The Mutual Effect of Microparticles and Antidepressants on the Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microparticles
2.2. Antidepressants
2.3. Liquid Chromatography Analyses (HPLC-PDA)
2.4. Microscopic Imaging
2.5. Preparation of S. ambiguum
2.6. Standard Toxicity Measurements
2.7. Ingestion Studies
2.8. Calculations and Statistics
3. Results
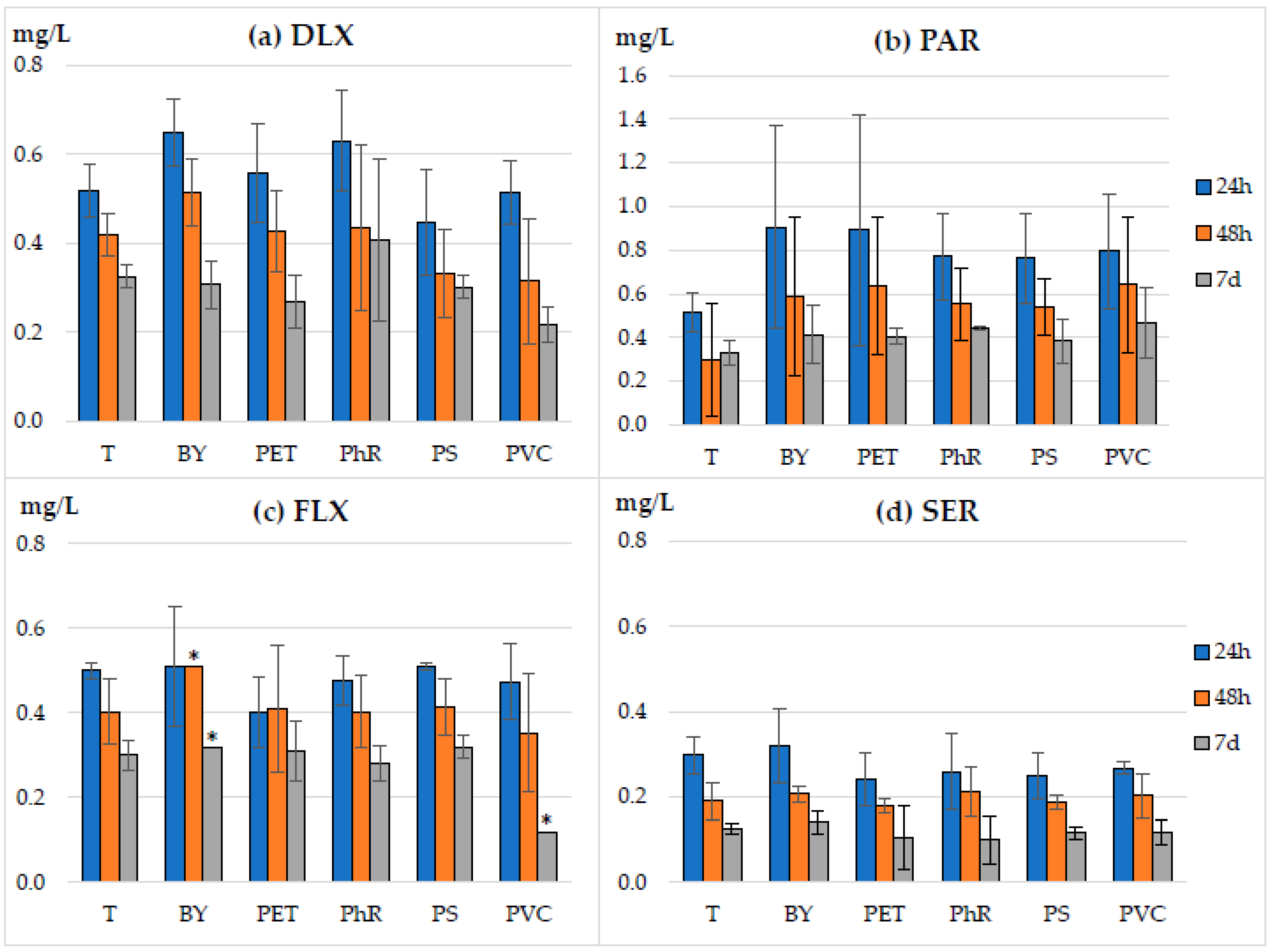
3.1. Sorption of Antidepressants on Microparticles
