Research Progress on the Decomposition Process of Plant Litter in Wetlands: A Review
Abstract
:1. Introduction
2. Studies on the Decomposition of Litter in Wetlands
2.1. Research Methodologies for Exploring the Decomposition of Wetland Plant Litter
2.2. Decomposition of Wetland Standing Dead Matter
2.3. Decomposition of Traditional Litter in Wetlands
2.3.1. The Decomposition Process of Wetland Litter
2.3.2. Modeling the Decomposition of Wetland Litter
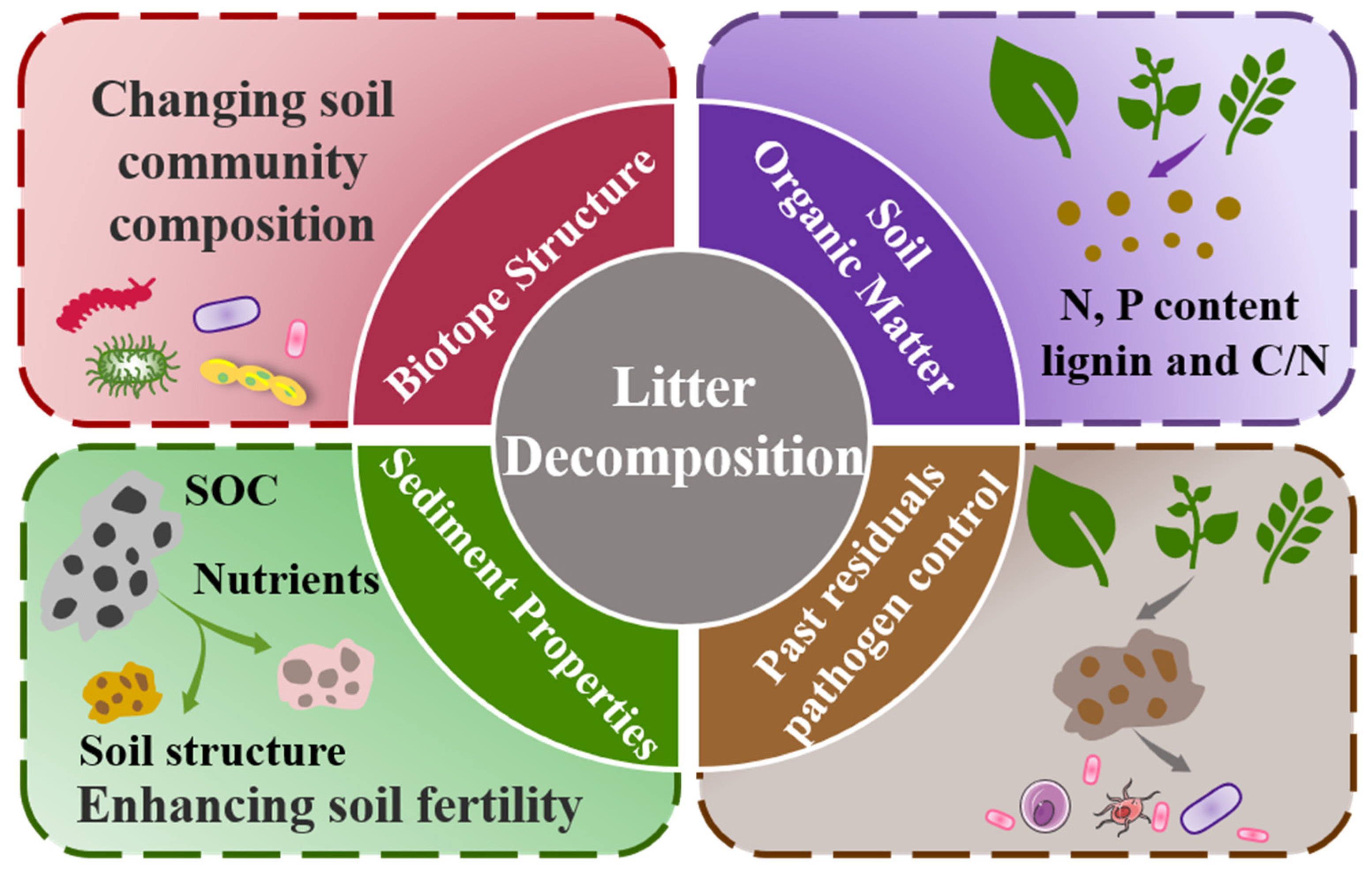
3. Effect of Decomposition of Litter on the Performance of Wetlands
3.1. Effects of Wetland Soil Biotope Structure
3.2. Effects of Wetland Soil Organic Matter
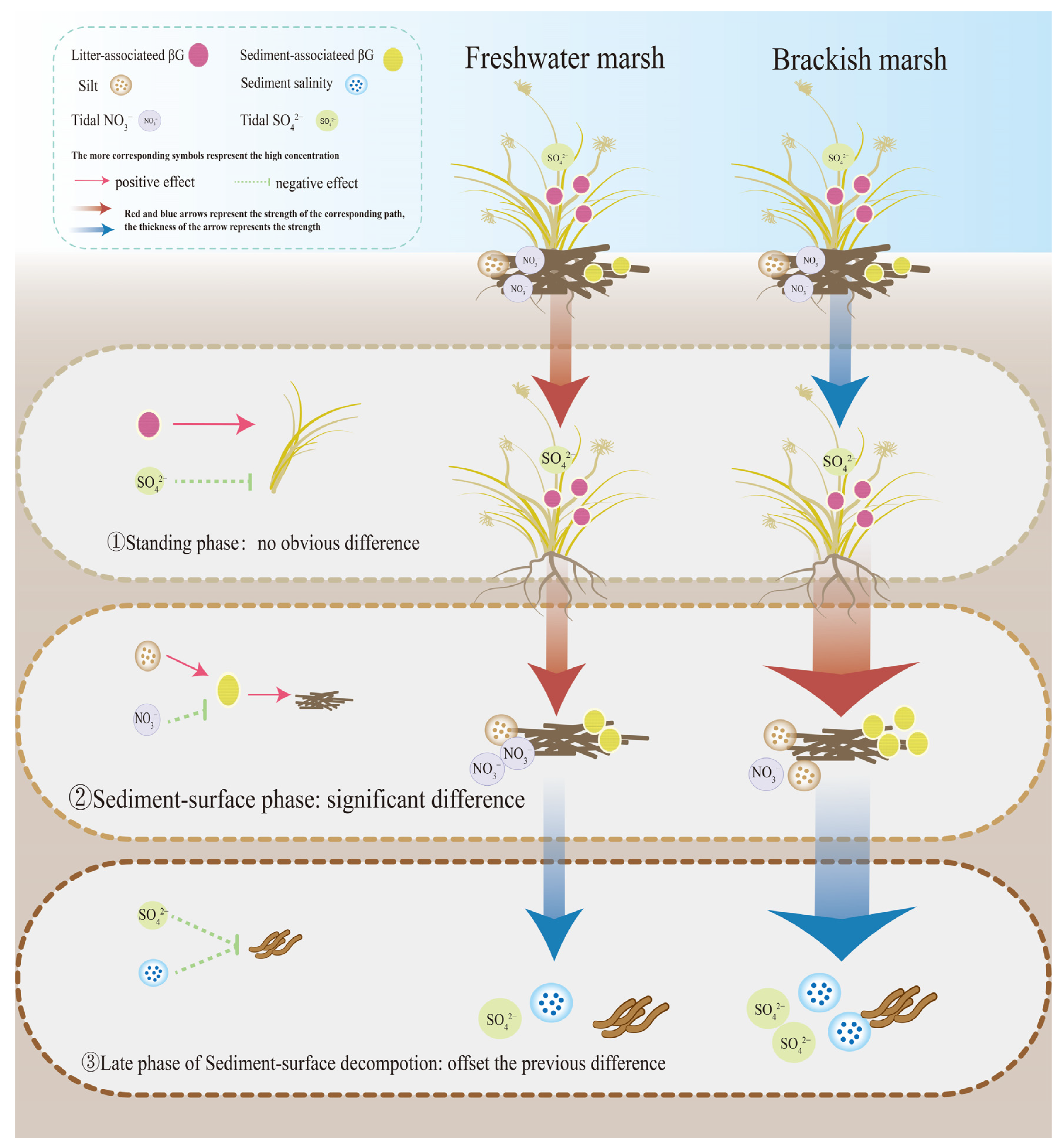
3.3. Impacts of Wetland Sediment Properties
4. Conclusions and Future Perspectives
- (1)
- Decomposition of matter above- and belowground. Most studies have revealed that the rate of litter decomposition is considerably faster than that of root litter decomposition and that there is a substantial difference between above- and belowground litter decomposition [102]; this difference and its effects are not well understood, and the influences of root litter decomposition and soil physicochemical properties remain unclear.
- (2)
- Mixed litter decomposition. The decomposition rate of mixed litter is usually more unstable than that of single litter [103]. Few studies have been conducted on the mechanism and model of hybrid litter decomposition, though the decomposition process has little relevance for wetlands.
