Highlights
What are the main finding?
- Spirulina platensis was applied to treat mariculture wastewater for N and P removal.
- Microalgal aggregates were formed under air velocity of 1.75 cm/s or higher.
What is the implication of the main finding?
- Microalgal aggregation was promoted, which was attributed to the protein contents in TB-EPS.
- P accumulation in biomass was enhanced accompanied by the aggregation process.
Abstract
As an important alternative to alleviate the shortage of wild fishery resources, mariculture is facing increasing challenges on the wastewater treatment, mainly due to the salinity brought from seawater and low nutrient concentration. In this study, Spirulina platensis (S. platensis) was adopted as the target algae stain for synthetic mariculture wastewater treatment, which exhibited excellent adaptability to high-saline wastewater during a 40 days’ adaptive culture. Then, the microalgae stain was inoculated into photo-bioreactors with different uplift airflow velocity (UAV) to achieve microalgal aggregation. After 10 days of pre-cultivation and a 30-day granulation process, the maximum nutrient removal rates by S. platensis were 86.5% of TN (to 3.4 mg/L), 98.1% of TP (to 0.1 mg/L) and 95.8% of DOC (to 5.5 mg/L), with 3.5 g/L of biomass content in synthetic wastewater. Analysis of phosphorus and EPS content showed that higher protein content in tightly bound EPS and enhanced P accumulation was accompanied with the microalgal aggregation processes, and P was mainly distributed in the residual rather than the EPS part of microalgal aggregates, indicating that the development of aggregates from suspension S. platensis would benefit not only biomass separation, but also phosphorus recovery, being a potential treatment for simultaneously removing nutrients and recovering mariculture wastewater.
1. Introduction
Accompanying the decrease in wild fishery resources and the increasing demand for seafood, is the rapid development of the mariculture industry, which has generated large amounts of saline wastewater (referred to as mariculture wastewater), which is rich in nutrients, such as carbon, nitrogen, and phosphorus, that may lead to the eutrophication of water bodies without treatment and cause serious damage to the aquatic resources [1]. This necessitates the development of sustainable technologies for mariculture wastewater treatment to maintain the sustainability of aquatic environments [2].
Compared to physicochemical technologies, the biological process is considered a desirable and economic option for mariculture wastewater. However, the characteristics of this type of wastewater, such as high salinity, low carbon/nitrogen and insufficient nutrients for microbial growth, have become the main challenges for biological mariculture wastewater treatment, resulting in ineffective nutrient removal, especially for N [3,4]. Therefore, alternative salt-tolerant biological processes should be developed in the mariculture wastewater treatment.
Biotreatments utilizing photosynthetic organisms, e.g., microalgae-systems, as technologies capable of treating wastewater without additional carbon sources, have recently been proposed to be of excellent economical value and environmental potential [5,6]. As a salt-tolerant organism, Spirulina platensis (S. platensis) possesses admirable performance in N recovery, superior efficiency of CO2 capture, which is 10 times that of terrestrial plants [7], and contains high contents of proteins, polysaccharides, essential fatty acids, and vitamins as well [8,9]. With respect to wastewater treatment, municipal wastewater [10], wastewater of monosodium factory [11], and toilet flushing wastewater [12] have been used for S. platensis cultivation with effective carbon and nutrients removal. However, S. platensis was usually cultured in suspension to achieve high biomass production and nutrient assimilation, while biomass separation after wastewater treatment became a great challenge due to the poor settling properties of suspended algal cells, restricting its large-scale application. As reported, harvesting of microalgae biomass accounts for more than 20–30% of the total cultivation expenses [13].
Microalgal aggregation would be an alternative that can solve this dilemma. The formation of dense aggregates or granules through the aggregation of bacteria and microalgae through extracellular polymers enables the excellent settling property of this type of algal–bacterial consortium [14]. The generated photogranules consist of phototrophs (microalgae and cyanobacteria) and other non-phototrophic microorganisms (nitrifiers and denitrifiers) that can remove carbon, nitrogen, and phosphorus simultaneously from wastewater [15]. Meanwhile, a considerable part of nitrogen and phosphorus were assimilated by algae for photosynthesis, rather than being converted into N2 via nitrifying–denitrifying processes, bringing about not only the effective removal of nutrients from wastewater, but also a high potential of resource recovery, such as alginate-like exopolymers (ALE) and lipids [4,6].
Currently, granulation can be achieved successfully from single-cell microalgae, e.g., Ankistrodesmus falcatus, in lab-scale airlift sequential batch reactors (SBRs) with the target microalgae strain dominating the biological communities, and shear force in airlift reactors provided by aeration is considered the pivotal factor affecting the formation of this type of microalgal aggregates [14,15,16]. Nevertheless, very limited information is available in the optimal conditions for microalgal granulation from single-strain microalgae, the underlying mechanisms for nutrients removal, as well as value-added product recovery. Accordingly, the present study aims at investigating the potential of a value-adding algal strain, i.e., S. platensis, in treating mariculture wastewater with the development of microalgal aggregates. The outcome of this study will provide a new perspective on microalgal-aggregate cultivation to accomplish efficient mariculture wastewater treatment.
2. Materials and Methods
2.1. Algal Culture and Synthetic Mariculture Wastewater
The target microalgae S. platensis purchased from Shanghai Guangyu Biotechnology Co., Ltd. (Shanghai, China) was used in this study. Stock algae strains were grown in 1.0 L flasks under a complete cycle of 12:12 h light/dark periods at a controlled temperature of 25 ± 0.5 °C. Light source was supplied by a LED (light emitting diode) lamp, with the light intensity of 80 μmol/m2/s at the flasks’ surface [17]. The growth media (Zarrouk medium, ZM) was prepared with deionized water (DW), sterilized at 121 °C for 30 min with the composition as following (g/L): 16.8 NaHCO3, 2.5 NaNO3, 0.5 K2HPO4, 1.0 K2SO4, 1.0 NaCl, 0.2 MgSO4.7H2O, 0.04 CaCl2.2H2O, 0.01 FeSO4.7H2O and 0.08 EDTA, the pH was controlled to be around 7.5 by using 1 M NaOH or 1 N HCl [8,18].
The synthetic mariculture wastewater (SMW) was prepared according to a previous study [19], consisting of a 250 mg/L chemical oxygen demand (COD), 3 mg/L PO43−-P, and 26 mg/L NH4+-N prepared from sodium acetate, KH2PO4 and NH4Cl, respectively, in addition to 3.2% NaCl, 250 mg/L NaHCO3, 75 mg/L MgSO4·7H2O, 36 mg/L CaCl2·2H2O, 50 mg/L FeSO4·7H2O, and 1 mL/L trace elements solution (2.86 g/L H3BO3, 1.86 g/L MnCl2·4H2O, 0.22 g/L Na2MoO4·2H2O, 0.08 g/L CuSO4·5H2O, 0.05 g/L Co(NO3)2·6H2O, 0.021 g/L ZnSO4·7H2O). The pH of the SMW was controlled to be around 7.5.
Before the granulation processes, an adaptive cultivation was conducted for 40 days (four culture cycles and 10 days for each) to enhance the adaptation of S. platensis to SMW used in this study. The batch experiments were conducted in 1.0 L sterile conical flasks with the supplement of SMW with different salinity, and the cultures consisted of 400 mL synthetic wastewater and 100 mL preculture algae. Firstly, S. platensis grown in ZM was inoculated into SMW with the NaCl concentration of 1.0 g/L, which was consistent with that in ZM. When cells were grown to the exponential phase after 10 days of cultivation (first culture cycle), the microalgae were inoculated into SMW with NaCl concentration of 3.2 g/L and cultivated for 30 days (microalgae were inoculated into a new medium every 10 days) to ensure the adaptation of this algal strain to high saline SMW.
