Stripping of Cu Ion from Aquatic Media by Means of MgY2O4@g-C3N4 Nanomaterials
Abstract
:1. Introduction
2. Experimental Procedures
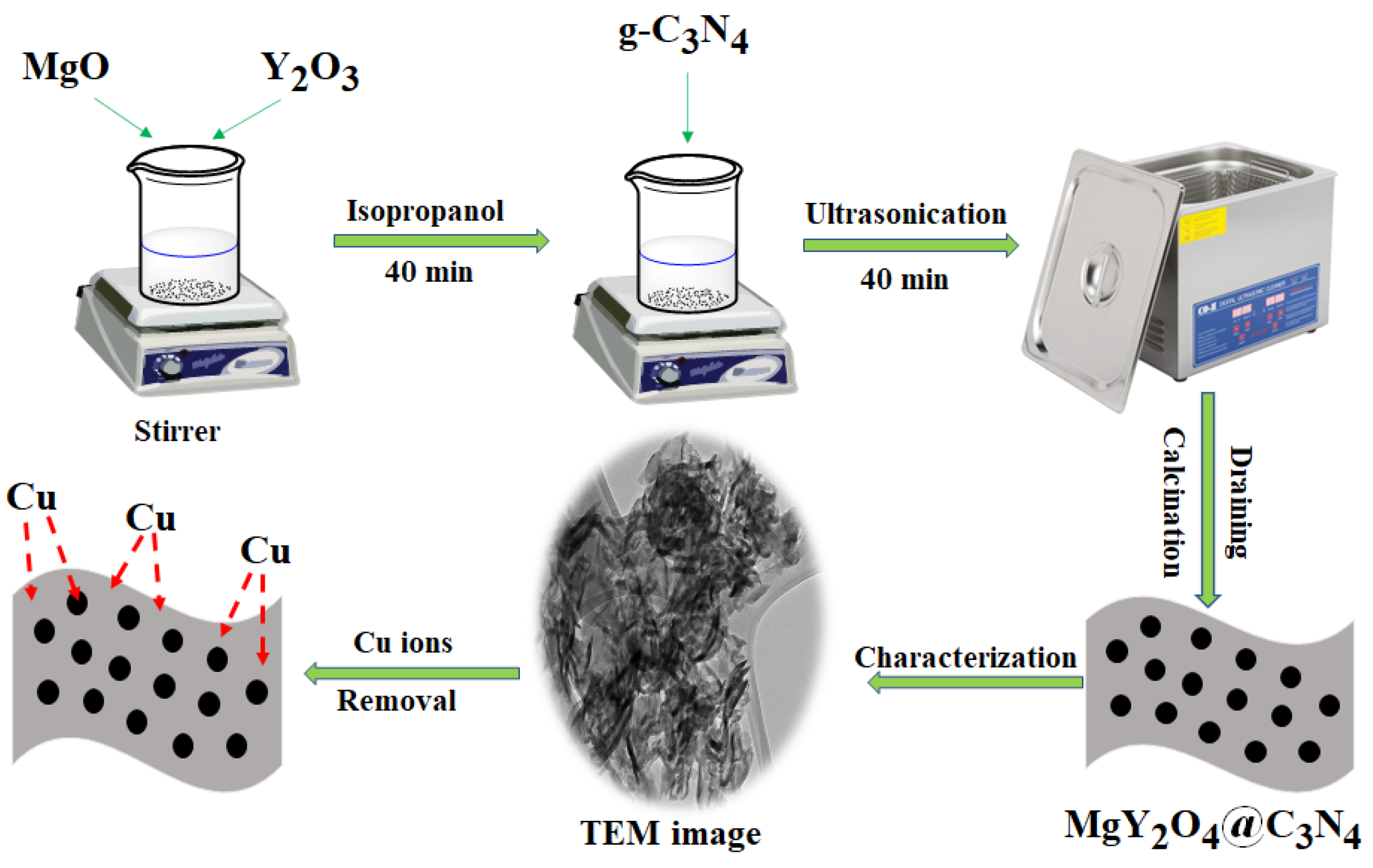
2.1. Preparation of MgY2O4@g-C3N4 Nanostructures
2.2. Characterizations of MgY2O4@g-C3N4 Nanostructures
2.3. Cu (II) Adsorption Test
| Kinetics Model | Equation | Plots | Ref. | |
| Pseudo-first-order | (7) | [1] | ||
| Pseudo-second-order | (8) | [2,3] | ||
| Elovich | (9) | [4] | ||
| Intra-particle Diffusion | (10) | [5,6] | ||
| Mass Transfer | (11) | [5,6] | ||
3. Results and Discussions
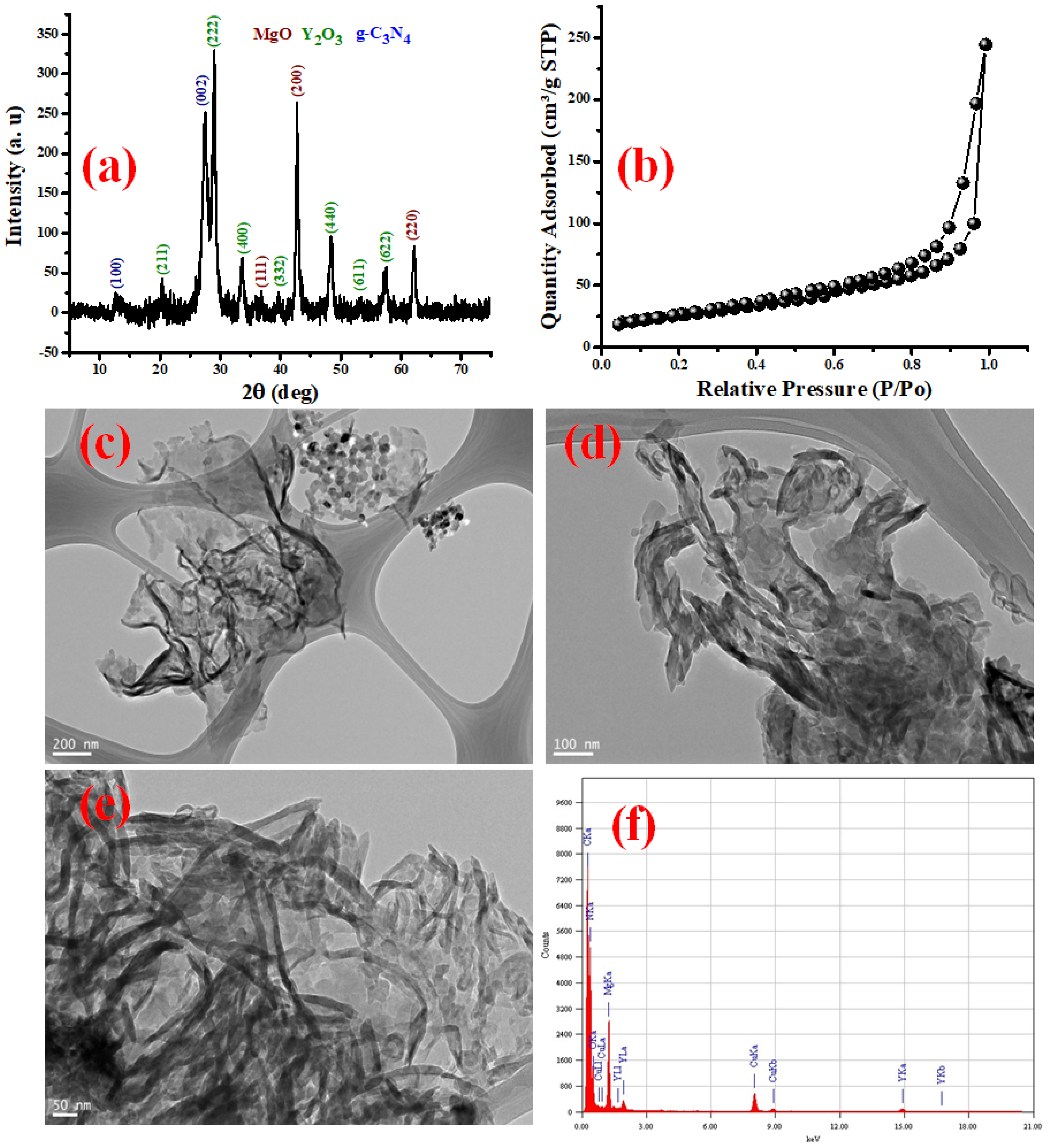
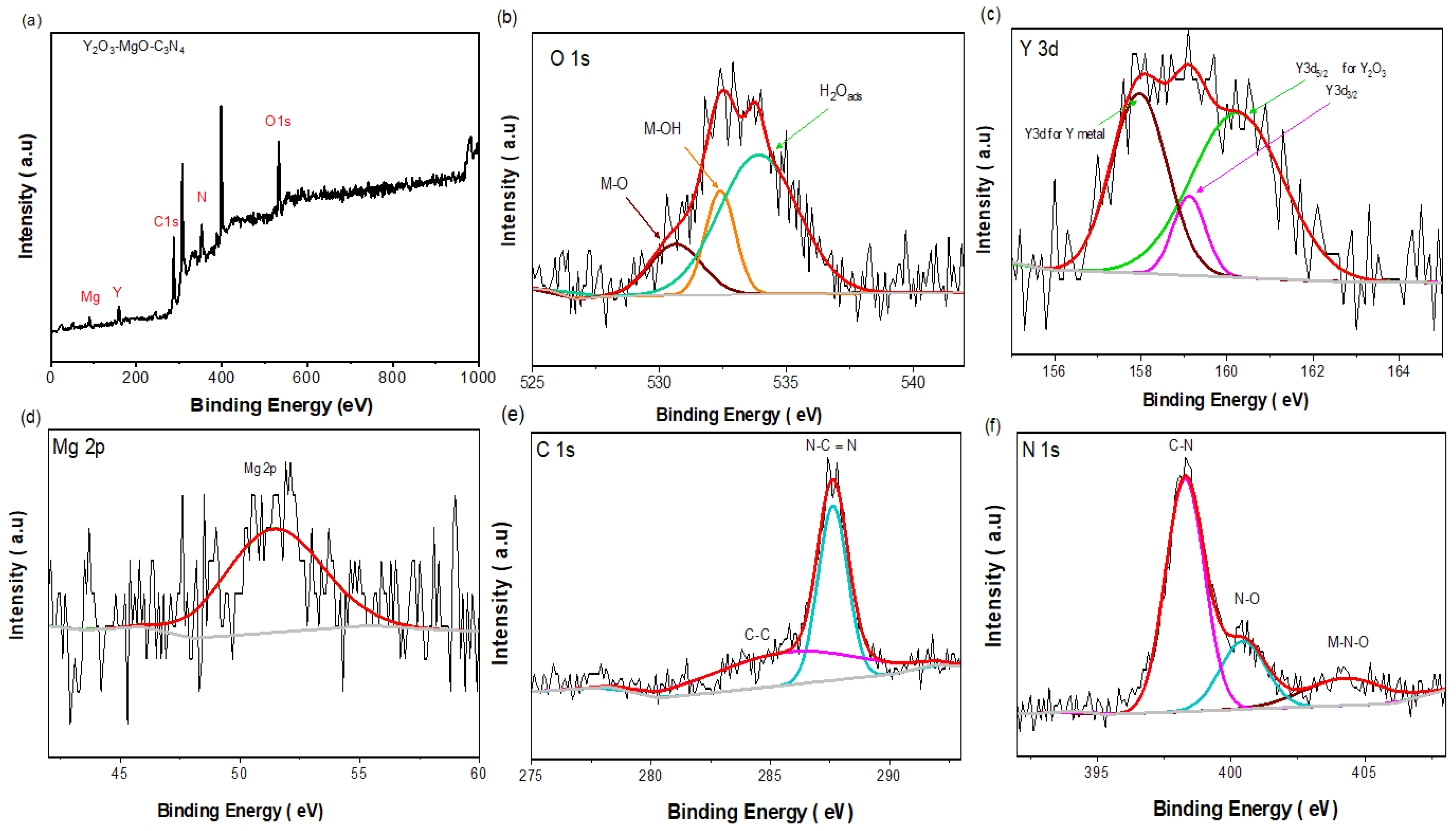
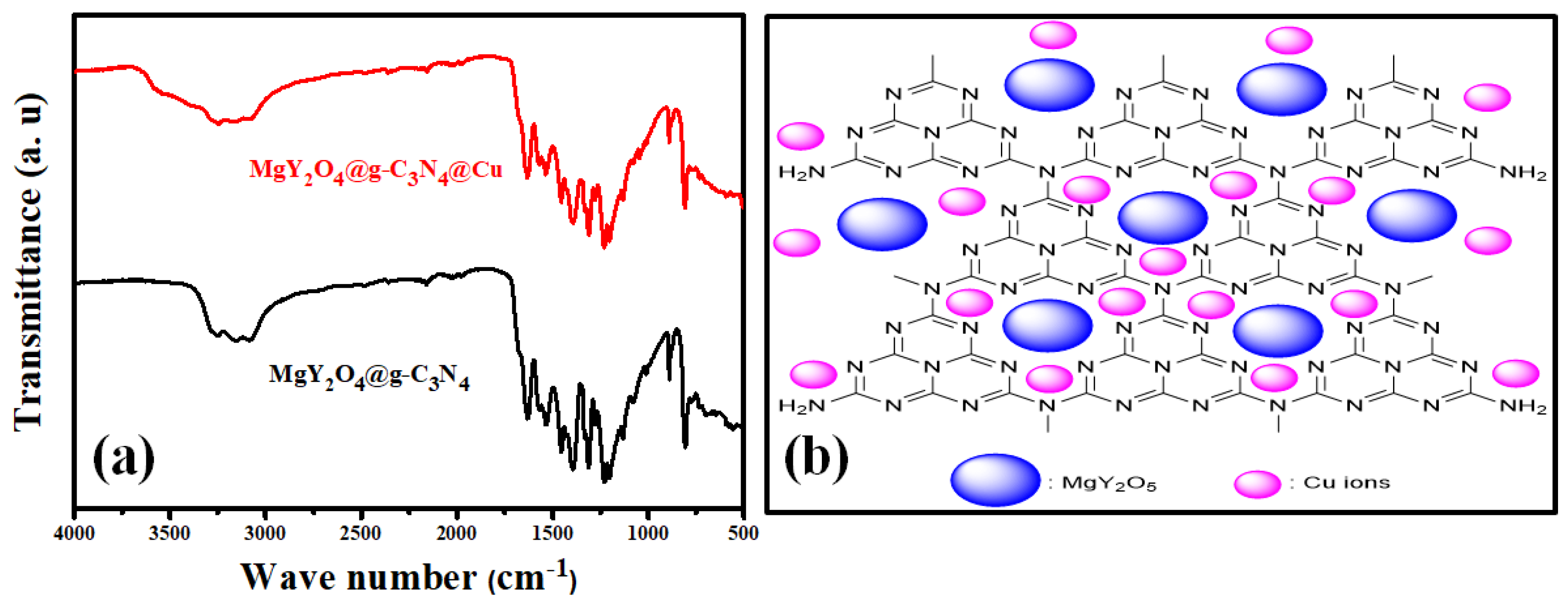
3.1. MgY2O4@g-C3N4 Nanomaterial Structural Analysis
3.2. Cu (II) Removal onto MgY2O4@g-C3N4 Nanomaterial
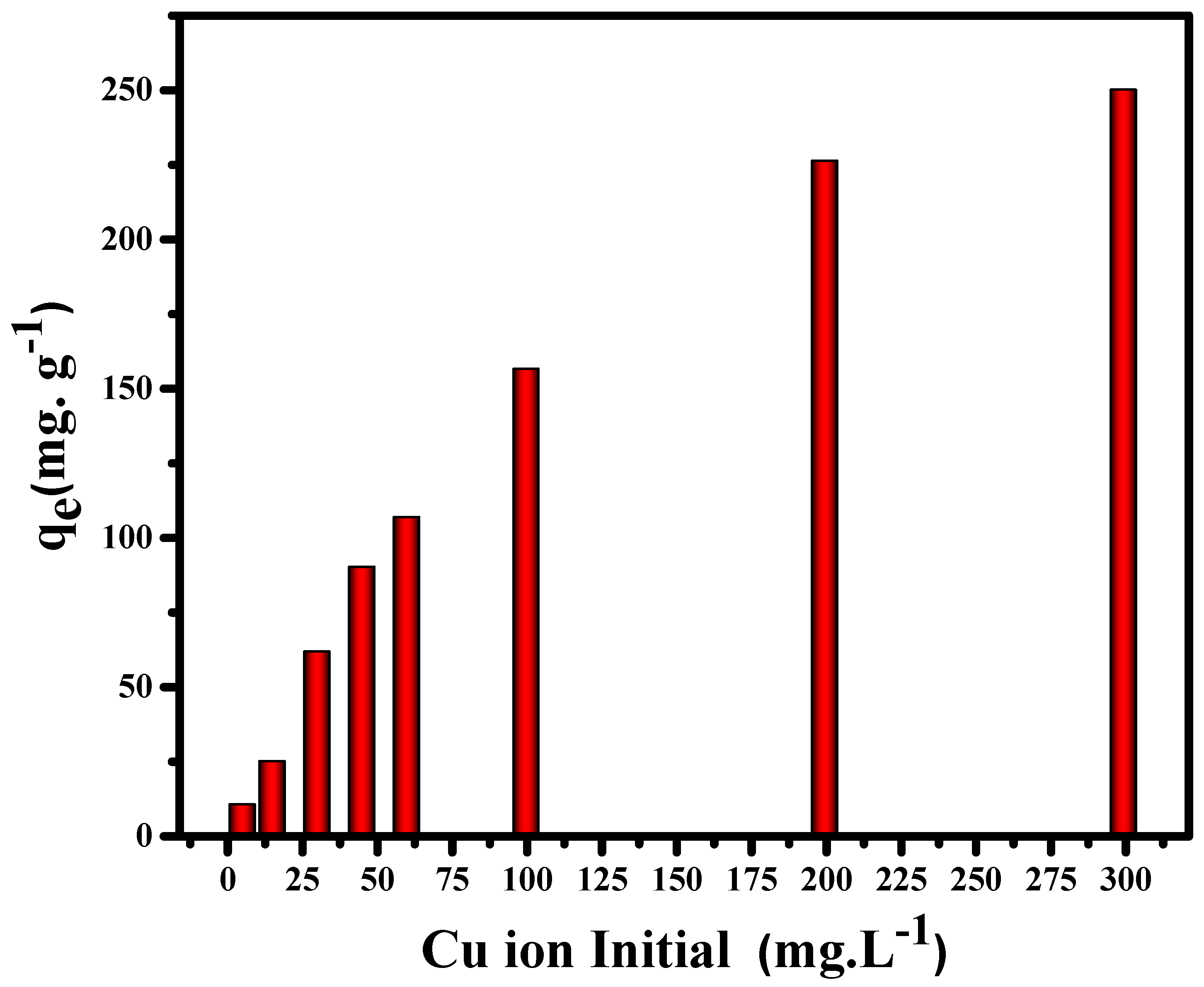
3.2.1. Effect of Cu (II) Initial Concentration
3.2.2. Cu (II) Removal and pH
3.2.3. Cu (II) Elimination and Equilibrium Contact Time
3.2.4. Adsorption Isotherms Modeling
3.2.5. Adsorption Kinetics Modeling
| Intra-Particle Diffusion/Transport Model | |||||
| kdif (mg·g−1·min−1/2) | C1 | r2 | kdif (mg·g−1·min−1/2) | C2 | r2 |
| 17.33 | 14.81 | 0.9987 | 1.30 | 49.78 | 0.9804 |
3.3. Mechanism of Cu (II) Adsorption onto MgY2O4@g-C3N4 Nanomaterial
3.4. Reusability and Stability
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saravanan, A.; Kumar, P.S.; Ramesh, B.; Srinivasan, S. Removal of toxic heavy metals using genetically engineered microbes: Molecular tools, risk assessment and management strategies. Chemosphere 2022, 298, 134341. [Google Scholar] [CrossRef]
- Mukherjee, A.G.; Wanjari, U.R.; Renu, K.; Vellingiri, B.; Gopalakrishnan, A.V. Heavy metal and metalloid-induced reproductive toxicity. Environ. Toxicol. Pharmacol. 2022, 92, 103859. [Google Scholar] [CrossRef] [PubMed]
- Burnase, N.; Jaiswal, S.; Barapatre, A. Metal Toxicity in Humans Associated with Their Occupational Exposures Due to Mining. In Medical Geology in Mining: Health Hazards Due to Metal Toxicity; Springer: Cham, Switzerland, 2022; pp. 127–186. [Google Scholar] [CrossRef]
- Xu, R.; Yang, Z.; Niu, Y.; Xu, D.; Wang, J.; Han, J.; Wang, H. Removal of microplastics and attached heavy metals from secondary effluent of wastewater treatment plant using interpenetrating bipolar plate electrocoagulation. Sep. Purif. Technol. 2022, 290, 120905. [Google Scholar] [CrossRef]
- Chakraborty, S.C.; Zaman, W.U.; Hoque, M.; Qamruzzaman; Zaman, J.U.; Hossain, D.; Pramanik, B.K.; Nguyen, L.N.; Nghiem, L.D.; Mofijur; et al. Metals extraction processes from electronic waste: Constraints and opportunities. Environ. Sci. Pollut. Res. 2022, 29, 32651–32669. [Google Scholar] [CrossRef]
