A New Acidity-Based Approach for Estimating Total Dissolved Solids in Acidic Mining Influenced Water
Abstract
1. Introduction
2. Methodology
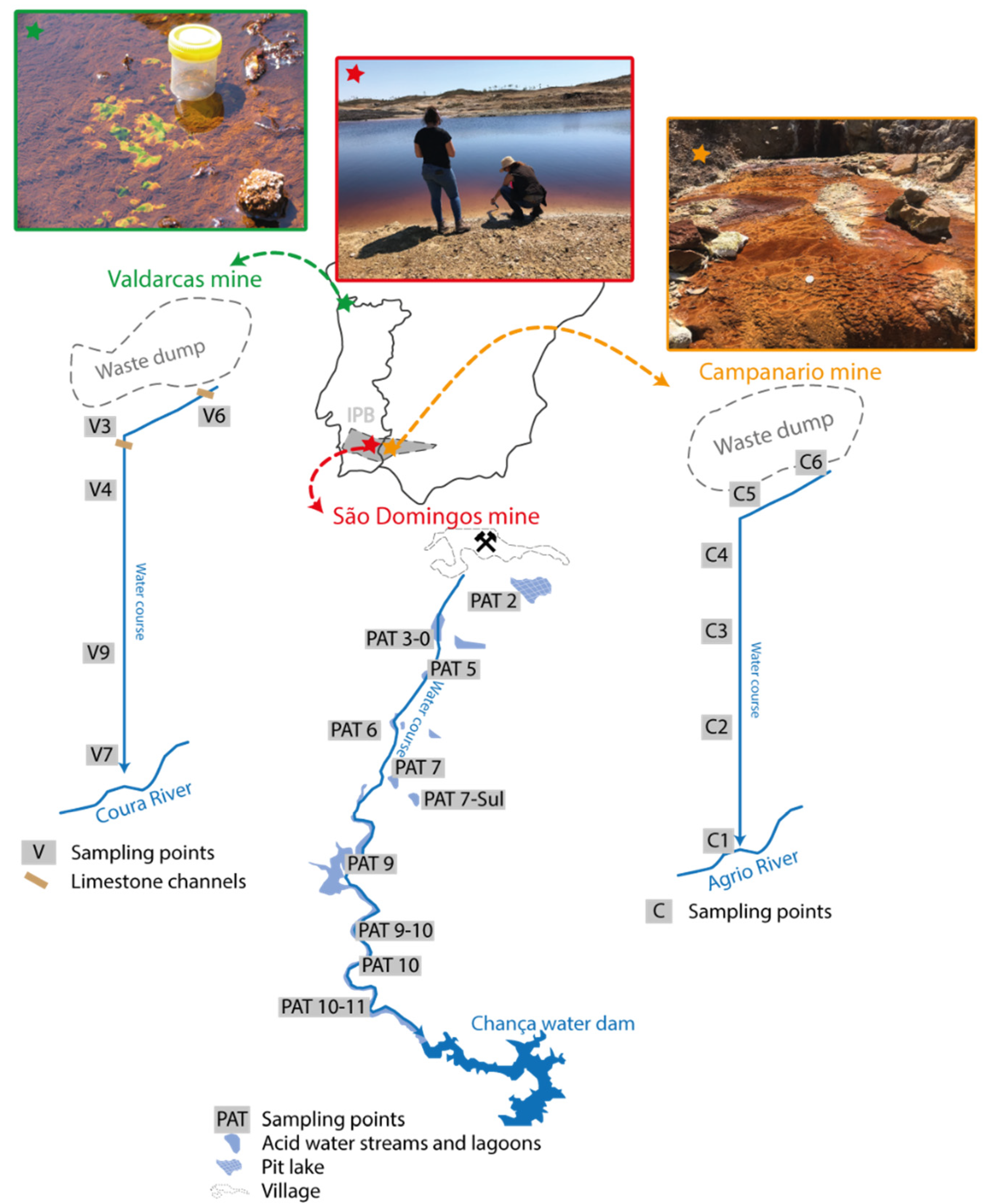
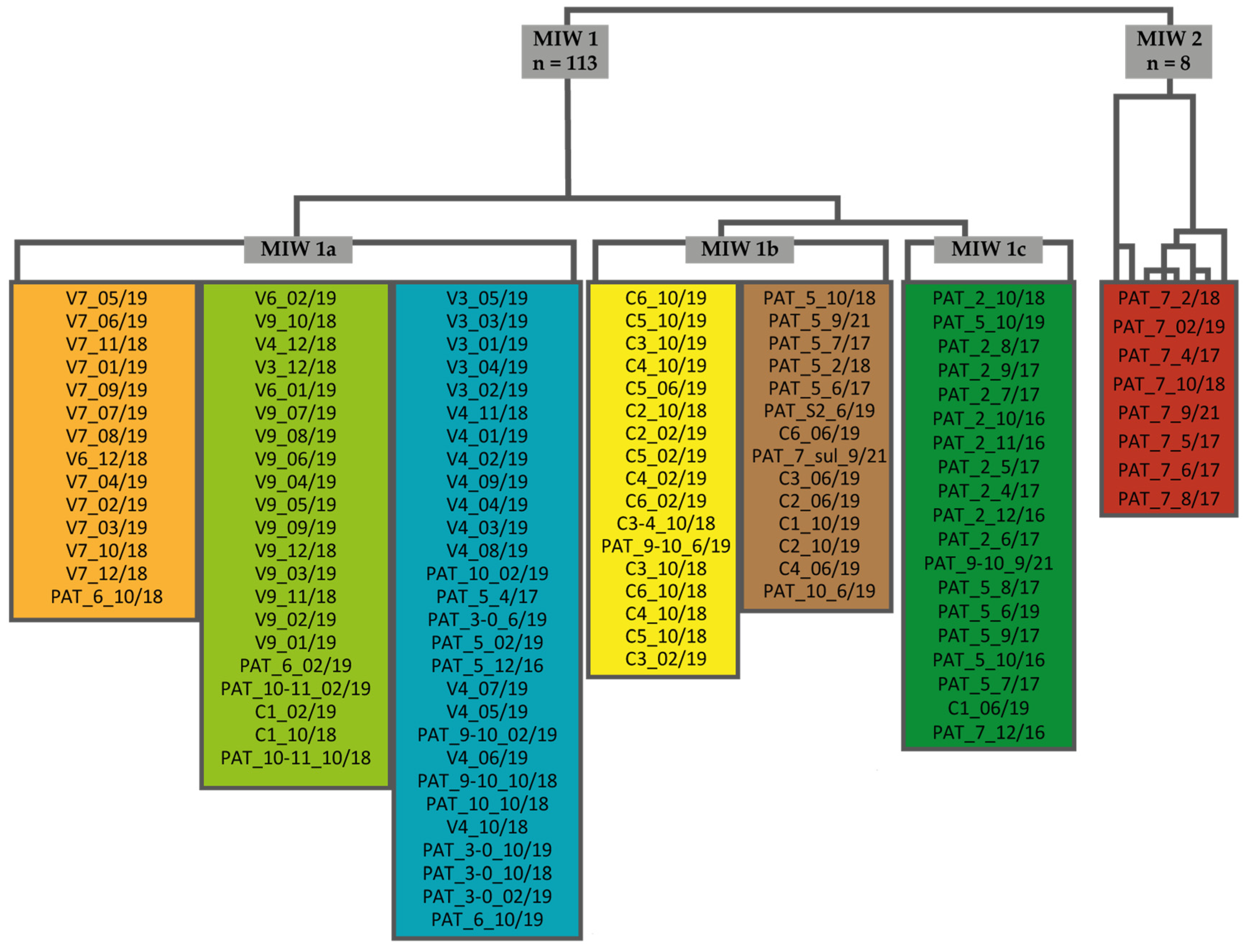
2.1. Study Areas
2.2. Water Sampling and Analysis
2.3. Statistical Treatment
3. Results and Discussion
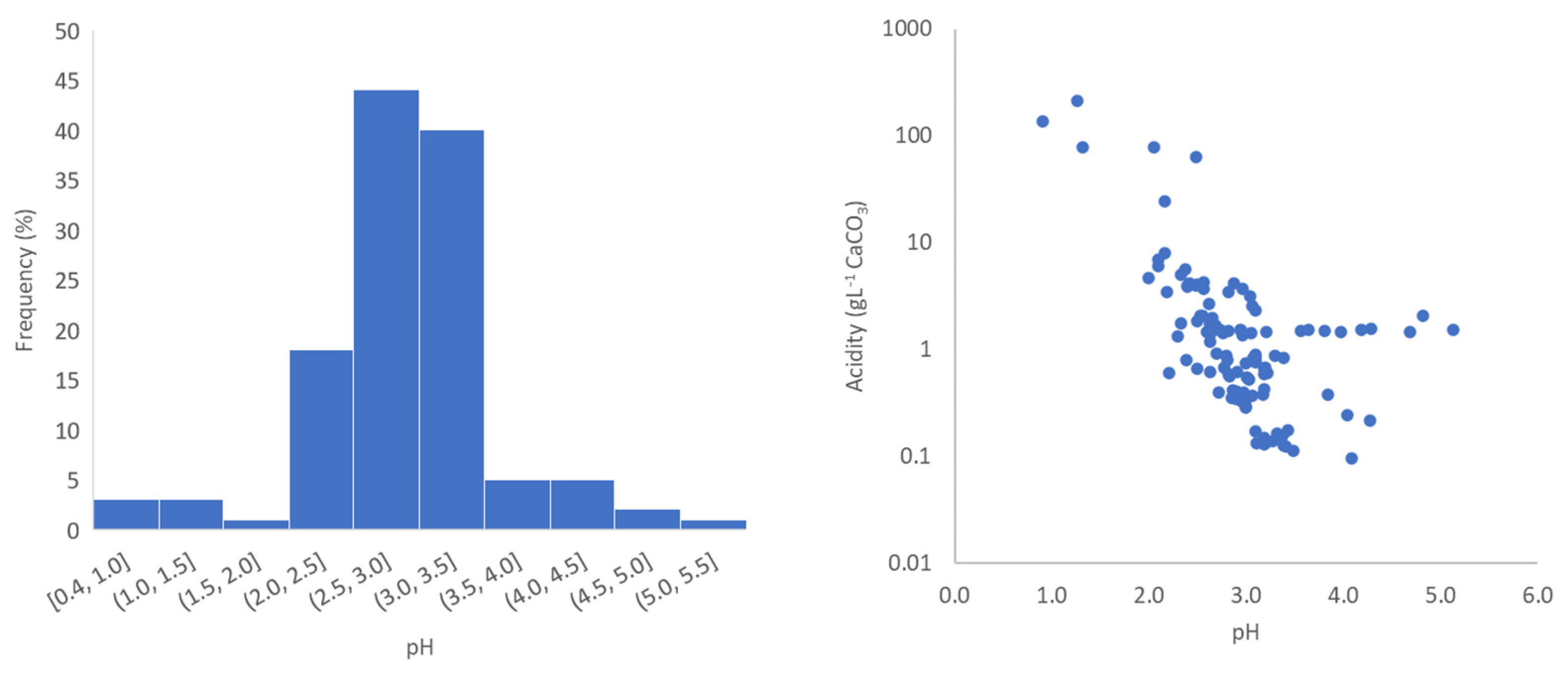
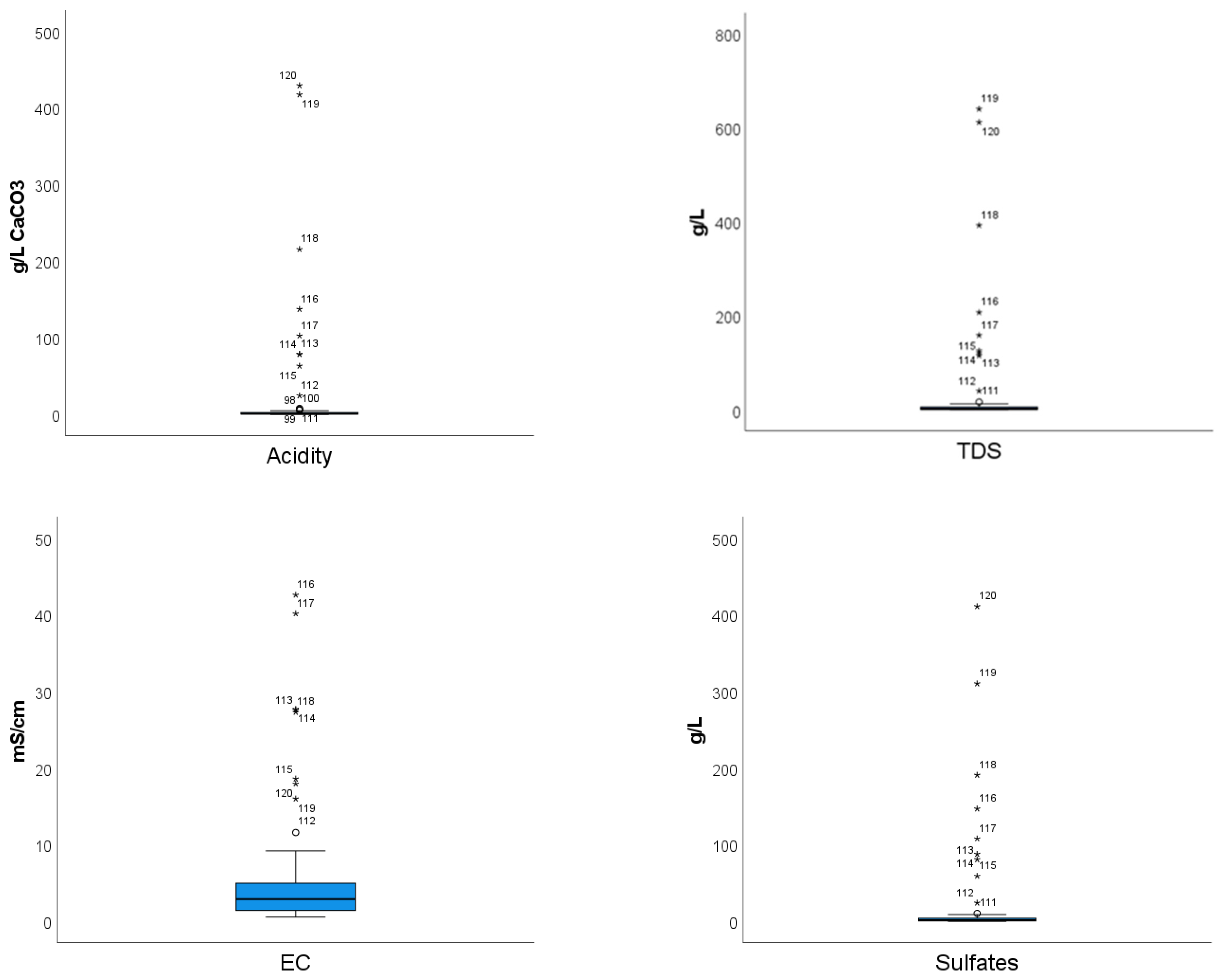
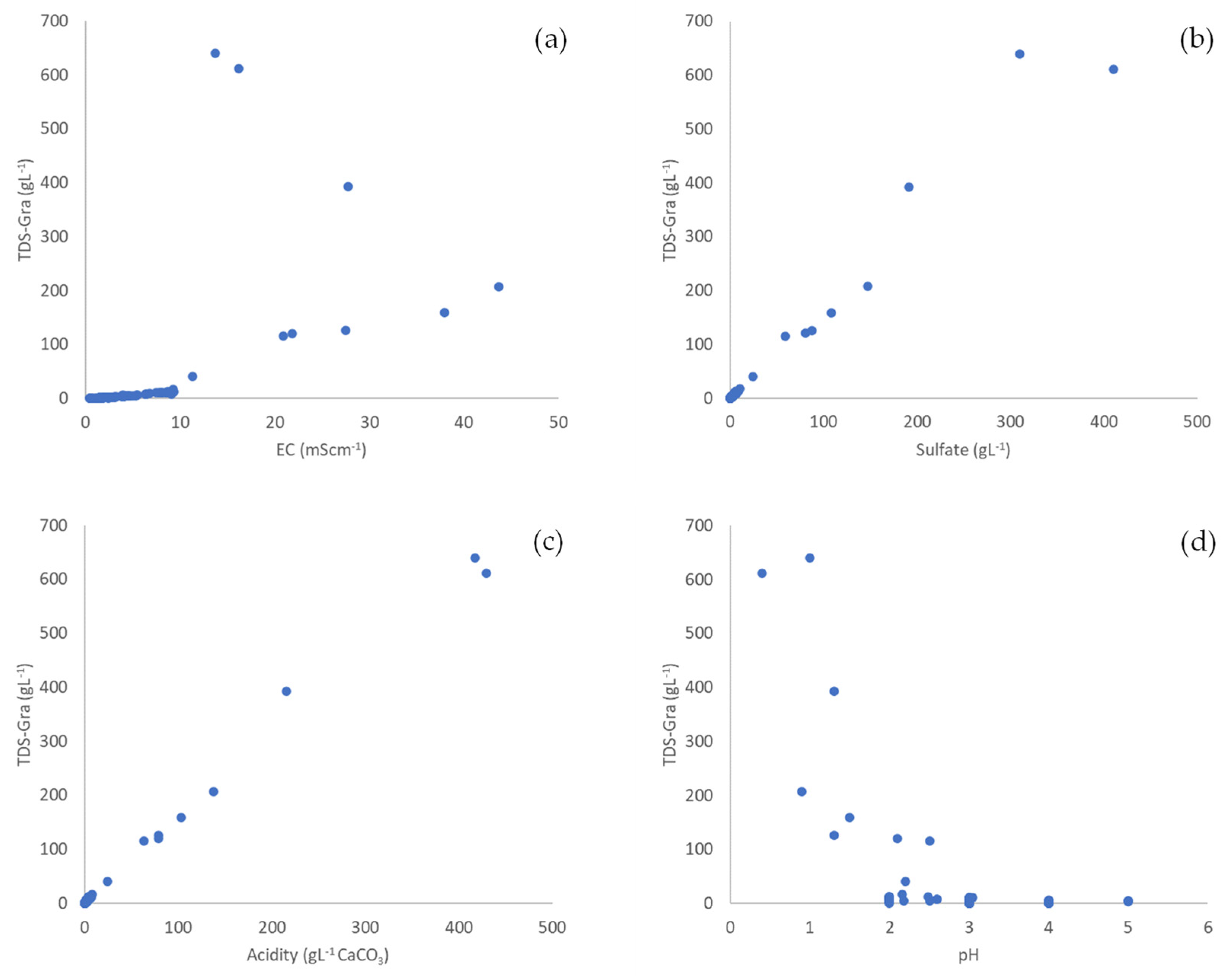
3.1. General Properties and Relationships in Mine Water
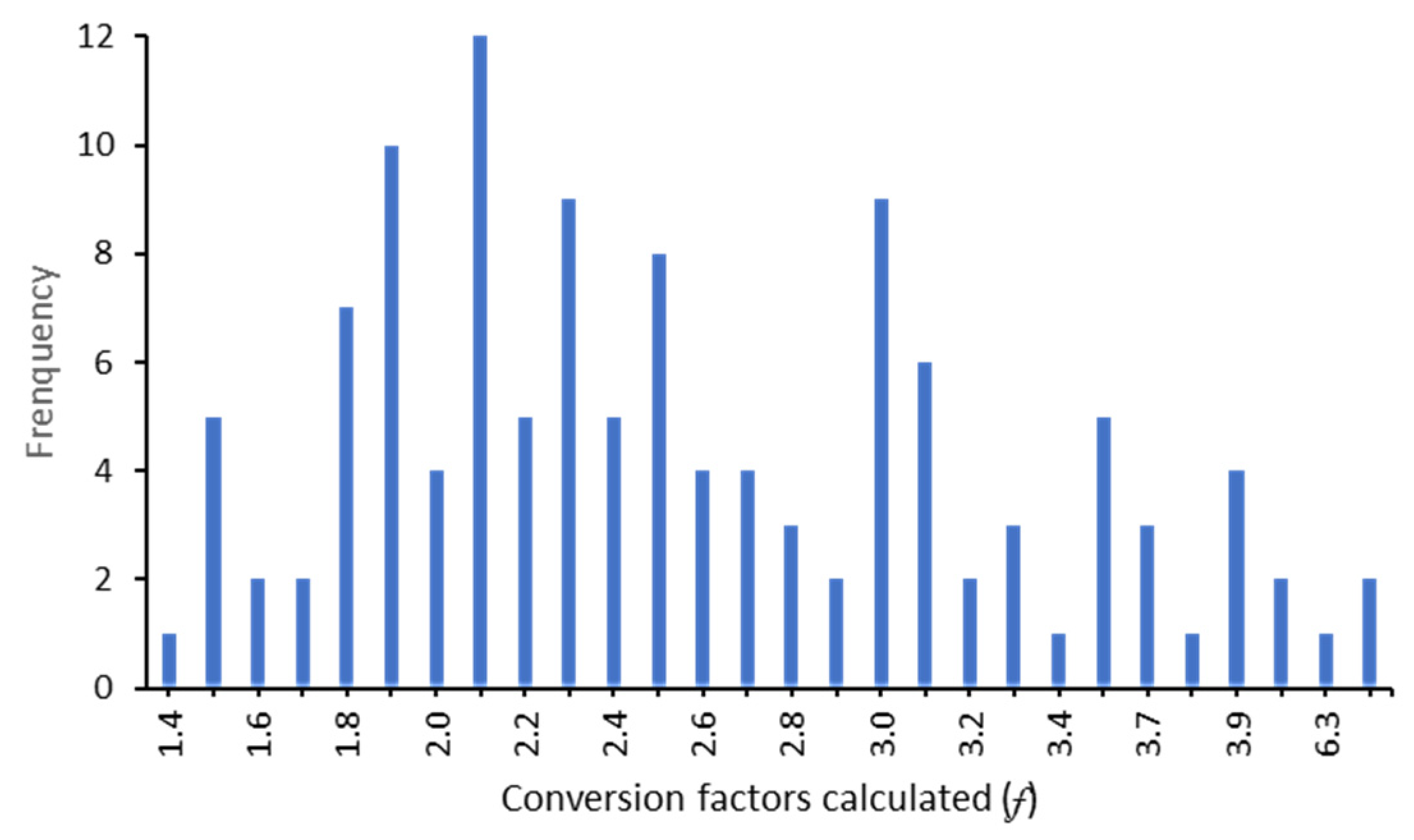
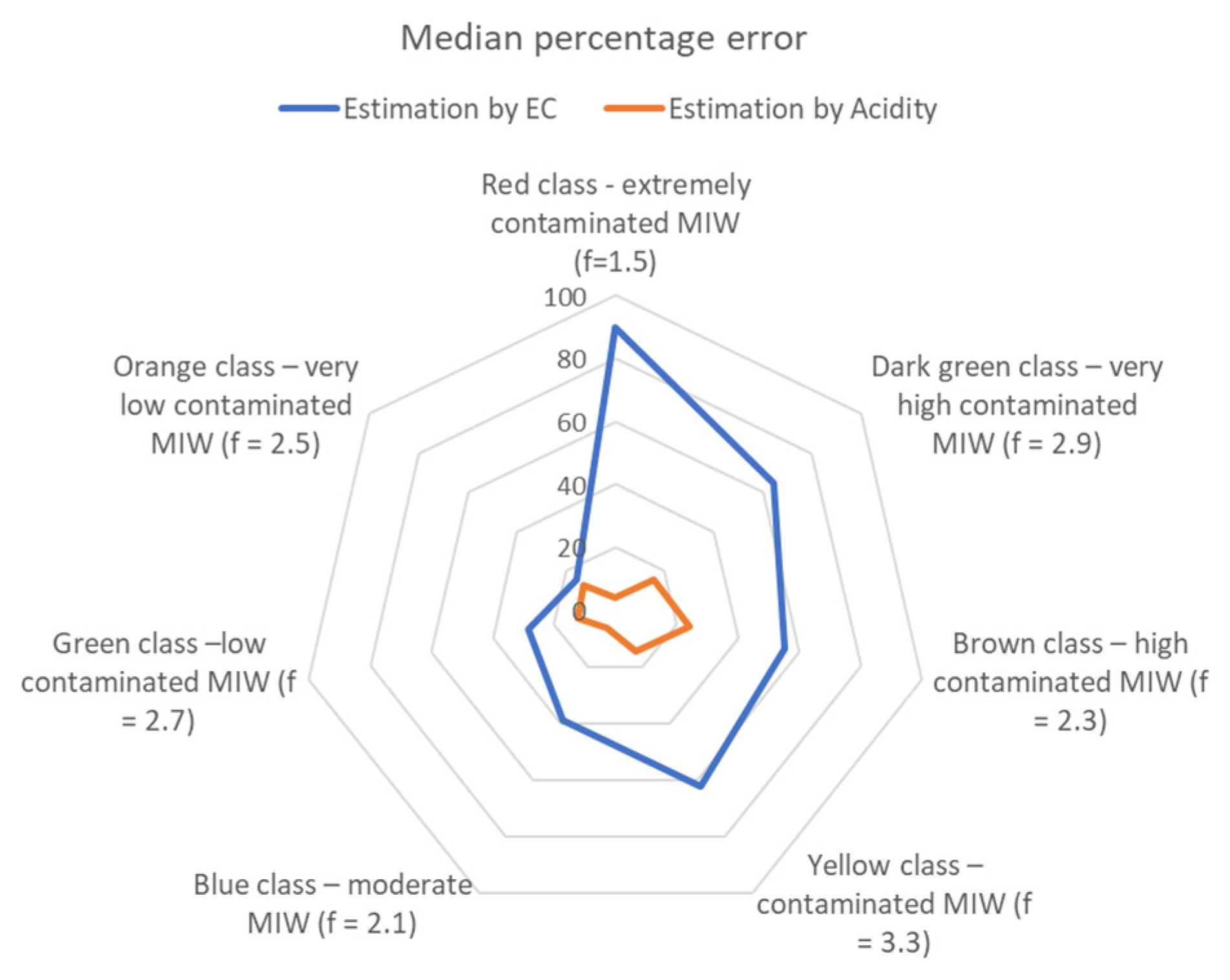
3.2. Conversion Factors
- 2.5 for very low MIW contamination, with very low sulfate (<0.250 gL−1) and acidity (<0.200 gL−1 of CaCO3);
- 2.7 for low MIW contamination, characterized by low sulfate (<0.800 gL−1) and acidity (<0.525 gL−1 of CaCO3);
- 2.1 for moderate MIW contamination, with sulfate concentration between 0.700 and 2.000 gL−1and acidity of 0.400 to 1.600 gL−1 of CaCO3;
- 3.3 for MIW contamination, with sulfate concentration range of 2.250 to 3.300 gL−1and acidity of 1.300 to 1.600 gL−1 of CaCO3;
- 2.3 for high MIW contamination, with high sulfate (2.250 to 5.200 gL−1) and acidity (1.400 to 4.300 gL−1 of CaCO3);
- 2.9 for very high MIW contamination, with sulfate concentration between 4.700 and 10.400 gL−1 and acidity of 2.000 to 8.000 gL−1 of CaCO3;
