Anxiety in Duckweed–Metabolism and Effect of Diazepam on Lemna minor
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Lemna Cultivation
2.3. Chlorophyll Fluorescence Measurement
2.4. ROS Analysis
2.5. Medium Extraction
2.6. LC-MS/MS Analysis
2.7. Statistical Analysis
3. Results
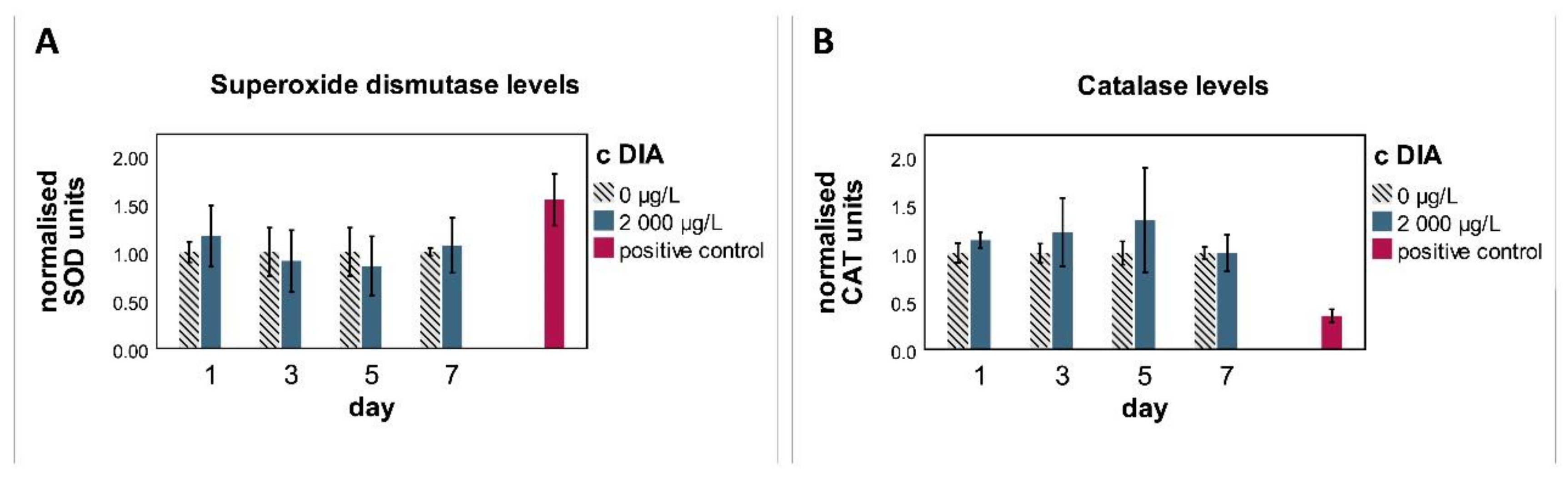
3.1. Biochemistry
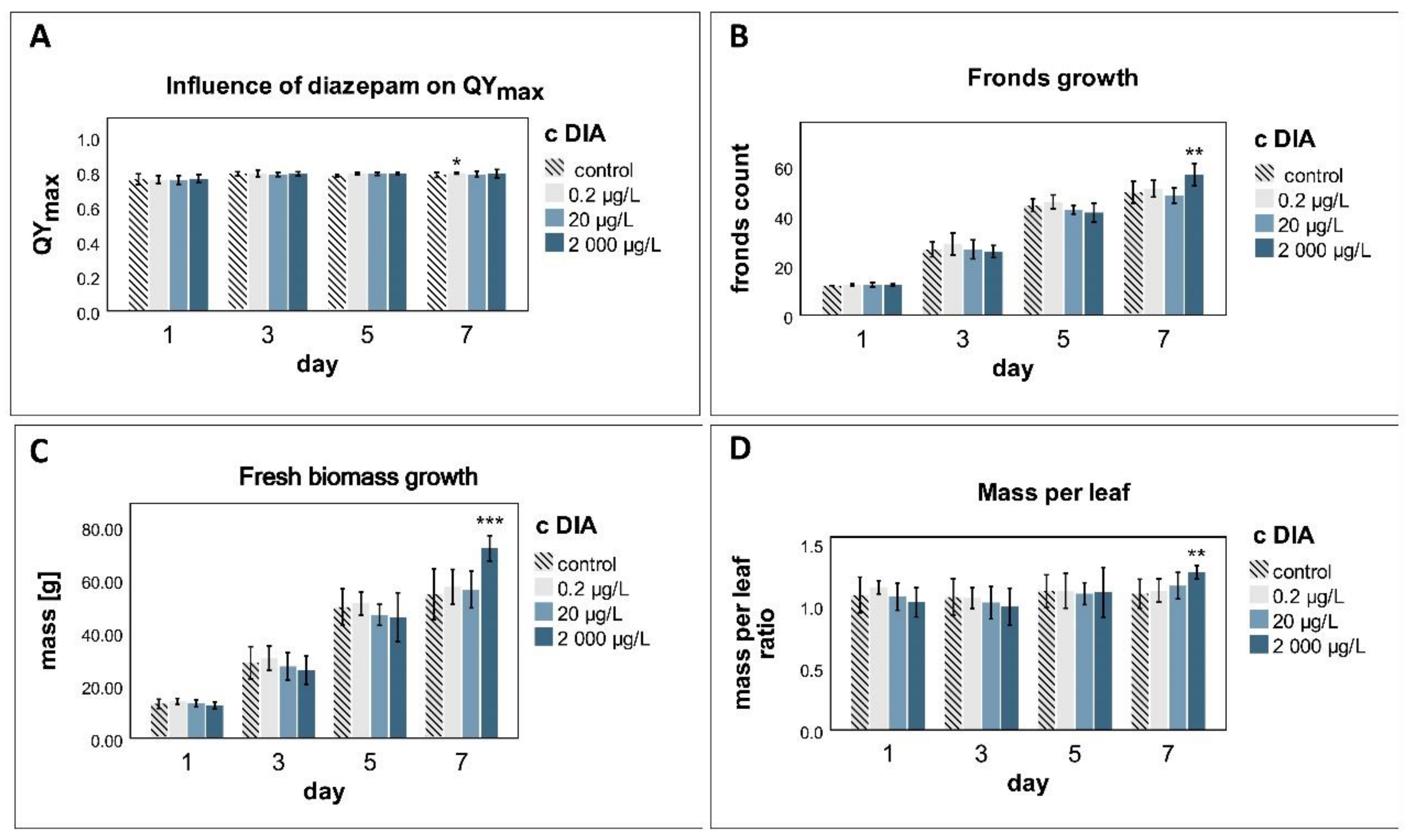
3.2. QYmax
3.3. Fronds Growth
3.4. Mass Growth
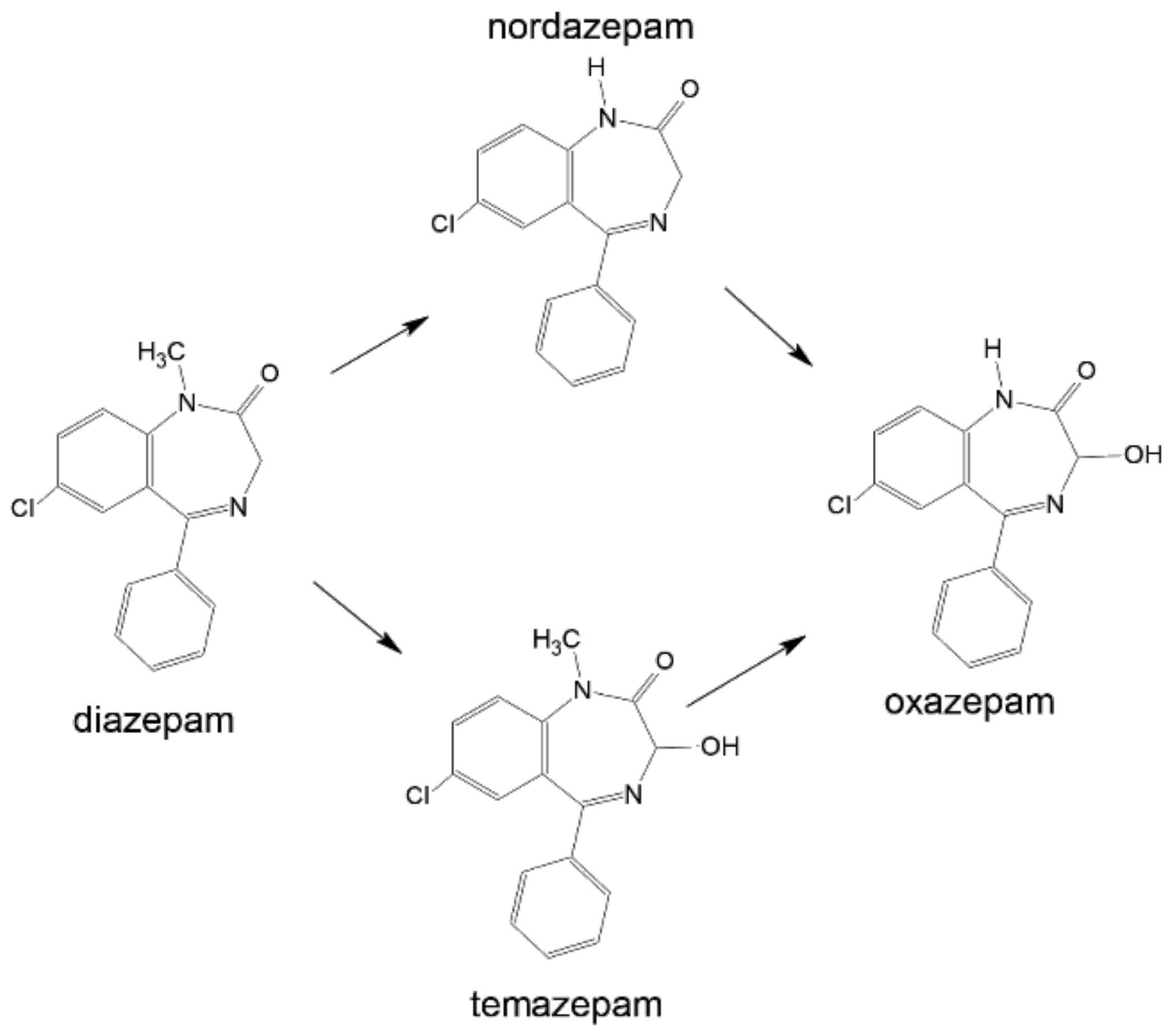
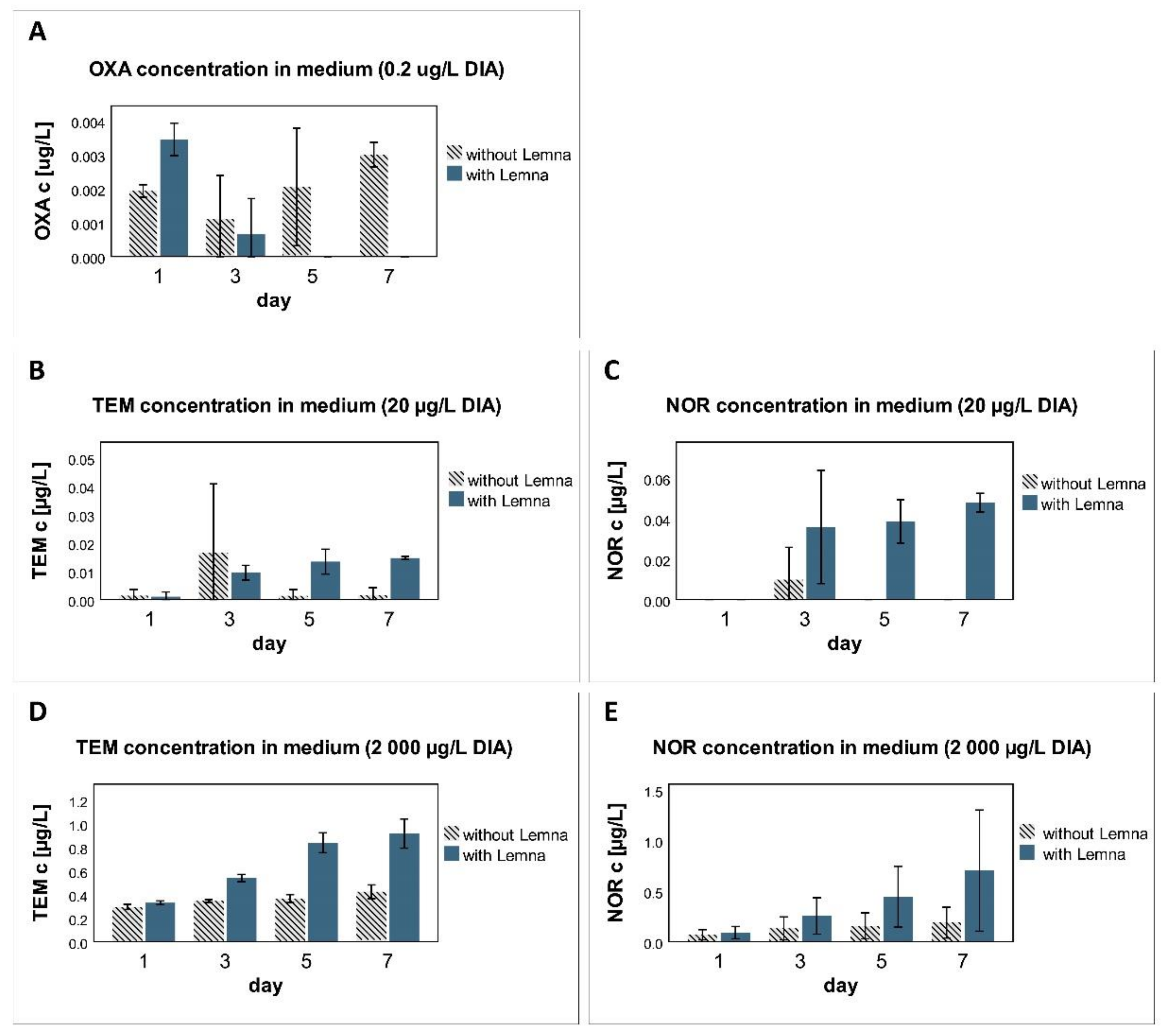