- Liaqat, A.; Zahra, I.; Abbas, S.Z.; Wabaidur, S.M.; Eldesoky, G.E.; Islam, A.; Rafatullah, M.; Shakoori, F.R.; Shakoori, A.R. Copper Bioremediation Ability of Ciliate Paramecium multimicronucleatum Isolated from Industrial Wastewater. Water 2022, 14, 1419. [Google Scholar] [CrossRef]
- Ismail, M.; Jobara, A.; Bekouche, H.; Allateef, M.A.; Ben Aissa, M.A.; Modwi, A. Impact of Cu Ions removal onto MgO nanostructures: Adsorption capacity and mechanism. J. Mater. Sci. Mater. Electron. 2022, 33, 12500–12512. [Google Scholar] [CrossRef]
- Abaker, Z.; Hussein, T.; Makawi, S.; Mustafa, B.; Modwi, A. Superior uptake of Cu (II) from aquatic media via Y2O3-ZnO nanostructures. Nano-Struct. Nano-Objects 2022, 30, 100879. [Google Scholar] [CrossRef]
- Lou, X.-Y.; Boada, R.; Verdugo, V.; Simonelli, L.; Pérez, G.; Valiente, M. Decoupling the adsorption mechanisms of arsenate at molecular level on modified cube-shaped sponge loaded superparamagnetic iron oxide nanoparticles. J. Environ. Sci. 2022, 121, 1–12. [Google Scholar] [CrossRef]
- Khulbe, K.C.; Matsuura, T. Removal of heavy metals and pollutants by membrane adsorption techniques. Appl. Water Sci. 2018, 8, 19. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Li, H.; Wu, Z.; Post, J.E.; Lanson, B.; Liu, Y.; Hu, B.; Wang, M.; Zhang, L.; Hong, M.; et al. Effects of cobalt doping on the reactivity of hausmannite for As(III) oxidation and As(V) adsorption. J. Environ. Sci. 2022, 122, 217–226. [Google Scholar] [CrossRef]
- Zaimee, M.Z.A.; Sarjadi, M.S.; Rahman, L. Heavy Metals Removal from Water by Efficient Adsorbents. Water 2021, 13, 2659. [Google Scholar] [CrossRef]
- Zamora-Ledezma, C.; Negrete-Bolagay, D.; Figueroa, F.; Zamora-Ledezma, E.; Ni, M.; Alexis, F.; Guerrero, V.H. Heavy metal water pollution: A fresh look about hazards, novel and conventional remediation methods. Environ. Technol. Innov. 2021, 22, 101504. [Google Scholar] [CrossRef]
- Lou, X.-Y.; Boada, R.; Simonelli, L.; Valiente, M. Enhanced arsenite removal by superparamagnetic iron oxide nanoparticles in-situ synthesized on a commercial cube-shape sponge: Adsorption-oxidation mechanism. J. Colloid Interface Sci. 2022, 614, 460–467. [Google Scholar] [CrossRef]
- Qi, X.; Tong, X.; Pan, W.; Zeng, Q.; You, S.; Shen, J. Recent advances in polysaccharide-based adsorbents for wastewater treatment. J. Clean. Prod. 2021, 315, 128221. [Google Scholar] [CrossRef]
- Rajasulochana, P.; Preethy, V. Comparison on efficiency of various techniques in treatment of waste and sewage water—A comprehensive review. Resour. Technol. 2016, 2, 175–184. [Google Scholar] [CrossRef] [Green Version]