- and, for extreme MIW contamination, rich in sulfate (>24.000 gL−1) and very acidic (>24.400 gL−1 of CaCO3), the f is around 1.5.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Omer, N.H. Water quality parameters. In Water Quality—Science, Assessments and Policy; IntechOpen: London, UK, 2019; Volume 18, pp. 1–34. [Google Scholar]
- McNeil, V.H.; Cox, M.E. Relationship between conductivity and analysed composition in a large set of natural surface-water samples, Queensland, Australia. Environ. Geol. 2000, 39, 1325–1333. [Google Scholar] [CrossRef]
- Hubert, E.; Wolkersdorfer, C. Establishing a conversion factor between electrical conductivity and total dissolved solids in South African mine waters. Water SA 2015, 41, 490–500. [Google Scholar] [CrossRef]
- Rebello, L.R.B.; Siepman, T.; Drexler, S. Correlations between TDS and electrical conductivity for high-salinity for-mation brines characteristic of South Atlantic pre-salt basins. Water SA 2020, 46, 602–609. [Google Scholar]
- Hem, J.D. Study and Interpretation of the Chemical Characteristics of Natural Water; Department of the Interior, US Geological Survey: Reston, CA, USA, 1985; 2254, p. 263.
- APHA Standard Methods for the Examination of Water and Wastewater, 20th ed.; American Public Health Association, American Water Works Association, Water Environment Federation: Washington, DC, USA, 2012.
- Singh, T.; Kalra, Y.P. Specific conductance method for in situ estimation of total dissolved solids. J. Am. Water Work. Assoc. 1975, 67, 99–100. [Google Scholar] [CrossRef]