3.2. Influence of Microparticles on the Toxicity of Antidepressants Evaluated in the Spirotox Assay
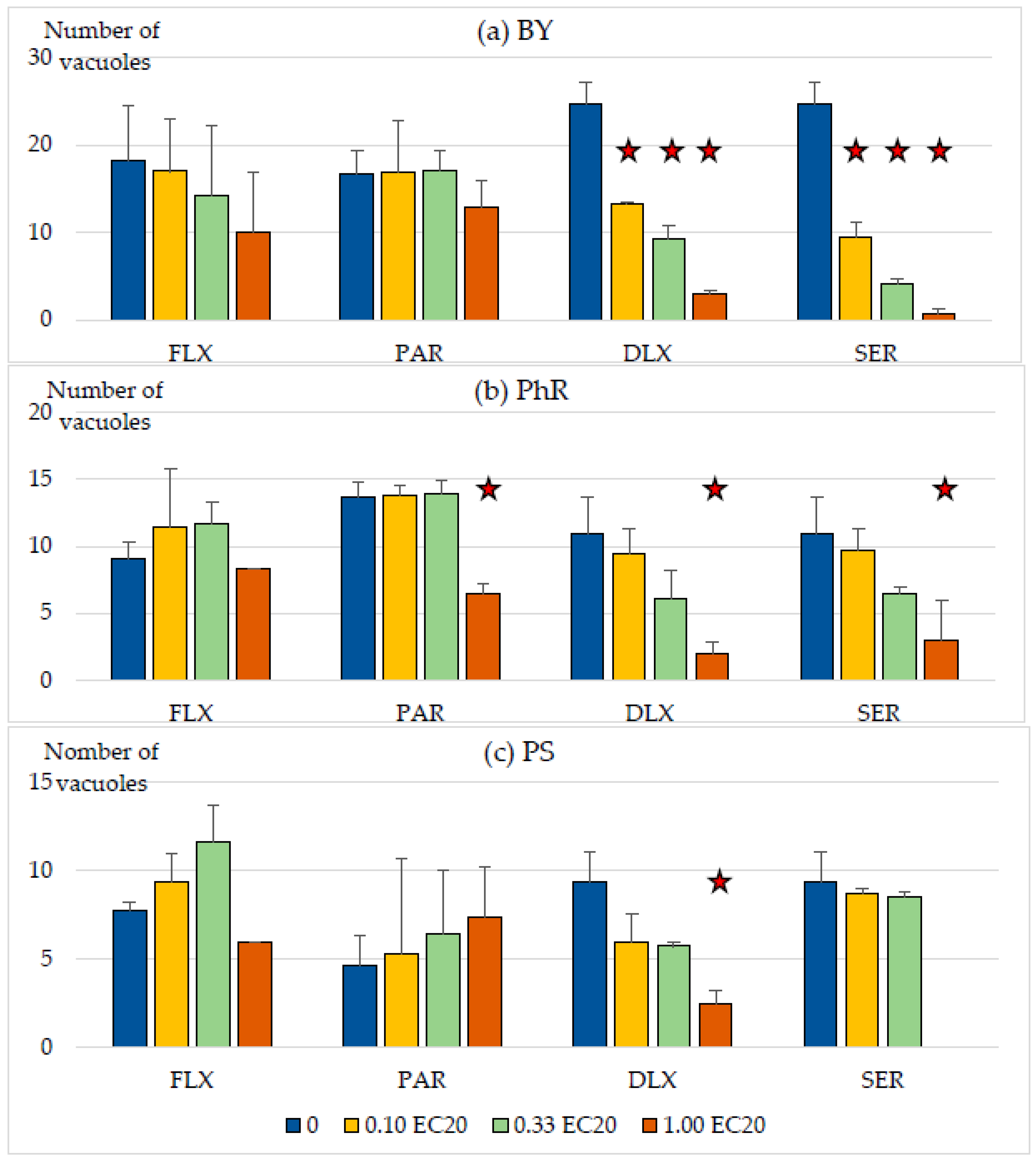
3.3. Influence of Low Levels of Antidepressants on the Formation of Vacuoles by S. ambiguum
4. Discussion
5. Conclusions and Recommendations
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Available online: www.clincalc.com (accessed on 29 August 2022).