3.5. Metabolism
4. Discussion
4.1. Diazepam Concentration Levels
4.2. Biochemistry
4.3. Photosynthesis
4.4. Physiological Changes
4.5. Metabolism
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Parent ion(m/z) | Quantifier/Qualifier | Fragmentor | Collision Energy (V) | |
|---|---|---|---|---|
| diazepam | 285 | 154/193 | 150 | 30/30 |
| diazepam-D5 | 290 | 154/198 | 150 | 30/30 |
| nordiazepam | 271 | 140/165 | 150 | 30/30 |
| oxazepam | 287 | 241/104 | 100 | 30/40 |
| temazepam | 301 | 255/177 | 100 | 25/45 |
References
- Barreto, E.F.; Larson, T.R.; Koubek, E.J. Drug Excretion. In Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Nikolaou, A.; Meric, S.; Fatta, D. Occurrence patterns of pharmaceuticals in water and wastewater environments. Anal. Bioanal. Chem. 2007, 387, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- OECD. Toolkit for Water Policies and Governance; OECD Council Recommendation on Water; OECD Publishing: Paris, France, 2021. [Google Scholar]
- Council of the European Union. Directive 2013/39/EU of the European Parliament and of the Council of 12 August 2013 amending Directives 2000/60/EC and 2008/105/EC as regards priority substances in the field of water policy. Text with EEA relevance. Off. J. Eur. Union 2013, 56, 1–17. [Google Scholar] [CrossRef]
- Eurostat. Medicine Use Statistics. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php?title=Medicine_use_statistics#Prescribed_medicines (accessed on 26 December 2021).