- (3)
- The effects of matter decomposition on the sorption properties of wetland soils. Litter decomposition promotes the decomposition of organic matter in the soil, changes the physical and chemical properties of the soil, and improves soil structure. With the sub-discharge of industrial wastewater, wetland soils are increasingly polluted by heavy metals, and soil adsorption of heavy metals can reduce their mobility. However, the effect of litter decomposition on the ability of soil to fix heavy metals and other pollutants remains unclear.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tingyu, Z.; Yang, X.; Qingyang, H.; Chen, X.; You, L. Forest Litter Decomposition: Research Progress and Prospect. Chin. Agric. Sci. Bull. 2022, 38, 44–51. [Google Scholar]
- Berg, B. Decomposition patterns for foliar litter—A theory for influencing factors. Soil Biol. Biochem. 2014, 78, 222–232. [Google Scholar] [CrossRef]
- Hobbie, S.E. Plant species effects on nutrient cycling: Revisiting litter feedbacks. Trends Ecol. Evol. 2015, 30, 357–363. [Google Scholar]
- Salimi, S.; Almuktar, S.A.A.A.N.; Scholz, M. Impact of climate change on wetland ecosystems: A critical review of experimental wetlands. J. Environ. Manag. 2021, 286, 112160. [Google Scholar]
- Ding, Y.; Wang, D.; Zhao, G.; Chen, S.; Sun, T.; Sun, H.; Wu, C.; Li, Y.; Yu, Z.; Li, Y.; et al. The contribution of wetland plant litter to soil carbon pool: Decomposition rates and priming effects. Environ. Res. 2023, 224, 115575. [Google Scholar] [CrossRef]
- Duan, H.; Wang, L.; Zhang, Y.; Fu, X.; Tsang, Y.; Wu, J.; Le, Y. Variable decomposition of two plant litters and their effects on the carbon sequestration ability of wetland soil in the Yangtze River estuary. Geoderma 2018, 319, 230–238. [Google Scholar]
- Cornelissen, J.H.C.; Cornwell, W.K.; Freschet, G.T.; Weedon, J.T.; Berg, M.P.; Zanne, A.E. Coevolutionary legacies for plant decomposition. Trends Ecol. Evol. 2023, 38, 44–54. [Google Scholar] [CrossRef]
- Zhang, W.-P.; Fornara, D.; Yang, H.; Yu, R.-P.; Callaway, R.M.; Li, L. Plant litter strengthens positive biodiversity–ecosystem functioning relationships over time. Trends Ecol. Evol. 2023, 38, 473–484. [Google Scholar] [CrossRef]
- Hassan, N.; Sher, K.; Rab, A.; Abdullah, I.; Zeb, U.; Naeem, I.; Shuaib, M.; Khan, H.; Khan, W.; Khan, A. Effects and mechanism of plant litter on grassland ecosystem: A review. Acta Ecol. Sin. 2021, 41, 341–345. [Google Scholar] [CrossRef]
- Li, F.; Zi, H.; Sonne, C.; Li, X. Microbiome sustains forest ecosystem functions across hierarchical scales. Eco-Environ. Health 2023, 2, 24–31. [Google Scholar]
- Mishra, S.; Hättenschwiler, S.; Yang, X. The plant microbiome: A missing link for the understanding of community dynamics and multifunctionality in forest ecosystems. Appl. Soil Ecol. 2020, 145, 103345. [Google Scholar] [CrossRef]
- Chen, H.; Harmon, M.E.; Griffiths, R.P.; Hicks, W. Effects of temperature and moisture on carbon respired from decomposing woody roots. For. Ecol. Manag. 2000, 138, 51–64. [Google Scholar] [CrossRef]
- Villar, C.A.; de Cabo, L.; Vaithiyanathan, P.; Bonetto, C. Litter decomposition of emergent macrophytes in a floodplain marsh of the Lower Parana River. Aquat. Bot. 2001, 70, 105–116. [Google Scholar] [CrossRef]
- Knacker, T.; Forster, B.; Rombke, J.; Frampton, G.K. Assessing the effects of plant protection products on organic matter breakdown in arable fields-litter decomposition test systems. Soil Biol. Biochem. 2003, 35, 1269–1287. [Google Scholar] [CrossRef]
- Zhang, X.; Song, C.; Mao, R.; Yang, G.; Tao, B.; Shi, F.; Zhu, X.; Hou, A. Litter mass loss and nutrient dynamics of four emergent macrophytes during aerial decomposition in freshwater marshes of the Sanjiang plain, Northeast China. Plant Soil 2014, 385, 139–147. [Google Scholar] [CrossRef]
- Zhang, X.; Jiang, W.; Jiang, S.; Tan, W.; Mao, R. Differential responses of litter decomposition in the air and on the soil surface to shrub encroachment in a graminoid-dominated temperate wetland. Plant Soil 2021, 462, 477–488. [Google Scholar] [CrossRef]
- Mao, R.; Wu, P.-P.; Xu, J.-W.; Wan, S.-Z.; Zhang, Y. Leaf litter decomposition in the air should not be ignored in subtropical plantations of China. For. Ecol. Manag. 2021, 499, 119614. [Google Scholar] [CrossRef]
- Lodato, M.B.; Boyette, J.S.; Smilo, R.A.; Jackson, C.R.; Halvorson, H.M.; Kuehn, K.A. Functional importance and diversity of fungi during standing grass litter decomposition. Oecologia 2021, 195, 499–512. [Google Scholar] [CrossRef]