- Lajeunesse, A.; Gagnon, C.; Sauve, S. Determination of basic antidepressants and their N-desmethyl metabolites in raw sewage and wastewater using solid-phase extraction and liquid chromatography-tandem mass spectrometry. Anal. Chem. 2008, 80, 5325–5333. [Google Scholar] [CrossRef] [PubMed]
- Schultz, M.M.; Furlong, E.T.; Kolpin, D.W.; Werner, S.L.; Schoenfuss, H.L.; Barber, L.B.; Blazer, V.S.; Norris, D.O.; Vajda, A.M. Antidepressant pharmaceuticals in two U.S. effluent-impacted streams: Occurrence and fate in water and sediment, and selective uptake in fish neural tissue. Environ. Sci. Technol. 2010, 44, 1918–1925. [Google Scholar] [CrossRef] [PubMed]
- Scircle, A.; Cizdziel, J.V.; Tisinger, L.; Anumol, T.; Robey, D. Occurrence of Microplastic Pollution at Oyster Reefs and Other Coastal Sites in the Mississippi Sound, USA: Impacts of Freshwater Inflows from Flooding. Toxics 2020, 8, 35. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.R.; Shashoua, Y.; Crawford, A.; Drury, A.; Sheppard, K.; Stewart, K.; Sculthorp, T. The Plastic Nile’: First Evidence of Microplastic Contamination in Fish from the Nile River (Cairo, Egypt). Toxics 2020, 8, 22. [Google Scholar] [CrossRef] [Green Version]
- Giebułtowicz, J.; Nałęcz-Jawecki, G. Occurrence of antidepressant residues in the sewage-impacted Vistula and Utrata rivers and in tap water in Warsaw (Poland). Ecotoxicol. Environ. Saf. 2014, 104, 103–109. [Google Scholar] [CrossRef]
- Grabicova, K.; Grabic, R.; Fedorova, G.; Fick, J.; Cerveny, D.; Kolarova, J.; Turek, J.; Zlabek, V.; Randak, T. Bioaccumulation of psychoactive pharmaceuticals in fish in an effluent dominated streams. Water Res. 2017, 124, 654–662. [Google Scholar] [CrossRef]
- Bojanowska-Czajka, A.; Pyszyńska, M.; Majkowska-Pilip, A.; Wawrowicz, K. Degradation of selected antidepressants sertraline and citalopram in ultrapure water and surface water using gamma radiation. Processes 2022, 10, 63. [Google Scholar] [CrossRef]
- Howard, P.H.; Muir, D.C.G. Identifying new persistent and bioaccumulative organics among chemicals in commerce II: Pharmaceuticals. Environ. Sci. Technol. 2011, 45, 6938–6946. [Google Scholar] [CrossRef]