- Ali, M.; Modwi, A.; Idriss, H.; Aldaghri, O.; Ismail, M.; Ibnaouf, K. Detoxification of Pb (II) from aquatic media via CaMgO2@g-C3N4 nanocomposite. Mater. Lett. 2022, 322, 132501. [Google Scholar] [CrossRef]
- Nair, R.R.; Mary, B.C.J.; Vijaya, J.J.; Mustafa, A.; Khezami, L.; Modwi, A.; Ismail, M.; Bououdina, M.; Lemine, O.M. Reduced graphene oxide/spinel ferrite nanocomposite as an efficient adsorbent for the removal of Pb (II) from aqueous solution. J. Mater. Sci. Mater. Electron. 2021, 32, 28253–28274. [Google Scholar] [CrossRef]
- Idriss, H.A.; Alakhras, A.; El Khair, H. Malachite green removal from aqueous solutions by MgO (86%). Se (7%). Te (7%) nanocomposites. Chalcogenide Lett. 2021, 18, 629–638. [Google Scholar] [CrossRef]
- Khezami, L.; Ben Aissa, M.A.; Modwi, A.; Ismail, M.; Guesmi, A.; Algethami, F.K.; Ben Ticha, M.; Assadi, A.A.; Nguyen-Tri, P. Harmonizing the photocatalytic activity of g-C3N4 nanosheets by ZrO2 stuffing: From fabrication to experimental study for the wastewater treatment. Biochem. Eng. J. 2022, 182, 108411. [Google Scholar] [CrossRef]
- Idriss, H. Decolorization of malachite green in aqueous solution via MgO nanopowder. J. Optoelectron. Biomed. Mater. 2021, 13, 183–192. [Google Scholar] [CrossRef]
- Ben Aissa, M.A.; Khezami, L.; Taha, K.; Elamin, N.; Mustafa, B.; Al-Ayed, A.S.; Modwi, A. Yttrium oxide-doped ZnO for effective adsorption of basic fuchsin dye: Equilibrium, kinetics, and mechanism studies. Int. J. Environ. Sci. Technol. 2021, 19, 9901–9914. [Google Scholar] [CrossRef]
- Modwi, A.; Khezami, L.; Ghoniem, M.G.; Nguyen-Tri, P.; Baaloudj, O.; Guesmi, A.; AlGethami, F.K.; Amer, M.S.; Assadi, A.A. Superior removal of dyes by mesoporous MgO/g-C3N4 fabricated through ultrasound method: Adsorption mechanism and process modeling. Environ. Res. 2022, 205, 112543. [Google Scholar] [CrossRef] [PubMed]
- Toghan, A.; Abd El-Lateef, H.M.; Taha, K.K.; Modwi, A. Mesoporous TiO2@g-C3N4 composite: Construction, characterization, and boosting indigo carmine dye destruction. Diam. Relat. Mater. 2021, 118, 108491. [Google Scholar] [CrossRef]
- Osorio-Vargas, P.; Pais-Ospina, D.; Marin-Silva, D.A.; Pinotti, A.; Damonte, L.; Cánneva, A.; Donadelli, J.A.; da Costa, L.P.; Pizzio, L.R.; Torres, C.C.; et al. TiO2 nanorods doped with g-C3N4—Polyethylene composite coating for self-cleaning applications. Mater. Chem. Phys. 2022, 288, 126356. [Google Scholar] [CrossRef]
- Reddy, K.R.; Reddy, C.V.; Nadagouda, M.N.; Shetti, N.P.; Jaesool, S.; Aminabhavi, T.M. Polymeric graphitic carbon nitride (g-C3N4)-based semiconducting nanostructured materials: Synthesis methods, properties and photocatalytic applications. J. Environ. Manag. 2019, 238, 25–40. [Google Scholar] [CrossRef]
