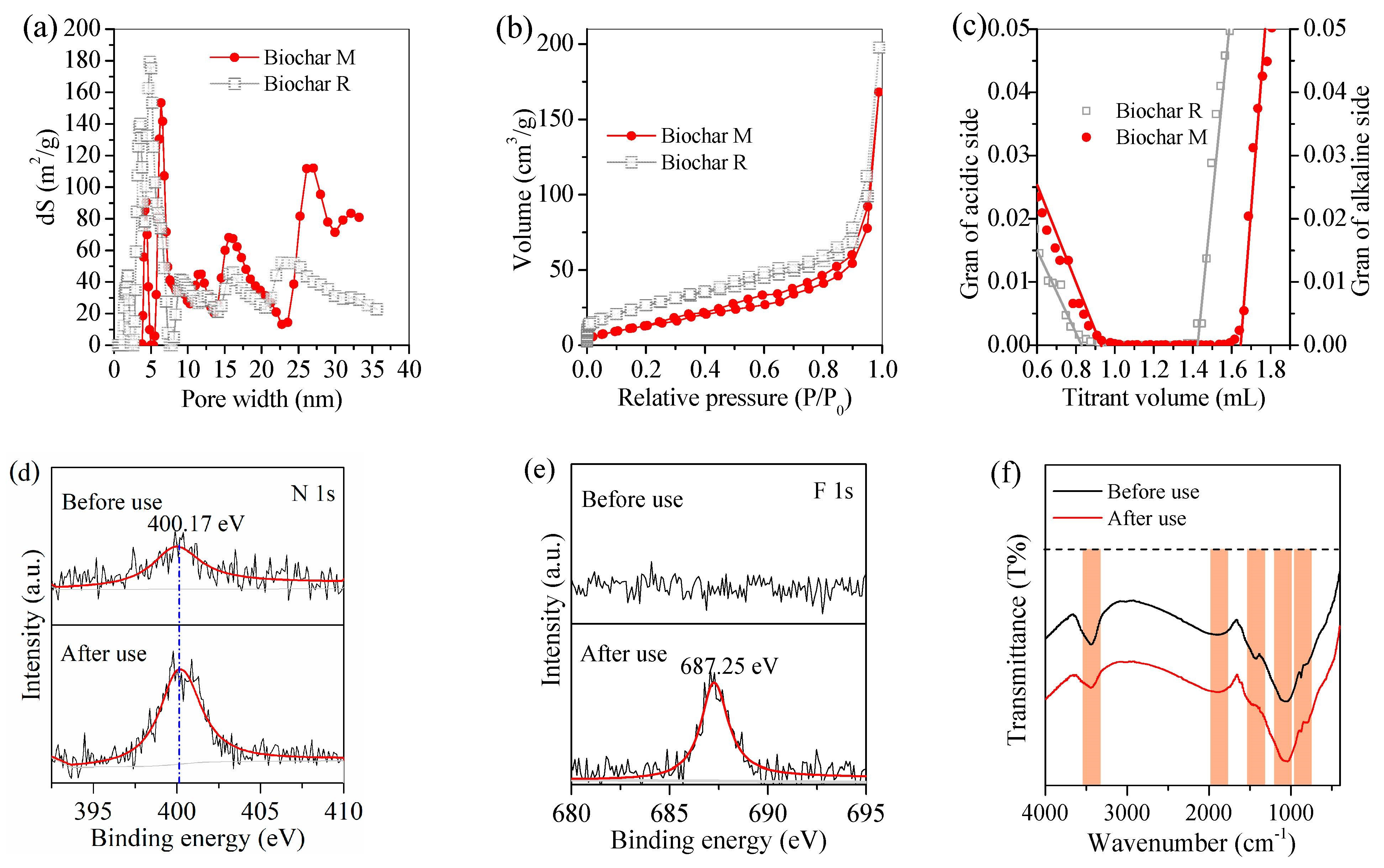
2.4. Characterization
3. Results and Discussion
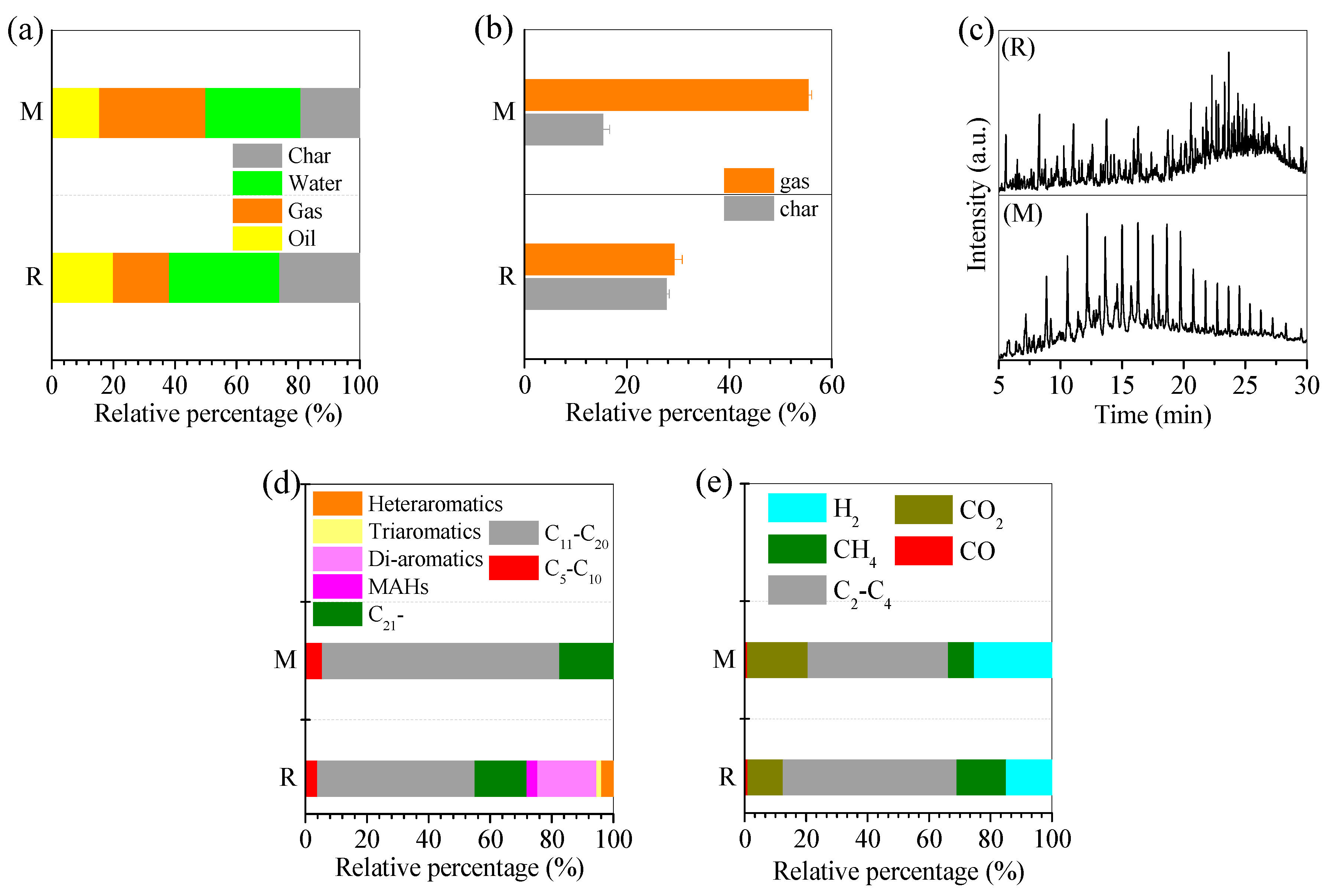
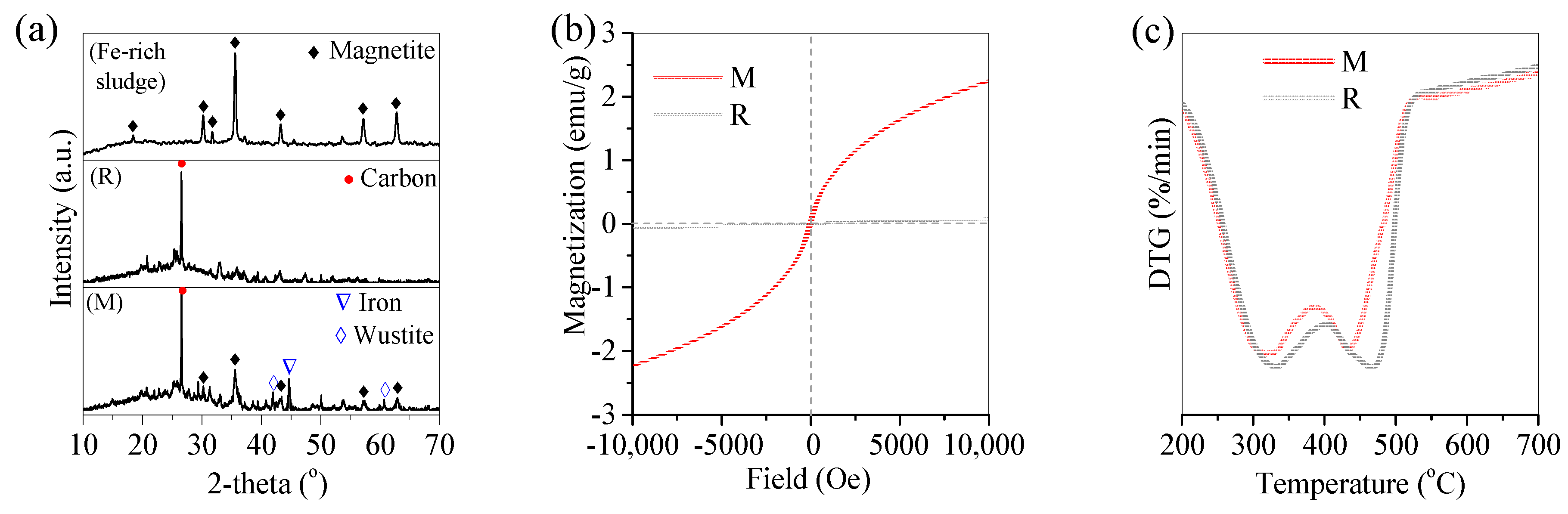
3.1. Pyrolysis of Oil Scum by Fe-Rich Sludge
3.2. Pyrolysis Mechanism of Oil Scum
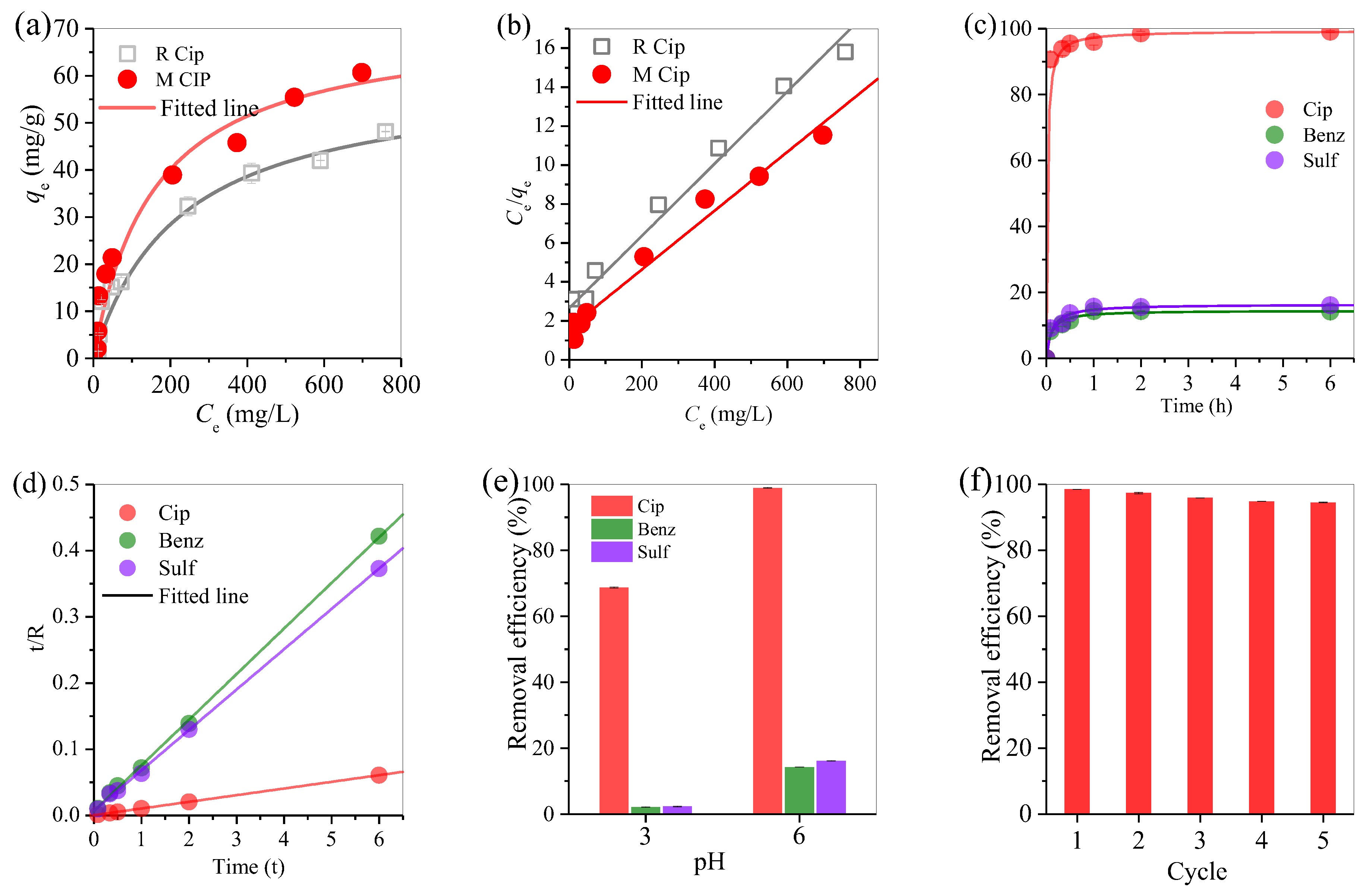
3.3. Adsorption Performance of Biochar
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sharma, N.K.; Suganya, K.; Sivapragsam, C.; Vanitha, S. Current Trends on Oil Sludge Characterization, Toxicity and Treatment Systems. Int. J. Recent Technol. Eng. 2020, 8, 13–17. [Google Scholar]
- Haghighat, M.; Majidian, N.; Hallajisani, A. Production of bio-oil from sewage sludge: A review on the thermal and catalytic conversion by pyrolysis. Sustain. Energy Technol. Assess. 2020, 42, 100870. [Google Scholar] [CrossRef]
- Sankaran, S.; Pandey, S.; Sumathy, K. Experimental investigation on waste heat recovery by refinery oil sludge incineration using fluidised-bed technique. J. Environ. Sci. Health Part A 1998, 33, 829–845. [Google Scholar] [CrossRef]
- Tsiligiannis, A.; Tsiliyannis, C. Oil refinery sludge and renewable fuel blends as energy sources for the cement industry. Renew. Energy 2020, 157, 55–70. [Google Scholar] [CrossRef]
- Hu, G.; Li, J.; Zeng, G. Recent development in the treatment of oily sludge from petroleum industry: A review. J. Hazard. Mater. 2013, 261, 470–490. [Google Scholar] [CrossRef]