- Puckowski, A.; Mioduszewska, K.; Łukaszewicz, P.; Borecka, M.; Caban, M.; Maszkowska, J.; Stepnowski, P. Bioaccumulation and analytics of pharmaceutical residues in the environment: A review. J. Pharm. Biomed. Anal. 2016, 127, 232–255. [Google Scholar] [CrossRef]
- Nałęcz-Jawecki, G.; Wawryniuk, M.; Giebułtowicz, J.; Olkowski, A.; Drobniewska, A. Influence of selected antidepressants on the ciliated protozoan Spirostomum ambiguum: Toxicity, bioaccumulation, and biotransformation products. Molecules 2020, 25, 1476. [Google Scholar] [CrossRef]
- Silva, L.J.G.; Pereira, A.; Meisel, L.M.; Lino, C.M.; Pena, A. Reviewing the serotonin reuptake inhibitors (SSRIs) footprint in the aquatic biota: Uptake, bioaccumulation and ecotoxicology. Environ. Pollut. 2015, 197, 127–143. [Google Scholar] [CrossRef] [PubMed]
- Akdogan, Z.; Guven, B. Microplastics in the environment: A critical review of current understanding and identification of future research needs. Environ. Pollut. 2019, 254, 113011. [Google Scholar] [CrossRef] [PubMed]
- De Hann, W.P.; Sanchez-Vidal, A.; Canalas, M. Floating the microplastics and aggregate formation in the Western Mediterranean Sea Marine Pollut. Bull. 2019, 140, 523–535. [Google Scholar] [CrossRef]
- Hidalgo-Ruz, V.; Gutow, L.; Thompson, R.C.; Thiel, M. Microplastics in the Marine Environment: A Review of the Methods Used for Identification and Quantification. Environ. Sci. Technol. 2012, 46, 3060–3075. [Google Scholar] [CrossRef] [PubMed]
- Abeynayaka, A.; Werellagama, I.; Ngoc-Bao, P.; Hengesbaugh, M.; Gajanayake, P.; Nallaperuma, B.; Karkour, S.; Bui, X.T.; Itsubo, N. Microplastics in Wastewater Treatment Plants. In Current Developments in Biotechnology and Bioengineering; Elsevier: Amsterdam, The Netherlands, 2022; pp. 311–337. [Google Scholar] [CrossRef]
- Rehse, S.; Kloas, W.; Zarfl, C. Short-term exposure with high concentrations of pristine microplastic particles leads to immobilisation of Daphnia magna. Chemosphere 2016, 153, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Kaposi, K.L.; Mos, B.; Kelaher, B.P.; Dworjanyn, S.A. Ingestion of microplastic has limited impact on a marine larva. Environ. Sci. Technol. 2014, 48, 1636–1645. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y.; Huang, A.; Cao, S.; Sun, F.; Wang, L.; Guo, H. Effects of nanoplastics and microplastics on toxicity, bioaccumulation, and environmental fate of phenanthrene in fresh water. Environ. Pollut. 2016, 219, 166–173. [Google Scholar] [CrossRef]
