A Novel Layered and Advanced Nitrogen Removal Filter with Gravel and Embedded Bio-Organic Carrier Based on Autotrophic and Heterotrophic Pathways
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Setup
2.2. Experimental Materials
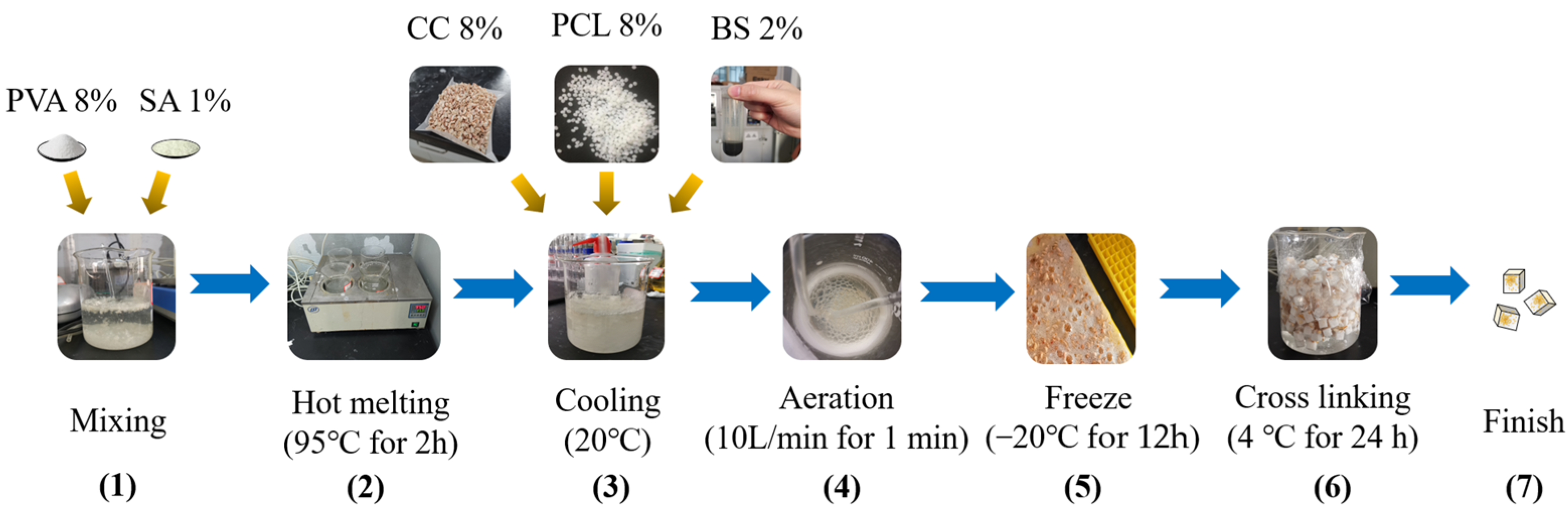
2.3. Preparation of Embedded Bio-Organic Carrier and Filmed Gravel
2.4. Experimental Design and Operation
2.5. Analytical Methods
2.5.1. Sampling and Analytical Methods
2.5.2. Anammox Activity Assay
2.5.3. High-Throughput Sequencing
2.5.4. Nitrogen Conversion Pathway
3. Results and Discussion
3.1. Pollutant Removal Performance
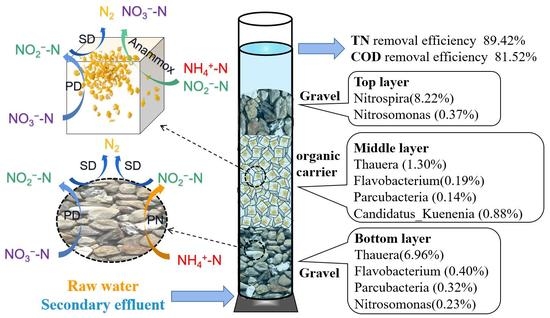
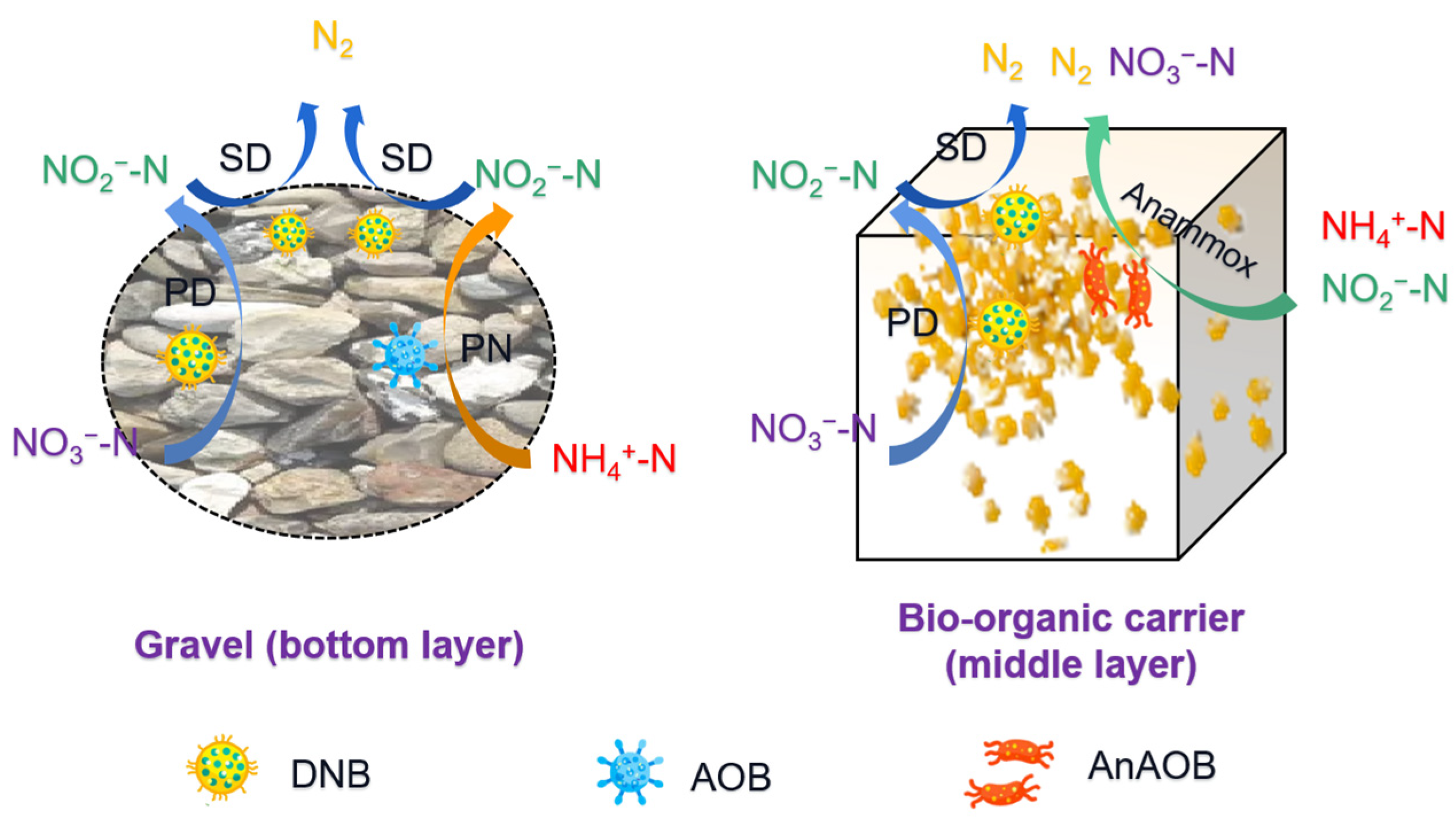
3.2. Comparison of Nitrogen Removal between Each Layer
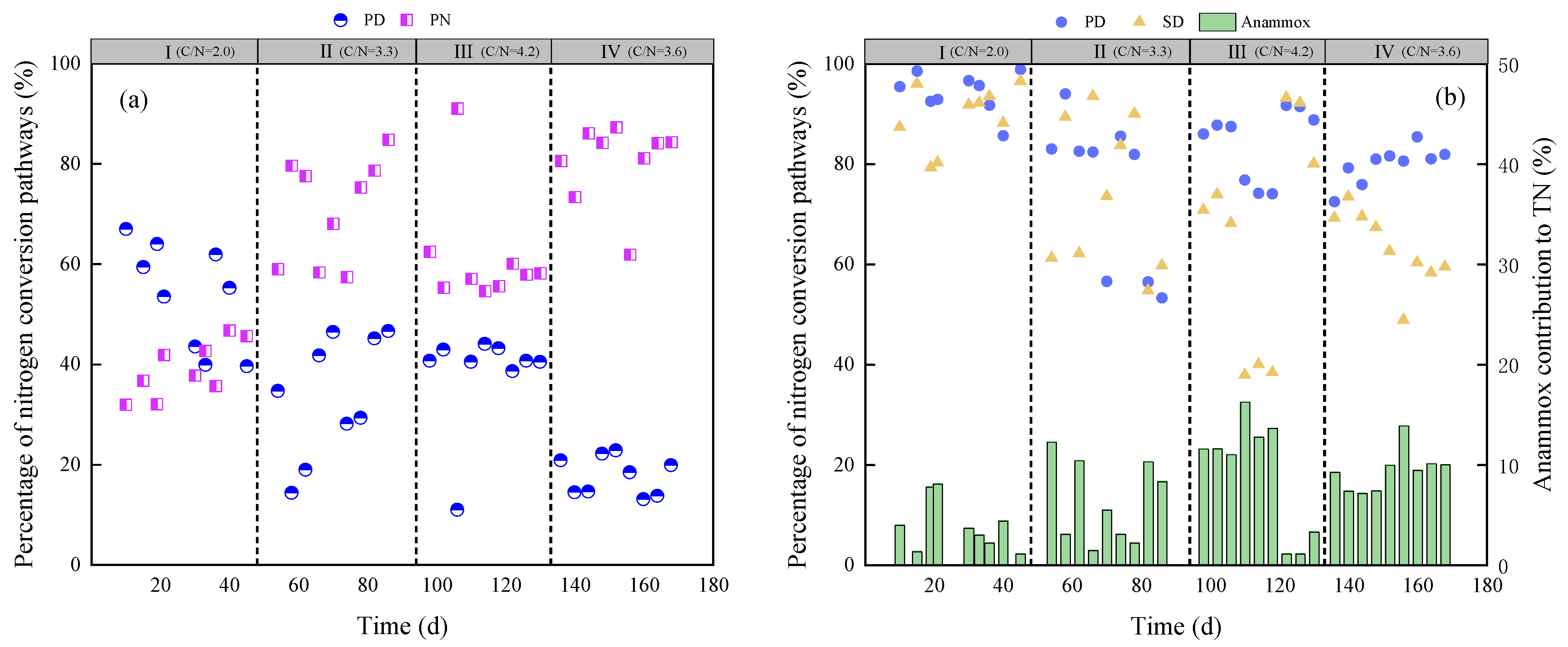
3.3. Nitrogen Conversion Pathway
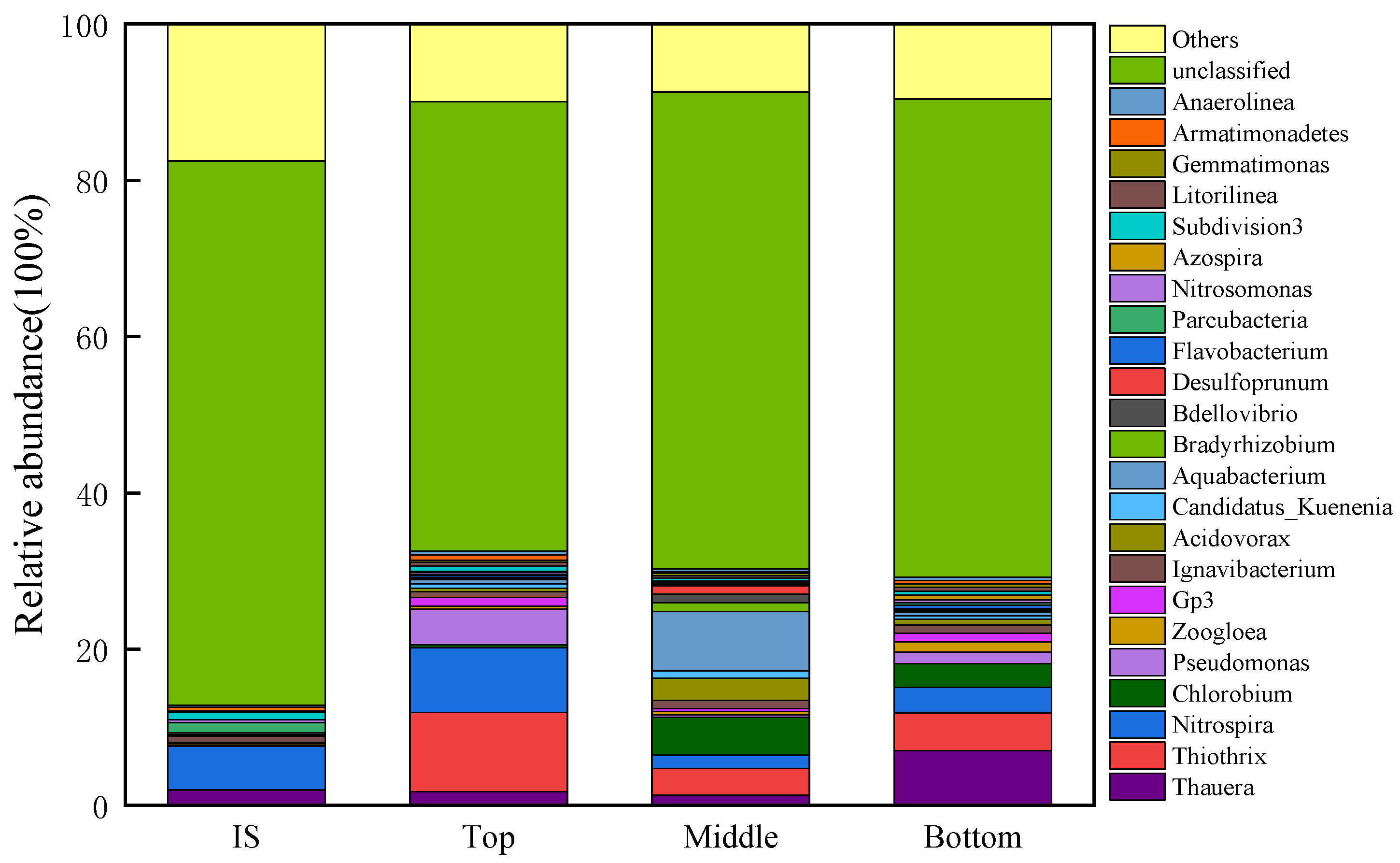
3.4. Microbial Communities in Each Layer
3.5. Influent Mixing Ratio Model
- NH4+-N in the layered filter comes from the raw water (RW) and secondary effluent (SE); its transfer pathway includes PN and Anammox.
- NO3−-N comes from the SE and Anammox.
- COD comes from the RW and SE.
- NH4+-N in the layered filter is removed completely.