- Kuehn, K.A.; Suberkropp, K. Decomposition of standing litter of the freshwater emergent macrophyte Juncus effusus. Freshw. Biol. 1998, 40, 717–727. [Google Scholar] [CrossRef]
- Zhang, X.; Mao, R.; Gong, C.; Qiao, T.; Song, C. CO2 evolution from standing litter of the emergent macrophyte Deyeuxia angustifolia in the Sanjiang Plain, Northeast China. Ecol. Eng. 2014, 63, 45–49. [Google Scholar] [CrossRef]
- Kuehn, K.A.; Steiner, D.; Gessner, M.O. Diel mineralization patterns of standing-dead plant litter: Implications for CO2 flux from wetlands. Ecology 2004, 85, 2504–2518. [Google Scholar] [CrossRef]
- Gao, J.; Zhou, W.; Liu, Y.; Sha, L.; Song, Q.; Lin, Y.; Yu, G.; Zhang, J.; Zheng, X.; Fang, Y.; et al. Litter-derived nitrogen reduces methane uptake in tropical rainforest soils. Sci. Total Environ. 2022, 849, 157891. [Google Scholar] [CrossRef]
- Su, Y.; Dong, K.; Wang, C.; Liu, X. Grazing promoted plant litter decomposition and nutrient release: A meta-analysis. Agric. Ecosyst. Environ. 2022, 337, 108051. [Google Scholar] [CrossRef]
- Su, Y.; Ma, X.; Gong, Y.; Ahmed, Z.; Han, W.; Li, K.; Liu, X. Global Patterns and Drivers of Litter Decomposition Under Nitrogen Enrichment: A Meta-Analysis. Front. For. Glob. Change 2022, 5, 895774. [Google Scholar] [CrossRef]
- Couteaux, M.M.; Bottner, P.; Berg, B. Litter decomposition, climate and liter quality. Trends Ecol. Evol. 1995, 10, 63–66. [Google Scholar] [CrossRef]
- Hattenschwiler, S.; Tiunov, A.V.; Scheu, S. Biodiversity and litter decomposition interrestrial ecosystems. Annu. Rev. Ecol. Evol. Syst. 2005, 36, 191–218. [Google Scholar] [CrossRef]
- Cepakova, S.; Frouz, J. Changes in chemical composition of litter during decomposition: A review of published C-13 NMR spectra. J. Soil Sci. Plant Nutr. 2015, 15, 805–815. [Google Scholar]
- Chapin, F.S., III; McFarland, J.; McGuire, A.D.; Euskirchen, E.S.; Ruess, R.W.; Kielland, K. The changing global carbon cycle: Linking plant-soil carbon dynamics to global consequences. J. Ecol. 2009, 97, 840–850. [Google Scholar]
- Yang, Y.; Shi, Y.; Sun, W.; Chang, J.; Zhu, J.; Chen, L.; Wang, X.; Guo, Y.; Zhang, H.; Yu, L.; et al. Terrestrial carbon sinks in China and around the world and their contribution to carbon neutrality. Sci. China Life Sci. 2022, 65, 861–895. [Google Scholar] [CrossRef]
- Bouchard, V.; Gillon, D.; Joffre, R.; Lefeuvre, J.C. Actual litter decomposition rates in salt marshes measured using near-infrared reflectance spectroscopy. J. Exp. Mar. Biol. Ecol. 2003, 290, 149–163. [Google Scholar] [CrossRef]
- Gillon, D.; Joffre, R.; Ibrahima, A. Can litter decomposability be predicted by near infrared reflectance spectroscopy? Ecology 1999, 80, 175–186. [Google Scholar] [CrossRef]
- Couteaux, M.; Sarmiento, L.; Herve, D.; Acevedo, D. Determination of water-soluble and total extractable polyphenolics in biomass, necromass and decomposing plant material using near-infrared reflectance spectroscopy (NIRS). Soil Biol. Biochem. 2005, 37, 795–799. [Google Scholar] [CrossRef]
- Mancinelli, G.; Costantini, M.L.; Rossi, L. Predicting ergosterol in leaf litter by near-infrared spectroradiometry: A preliminary assessment. Eur. J. Soil Biol. 2014, 63, 49–54. [Google Scholar] [CrossRef]
- Zhang, K.; Xu, Y.; Johnson, L.; Yuan, W.; Pei, Z.; Wang, D. Development of near-infrared spectroscopy models for quantitative determination of cellulose and hemicellulose contents of big bluestem. Renew. Energy 2017, 109, 101–109. [Google Scholar] [CrossRef]
- Ferreira, G.W.D.; Roque, J.V.; Soares, E.M.B.; Silva, I.R.; Silva, E.F.; Vasconcelos, A.A.; Teofilo, R.F. Temporal decomposition sampling and chemical characterization of eucalyptus harvest residues using NIR spectroscopy and chemometric methods. Talanta 2018, 188, 168–177. [Google Scholar] [CrossRef]
- Yunshuo, L.; Renjie, C.; Hui, L.; Chenli, W.; Kun, T.; Hang, W. Study on Decomposition Characteristics of Key Indexes of Wetland Emergent Aquatic Plant. J. Southwest For. Univ. 2021, 41, 93–102. [Google Scholar]
- Kuehn, K.A.; Gessner, M.O.; Wetzel, R.G.; Suberkropp, K. Decomposition and CO2 evolution from standing litter of the emergent macrophyte Erianthus giganteus. Microb. Ecol. 1999, 38, 50–57. [Google Scholar] [CrossRef]
- Welsch, M.; Yavitt, J.B. Early stages of decay of Lythrum salicaria L. and Typha latifolia L. in a standing-dead position. Aquat. Bot. 2003, 75, 45–57. [Google Scholar] [CrossRef]