- Teuten, E.L.; Rowland, S.J.; Galloway, T.S.; Thompson, R.C. Potential for plastics to transport hydrophobic contaminants. Environ. Sci. Technol. 2007, 41, 7759–7764. [Google Scholar] [CrossRef]
- Hartmann, N.B.; Rist, S.; Bodin, J.; Jensen, L.H.S.; Schmidt, S.N.; Mayer, P.; Meibom, A.; Baun, A. Microplastics as vectors for environmental contaminants: Exploring sorption, desorption, and transfer to biota. Integr. Environ. Assess. Manag. 2017, 13, 488–493. [Google Scholar] [CrossRef]
- Ma, J.; Zhao, J.; Zhu, Z.; Li, L.; Yu, F. Effect of microplastic size on the adsorption behavior and mechanism of triclosan on polyvinyl chloride. Environ. Pollut. 2019, 254, 113104. [Google Scholar] [CrossRef]
- Liu, G.; Zhu, Z.; Yang, Y.; Sun, Y.; Yu, F.; Ma, J. Sorption behavior and mechanism of hydrophilic organic chemicals to virgin and aged microplastics in freshwater and seawater. Environ. Pollut. 2019, 246, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Frydkjær, C.K.; Iversen, N.; Roslev, P. Ingestion and Egestion of Microplastics by the Cladoceran Daphnia magna: Effects of Regular and Irregular Shaped Plastic and Sorbed Phenanthrene. Bull. Environ. Contam. Toxicol. 2017, 99, 655–661. [Google Scholar] [CrossRef]
- Wei, S.; Yu, H.; Shuge, S.; Yu, T.; Weishang, Z.; Xueying, D.; Guangxu, L. Immunotoxicities of microplastics and sertraline, alone and in combination, to a bivalve species: Size-dependent interaction and potential toxication mechanism. J. Hazard. Mater. 2020, 396, 122603. [Google Scholar] [CrossRef]
- Dürichen, H.; Siegmund, L.; Burmester, A.; Fischer, M.S.; Wöstemeyer, J. Ingestion and digestion studies in Tetrahymena pyriformis based on chemically modified microparticles. Eur. J. Protistol. 2016, 52, 45–57. [Google Scholar] [CrossRef] [PubMed]
- Razanajatovo, R.M.; Ding, J.; Zhang, S.; Jiang, H.; Zou, H. Sorption and desorption of selected pharmaceuticals by polyethylene microplastics. Mar. Pollut. Bull. 2018, 136, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Christaki, U.; Dolan, J.R.; Pelegri, S.; Rassoulzadegan, F. Consumption of picoplankton-size particles by marine ciliates: Effects of physiological state of the ciliate and particle quality. Limnol. Oceanogr. 2003, 43, 458–464. [Google Scholar] [CrossRef]