2.2. Experimental Set-Up for Microalgal Aggregation
Three identical lab-scale cylindrical SBRs (50 mm in diameter) with the effective working volume of 790 mL were used in this work to study the effect of aeration intensity on algal granulation from S. platensis. These SBRs were operated at room temperature (25 ± 2 °C) and illuminated under four LED lamps (80 μmol/m2/s) (15h light per day).
Prior to the start of the experiments, the axenic Spirulina platensis at its logarithmic growth were taken from ZM by centrifuging the bulk of algal suspension at 10,000 rpm for 10 min; after discharging the supernatant, the pellet was washed by DW twice. Then, the separated S. platensis was resuspended and pre-cultivated in the SMW for the 10 days of pre-cultivation to achieve rapid adaptation and biomass accumulation. During the pre-cultivation period, non-aeration and aeration phases were alternatively operated every 3 h to enhance the settleability of suspended S. platensis, and the SMW was exchanged every 12 h manually. After pre-cultivation, the initial concentration of algal biomass in reactors was approximately 1.28 g/L. Then, the 12 h cycles were operated automatically, including 2 min filling, 600 min aeration/non-aeration phase (120 min of aeration and 120 min of non-aeration was operated alternatively), 116 min settling (the settling time was shortened gradually from 120 to 10 min according to the settleability of algal biomass in reactors), 1 min discharging and 1 min idling. Air was introduced by an air diffuser placed on the bottom of each reactor with air pumps, and the uplift airflow velocity was controlled at 0.65, 1.75, 2.75 cm/s by air flowmeters for reactors R1, R2, and R3, respectively. Throughout the whole experiment, the hydraulic retention time (HRT) was 24 h with the exchange ratio of 50%; sludge retention time (SRT) was around 20 days, discharging of 80 mL of mixed liquor every two days.
2.3. Analytical Methods
During the operation period, influent and effluent samples were taken from the bioreactors to determine nutrient removal efficiency. After filtration through 0.22 μm membrane filters, the water samples were utilized to determine the concentration of NH4+-N, NO2−-N, NO3−-N and PO43−-P, according to the standard method [20]. Total dissolved organic carbon (DOC) and dissolved inorganic carbon (DIC) were analyzed by a total organic carbon (TOC) analyzer (TOC-VCSN, Shimadzu, Kyoto, Japan). To evaluate the characteristics of algal biomass; the quantification of total mixed liquor suspended solids (TS), mixed liquor volatile suspended solids (VS), sludge volume index at 5 or 30 min (SVI5 and SVI30), and chlorophyll-a (Chl-a) in biomass was conducted in accordance with the standard methods [20].
To analyze the extracellular polymeric substances’ (EPS) content of algal biomass, 20 mL mixed liquor taken from reactors at the end of the aeration period in each cycle and centrifuged at 3000 rpm for 10 min to separate the solid part, the pellet was resuspended in 0.9% NaCl solution and homogenized by a glass homogenizer, then the mixture was centrifuged at 7500 rpm for 20 min and the supernatant filtered through a 0.22 μm membrane filter for the determination of loosely bonded EPS (LB-EPS). The residue left was further suspended in 0.9% NaCl solution and heated under 80 °C for 30 min, then centrifuged at 8500 rpm for 20 min. The supernatant was collected and filtered through a 0.22 μm membrane filter for the determination of tightly bound EPS (TB-EPS). The content of polysaccharides (PS) and proteins (PN) in the LB-EPS and TB-EPS were determined by the phenol-sulfuric acid and the Lowry–Folin methods, respectively [21,22]. P content in EPS or intact algal–bacterial was analyzed according to the standards, measurements and testing (SMT) protocol [16]. The surface characteristics of the microalgal aggregates generated from S. platensis were recorded by a stereomicroscope (Olympus, Tokyo, Japan) and a scanning electron microscope (SEM) (Zeiss, Jena, Germany).
3. Results
3.1. Adaptation of S. platensis to Salinity-Stress
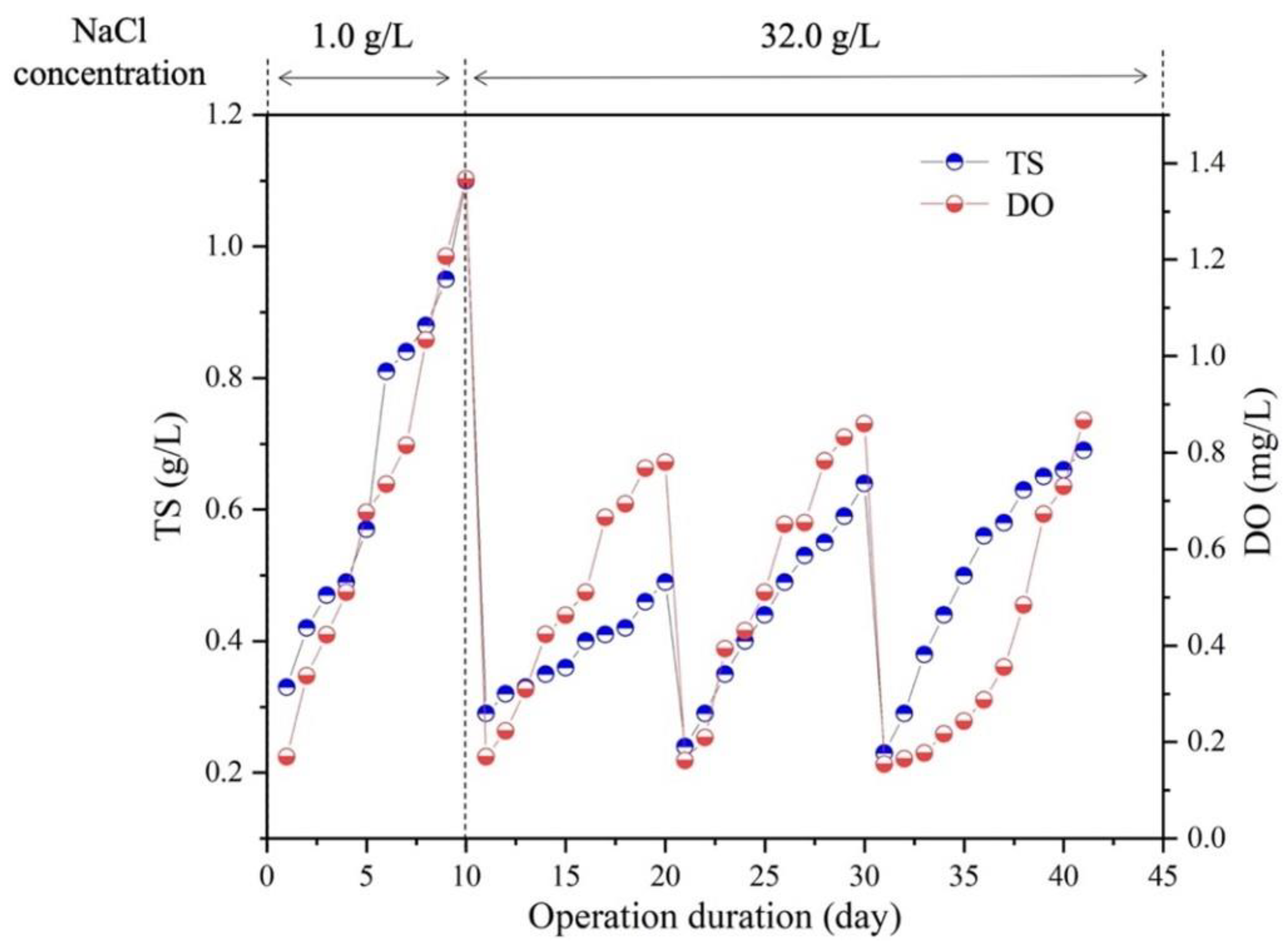
In this study, the purchased S. platensis was cultivated in ZM with NaCl concentration of 1.0 g/L, to enhance its tolerance ability to higher salinity of 3.2% NaCl in SMW, an adaptive culture of 40 days was conducted as depicted in Figure 1. The results showed that in the first culture cycle, the microalgae could grow well in SMW under the salinities of 1.0 g/L NaCl (Figure 1), algal biomass increased exponentially with the maximum biomass concentration of 1.18 g/L achieved on day 10, which was close to that in ZM. When NaCl content was increased to 3.2%, algal growth decreased significantly during the second 10 days’ cultivation, while recovered slightly in the third culture cycle. Then, in the last culture cycle of day 30 to 40, the maximum biomass concentration of 0.78 g/L was achieved at the end. Consistent with algal growth, the change of dissolved oxygen (DO) concentration of all the groups showed similar trends with the growth of algal biomass, which was 0.9 mg/L in the end of adaptive cultivation. The above results indicated that S. platensis was able to adapt to a higher salinity environment as 3.2% NaCl after a short time acclimation, suggesting the potential of S. platensis for effective SMW treatment.