- Eurostat. Self-Reported Use of Prescribed Medicines by Sex, Age and Educational Attainment Level. Available online: https://ec.europa.eu/eurostat/databrowser/view/HLTH_EHIS_MD1E__custom_1718334/default/table?lang=en (accessed on 26 December 2021).
- Sengar, A.; Vijayanandan, A. Human health and ecological risk assessment of 98 pharmaceuticals and personal care products (PPCPs) detected in Indian surface and wastewaters. Sci. Total Environ. 2022, 807, 18. [Google Scholar] [CrossRef] [PubMed]
- Kay, P.; Hughes, S.R.; Ault, J.R.; Ashcroft, A.E.; Brown, L.E. Widespread, routine occurrence of pharmaceuticals in sewage effluent, combined sewer overflows and receiving waters. Environ. Pollut. 2017, 220, 1447–1455. [Google Scholar] [CrossRef] [Green Version]
- Wahlberg, C.; Bjorlenius, B.; Paxeus, N. Fluxes of 13 selected pharmaceuticals in the water cycle of Stockholm, Sweden. Water Sci. Technol. 2011, 63, 1772–1780. [Google Scholar] [CrossRef]
- Reberski, J.L.; Terzic, J.; Maurice, L.D.; Lapworth, D.J. Emerging organic contaminants in karst groundwater: A global level assessment. J. Hydrol. 2022, 604, 15. [Google Scholar] [CrossRef]
- Fram, M.S.; Belitz, K. Occurrence and concentrations of pharmaceutical compounds in groundwater used for public drinking-water supply in California. Sci. Total Environ. 2011, 409, 3409–3417. [Google Scholar] [CrossRef] [Green Version]
- Wu, M.; Xiang, J.; Que, C.; Chen, F.; Xu, G. Occurrence and fate of psychiatric pharmaceuticals in the urban water system of Shanghai, China. Chemosphere 2015, 138, 486–493. [Google Scholar] [CrossRef]
- Cerveny, D.; Brodin, T.; Cisar, P.; McCallum, E.S.; Fick, J. Bioconcentration and behavioral effects of four benzodiazepines and their environmentally relevant mixture in wild fish. Sci. Total Environ. 2020, 702, 134780. [Google Scholar] [CrossRef]
- David, A.; Lange, A.; Tyler, C.R.; Hill, E.M. Concentrating mixtures of neuroactive pharmaceuticals and altered neurotransmitter levels in the brain of fish exposed to a wastewater effluent. Sci. Total Environ. 2018, 621, 782–790. [Google Scholar] [CrossRef]
- Gomez, E.; Bachelot, M.; Boillot, C.; Munaron, D.; Chiron, S.; Casellas, C.; Fenet, H. Bioconcentration of two pharmaceuticals (benzodiazepines) and two personal care products (UV filters) in marine mussels (Mytilus galloprovincialis) under controlled laboratory conditions. Environ. Sci. Pollut. Res. 2012, 19, 2561–2569. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, U.; Mohler, H. GABA-based therapeutic approaches: GABA(A) receptor subtype functions. Curr. Opin. Pharmacol. 2006, 6, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Pang, K.S.; Morris, M.E.; Sun, H.D. Formed and preformed metabolites: Facts and comparisons. J. Pharm. Pharmacol. 2008, 60, 1247–1275. [Google Scholar] [CrossRef] [PubMed]