- Kuehn, K.A.; Ohsowski, B.M.; Francoeur, S.N.; Neely, R.K. Contributions of fungi to carbon flow and nutrient cycling from standing dead Typha angustifolia leaf litter in a temperate freshwater marsh. Limnol. Oceanogr. 2011, 56, 529–539. [Google Scholar] [CrossRef]
- Hebert, T.A.; Halvorson, H.M.; Kuehn, K.A. A literature synthesis resolves litter intrinsic constraints on fungal dynamics and decomposition across standing dead macrophytes. Oikos 2021, 130, 958–968. [Google Scholar] [CrossRef]
- Zhang, X.; Mao, R.; Song, C.; Song, Y.; Finnegan, P.M. Nitrogen addition in a freshwater marsh alters the quality of senesced leaves, promoting decay rates and changing nutrient dynamics during the standing-dead phase. Plant Soil 2017, 417, 511–521. [Google Scholar] [CrossRef]
- Liao, C.Z.; Luo, Y.Q.; Fang, C.M.; Chen, J.K.; Li, B. Litter pool sizes, decomposition, and nitrogen dynamics in Spartina alterniflora-invaded and native coastal marshlands of the Yangtze Estuary. Oecologia 2008, 156, 589–600. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Chen, H.Y.H. Global effects of plant litter alterations on soil CO2 to the atmosphere. Glob. Change Biol. 2018, 24, 3462–3471. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Deng, Z.; Xie, Y.; Zhang, C.; Chen, X.; Li, X.; Li, F.; Chen, X.; Zou, Y.; Wang, W. Effects of hydrological environment on litter carbon input into the surface soil organic carbon pool in the Dongting Lake floodplain. Catena 2022, 208, 105761. [Google Scholar] [CrossRef]
- Hong-juan, H.; Shui-jing, Z.; Wei-ping, H. Modelling Nitrogen and Phosphorus Transfer in Potamogeton malaianus Miq. Decompostion. Environ. Sci. 2010, 31, 1483–1488. [Google Scholar]
- Zeng, Q.; Liu, Y.; Zhang, H.; An, S. Fast bacterial succession associated with the decomposition of Quercus wutaishanica litter on the Loess Plateau. Biogeochemistry 2019, 144, 119–131. [Google Scholar] [CrossRef]
- Zhan, P.; Li, H.; Cui, W.; Wang, Y.; Liu, Z.; Xiao, D.; Wang, H. Functional insights into succession in a phyllospheric microbial community across a full period of aquatic plant litter decomposition. Freshw. Sci. 2023, 42, 13–32. [Google Scholar] [CrossRef]
- Ma, A.; Liu, H.; Song, C.; Tian, E.; Wang, X. Home-field advantage in litter decomposition: A critical review from a microbial perspective. J. Soils Sediments 2023, 63, 709–721. [Google Scholar] [CrossRef]
- Yan, W.; Zhong, Y.; Zhu, G.; Liu, W.; Shangguan, Z. Nutrient limitation of litter decomposition with long-term secondary succession: Evidence from controlled laboratory experiments. J. Soils Sediments 2020, 20, 1858–1868. [Google Scholar] [CrossRef]
- Weintraub, M.N.; Schimel, J.P. Interactions between carbon and nitrogen mineralization and soil organic matter chemistry in arctic tundra soils. Ecoststems 2003, 6, 129–143. [Google Scholar] [CrossRef]
- Harmon, M.E.; Silver, W.L.; Fasth, B.; Chen, H.; Burke, I.C.; Parton, W.J.; Hart, S.C.; Currie, W.S.; Lidet. Long-term patterns of mass loss during the decomposition of leaf and fine root litter: An intersite comparison. Glob. Change Biol. 2009, 15, 1320–1338. [Google Scholar] [CrossRef]
- Currie, W.S.; Harmon, M.E.; Burke, I.C.; Hart, S.C.; Parton, W.J.; Silver, W. Cross-biome transplants of plant litter show decomposition models extend to a broader climatic range but lose predictability at the decadal time scale. Glob. Change Biol. 2010, 16, 1744–1761. [Google Scholar] [CrossRef]
- Bonanomi, G.; Incerti, G.; Antignani, V.; Capodilupo, M.; Mazzoleni, S. Decomposition and nutrient dynamics in mixed litter of Mediterranean species. Plant Soil 2010, 331, 481–496. [Google Scholar] [CrossRef]
- Maisto, G.; De Marco, A.; Meola, A.; Sessa, L.; De Santo, A.V. Nutrient dynamics in litter mixtures of four Mediterranean maquis species decomposing in situ. Soil Biol. Biochem. 2011, 43, 520–530. [Google Scholar] [CrossRef]
- Shao-yong, L.; Peng-yi, Z.; Gang, Y.; Wan-peng, Z.; Chang-sheng, X. The contaminants release rule of Zizania caduciflora, Phragmites austrails and Eichhornia crassipes. China Environ. Sci. 2005, 25, 554–557. [Google Scholar]
- Lan, Y.; Cui, B.; You, Z.; Li, X.; Han, Z.; Zhang, Y.; Zhang, Y. Litter decomposition of six macrophytes in a eutrophic shallow lake (Baiyangdian Lake, China). Clean–Soil Air Water 2012, 40, 1159–1166. [Google Scholar] [CrossRef]
- Eid, E.M.; Shaltout, K.H.; Al-Sodany, Y.M. Decomposition dynamics of Phragmites australis litter in Lake Burullus, Egypt. Plant Species Biol. 2014, 29, 47–56. [Google Scholar] [CrossRef]