- Walton, N.R.G. Electrical Conductivity and Total Dissolved Solids—What is Their Precise Relationship? Desalination 1989, 72, 275–292. [Google Scholar] [CrossRef]
- Atekwana, E.A.; Atekwana, E.A.; Rowe, R.S.; Werkema, D.D., Jr.; Legall, F.D. The relationship of total dissolved solids measurements to bulk electrical conductivity in an aquifer contaminated with hydrocarbon. J. Appl. Geophys. 2004, 56, 281–294. [Google Scholar] [CrossRef]
- Marandi, A.; Polikarpus, M.; Jõeleht, A. A new approach for describing the relationship between electrical con-ductivity and major anion concentration in natural waters. Appl. Geochem. 2013, 38, 103–109. [Google Scholar] [CrossRef]
- Rusydi, A.F. Correlation between conductivity and total dissolved solid in various type of water: A review. IOP Conf. Ser. Earth Environ. Sci. 2018, 118, 012019. [Google Scholar] [CrossRef]
- Thirumalini, S.; Joseph, K. Correlation between Electrical Conductivity and Total Dissolved Solids in Natural Waters. Malays. J. Sci. 2009, 28, 55–61. [Google Scholar] [CrossRef]
- Weiner, E.R. Applications of Environmental Aquatic Chemistry—A Practical Guide; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Ali, N.S.; Mo, K.; Kim, M. A case study on the relationship between conductivity and dissolved solids to evaluate the potential for reuse of reclaimed industrial wastewater. KSCE J. Civ. Eng. 2012, 16, 708–713. [Google Scholar] [CrossRef]
- Taylor, M.; Elliott, H.A.; Navitsky, L.O. Relationship between total dissolved solids and electrical conductivity in Marcellus hydraulic fracturing fluids. Water Sci. Technol. 2018, 77, 1998–2004. [Google Scholar] [CrossRef] [PubMed]