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tong, Y.; Zhang, W.; Wang, X.; Couture, R.-M.; Larssen, T.; Zhao, Y.; Li, J.; Liang, H.; Liu, X.; Bu, X.; et al. Decline in Chinese lake phosphorus concentration accompanied by shift in sources since 2006. Nat. Geosci. 2017, 10, 507–511. [Google Scholar] [CrossRef]
- Zheng, X.; Zhang, S.; Zhang, J.; Huang, D.; Zheng, Z. Advanced nitrogen removal from municipal wastewater treatment plant secondary effluent using a deep bed denitrification filter. Water Sci. Technol. 2018, 77, 2723–2732. [Google Scholar] [CrossRef] [PubMed]
- Zhu-Barker, X.; Bailey, S.K.; Paw, U.K.; Burger, M.; Horwath, W.R. Greenhouse gas emissions from green waste composting windrow. Waste Manag. 2017, 59, 70–79. [Google Scholar] [CrossRef]
- Qiao, S.; Tian, T.; Duan, X.; Zhou, J.; Cheng, Y. Novel single-stage autotrophic nitrogen removal via co-immobilizing partial nitrifying and anammox biomass. Chem. Eng. J. 2013, 230, 19–26. [Google Scholar] [CrossRef]
- Tomaszewski, M.; Cema, G.; Ziembinska-Buczynska, A. Influence of temperature and pH on the anammox process: A review and meta-analysis. Chemosphere 2017, 182, 203–214. [Google Scholar] [CrossRef]
- Du, R.; Cao, S.; Peng, Y.; Zhang, H.; Wang, S. Combined Partial Denitrification (PD)-Anammox: A method for high nitrate wastewater treatment. Environ. Int. 2019, 126, 707–716. [Google Scholar] [CrossRef]
- Cui, B.; Yang, Q.; Liu, X.; Wu, W.; Liu, Z.; Gu, P. Achieving partial denitrification-anammox in biofilter for advanced wastewater treatment. Environ. Int. 2020, 138, 105612. [Google Scholar] [CrossRef]