- Cao, P.; Liu, M.; Tang, J.; Teng, S.; Xu, C. A comparative study on the decomposition processes among some aquatic plants. Acta Ecol. Sin 2014, 34, 3848–3858. [Google Scholar]
- Kuehn, K.; Lemke, M.; Suberkropp, K.; Wetzel, R. Microbial biomass and production associated with decaying leaf litter of the emergent macrophyte Juncus effusus. Limnol. Oceanogr. 2000, 45, 862–870. [Google Scholar] [CrossRef]
- Zhang, L.; Tong, C.; Marrs, R.; Wang, T.; Zhang, W.; Zeng, C. Comparing litter dynamics of Phragmites australis and Spartina alterniflora in a sub-tropical Chinese estuary: Contrasts in early and late decomposition. Aquat. Bot. 2014, 117, 1–11. [Google Scholar] [CrossRef]
- Turner, R.E.; Bodker, J.E. The effects of N, P and crude oil on the decomposition of Spartina alterniflora belowground biomass. Wetl. Ecol. Manag. 2016, 24, 373–380. [Google Scholar] [CrossRef]
- Wu, W.; Huang, H.; Biber, P.; Bethel, M. Litter decomposition of Spartina alterniflora and Juncus roemerianus: Implications of climate change in salt marshes. J. Coast. Res. 2017, 33, 372–384. [Google Scholar] [CrossRef]
- Purahong, W.; Hyde, K.D. Effects of fungal endophytes on grass and non-grass litter decomposition rates. Fungal Divers. 2011, 47, 1–7. [Google Scholar] [CrossRef]
- Bray, S.R.; Kitajima, K.; Mack, M.C. Temporal dynamics of microbial communities on decomposing leaf litter of 10 plant species in relation to decomposition rate. Soil Biol. Biochem. 2012, 49, 30–37. [Google Scholar] [CrossRef]
- Zheng, H.; Yang, T.; Bao, Y.; He, P.; Yang, K.; Mei, X.; Wei, Z.; Xu, Y.; Shen, Q.; Banerjee, S. Network analysis and subsequent culturing reveal keystone taxa involved in microbial litter decomposition dynamics. Soil Biol. Biochem. 2021, 157, 108230. [Google Scholar] [CrossRef]
- Zhan, P.; Liu, Y.; Wang, H.; Wang, C.; Xia, M.; Wang, N.; Cui, W.; Xiao, D.; Wang, H. Plant litter decomposition in wetlands is closely associated with phyllospheric fungi as revealed by microbial community dynamics and co-occurrence network. Sci. Total Environ. 2021, 753, 142194. [Google Scholar] [CrossRef]
- Torres, P.A.; Abril, A.B.; Bucher, E.H. Microbial succession in litter decomposition in the semi-arid Chaco woodland. Soil Biol. Biochem. 2005, 37, 49–54. [Google Scholar] [CrossRef]
- Veen, G.F.; Snoek, B.L.; Bakx-Schotman, T.; Wardle, D.A.; van der Putten, W.H. Relationships between fungal community composition in decomposing leaf litter and home-field advantage effects. Funct. Ecol. 2019, 33, 1524–1535. [Google Scholar] [CrossRef]
- Tennakoon, D.S.; Gentekaki, E.; Jeewon, R.; Kuo, C.H.; Promputtha, I.; Hyde, K.D. Life in leaf litter: Fungal community succession during decomposition. Mycosphere 2021, 12, 406–429. [Google Scholar] [CrossRef]
- Asemaninejad, A.; Thorn, R.G.; Lindo, Z. Vertical distribution of fungi in hollows and hummocks of boreal peatlands. Fungal Ecol. 2017, 27, 59–68. [Google Scholar] [CrossRef]
- Baker, B.J.; Lazar, C.S.; Teske, A.P.; Dick, G.J. Genomic resolution of linkages in carbon, nitrogen, and sulfur cycling among widespread estuary sediment bacteria. Microbiome 2015, 3, 1–12. [Google Scholar] [CrossRef]
- Ligi, T.; Oopkaup, K.; Truu, M.; Preem, J.-K.; Nolvak, H.; Mitsch, W.J.; Mander, U.; Truu, J. Characterization of bacterial communities in soil and sediment of a created riverine wetland complex using high-throughput 16S rRNA amplicon sequencing. Ecol. Eng. 2014, 72, 56–66. [Google Scholar] [CrossRef]
- Peralta, R.M.; Ahn, C.; Gillevet, P.M. Characterization of soil bacterial community structure and physicochemical properties in created and natural wetlands. Sci. Total Environ. 2013, 443, 725–732. [Google Scholar] [CrossRef]
- Hartman, W.H.; Richardson, C.J.; Vilgalys, R.; Bruland, G.L. Environmental and anthropogenic controls over bacterial communities in wetland soils. Proc. Natl. Acad. Sci. USA 2008, 105, 17842–17847. [Google Scholar] [CrossRef] [PubMed]
- Prasse, C.E.; Baldwin, A.H.; Yarwood, S.A. Site History and Edaphic Features Override the Influence of Plant Species on Microbial Communities in Restored Tidal Freshwater Wetlands. Appl. Environ. Microbiol. 2015, 81, 3482–3491. [Google Scholar] [CrossRef] [PubMed]
- Palozzi, J.E.; Lindo, Z. Are leaf litter and microbes team players? Interpreting home-field advantage decomposition dynamics. Soil Biol. Biochem. 2018, 124, 189–198. [Google Scholar]
- Yuan, X.; Niu, D.; Wang, Y.; Boydston, A.; Guo, D.; Li, X.; Wen, H.; Qin, Y.; Fu, H. Litter decomposition in fenced and grazed grasslands: A test of the home-field advantage hypothesis. Geoderma 2019, 354, 113876. [Google Scholar] [CrossRef]