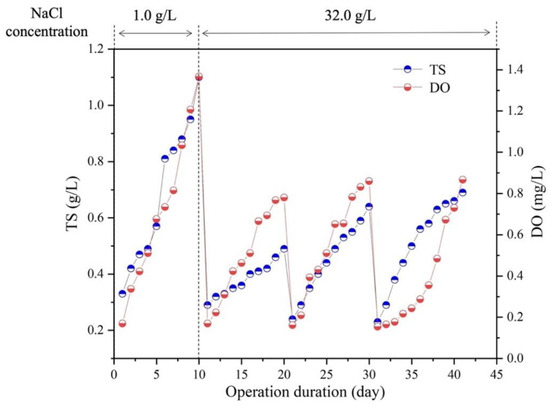
Figure 1.
Performance of microalgal growth and oxygen production in synthetic mariculture wastewater. All experiments were operated under a 12/12 h light/dark cycle and illumination of 80 μmol/m2/s at 25 °C.
3.2. Formation of Microalgal Aggregates
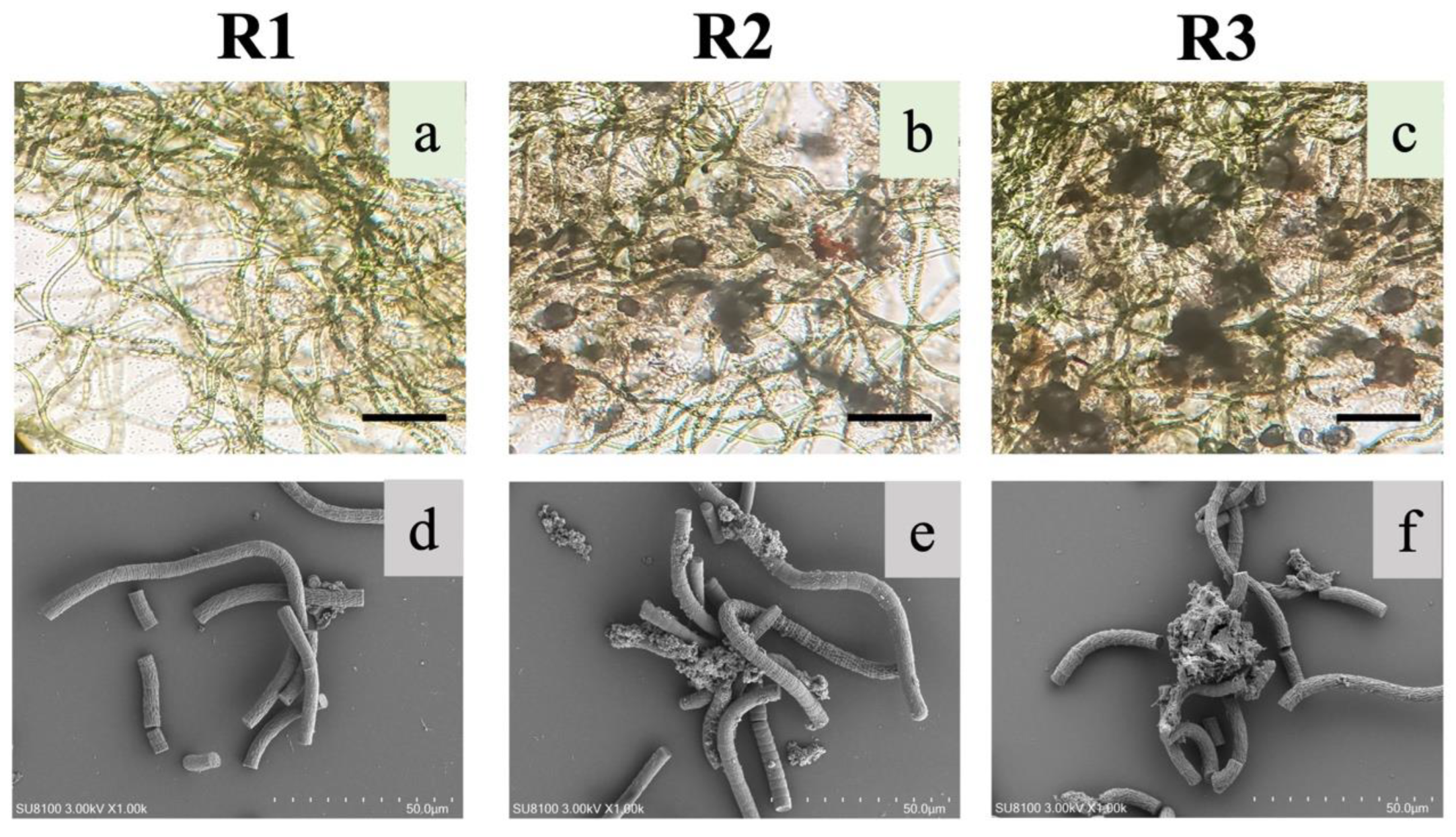
In this study, S. platensis, after 40 days of adaptive cultivation, was inoculated into three photo-bioreactors; after pre-cultivation (described in Section 2.2) and adjustments, biomass concentration in reactors was around 1.28 g/L, with SVI30 of 781.3 mL/g and Chl-a content of 1.23 mg/g-VS. Then, the seeded microalgae were cultivated in SBRs with different aeration intensity (UAV) of 0.65, 1.75, 2.75 cm/s in R1, R2 and R3, respectively. After the 30-day operation, microalgal aggregates were formed in R3 with diameter around 100 μm, as shown in Figure 2c. Relatively smaller aggregates could be observed in R2 with loose and irregular structure, while S. platensis in R1 was still filamentous, with less microalgal flocs and more free microalgae (Figure 2a,c). From the images observed from SEM, aggregates formed in R3 were consisted of both S. platensis and bacteria (rod- or cocci-shaped).