- Cao, S.; Oehmen, A.; Zhou, Y. Denitrifiers in Mainstream Anammox Processes: Competitors or Supporters? Environ. Sci. Technol. 2019, 53, 11063–11065. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, Q.; Peng, Y.; Peng, Y.; Li, X.; Jiang, H. Advanced nitrogen elimination from domestic sewage through two stage partial nitrification and denitrification (PND) coupled with simultaneous anaerobic ammonia oxidation and denitrification (SAD). Bioresour. Technol. 2022, 343, 125986. [Google Scholar] [CrossRef]
- Wu, W.; Yang, F.; Yang, L. Biological denitrification with a novel biodegradable polymer as carbon source and biofilm carrier. Bioresour. Technol. 2012, 118, 136–140. [Google Scholar] [CrossRef]
- Pierro, L.; Matturro, B.; Rossetti, S.; Sagliaschi, M.; Sucato, S.; Alesi, E.; Bartsch, E.; Arjmand, F.; Papini, M.P. Polyhydroxyalkanoate as a slow-release carbon source for in situ bioremediation of contaminated aquifers: From laboratory investigation to pilot-scale testing in the field. New Biotechnol. 2017, 37, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.M.; Yang, F. Biological Denitrification Using Slow-Release Carbon Source Composite Materials as Solid Carbon Source and Biofilm Carrier. Adv. Mater. Res. 2013, 702, 218–223. [Google Scholar] [CrossRef]