- Al-Jabari, M.H.; Sulaiman, S.; Ali, S.; Barakat, R.; Mubarak, A.; Khan, S.A. Adsorption study of levofloxacin on reusable magnetic nanoparticles: Kinetics and antibacterial activity. J. Mol. Liq. 2019, 291, 111249. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef] [Green Version]
- Hu, Q.; Zhang, Z. Application of Dubinin–Radushkevich isotherm model at the solid/solution interface: A theoretical analysis. J. Mol. Liq. 2019, 277, 646–648. [Google Scholar] [CrossRef]
- Temkin, M.I. Adsorption equilibrium and the kinetics of processes on nonhomogeneous surfaces and in the interaction between adsorbed molecules. Zh. Fiz. Chim. 1941, 15, 296–332. [Google Scholar]
- Ghorbani, M.; Ariavand, S.; Aghamohammadhasan, M.; Seyedin, O. Synthesis and optimization of a green and efficient sorbent for removal of three heavy metal ions from wastewater samples: Kinetic, thermodynamic, and isotherm studies. J. Iran. Chem. Soc. 2021, 18, 1947–1963. [Google Scholar] [CrossRef]
- Yu, Y.; Yan, W.; Wang, X.; Li, P.; Gao, W.; Zou, H.; Wu, S.; Ding, K. Surface engineering for extremely enhanced charge separation and photocatalytic hydrogen evolution on g-C3N4. Adv. Mater. 2018, 30, 1705060. [Google Scholar] [CrossRef] [PubMed]
- Vesali-Kermani, E.; Habibi-Yangjeh, A.; Ghosh, S. Visible-light-induced nitrogen photofixation ability of g-C3N4 nanosheets decorated with MgO nanoparticles. J. Ind. Eng. Chem. 2020, 84, 185–195. [Google Scholar] [CrossRef]
- Chen, G.; Lei, R.; Wang, H.; Huang, F.; Zhao, S.; Xu, S. Temperature-dependent emission color and temperature sensing behavior in Tm3+/Yb3+: Y2O3 nanoparticles. Opt. Mater. 2018, 77, 233–239. [Google Scholar] [CrossRef]
- Sotomayor, F.J.; Cychosz, K.A.; Thommes, M. Characterization of micro/mesoporous materials by physisorption: Concepts and case studies. Acc. Mater. Surf. Res 2018, 3, 34–50. [Google Scholar]
- Tran, C.V.; Quang, D.V.; Nguyen Thi, H.P.; Truong, T.N.; La, D.D. Effective removal of Pb (II) from aqueous media by a new design of Cu–Mg binary ferrite. ACS Omega 2020, 5, 7298–7306. [Google Scholar] [CrossRef]
- Mao, N.; Jiang, J.-X. MgO/g-C3N4 nanocomposites as efficient water splitting photocatalysts under visible light irradiation. Appl. Surf. Sci. 2019, 476, 144–150. [Google Scholar] [CrossRef]
- van der Heide, P. Photoelectron binding energy shifts observed during oxidation of group IIA, IIIA and IVA elemental surfaces. J. Electron Spectrosc. Relat. Phenom. 2006, 151, 79–91. [Google Scholar] [CrossRef]