- Ho, C.; Show, M.; Ong, S. Recovery of residual oil from the centrifuge sludge of a palm oil mill: Effect of enzyme digestion and surfactant treatment. J. Am. Oil Chem. Soc. 1992, 69, 276–282. [Google Scholar] [CrossRef]
- Yu, Y.; Yang, C.; Li, J.; Zhu, Y.; Yan, Z.; Zhang, H. Screening of inexpensive and efficient catalyst for microwave-assisted pyrolysis of ship oil sludge. J. Anal. Appl. Pyrolysis 2020, 152, 104971. [Google Scholar] [CrossRef]
- Lin, B.; Wang, J.; Huang, Q.; Ali, M.; Chi, Y. Aromatic recovery from distillate oil of oily sludge through catalytic pyrolysis over Zn modified HZSM-5 zeolites. J. Anal. Appl. Pyrolysis 2017, 128, 291–303. [Google Scholar] [CrossRef]
- Wahab, M.A.; Ates, F.; Yildirir, E.; Miskolczi, N. Investigation of thermal degradation kinetics and catalytic pyrolysis of industrial sludge produced from textile and leather industrial wastewater. Biomass Convers. Biorefin. 2022. [Google Scholar] [CrossRef]
- Milato, J.V.; Frana, R.J.; Rocha, A.S.; Calderari, M. Catalytic co-pyrolysis of oil sludge with HDPE to obtain paraffinic products over HUSY zeolites prepared by dealumination and desilication. J. Anal. Appl. Pyrolysis 2020, 151, 104928. [Google Scholar] [CrossRef]
- Hu, G.; Li, J.; Hou, H. A combination of solvent extraction and freeze thaw for oil recovery from petroleum refinery wastewater treatment pond sludge. J. Hazard. Mater. 2015, 283, 832–840. [Google Scholar] [CrossRef]
- Abouelnasr, D.; Al Zubaidy, E.A. Treatment and Recovery of Oil-Based Sludge Using Solvent Extraction. In International Petroleum Exhibition and Conference; OnePetro: Abu Dhabi, United Arab Emirates, 2008. [Google Scholar]