- Bao, T.; Chen, T.; Wille, M.-L.; Chen, D.; Yu, S.; Bian, J.; Qing, C.; Wu, W.; Frost, R.L. Production of zeolite composite filters using waste paper pulp as slow release carbon source and performance investigation in a biological aerated filter. J. Water Process Eng. 2016, 9, 38–46. [Google Scholar] [CrossRef]
- Reyes-Alvarado, L.C.; Okpalanze, N.N.; Rene, E.R.; Rustrian, E.; Houbron, E.; Esposito, G.; Lens, P.N.L. Carbohydrate based polymeric materials as slow release electron donors for sulphate removal from wastewater. J. Environ. Manag. 2017, 200, 407–415. [Google Scholar] [CrossRef]
- Lin, L.; Li, X.Y. Acidogenic fermentation of iron-enhanced primary sedimentation sludge under different pH conditions for production of volatile fatty acids. Chemosphere 2018, 194, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.X.; Chen, L.; Zhu, J.; Walquist, M.; Christian, D. Pre-digestion to enhance volatile fatty acids (VFAs) concentration as a carbon source for denitrification in treatment of liquid swine manure. J. Environ. Sci. Health Part A Toxic Hazard. Subst. Environ. Eng. 2018, 53, 891–898. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liang, J.; Ning, D.; Zhang, T.; Wang, M. A review of biomass immobilization in anammox and partial nitrification/anammox systems: Advances, issues, and future perspectives. Sci. Total Environ. 2022, 821, 152792. [Google Scholar] [CrossRef] [PubMed]