- Nałęcz-Jawecki, G. Spirotox—Spirostomum ambiguum acute toxicity test—10 years of experience. Environ. Toxicol. 2004, 19, 359–364. [Google Scholar] [CrossRef]
- Nałęcz-Jawecki, G.; Chojnacka, J.; Wawryniuk, M.; Drobniewska, A. Influence of nano- and small microplastics on ciliated protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835. Water 2021, 13, 2857. [Google Scholar] [CrossRef]
- Csaba, G. Presence in and effects of pineal indoleamines at very low level of phylogeny. Experientia 1993, 49, 627–634. [Google Scholar] [CrossRef]
- Csaba, G.; Kovacs, P.; Pallinger, E. How does the unicellular Tetrahymena utilize the hormones that it produces? Paying a visit to the realm of atto- and zeptomolar concentrations. Cell Tissue Res. 2007, 327, 199–203. [Google Scholar] [CrossRef]
- Ogonowski, M.; Schür, C.; Jarsen, A.; Gorokhova, E. The effects of natural and anthropogenetic microparticles on individual fitness in Daphnia magna. PLoS ONE 2016, 11, e01550632016. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nałęcz-Jawecki, G.; Sawicki, J. Toxicity of inorganic compounds in the Spirotox test—A miniaturized version of the Spirostomum ambiguum test. Arch. Environ. Contam. Toxicol. 1998, 34, 1–5. [Google Scholar] [CrossRef]
- Trawiński, J.; Skibiński, R. Studies on photodegradation process of psychotropic drugs: A review. Environ. Sci. Pollut. Res. 2017, 24, 1152–1199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meredith-Williams, M.; Carter, L.J.; Fussell, R.; Raffaelli, D.; Ashauer, R.; Boxall, A.B. Uptake and depuration of pharmaceuticals in aquatic invertebrates. Environ. Pollut. 2012, 165, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Kucharski, D.; Nałęcz-Jawecki, G.; Drzewicz, P.; Skowronek, A.; Mianowicz, K.; Strzelecka, A.; Giebułtowicz, J. The assessment of environmental risk related to the occurrence of pharmaceuticals in bottom sediments of the Odra River estuary (SW Baltic Sea). Sci. Total Environ. 2022, 828, 154446. [Google Scholar] [CrossRef]