- Zhao, K.; Shu, Y.; Li, F.; Peng, G. Bimetallic catalysts as electrocatalytic cathode materials for the oxygen reduction reaction in microbial fuel cell: A review. Green Energy Environ. 2022. [Google Scholar] [CrossRef]
- Liu, J.; Yu, Y.; Zhu, S.; Yang, J.; Song, J.; Fan, W.; Yu, H.; Bian, D.; Huo, M. Synthesis and characterization of a magnetic adsorbent from negatively-valued iron mud for methylene blue adsorption. PLoS ONE 2018, 13, e0191229. [Google Scholar] [CrossRef] [PubMed]
- Qu, Z.; Dong, G.; Zhu, S.; Yu, Y.; Huo, M.; Xu, K.; Liu, M. Recycling of groundwater treatment sludge to prepare nano-rod erdite particles for tetracycline adsorption. J. Clean. Prod. 2020, 257, 120462. [Google Scholar] [CrossRef]
- Zhu, S.; Wang, Z.; Lin, X.; Sun, T.; Qu, Z.; Chen, Y.; Su, T.; Huo, Y. Effective recycling of Cu from electroplating wastewater effluent via the combined Fenton oxidation and hydrometallurgy route. J. Environ. Manag. 2020, 271, 110963. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Su, T.; Wang, J.; Zhu, S.; Zhang, Y.; Geng, Z.; Wang, X.; Gao, Y. Simultaneous removal of sulfate and nitrate from real high-salt flue gas wastewater concentrate via a waste heat crystallization route. J. Clean. Prod. 2023, 382, 135262. [Google Scholar] [CrossRef]