- Qiao, D.; Liu, H.; Yu, L.; Bao, X.; Simon, G.P.; Petinakis, E.; Chen, L. Preparation and characterization of slow-release fertilizer encapsulated by starch-based superabsorbent polymer. Carbohydr. Polym. 2016, 147, 146–154. [Google Scholar] [CrossRef]
- Yang, Y.; Li, Y.; Gu, Z.; Lu, F.; Xia, S.; Hermanowicz, S. Quick start-up and stable operation of a one-stage deammonification reactor with a low quantity of AOB and ANAMMOX biomass. Sci. Total Environ. 2019, 654, 933–941. [Google Scholar] [CrossRef]
- Ahmad, M.; Liu, S.; Mahmood, N.; Mahmood, A.; Ali, M.; Zheng, M.; Ni, J. Effects of porous carrier size on biofilm development, microbial distribution and nitrogen removal in microaerobic bioreactors. Bioresour. Technol. 2017, 234, 360–369. [Google Scholar] [CrossRef]
- Van de Graaf, A.A.; de Bruijn, P.; Robertson, L.A.; Jetten, M.S.M.; Kuenen, J.G. Autotrophic growth of anaerobic ammonium-oxidizing micro-organisms in a fluidized bed reactor. Microbiology 1996, 142, 2187–2196. [Google Scholar] [CrossRef]
- Kalyuzhnyi, S.; Gladchenko, M. DEAMOX—New microbiological process of nitrogen removal from strong nitrogenous wastewater. Desalination 2009, 248, 783–793. [Google Scholar] [CrossRef]