- Wolkersdorfer, C. Mine Water Treatment—Active and Passive Methods; Springer: Berlin/Heidelberg, Germany, 2022; p. 328. ISBN 978-3-662-65770-6. [Google Scholar] [CrossRef]
- Nordstrom, D.K.; Alpers, C. Negative pH, efflorescent mineralogy, and consequences for environmental restoration at the Iron Mountain Superfund site, California. Proc. Natl. Acad. Sci. USA 1999, 96, 3455–3462. [Google Scholar] [CrossRef] [PubMed]
- Nordstrom, D.K. Mine Waters: Acidic to Circmneutral. Elements 2011, 7, 393–398. [Google Scholar] [CrossRef]
- Wolkersdorfer, C.; Nordstrom, D.K.; Beckie, R.D.; Cicerone, D.S.; Elliot, T.; Edraki, M.; Valente, T.; França, S.C.; Kumar, P.; Oyarzún Lucero, R.A.; et al. Guidance for the Integrated Use of Hydrological, Geochemical, and Isotopic Tools in Mining Operations. Mine Water Environ. 2020, 39, 204–228. [Google Scholar] [CrossRef]
- Fink, C.G. Chemical Composition versus Electrical Conductivity. Phys. Chem. 1917, 21, 32–36. [Google Scholar] [CrossRef][Green Version]
- Appelo, C.; Postma, D. Geochemistry, Groundwater and Pollution, 2nd ed.; Balkema: Rotterdam, The Netherlands, 2005; p. 649. ISBN 04 1536 428 0. [Google Scholar] [CrossRef]
- Hedin, R.S.; Watzlaf, G.R. The effects of anoxic limestone drains on mine water chemistry. J. Am. Soc. Min. Reclam. 1994, 6, 185–194. [Google Scholar] [CrossRef]