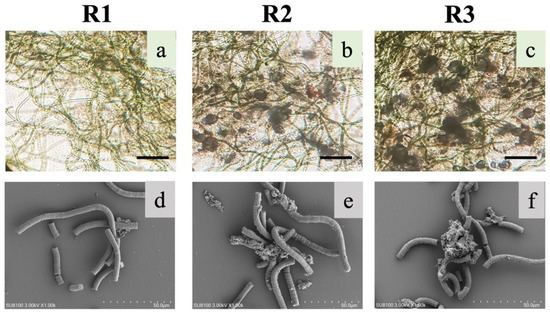
Figure 2.
Images of suspension S. platensis and microalgal aggregates in bioreactor R1, R2, and R3 obtained with light microscope (a–c) or SEM (d–f). Scale bar for panels is (a–c) 100 μm (solid lines) and (d–f) 50 μm (dotted lines).
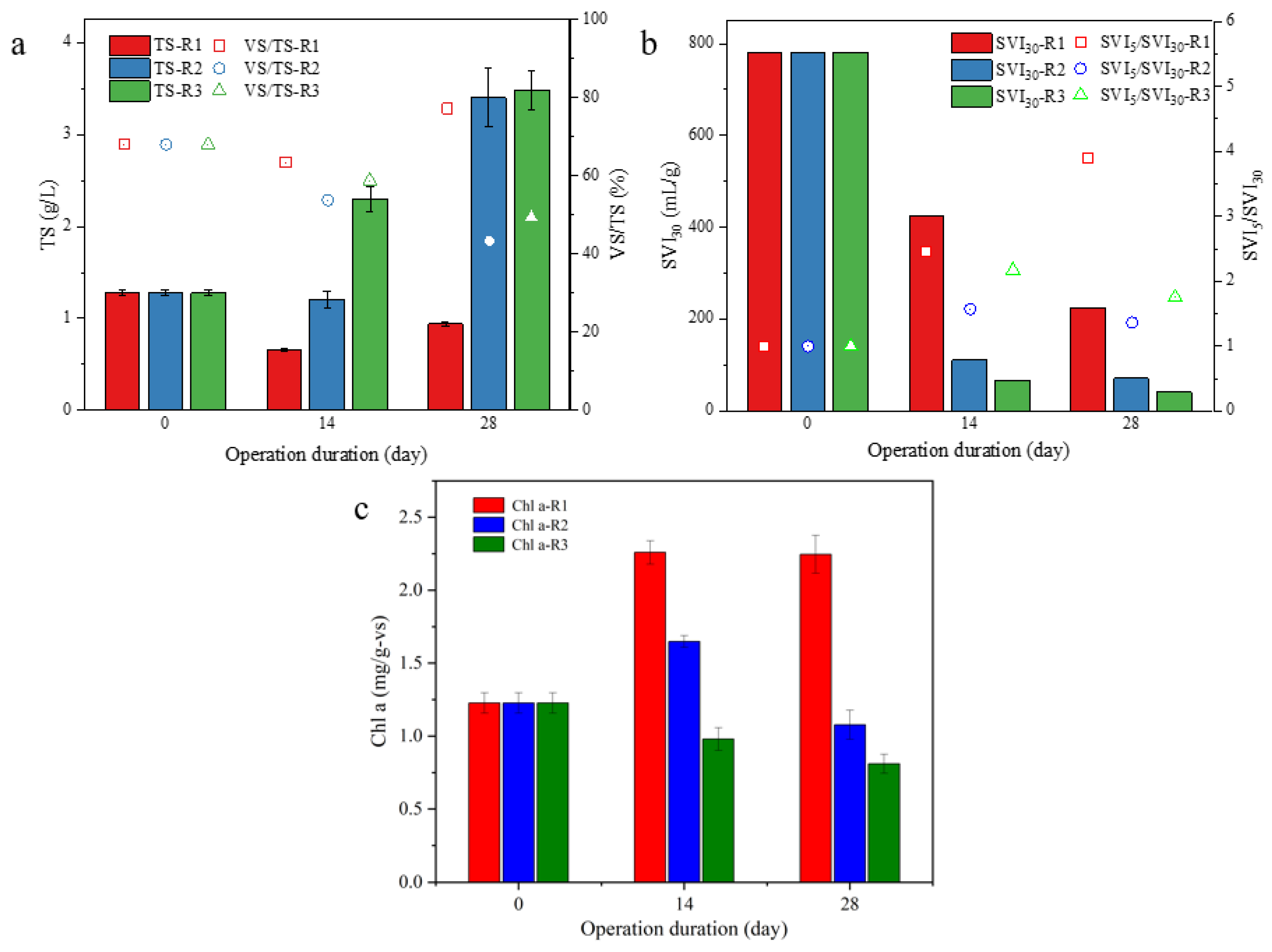
Together with the aggregation process, the biomass accumulation in R3 was also superior to other groups. As shown in Figure 3a, TS in three reactors were firstly decreased after 2 weeks of cultivation, and then, recovered to a different degree. The final TS in R2 (3.40 g/L) and R3 (3.48 g/L) was significantly higher than that in R1 (0.94 g/L). More noticeably, the results about SVI, SVI30 for the initial microalgae were as high as 781.3 mL/g, which decreased dramatically in R2 and R3 to 70.2 and 40.7 mL/g, respectively, while it maintained the high value of 224.1 mL/g in R1. However, dissimilarly, the result of the Chl-a content, which was initially 1.23 mg/g-VS, the Chl-a content in R1 increased significantly to 2.26 mg/g-VS after 2 weeks of operation, and then, the value was maintained after 2 more weeks. In R2 and R3, Chl-a content continued to decrease, and the lowest value was 0.81 mg/g-VS in R3. According to the above results, it could be concluded that the formation of the microalgal aggregate will bring with its superiors in biomass accumulation and sludge settleability, accompanied with a decline in microalgae content.
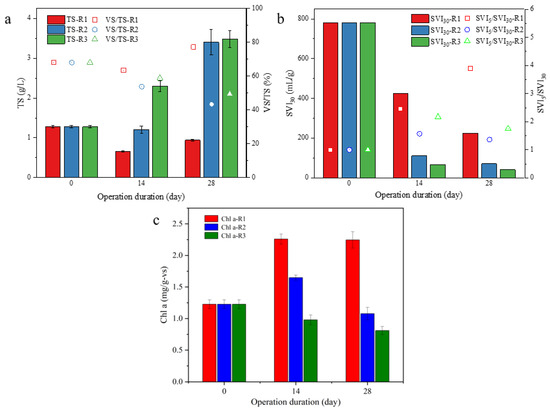
Figure 3.
Variations in TS (a), SVI30 (b), and chlorophyll a content (c) from R1, R2 and R3 during microalgal aggregation process.