- Puckowski, A.; Ćwięk, W.; Mioduszewska, K.; Stepnowski, P.; Białk-Bielińska, A. Sorption of pharmaceuticals on the surface of microplastics. Chemosphere 2021, 263, 127976. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Zhang, M.; Sha, W.; Wang, Y.; Hao, H.; Dou, Y.; Li, Y. Sorption behavior and mechanisms of organic contaminants to nano and microplastics. Molecules 2020, 25, 1827. [Google Scholar] [CrossRef]
- Atugoda, T.; Vithanage, M.; Wijesekara, H.; Bolan, N.; Sarmah, A.K.; Bank, M.S.; You, S.; Ok, Y.S. Interactions between microplastics, pharmaceuticals and personal care products: Implications for vector transport. Environ. Int. 2021, 149, 106367. [Google Scholar] [CrossRef]
- Rhodes, C.J. Plastic pollution and potential solutions. Sci. Prog. 2018, 101, 207–226. [Google Scholar] [CrossRef]
- Koelmans, A.A.; Nor, N.H.M.; Hermsen, E.; Kooi, M.; Mintenig, S.M.; De France, J. Microplastics in Freshwaters and Drinking Water: Critical Review and Assessment of Data Quality. Water Res. 2019, 155, 410–422. [Google Scholar] [CrossRef] [PubMed]
- Schmieg, H.; Burmester, J.K.Y.; Krais, S.; Ruhl, A.S.; Tisler, S.; Zwiener, C.; Köhler, H.R.; Triebskorn, R. Interacting effects of polystyrene microplastics and the antidepressant amitriptyline on early life stages of brown trout (Salmo trutta f. fario). Water 2020, 12, 2361. [Google Scholar] [CrossRef]
- Zhang, S.; Ding, J.; Razanajatovo, R.M.; Jiang, H.; Zou, H.; Zhu, W. Interactive effects of polystyrene microplastics and roxithromycin on bioaccumulation and biochemical status in the freshwater fish red tilapia (Oreochromis niloticus). Sci. Total Environ. 2019, 648, 1431–1439. [Google Scholar] [CrossRef] [PubMed]
- Wloga, G.; Frankel, J. From molecules to morphology: Cellular organization of Tetrahymena thermophila. Methods Cell Biol. 2012, 109, 83–140. [Google Scholar] [CrossRef] [PubMed]
- Ishida, M.; Allen, R.D.; Fok, A.K. Phagosome formation in Paramecium: Roles of somatic and oral cilia and of solid particles as revealed by video microscopy. J. Eucaryot. Microbiol. 2001, 48, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Buduma, N.; Balabanian, J.; Dalvi, P.; Chia, S.; Dhaliwal, A.; Eliya, D.; Boothby, J.; Bros-Seemann, S.; Kibler, R.; Khuri, S.; et al. Modulation of phagocytosis in Tetrahymena thermophila by histamine and the antihistamine diphenhydramine. Acta Protozool. 2013, 52, 317–323. [Google Scholar] [CrossRef]