- Langmuir, D. Aqueous Environmental Geochemistry; Prentice Hall: Upper Saddle River, NJ, USA, 1997; p. 600. ISBN 0-02-367412-1. [Google Scholar]
- Barriga, F.J.A.S. Metallogenesis in the Iberian Pyrite Belt. In Premesozoic Geology of Iberia; Dallmeyer, R.D., Martinez-Garcia, E., Eds.; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1990; pp. 369–379. [Google Scholar] [CrossRef]
- Inverno, C.; Diez-Montes, A.; Rosa, C.; García-Crespo, J.; Matos, J.; García-Lobón, J.L.; Carvalho, J.; Bellido, F.; Caste-llo-Branco, J.M.; Ayala, C.; et al. Introduction and geological setting of the Iberian Pyrite Belt. In 3D, 4D and Predictive Modelling of Major Mineral Belts in Europe; Weihed, P., Ed.; Springer: Berlin/Heidelberg, Germany, 2015; Volume 9, pp. 191–208. [Google Scholar] [CrossRef]
- Gomes, P.; Valente, T.M.; Pereira, P. Addressing quality and usability of surface water bodies in semi-arid regions with mining influences. Environ. Process. 2018, 5, 707–725. [Google Scholar] [CrossRef]
- Valente, T.M.; Gomes, C.L. Fuzzy modelling of acid mine drainage environments using geochemical, ecological and mineralogical indicators. Environ. Geol. 2009, 57, 653. [Google Scholar] [CrossRef]
- Valente, T.M.; Gomes, C.L. Occurrence, properties and pollution potential of environmental minerals in acid mine drainage. Sci. Total Environ. 2009, 407, 1135–1152. [Google Scholar] [CrossRef]