- Pan, T.-M.; Lee, J.-D. Physical and Electrical Properties of Yttrium Oxide Gate Dielectrics on Si Substrate with NH3 Plasma Treatment. J. Electrochem. Soc. 2007, 154, H698–H703. [Google Scholar] [CrossRef]
- Amer, M.S.; Arunachalam, P.; Al-Mayouf, A.M.; Prasad, S.; Alshalwi, M.N.; Ghanem, M.A. Mesoporous Tungsten Trioxide Photoanodes Modified with Nitrogen-Doped Carbon Quantum Dots for Enhanced Oxygen Evolution Photo-Reaction. Nanomaterials 2019, 9, 1502. [Google Scholar] [CrossRef] [Green Version]
- He, Y.; Wang, Y.; Zhang, L.; Teng, B.; Fan, M. High-efficiency conversion of CO2 to fuel over ZnO/g-C3N4 photocatalyst. Appl. Catal. B Environ. 2015, 168, 1–8. [Google Scholar] [CrossRef]
- Sun, Q.; Lv, K.; Zhang, Z.; Li, M.; Li, B. Effect of contact interface between TiO2 and g-C3N4 on the photoreactivity of g-C3N4/TiO2 photocatalyst: (0 0 1) vs (1 0 1) facets of TiO2. Appl. Catal. B Environ. 2015, 164, 420–427. [Google Scholar]
- Hamdy, A. Experimental study of the relationship between dissolved iron, turbidity, and removal of Cu (II) ion from aqueous solutions using zero-valent iron nanoparticles. Arab. J. Sci. Eng. 2021, 46, 5543–5565. [Google Scholar] [CrossRef]
- Zhu, S.; Xu, J.; Wang, B.; Xie, J.; Ying, G.; Li, J.; Cheng, Z.; Li, J.; Chen, K. Highly efficient and rapid purification of organic dye wastewater using lignin-derived hierarchical porous carbon. J. Colloid Interface Sci. 2022, 625, 158–168. [Google Scholar] [CrossRef]
- Biswas, M.C.; Chowdhury, A.; Hossain, M. Applications, drawbacks, and future scope of nanoparticle-based polymer composites. In Nanoparticle-Based Polymer Composites; Elsevier: Amsterdam, The Netherlands, 2022; pp. 243–275. [Google Scholar] [CrossRef]
- García-Diéguez, M.; Hibbitts, D.D.; Iglesia, E. Hydrogen Chemisorption Isotherms on Platinum Particles at Catalytic Temperatures: Langmuir and Two-Dimensional Gas Models Revisited. J. Phys. Chem. C 2019, 123, 8447–8462. [Google Scholar] [CrossRef]
- Baaloudj, O.; Nasrallah, N.; Kebir, M.; Guedioura, B.; Amrane, A.; Nguyen-Tri, P.; Nanda, S.; Assadi, A.A. Artificial neural network modeling of cefixime photodegradation by synthesized CoBi2O4 nanoparticles. Environ. Sci. Pollut. Res. 2021, 28, 15436–15452. [Google Scholar] [CrossRef]
- Karri, R.R.; Sahu, J. Modeling and optimization by particle swarm embedded neural network for adsorption of zinc (II) by palm kernel shell based activated carbon from aqueous environment. J. Environ. Manag. 2018, 206, 178–191. [Google Scholar] [CrossRef]
- Charaabi, S.; Absi, R.; Pensé-Lhéritier, A.-M.; Le Borgne, M.; Issa, S. Adsorption studies of benzophenone-3 onto clay minerals and organosilicates: Kinetics and modelling. Appl. Clay Sci. 2021, 202, 105937. [Google Scholar] [CrossRef]