- Meng, Y.; Hui, D.; Huangfu, C. Site conditions interact with litter quality to affect home-field advantage and rhizosphere effect of litter decomposition in a subtropical wetland ecosystem. Sci. Total Environ. 2020, 749, 141442. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.; Wang, M.; Zheng, Y.; Lv, M.; Zhu, G.; Zhong, Q.; Cheng, D. Leaf litter phosphorus regulates the soil meso- and micro-faunal contribution to home-field advantage effects on litter decomposition along elevation gradients. Catena 2021, 207, 105673. [Google Scholar] [CrossRef]
- Pugnaire, F.I.; Aares, K.H.; Alifriqui, M.; Bråthen, K.A.; Kindler, C.; Schöb, C.; Manrique, E. Home-field advantage effects in litter decomposition is largely linked to litter quality. Soil Biol. Biochem. 2023, 184, 109069. [Google Scholar] [CrossRef]
- Pastorelli, R.; Costagli, V.; Forte, C.; Viti, C.; Rompato, B.; Nannini, G.; Certini, G. Litter decomposition: Little evidence of the “home-field advantage” in a mountain forest in Italy. Soil Biol. Biochem. 2021, 159, 108300. [Google Scholar]
- Wang, Y.; Li, F.Y.; Song, X.; Wang, X.; Suri, G.; Baoyin, T. Changes in litter decomposition rate of dominant plants in a semi-arid steppe across different land-use types: Soil moisture, not home-field advantage, plays a dominant role. Agric. Ecosyst. Environ. 2020, 303, 107119. [Google Scholar]
- Yarwood, S.A. The role of wetland microorganisms in plant-litter decomposition and soil organic matter formation: A critical review. FEMS Microbiol. Ecol. 2018, 94, 11. [Google Scholar]
- Ma, Y.; Cai, R.; Zhong, H.; Wu, L.; Ge, G. The home-field advantage of litter decomposition in lake wetlands and the community characteristics of bacterial and eukaryotic decomposers. Plant Soil 2023, 483, 109–130. [Google Scholar]
- Lam, W.N.; Lian, J.J.; Chan, P.J.; Ting, Y.Y.; Chong, R.; Rahman, N.E.; Tan, L.W.A.; Ho, Q.Y.; Ramchunder, S.J.; Peh, K.S.H.; et al. Leaf litter decomposition in tropical freshwater swamp forests is slower in swamp than non-swamp conditions. Biotropica 2021, 53, 920–929. [Google Scholar]
- Homet, P.; Gomez-Aparicio, L.; Matias, L.; Godoy, O. Soil fauna modulates the effect of experimental drought on litter decomposition in forests invaded by an exotic pathogen. J. Ecol. 2021, 109, 2963–2980. [Google Scholar]
- Laiho, R.; Laine, J.; Trettin, C.C.; Finer, L. Scots pine litter decomposition along drainage succession and soil nutrient gradients in peatland forests, and the effects of inter-annual weather variation. Soil Biol. Biochem. 2004, 36, 1095–1109. [Google Scholar]
- Stoler, A.B.; Relyea, R.A. Reviewing the role of plant litter inputs to forested wetland ecosystems: Leafing through the literature. Ecol. Monogr. 2020, 90, e01400. [Google Scholar]
- Zhang, G.; Yu, X.; Xu, J.; Duan, H.; Rafay, L.; Zhang, Q.; Li, Y.; Liu, Y.; Xia, S. Effects of environmental variation on stable isotope abundances during typical seasonal floodplain dry season litter decomposition. Sci. Total Environ. 2018, 630, 1205–1215. [Google Scholar] [PubMed]
- Macreadie, P.I.; Ollivier, Q.R.; Kelleway, J.J.; Serrano, O.; Carnell, P.E.; Lewis, C.J.E.; Atwood, T.B.; Sanderman, J.; Baldock, J.; Connolly, R.M.; et al. Carbon sequestration by Australian tidal marshes. Sci. Rep. 2017, 7, 44071. [Google Scholar]
- Peng, Y.; Zhou, C.; Jin, Q.; Ji, M.; Wang, F.; Lai, Q.; Shi, R.; Xu, X.; Chen, L.; Wang, G. Tidal variation and litter decomposition co-affect carbon emissions in estuarine wetlands. Sci. Total Environ. 2022, 839, 156357. [Google Scholar] [CrossRef] [PubMed]
- Day, T.A.; Guenon, R.; Ruhland, C.T. Photodegradation of plant litter in the Sonoran Desert varies by litter type and age. Soil Biol. Biochem. 2015, 89, 109–122. [Google Scholar] [CrossRef]
- Liyan, D.; Fanlong, K.; Sen, W.; Yue, L.; Min, X. Effect of litter decomposition on mineralization of soil organic carbon in the Jiaozhou Bay coastal wetlands. Acta Ecol. Sin. 2019, 39, 8483–8493. [Google Scholar]
- Patoine, G.; Thakur, M.P.; Friese, J.; Nock, C.; Hoenig, L.; Haase, J.; Scherer-Lorenzen, M.; Eisenhauer, N. Plant litter functional diversity effects on litter mass loss depend on the macro-detritivore community. Pedobiologia 2017, 65, 29–42. [Google Scholar] [CrossRef]
- Hanxia, Y.; Jiayi, W.; Fanghao, W.; Xiaoyan, Z.; Minling, C.; Qiaojing, O.; Weihua, L. Research progress on effects of plant litter on the decomposition of soil organic matter. J. Biosaf. 2018, 27, 88–94. [Google Scholar]