- Alves, R.C.; Valente, T.M.F.; Braga, M.A.; Gomes, C.L. Mineralogical composition and metals retention in the fine-fraction streambed precipitates of on AMD affected system. In Proceedings of the International Mine Water Association Congress-IMWA, Aachen, Germany, 4–11 September 2011; ISBN 9781618393050. [Google Scholar]
- Gomes, P.; Valente, T.; Geraldo, D.; Ribeiro, C. Photosynthetic pigments in acid mine drainage: Seasonal patterns and associations with stressful abiotic characteristics. Chemosphere 2020, 239, 124774. [Google Scholar] [CrossRef] [PubMed]
- Lobo, A.; Valente, T.; de la Torre, M.L.; Grande, J.A.; Santisteban, M.; Salmerón, I.; Sánchez Requena, J. Spatial behavior of acid mine drainage in a peculiar stream: Physical-chemical evolution from the source until the temporarily receptor in the Iberian Pyrite Belt. In Proceedings of the Energy and Environment Knowledge Week, Toledo, Spain, 30–31 October 2014; ISBN 978-84-697-1162-0. [Google Scholar]
- Egbueri, J.C. Groundwater quality assessment using pollution index of groundwater (PIG), ecological risk index (ERI) and hierarchical cluster analysis (HCA): A case study. Groundw. Sustain. Dev. 2020, 10, 100292. [Google Scholar] [CrossRef]
- Gomes, P.; Valente, T.; Albuquerque, T.; Henriques, R.; Flor-Arnau, N.; Pamplona, J.; Macías, F. Algae in acid mine drainage and relationships with pollutants in a degraded mining ecosystem. Minerals 2021, 11, 110. [Google Scholar] [CrossRef]
| pH | TDS–Gra (g L−1) | TDS–est (g L−1) | EC (mS cm−1) | Sulfate (g L−1) | Acidity (g L−1 of CaCO3) | |
|---|---|---|---|---|---|---|