- Xiong, R.; Yu, X.; Yu, L.; Peng, Z.; Cheng, L.; Li, T.; Fan, P. Biological denitrification using polycaprolactone-peanut shell as slow-release carbon source treating drainage of municipal WWTP. Chemosphere 2019, 235, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Xiong, R.; Yu, X.; Zhang, Y.; Peng, Z.; Yu, L.; Cheng, L.; Li, T. Comparison of agricultural wastes and synthetic macromolecules as solid carbon source in treating low carbon nitrogen wastewater. Sci. Total Environ. 2020, 739, 139885. [Google Scholar] [CrossRef]
- Jenni, S.; Vlaeminck, S.E.; Morgenroth, E.; Udert, K.M. Successful application of nitritation/anammox to wastewater with elevated organic carbon to ammonia ratios. Water Res. 2014, 49, 316–326. [Google Scholar] [CrossRef]
- Hu, H.; Liao, K.; Geng, J.; Xu, K.; Huang, H.; Wang, J.; Ren, H. Removal Characteristics of Dissolved Organic Nitrogen and Its Bioavailable Portion in a Postdenitrifying Biofilter: Effect of the C/N Ratio. Environ. Sci. Technol. 2018, 52, 757–764. [Google Scholar] [CrossRef]
- Yu, G.; Peng, H.; Fu, Y.; Yan, X.; Du, C.; Chen, H. Enhanced nitrogen removal of low C/N wastewater in constructed wetlands with co-immobilizing solid carbon source and denitrifying bacteria. Bioresour. Technol. 2019, 280, 337–344. [Google Scholar] [CrossRef]
- Wang, W.; Wang, X.; Wang, S.; Li, J. Partial denitrification coupled with immobilization of anammox in a continuous upflow reactor. RSC Adv. 2018, 8, 32016–32021. [Google Scholar] [CrossRef]