- Liu, Y.; Khan, A.; Wang, Z.; Chen, Y.; Zhu, S.; Sun, T.; Liang, D.; Yu, H. Upcycling of Electroplating Sludge to Prepare Erdite-Bearing Nanorods for the Adsorption of Heavy Metals from Electroplating Wastewater Effluent. Water 2020, 12, 1027. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, T.; Tong, L.; Gao, Y.; Zhang, H.; Zhang, Y.; Wang, Z.; Zhu, S. Non-free Fe dominated PMS activation for enhancing electro-Fenton efficiency in neutral wastewater. J. Electroanal. Chem. 2023, 928, 117062. [Google Scholar] [CrossRef]
- Yu, C.; Dongxu, L.; Hongyu, C.; Suiyi, Z.; Xianze, W.; Jiakuan, Y.; Xinfeng, X.; Eskola, J.; Dejun, B. Review of resource utilization of Fe-rich sludges: Purification, upcycling, and application in wastewater treatment. Environ. Rev. 2022, 30, 460–484. [Google Scholar] [CrossRef]
- Hou, J.; Wang, C.; Mao, D.; Luo, Y. The occurrence and fate of tetracyclines in two pharmaceutical wastewater treatment plants of Northern China. Environ. Sci. Pollut. Res. 2016, 23, 1722–1731. [Google Scholar] [CrossRef]
- Sarmah, A.K.; Meyer, M.T.; Boxall, A.B. A global perspective on the use, sales, exposure pathways, occurrence, fate and effects of veterinary antibiotics (VAs) in the environment. Chemosphere 2006, 65, 725–759. [Google Scholar] [CrossRef] [PubMed]
- Reis, A.C.; Kolvenbach, B.A.; Nunes, O.C.; Corvini, P.F. Biodegradation of antibiotics: The new resistance determinants—Part I. New Biotechnol. 2020, 54, 34–51. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Li, Y.; Guo, J.; Kan, Z.; Jia, Y. Catalytic ozonation mechanisms of Norfloxacin using Cu–CuFe2O4. Environ. Res. 2023, 216, 114521. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, S.A.; Janjani, H. Antibiotics adsorption from aqueous solutions using carbon nanotubes: A systematic review. Toxin Rev. 2018, 39, 87–98. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhou, J.; Zhang, L.; Zhong, S.; Ru, X.; Shu, X. Sulfur defect and Fe (III)(hydr) oxides on pyrite surface mediate tylosin adsorption in lake water: Effect of solution chemistry and dissolved organic matter. Environ. Sci. Pollut. Res. 2022, 29, 90248–90258. [Google Scholar] [CrossRef]
- Fakhri, A.; Adami, S. Adsorption and thermodynamic study of Cephalosporins antibiotics from aqueous solution onto MgO nanoparticles. J. Taiwan Inst. Chem. Eng. 2014, 45, 1001–1006. [Google Scholar] [CrossRef]
- Zhu, S.; Dong, G.; Yu, Y.; Yang, J.; Yang, W.; Fan, W.; Zhou, D.; Liu, J.; Zhang, L. Hydrothermal synthesis of a magnetic adsorbent from wasted iron mud for effective removal of heavy metals from smelting wastewater. Environ. Sci. Pollut. Res. 2018, 25, 22710–22724. [Google Scholar] [CrossRef]
- Abdrakhimov, V.; Abdrakhimova, E. Study of the distribution of iron oxides in intershale clay and oil sludge porous filler with Mossbauer spectroscopy. Theor. Found. Chem. Eng. 2019, 53, 703–707. [Google Scholar] [CrossRef]
- Lin, B.; Huang, Q.; Yang, Y.; Chi, Y. Preparation of Fe-char catalyst from tank cleaning oily sludge for the catalytic cracking of oily sludge. J. Anal. Appl. Pyrolysis 2019, 139, 308–318. [Google Scholar] [CrossRef]