- Andersson, T.; Miners, J.O.; Veronese, M.E.; Birkett, D.J. Diazepam Metabolism by Human Liver-Microsomes Is Mediated by both S-Mephenytoin Hydroxylase and CYP3A Isoforms. Br. J. Clin. Pharmacol. 1994, 38, 131–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calcaterra, N.E.; Barrow, J.C. Classics in Chemical Neuroscience: Diazepam (Valium). ACS Chem. Neurosci. 2014, 5, 253–260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López-García, E.; Mastroianni, N.; Postigo, C.; Barceló, D.; López de Alda, M. A fully automated approach for the analysis of 37 psychoactive substances in raw wastewater based on on-line solid phase extraction-liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2018, 1576, 80–89. [Google Scholar] [CrossRef]
- Fernández-Rubio, J.; Rodríguez-Gil, J.L.; Postigo, C.; Mastroianni, N.; López de Alda, M.; Barceló, D.; Valcárcel, Y. Psychoactive pharmaceuticals and illicit drugs in coastal waters of North-Western Spain: Environmental exposure and risk assessment. Chemosphere 2019, 224, 379–389. [Google Scholar] [CrossRef]
- Huerta-Fontela, M.; Galceran, M.T.; Ventura, F. Fast liquid chromatography–quadrupole-linear ion trap mass spectrometry for the analysis of pharmaceuticals and hormones in water resources. J. Chromatogr. A 2010, 1217, 4212–4222. [Google Scholar] [CrossRef]
- Loffler, D.; Rombke, J.; Meller, M.; Ternes, T.A. Environmental fate of pharmaceuticals in water/sediment systems. Environ. Sci. Technol. 2005, 39, 5209–5218. [Google Scholar] [CrossRef]
- Bellot, M.; Barata, C.; Gomez-Canela, C. Aqueous stability and degradation of psychiatric and neuroactive compounds and its biological activity in Daphnia magna. Sci. Total Environ. 2021, 798, 8. [Google Scholar] [CrossRef]
- Chen, C.; Li, L.X.; Li, M.; Wu, M.; Liang, W.H.; Takai, Y.K.; Qiu, X.C.; Shimasaki, Y.; Oshima, Y.J. Impacts of Diazepam on the Survival, Development, and Response to Light Stimulation in Early-life stages of Zebrafish (Danio rerio). J. Fac. Agric. Kyushu Univ. 2021, 66, 205–210. [Google Scholar] [CrossRef]
- Gagne, F.; Andre, C.; Gelinas, M. Neurochemical effects of benzodiazepine and morphine on freshwater mussels. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2010, 152, 207–214. [Google Scholar] [CrossRef]
- Brodin, T.; Fick, J.; Jonsson, M.; Klaminder, J. Dilute Concentrations of a Psychiatric Drug Alter Behavior of Fish from Natural Populations. Science 2013, 339, 814–815. [Google Scholar] [CrossRef] [PubMed]
- Brodin, T.; Nordling, J.; Lagesson, A.; Klaminder, J.; Hellstrom, G.; Christensen, B.; Fick, J. Environmental relevant levels of a benzodiazepine (oxazepam) alters important behavioral traits in a common planktivorous fish, (Rutilus rutilus). J. Toxicol. Environ. Health Part A Curr. Issues 2017, 80, 963–970. [Google Scholar] [CrossRef] [PubMed]
- OECD. OECD Guidelines for the Testing of Chemicals, Section 2: Effects on Biotic Systems, Test No. 221: Lemna sp. Growth Inhibition Test. 2006. Available online: https://read.oecd-ilibrary.org/environment/test-no-221-lemna-sp-growth-inhabition-test_9789264016194-en#page1 (accessed on 17 August 2020).