3.3. Performance of Nutrients Removal
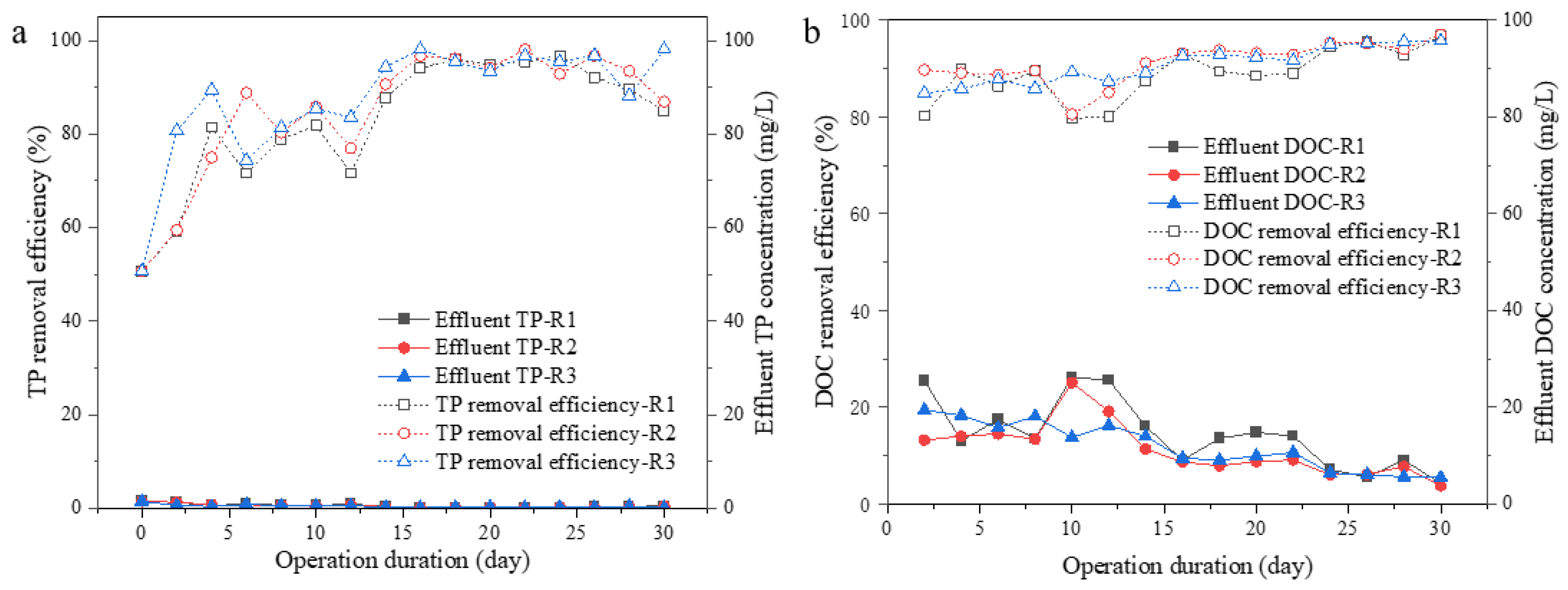
During the aggregation process, N and P removal was also recorded, as shown in Figure 4 and Figure 5. All reactors were operated under ambient conditions with different aeration intensities using the same synthetic wastewater.
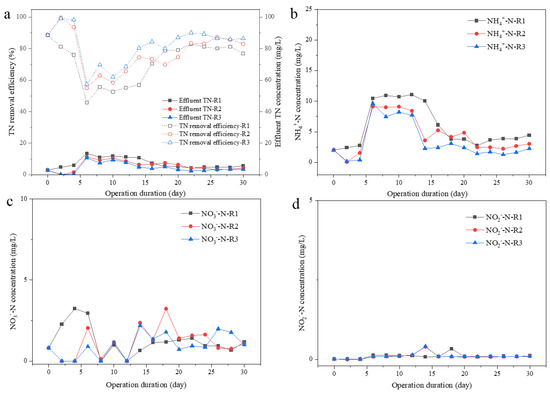
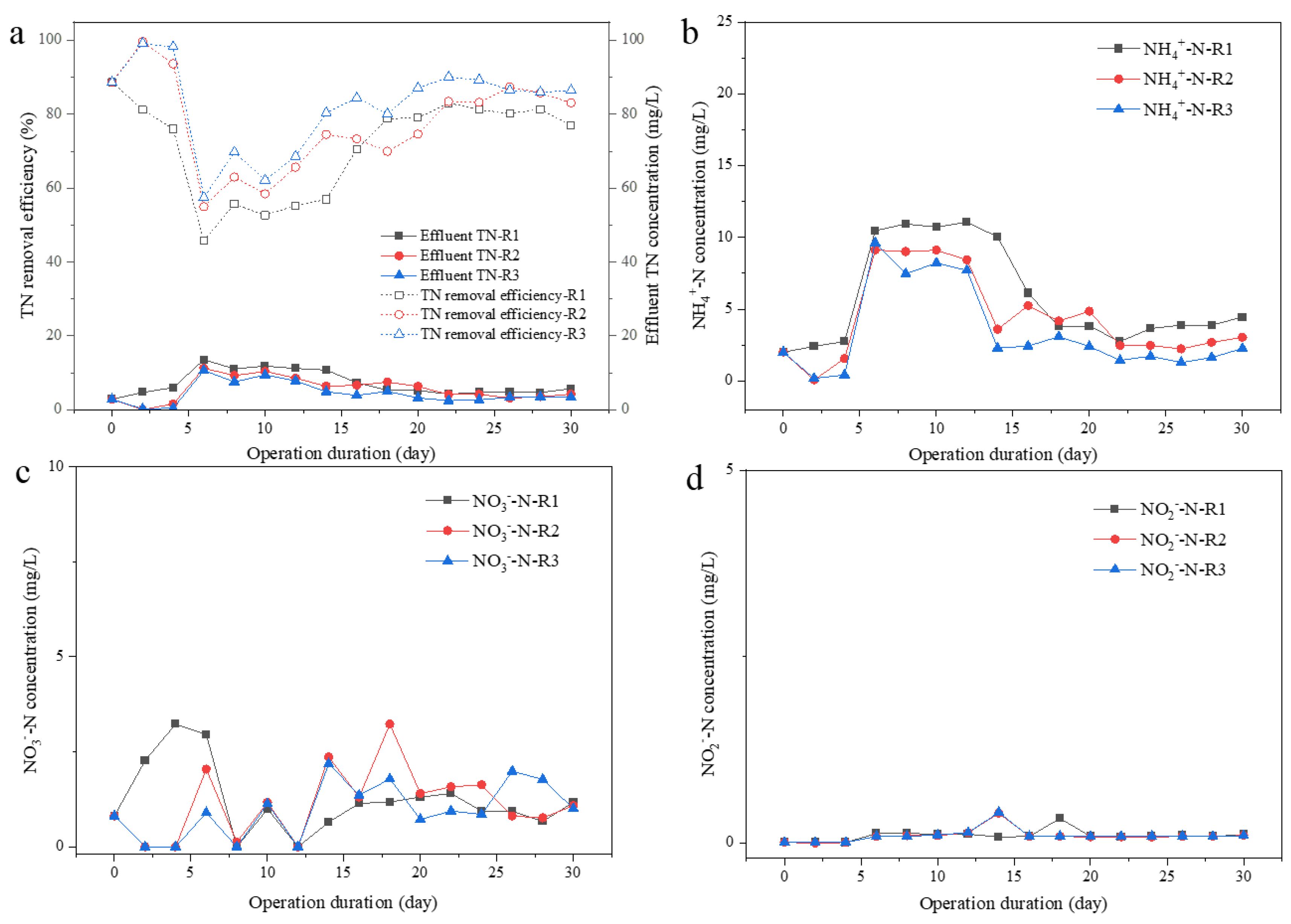
Figure 4.
Effluent nitrogen concentrations and TN removal efficiency. Effluent TN and TN removal efficiency (a), effluent ammonia nitrogen (NH4+-N) (b), effluent nitrate nitrogen (NO3−-N) (c), and effluent nitrite nitrogen (NO2−-N) (d) concentrations.
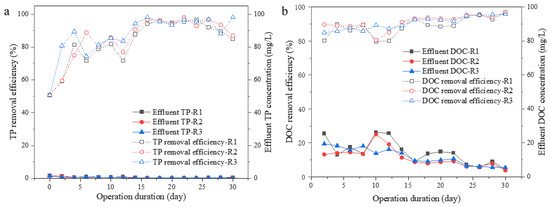
Figure 5.