- Park, S.; Bae, W.; Rittmann, B.E. Multi-species nitrifying biofilm model (MSNBM) including free ammonia and free nitrous acid inhibition and oxygen limitation. Biotechnol. Bioeng. 2010, 105, 1115–1130. [Google Scholar] [CrossRef]
- Li, J.; Zhang, L.; Peng, Y.; Zhang, Q. Effect of low COD/N ratios on stability of single-stage partial nitritation/anammox (SPN/A) process in a long-term operation. Bioresour. Technol. 2017, 244, 192–197. [Google Scholar] [CrossRef]
- Li, B.; Wang, Y.; Guo, Y.; Wang, W.; Huang, X.; Wang, Z. Partial nitrification coupled with anammox in a biofilter reactor (BR) of large height-to-diameter ratio for treatment of wastewater with low C/N. Process Biochem. 2021, 104, 19–26. [Google Scholar] [CrossRef]
- Niu, W.; Guo, J.; Lian, J.; Song, Y.; Lu, C.; Li, H.; Han, Y.; Yin, P. Rapid start-up of denitrifying granular sludge by dosing with semi-starvation fluctuation C/N ratio strategy. Bioresour. Technol. 2017, 241, 945–950. [Google Scholar] [CrossRef] [PubMed]

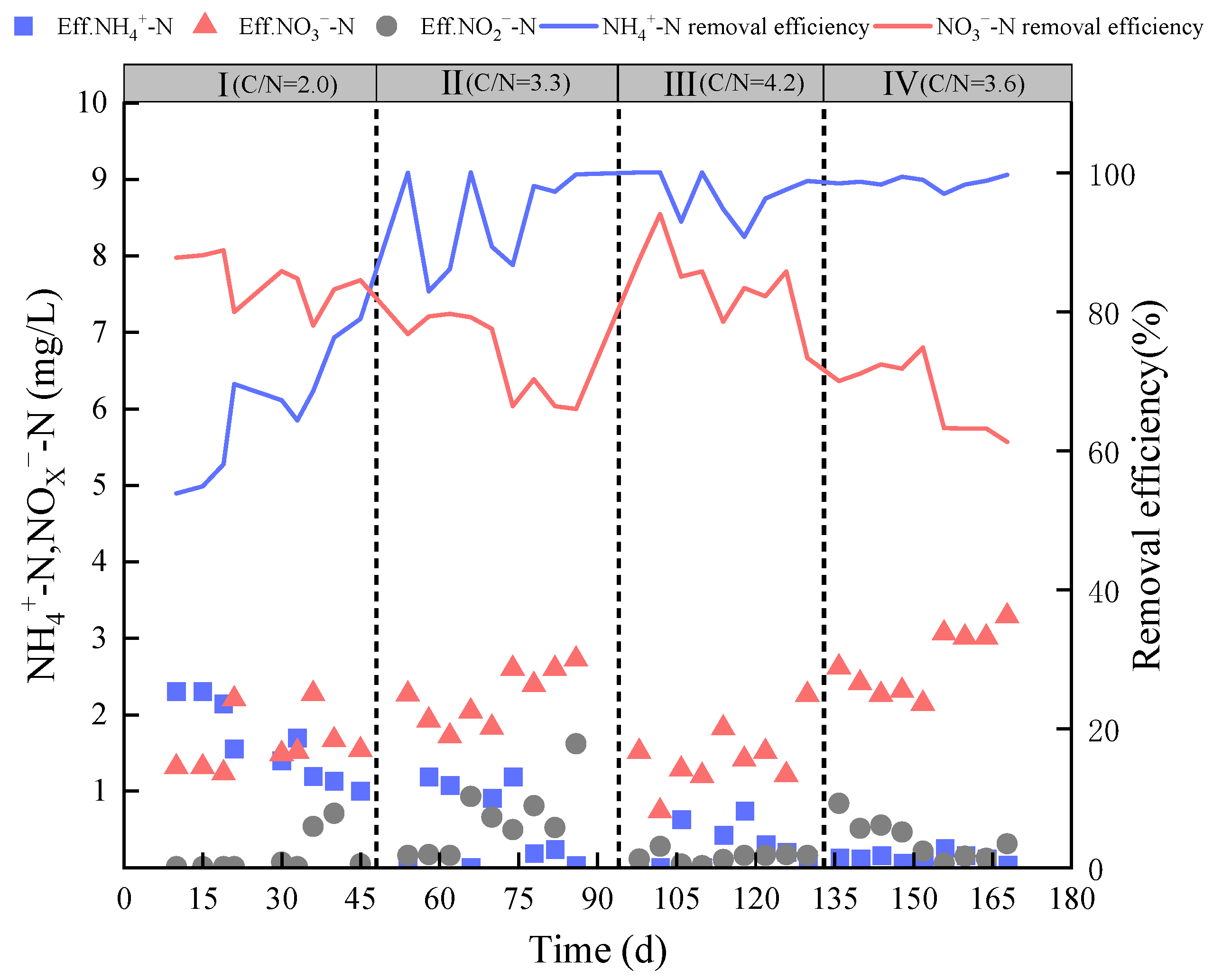
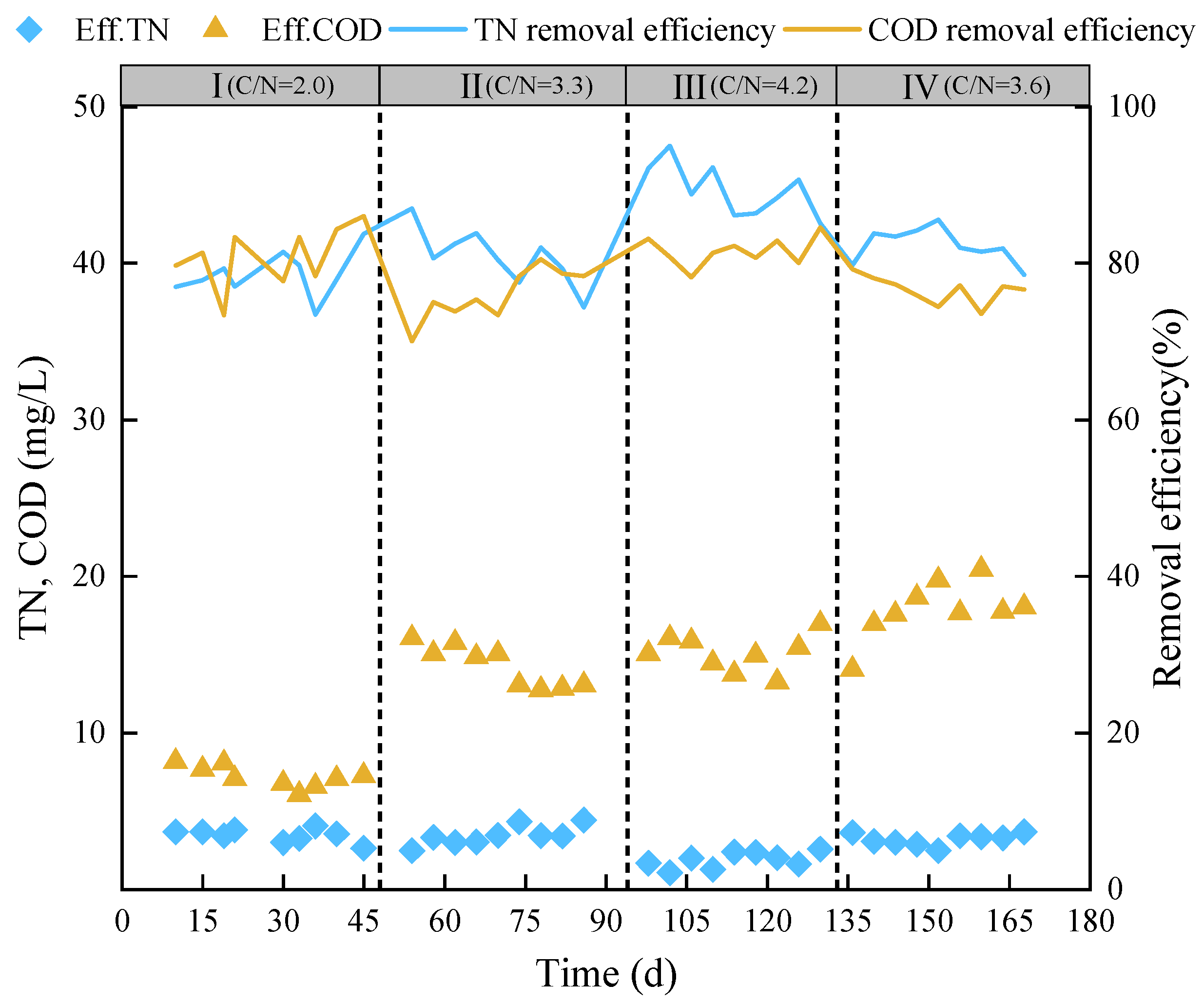

| Experimental Phase | NH4+-N (mg·L−1) | NO2−-N (mg·L−1) | NO3−-N (mg·L−1) | COD (mg·L−1) | TP (mg·L−1) | C/N | Time (d) |
|---|---|---|---|---|---|---|---|
| I | 5 | 0 | 10 | 30 | 0.50 | 2.00 | 1–48 |