- Markovic, M.; Neale, P.A.; Nidumolu, B.; Kumar, A. Combined toxicity of therapeutic pharmaceuticals to duckweed, Lemna minor. Ecotoxicol. Environ. Saf. 2021, 208, 8. [Google Scholar] [CrossRef] [PubMed]
- Seifi, A.; Dehghani, M. Influence of nano-titanium dioxide particles (TiO2 NPs) on improving phytoremediation efficiency of As/Cu/Cd from copper mine wastewaters using Lemna minor. Arab. J. Geosci. 2021, 14, 14. [Google Scholar] [CrossRef]
- Ali, S.; Abbas, Z.; Rizwan, M.; Zaheer, I.E.; Yava, I.; Unay, A.; Abdel-Daim, M.M.; Bin-Jumah, M.; Hasanuzzaman, M.; Kalderis, D. Application of Floating Aquatic Plants in Phytoremediation of Heavy Metals Polluted Water: A Review. Sustainability 2020, 12, 1927. [Google Scholar] [CrossRef] [Green Version]
- Matuszewski, B.K.; Constanzer, M.L.; Chavez-Eng, C.M. Strategies for the assessment of matrix effect in quantitative bioanalytical methods based on HPLC-MS/MS. Anal. Chem. 2003, 75, 3019–3030. [Google Scholar] [CrossRef]
- Bergmann, R.; Kongsbak, K.; Sorensen, P.L.; Sander, T.; Balle, T. A Unified Model of the GABA(A) Receptor Comprising Agonist and Benzodiazepine Binding Sites. PLoS ONE 2013, 8, e52323. [Google Scholar] [CrossRef] [Green Version]
- Ramesh, S.A.; Tyerman, S.D.; Xu, B.; Bose, J.; Kaur, S.; Conn, V.; Domingos, P.; Ullah, S.; Wege, S.; Shabala, S.; et al. GABA signalling modulates plant growth by directly regulating the activity of plant-specific anion transporters. Nat. Commun. 2015, 6, 7879. [Google Scholar] [CrossRef] [Green Version]
- Žárský, V. Signal transduction: GABA receptor found in plants. Nat. Plants 2015, 1, 15115. [Google Scholar] [CrossRef] [PubMed]
- Bourioug, M.; Mazzitelli, J.Y.; Marty, P.; Budzinski, H.; Aleya, L.; Bonnafe, E.; Geret, F. Assessment of Lemna minor (duckweed) and Corbicula fluminea (freshwater clam) as potential indicators of contaminated aquatic ecosystems: Responses to presence of psychoactive drug mixtures. Environ. Sci. Pollut. Res. 2018, 25, 11192–11204. [Google Scholar] [CrossRef] [PubMed]
- Carter, L.J.; Williams, M.; Martin, S.; Kamaludeen, S.P.B.; Kookana, R.S. Sorption, plant uptake and metabolism of benzodiazepines. Sci. Total Environ. 2018, 628–629, 18–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dudley, S.; Sun, C.L.; McGinnis, M.; Trumble, J.; Gan, J. Formation of biologically active benzodiazepine metabolites in Arabidopsis thaliana cell cultures and vegetable plants under hydroponic conditions. Sci. Total Environ. 2019, 662, 622–630. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lindemann, P.; Koch, A.; Degenhardt, B.; Hause, G.; Grimm, B.; Papadopoulos, V. A Novel Arabidopsis thaliana Protein is a Functional Peripheral-Type Benzodiazepine Receptor. Plant Cell Physiol. 2004, 45, 723–733. [Google Scholar] [CrossRef] [Green Version]
- Frank, W.; Baar, K.-M.; Qudeimat, E.; Woriedh, M.; Alawady, A.; Ratnadewi, D.; Gremillon, L.; Grimm, B.; Reski, R. A mitochondrial protein homologous to the mammalian peripheral-type benzodiazepine receptor is essential for stress adaptation in plants. Plant J. 2007, 51, 1004–1018. [Google Scholar] [CrossRef] [PubMed]