Effluent TP and DOC concentration or removal efficiency. Effluent TP and TP removal efficiency (a); effluent DOC and DOC removal efficiency (b).
N removal was recorded, as shown in Figure 4, over 80% of total nitrogen (TN) could be successfully removed after 2 weeks of operation; the TN removal efficiency was 77.0, 83.1 and 86.5% in R1, R2, and R3, respectively. The nitrogen in the effluent was mainly NH4+-N, with the concentration of 4.46, 3.04, and 2.27 mg/L at different UAV of 0.65, 1.75, 2.75 cm/s in R1, R2, and R3 on Day 30 (Figure 4b). No significant accumulation of nitrate and nitrite was found. However, slightly higher concentration of NO3−-N could be observed in R2 or R3 than that in R1 during Day 16 to 30, which was 0.76–3.23 mg/L in R2, 0.72–1.99 mg/L in R3, and 0.67–1.40 mg/L in R1 (Figure 4c). The higher concentration of residual NH4+-N in R1 effluent and more accumulation of NO3−-N in effluent from R2 and R3 indicated that a relatively stable nitrification system was established in the three reactors, especially in R2 and R3.
As for P removal, small fluctuations in P removal efficiency were observed during the first 2 weeks of operation and then stabilized; all reactors exhibited an excellent P removal rate of over 85.0% from Day 16 to 30. As shown in Figure 6, P was mainly distributed in the residual rather than the EPS part of microalgal aggregates. This result suggests that the microalgal systems applying different aeration intensities did not show much difference in P removal capacity, probably due to the relatively long HRT (24 h) applied to the systems. Similar trends could be found in terms of DOC removal. All reactors showed a gradual increase in DOC removal capacity during the 30 days of operation. At the end of the operation, the DOC removal capacity in the three reactors was stable at more than 90% with no significant difference. It could be summarized from the above results that a higher aeration intensity would benefit TN removal in terms of ammonia and nitrite oxidizing, while it would have no obvious changes in TP and DOC removal.
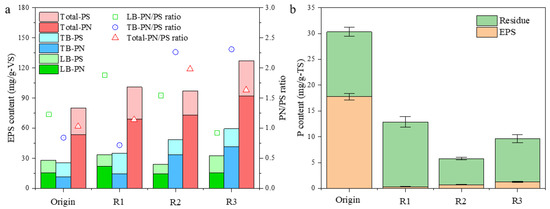
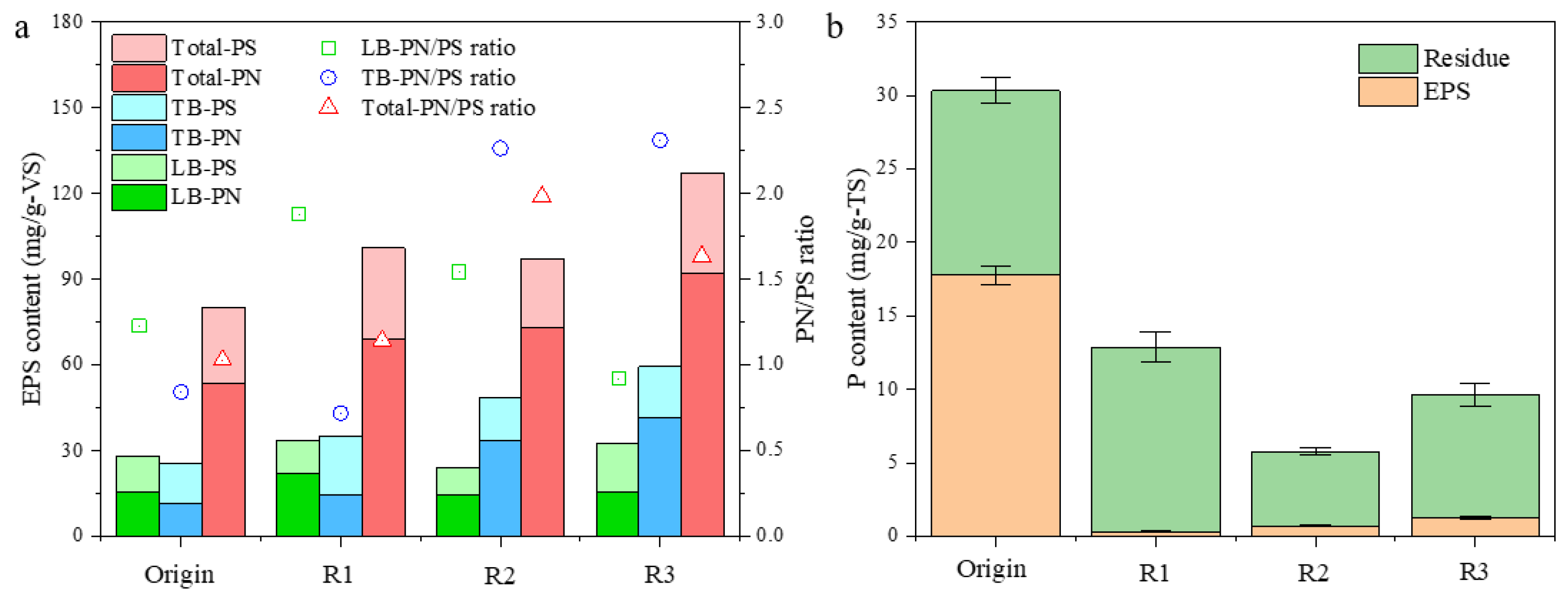
Figure 6.
Changes of EPS (a) and phosphorus (b) content in biomass from R1, R2, and R3 after a 30-day aggregation process. LB-EPS: loosely bound EPS; TB-EPS: tightly bound EPS; residue: the residual part of microalgal biomass after EPS extraction.
3.4. Contents of EPS and Phosphorus
To further evaluate the effect of aeration intensity on microalgal aggregation, EPS was extracted from the original single-cell microalgae and the finally formed aggregates. The EPS content in all reactors increased to a different degree in comparison to that in the seeded microalgae after 30 days of operation (Figure 6), which was significantly higher in R3 (127.2 mg/g-VS), being 58.9% elevated. The proteins-to-polysaccharides (PN/PS) ratio in EPS from all reactors also increased, indicating the enhanced hydrophobicity of microalgal aggregates, especially in R2 and R3, which was more favorable for microalgae pelletizing [15]. Moreover, it is evident from the results that proteins in TB-EPS were more sensitive to microalgal aggregation throughout the experiment compared to LB-EPS, showing an increase in TB-PN content and PN/PS ratio in accordance to aggregation stages among R1, R2, and R3.
The P content in the extracted EPS and the residual part were also determined. As shown in Figure 6b, the P content in the seeded S. platensis was as high as 30.3 mg/g-TS, which was mainly due to the high PO43−-P concentration in ZM (89.0 mg/L in ZM and 3.0 mg/L in SMW) and the P may have accumulated on the EPS part of algal cells. Then, in all reactors, P in EPS was highest in R3, while P content in the residue part was the highest in R1. Interestingly, P content in the residue part of the original microalgae and the biomass in R1 (mainly filamentous microalgae) was very close, indicating that P in algal cells would be stable during the aggregation process, and the decline in P content could be ascribed to the higher mineral part in the formed microalgal aggregates.