| Minimum | 0.44 | 296 | 0.276 | 0.412 | 0.153 | 0.096 |
| Maximum | 4.82 | 640.086 | 20.850 | 43.710 | 41.0601 | 429.250 |
| Mean | 2.90 | 23.475 | 2.507 | 4.963 | 13.806 | 14.172 |
| Median | 2.92 | 2.414 | 1.413 | 3.052 | 1.389 | 1.180 |
| Standard deviation | 0.70 | 90.358 | 3.512 | 6.663 | 52.291 | 59.430 |
| pH | TDS | EC | Sulfate | Acidity | |
|---|---|---|---|---|---|
| pH | 1 | ||||
| TDS | −0.599 * | 1 | |||
| ρ < 0.001 | |||||
| EC | −0.652 * | 0.978 * | 1 | ||
| ρ < 0.001 | ρ < 0.001 | ||||
| Sulfate | −0.624 * | 0.981 * | 0.984 * | 1 | |
| ρ < 0.001 | ρ < 0.001 | ρ < 0.001 | |||
| Acidity | −0.637 * | 0.964 * | 0.971 * | 0.973 * | 1 |
| ρ < 0.001 | ρ < 0.001 | ρ < 0.001 | ρ < 0.001 |
| Orange Class f = 2.5 | ||||
|---|---|---|---|---|
| pH | CE | Sulfate | Acidity | |
| Median | 3 | 0.564 | 0.210 | 0.143 |
| Range | [3.0–4.0] | [0.412–0.872] | [0.153–0.247] | [0.096–0.173] |
| Green Class f = 2.7 | ||||
| pH | CE | Sulfate | Acidity | |
| Median | 3 | 1.302 | 0.553 | 0.325 |
| Range | [3.0–4.0] | [1.091–1.565] | [0.380–0.731] | [0.113–0.525] |
| Blue Class f = 2.1 | ||||
| pH | CE | Sulfate | Acidity | |
| Median | 3 | 2.236 | 1.034 | 0.754 |
| Range | [2.0–3.0] | [1.751–3.291] | [0.762–1.969] | [0.406–1.605] |
| Yellow Class f = 3.3 | ||||
| pH | CE | Sulfate | Acidity | |
| Median | 3 | 3.948 | 2.675 | 1.500 |
| Range | [3.0–5.0] | [3.848–4.189] | [2.255–3.265] | [1.360–1.560] |
| Brown Class f = 2.3 | ||||
| pH | CE | Sulfate | Acidity | |