| Ⅱ | 10 | 0 | 8 | 60 | 1.25 | 3.33 | 49–94 |
| Ⅲ | 10 | 0 | 8 | 77 | 1.40 | 4.23 | 95–133 |
| Ⅳ | 13 | 0 | 8 | 77 | 1.90 | 3.67 | 134–168 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peng, Z.; Liu, M.; Li, T.; Zhang, W.; Wang, Y.; Yu, L.; Ji, J. A Novel Layered and Advanced Nitrogen Removal Filter with Gravel and Embedded Bio-Organic Carrier Based on Autotrophic and Heterotrophic Pathways. Water 2023, 15, 1919. https://doi.org/10.3390/w15101919
Peng Z, Liu M, Li T, Zhang W, Wang Y, Yu L, Ji J. A Novel Layered and Advanced Nitrogen Removal Filter with Gravel and Embedded Bio-Organic Carrier Based on Autotrophic and Heterotrophic Pathways. Water. 2023; 15(10):1919. https://doi.org/10.3390/w15101919
Chicago/Turabian StylePeng, Zhaoxu, Minghui Liu, Tingmei Li, Wangcheng Zhang, Yanpeng Wang, Luji Yu, and Jiantao Ji. 2023. "A Novel Layered and Advanced Nitrogen Removal Filter with Gravel and Embedded Bio-Organic Carrier Based on Autotrophic and Heterotrophic Pathways" Water 15, no. 10: 1919. https://doi.org/10.3390/w15101919
APA StylePeng, Z., Liu, M., Li, T., Zhang, W., Wang, Y., Yu, L., & Ji, J. (2023). A Novel Layered and Advanced Nitrogen Removal Filter with Gravel and Embedded Bio-Organic Carrier Based on Autotrophic and Heterotrophic Pathways. Water, 15(10), 1919. https://doi.org/10.3390/w15101919