- Modwi, A.; Khezami, L.; Taha, K.; Al-Duaij, O.; Houas, A. Fast and high efficiency adsorption of Pb(II) ions by Cu/ZnO composite. Mater. Lett. 2017, 195, 41–44. [Google Scholar] [CrossRef]
- Khezami, L.; Taha, K.K.; Modwi, A. Efficient Removal of Cobalt from Aqueous Solution by Zinc Oxide Nanoparticles: Kinetic and Thermodynamic Studies. Z. Für Nat. A 2017, 72, 409–418. [Google Scholar] [CrossRef]
- Usman, A.R.; Sallam, A.S.; Al-Omran, A.; El-Naggar, A.H.; Alenazi, K.K.; Nadeem, M.; Al-Wabel, M.I. Chemically Modified Biochar Produced from Conocarpus Wastes: An Efficient Sorbent for Fe(II) Removal from Acidic Aqueous Solutions. Adsorpt. Sci. Technol. 2013, 31, 625–640. [Google Scholar] [CrossRef]
- Kołodyńska, D.; Wnętrzak, R.; Leahy, J.; Hayes, M.; Kwapiński, W.; Hubicki, Z. Kinetic and adsorptive characterization of biochar in metal ions removal. Chem. Eng. J. 2012, 197, 295–305. [Google Scholar] [CrossRef]
- Usman, A.; Sallam, A.; Zhang, M.; Vithanage, M.; Ahmad, M.; Al-Farraj, A.; Ok, Y.S.; Abduljabbar, A.; Al-Wabel, M. Sorption Process of Date Palm Biochar for Aqueous Cd (II) Removal: Efficiency and Mechanisms. Water Air Soil Pollut. 2016, 227, 449. [Google Scholar] [CrossRef]
- Gibson, K.; Glaser, J.; Milke, E.; Marzini, M.; Tragl, S.; Binnewies, M.; Mayer, H.A.; Meyer, H.-J. Preparation of carbon nitride materials by polycondensation of the single-source precursor aminodichlorotriazine (ADCT). Mater. Chem. Phys. 2008, 112, 52–56. [Google Scholar] [CrossRef]
- Shen, C.; Chen, C.; Wen, T.; Zhao, Z.; Wang, X.; Xu, A. Superior adsorption capacity of g-C3N4 for heavy metal ions from aqueous solutions. J. Colloid Interface Sci. 2015, 456, 7–14. [Google Scholar] [CrossRef]
- Cui, Y.; Zhang, J.; Zhang, G.; Huang, J.; Liu, P.; Antonietti, M.; Wang, X. Synthesis of bulk and nanoporous carbon nitride polymers from ammonium thiocyanate for photocatalytic hydrogen evolution. J. Mater. Chem. 2011, 21, 13032–13039. [Google Scholar] [CrossRef]
- Feng, Y.; Chen, G.; Zhang, Y.; Li, D.; Ling, C.; Wang, Q.; Liu, G. Superhigh co-adsorption of tetracycline and copper by the ultrathin g-C3N4 modified graphene oxide hydrogels. J. Hazard. Mater. 2021, 424, 127362. [Google Scholar] [CrossRef]
- Abou-Zeid, R.E.; Ali, K.A.; Gawad, R.M.; Kamal, K.H.; Kamel, S.; Khiari, R. Removal of Cu (II), Pb (II), Mg (II), and Fe (II) by adsorption onto alginate/nanocellulose beads as bio-sorbent. J. Renew. Mater. 2021, 9, 601. [Google Scholar] [CrossRef]
- Azzaz, A.A.; Jellali, S.; Akrout, H.; Assadi, A.A.; Bousselmi, L. Dynamic investigations on cationic dye desorption from chemically modified lignocellulosic material using a low-cost eluent: Dye recovery and anodic oxidation efficiencies of the desorbed solutions. J. Clean. Prod. 2018, 201, 28–38. [Google Scholar] [CrossRef]