| Median | 3 | 4.579 | 3.324 | 1.954 |
| Range | [2.0–5.0] | [3.856–6.330] | [2.244–5.152] | [1.405–4.305] |
| Dark green Class f = 2.9 | ||||
| pH | CE | Sulfate | Acidity | |
| Median | 2.3 | 8.306 | 6.158 | 4.128 |
| Range | [2.0–3.0] | [6.330–9.298] | [4.742–10.399] | [2.080–7.935] |
| Red Class f = 1.5 | ||||
| pH | CE | Sulfate | Acidity | |
| Median | 1.3 | 21.830 | 107.962 | 102.700 |
| Range | [0.4–2.5] | [11.320–43.710] | [24.075–410.601] | [24.438–429.250] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barroso, A.; Valente, T.; Marinho Reis, A.P.; Antunes, I.M.H.R. A New Acidity-Based Approach for Estimating Total Dissolved Solids in Acidic Mining Influenced Water. Water 2023, 15, 2995. https://doi.org/10.3390/w15162995
Barroso A, Valente T, Marinho Reis AP, Antunes IMHR. A New Acidity-Based Approach for Estimating Total Dissolved Solids in Acidic Mining Influenced Water. Water. 2023; 15(16):2995. https://doi.org/10.3390/w15162995
Chicago/Turabian StyleBarroso, Ana, Teresa Valente, Amélia Paula Marinho Reis, and Isabel Margarida H. R. Antunes. 2023. "A New Acidity-Based Approach for Estimating Total Dissolved Solids in Acidic Mining Influenced Water" Water 15, no. 16: 2995. https://doi.org/10.3390/w15162995
APA StyleBarroso, A., Valente, T., Marinho Reis, A. P., & Antunes, I. M. H. R. (2023). A New Acidity-Based Approach for Estimating Total Dissolved Solids in Acidic Mining Influenced Water. Water, 15(16), 2995. https://doi.org/10.3390/w15162995