- El-Bendary, N.; El-Etriby, H.K.; Mahanna, H. Reuse of adsorption residuals for enhancing removal of ciprofloxacin from wastewater. Environ. Technol. 2021, 26, 1–17. [Google Scholar] [CrossRef]
- Li, J.; Yu, G.; Pan, L.; Li, C.; You, F.; Xie, S.; Wang, Y.; Ma, J.; Shang, X. Study of ciprofloxacin removal by biochar obtained from used tea leaves. J. Environ. Sci. 2018, 73, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.; Karthikeyan, K.G. Sorption of the Antimicrobial Ciprofloxacin to Aluminum and Iron Hydrous Oxides. Environ. Sci. Technol. 2005, 39, 9166–9173. [Google Scholar] [CrossRef] [PubMed]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Ji, M.; Qin, J.; Zhu, J.; Zhu, S. Catalytic Performance of Fe-Rich Sludge in Pyrolysis of Waste Oil Scum as Volatiles and Magnetic Char. Water 2023, 15, 2637. https://doi.org/10.3390/w15142637
Liu J, Ji M, Qin J, Zhu J, Zhu S. Catalytic Performance of Fe-Rich Sludge in Pyrolysis of Waste Oil Scum as Volatiles and Magnetic Char. Water. 2023; 15(14):2637. https://doi.org/10.3390/w15142637
Chicago/Turabian StyleLiu, Jiancong, Manhong Ji, Jiabao Qin, Jia Zhu, and Suiyi Zhu. 2023. "Catalytic Performance of Fe-Rich Sludge in Pyrolysis of Waste Oil Scum as Volatiles and Magnetic Char" Water 15, no. 14: 2637. https://doi.org/10.3390/w15142637
APA StyleLiu, J., Ji, M., Qin, J., Zhu, J., & Zhu, S. (2023). Catalytic Performance of Fe-Rich Sludge in Pyrolysis of Waste Oil Scum as Volatiles and Magnetic Char. Water, 15(14), 2637. https://doi.org/10.3390/w15142637