4. Discussion
4.1. Suitable Aeration Intensity for Microalgal Aggregation from Filamentous S. platensis
In the current study, filamentous S. platensis microalgae were cultivated in three SBRs under different aeration intensity, UAV that was set as 0.65 cm/s, 1.75 cm/s and 2.75 cm/s, respectively. Obvious microalgal aggregates could be observed in R2 and R3, while the microalgae in R1 remained filamentous (Figure 2a). Accordingly, SVI30 in R2 and R3 also dropped dramatically from the initial 781.2 mL/g to lower than 71.0 mL/g, indicating the great elevation in the settleability of the microalgae. As reported, the SVI30 value of the seed algae cells declined from over 3000 mL/g to 53.4 mL/g for the mature granules after 150 days of the granulation process at a UAV of 0.98 cm/s [14]. Moreover, protein content of the microalgal aggregates, as well as PN/PS ratios in TB- or total EPS in R2 and R3 were also significantly higher than that in R1, which also indicated the better granular structure of aggregates. These results were consistent with the previous studies that enhanced TB-EPS content, favored the integrity of microalgal aggregates [15], and more EPS excretion with high PN/PS ratio was detected in algae granules formed from pure algae [16].
Interestingly, the reduction in Chl-a content in algal biomass was accompanied by the aggregation process from R2 and R3 (Figure 3c), which could be ascribed to the accumulation of mineral particles in these microalgal aggregates. This deduction could be proven by the results of the MLVSS/MLSS ratios (Figure 3a), where the percentage of MLVSS in the total biomass decreased along with the aggregation process. At the end of cultivation, the MLVSS/MLSS ratios were only 43.3% or 49.4% in R2 and R3, respectively, suggesting a large amount of inorganic parts in the formed microalgal aggregates. Therefore, it is suggested from the above results that UAV in SBRs should be higher than 1.75 cm/s for microalgal aggregates, and a UAV of 2.75 cm/s would be suitable to achieve successful microalgal aggregation in the short duration of 30 days.
4.2. Nutrients’ Removal by Filamentous or Aggregated S. platensis
The formation of microalgal aggregates does not only benefit the enhancement of the settleability of filamentous S. platensis, but nutrient removal efficiency would also be elevated. On the one hand, microalgae can use carbon, nitrogen, phosphorus and other nutrients from mariculture wastewater to accumulate biomass; on the other hand, the growth of functional microorganisms, such as ammonia-oxidizing bacteria (AOB), nitrite-oxidizing bacteria (NOB), denitrifying bacteria and phosphorus-accumulating organisms (PAO), inside or on the surface of the aggregates would also improve the removal efficiency of nutrients [14,23,24].
Nitrogen and phosphorus’ removal rates by filamentous or aggregated S. platensis were elevated soon after a short adaptive duration (Day 8 to 14), then maintained high efficiency in the following 2 weeks (Figure 4 and Figure 5). High nutrient removal capacity was obtained for all systems with different aeration intensities at 12 h HRT, demonstrating the high potential of S. platensis for wastewater treatment applications. Even though both TN and TP removal rate in R3 was slightly higher than that in R2 or R1, the differences were less than 5% and not significant, suggesting that elevation in nutrients’ removal was mainly due to the adaptation of S. platensis to SMW, the aggregation process had only a very limited promotion effect. However, nitrogen removal pathway would be different between R1 and R2 or 3. Firstly, lower ammonia N but higher nitrate N could be found in the effluent from R2 and R3 than that from R1, suggesting the higher efficiency of ammonia/nitrite oxidizing. As reported, Nitrosomonas-related AOB were the predominant functional bacteria under the 200 μmol/(m2·s) illuminance condition for algal–bacterial granulation [25]. Nitrospina-related NOB were dominant nitrifying genera contributed to bacterial ammonium oxidation in an algal–bacterial consortium process [26]. Moreover, the formation of aggregates could provide niches for the growth of anaerobic denitrifying bacteria due to the layered structure of granules, benefiting by the denitrifying process as well [27,28]. In terms of P content, significantly higher P content could be found in the EPS of microalgal aggregates in R3, than that in R1 or R2, which could be ascribed to the accumulation of PAOs in the aggregates [24]. Therefore, the aggregation process of S. platensis not only increased the settleability of algal biomass, but the nutrient removal efficiency was also elevated due to the diversity of microorganisms, making this algal–bacterial symbiosis system more adaptable to various environmental conditions during wastewater treatment.
5. Conclusions
S. platensis is a potential organism for a cost-effective and environmentally friendly mariculture wastewater treatment, while the biomass harvesting remained the bottleneck of its large-scale application. In this study, the filamentous S. platensis was cultivated in SBRs under three stages of aeration intensity to form well-settled microalgal aggregates; the nutrients’ removal efficiency from SMW was also evaluated. It could be concluded from this study that UAV should be set to be higher than 1.75 cm/s for microalgal aggregation, and the UAV of 2.75 cm/s was suitable to achieve successful microalgal aggregation in the short duration of 30 days. The formation of a microalgal aggregate from filamentous S. platensis not only elevated TN and TP removal efficiency, but the P content in biomass was also increased, reflecting its great potential for P recovery from wastewater. The aggregation of S. platensis proposed in this study could be an efficient and feasible method for high saline wastewater treatment, particularly in the significance of microalgal nutrient recovery adapted to resource-oriented mariculture wastewater treatment schemes.
Author Contributions
Conceptualization, G.L. and Z.Z.; Methodology, L.L. (Lihong Liu) and X.Y.; Formal Analysis, S.L., J.L. and Z.Z; Investigation, Q.J. and L.L. (Li Liu); Writing—Original Draft Preparation, X.F. and Q.J.; Writing—Review and Editing, X.Y. and Z.Z.; Supervision, Q.L. and J.C.; Funding Acquisition, G.L. and Z.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Special Basic Research Fund for the Central Public Research Institutes of China (No. PM-zx703-202104-130 and No. PM-zx703-202204-152), Science and Technology Research Project of China Construction Fourth Division (CSCEC4B-2021-KTA-6), and Joint Research on the Protection and Restoration of the Ecological Environment of the Yangtze River (II) (2022-LHYJ-02-0511-04).