- Eldeeb, T.M.; El Nemr, A.; Khedr, M.H.; El-Dek, S.I. Efficient removal of Cu(II) from water solution using magnetic chitosan nanocomposite. Nanotechnol. Environ. Eng. 2021, 6, 34. [Google Scholar] [CrossRef]
- Chen, X.; Chen, G.; Chen, L.; Chen, Y.; Lehmann, J.; McBride, M.B.; Hay, A.G. Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour. Technol. 2011, 102, 8877–8884. [Google Scholar] [CrossRef]
- Wang, X.; Liang, X.; Wang, Y.; Wang, X.; Liu, M.; Yin, D.; Xia, S.; Zhao, J.; Zhang, Y. Adsorption of Copper (II) onto activated carbons from sewage sludge by microwave-induced phosphoric acid and zinc chloride activation. Desalination 2011, 278, 231–237. [Google Scholar] [CrossRef]
- Han, Y.; Boateng, A.A.; Qi, P.X.; Lima, I.M.; Chang, J. Heavy metal and phenol adsorptive properties of biochars from pyrolyzed switchgrass and woody biomass in correlation with surface properties. J. Environ. Manag. 2013, 118, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.-X.; Jiang, H. Amino modification of biochar for enhanced adsorption of copper ions from synthetic wastewater. Water Res. 2014, 48, 396–405. [Google Scholar] [CrossRef]
- Xiao, F.; Cheng, J.; Cao, W.; Yang, C.; Chen, J.; Luo, Z. Removal of heavy metals from aqueous solution using chitosan-combined magnetic biochars. J. Colloid Interface Sci. 2019, 540, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Kuang, Y.; Zhang, Z.; Wu, D. Synthesis of graphene oxide/polyethyleneimine sponge and its performance in the sustainable removal of Cu(II) from water. Sci. Total. Environ. 2021, 806, 151258. [Google Scholar] [CrossRef] [PubMed]
- Bilgic, A. Fabrication of monoBODIPY-functionalized Fe3O4@ SiO2@ TiO2 nanoparticles for the photocatalytic degradation of rhodamine B under UV irradiation and the detection and removal of Cu (II) ions in aqueous solutions. J. Alloy. Compd. 2022, 899, 163360. [Google Scholar] [CrossRef]
- Ismael, M. A review on graphitic carbon nitride (g-C3N4) based nanocomposites: Synthesis, categories, and their application in photocatalysis. J. Alloy. Compd. 2020, 846, 156446. [Google Scholar] [CrossRef]
- Meramo-Hurtado, S.I.; González-Delgado, A.D. Application of techno-economic and sensitivity analyses as decision-making tools for assessing emerging large-scale technologies for production of chitosan-based adsorbents. ACS Omega 2020, 5, 17601–17610. [Google Scholar] [CrossRef]