- Santos, L.H.M.L.M.; Rodríguez-Mozaz, S.; Barceló, D. Microplastics as vectors of pharmaceuticals in aquatic organisms—An overview of their environmental implications. CSCEE 2021, 3, 100079. [Google Scholar] [CrossRef]
- Csaba, G. The hormonal system of the unicellular Tetrahymena: A review with evolutionary aspects. A Microbiol. Immunol. Hung. 2012, 59, 131–156. [Google Scholar] [CrossRef]



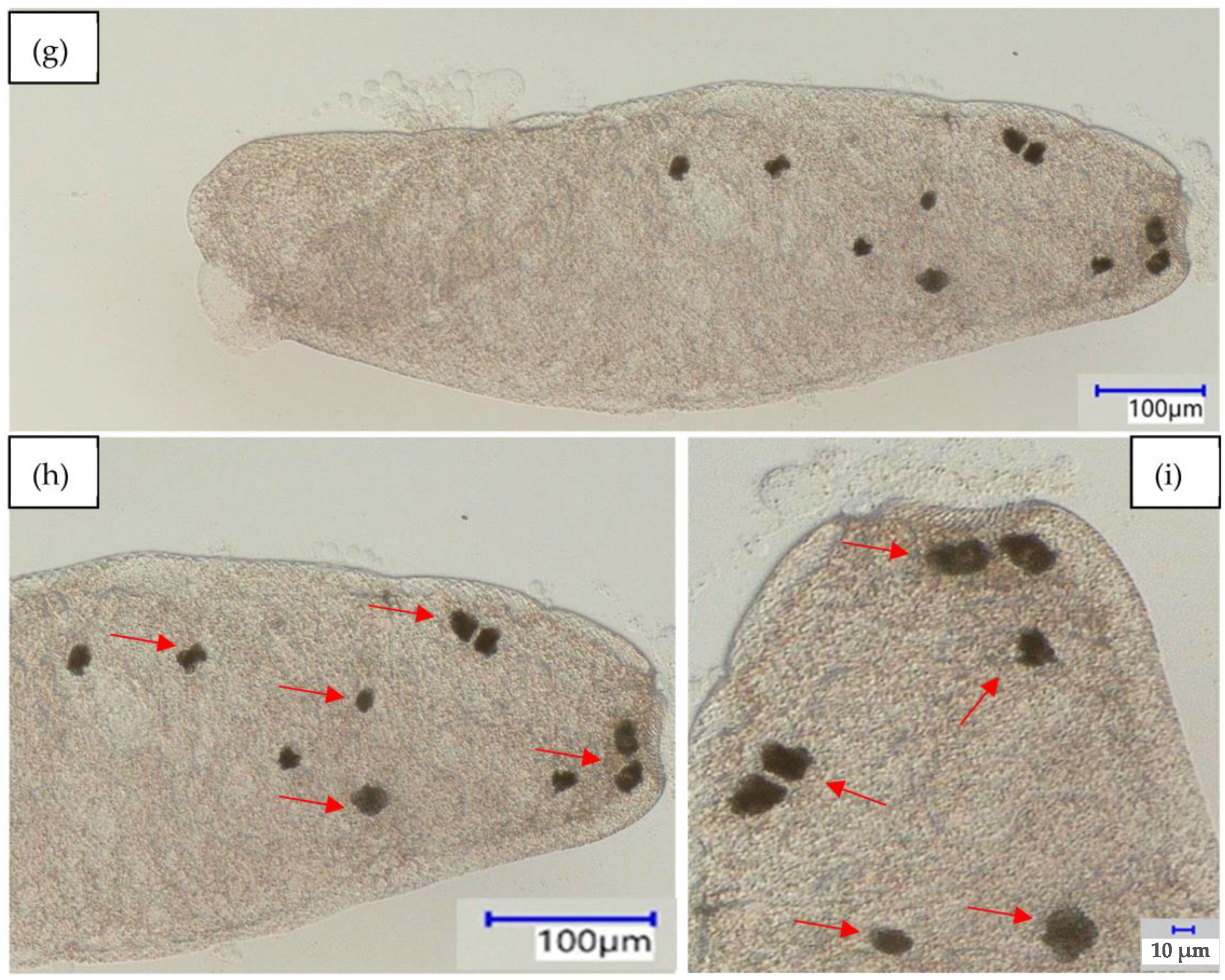
 enlargement and mark food vacuoles (b,c) baker’s yeast; (e,f) phenolic resin microparticles; and (h,i) polystyrene microparticles.
enlargement and mark food vacuoles (b,c) baker’s yeast; (e,f) phenolic resin microparticles; and (h,i) polystyrene microparticles.
 enlargement and mark food vacuoles (b,c) baker’s yeast; (e,f) phenolic resin microparticles; and (h,i) polystyrene microparticles.
enlargement and mark food vacuoles (b,c) baker’s yeast; (e,f) phenolic resin microparticles; and (h,i) polystyrene microparticles.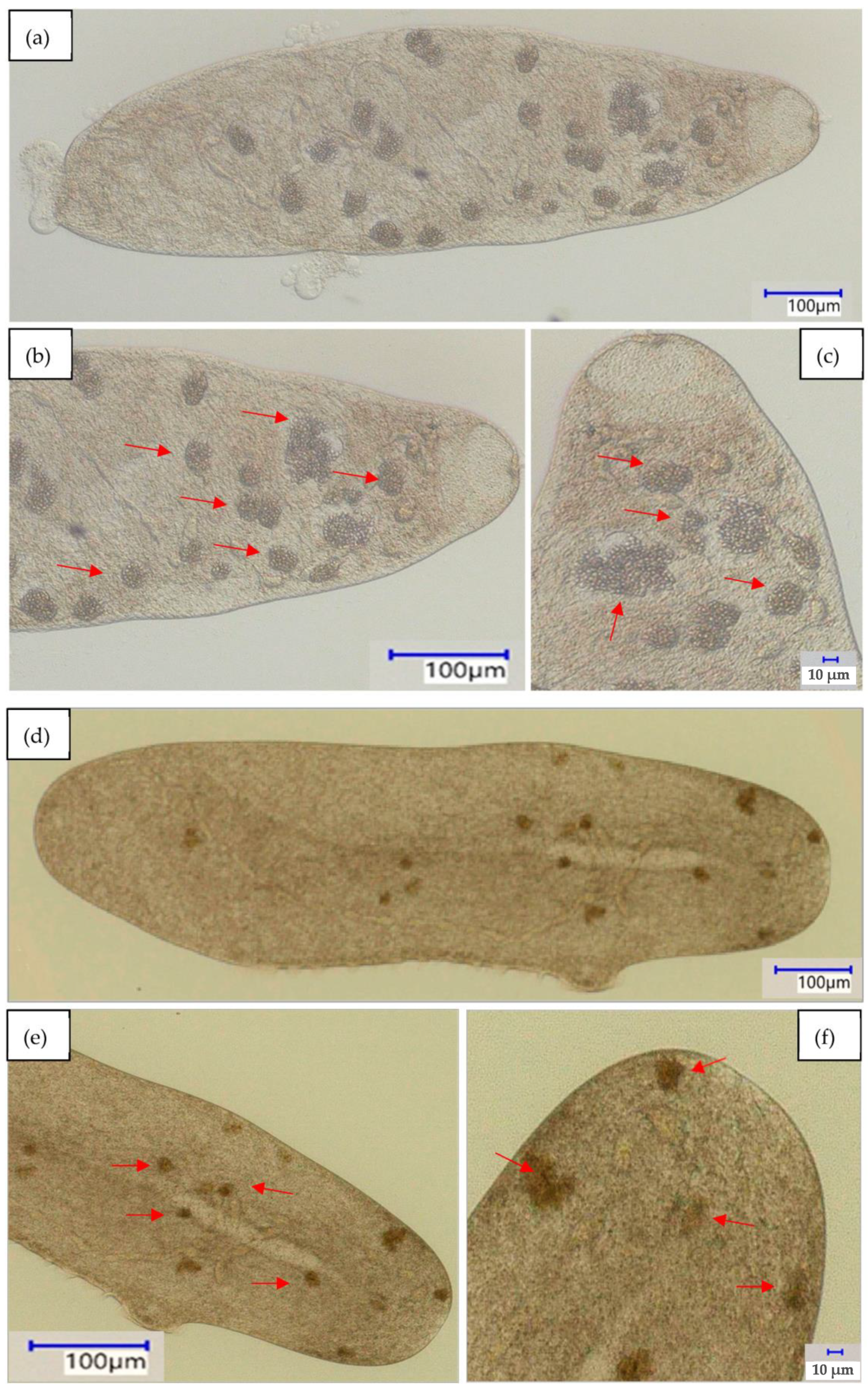
| Pharmaceutical | 1.00 EC20 [mg L−1] | 0.33-EC20 [mg L−1] | 0.10-EC20 [mg L−1] |
|---|---|---|---|
| Sertraline | 0.25 | 0.076 | 0.025 |
| Duloxetine | 0.63 | 0.19 | 0.063 |
| Fluoxetine | 0.65 | 0.20 | 0.064 |
| Paroxetine | 0.88 | 0.26 | 0.088 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chojnacka, J.; Drobniewska, A.; Lenga, W.; Misztal, J.; Wawryniuk, M.; Nałęcz-Jawecki, G. The Mutual Effect of Microparticles and Antidepressants on the Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835. Water 2023, 15, 552. https://doi.org/10.3390/w15030552
Chojnacka J, Drobniewska A, Lenga W, Misztal J, Wawryniuk M, Nałęcz-Jawecki G. The Mutual Effect of Microparticles and Antidepressants on the Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835. Water. 2023; 15(3):552. https://doi.org/10.3390/w15030552
Chicago/Turabian StyleChojnacka, Justyna, Agata Drobniewska, Wioletta Lenga, Joanna Misztal, Milena Wawryniuk, and Grzegorz Nałęcz-Jawecki. 2023. "The Mutual Effect of Microparticles and Antidepressants on the Protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835" Water 15, no. 3: 552. https://doi.org/10.3390/w15030552