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Zheng, L.; Liu, Q.; Liu, J.; Xiao, J.; Xu, G. Pollution Control of Industrial Mariculture Wastewater: A Mini-Review. Water 2022, 14, 1390. [Google Scholar] [CrossRef]
- Webber, J.L.; Tyler, C.R.; Carless, D.; Jackson, B.; Tingley, D.; Stewart-Sinclair, P.; Artioli, Y.; Torres, R.; Galli, G.; Miller, P.I. Impacts of Land Use on Water Quality and the Viability of Bivalve Shellfish Mariculture in the UK: A Case Study and Review for SW England. Environ. Sci. Policy 2021, 126, 122–131. [Google Scholar] [CrossRef]
- Zhang, C.; Hasunuma, T.; Lam, S.S.; Kondo, A.; Ho, S.-H. Salinity-induced Microalgal-based Mariculture Wastewater Treatment Combined with Biodiesel Production. Bioresour. Technol. 2021, 340, 125638. [Google Scholar] [CrossRef]
- Gao, Y.; Guo, L.; Liao, Q.; Zhang, Z.; Zhao, Y.; Gao, M.; Jin, C.; She, Z.; Wang, G. Mariculture Wastewater Treatment with Bacterial-Algal Coupling System (BACS): Effect of Light Intensity on Microalgal Biomass Production and Nutrient Removal. Environ. Res. 2021, 201, 111578. [Google Scholar] [CrossRef] [PubMed]
- Maity, J.P.; Bundschuh, J.; Chen, C.-Y.; Bhattacharya, P. Microalgae for Third Generation Biofuel Production, Mitigation of Greenhouse Gas Emissions and Wastewater Treatment: Present and Future Perspectives—A Mini Review. Energy 2014, 78, 104–113. [Google Scholar] [CrossRef]
- Vo, H.N.P.; Ngo, H.H.; Guo, W.; Chang, S.W.; Nguyen, D.D.; Chen, Z.; Wang, X.C.; Chen, R.; Zhang, X. Microalgae for Saline Wastewater Treatment: A Critical Review. Crit. Rev. Environ. Sci. Technol. 2020, 50, 1224–1265. [Google Scholar] [CrossRef]
- Skjånes, K.; Lindblad, P.; Muller, J. BioCO2-A Multidisciplinary, Biological Approach Using Solar Energy to Capture CO2 while Producing H2 and High Value Products. Biomol. Eng. 2007, 24, 405–413. [Google Scholar] [CrossRef]
- Madkour, F.F.; Kamil, A.E.W.; Nasr, H.S. Production and Nutritive Value of Spirulina platensis in Reduced Cost Media. Egypt. J. Aquat. Res. 2012, 38, 51–57. [Google Scholar] [CrossRef]
- Masten Rutar, J.; Jagodic Hudobivnik, M.; Nečemer, M.; Vogel Mikuš, K.; Arčon, I.; Ogrinc, N. Nutritional Quality and Safety of the Spirulina Dietary Supplements Sold on the Slovenian Market. Foods 2022, 11, 849. [Google Scholar] [CrossRef]
- Park, Y.I.; Labrecque, M.; Lavoie, J.M. Influence of Elevated CO2 and Municipal Wastewater Feed on the Productivity, Morphology, and Chemical Composition of Arthrospira (Spirulina) platensis. ACS Sustain. Chem. Eng. 2013, 1, 1348–1356. [Google Scholar] [CrossRef]
- Jiang, L.; Pei, H.; Hu, W.; Ji, Y.; Han, L.; Ma, G. The Feasibility of Using Complex Wastewater from a Monosodium Glutamate Factory to Cultivate Spirulina subsalsa and Accumulate Biochemical Composition. Bioresour. Technol. 2015, 180, 304–310. [Google Scholar] [CrossRef]
- Zhou, W.; Li, Y.; Gao, Y.; Zhao, H. Nutrients Removal and Recovery from Saline Wastewater by Spirulina platensis. Bioresour. Technol. 2017, 245, 10–17. [Google Scholar] [CrossRef]
- Lu, Z.; Beal, C.M.; Johnson, Z.I. Comparative Performance and Technoeconomic Analyses of Two Microalgae Harvesting Systems Evaluated at aCommercially Relevant Scale. Algal Res. 2022, 64, 102667. [Google Scholar] [CrossRef]
- Wang, Q.; Shen, Q.; Wang, J.; Zhang, Y.; Zhang, Z.; Lei, Z.; Shimizu, K.; Lee, D.-J. Fast Cultivation and Harvesting of Oil-producing Microalgae Ankistrodesmus falcatus var. Acicularis Fed with Anaerobic Digestion Liquor via Biogranulation in Addition to Nutrients Removal. Sci. Total Environ. 2020, 741, 140183. [Google Scholar]
- Wang, Q.; Shen, Q.; Wang, J.; Zhao, J.; Zhang, Z.; Lei, Z.; Yuan, T.; Shimizu, K.; Liu, Y.; Lee, D.-J. Insight into the Rapid Biogranulation for Suspended Single-cell Microalgae Harvesting in Wastewater Treatment Systems: Focus on the Role of Extracellular Polymeric Substances. Chem. Eng. J. 2022, 430, 132631. [Google Scholar] [CrossRef]
- Cai, W.; Zhao, Z.; Li, D.; Lei, Z.; Zhang, Z.; Lee, D.-J. Algae Granulation for Nutrients Uptake and Algae Harvesting during Wastewater Treatment. Chemosphere 2019, 214, 55–59. [Google Scholar] [CrossRef]
- Bazdar, E.; Roshandel, R.; Yaghmaei, S.; Mardanpour, M.M. The Effect of Different Light Intensities and Light/Dark Regimes on the Performance of Photosynthetic Microalgae Microbial Fuel Cell. Bioresour. Technol. 2018, 261, 350–360. [Google Scholar] [CrossRef]
- Leema, J.T.M.; Kirubagaran, R.; Vinithkumar, N.V.; Dheenan, P.S.; Karthikayulu, S. High Value Pigment Production from Arthrospira (Spirulina) platensis Cultured in Seawater. Bioresour. Technol. 2010, 101, 9221–9227. [Google Scholar] [CrossRef]
- Zhu, J.; You, H.; Li, Z.; Xie, B.; Chen, H.; Ding, Y.; Qi, S.; Li, W.; Ma, B.; Qu, X. Comparison on the Photogranules Formation and Microbial Community Shift between the Batch and Continuous-flow Mode for the High Saline Wastewater Treatment. Chem. Eng. J. 2022, 446, 137284. [Google Scholar] [CrossRef]
- Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA; American Water Work Association: Washington, DC, USA; Water Environment Federation: Washington, DC, USA, 2012; Available online: https://beta-static.fishersci.com/content/dam/fishersci/en_US/documents/programs/scientific/technical-documents/white-papers/apha-water-testing-standard-methods-introduction-white-paper.pdf (accessed on 14 December 2022).
- Lowry, O.H. Protein Measurement with the Folin Phenol Reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.T.; Smith, F. Colorimetric Method for Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- de Sousa Rollemberg, S.L.; Barros, A.R.M.; Firmino, P.I.M.; Dos Santos, A.B. Aerobic Granular Sludge: Cultivation Parameters and Removal Mechanisms. Bioresour. Technol. 2018, 270, 678–688. [Google Scholar] [CrossRef]
- Zhang, B.; Lens, P.N.; Shi, W.; Zhang, R.; Zhang, Z.; Guo, Y.; Bao, X.; Cui, F. Enhancement of Aerobic Granulation and Nutrient Removal by an Algal-bacterial Consortium in aLab-scale Photobioreactor. Chem. Eng. J. 2018, 334, 2373–2382. [Google Scholar] [CrossRef]
- Si, G.; Liu, B.; Liu, Y.; Yan, T.; Wei, D. Light-introduced Partial Nitrification in an Algal-bacterial Granular Sludge Bioreactor: Performance Evolution and Microbial Community Shift. Bioresour. Technol. 2022, 354, 127226. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, Q.; Xu, G.; Gao, F. Simultaneous Nitritation, Denitritation and Phosphorus Removal in an Algal-bacterial Consortium System Treating Low-strength Mariculture Wastewater. J. Water Process Eng. 2022, 49, 103056. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhou, Y.; Chen, S.; Guo, N.; Xiang, P.; Lin, S.; Bai, Y.; Hu, X.; Zhang, Z. Evaluating the Role of Algae in Algal-bacterial Granular Sludge: Nutrient Removal, Microbial Community and Granular Characteristics. Bioresour. Technol. 2022, 365, 128165. [Google Scholar] [CrossRef]
- Xiong, W.; Wang, S.; Jin, Y.; Wu, Z.; Liu, D.; Su, H. Insights into Nitrogen and Phosphorus Metabolic Mechanisms of Algal-bacterial Aerobic Granular Sludge via Metagenomics: Performance, Microbial Community and Functional Genes. Bioresour. Technol. 2023, 369, 128442. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).