- Aziz, F.; El Achaby, M.; Lissaneddine, A.; Aziz, K.; Ouazzani, N.; Mamouni, R.; Mandi, L. Composites with alginate beads: A novel design of nano-adsorbents impregnation for large-scale continuous flow wastewater treatment pilots. Saudi J. Biol. Sci. 2019, 27, 2499–2508. [Google Scholar] [CrossRef] [PubMed]


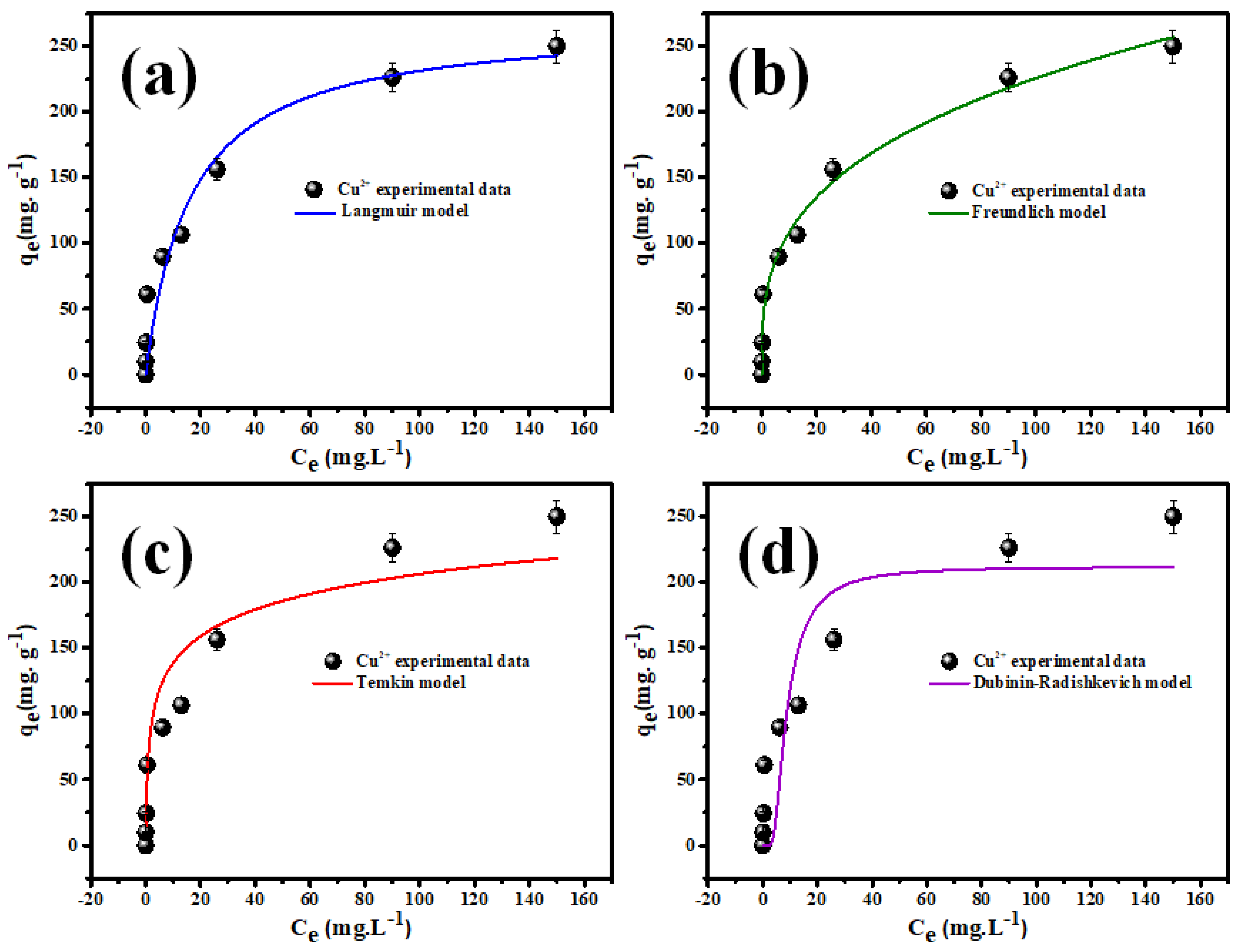



| Equilibrium Model | Parameters | Cu2+ |
|---|---|---|
| Langmuir | qm (mg·g−1) | 290.7 |
| KL (mg·g−1) | 0.028 | |
| RL (L.mg−1) | 0.125 | |
| R2 | 0.9965 | |
| Freundlich | n | 1.48 |
| KF (L.mg−1) | 11.36 | |
| R2 | 0.9644 | |
| Temkin | B (J.mol−1) | 85.65 |
| KT (L.mg−1) | 5.46 | |
| R2 | 0.9302 | |
| Dubinin-Radushkevich | β (mol2.J−2) | 2.0.95 × 10−8 |
| q (mg·g−1) | 186.32 | |
| E (J.mol−1) | 4886.5 | |
| R2 | 0.9833 |
| Pseudo-Second-Order Model | ||||||
|---|---|---|---|---|---|---|
| Cu2+ | qe(Exp) (mg·g−1) | t1/2 (min) | h0 (mg·g−1·min−1) | qe(Cal) (mg·g−1) | K2 × 104 (g·mg−1·min−1) | r2 |
| 89.75 ± 1.57 | 24.31 ± 0.86 | 3.57 ± 0.12 | 86.88 ± 1.23 | 4.73 ± 0.23 | 0.9978 | |
| Pseudo-First-order model | Elovich model | |||||
| qe(Cal) (mg·g−1) | K1 × 103 (min−1) | r2 | β × 102 (g·mg−1) | α | r2 | |
| Cu2+ | 37.66 ± 1.98 | 2.31 ± 0.18 | 0.9378 | 8.38 ± 0.18 | 21.52 ± 1.26 | 0.9422 |
| Adsorbents Used | Removal Capacity (mg/g) | Refs. |
|---|---|---|
| Alg + CNC | 53.4 | [59] |
| CNC/Sulfate (−SO3−) | 17.9 | [60] |
| CNF/Tempo | 49 | [60] |
| MWCNTs/Chitosan nanocomposite | 12.12 | [61] |
| Corn straw | 12.5 | [62] |
| Sewage sludge | 10.6 | [63] |
| Hardwood | 7.4 | [64] |
| Pristine biochar (saw dust char) | 16.1 | [65] |
| Spartina alternifora | 48.5 | [66] |
| GO/PEI | 150.9 | [67] |
| Fe3O4@SiO2@TiO2-APTMS | 50.5 | [68] |
| MgY2O4@g-C3N4 | 290.7 | Current study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Modwi, A.; Idriss, H.; Khezami, L.; Albadri, A.; Ismail, M.; Assadi, A.A.; Nguyen-Tri, P. Stripping of Cu Ion from Aquatic Media by Means of MgY2O4@g-C3N4 Nanomaterials. Water 2023, 15, 1188. https://doi.org/10.3390/w15061188
Modwi A, Idriss H, Khezami L, Albadri A, Ismail M, Assadi AA, Nguyen-Tri P. Stripping of Cu Ion from Aquatic Media by Means of MgY2O4@g-C3N4 Nanomaterials. Water. 2023; 15(6):1188. https://doi.org/10.3390/w15061188
Chicago/Turabian StyleModwi, Abueliz, Hajo Idriss, Lotfi Khezami, Abuzar Albadri, Mukhtar Ismail, Aymen Amine Assadi, and Phuong Nguyen-Tri. 2023. "Stripping of Cu Ion from Aquatic Media by Means of MgY2O4@g-C3N4 Nanomaterials" Water 15, no. 6: 1188. https://doi.org/10.3390/w15061188
APA StyleModwi, A., Idriss, H., Khezami, L., Albadri, A., Ismail, M., Assadi, A. A., & Nguyen-Tri, P. (2023). Stripping of Cu Ion from Aquatic Media by Means of MgY2O4@g-C3N4 Nanomaterials. Water, 15(6), 1188. https://doi.org/10.3390/w15061188