- Corsi, L. Peripheral benzodiazepine receptors in potatoes (Solanum tuberosum). Biochem. Biophys. Res. Commun. 2004, 313, 62–66. [Google Scholar] [CrossRef]
| Oxazepam | Temazepam | Nordazepam | Diazepam | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Concentration [µg/L] | 0.2 | 20 | 2000 | 0.2 | 20 | 2000 | 0.2 | 20 | 2000 | 0.2 | 20 | 2000 |
| Process efficiency * | 102.99 ± 5.96 | 77.89 ± 10.00 | 103.59 ± 19.06 | 111.15 ± 6.14 | 86.00 ± 5.75 | 104.56 ± 15.65 | 104.74 ± 2.81 | 85.10 ± 7.67 | 104.10 ± 15.80 | 134.76 ± 9.38 | 82.90 ± 3.61 | 102.05 ± 14.51 |
| Spiked recoveries * | 113.58 ± 4.71 | 102.76 ± 9.99 | 124.23 ± 18.37 | 115.76 ± 5.86 | 109.05 ± 5.62 | 122.98 ± 14.93 | 113.50 ± 5.50 | 110.00 ± 7.57 | 121.29 ± 15.08 | 110.14 ± 8.32 | 105.15 ± 3.30 | 119.57 ± 13.91 |
| Organism | Compound | Dose | Exposure | Effect | Reference |
|---|---|---|---|---|---|
| Lemna minor | mixture of psychoactive drugs incl. oxazepam | 0.5 µg/L | 7days | No effect on frond number, fresh biomass or frond area | [37] |
| 5 µg/L | 7days | ||||
| 50 µg/L | 7days | ||||
| Corbicula fluminea | mixture of psychoactive drugs including oxazepam | 50 µg/L | 3 days | Elevated EROD, GST, CAT, otherwise no effect | |
| Perca fluviatilis | oxazepam | 1.8 µg/L | 7 days | Increased activity, reduced sociality | [27] |
| 910 µg/L | 7 days | Increased activity, reduced sociality, increased boldness, higher feeding rate | |||
| Rutilus rutilus | oxazepam | 280 µg/L | - | Increased boldness | [28] |
| 0.84 µg/L | - | Increased boldness and activity | |||
| Perca fluviatilis | benzodiazepine mix (OXA, BRO, TEM, CLO) | 9.1, 6.9, 5.7, 8.1 µg/L | 7 days | Increased boldness and activity | [13] |
| 0.5, 0.5, 0.3, 0.4 µg/L | 7 days | Increased boldness and activity |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lamaczová, A.; Malina, T.; Maršálková, E.; Odehnalová, K.; Opatřilová, R.; Přibilová, P.; Zezulka, Š.; Maršálek, B. Anxiety in Duckweed–Metabolism and Effect of Diazepam on Lemna minor. Water 2022, 14, 1484. https://doi.org/10.3390/w14091484
Lamaczová A, Malina T, Maršálková E, Odehnalová K, Opatřilová R, Přibilová P, Zezulka Š, Maršálek B. Anxiety in Duckweed–Metabolism and Effect of Diazepam on Lemna minor. Water. 2022; 14(9):1484. https://doi.org/10.3390/w14091484
Chicago/Turabian StyleLamaczová, Adéla, Tomáš Malina, Eliška Maršálková, Klára Odehnalová, Radka Opatřilová, Petra Přibilová, Štěpán Zezulka, and Blahoslav Maršálek. 2022. "Anxiety in Duckweed–Metabolism and Effect of Diazepam on Lemna minor" Water 14, no. 9: 1484. https://doi.org/10.3390/w14091484
APA StyleLamaczová, A., Malina, T., Maršálková, E., Odehnalová, K., Opatřilová, R., Přibilová, P., Zezulka, Š., & Maršálek, B. (2022). Anxiety in Duckweed–Metabolism and Effect of Diazepam on Lemna minor. Water, 14(9), 1484. https://doi.org/10.3390/w14091484