- Lynch, J.M.; Whipps, J.M. Substrate flow in the rhizosphere. Plant Soil 1990, 129, 1–10. [Google Scholar] [CrossRef]
- Xue, B.; Yan, C.; Lu, H.; Bai, Y. Mangrove-Derived Organic Carbon in Sediment from Zhangjiang Estuary (China) Mangrove Wetland. J. Coast. Res. 2009, 25, 949–956. [Google Scholar] [CrossRef]
- Hu, W.; Zhang, L.; Lai, D.Y.F.; Gao, J.; Sun, Z.; Tong, C.; Chen, Y.; Zeng, C. The Difference of Litter Decay, Litter- and Sediment-Associated Hydrolytic Enzymes between Brackish and Freshwater Tidal Marshes. Estuaries Coasts 2019, 42, 1328–1341. [Google Scholar] [CrossRef]
- Cui, Y.; Luo, F.-L.; Chen, Y.-H.; Zhang, M.-X.; Yu, F.-H. Rhizodeposition and litter decomposition of Phragmites australis play important roles in composition and properties of soil dissolved organic matter. Ecol. Indic. 2022, 142, 109275. [Google Scholar] [CrossRef]
- Yang, K.; Zhu, J.; Zhang, W.; Zhang, Q.; Lu, D.; Zhang, Y.; Zheng, X.; Xu, S.; Wang, G.G. Litter decomposition and nutrient release from monospecific and mixed litters: Comparisons of litter quality, fauna and decomposition site effects. J. Ecol. 2022, 110, 1673–1686. [Google Scholar] [CrossRef]
- Ren, Y.; Peng, Q.; You, C.; Li, H.; Zhang, L.; Liu, S.; Wang, L.; Tan, B.; Liu, Y.; Xu, Z. Diameter-associated dynamics of multiple metallic elements during the root decomposition of two dominant subalpine trees in southwestern China. Biogeochemistry 2023, 164, 415–430. [Google Scholar] [CrossRef]
- Wu, Q.; Wu, F.; Zhu, J.; Ni, X. Leaf and root inputs additively contribute to soil organic carbon formation in various forest types. J. Soils Sediments 2023, 23, 1135–1145. [Google Scholar] [CrossRef]
- Xie, Y.; Xie, Y.; Chen, X.; Li, F.; Hou, Z.; Li, X. Non-additive effects of water availability and litter quality on decomposition of litter mixtures. J. Freshw. Ecol. 2016, 31, 153–168. [Google Scholar]
| The Decomposition Process of Wetland Litter | Specific Performance |
|---|---|
| Initial period | First decomposition of water-soluble material with non-lignin carbohydrates, where lignin increases; the degree of material loss is more significant and the nutrient level limits the decomposition rate. |
| Medium term | Decomposers break down carbohydrates and lignin, lignin content decreases, decomposing litter composition tends to stabilize, material loss tends to slow down, and the decomposition rate is constrained by lignin. |
| End of the period | The change in lignin content in the decomposing litter gradually decreases, decomposition is almost at a standstill, and the remaining material is gradually eroded by humus. |
| Wetland Plant Species | Mean k/% | Median k/% | CV/% | t50/d | Reference |
|---|---|---|---|---|---|
| Typha orientalis | 0.0086 | 0.0086 | — | 81 | [36,53,54] |
| Acorus calamus L. | 0.0110 | 0.0110 | — | 63 | [55] |
| Phragmites australis | 0.0039 | 0.0018 | — | 180 | [56,57,58] |
| Juncus effusus | 0.0021 | 0.0021 | 65 | 338 | [59] |
| Spartina alterniflora Loisel | 0.0025 | 0.0028 | 118 | 21 | [60,61,62] |
| Categories | Wetland Types | Dominant Species | References |
|---|---|---|---|
| Fungi | Forest Marsh Wetlands | Ericoid mycorrhizae | [70] |
| Cantharellales | |||
| Graminoids | |||
| Bacteria | Coastal Estuarine Wetlands | Bacteroides | [71] |
| Planctomycetes | |||
| Gemmatiomonas | |||
| Artificial Wetland (15 years) | Proteobacteria | [72] | |
| Acidobacteria | |||
| Actinobacteria | |||
| Bacteriodetes | |||
| Verrucomicrobia | |||
| Created and Natural Wetlands | Proteobacteria | [73] | |
| Acidobacteria | |||
| Bacteria and Archaea | Tidal Freshwater Wetland | Flavobacteria-Bacteroides | [74] |
| Acidobacteria | |||
| Proteobacteria | |||
| Euryarchaeota | |||
| Restored Tidal Freshwater Wetlands | Proteobacteria | [75] | |
| Bacteroides | |||
| Euryarchaeota |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, X.; Dong, K.; Tang, Y.; Huang, H.; Peng, G.; Wang, D. Research Progress on the Decomposition Process of Plant Litter in Wetlands: A Review. Water 2023, 15, 3246. https://doi.org/10.3390/w15183246
Zhou X, Dong K, Tang Y, Huang H, Peng G, Wang D. Research Progress on the Decomposition Process of Plant Litter in Wetlands: A Review. Water. 2023; 15(18):3246. https://doi.org/10.3390/w15183246
Chicago/Turabian StyleZhou, Xinyu, Kun Dong, Yukun Tang, Haoyu Huang, Guosen Peng, and Dunqiu Wang. 2023. "Research Progress on the Decomposition Process of Plant Litter in Wetlands: A Review" Water 15, no. 18: 3246. https://doi.org/10.3390/w15183246





